Abstract
The aim of this study was to determine whether the vascular endothelial growth factor (VEGF) +936C/T polymorphism confers susceptibility to gastric cancer (GC) by conducting a meta-analysis. Publications addressing the association between the VEGF +936C/T polymorphism and GC risk were selected from the Pubmed, Embase and CBM databases. Data were extracted from the studies by two independent reviewers. The meta-analysis was performed using RevMan 5.0.25 and STATA 9.2 software. From these data, the odds ratio (OR) with 95% confidence interval (CI) was calculated. Finally, 8 case-control studies were retrieved reporting a total of 2,131 gastrointestinal cancer patients and 2,670 controls. Meta-analysis results showed that there was no significant association between the VEGF +936C/T polymorphism and GC risk in all comparisons of the T allele vs. C allele (OR=1.08, 95% CI 0.90–1.30, P=0.42), CT+TT vs. CC (OR=1.08, 95% CI 0.87–1.34, P=0.49), TT vs. CC+CT (OR=1.14, 95% CI 0.85–1.53, P=0.37), TT vs. CC (OR=1.18, 95% CI 0.87–1.59, P=0.28) and TT vs. CT (OR=1.11, 95% CI 0.79–1.56, P=0.56). This meta-analysis confirms that there is a lack of association between the VEGF +936C/T polymorphism and GC risk.
Keywords: gastric cancer, polymorphism, vascular endothelial growth factor, meta-analysis
Introduction
Despite a major decline in the incidence and mortality of gastric cancer (GC) over the past several decades, GC remains the fourth most common cancer and the second leading cause of cancer-related death in the world (1). The incidence of GC is particularly high in East Asia, Eastern Europe and parts of Central and South America (2). Male-to-female incidence ratios generally are in the 1.5–2.5 range, with higher ratios for intestinal than diffuse cancers and higher-risk populations (3). GC is a major health problem worldwide due to its high incidence, poor prognosis and limited treatment options (4). GC is a multifactorial disease with environmental and lifestyle factors as major contributors (5). Epidemiological investigations have identified many risk factors for GC, including age, male gender, Helicobacter pylori (H. pylori) bacteria infection, smoking, obesity, radiation, diet and hereditary factors (4–6). The most important etiological factors implicated in gastric carcinogenesis are H. pylori infection, and dietary and hereditary factors (7–9). Individual variations in cancer risk have been associated with specific variant alleles of different genes (polymorphisms) that are present in a significant proportion of the normal population (10). Polymorphisms in a wide variety of genes may modify the effect of environmental exposures, and these gene-environmental interactions could explain the high variation in the GC incidence observed worldwide (11,12). However, the interaction between environmental factors and genetic susceptibility has not yet been adequately addressed.
Tumor angiogenesis is a vital process for the progression of a neoplasm from a small localized tumor to an enlarged tumor with the ability to metastasize (13). Vascular endothelial growth factor (VEGF) as a vascular permeability factor is the major mediator of physiologic and pathologic angiogenesis (14). Evidence from in vitro and in vivo experiments has shown that increased VEGF expression is associated with tumor growth and metastasis, whereas the inhibition of VEGF signaling results in suppression of both tumor angiogenesis and tumor growth (15). The VEGF gene is assigned to chromosome 6p12-p21 and consists of eight exons separated by seven introns that exhibit alternative splicing to form a family of proteins (16). In mammals, the VEGF family consists of seven secreted glycoproteins that are designated VEGF-A, VEGF-B, VEGF-C, VEGF-D, VEGF-E, placental growth factor (PIGF) and VEGF-F (17). VEGF and its receptors play an important role in the development of the vascular system, via angiogenesis mechanisms, as well as in the formation of the lymphatic vascular system (18). In the stomach, GC frequently displays high levels of VEGF expression which has been correlated with vascular involvement, and lymph node and liver metastasis (19,20). Moreover, high levels of VEGF expression have also been observed in gastric premalignant lesions, such as chronic atrophic gastritis and intestinal metaplasia, suggesting that alterations in VEGF expression may also contribute to the process of gastric carcinogenesis via angiogenesis (21).
Several polymorphisms in the VEGF gene involved in the development of GC have been described in the literature (22). Among these, VEGF +936C/T (rs3025039) in the 3′-untranslated region is one of the most common polymorphisms. Numerous studies have evaluated the association between the VEGF +936C/T polymorphism and GC risk. However, the results have been inconsistent. The aim of this meta-analysis was to investigate the association of VEGF polymorphisms with GC risk by conducting a meta-analysis from all eligible case-control studies published to date.
Materials and methods
Literature search
An electronic search of the Pubmed, Embase and CBM was performed to retrieve studies linking the VEGF +936C/T polymorphism and GC risk available by December 2010 without language restrictions, using the following query: [‘Vascular Endothelial Growth Factor A’ or ‘Vascular Endothelial Growth Factor B’ or ‘Vascular Endothelial Growth Factor C’ or ‘Vascular Endothelial Growth Factor D’ or ‘Vascular Endothelial Growth Factor, Endocrine-Gland-Derived’ or ‘Vascular Endothelial Growth Factors’ (Mesh)] and [‘Polymorphism, Single Nucleotide’ or ‘Polymorphism, Restriction Fragment Length’ or ‘Polymorphism, Single-Stranded Conformational’ or ’Genomic Structural Variation’ or ‘Polymorphism, Genetic’ (Mesh)] and [‘Gastric Cancer’ or ‘Gastric Neoplasm’ or ‘Stomach Neoplasms’ (Mesh)]. The reference lists of major textbooks, reviews and included articles were identified through manual searches to find other potentially eligible studies. Studies reported by the same authors, yet published in different journals, were checked for possible overlapping participant groups. When pertinent data were not included or data that were presented were unclear, the authors were directly contacted.
Inclusion criteria and exclusion criteria
To be eligible for inclusion in this meta-analysis, the following criteria were established: i) case-control studies that addressed GC cases and healthy or benign disease controls; ii) studies that evaluated the association between the VEGF +936C/T polymorphism and GC risk; iii) studies that included sufficient genotype data for extraction. Studies excluded were: i) not case-control studies that evaluated the association between the VEGF +936C/T polymorphism and GC risk; ii) case reports, letters, reviews and editorial articles; iii) studies based on incomplete raw data and no usable data reported; iv) studies containing duplicate data; v) family-based design.
Data extraction
Using a standardized form, data from the published studies were extracted independently by two reviewers (X.H. Dong and G.J. Jin) to populate the necessary information. From each of the included articles, the following information was extracted: first author, year of publication, country, language, ethnicity, study design, diagnostic criteria, source of cases and controls, number of cases and controls, sample, detection methods, polymorphisms and evidence of Hardy-Weinberg equilibrium (HWE) in controls. For conflicting evaluations, an agreement was reached following discussion.
Quality assessment of the included studies
The quality of the studies was also independently assessed by two reviewers (X.H. Dong and G.J. Jin) based on the STROBE quality score systems (23). Thirty items relevant to the quality appraisal were used for assessment in this meta-analysis, with scores ranging from 0 to 30. Any discrepancies between the two reviewers were resolved by discussion and consultation with a third reviewer (H. Shang).
Statistical analysis
The meta-analysis was carried out using the Review Manager version 5.0.25 (provided by The Cochrane Collaboration) and STATA package version 9.2 (Stata Corporation, College Station, TX, USA). The strength of the associations between the VEGF +936C/T polymorphism and GC risk was estimated using odds ratio (OR) and 95% confidence interval (95% CI). The following contrasts for the VEGF +936C/T polymorphism were evaluated: comparison of the variant allele with ancestral allele (T vs. C); comparison of the variant homozygote combined with the heterozygote vs. ancestral homozygote (T/T+C/T vs. C/C); comparison of the variant homozygote combined with the heterozygote vs. ancestral homozygote (T/T vs. C/C+C/T); comparison of the variant homozygote vs. ancestral homozygote (T/T vs. C/C); comparison of the variant homozygote vs. heterozygote (T/T vs. C/T). Between-study heterogeneities were estimated using Cochran’s Q test (24,25). We also quantified the effect of heterogeneity using the I2 test. I2 ranges between 0 and 100% and represents the proportion of inter-study variability that can be attributed to heterogeneity rather than chance. I2 values of 25, 50 and 75% were defined as low, moderate and high estimates, respectively. When a significant Q test (P<0.10) or I2<50% indicated heterogeneity across studies, the random effects model was used for meta-analysis, or else the fixed effects model was used (26). Before the effect estimation of associations between the VEGF +936C/T polymorphism and GC risk, we tested whether genotype frequencies of the controls were in HWE using the χ2 test. Subgroup analysis based on ethnicity was used to explore and to explain the diversity among the results of different studies. Sensitivity analysis was mainly performed by sequential omission of individual studies. Publication bias was investigated by Begg’s funnel plot, and funnel plot asymetry was assessed by Egger’s linear regeression test (27). Statistical significance was considered when the P-value of Egger’s test was ≤0.10. All P-values were two-sided. To ensure the reliability and the accuracy of the results, two reviewers (L.P. Zhou and H. Luan) populated the data in the statistical software programs independently and obtained the same results.
Results
Characteristics of the included studies
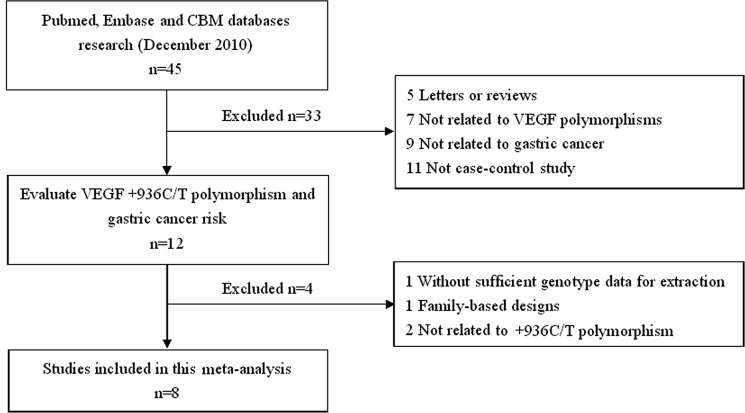
The search strategy retrieved 45 potentially relevant studies. According to the inclusion criteria, only 8 studies (28–35) with full-text were included in this meta-analysis and 37 studies were excluded. The flow chart of the study selection is summarized in Fig. 1. These 8 case-control studies selected included a total of 2,131 GC cases and 2,670 healthy or benign disease controls. All were case-control studies which evaluated the association between VEGF +936C/T polymorphism and GC risk. The year of publication of the included studies ranged from 2006 to 2010. All included articles were written in English except one in Chinese (35). The source of controls was mainly based on healthy population. Diverse genotyping methods mainly used polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). The baseline characteristics and methodological quality of all included studies are summarized in Table I. The genotype distribution and risk allele frequency are summarized in Table II.
Figure 1.
Flow chart showing the study selection procedure. Finally, 8 case-control studies were retrieved reporting a total of 2,131 gastrointestinal cancer patients and 2,670 controls.
Table I.
Baseline characteristics of the studies included in the meta-analysis.
| First author (ref.) | Year | Country | Ethnicity | Source of controls | Source of cases | Detection method | No. of subjects
|
Gender (male/female)
|
Age, in years (mean±SD)
|
|||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cases | Controls | Cases | Controls | Cases | Controls | |||||||
| Tzanakis, et al (28) | 2006 | Greece | Caucasian | Population-based | Hospital-based | PCR-RFLP | 100 | 100 | 58/42 | - | 65.0±12.3 | - |
| Chae, et al (29) | 2006 | Korea | Asian | Population-based | Hospital-based | PCR-RFLP | 413 | 413 | 313/100 | 333/80 | 60.2±10.3 | 60.2±9.0 |
| Bae, et al (30) | 2008 | Korea | Asian | Population-based | Hospital-based | PCR-RFLP | 154 | 229 | 91/63 | 112/117 | 58.1±12.7 | 59.6±11.8 |
| Ke, et al (31) | 2008 | China | Asian | Population-based | Population-based | PCR-RFLP | 540 | 561 | 371/169 | 384/177 | 60.4±9.5 | 59.5±10.2 |
| Al-Moundhri, et al (32) | 2009 | Oman | Caucasian | Population-based | Hospital-based | PCR-RFLP | 130 | 130 | 75/55 | - | 56.8±12.2 | 41.3±11.6 |
| Guan, et al (33) | 2009 | USA | Caucasian | Population-based | Hospital-based | PCR-RFLP | 171 | 353 | 56/115 | 104/249 | 59.7±12.6 | 57.6±11.2 |
| Tahara, et al (34) | 2009 | Japan | Asian | Hospital-based | Hospital-based | PCR-RFLP | 385 | 459 | 271/114 | 267/192 | 65.5±11.0 | 61.5±13.0 |
| Xia, et al (35) | 2010 | China | Asian | Population-based | Hospital-based | - | 238 | 425 | 191/47 | 336/89 | 61.0±9.4 | 60.6±8.4 |
PCR-RFLP, polymerase chain reaction-restriction fragment length polymorphism; SD, standard deviation.
Table II.
Genotype distribution and risk allele frequency of all studies included.
| First author (Ref.) | Cases
|
Controls
|
HWE test (P-value)
|
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | CC | CT | TT | Total allele | C | T | AF | Total | CC | CT | TT | Total allele | C | T | AF | ||
| Tzanakis, et al (28) | 100 | 41 | 33 | 26 | 200 | 115 | 85 | 0.425 | 100 | 51 | 27 | 22 | 200 | 129 | 71 | 0.355 | 0.000 |
| Chae, et al (29) | 413 | 283 | 122 | 8 | 826 | 688 | 138 | 0.167 | 413 | 252 | 149 | 12 | 826 | 653 | 173 | 0.209 | 0.069 |
| Bae, et al (30) | 154 | 89 | 58 | 7 | 308 | 236 | 72 | 0.234 | 229 | 169 | 57 | 3 | 458 | 395 | 63 | 0.138 | 0.458 |
| Ke, et al (31) | 540 | 373 | 152 | 15 | 1,080 | 898 | 182 | 0.169 | 561 | 386 | 164 | 11 | 1,122 | 936 | 186 | 0.166 | 0.177 |
| Al-Moundhri, et al (32) | 130 | 109 | 2 | 19 | 260 | 220 | 40 | 0.154 | 130 | 110 | 0 | 20 | 260 | 220 | 40 | 0.154 | 0.000 |
| Guan, et al (33) | 171 | 127 | 41 | 3 | 342 | 295 | 47 | 0.137 | 353 | 276 | 70 | 7 | 706 | 622 | 84 | 0.119 | 0.309 |
| Tahara, et al (34) | 385 | 256 | 118 | 11 | 770 | 630 | 140 | 0.182 | 459 | 300 | 140 | 19 | 918 | 740 | 178 | 0.194 | 0.603 |
| Xia, et al (35) | 238 | 155 | 63 | 10 | 476 | 373 | 83 | 0.174 | 425 | 276 | 131 | 6 | 850 | 683 | 143 | 0.168 | 0.021 |
AF, allele frequency; HWE, Hardy-Weinberg equilibrium.
Main meta-analysis results
A summary of the meta-analysis findings of the association between the VEGF +936C/T polymorphism and GC risk is provided in Table III. Meta-analysis results identified no significant association between the VEGF +936C/T polymorphism and GC risk in all comparisons of T allele vs. C allele (OR=1.08, 95% CI 0.90–1.30, P=0.42), CT+TT vs. CC (OR=1.08, 95% CI 0.87–1.34, P=0.49), TT vs. CC+CT (OR=1.14, 95% CI 0.85–1.53, P=0.37), TT vs. CC (OR=1.18, 95% CI 0.87–1.59, P=0.28) and TT vs. CT (OR=1.11, 95% CI 0.79–1.56, P=0.56). In the subgroup analysis based on ethnicity, the subjects of all included studies were divided into Caucasian and Asian populations. Results of subgroup analysis confirmed that there was also no association between the VEGF +936C/T polymorphism and GC risk in both Caucasian and Asian populations. Sensitivity analysis was performed by sequential omission of individual studies. The significance of the pooled OR in all individual analyses and subgroup analyses was not influenced excessively by omitting any single study. In addition, we also performed sensitivity analysis by omission of studies without HWE, but the results were also not influenced.
Table III.
Meta-analysis of the association between the VEGF +936C/T polymorphism and gastric cancer risk.
| Comparisons | OR | 95% CI | P-value | Heterogeneity
|
Effects model | |
|---|---|---|---|---|---|---|
| I2 | P-value | |||||
| T vs. C | 1.08 | 0.90–1.30 | 0.42 | 64% | 0.007 | Random |
| Caucasian | 1.18 | 0.93–1.51 | 0.17 | 0% | 0.650 | |
| Asian | 1.05 | 0.82–1.34 | 0.72 | 76% | 0.002 | |
| T/T+C/T vs. C/C | 1.08 | 0.87–1.34 | 0.49 | 63% | 0.008 | Random |
| Caucasian | 1.27 | 0.94–1.72 | 0.12 | 0% | 0.730 | |
| Asian | 1.02 | 0.77–1.35 | 0.88 | 75% | 0.003 | |
| T/T vs. C/C+C/T | 1.14 | 0.85–1.53 | 0.37 | 32% | 0.170 | Fixed |
| Caucasian | 1.07 | 0.68–1.66 | 0.78 | 0% | 0.810 | |
| Asian | 1.21 | 0.82–1.79 | 0.34 | 59% | 0.050 | |
| T/T vs. C/C | 1.18 | 0.87–1.59 | 0.28 | 41% | 0.100 | Fixed |
| Caucasian | 1.15 | 0.73–1.82 | 0.55 | 0% | 0.660 | |
| Asian | 1.20 | 0.81–1.77 | 0.37 | 64% | 0.030 | |
| T/T vs. C/T | 1.11 | 0.79–1.56 | 0.56 | 27% | 0.220 | Fixed |
| Caucasian | 0.82 | 0.43–1.57 | 0.55 | 0% | 0.590 | |
| Asian | 1.25 | 0.83–1.87 | 0.28 | 47% | 0.110 | |
OR, odds ratio; 95% CI, 95% confidence interval.
Publication bias
Publication bias of the literature was assessed using the Begg’s funnel plot and Egger’s linear regression test. Egger’s linear regression test was used to measure the asymmetry of the funnel plot. The results of Egger’s linear regression test are shown in Table IV. Results showed that there was no publication bias (all P>0.05).
Table IV.
Evaluation of publication bias by Egger’s linear regression test.
| Comparison | Coefficient | Standard error | t | P> |t| | 95% CI |
|---|---|---|---|---|---|
| T vs. C | 3.69 | 2.07 | 1.78 | 0.13 | −1.38–8.76 |
| T/T+C/T vs. C/C | 3.12 | 1.77 | 1.76 | 0.13 | −1.22–7.46 |
| T/T vs. C/C+C/T | 1.87 | 1.71 | 1.09 | 0.32 | −2.32–6.06 |
| T/T vs. C/C | 2.11 | 1.91 | 1.11 | 0.31 | −2.57–6.80 |
| T/T vs. C/T | −0.21 | 1.37 | −0.16 | 0.88 | −3.58–3.15 |
95% CI, 95% confidence interval.
Discussion
Evidence suggests that VEGF plays an important role in the carcinogenesis pathway, such as in the inhibition of apoptosis, tumor growth, angiogenesis, invasion and metastasis (36). The specific function of VEGF in the formation of prostaglandins makes it a strong candidate for increasing the susceptibility to common cancers, such as GC, colorectal, lung, breast, prostate cancer and other solid tumors (37–40). As is known, genetic polymorphisms altering the level of protein expressed are anticipated to have a substantial influence on disease activity (41). Several polymorphisms in VEGF have been previously reported, although some of these polymorphisms are not functionally significant and not associated with a susceptibility to GC.
Our meta-analysis quantitatively assessed the association between the VEGF +936C/T polymorphism and GC risk. Finally, 8 case-control studies were included and comprised a total of 2,131 GC cases and 2,670 healthy or benign disease controls. The main meta-analysis results showed that there was no association between the VEGF +936C/T polymorphism and GC risk in all comparisons of T allele vs. C allele, CT+TT vs. CC, TT vs. CC+CT, TT vs. CC and TT vs. CT, suggesting that the VEGF +936C/T polymorphism is not a risk factor for GC. Similarly, in the subgroup analysis by ethnicity, no association was found between the VEGF +936C/T polymorphism and GC risk in all comparisons in both Caucasian and Asian populations. Between-study heterogeneity was found in the comparisons of the T allele vs. C allele and CT+TT vs. CC; the random effects model was used. No heterogeneity was found in other comparisons; the fixed effects model was used. No evidence of publication bias was noted in this meta-analysis for the VEGF +936C/T polymorphism.
There were some limitations in our meta-analysis. Firstly, due to incomplete raw data or publication limitations, several relevant studies could not be included. Secondly, we were not able to address the sources of heterogeneity existing among studies for most polymorphisms. However, we could not perform subgroup stratifications analysis for the limited number of published studies. Thirdly, the lack of genotype frequency information provided by some published studies did not allow the estimation of the best genetic model of inheritance to follow. Although we actively contacted the authors, they did not provide a comprehensive set of data. In addition, the small sample size available was not ideal for detecting small genetic effects. Finally, our systematic review was based on unadjusted data, as the genotype information stratified for the main confounding variables was not available in the original studies and also the confounding factors addressed across the different studies were variable.
In conclusion, our meta-analysis of 8 case-control studies demonstrated that there was lack of association between the VEGF +936C/T polymorphism and GC risk. Therefore, the necessity to conduct large studies with an adequate methodological quality, properly controlling for possible confounds in order to obtain valid results should be emphasized.
Acknowledgments
This study was supported by a grant from the Science and Technology Research Project of the Higher Education Department of Liaoning Province (no. L2010695).
References
- 1.Brenner H, Rothenbacher D, Arndt V. Epidemiology of stomach cancer. Methods Mol Biol. 2009;472:467–477. doi: 10.1007/978-1-60327-492-0_23. [DOI] [PubMed] [Google Scholar]
- 2.Crew KD, Neugut AI. Epidemiology of gastric cancer. World J Gastroenterol. 2006;12:354–362. doi: 10.3748/wjg.v12.i3.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Roder DM. The epidemiology of gastric cancer. Gastric Cancer. 2002;5(Suppl 1):5–11. doi: 10.1007/s10120-002-0203-6. [DOI] [PubMed] [Google Scholar]
- 4.Compare D, Rocco A, Nardone G. Risk factors in gastric cancer. Eur Rev Med Pharmacol Sci. 2010;14:302–308. [PubMed] [Google Scholar]
- 5.Krejs GJ. Gastric cancer: epidemiology and risk factors. Dig Dis. 2010;28:600–603. doi: 10.1159/000320277. [DOI] [PubMed] [Google Scholar]
- 6.Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- 7.Uemura N, Okamoto S, Yamamoto S, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784–789. doi: 10.1056/NEJMoa001999. [DOI] [PubMed] [Google Scholar]
- 8.Liu C, Russell RM. Nutrition and gastric cancer risk: an update. Nutr Rev. 2008;66:237–249. doi: 10.1111/j.1753-4887.2008.00029.x. [DOI] [PubMed] [Google Scholar]
- 9.Oliveira C, Seruca R, Carneiro F. Hereditary gastric cancer. Best Prac Res Clin Gastroenterol. 2009;23:147–157. doi: 10.1016/j.bpg.2009.02.003. [DOI] [PubMed] [Google Scholar]
- 10.González CA, Sala N, Capellá G. Genetic susceptibility and gastric cancer risk. Int J Cancer. 2002;100:249–260. doi: 10.1002/ijc.10466. [DOI] [PubMed] [Google Scholar]
- 11.Perera FP, Weinstein IB. Molecular epidemiology: recent advances and future directions. Carcinogenesis. 2000;21:517–524. doi: 10.1093/carcin/21.3.517. [DOI] [PubMed] [Google Scholar]
- 12.Ames BN. Cancer prevention and diet: help from single nucleotide polymorphisms. Proc Natl Acad Sci USA. 1999;96:12216–12218. doi: 10.1073/pnas.96.22.12216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McMahon G. VEGF receptor signaling in tumor angiogenesis. Oncologist. 2000;5(Suppl 1):3–10. doi: 10.1634/theoncologist.5-suppl_1-3. [DOI] [PubMed] [Google Scholar]
- 14.Gaur P, Bose D, Samuel S, Ellis LM. Targeting tumor angiogenesis. Semin Oncol. 2009;36(Suppl 1):12–19. doi: 10.1053/j.seminoncol.2009.02.002. [DOI] [PubMed] [Google Scholar]
- 15.Ferrara N. VEGF and the quest for tumour angiogenesis factors. Nat Rev Cancer. 2002;2:795–803. doi: 10.1038/nrc909. [DOI] [PubMed] [Google Scholar]
- 16.Vincenti V, Cassano C, Rocchi M, Persico G. Assignment of the vascular endothelial growth factor gene to human chromosome 6p21.3. Circulation. 1996;93:1493–1495. doi: 10.1161/01.cir.93.8.1493. [DOI] [PubMed] [Google Scholar]
- 17.Otrock ZK, Makarem JA, Shamseddine AI. Vascular endothelial growth factor family of ligands and receptors: review. Blood Cells Mol Dis. 2007;38:258–268. doi: 10.1016/j.bcmd.2006.12.003. [DOI] [PubMed] [Google Scholar]
- 18.Zhang Z, Neiva KG, Lingen MW, et al. VEGF-dependent tumor angiogenesis requires inverse and reciprocal regulation of VEGFR1 and VEGFR2. Cell Death Differ. 2010;17:499–512. doi: 10.1038/cdd.2009.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ichikura T, Tomimatsu S, Ohkura E, et al. Prognostic significance of the expression of vascular endothelial growth factor (VEGF) and VEGF-C in gastric carcinoma. J Surg Oncol. 2001;78:132–137. doi: 10.1002/jso.1133. [DOI] [PubMed] [Google Scholar]
- 20.Lieto E, Ferraraccio F, Orditura M, et al. Expression of vascular endothelial growth factor (VEGF) and epidermal growth factor receptor (EGFR) is an independent prognostic indicator of worse outcome in gastric cancer patients. Ann Surg Oncol. 2008;15:69–79. doi: 10.1245/s10434-007-9596-0. [DOI] [PubMed] [Google Scholar]
- 21.Smith MG, Hold GL, Tahara E, et al. Cellular and molecular aspects of gastric cancer. World J Gastroenterol. 2006;12:2979–2990. doi: 10.3748/wjg.v12.i19.2979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Watson CJ, Webb NJ, Bottomley MJ, et al. Identification of polymorphisms within the vascular endothelial growth factor (VEGF) gene: correlation with variation in VEGF protein production. Cytokine. 2006;12:1232–1235. doi: 10.1006/cyto.2000.0692. [DOI] [PubMed] [Google Scholar]
- 23.Vandenbroucke JP, von Elm E, Altman DG, et al. Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. Epidemiology. 2007;18:805–835. doi: 10.1097/EDE.0b013e3181577511. [DOI] [PubMed] [Google Scholar]
- 24.Zintzaras E, Ioannidis JP. Heterogeneity testing in meta-analysis of genome searches. Genet Epidemiol. 2005;28:123–137. doi: 10.1002/gepi.20048. [DOI] [PubMed] [Google Scholar]
- 25.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–1158. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 26.Viechtbauer W. Confidence intervals for the amount of heterogeneity in meta-analysis. Stat Med. 2007;26:37–52. doi: 10.1002/sim.2514. [DOI] [PubMed] [Google Scholar]
- 27.Peters JL, Sutton AJ, Jones DR, et al. Comparison of two methods to detect publication bias in meta-analysis. JAMA. 2006;295:676–680. doi: 10.1001/jama.295.6.676. [DOI] [PubMed] [Google Scholar]
- 28.Tzanakis N, Gazouli M, Rallis G, et al. Vascular endothelial growth factor polymorphisms in gastric cancer development, prognosis, and survival. J Surg Oncol. 2006;94:624–630. doi: 10.1002/jso.20619. [DOI] [PubMed] [Google Scholar]
- 29.Chae YS, Kim JG, Sohn SK, et al. Investigation of vascular endothelial growth factor gene polymorphisms and its association with clinicopathologic characteristics in gastric cancer. Oncology. 2006;71:266–272. doi: 10.1159/000106788. [DOI] [PubMed] [Google Scholar]
- 30.Bae SJ, Ahn DH, Hong SP, et al. Gender-specific association between polymorphism of vascular endothelial growth factor (VEGF 936C>T) gene and patients with stomach cancer. Yonsei Med J. 2008;49:783–791. doi: 10.3349/ymj.2008.49.5.783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ke Q, Liang J, Wang LN, et al. Potentially functional polymorphisms of the vascular endothelial growth factor gene and risk of gastric cancer. Mol Carcinog. 2008;47:647–651. doi: 10.1002/mc.20435. [DOI] [PubMed] [Google Scholar]
- 32.Al-Moundhri MS, Al-Nabhani M, Burney IA, et al. Gastric cancer risk predisposition and prognostic significance of vascular endothelial growth factor (VEGF) gene polymorphisms: a case-control study in an Omani population. Mol Carcinog. 2009;48:1170–1176. doi: 10.1002/mc.20572. [DOI] [PubMed] [Google Scholar]
- 33.Guan X, Zhao H, Niu J, et al. The VEGF -634G>C promoter polymorphism is associated with risk of gastric cancer. BMC Gastroenterol. 2009;9:77. doi: 10.1186/1471-230X-9-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tahara T, Shibata T, Nakamura M, et al. Effect of polymorphisms in the 3′ untranslated region (3′-UTR) of vascular endothelial growth factor gene on gastric cancer and peptic ulcer diseases in Japan. Mol Carcinog. 2009;48:1030–1037. doi: 10.1002/mc.20554. [DOI] [PubMed] [Google Scholar]
- 35.Xia HZ, Wu Q, Liu Y, et al. Association ofVEGF rs3025039 and rs3025021 with VEGF and COX-2 expression in gastric cancer tissues. J Clin Exp Pathol. 2010;26:139–143. [Google Scholar]
- 36.Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol. 2005;23:1011–1027. doi: 10.1200/JCO.2005.06.081. [DOI] [PubMed] [Google Scholar]
- 37.Uthoff SM, Duchrow M, Schmidt MH, et al. VEGF isoforms and mutations in human colorectal cancer. Int J Cancer. 2002;101:32–36. doi: 10.1002/ijc.10552. [DOI] [PubMed] [Google Scholar]
- 38.Yuan A, Yu CJ, Chen WJ, et al. Correlation of total VEGF mRNA and protein expression with histologic type, tumor angiogenesis, patient survival and timing of relapse in non-small-cell lung cancer. Int J Cancer. 2000;89:475–483. doi: 10.1002/1097-0215(20001120)89:6<475::aid-ijc2>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 39.Krippl P, Langsenlehner U, Renner W, et al. A common 936 C/T gene polymorphism of vascular endothelial growth factor is associated with decreased breast cancer risk. Int J Cancer. 2003;106:468–471. doi: 10.1002/ijc.11238. [DOI] [PubMed] [Google Scholar]
- 40.Lin CC, Wu HC, Tsai FJ, et al. Vascular endothelial growth factor gene -460 C/T polymorphism is a biomarker for prostate cancer. Urology. 2003;62:374–377. doi: 10.1016/s0090-4295(03)00268-1. [DOI] [PubMed] [Google Scholar]
- 41.Taylor JG, Choi EH, Foster CB, et al. Using genetic variation to study human disease. Trends Mol Med. 2001;7:507–512. doi: 10.1016/s1471-4914(01)02183-9. [DOI] [PubMed] [Google Scholar]