Abstract
The aim of the present study was to examine the expression of glutamine synthetase (GS) in rat retinal Müller cells induced by different levels of hydrostatic pressure using a novel pressure mechanism. pH, PCO2 or PO2 in culture medium as determined by gas analysis was used to examine the pressure mechanism. GS expression in the Müller cells at different levels of hydrostatic pressure (0, 20, 40, 60 and 80 mmHg/24 h) was examined using immunofluorescence, real-time PCR and Western blotting. There was no significant difference in pH, PCO2 or PO2 in the culture medium by gas analysis at the different hydrostatic pressure levels (p>0.05). Immunofluorescence staining showed that GS was expressed in the Müller cells. The expression of GS in the 40 mmHg/24 h and the 60 mmHg/24 h groups was increased significantly compared to that in the 0 mmHg/24 h group (p<0.05). These results suggest that the pressure mechanism which was constructed was effective and that moderate pressure promotes the up-regulation of GS in active Müller cells.
Keywords: glutamine synthetase, pressure mechanism, Müller cells
Introduction
Glaucoma is an irreversible, chronic, blinding disease characterized by visual field loss, optic nerve head cupping and retinal ganglion cell loss. Glaucoma is considered the second most prevalent cause of blindness in the world. There are several risk factors associated with glaucoma, such as intraocular pressure (IOP), age, race, family history and vascular disease. Among these, increased IOP is the most common risk factor. Reducing IOP is presently the most accepted and most practiced therapeutical approach for glaucoma patients. However, some patients continue to lose their sight despite apparently adequate pressure control. This suggests that reducing IOP does not provide complete protection, whereas other factors unrelated to IOP may be active (1–4).
Glutamate is a normal constituent of the retina, but in high non-physiological concentrations it causes neuronal damage and death. Normally, glutamate is rapidly removed by Müller cells in the retina (5–8). Glutamine synthetase (GS), an important enzyme located mainly in Müller cells, catalyzes the amidation of glutamate to glutamine. It is one of the major mechanisms for the clearance of extracellular glutamate and in the past has been used as a specific label for Müller cells (8–13).
Although various studies suggest that an increase in vitreal and retinal glutamate levels causes glaucomatous damage (14–16), the issue remains controversial with research data both supporting or opposing this theory. We would expect that elevated IOP leads to an increase in glutamate, or that Müller cells respond normally, which would lead to an increased expression of GS (9,17–22). This study investigated the expression of GS in Müller cells under different pressure in vitro.
There are many animal models of glaucoma in vivo (23). However, only one type of method is used in vitro (24–27). Briefly, a glass chamber or oven is re-equipped into an incubator. This pressure system is complicated and expensive inspite of its classical and extensive use. In the present experiment, a novel pressure mechanism was used, which was simpler and less costly than the previous described method.
Materials and methods
Pressure mechanism
T75 culture flasks were used to construct the pressure mechanism (Fig. 1). An air mixture of 95% air and 5% CO2 was pumped to obtain the relevant pressure. The pressure level of the model was 0, 20, 40, 60 and 80 mmHg, respectively, in accordance with human IOP in glaucoma, and it was adjusted every 4 h. The time period of the pressure was 24 h. All of the flasks were placed in an incubator at 37˚C.
Figure 1.

Pressure mechanism. Laboratory film (Pechiney) was used to seal up the interfaces. All of the operations involving refreshment of the medium or adjustment of the pressure were rapidly performed.
The possibility that the elevated hydrostatic pressure could alter the gas exchange was assessed by analyzing the blood gas in the culture medium in the pressure (20, 40, 60 and 80 mmHg) and control (0 mmHg) culture groups before and after 12 h of pressurization. The tightness was assessed by a pressure gauge.
Cell culture
Experiments were performed on newborn (0–3 days) Sprague-Dawley (SD) rats (Slaccas Laboratory Animal Co., Ltd.). SD rats were decapitated, and their retinas were quickly dissected in cold D-Hank’s solution (Anresco). The mixture was transferred to sterile centrifuge tubes and centrifuged at 1,000 rpm for 10 min. The supernatant was discarded. Trypsin (0.125%) (Anresco) was added for digestion. The mixture was incubated at 37˚C for 15 min. At the end of the designated time, DMEM/F12 medium (Gibco) supplemented with 2 mM glutamine, 100 U/ml penicillin and 100 μg/ml streptomycin and 10% fetal bovine serum (FBS; Sijiqing) was added to terminate the digestion. The digests were filtered using a 200-mesh nylon sieve and centrifuged at 1,000 rpm for 10 min. The supernatant was discarded. The cell suspension was cultured in T75 culture flasks at 37˚C in humidified air containing 5% CO2. When confluent, the cells were washed once with D-Hank’s solution, detached from the flask by treatment with 0.125% trypsin, washed with complete cell culture medium and split 1:2 into fresh flasks.
Immunofluorescence
The cells on the coverslips were incubated at 37˚C for 1 day and washed three times (10 min/wash) with PBS. The cells were fixed with 4% paraformaldehyde at room temperature for 10 min and incubated with 0.3% Triton X-100 at 37˚C for 10 min. The cells were washed three times (10 min/wash) with PBS, blocked with 10% goat serum in PBS and subsequently incubated with a rabbit anti-rat polyclonal antibody against GS (1:5,000; Abcam) as an identity marker for Müller cells. The cells were then incubated overnight at 4˚C. The following day, the cells were incubated with the secondary anti-rabbit IgG-Cy3 antibodies (1:200; BioLegend) at 37˚C in darkness for 1 h. After three washes with PBS, the cells on the coverslips were mounted on glass slides with Histomount. The cells were viewed under an Axio microscope, and images were acquired with a digital camera.
Western blot analysis
Fifteen samples, three at each pressure (0, 20, 40, 60 and 80 mmHg/24 h), were used for Western blotting. Cells were washed twice in PBS. The protein concentration was determined by a radioimmunoprecipitation assay and lysed in 2X Laemmli buffer. Protein extracts were boiled for 10 min and centrifuged at 16,000 x g. Proteins were separated on 12% SDS-PAGE and transferred to PVDF membranes (Millipore). The membranes were soaked in Tris-buffered saline (20 mmol/l Tris-Cl, 140 mmol/l NaCl, pH 7.5) containing 5% skimmed milk and 0.1% Tween-20 for 1 h at room temperature. The primary antibody used was GS (1:2,500; Abcam). Blots were incubated with the primary antibody overnight at 4˚C. Anti-β-actin antibody (1:3,000; Abcam) was used as a reference to normalize the intensities of the immunoreactions with the different antibodies. Then, after several washes, the membranes were incubated with a secondary antibody against rabbit-IgG (Invitrogen) for 1 h at room temperature in darkness. The protein bands were scanned, and images were quantified with Odyssey (Li-CDR).
Real-time PCR (RT-PCR)
Total RNA was isolated from the individual samples using the TRIzol® reagent (Invitrogen). The concentration and purity of the RNA preparations were determined by measuring the absorbance at 260/280 nm in a spectrophotometer (Beckman). Total RNA was reversetranscribed into cDNA in a 20-μl reaction containing 2 μg RNA, 4 μl 5X M-MLV buffer, 2 μl of dNTP, 1 μl random hexamer primer, 0.5 μl of RNase inhibitor and 1 μl of M-MLV RTase. Reactions were performed at 25˚C for 10 min, at 42˚C for 60 min and at 70˚C for 10 min.
The following targets were analyzed by RT-PCR. The nucleotide sequences of the primers were based on published cDNA sequences (Gene Bank). The primer sequences used for RT-PCR were as follows: GS, 5′-ccgctcttcgtctcgttc-3′ and 5′-ctg cttgatgcctttgtt-3′; β-actin, 5′-cccatctatgagggttacgc-3′ and 5′-ttta atgtcacgcacgatttc-3′. RT-PCRs were performed on a LightCycler instrument (Rotor Gene) with SYBR Green I, according to the manufacturer’s recommendations. Cycling conditions were as follows: initial denaturation at 94˚C for 5 min, followed by 40 cycles at 94˚C for 30 sec, 55˚C for 30 sec and 72˚C for 30 sec. The melting curve analysis of the final PCR products was carried out from 50 to 99˚C at 1˚C intervals.
Statistical analysis
Data are presented as the mean ± SD (n=3–6 in each group). Data were analyzed using one-way ANOVA followed by the Turkey’s test. A p-value of <0.05 was accepted as indicative of statistical significance.
Results
Pressure mechanism
We analyzed pH, PCO2 or PO2 from samples of the culture medium 12 h after exposure to the different pressure levels (0, 20, 40, 60 and 80 mmHg). There was no significant difference in these values between the control (0 mmHg) and each pressure (20, 40, 60 and 80 mmHg) group (Table I).
Table I.
Measurement of pressure, pH, PCO2 and PO2 after 12 h.
| Treatment (mmHg)
|
|||||
|---|---|---|---|---|---|
| Control | 20 | 40 | 60 | 80 | |
| Time (h) | 12 | 12 | 12 | 12 | 12 |
| Total pressure (mmHg) | 0 | 19±3 | 41±2 | 60±2 | 78±2 |
| pH | 7.69±0.03 | 7.64±0.02 | 7.67±0.04 | 7.65±0.02 | 7.61±0.03 |
| PO2 (mmHg) | 168.0±1.9 | 166.4±1.6 | 168.0±1.6 | 164.5±1.5 | 169.1±2.0 |
| PCO2 (mmHg) | 27.5±0.4 | 27.1±0.1 | 26.5±0.4 | 25.9±0.4 | 5.6±0.2 |
Statistical analysis (Student’s unpaired t-test) showed no significant difference in pH, PCO2 and PO2 in the different samples (p>0.05, n=3).
Immunofluorescence
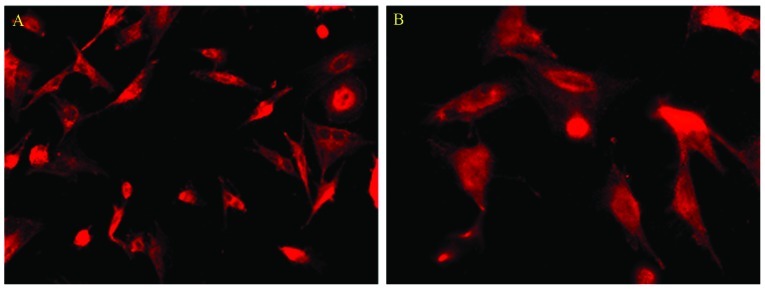
We characterized the cultured rat Müller cells by their expression of GS as assessed by immunocytochemical staining. Cells in the culture system showed positive labeling for GS (Fig. 2). Using this immunocytochemical labeling, the cultured cells were determined to be Müller cells.
Figure 2.
Identification of the Müller cells. Glutamine synthetase (red) was used to label the Müller cells.
Western blot analysis
Western blot analyses of the samples at different pressure showed that the expression of GS protein was variable. The expression of the GS protein in the 20 mmHg/24 h group was decreased compared to the control (0 mmHg/24 h) group. The expression of GS protein was increased in the 40 mmHg/24 h and the 60 mmHg/24 h groups. The expression of GS protein was slightly decreased in the 80 mmHg/24 h group (Fig. 3).
Figure 3.
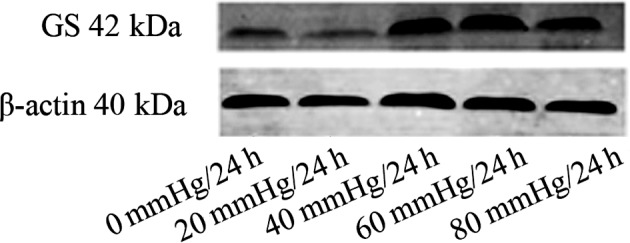
Glutamine synthetase protein expression at different pressures.
RT-PCR analysis
RT-PCR was performed to examine whether the mRNA expression levels of GS were different among the different pressure groups. Data showed that the expression of GS mRNA was decreased in the 20 mmHg/24 h group compared to the control (0 mmHg/24 h) group. Hereafter, the GS mRNA expression was increased in the 40 mmHg/24 h group which further increased in the 60 mmHg/24 h group, and then decreased in the 80 mmHg/24 h group. However, the difference was only statistically significant between the control (0 mmHg/24 h) and the 40 mmHg/24 h and 60 mmHg/24 h groups, and there was no statistical significance between the control (0 mmHg/24 h) group and the 20 mmHg/24 h and 80 mmHg/24 h groups (Fig. 4). The results of the expression of GS mRNA were in accord with the results of the expression of the GS protein.
Figure 4.
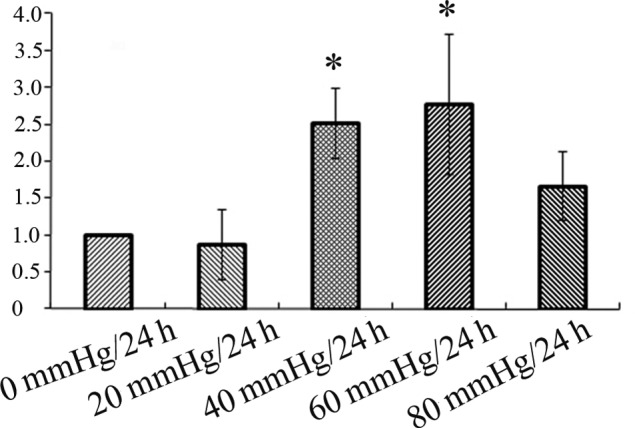
Glutamine synthetase mRNA expression at different pressures (0, 20, 40, 60 and 80 mmHg/24 h). *p<0.05 vs. 0 mmHg/24 h.
Discussion
The present results demonstrated that i) the pressure mechanism that we constructed was effective and that ii) moderate pressure promotes the up-regulation of GS in active Müller cells.
In recent years, a number of experimental techniques have been used to chronically raise the IOP in a short period of time. These techniques include laser photocoagulation of the trabecular meshwork, episcleral vein cauterization, episcleral vein injection of hypertonic saline and injection of substances into the anterior chamber to obstruct aqueous outflow (28). Compared to glaucomatous models in vivo, in vitro models are fewer. In this study, many precautions were taken to limit artifacts from the experimental method. Laboratory film (Pechiney) was used to seal up the interfaces. To avoid artifacts due to ‘on-off’ changes in pressure, all of the operations involving refreshment of the medium or adjustment of pressure were carried out within 5 min. The results (Table I) showed that the pressure mechanism was effective.
Müller cells are the major glial cells of the retina and constitute a functional link between neurons and vessels. They maintain the integrity of the blood-retinal barrier, remove metabolic waste and maintain the balance of the retinal extracellular environment (ions, water, pH, neurotransmitters). In the normal retina, Müller cells play a crucial role in degrading the level of glutamate. Glutamate taken up by Müller cells is converted, in large part, intracellularly, to glutamine through GS (29–32).
In the present study, the change in GS expression in the Müller cells was variable depending on the change of pressure. At 20 mmHg of pressure, the expression of GS was lower than that in the control. When the pressure was increased to 40 mmHg, the expression of GS was markedly higher (Figs. 3 and 4). The expression level of GS was the highest at a pressure of 60 mmHg and from then the level declined. The present results showed that at an increased hydrostatic pressure, Müller cells may malfunction. However, by further elevating the hydrostatic pressure, Müller cells reacted rapidly, with a return of functionality. Yet, at an excessively high pressure, Müller cells may be damaged. Since Müller cells are the main storage of glycogen (33) and obtain ATP from glycolysis rapidly, longterm exposure to noxious stress (extensively high hydrostatic pressure) which induces ATP depletion, inhibits GS activity.
Although the relationship between the elevated hydrostatic pressure and the increase in glutamate was not determined in the present study, GS up-regulation in Müller cells at a moderate hydrostatic pressure suggests that conversion ability of glutamate in the Müller cells was reinforced. Therefore, we propose that Müller cells adaptively react to GS up-regulation to degrade the level of glutamate at an elevated but moderate hydrostatic pressure.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (no. 81070728), the Shanghai Leading Academic Discipline Project (no. S30205), the Shanghai Municipal Education Committee Project (no. 10YZ38) and the Shanghai Natural Science Foundation (no. 08RZ1413900).
References
- 1.John C, Morrison MD. Integrins in the optic nerve head: potential roles in glaucomatous optic neuropathy. Trans Am Ophthalmol Soc. 2006;104:453–477. [PMC free article] [PubMed] [Google Scholar]
- 2.Bull ND, Limb GA, Martin KR. Human Müller stem cell (MIO-M1) transplantation in a rat model of glaucoma: survival, differentiation and integration. Invest Ophthalmol Vis Sci. 2008;49:3449–3456. doi: 10.1167/iovs.08-1770. [DOI] [PubMed] [Google Scholar]
- 3.Quigley HA. New paradigms in the mechanisms and management of glaucoma. Eye. 2005;19:1241–1248. doi: 10.1038/sj.eye.6701746. [DOI] [PubMed] [Google Scholar]
- 4.Sommer A. Intraocular pressure and glaucoma. Am J Ophthalmol. 1989;107:186–188. doi: 10.1016/0002-9394(89)90221-3. [DOI] [PubMed] [Google Scholar]
- 5.Carter-Dawson L, Shen F, Harwerth RS, Smith EL, Crawford ML, Chang A. Glutamine immunoreactivity in Müller cells of monkey eyes with experimental glaucoma. Exp Eye Res. 1998;66:537–545. doi: 10.1006/exer.1997.0447. [DOI] [PubMed] [Google Scholar]
- 6.Massey SC. Cell types using glutamate as a neurotransmitter in the vertebrate retina. In: Osborne NN, Chader CJ, editors. Progress in Retinal Research. Pergamon; Oxford: 1990. pp. 399–425. [Google Scholar]
- 7.Pow DV, Robinson SR. Glutamate in some retinal neurons is derived solely from glia. Neuroscience. 1994;60:355–366. doi: 10.1016/0306-4522(94)90249-6. [DOI] [PubMed] [Google Scholar]
- 8.Ambati J, Chalam KV, Chawla DK. Elevated gammaaminobutyric acid, glutamate, and vascular endothelial growth factor levels in the vitreous of patients with proliferative diabetic retinopathy. Arch Ophthalmol. 1997;115:1161–1166. doi: 10.1001/archopht.1997.01100160331011. [DOI] [PubMed] [Google Scholar]
- 9.Shen F, Chen B, Danias J, Lee KC, Lee H, Su Y. Glutamate-induced glutamine synthetase expression in retinal Müller cells after short-term ocular hypertension in the rat. Invest Ophthalmol Vis Sci. 2004;45:3107–3112. doi: 10.1167/iovs.03-0948. [DOI] [PubMed] [Google Scholar]
- 10.Riepe RE, Norenberg MD. Müller cell localization of glutamine synthetase in the rat retina. Nature. 1977;268:654–655. doi: 10.1038/268654a0. [DOI] [PubMed] [Google Scholar]
- 11.Rauen T, Wiebner M. Fine tuning of glutamate uptake and degradation in glial cells: common transcriptional regulation of GLAST1 and GS. Neurochem Int. 2000;37:179–189. doi: 10.1016/s0197-0186(00)00021-8. [DOI] [PubMed] [Google Scholar]
- 12.Linser PJ, Sorrentino M, Moscona AA. Cellular compartmentalization of carbonic anhydrase-C and glutamine synthethase in developing and mature mouse neural retina. Brain Res. 1984;315:65–71. doi: 10.1016/0165-3806(84)90077-4. [DOI] [PubMed] [Google Scholar]
- 13.Newman E, Reichenbach A. The Müller cells: a functional element of the retina. Trends Neurosci. 1996;19:307–312. doi: 10.1016/0166-2236(96)10040-0. [DOI] [PubMed] [Google Scholar]
- 14.Dreyer EB, Zurakowski D, Schumer RA, Podos SM, Lipton SA. Elevated glutamate in vitreous body of humans and monkeys with glaucoma. Arch Opthalmol. 1996;114:299–305. doi: 10.1001/archopht.1996.01100130295012. [DOI] [PubMed] [Google Scholar]
- 15.Brooks DE, Garcia GA, Dreyer EB, Zurakowski D, Franco-Bourland RE. Vitreous body glutamate concentration in dogs with glaucoma. Am J Vet Res. 1997;58:864–867. [PubMed] [Google Scholar]
- 16.Dkhissi O, Chanut E, Wasowicz M. Retinal TUNEL-positive cells and high glutamate levels in vitreous humor of mutant quail with a glaucoma-like disorder. Invest Ophthalmol Vis Sci. 1999;40:990–995. [PubMed] [Google Scholar]
- 17.Carter-Dawson L, Shen F, Harwerth RS, Smith EL, Crawford ML, Chuang A. Glutamine immunoreactivity in Müller cells of monkey eyes with experimental glaucoma. Exp Eye Res. 1998;66:537–545. doi: 10.1006/exer.1997.0447. [DOI] [PubMed] [Google Scholar]
- 18.Li S, Wang J, Wang D, Bai H. The experimental study of effect of pressure on rat Müller cells in vitro. Chin J Ophthamol. 2005;41:325–329. [PubMed] [Google Scholar]
- 19.Woldemussie E, Wijono M, Ruiz G. Müller cell response to laser-induced increase in intraocular pressure in rats. Glia. 2004;47:109–119. doi: 10.1002/glia.20000. [DOI] [PubMed] [Google Scholar]
- 20.Dreyer EB, Zurakowski D, Schumer RA, Podos SM, Lipton SA. Elevated glutamate in vitreous body of humans and monkeys with glaucoma. Arch Opthalmol. 1996;114:299–305. doi: 10.1001/archopht.1996.01100130295012. [DOI] [PubMed] [Google Scholar]
- 21.Carter-Dawson L, Crauford ML, Harwerth RS. Vitreal glutamate concentration in monkeys with experimental glaucoma. Invest Opthalmol Vis Sci. 2002;43:2633–2637. [PubMed] [Google Scholar]
- 22.Honkanen RA, Baruah S, Zimmerman MB. Vitreous amino acid levels in patients with glaucoma undergoing vitrectomy. Arch Ophthalmol. 2003;121:183–188. doi: 10.1001/archopht.121.2.183. [DOI] [PubMed] [Google Scholar]
- 23.Johnson TV, Tomarev SI. Rodent models of glaucoma. Brain Res Bull. 2010;81:349–358. doi: 10.1016/j.brainresbull.2009.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Agar A, Yip SS, Hill MA, Coroneo MT. Pressure-related apoptosis in neuronal cell lines. J Neurosci Res. 2000;60:495–503. doi: 10.1002/(SICI)1097-4547(20000515)60:4<495::AID-JNR8>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 25.Sappington RM, Chan M, Calkins DJ. Interleukin-6 protects retinal ganglion cells from pressure-induced death. Invest Opthalmol Vis Sci. 2006;47:2932–2942. doi: 10.1167/iovs.05-1407. [DOI] [PubMed] [Google Scholar]
- 26.Salvador-Silva M, Ricard CS, Agapova OA, Yang P, Hernandez MR. Expression of small heat shock proteins and intermediate filaments in the human optic nerve head astrocytes exposed to elevated hydrostatic pressure in vitro. J Neurosci Res. 2001;66:59–73. doi: 10.1002/jnr.1197. [DOI] [PubMed] [Google Scholar]
- 27.Tezel G, Wax MB. Hypoxia-inducible factor 1α in the glaucomatous retina and optic nerve head. Arch Ophthalmol. 2004;122:1348–1356. doi: 10.1001/archopht.122.9.1348. [DOI] [PubMed] [Google Scholar]
- 28.Reichenbach A, Wurm A, Pannicke T, Landiev I, Wiedemann P, Bringmann A. Müller cells as players in retinal degeneration and edema. Graefe’s Arch Clin Exp Ophthalmol. 2007;245:627–636. doi: 10.1007/s00417-006-0516-y. [DOI] [PubMed] [Google Scholar]
- 29.Walsh N, Valter K, Stone J. Cellular and subcellular patterns of expression of bFGF and CNTF in the normal and light stressed adult rat retina. Curr Eye Res. 2001;72:495–501. doi: 10.1006/exer.2000.0984. [DOI] [PubMed] [Google Scholar]
- 30.Harada T, Harada C, Kohsaka S. Microglia-Müller glia cell interactions control neurotrophic factor production during lightinduced retinal degeneration. Neurosei. 2002;22:9228–9236. doi: 10.1523/JNEUROSCI.22-21-09228.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pow DV, Crook DK. Direct immunocytochemical evidence for the transfer of glutamine from glial cells to neurons: use of specific antibodies directed against the D-steroisomers of glutamate and glutamine. Neuroscience. 1996;70:295–302. doi: 10.1016/0306-4522(95)00363-n. [DOI] [PubMed] [Google Scholar]
- 32.Johnson TV, Tomarev SI. Rodent models of glaucoma. Brain Research Bulletin. 2010;81:349–358. doi: 10.1016/j.brainresbull.2009.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tsacopoulos M. Metabolic signaling between neurons and glial cells: a short review. J Physiol Paris. 2002;96:283–288. doi: 10.1016/s0928-4257(02)00017-7. [DOI] [PubMed] [Google Scholar]