Abstract
External genitalia development occurs through a combination of hormone independent, hormone dependent, and endocrine pathways. Perturbation of these pathways can lead to abnormal external genitalia development. We review human and animal mechanisms of normal and abnormal external genitalia development, and we evaluate abnormal mechanisms that lead to hypospadias. We also discuss recent laboratory findings that further our understanding of animal models of hypospadias.
Keywords: external genitalia, development, hypospadias
Introduction
Our knowledge of the embryology of external genitalia development is augmented by understanding the molecular pathways that coordinate sex differentiation. Elucidation of molecular mechanisms of genital development has stemmed from gene analysis, evaluation of disruptions of normal endocrine pathways, congenital abnormalities, and animal models. In this paper we review embryologic sex differentiation and external genitalia development in both humans and animals. Since many pathways that affect gonadal sex determination and development also affect external genitalia development and may lead to abnormalities of external genitalia, we also briefly discuss gonadal sex determination and development.
Development of external genitalia occurs via three main pathways: 1) androgen-independent, 2) androgen-dependent, and 3) endocrine/environmental influence. A complex interaction of these three pathways exists, and external genitalia development should be evaluated in the context of all three (Figure 1). Endocrine and environmental influences affect both androgen-independent and androgen-dependent pathways on a genetic and epigenetic basis.
Figure 1. External Genitalia Development Processes.

The development of external genitalia occurs through androgen dependent, androgen independent, and endocrine/environmental influences. These processes sometimes overlap and interact during development.
Human Embryologic Gonadal Sex Determination and Development
In human fetuses during the ambisexual stage, primordial germ cells migrate beginning in the fifth week from their site of origin in the hindgut/allantois into the genital ridges. Sex cords are formed within the gonads in the sixth embryonic week, and the urogenital folds and labioscrotal folds begin to form in the perineum. The Mullerian and Wolffian ducts form around the seventh embryonic week.
Gonadal differentiation occurs from the tenth through twelfth embryonic weeks. The Sex-determining Region of the Y chromosome (SRY) gene is required to initiate signaling for male gonadal differentiation (Sinclair et al, 1990). SRY gene expression results in differentiation of Sertoli cells, which then produce Mullerian inhibiting substance (MIS). SRY box-related gene 9 (SOX9) is an SRY-related transcription factor found only in male embryos during gonadal sex-determination (Agrawal et al, 2009). SOX9 is upregulated by SRY, and is thought to aid in Sertoli cell differentiation (Sekido et al, 2004). Additional genes involved in gonadogenesis and normal SRY levels of expression are GATA4 and FOG2 (Tevosian et al, 2002).
During embryonic weeks nine and ten SRY causes differentiation of Leydig cells, which produce testosterone. In the presence of fetal testicular androgens the Wolffian ducts persist and develop into the epididymis, vas deferens, and seminal vesicles. Testosterone is also a substrate for the enzyme, 5α-reductase, which converts testosterone to dihydrotestosterone. This even more potent androgen drives growth of the external genitalia and prostate (Wilson et al, 1981). The trophic and organizational effects of both testosterone and dihydrotestosterone are elicited following interaction of these hormones with the hormone-binding pocket of the androgen receptor, a nuclear transcription factor. In the absence of functional androgen receptors, the Wolffian ducts degenerate, the prostate does not develop from the urogenital sinus, and the external genitalia develop according to the female pattern. Conversely, exposure of the female urogenital sinus to endogenous or exogenous androgens elicits prostatic development in females and masculinization of the external genitalia (Hotchkiss et al, 2007a).
Absence of the SRY gene results in ovarian differentiation. In females, in which testosterone, dihydrotesterone, and MIS are absent during critical periods, the Mullerian ducts persist and develop into the Fallopian tubes, uterus, cervix, and the cranial portion of the vagina, and due to the absence of androgens, the Wolffian ducts regress. WNT4 is required for Mullerian duct formation (Vainio et al, 1999), and DAX1 (dosage-sensitive sex reversal, adrenal hypoplasia congenita, X chromosome) has been demonstrated to suppress SF1 transcriptional activity, which is required for adrenal and gonadal development (Ito et al, 1997).
Human Ambisexual External Genitalia Development
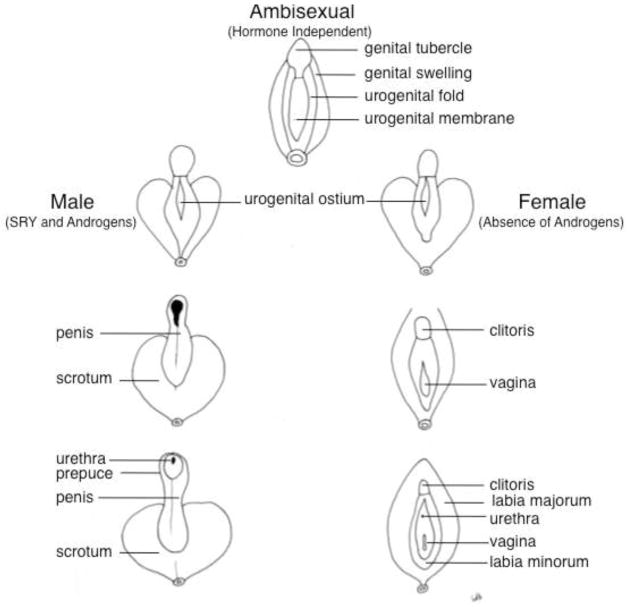
Development of male and female external genitalia begins with the formation of structures constituting the ambisexual stage that in turn undergo sex differentiation to generate the male and female forms of external genitalia. In the fifth embryonic week, the cloacal folds form from mesenchymal cells migrating into the perineum. These mesenchymal cells pile up in the midline and form an elevation in the perineum called the genital tubercle. The genital tubercle is located just cranial to the midline opening of the endodermal urogenital sinus called the urogenital ostium. The urogenital ostium is flanked laterally by the urogenital folds and the genital swellings/labioscrotal folds (Figure 2). Endodermal epithelial cells from the urogenital sinus are thought to invade into the genital tubercle to form the solid midline epithelial urethral plate. Formation of these structures occurs identically in male and female fetuses in the ambisexual stage of external genitalia development through a hormone-independent process. In humans this ambisexual stage of development occurs between gestational weeks 8 and 12. In the mouse, the ambisexual stage occurs between days E11 and E16 of gestation (Yamada et al, 2003).
Figure 2.
Genital tubercle differentiation from ambisexual stage to male and female external genitalia. The ambisexual stage is hormone independent. SRY and androgens are required for male development, while female development occurs in the absence of androgens.
Genes Involved in External Genitalia Development
Sonic hedge hog (Shh) is a gene that regulates development of two major body appendages, limbs and the genital tubercle. Shh is expressed within the genital tubercle in urethral plate epithelium in mice and has been demonstrated to be involved in formation of the ambisexual stage and subsequent initiation of sex differentiation of the penis (Miyagawa et al, 2011). Shh signaling is required for genital tubercle development in mice (Lin et al, 2009, Haraguchi et al, 2001) and for formation of the genital appendage (gonopodium) in male mosquitofish (Ogino et al, 2004). Shh is also expressed in hindgut, urethral plate, and cloacal epithelium, and it has been suggested Shh functions in proper cloacal formation (Haraguchi et al, 2000, Haraguchi et al, 2001). Its absence in Shh-null mice results in disruption of genital tubercle development and a persistent cloaca (Haraguchi et al, 2001, Seifert et al, 2009a). In the absence of Shh, the following genes are downregulated: Wnt5a, Fibroblast growth factor 8 (Fgf8), Fgf10, Bmp2, and Bmp4 (Haraguchi et al 2001, Perriton et al, 2002). Wnt5a, beta-catenin, and Fgf8 act downstream of Shh, and beta-catenin gain-of-function mutations or exogenous upregulation of beta-catenin can rescue genital tubercle development in Shh-null mice and “rescue or recover” Fgf8 expression (Miyagawa et al, 2009a). Fgf8 is also expressed in the urethral plate, and ectopic Fgf8 beads in vitro promote genital tubercle outgrowth (Haraguchi et al, 2000). The Wnt/beta-catenin pathway has been demonstrated to be essential in androgen regulated pathways of genital tubercle development in embryonic mice, and overexpression of beta-catenin resulted in “masculinization” of female mice characterized by prepuce hypertrophy and enlargement of the external genitalia (Miyagawa et al, 2009b). Although Fgf8 has been proposed as a mediator of genital tubercle outgrowth, additional research has shown that Fgf8 may only be a marker of external genitalia development since germline deletion of Fgf8 in embryonic mice resulted in normal external genitalia (Seifert et al, 2009b)(Table 1). Whether or not Fgf8 functions in genital tubercle development awaits further analysis. Human studies confirm SHH involvement in penile development. In human embryos, SHH is expressed in human fetal penises during development with the greatest expression demonstrated with immunohistochemistry during urethral tubularization at 14 weeks of gestation (Shehata et al, 2011).
Table 1.
Genes and their involvement in external genitalia development
| Gene | Action |
|---|---|
| SRY | Male gonadal differentiation |
| SOX9 | Facilitates Sertoli cell differentiation |
| GATA4 | Male gonandogenesis |
| FOG2 | Male gonandogenesis |
| WNT4 | Mullerian duct formation |
| DAX1 | Female gonandogenesis |
| SHH | Genital tubercle development |
| WNT5a | Acts downstream of SHH in genital tubercle development |
| FGF8 | Acts downstream of SHH in genital tubercle development |
| ATF3 | Upregulated in hypospadias |
Genetic Syndromes
Molecular mechanisms of external genitalia development have been elucidated from understanding the genes involved in congenital anomaly syndromes affecting external genitalia. An example of this is the CHARGE syndrome, which results in coloboma (defect or gap which may occur in multiple structures in the eye and may result in blindness), heart defects, atretic choanae (lack of development of the nasal passage), retarded growth and development (growth hormone deficiency or short stature), genital hypoplasia, and ear anomalies/deafness. Autosomal dominant mutations in the chromodomain-helicase-DNA-binding protein 7 (CHD7) gene result in the CHARGE syndrome, and a variety of mutations in CHD7 are found in over 75% of CHARGE syndrome patients (Blake and Prasad, 2006). Genital hypoplasia is more evident in males with CHARGE syndrome and may result in cryptorchidism, micropenis, and/or hypogonadotrophic hypogonadism (Pinto et al, 2005). The syndrome of Wilms tumor, aniridia, genitourinary abnormalities, and mental retardation (WAGR), which results from chromosomal deletion at 11p13 and affects the WT1 gene, may result in hypospadias, cryptorchidism, or ambiguous genitalia. The WT1 gene is expressed in the fetal gonadal ridges and is thought to interact in the Wnt/beta-catenin pathway, which plays an essential role in androgenic genital tubercle development. Denys-Drash syndrome and Frasier syndrome are both associated with defects in WT1 and a spectrum of external genitalia abnormalities including ambiguous genitalia, hypospadias, and cryptorchidism (Le Caignec et al, 2007). Mutations in WT1 have been associated with hypospadias and micropenis (Wang et al, 2004).
Human External Genitalia Sexual Differentiation
The genital tubercle elongates to become the penis in males under the influence of fetal testicular androgens. In the absence of androgens in females the genital tubercle exhibits minimal growth in size and becomes the clitoris. A portion of the cloacal folds becomes the urogenital folds, which laterally bound the urogenital ostium with the labioscrotal folds developing laterally. The labioscrotal folds fuse in the midline to form the scrotum in males, but remain separate forming the labia majora in females (Figure 2). As the solid epithelial urethral plate elongates towards the tip of the genital tubercle, it canalizes to form a groove on the ventral surface of the genital tubercle bounded by urethral folds in males. These urethral folds fuse in the midline converting the urethral groove into the penile urethra. Failure of fusion of the penile urethral folds from embryonic weeks eleven to sixteen results in hypospadias, an abnormal opening of the urethra proximal to its normal location at the tip of the penis.
Hormones Involved in External Genitalia Development
Sexual differentiation of external genitalia is hormone dependent. Thus, disruption of normal endocrine function can adversely affect external genitalia development. Normal endocrine function entails production of the appropriate hormones by the gonads, steroid metabolizing enzymes within the external genitalia, and functional sex hormone receptors. Absence or impairment of any of these essential elements through genetic alterations or environmental influence may adversely affect external genitalia development.
Congenital/genetic disorders resulting in abnormal external genitalia development in humans includes absence of the Y chromosome containing SRY or mutations in the SRY gene promoter or coding region in males, which prevents testicular development and thus eliminates testosterone production (Assumpcao, 2005). Defective androgen receptors result in androgen insensitivity syndrome, which has a spectrum of phenotypes depending on the degree of defectiveness of the androgen receptor. A fully non-functional androgen receptor will result in an XY male with phenotypically female external genitalia (Galani et al, 2008). Mutations in the androgen receptor gene are associated with defects in masculinization of the external genitalia and hypospadias, a triad of abnormal urethral, penile, and foreskin development (Wang et al, 2004). Defects in the aromatase gene, which converts testosterone to estrogen results in elevated testosterone, which causes ambiguous genitalia in 46 XX females with virilization of the clitoris to a phallus-like structure (Lin et al, 2007).
Animal studies have solidified our knowledge of the hormone-dependent pathways of external genitalia development. Blocking dihydrotestosterone production by administration of a 5 alpha-reductase inhibitor to fetal rabbits results in feminization of the phallus with abnormal urethral plate fusion and deficient foreskin in males (Kurzrock et al, 2000). Prenatal exposure to antiandrogens (cyproterone or flutamide) causes feminization of the phallus, a proximal urethral opening, decreased anogential distance (Goldman and Baker, 1971), and cryptorchidism in male rats (McIntyre et al, 2001) and feminization of the genital tubercle in male mice (Suzuki et al, 2002). Pesticides with antiandrogenic properties such as linuron and vinclozolin cause feminization of the external genitalia and an abnormal proximal urethral opening with fetal exposure in male rats (Gray et al, 1999a) and male mice (Buckley et al, 2006). Phthalates act as antiandrogens by affecting the hypothalamic pituitary/gonadal axis and reduce testosterone production. Fetal male rats exposed to phthalates are feminized and have “hypospadias” (Gray et al, 1999b), abnormal epididymal and gubernacular development (Rider et al, 2009) and down-regulation of expression of beta-catenin, which has been demonstrated to be crucial to genital tubercle development through its involvement in the Wnt/beta-catenin pathway (Zhang et al, 2011). Phthalate exposure of fetal mice also elicits an abnormal proximal urethral opening in males and upregulation of TGF-beta1 (Liu et al, 2008).
While blocking the androgen pathway results in feminization of external genitalia of male animals, administration of exogenous androgens results in masculinization of female animals. Administration of androgens to fetal sheep results in masculinization of the external genitalia in females, which involves lengthening of the urethra, formation and elongation of a phallus, and development of a scrotal sac (Lamm, 2011). Similarly, prenatal androgen administration masculinizes external genitalia of female mice and rats and causes prostate development in female rats (Yucel et al, 2003a, Hotchkiss et al, 2007a, Hotchkiss et al, 2007b).
Both androgenic and estrogenic pathways when perturbed alter external genitalia development. Prenatal administration of estrogens (Kim et al, 2004, Yucel et al, 2003b) and estrogenic compounds, including benzophenone-2, an additive used to filter ultraviolet light, elicited feminization of the external genitalia and an abnormal proximal urethral opening in male mice (Hsieh et al, 2007). Similar feminization in male rats was noted following prenatal exposure to estrogenic styrene trimers found in polystyrene food containers (Ohyama et al, 2007). Curiously, female estrogen receptor alpha knockout mice develop a profoundly masculinized clitoris, perhaps as a result of perturbation of the balance between androgen and estrogen signaling (Yang et al, 2010).
Human Hypospadias
Hypospadias is the most common congenital malformation in boys occurring in 1 in 200 to 1 in 300 live births and may be utilized as a clinical example of the hormonal and genetic factors that interact during normal and abnormal external genitalia development. Hypospadias in humans is defined by three penile abnormalities: 1) abnormal urethral orifice location, 2) penile curvature (chordee), and 3) abnormal foreskin. In hypospadias the urethral orifice is located more proximally than its normal location at the tip of the penis. Chordee is an abnormal ventral penile curvature. Males with hypospadias also have a dorsal hooded foreskin with absence of ventral foreskin. Endocrine abnormalities have been linked to some cases of hypospadias in humans, but the etiology of hypospadias is multifactorial (Baskin, 2000, Silver, 2004). Hypospadias in humans has also been linked to exposure to endocrine disrupting compounds (Wang and Baskin, 2008, Nassar et al, 2010). Exposure of pregnant women to estrogenic and anti-androgenic endocrine disrupting compounds is associated with hypospadias and reduced anogenital distance in their male offspring. Anogenital distance is a recognized metric of androgen action. Several studies have demonstrated that exposure to phthalates results in decreased anogenital distance in human males, presumably due to lowered testosterone (Suzuki et al, 2011, Jurewicz and Hanke, 2011, Swan et al, 2005). Similarly, an anti-androgenic metabolite of the pesticide DDT reduces anogenital distance in human males (Torres-Sanchez et al, 2008). Exposure to bisphenol-A, a weakly estrogenic compound in plastic, has also been shown to reduce anogenital distance in human male offspring in a dose dependent fashion (Miao et al, 2011). Decreased anogenital distance in human males has been associated with decreased fertility in adulthood (Eisenberg et al, 2011). Exposure of pregnant women to pesticides has been linked to cryptorchidism, hypospadias, and micropenis with a non-statistically significant association with family history and maternal medications (Gaspari et al, 2011). Similarly, maternal exposure to estrogenic and antiandrogenic endocrine disrupting compounds has been implicated in increased risk of cryptorchidism and hypospadias in human male offspring without statistical significance (Morales-Suarez-Varela et al, 2011).
Other causes of hypospadias in humans include genetic defects, which have been demonstrated in a small percentage of cases. The estrogen receptor 2 (ERβ) gene has been linked to hypospadias (Beleza-Meireles, 2007a). Activating transcription factor 3 (ATF3) has been demonstrated to be upregulated in hypospadias tissue in humans and is known to be estrogen-responsive (Liu et al, 2006). ATF3 genomic variants have been found in patients with hypospadias (Beleza-Meireles et al, 2008, Kalfa et al, 2008), and ATF3 overexpression in the urethral plate is found in hypospadias (Kalfa et al, 2008). Mutations in SRD5A2, a gene involved in conversion of testosterone to dihydrotestosterone have been linked to an increased risk in hypospadias (Samtani et al, 2011, Wang et al, 2004). Mutations in FGF8 and FGFR2 were found in patients with hypospadias but not in age matched controls (Beleza-Meireles et al, 2007b).
Animal Models of Hypospadias
Human hypospadias consists of an abnormal opening of the penile urethra, defective prepuce, and chordee, an abnormal curvature of the penis. In mice the term “hypospadias” has been used to describe everything from urogenital sinus/persistent cloaca and imperforate anus abnormalities to nebulous preputial defects with rare references to actual urethral defects. While some reports are accurate, there is considerable confusion in the literature, rendering the term, murine (or ratine) “hypospadias”, difficult to interpret and non-comparable from study to study. Another problem is that many reports of rat or mouse hypospadias are reported as text only without illustrations or detailed descriptions as to what the authors recognize as hypospadias. In all cases, “urethral hypospadias” and “preputial hypospadias” should be specified. The dire state of the mouse (and rat) hypospadias literature is in part attributable to the paucity of knowledge of normal adult anatomy of mouse and rat external genitalia. Finally, an ideal animal model of hypospadias having translational relevance to human hypospadias should as closely as possible mimic the human malformation.
Hypospadias is fundamentally a disturbance in pattern, that is, morphological elements are either absent or mal-positioned in an absolute or relative sense. In the case of the urethra, the external orifice is in an abnormal proximal position. Consequently, the urethral orifice is absolutely mal-positioned as well as being mal-positioned relative to other elements within the penis. In humans the ventral portion of the prepuce is missing. Thus, spatial relationships of one component to others are disturbed, a concept that is particularly apparent in the mouse penis, which contains many more internal elements (bone, cartilage and several erectile bodies) and thus is vastly more complex than the human penis (Rodriguez et al, 2001, Weiss et al, 2012). The main problem hampering animal studies on hypospadias, especially in mouse models, is confusion regarding the term hypospadias. This problem has also been voiced for human hypospadias, as “determining whether there is a real increase in hypospadias is confounded by diagnostic criteria” (Fisher 2004). The field is in need of a standardized, accurate and precise anatomical description of mouse hypospadias that closely, unequivocally and accurately mimics human hypospadias so that translational inferences are possible. First, there must be agreement as to what constitutes mouse hypospadias and when it can and cannot be accurately and unequivocally identified. Of the three elements that characterize human hypospadias, chordee has not been reported in mice nor have we seen chordee in our experimental mouse studies. Thus, mouse hypospadias is an abnormality in the penile urethra and prepuce, separately or in combination. Such malformations can only be defined when an unambiguous penile urethra and prepuce actually exist (and not before). The penile urethra is by definition an epithelial lined tube transmitting urine that is located within the penis and therefore surrounded by and associated with elements of penile mesenchyme in the embryo or elements of penile stroma in the adult. The mature penis is an organ, which is radically different from and bears little resemblance to the embryonic or early postnatal genital tubercle. In cross sections it is evident that the adult mouse penile urethra is an independent tubular structure, not attached to other epithelia except at is distal termination, and surrounded by differentiated adult penile stroma containing bone, cartilage and erectile bodies (Figure 3D). Therefore, the adult penile urethra is a “differentiated penile urethra”. Such definitive adult penile urethral morphology is seen throughout the adult glans penis from proximal to distal and is also seen in proximal areas within 10- and 21-day penises (Figure 3B–C), but is completely absent in the undifferentiated genital tubercle prenatally and at birth (Figure 3A). This simple observation carries with it the profound implication that murine malformations of external genitalia observed at birth or earlier fail to meet a strict definition of hypospadias with direct translational relevance to human malformations because of an absence of a mature or terminally differentiated penile urethra or prepuce. The embryonic genital tubercle does contain an epithelial tube which transmits urine, which we designate as “undifferentiated penile urethra” since it is not surrounded by differentiated elements of penile stroma. The embryonic “undifferentiated penile urethra” bears only partial resemblance to the adult “differentiated penile urethra”. These are important distinctions. Malformations in external genitalia reported in newborn or embryonic mice are not strictly comparable to adult urethral or preputial defects observed in adulthood when the penis is fully formed/differentiated. An important point to recognize is that murine or ratine urethral or preputial defects comparable to human hypospadias can be accurately and definitively diagnosed in adulthood, which is the preferred stage to diagnose hypospadias. Malformations observed during embryonic periods are perhaps best interpreted as possible precursors of “definitive” hypospadias provided that it is possible to verify that embryonic malformations actually progress to adult urethral or preputial defects. Unfortunately, this is not possible in mutants that cannot survive to adulthood.
Figure 3. Comparison of Day 1, 10, 21, and Adult Mouse Penises.
Penile mes = penile mesenchyme, UGS = urogenital sinus, U = urethra, Ur = urethra, B = bone, Cart = cartilage, CCG or double- headed arrows = corpus cavernosum glandis, MUMPCC = MUMP corpus cavernosum, CCU = corpus cavernosum urethrae, PPL = preputial epithelial lamina
At birth, the 1-day mouse genital tubercle (3A) contains undifferentiated mesenchyme, which will give rise to preputial stroma and the differentiated elements of adult penile stroma. The epithelial tube, “undifferentiated penile urethra”, (labeled urogenital sinus) is not a “differentiated penile urethra” because it is not located in mature penile stroma and is attached to other epithelial structures. Instead, the epithelial urine-transmitting tube observed prenatally and in the neonatal period should more appropriately be designated as “undifferentiated penile urethra”. At postnatal day 10 (3B) and day 21 (3C) a differentiated penile urethra can be identified in certain restricted proximal regions within the developing penis. In such areas the penile urethra is located within differentiated penile mesenchyme containing bone, cartilage, corpus cavernosum glandis, MUMP corpus cavernosum, and corpus cavernosum urethrae. Compare the 10- and 21-day images (3B & 3C) with the adult penile urethra (3D) versus the day 1 genital tubercle (3A), whose urogenital sinus falls far short of the definition of “differentiated penile urethra”. Likewise, in the 1-, 10- and 21-day specimens the preputial epithelial lamina has not yet canalized and thus a defined prepuce is still not present even out to 21 days postnatal. Defects in the penile urethra (if present) could be recognized and interpreted as hypospadias at 10 and 21 days postnatal only in areas where a “differentiated penile urethra” exists, but not at day 1 (3A), in which an “undifferentiated penile urethra” is present. The 21-day specimen is identical to adult penis except that the preputial lamina has not yet canalized to create the preputial space and to define the prepuce.
It is important to recognize that two epithelial tubes develop within the developing penis: 1) the penile urethra and 2) the epithelial-lined prepuce, housing the penis. Both emerge from a precursor epithelial tube. The embryonic/neonatal urethral precursor tube extends to near the distal tip of the genital tubercle and transmits urine, but is not a differentiated “penile urethra” because it is not surrounded by or associated with differentiated elements of adult penile stroma. Accordingly, at 10 and 21 days postnatal a well-defined penile urethra is identifiable only in proximal regions of the developing penis, while in the distal part of the external genitalia the precursor epithelial tube remains undifferentiated. Comparison of the newborn genital tubercle to 10-, 21- and 60-day mouse penises reveals the vast differences between definitive penile urethra seen in 10-, 21- and 60-day penises versus the ”undifferentiated penile urethra” of the newborn mouse (Figure 3). Defects, if present, within the fully developed adult mouse penile urethra can be definitively judged to be equivalent to human hypospadias. However, defects seen embryonically or at birth are true malformations, but not strictly comparable to definitive hypospadias relevant to human hypospadias (Figure 3).
Our aim is to precisely define hypospadias so that the concept is unequivocally transferrable across studies and across species lines. In this regard reports of hypospadias should ideally be accompanied with photographs that allow discrimination between “urethral hypospadias” versus “preputial hypospadias’. Reference to hypospadias only in text should be avoided. With this as a background, we now turn our attention to genital tubercle defects in mutant mouse models. Since all of the models discussed below are derived from mice examined prenatally, the term hypospadias used liberally in the original publications of embryonic mice needs to be put in a more appropriate developmental context as possible precursors of hypospadias. Actual defects need to be described in detail to get a sense of the impact of gene deletion globally on development of the external genitalia.
Fibroblast growth factor receptor 2 (Fgfr2)-null mice examined at 13 through 19 days of gestation have an open urethral groove and absence of ventral prepuce, indicating that Fgfr2 is essential in development of the external genitalia and specifically in development of the urethra (Petiot et al, 2005, Satoh et al, 2004). Fgf-10 deficient male mice examined at embryonic days 13 through 19 and at 7 days postnatal have an open urethral groove due to failure of fusion of the urethral folds (Yucel et al, 2004b). Bone morphogenetic protein 7 (Bmp7)-null mice have an abnormal urethral plate (Suzuki et al, 2008), proximal urethral opening, rectourethral fistula and arrest of normal cloacal septation when examined at embryonic day 17.5 (Wu et al, 2009). In beta-catenin knockout mice the “urethra/urogenital sinus” opens to the exterior in a more proximal region when examined at embryonic day 14.5, reinforcing the role of Wnt/beta-catenin in genital tubercle and urethral development (Lin et al, 2008). In Fkbp52-deficient mice examined at E18.5 and adulthood the urethra also opens in a more proximal region, and prostate development is abnormal (Yong et al, 2007). Wnt5a deficient mice have agenesis of the genital tubercle when examined at embryonic day 18.5 (Suzuki et al, 2003). Expression of Prolactin-induced protein (Pip) was found to be decreased in a flutamide-induced “hypospadias” model in rats, and Pip expression was found to be localized to the prepuce in rats and in humans with hypospadias and phimosis, an inability to retract the foreskin over the glans penis (Kurokawa et al, 2011). Targeted ablation of FK506-binding protein-52 (Fkbp52), a co-chaperone of the androgen receptor, results in ectopic urethral seam fusion in male mice at embryonic day 18.5 (Chen et al, 2010). Inactivation of the Dlx5, Dlx6, p63, and Bmp7 genes results in abnormal urethral formation in embryonic mice (Suzuki et al, 2008) with p63 null mice demonstrating hypoplastic genitalia and persistent cloaca in females (Ince et al, 2002). Foxf2 knockout mice have cleft palates and hypoplastic genital tubercles when examined at birth (Jochumsen et al, 2008). Unfortunately, almost all mutant mice described above are not viable postnatally and, thus, it is impossible to determine the final adult consequences of these fetal malformations. The exception is the Fkbp52-deficient mice, which can be examined in adulthood, which is optimal for diagnosis of hypospadias. Perhaps use of conditional gene knockout will define mutations that culminate in definitive hypospadias.
Additional genes for study have been identified based on expression during external genitalia development. Genes expressed during genital tubercle development in mice include Hoxa13 (Lehoczky and Innis, 2008) with downstream expression of Fgf8 and Bmp7 (Morgan et al, 2003). The mouse genital tubercle also expresses TGFbeta1, TGFbeta receptor III, and Frizzled1 during development (Li et al, 2006). Cyp1b1, Fkbp51, and MafB were expressed during androgen-dependent masculinization of mouse genital tubercles (Nishida et al, 2008), however, Fkbp51-deficient mice have normal phenotypes (Yong et al, 2007).
Future studies will continue to identify genes present during normal genital development or presence or abnormal expression of genes in hypospadias and disorders of sex development. Identification of these genes and further elucidation of the pathways involved in sex development will help augment our understanding and may aid in development of directed therapeutics for hypospadias.
Acknowledgments
This work was supported by NSF Grant IOS-0920793 and NIH grant RO1 DK0581050.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Agrawal R, Wessely O, Anand A, Singh L, Aggarwal RK. Male-specific expression of Sox9 during gonad development of crocodile and mouse is mediated by alternative splicing of its proline-glutamine-alanine rich domain. FEBS J. 2009;276(15):4184–96. doi: 10.1111/j.1742-4658.2009.07127.x. [DOI] [PubMed] [Google Scholar]
- Assumpcao JG, Ferraz LF, Benedetti CE, Maciel-Guerra AT, Guerra G, Jr, Marques-de-Faria AP, Baptista MT, de Mello MP. A naturally occurring deletion in the SRY promoter region affecting the SP1 binding site is associated with sex reversal. J Endocrinol Invest. 2005;28(7):651–6. doi: 10.1007/BF03347266. [DOI] [PubMed] [Google Scholar]
- Baskin LS. Hypospadias and urethral development. J Urol. 2000;163(3):951–6. [PubMed] [Google Scholar]
- Beleza-Meireles A, Tohonen V, Soderhall C, Schwentner C, Radmayr C, Kockum I, Nordenskjold A. Activating transcription factor 3: a hormone responsive gene in the etiology of hypospadias. Eur J Endocrinol. 2008;158(5):729–39. doi: 10.1530/EJE-07-0793. [DOI] [PubMed] [Google Scholar]
- Beleza-Meireles A, Kockum I, Lundberg F, Soderhall C, Nordenskjold A. Risk factors for hypospadias in the estrogen receptor 2 gene. J Clin Endocrinol Metab. 2007;92(9):3712–8. doi: 10.1210/jc.2007-0543. [DOI] [PubMed] [Google Scholar]
- Beleza-Meireles A, Lundberg F, Lagerstedt K, Zhou X, Omrani D, Frisen L, Nordenskjold A. FGFR2, FGF8, FGF10 and BMP7 as candidate genes for hypospadias. Eur J Hum Genet. 2007;15(4):405–10. doi: 10.1038/sj.ejhg.5201777. [DOI] [PubMed] [Google Scholar]
- Blake KD, Prasad C. CHARGE syndrome. Orphanet J Rare Dis. 2006 Sep 7;1:34. doi: 10.1186/1750-1172-1-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckley J, Willingham E, Agras K, Baskin LS. Embryonic exposure to the fungicide vinclozolin causes virilization of females and alteration of progesterone receptor expression in vivo: an experimental study in mice. Environ Health. 2006 Feb 21;5:4. doi: 10.1186/1476-069X-5-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H, Yong W, Hinds TD, Jr, Yang Z, Zhou Y, Sanchez ER, Shou W. Fkbp52 regulates androgen receptor transactivation activity and male urethra morphogenesis. J Biol Chem. 2010;285(36):27776–84. doi: 10.1074/jbc.M110.156091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher JS. Environmental anti-androgens and male reproductive health: focus on phthalates and testicular dysgenesis syndrome. Reproduction. 2004;127(3):305–15. doi: 10.1530/rep.1.00025. [DOI] [PubMed] [Google Scholar]
- Galani A, Kitsiou-Tzeli S, Sofokleous C, Kanavakis E, Kalpini-Mavrou A. Androgen insensitivity syndrome: clinical features and molecular defects. Hormones. 2008;7(3):217–29. doi: 10.14310/horm.2002.1201. [DOI] [PubMed] [Google Scholar]
- Gaspari L, Paris F, Jandel C, Kalfa N, Orsini M, Daures JP, Sultan C. Prenatal environmental risk factors for genital malformations in a population of 1442 French male newborns: a nested case-control study. Hum Reprod. 2011;26(11):3155–62. doi: 10.1093/humrep/der283. [DOI] [PubMed] [Google Scholar]
- Goldman AS, Baker MK. Androgenicity in the rat fetus of metabolites of testosterone and antagonism by cyproterone acetate. Endocrinology. 1971;89(1):276–80. doi: 10.1210/endo-89-1-276. [DOI] [PubMed] [Google Scholar]
- Gray LE, Jr, Ostby J, Monosson E, Kelce WR. Environmental antiandrogens: low doses of the fungicide vinclozolin alter sexual differentiation of the male rat. Toxicol Ind Health. 1999;15(1–2):48–64. doi: 10.1177/074823379901500106. [DOI] [PubMed] [Google Scholar]
- Gray LE, Jr, Wolf C, Lambright C, Mann P, Price M, Cooper RL, Ostby J. Administration of potentially antiandrogenic pesticides (procymidone, linuron, iprodione, chlozolinate, p,p′-DDE, and ketoconazole) and toxic substances (dibutyl-and diethylhexyl phthalate, PCB 169, and ethane dimethane sulphonate) during sexual differentiation produces diverse profiles of reproductive malformations in the male rat. Toxicol Ind Health. 1999;15(1–2):94–118. doi: 10.1177/074823379901500109. [DOI] [PubMed] [Google Scholar]
- Haraguchi R, Mo R, Hui C, Motoyama J, Makino S, Shiroshi T, Gaffield W, Yamada G. Unique functions of Sonic hedgehog signaling during external genitalia development. Development. 2001;128(21):4241–50. doi: 10.1242/dev.128.21.4241. [DOI] [PubMed] [Google Scholar]
- Haraguchi R, Suzuki K, Murakami R, Sakai M, Kamikawa M, Kengaku M, Sekine K, Kawano H, Kato S, Ueno N, Yamada G. Molecular analysis of external genitalia formation: the role of fibroblast growth factor (Fgf) genes during genital tubercle formation. Development. 2000;127(11):2471–9. doi: 10.1242/dev.127.11.2471. [DOI] [PubMed] [Google Scholar]
- Hotchkiss AK, Lambright CS, Ostby JS, Parks-Saldutti L, Vandenbergh JG, Gray LE., Jr Prenatal testosterone exposure permanently masculinizes anogenital distance, nipple development, and reproductive tract morphology in female Sprague-Dawley rats. Toxicol Sci. 2007;96(2):335–45. doi: 10.1093/toxsci/kfm002. [DOI] [PubMed] [Google Scholar]
- Hotchkiss AK, Furr J, Makynen EA, Ankley GT, Gray LE., Jr In utero exposure to the environmental androgen trenbolone masculinizes female Sprague-Dawley rats. Toxicol Lett. 2007;174(1–3):31–41. doi: 10.1016/j.toxlet.2007.08.008. [DOI] [PubMed] [Google Scholar]
- Hsieh MH, Grantham EC, Liu B, Macapagal R, Willingham E, Baskin LS. In utero exposure to benxophenone-2 causes hypospadias through an estrogen receptor dependent mechanism. J Urol. 2007;178(4 Pt 2):1637–42. doi: 10.1016/j.juro.2007.03.190. [DOI] [PubMed] [Google Scholar]
- Ince TA, Cviko AP, Quade BJ, Yang A, McKeon FD, Mutter GL, Crum CP. P63 Coordinates anogenital modeling and epithelial cell differentiation in the developing female urogenital tract. Am J Pathol. 2002;161(4):1111–7. doi: 10.1016/S0002-9440(10)64387-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito M, Yu R, Jameson JL. DAX-1 inhibits SF-1-mediated transactivation via a carboxy-terminal domain that is deleted in adrenal hypoplasia congenita. Mol Cell Biol. 1997;17(3):1476–83. doi: 10.1128/mcb.17.3.1476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jochumsen U, Werner R, Miura N, Richyer-Unruh A, Hiort O, Holterhus PM. Mutation analysis of FOXF2 in patients with disorders of sex development (DSD) in combination with cleft palate. Sex Dev. 2008;2(6):302–8. doi: 10.1159/000195679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jurewicz J, Hanke W. Exposure to phthalates: reproductive outcome and children health. A review of epidemiological studies. Int J Occup Med Environ Health. 2011;24(2):115–41. doi: 10.2478/s13382-011-0022-2. [DOI] [PubMed] [Google Scholar]
- Kalfa N, Liu B, Klein O, Wang MH, Cao M, Baskin LS. Genomic variants of ATF3 in patients with hypospadias. J Urol. 2008;180(5):2183–8. doi: 10.1016/j.juro.2008.07.066. [DOI] [PubMed] [Google Scholar]
- Kim KS, Torres CR, Jr, Yucel S, Raimondo K, Cunha GR, Baskin LS. Induction of hypospadias in a murine model by maternal exposure to synthetic estrogens. Environ Res. 2004;94(3):267–75. doi: 10.1016/S0013-9351(03)00085-9. [DOI] [PubMed] [Google Scholar]
- Kurokawa S, Kojima Y, Mizuno K, Kamisawa H, Tozawa K, Kohri K, Hayashi Y. Association of prolactin-induced protein with preputial development of hypospadias. BJU Int Epub ahead of print. 2011 Aug 25; doi: 10.1111/j.1464-410X.2011.10467.x. [DOI] [PubMed] [Google Scholar]
- Kurzrock EA, Jegatheesan P, Cunha GR, Baskin LS. Urethral development in the fetal rabbit and insuction of hypospadias: a model for human development. J Urol. 2000;164(5):1786–92. [PubMed] [Google Scholar]
- Lamm CG, Hastie PM, Evans NP, Robinson JE. Masculinization of the Distal Tubular and External Genitalia in Female Sheep with Prenatal Androgen Exposure. Vet Pathol. 2011 Sep 20; doi: 10.1177/0300985811419533. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- Le Caignec C, Delnatte C, Vermeesch JR, Boceno M, Joubert M, Lavenant F, David A, Rival JM. Complete sex reversal in a WAGR syndrome patient. Am J Med Genet A. 2007 Nov 15;143A(22):2692–5. doi: 10.1002/ajmg.a.31997. [DOI] [PubMed] [Google Scholar]
- Lehoczky JA, Innis JW. BAC transgenic analysis reveals enhancers sufficient for Hoxa13 and neighborhood gene expression in mouse embryonic distal limbs and genital bud. Evol Dev. 2008;10(4):421–32. doi: 10.1111/j.1525-142X.2008.00253.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Willingham E, Baskin LS. Gene expression profiles in mouse urethral development. BJU Int. 2006;98(4):880–5. doi: 10.1111/j.1464-410X.2006.06435.x. [DOI] [PubMed] [Google Scholar]
- Lin C, Yin Y, Veith GM, Fisher AV, Long F, Ma L. Temporal and spatial dissection of Shh signaling in genital tubercle development. Development. 2009;136(23):3959–67. doi: 10.1242/dev.039768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin C, Yin Y, Long F, Ma L. Tissue-specific requirements of beta-catenin in external genitalia development. Development. 2008;135(16):2815–25. doi: 10.1242/dev.020586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin L, Ercan O, Raza J, Burren CP, Creighton SM, Auchus RJ, Dattani MT, Achermann JC. Variable phenotypes associated with aromatase (CYP19) insufficiency in humans. J Clin Endocrinol Metab. 2007;92(3):982–90. doi: 10.1210/jc.2006-1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu B, Agras K, Willingham E, Vilela ML, Baskin LS. Activating transcription factor 3 is estrogen-responsive in utero and upregulated during sexual differentiation. Horm Res. 2006;65(5):217–22. doi: 10.1159/000092402. [DOI] [PubMed] [Google Scholar]
- Liu X, He DW, Zhang DY, Lin T, Wei GH. Di(2-ethylhexyl) phthalate (DEHP) increases transforming growth factor-beta 1 expression in fetal mouse genital tubercles. J Toxicol Environ Health A. 2008;71(19):1289–94. doi: 10.1080/15287390802114915. [DOI] [PubMed] [Google Scholar]
- McIntyre BS, Barlow NJ, Foster PM. Androgen-mediated development in male rat offspring exposed to flutamide in utero: permanence and correlation of early postnatal changes in anogenital distance and nipple retention with malformations in androgen-dependent tissues. Toxicol Sci. 2001 Aug;62(2):236–49. doi: 10.1093/toxsci/62.2.236. [DOI] [PubMed] [Google Scholar]
- Miao M, Yuan W, He Y, Zhou Z, Wang J, Gao E, Li G, Li DK. In utero exposure to bisphenol-A and anogenital distance in male offspring. Birth Defects Res A Clin Mol Teratol. 2011;91(10):867–72. doi: 10.1002/bdra.22845. [DOI] [PubMed] [Google Scholar]
- Miyagawa S, Matsumaru D, Murashima A, Omori A, Satoh Y, Haraguchi R, Motoyama J, Iguchi T, Nakagata N, Hui CC, Yamada G. The role of sonic hedgehog-Gli2 pathway in the masculinization of external genitalia. Endocrinology. 2011;152(7):2894–903. doi: 10.1210/en.2011-0263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyagawa S, Moon A, Haraguchi R, Inoue C, Harada M, Nakahara C, Suzuki K, Matsumaru D, Kaneko T, Matsuo I, Yang L, Taketo MM, Iguchi T, Evans SM, Yamada G. Dose-dependent hedgehog signals integrated with Wnt/beta-catenin signaling regulate external genitalia formation as an appendicular program. Development. 2009;136(23):3969–78. doi: 10.1242/dev.039438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyagawa S, Satoh Y, Haraguchi R, Suzuki K, Iguchi T, Taketo MM, Nakagata N, Matsumoto T, Takeyama K, Kato S, Yamada G. Genetic interactions of the androgen and Wnt/beta-catenin pathways for the masculinization of external genitalia. Mol Endocrinol. 2009;23(6):871–80. doi: 10.1210/me.2008-0478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morales-Suarez-Varela MM, Toft GV, Jensen MS, Ramlau-Hansen C, Kaerlev L, Thulstrup AM, Llopis-Gonzalez A, Olsen J, Bonde JP. Parental occupational exposure to endocrine disrupting chemicals and male genital malformations: a study in the Danish National Birth Cohort study. Environ Health. 2011;10(1):3. doi: 10.1186/1476-069X-10-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan EA, Nguyen SB, Scott V, Stadler HS. Loss of Bmp7 and Fgf8 signaling in Hoxa13-mutant mice causes hypospadia. Development. 2003;130(14):3095–109. doi: 10.1242/dev.00530. [DOI] [PubMed] [Google Scholar]
- Nassar N, Abeywardana P, Barker A, Bower C. Parental occupational exposure to potential endocrine disrupting chemicals and risk of hypospadias in infants. Occup Environ Med. 2010;67(9):585–9. doi: 10.1136/oem.2009.048272. [DOI] [PubMed] [Google Scholar]
- Nishida H, Miyagawa S, Matsumaru D, Wada Y, Satoh Y, Ogino Y, Fukuda S, Iguchi T, Yamada G. Gene expression analyses on embryonic external genitalia: identification of regulatory genes possibly involved in masculinization processes. Congenit Anom. 2008;48(2):63–7. doi: 10.1111/j.1741-4520.2008.00180.x. [DOI] [PubMed] [Google Scholar]
- Ogino Y, Katoh H, Yamada G. Androgen dependent development of a modified anal fin, gonopodium, as a model to understand the mechanism of secondary sexual character expression in vertebrates. FEBS Lett. 2004;575(1–3):119–26. doi: 10.1016/j.febslet.2004.08.046. [DOI] [PubMed] [Google Scholar]
- Ohyama K, Satoh K, Sakamoto Y, Ogata A, Nagai F. Effects of prenatal exposure to styrene trimers on genital organs and hormones in male rats. Exp Biol Med. 2007;232(2):301–8. [PubMed] [Google Scholar]
- Perriton CL, Powles N, Chiang C, Maconochie MK, Cohn MJ. Sonic hedgehog signaling from the urethral epithelium controls external genital development. Dev Biol. 2002;247(1):26–46. doi: 10.1006/dbio.2002.0668. [DOI] [PubMed] [Google Scholar]
- Petiot A, Perriton CL, Dickson C, Cohn MJ. Development of the mammalian urethra is controlled by FGFr2-IIIb. Development. 2005;132(10):2441–50. doi: 10.1242/dev.01778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinto G, Abadie V, Mesnage R, Blustajn J, Cabrol S, Amiel J, Hertz-Pannier L, Bertrand AM, Lyonnet S, Rappaport R, Netchine I. CHARGE syndrome includes hypogonadotropic hypogonadism and abnormal olfactory bulb development. J Clin Endocrinol Metab. 2005 Oct;90(10):5621–6. doi: 10.1210/jc.2004-2474. [DOI] [PubMed] [Google Scholar]
- Rodriguez E, Jr, Weiss DA, Yang JH, Menshenina J, Ferretti M, Cunha TJ, Barcellos D, Chan LY, Risbridger G, Cunha GR, Baskin LS. New insights on the morphology of adult mouse penis. Biol Reprod. 2011;85(6):1216–21. doi: 10.1095/biolreprod.111.091504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh Y, Haraguchi R, Wright TJ, Mansour SL, Partanen J, Hajihosseini MK, Eswarakumar VP, Lonai P, Yamada G. Regulation of external genitalia development by concerted actions of FGF ligands and FGF receptors. Anat Embryol. 2004;208(6):479–86. doi: 10.1007/s00429-004-0419-9. [DOI] [PubMed] [Google Scholar]
- Samtani R, Bajpai M, Vashisht K, Ghosh PK, Saraswathy KN. Hypospadias risk and polymorphism in SRD5A2 and CYP17 genes: case-control study among Indian children. J Urol. 2011;185(6):2334–9. doi: 10.1016/j.juro.2011.02.043. [DOI] [PubMed] [Google Scholar]
- Seifert AW, Bouldin CM, Choi KS, Harfe BD, Cohn MJ. Multiphasic and tissue-specific roles of sonic hedgehog in cloacal septation and external genitalia development. Development. 2009;136(23):3949–57. doi: 10.1242/dev.042291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seifert AW, Yamaguchi T, Cohn MJ. Functional and phylogenetic analysis shows that Fgf8 is a marker of genital induction in mammals but is not required for external genital development. Development. 2009;136(15):2643–51. doi: 10.1242/dev.036830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sekido R, Bar I, Narvaez V, Penny G, Lovell-Badge R. SOX9 is up-regulated by the transient expression of SRY specifically in Sertoli cell precursors. Dev Biol. 2004;15;274(2):271–9. doi: 10.1016/j.ydbio.2004.07.011. [DOI] [PubMed] [Google Scholar]
- Shehata BM, Elmore JM, Bootwala Y, Steelman CK, Bare JB, Shoffeitt CJ, Wang R, Zhau HE, He D, Zhu G, Chung LW. Immunohistochemical charicterization of sonic hedgehog and its downstream signaling molecules during human penile development. Fetal Pediatr Pathol. 2011;30(4):244–51. doi: 10.3109/15513815.2011.555809. [DOI] [PubMed] [Google Scholar]
- Silver RI. Endocrine abnormalities in boys with hypospadias. Adv Exp Med Biol. 2004;545:45–72. doi: 10.1007/978-1-4419-8995-6_4. [DOI] [PubMed] [Google Scholar]
- Sinclair AH, Berta P, Palmer MS, Hawkins JR, Griffiths BL, Smith MJ, Foster JW, Frischauf AM, Lovell-Badge R, Goodfellow PN. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature. 1990;346(6281):240–4. doi: 10.1038/346240a0. [DOI] [PubMed] [Google Scholar]
- Suzuki Y, Yoshinaga J, Mizumoto Y, Serizawa S, Shiraishi H. Foetal exposure to phthalate esters and anogenital distance in male newborns. Int J Androl. 2011 Jun 22; doi: 10.1111/j.1365-2605.2011.01190.x. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- Suzuki K, Haraguchi R, Ogata T, Barbieri O, Alegria O, Vieux-Rochas M, Nakagata N, Ito M, Mills AA, Kurita T, Levi G, Yamada G. Abnormal urethra formation in mouse models of split-hand/split-foot malformation type 1 and type 4. Eur J Hum Genet. 2008;16(1):36–44. doi: 10.1038/sj.ejhg.5201925. [DOI] [PubMed] [Google Scholar]
- Suzuki K, Bachiller D, Chen YP, Kamikawa M, Ogi H, Haraguchi R, Ogino Y, Minami Y, Mishina Y, Ahn K, Crenshaw EB, 3rd, Yamada G. Regulation of outgrowth and apoptosis for the terminal appendage: external genitalia development by concerted actions of BMP signaling. Development. 2003;130(25):6209–20. doi: 10.1242/dev.00846. [DOI] [PubMed] [Google Scholar]
- Suzuki K, Ogino Y, Murakami R, Satoh Y, Bachiller D, Yamada G. Embryonic development of mouse external genitalia: insights into a unique mode of organogenesis. Evol Dev. 2002;4(2):133–41. doi: 10.1046/j.1525-142x.2002.01061.x. [DOI] [PubMed] [Google Scholar]
- Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, Mao CS, Redmon JB, Ternand CL, Sullivan S, Teague JL Study for Future Families Research Team. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect. 2005;113(8):1056–61. doi: 10.1289/ehp.8100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Sanchez L, Zepeda M, Cebrian ME, Belkind-Gerson J, Garcia-Hernandez RM, Belkind-Valdovinos U, Lopez-Carrillo L. Dichlorodiphenyldichloroethylene exposure during the first trimester of pregnancy alters the anal position in male infants. Ann N Y Acad Sci. 2008 Oct;1140:155–62. doi: 10.1196/annals.1454.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang MH, Baskin LS. Endocrine disruptors, genital development, and hypospadias. J Androl. 2008;29(5):499–505. doi: 10.2164/jandrol.108.004945. [DOI] [PubMed] [Google Scholar]
- Wang Y, Li Q, Xu J, Liu Q, Wang W, Lin Y, Ma F, Chen T, Li S, Shen Y. Mutation analysis of five candidate genes in Chinese patients with hypospadias. Eur J Hum Genet. 2004;12(9):706–12. doi: 10.1038/sj.ejhg.5201232. [DOI] [PubMed] [Google Scholar]
- Weiss DA, Rodriguez E, Jr, Cunha T, Menshenina J, Barcellos D, Chan LY, Risbridger G, Baskin L, Cunha G. Morphology of the external genitalia of the adult male and female mice as an endpoint of sex differentiation. Mol Cell Endocrinol. 2012;354(1–2):94–102. doi: 10.1016/j.mce.2011.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson JD, Griffin JE, Leshin M, George FW. Role of gonadal hormones in development of the sexual phenotypes. Hum Genet. 1981;58(1):78–84. doi: 10.1007/BF00284153. [DOI] [PubMed] [Google Scholar]
- Wu X, Ferrara C, Shapiro E, Grishina I. Bmp7 expression and null phenotype in the urogenital system suggest a role in re-organization of the urethral epithelium. Gene Expr Patterns. 2009;9(4):224–30. doi: 10.1016/j.gep.2008.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada G, Satoh Y, Baskin LS, Cunha GR. Cellular and molecular mechanisms of development of the external genitalia. Differentiation. 2003;71(8):445–60. doi: 10.1046/j.1432-0436.2003.7108001.x. [DOI] [PubMed] [Google Scholar]
- Yang JH, Menshenina J, Cunha GR, Place N, Baskin LS. Morphology of mouse external genitalia: implications for a role of estrogen in sexual dimorphism of the mouse genital tubercle. J Urol. 2010;184(4 Suppl):1604–9. doi: 10.1016/j.juro.2010.03.079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yong W, Yang Z, Perivasamy S, Chen H, Yucel S, Li W, Lin LY, Wolf IM, Cohn MJ, Baskin LS, Sanchez ER, Shou W. Essential role for Co-chaperone Fkbp52 but not Fkbp51 in androgen receptor-mediated signaling and physiology. J Biol Chem. 2007;16;282(7):5026–36. doi: 10.1074/jbc.M609360200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yucel S, Liu W, Cordero D, Donjacour A, Cunha G, Baskin LS. Anatomical studies of the fibroblast growth gactor-10 mutant, Sonic Hedge Hog mutant and androgen receptor mutant mouse genital tubercle. Adv Exp Med Biol. 2004;545:123–48. doi: 10.1007/978-1-4419-8995-6_8. [DOI] [PubMed] [Google Scholar]
- Yucel S, Cavalcanti AG, Wang Z, Baskin LS. The impact of prenatal androgens on vaginal and urogenital sinus development in the female mouse. 2003;170(4 Pt 1):1432–6. doi: 10.1097/01.ju.0000084595.73653.da. [DOI] [PubMed] [Google Scholar]
- Yucel S, Cavalcanti AG, Desouza A, Wang Z, Baskin LS. The effect of oestrogen and testosterone on the urethral seam of the developing male mouse genital tubercle. BJU Int. 2003;92(9):1016–21. doi: 10.1111/j.1464-410x.2003.04511.x. [DOI] [PubMed] [Google Scholar]
- Zhang LF, Qin C, Wei YF, Wang Y, Chang JK, Mi YY, Ma L, Jiang JT, Feng NH, Wang ZJ, Zhang W. Differential expression of the Wnt/β-catenin pathway in the genital tubercle (GT) of fetal male rat following maternal exposure to di-n-butyl phthalate (DBP) Syst Biol Reprod Med. 2011;57(5):244–50. doi: 10.3109/19396368.2011.577509. [DOI] [PubMed] [Google Scholar]