Abstract
Objective
Cardiovascular disease is the number one killer of women. Identifying women at risk of cardiovascular disease has tremendous public health importance. Early menopause is associated with increased cardiovascular disease events in some predominantly white populations, but not consistently. Our objective was to determine if a self-reported early menopause (menopause at an age <46) identifies women as at risk for future coronary heart disease or stroke.
Methods
The study population came from the Multi-Ethnic Study of Atherosclerosis, a longitudinal, ethnically diverse cohort study of US men and women aged 45 to 84 years enrolled in 2000–2002 and followed up until 2008. The association between a personal history of early menopause (either natural menopause or surgical removal of ovaries at an age <46) and future coronary heart disease and stroke was assessed in 2509 women (ages 45–84, 987 White, 331 Chinese, 641 Black, 550 Hispanic) from the Multi-Ethnic Study Atherosclerosis, who were free of cardiovascular disease at baseline.
Results
693/2509 (28%) of women reported either surgical or natural early menopause. In survival curves, women with early menopause had worse coronary heart disease and stroke-free survival (log rank p=<0.008 and 0.0158). In models adjusted for age, race/ethnicity, Multi-Ethnic Study Atherosclerosis site and traditional cardiovascular disease risk factors, this risk for coronary heart disease and stroke remained (HR 2.08, 95% CI 1.17, 3.70 and 2.19, 95% CI 1.11, 4.32, respectively).
Conclusions
Early menopause is positively associated with coronary heart disease and stroke in a multiethnic cohort, independent of traditional cardiovascular disease risk factors.
Keywords: Early Menopause, Coronary Heart Disease, Stroke
INTRODUCTION
Cardiovascular disease (CVD) is the leading cause of death in US women.1 The early identification of women at high risk of CVD and implementation of appropriate lifestyle and medical therapy is of tremendous public health importance. Current strategies for identifying high risk women involve counting major coronary heart disease (CHD) factors and estimating 10-year risk with algorithms.2–4 The algorithm most commonly used to identify women at high risk of CHD, the Framingham risk score, relies heavily on age and biomarkers. Clinical history including smoking history is incorporated. However, reproductive health history is not, yet a number of cohort studies have identified an association between early menopause and cardiovascular risk.5–12
The second Adult Treatment Panel (ATP II) report included a history of premature menopause in global CVD risk assessment.13 It considered premature menopause without estrogen therapy a risk factor for CHD. This plus one other risk factor (family history of premature CHD, current cigarette smoking, hypertension, low high-density lipoprotein (HDL) HDL cholesterol, or diabetes) identified a woman as having high CHD risk. This was based on observational studies that indicated that increases in LDL with menopause were reversed by hormone therapy (HT).14 The panel posited that the presence of estrogen may protect women from CHD. However, subsequent panels (ATP III3, 15) dropped early menopause as a risk factor. This may have been because HT was not shown to confer protection from CHD in randomized trials.16,17
Whether HT benefits vascular health is intensely debated. However, observational studies consistently show that early age at menopause is associated with CHD7,10 and possibly stroke.9 These studies have included mostly white or European populations and may not be generalizable to US women not of European origin. Therefore, we investigated whether early menopause (menopause before age 46 yrs) was associated with CHD and stroke in a multi-ethnic population of US women. We further investigated whether this relationship was independent of traditional CVD risk factors.
METHODS
Design Overview, Setting, and Participants
Multi-Ethnic Study Atherosclerosis (MESA) is a multi-center, longitudinal cohort study of the prevalence and correlates of subclinical CVD and the factors that influence its progression.18 Between July 2000 and August 2002, 3213 men and 3601 women who identified themselves as white, black, Hispanic, or Chinese, reported that they were free of CVD, and were 45 to 84 years of age, were recruited from 6 US communities: Baltimore City and Baltimore County, Maryland; Chicago, Illinois; Forsyth County, North Carolina; Los Angeles County, California; Northern Manhattan and the Bronx, New York; and St. Paul Minnesota. Details on the sampling frames and the cohort examination procedures have been published previously.18 Informed consent was obtained from each participant and the study was approved by the Institutional Review Boards of each institution.
At the baseline exam (July 2000 to August 2002), medical history, anthropometric measurements, and laboratory data were obtained. Age, gender, ethnicity, and medical and medication histories were obtained by questionnaire. Family history of CVD was defined as having a self-reported history of a first-degree relative (parent, sibling, or child) with heart attack. Ever smoking was defined as having smoked ≥100 cigarettes in a lifetime. Diabetes mellitus was defined as a fasting glucose ≥126 mg/dL19 or use of a hypoglycemic medication.
Blood pressure, weight and height were measured using standardized protocols.18, 20 Hypertension was defined as a systolic blood pressure (BP) >140 mm Hg, diastolic BP >90 mm Hg (JNC VI21), or self report of hypertension plus the use of an antihypertensive medication. Body mass index (BMI) was calculated as the weight (kg) ÷ height squared (m2). Plasma total and HDL cholesterol and glucose were measured after a 12-hour fast.
As part of the in-person MESA baseline assessment questions relevant to menopausal status were asked of women participants. Natural menopause was determined with the questions: “Have you gone through menopause (change of life)?” or “Are you currently going through menopause?” Age at natural menopause was determined with the question “At what age did you go through menopause (year)?” Surgical menopause was determined with the question “Have you had surgery to remove your ovaries?” Age at surgical menopause was determined with the question “At what age?” All women were asked “Have you ever taken hormone replacement therapy?”
These questions were asked as part of an interviewer-administered form. For each question, possible responses are “Yes”, “No,” ‘Don’t Know,” and/or ‘Not Applicable” or “N/A” (not applicable). The interviewer was instructed to have the participant choose the appropriate responses for each question. The interviewers were instructed not to probe or to make interpretations about a participant’s specific symptoms. They were instructed to ask questions as written and record answers as given. Chart reviews were not performed to confirm menopausal status or surgical history.
For our study, early menopause was defined as a self-reported menopause at age <46. The use of the age cutpoint of 46 allowed for the categorization of any woman not reporting menopause at baseline as unexposed to early menopause. This was because all MESA women were at least age 45 at enrollment.
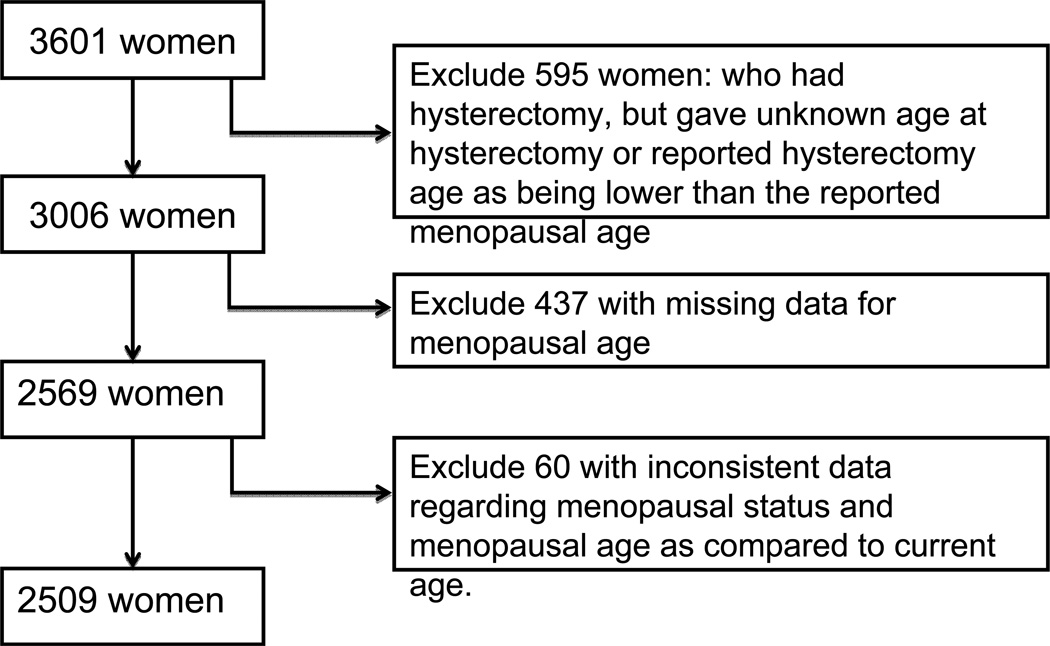
Among all MESA women (n=3601), 1261/3601 (35%) reported hysterectomy. This prevalence of hysterectomy appears consistent with a previously published report from the National Women’s Health Information Center that 1/3 of women age 60 in the US have undergone hysterectomy.22 In our study, participants with hysterectomy without oophorectomy, missing data, or inconsistent data regarding menopausal status were excluded from our sample (Figure 1). In our sample, 27% of women reported an early menopause (either natural or surgical). In comparison, in a cohort study of age at natural menopause and mortality by Snowden et al, age 46 represented the 25th percentile for age at natural menopause.12
Figure 1.
Flow of MESA Sample Selection of Women with and without Early Menopause.
CHD and Stroke Outcomes
The MESA cohort has been followed for incident cardiovascular events for a median of 57.3 months (range of 1.3 to 73.2 months). The primary means of identifying possible events in MESA is participant self-report via post-baseline contacts (follow-up calls) conducted by telephone. Field center staff may also learn of potential events in other ways: participants may notify the clinic when they experience an event; a MESA examination may identify a possible event; investigation of one event may identify another event; National Death Index search could identify a death; or field center staff may learn of a participant’s death through an obituary or other public notice.
At intervals of 9 to 12 months, a telephone interviewer contacted each participant regarding hospital admissions, cardiovascular outpatient diagnoses, and deaths. To verify self-reported diagnoses, copies of all death certificates and medical records for hospitalizations and outpatient cardiovascular diagnoses were requested. For out-of-hospital cardiovascular deaths, next-of-kin interviews were performed. Records on an estimated 98% of reported hospitalized cardiovascular events and some information on 95% of reported outpatient diagnostic encounters were obtained. Two physicians independently reviewed and classified CVD events and assigned incidence dates. If they disagreed, they adjudicated their differences via discussion.
Periodically, the Coordinating Center will search the National Death Index for participants with whom the study has lost touch. The Field Centers will then be notified of these deaths so that additional information can be obtained and so that the death can go to physician review. Criteria for events are available on the MESA website (http://www.mesa-nhlbi.org/Mesa-Internal/manuals.asp) and are described in published MESA manuscripts.23
Reviewers classified a myocardial infarction (MI) as definite or probable if either abnormal cardiac biomarkers (two times upper limits of normal) regardless of pain or ECG findings; evolving Q waves regardless of pain or biomarker findings; or a combination of chest pain, and ST-T evolution or new LBBB, and biomarker levels 1–2 times upper limits of normal was present. Reviewers classified a resuscitated cardiac arrest as present when a subject had successfully recovered from a full cardiac arrest through cardiopulmonary resuscitation (including cardioversion). The reviewers classified CHD death as present or absent based on hospital records and interviews with families. Definite fatal CHD required an MI within 28 days of death, chest pain within the 72 hours before death, or a history of CHD and the absence of a known non- atherosclerotic or non-cardiac cause of death. Neurologists reviewed and classified stroke as present if there was a focal neurologic deficit lasting 24 hours or until death, with a clinically relevant lesion on brain imaging and no nonvascular cause.
For this report, we defined incident CHD as definite or probable MI, resuscitated cardiac arrest, and definite CHD death. Incident stroke included fatal and nonfatal stroke. Follow-up was from the baseline examination until first CVD event, loss to follow-up, death, or October 14, 2008, whichever came first.
Statistical Analyses
The association between early menopause and incident CHD and stroke was examined in Kaplan Meier survival analyses and Cox proportional hazards models. Proportional hazards models were first adjusted only for age. The models were then adjusted for demographics (race/ethnicity and MESA site) and traditional CVD risk factors (hypertension, ever smoking, diabetes mellitus, total and HDL cholesterol). Additional models included BMI and family history of CHD. Secondary analyses included adjustment for HT use and type of menopause (natural vs. surgical). Interactions between early menopause and 1) HT use 2) type of menopause and 3) ever smoking were performed after adjustment for age, race/ethnicity, and MESA site. We also performed sensitivity analyses that included adjustment for education as a proxy for socioeconomic status.
To assess discrimination of a model including traditional CVD risk factors only (hypertension, ever smoking, diabetes mellitus, total and HDL cholesterol) verses a model that also includes early menopause, we performed multiple statistical tests. Hazard ratios were estimated for each regression model. The incremental statistical significance of early menopause when added to the traditional CVD risk factor model was evaluated with the Wald test of significance of the beta coefficient. Discrimination was assessed using the area under the receiver operator characteristic curve (ROC-AUC, or C-statistic). The C-statistic for each model was compared with the C-statistic for the baseline model using a binomial test (Mann-Whitney U test). All statistical analyses were performed with Stata version 8.0 (Stata Corp, Austin, Tex; http://www.stata.com) with significance set at P<l0.05 (2-tailed).
Results
Table 1 includes baseline characteristics of participants with early menopause (n=693) versus no early menopause (n=1816). Compared to participants without early menopause, a greater percentage of women with early menopause was black or Hispanic, age 75–84 at baseline, surgically menopausal, and had ever used HT.
Table 1.
Characteristics of Women with and without Early Menopause (n=2509)
| Early Menopause (n=693) Mean (SD) or No. (%) |
No Early Menopause (n=1816) Mean (SD) or No. (%) |
p-value | |
|---|---|---|---|
| Characteristics | |||
| Age, y 45–54 55–64 65–74 75–84 |
162 (23%) 189 (27%) 206 (30%) 136 (20%) |
368 (20%) 573 (32%) 615 (34%) 260 (14%) |
0.001 |
| Education < High school graduate ≥ High school graduate |
152 (22%) 537 (79%) |
268 (20%) 1443 (80%) |
0.34 |
| Race/ethnicity | |||
| • White | 241 (35%) | 746 (41%) | <0.001 |
| • Chinese | 56 (8%) | 275 (15%) | |
| • Black | 213 (31%) | 428 (24%) | |
| • Hispanic | 183 (26%) | 367 (20%) | |
| Live births median [IQR] | 3 [1 to 4] | 3 [2 to 4] | 0.31 |
| Pregnancies [IQR] | 3 [2 to 5] | 3 [2 to 5] | 0.40 |
| Type of Menopause • Natural • Surgical |
446 (64%) 247 (36%) |
1623 (89%) 193 (11%) |
<0.001 |
| Hormone Therapy • Ever Use • Never Use |
362 (53%) 319 (47%) |
837 (47%) 946 (53%) |
0.006 |
Table 2 includes baseline CVD risk characteristics of participants with or without early menopause. Women with early menopause were more often smokers, had diabetes, and had a higher average BMI.
Table 2.
Cardiovascular Disease Risk Factors of Women with and without Early Menopause (n=2509)
| Early Menopause (n=693) |
No Early Menopause (n=1816) |
p-value | |
|---|---|---|---|
| CVD Risk Factors (Baseline exam) | |||
| Smoking Never Past Current |
380 (55%) 209 (30%) 104 (15%) |
1110 (61%) 532 (29%) 174 (10%) |
<0.001 |
| Total cholesterol mg/dL | 203±37 | 201±36 | 0.36 |
| HDL Cholesterol mg/dL | 56±15 | 57±15 | 0.44 |
| Diabetes (%)a | 97 (14%) | 195 (11%) | 0.021 |
| Systolic Blood Pressure mm Hg | 129±25 | 128±23 | 0.29 |
| Diastolic Blood Pressure mm Hg | 69±11 | 69±10 | 0.68 |
| Hypertension (%)b | 337 (49%) | 863 (48%) | 0.25 |
| Family History of CVD | 323 (50%) | 811 (47%) | 0.23 |
| BMI | 28.9±6.3 | 28.2±6.0 | 0.023 |
ADA 2003 definition19 or use of diabetes medication.
JNC VI definition or use of antihypertensive medication.
Coronary Heart Disease and Stroke Events
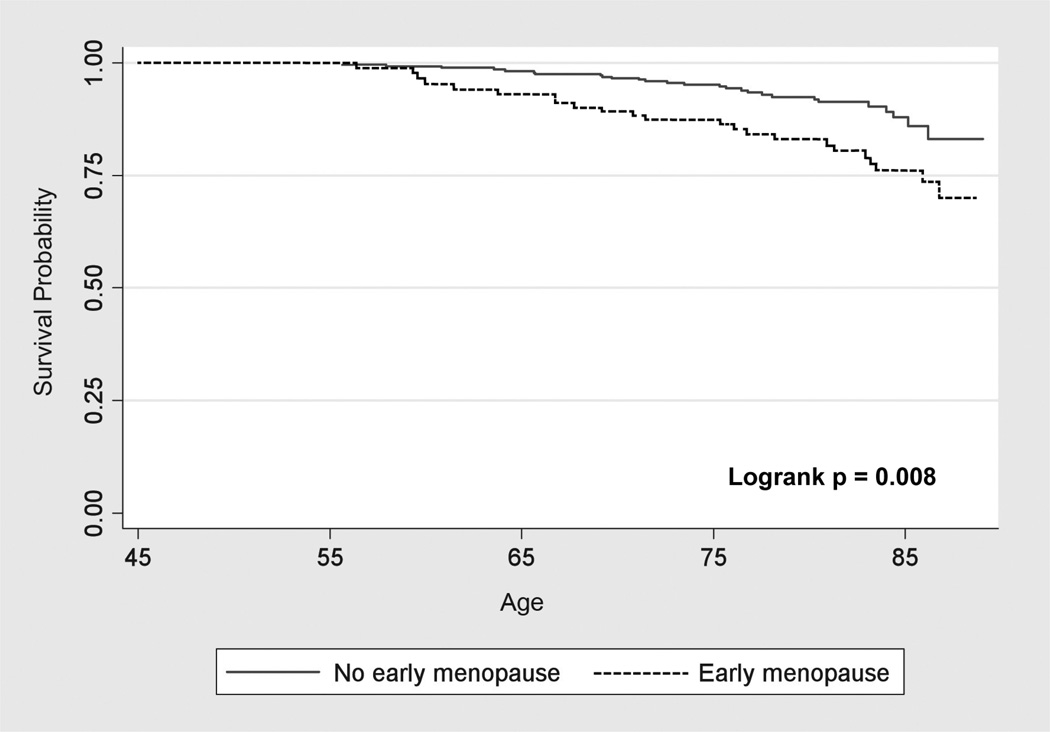
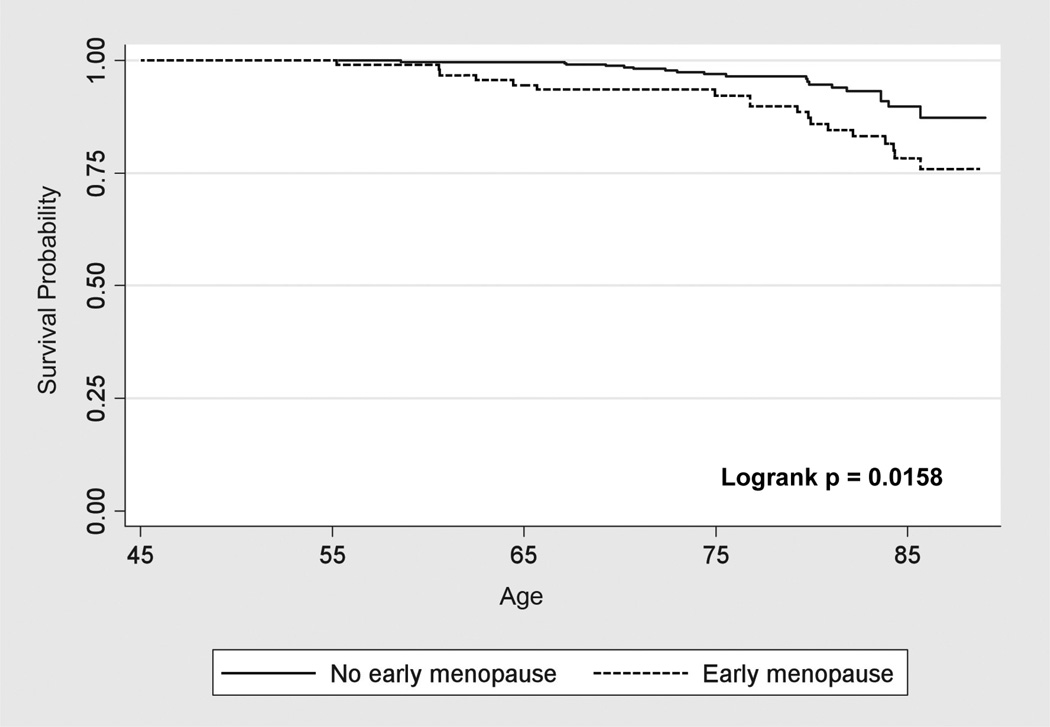
Fifty CHD events occurred (23 with early menopause vs. 27 without) and 37 stroke events (18 with early menopause vs. 19 without) during a median follow-up of 4.78 years (interquartile range 4.59 to 4.97 years). Traditional CVD risk factors were associated with incident CHD and stroke events (see Table, Supplemental Digital Content, http://links.lww.com/MENO/A28). CHD-free and stroke-free survival was significantly lower for women with early menopause (Figures 2 & 3).
Figure 2.
Kaplan Meier Survival Curves for Coronary Heart Disease in Women with and without Early Menopause.
Figure 3.
Kaplan Meier Survival Curves for Stroke in Women with and without Early Menopause.
Early Menopause as a Predictor of CHD and Stroke
Early menopause was an independent predictor of CHD and stroke after adjustment for age, race/ethnicity, and MESA site (2.11, 95% CI 1.19, 3.75 and 2.10, 95% CI 1.08, 4.07). It remained an independent predictor of CHD and stroke after further adjustment for traditional CVD risk factors (2.08, 95% CI 1.17, 3.70 and 2.19, 95% CI 1.11, 4.32). The HRs were attenuated after adjustment for family history of CVD (1.80, 95% CI 0.99, 3.29 and 1.98, 95% CI 0.98, 4.00). Further adjustment for HT use did not alter the HRs appreciably (1.85, 95% CI 1.01, 3.37 and 2.03, 95% CI 1.00, 4.10) (Table 3). In sensitivity analyses that also adjusted for education, the HRs were not significantly different.
Table 3.
Incident Coronary Heart Disease and Stroke Events in Women with and without Early Menopause (n=2509)
| Early Menopause n=693 |
No Early Menopause n=1816 |
|||
|---|---|---|---|---|
| Rate per 1000 (Annualized %) | Hazard ratio (95% CI) |
p-value | ||
| Coronary Heart Disease Events | 7.33/1000/yr | 3.22/1000/yr | ||
| Model 1 | 2.11 [1.20, 3.71] | 0.009 | ||
| Model 2 | 2.11 [1.19, 3.75] | 0.010 | ||
| Model 3 | 2.08 [1.17, 3.70] | 0.013 | ||
| Model 4 | 2.08 [1.17, 3.70] | 0.013 | ||
| Model 5 | 1.80 [0.99, 3.29] | 0.054 | ||
| Model 6 | 1.85 [1.01, 3.37] | 0.045 | ||
| Stroke Events | 5.72/1000/yr | 2.26/1000/yr | ||
| Model 1 | 2.19 [1.14, 4.21] | 0.018 | ||
| Model 2 | 2.10 [1.08, 4.07] | 0.028 | ||
| Model 3 | 2.19 [1.11, 4.32] | 0.025 | ||
| Model 4 | 2.20 [1.11, 4.35] | 0.023 | ||
| Model 5 | 1.98 [0.98, 4.00] | 0.058 | ||
| Model 6 | 2.03 [1.00, 4.10] | 0.049 | ||
Model 1: Early menopause + age (inherent time scale)
Model 2: Early Menopause + demographics (age, race/ethnicity, MESA site)
Model 3: Early Menopause + demographics + traditional CVD risk factors: ever smoke, total cholesterol, hdl cholesterol, hypertension (JNC VI or use of antihypertensive medication), diabetes (ADA 2003 or use of antidiabetic medication).
Model 4: Early Menopause + demographics + traditional CVD risk factors +BMI
Model 5: Early Menopause + demographics + traditional CVD risk factors + Family History of CVD
Model 6: Early Menopause + demographics + traditional CVD risk factors + Family History of CVD + HT use
Discrimination for CHD
The C-statistic for the traditional CVD risk factors was 0.68 in our sample. When the predicted hazard due to both early menopause and traditional CVD risk factors was used, the C-statistic was 0.70 (p=0.55 vs. traditional CVD risk factors alone).
Secondary Analyses
Adjustment for type of menopause did not alter the results significantly. Analyses did not provide evidence for interactions between early menopause and the covariates 1) HT use, 2) type of menopause, and 3) ever smoking. However, power was limited for these analyses.
Discussion
Early menopause was a significant predictor of future CHD and stroke in our population-based sample of multi-ethnic US women, independent of traditional CVD risk factors. We found that women with early menopause have approximately a 2-fold increased risk of a future CHD or stroke event. Our findings align with other large-scale epidemiologic studies of early age at natural menopause and CHD. However, most of these studies assessed CHD mortality and were predominantly in European or white cohorts.7, 8, 11, 24 For example, studies of Norwegian, Netherlands, and Seventh Day Adventist cohorts have all shown a 1.5 to 2-fold increase in CHD mortality in women with menopause at early ages (various ages <45) versus those with menopause at more average ages (various ages >49).7, 8, 11 In the Nurses’ Health Study, early natural menopause (age at menopause 40–44 vs. 50–54) and myocardial infarction were significantly related (RR 1.42, 95% CI 1.08–1.86) after adjustment for traditional risk factors.10
As compared with prior studies of early age at surgical menopause and CHD events, our findings are not as strong.6, 25 However prior studies did not always adjust for other risk factors5, 25 and included women close to the time of their surgical menopause. For our study, we defined early menopause as a self-reported menopause that occurred naturally or surgically before age 46. We lacked adequate power to test for interactions between type of menopause (natural vs. surgical) and early menopause and our CVD outcomes. Longer follow-up of the MESA cohort may provide sufficient power. This could provide valuable information for women weighing the risks and benefits of hysterectomy and oophorectomy. Currently, the risks and benefits of early hysterectomy and oophorectomy are unclear. A recent study of the Women’s Health Initiative (WHI) observational cohort found that, in women who underwent hysterectomy at age <40, oophorectomy is associated with a slightly lower risk of ovarian and possibly breast cancer without an increased risk of CVD.26
Early Menopause and Stroke
Our study showed that early menopause was associated with an increased risk of stroke. Prior studies have found a relationship between early menopause and stroke, although not consistently. Studies of a Japanese cohort27 and the Framingham cohort9 have found a two-fold increased risk of stroke in women with menopause age ages <42 as compared with women without early menopause. In the Framingham cohort, this increased risk persisted even when the sample was restricted to women who never smoked9. However, in a Norwegian cohort28 and the Nurses’ Health Study10 age at natural menopause and stroke appeared unrelated.
Factors affecting Early Menopause
Smoking and age at menopause are closely linked29–31 and smokers reach menopause on average two years earlier than non-smokers.32 Thus studies of the relationship between early menopause and CVD that do not adjust for smoking are likely confounded. Even in studies that control for smoking, significant interactions between smoking and early menopause may remain. For example, in the Nurses’ Health Study cohort (35,000 women), the significant relationship between early natural menopause and myocardial infarction disappeared among the non-smokers but remained in the smokers when these two groups were stratified. We did not have adequate power to test for an interaction between smoking history and early menopause when assessing our CVD outcomes. Longer follow-up of the MESA cohort may provide us with this information in the future.
In our study, after adjustment for family history of CVD, early menopause was no longer a statistically significant predictor of CVD events. This may be because 1) of insufficient power 2) family history of CVD is a better predictor of CVD than early menopause, or 3) the variables are highly related. The timing of menopause and CVD both appear highly heritable. Family history of premature CHD is included in CVD risk algorithms developed for postmenopausal women2. Family history of menopausal age is highly associated with age at natural menopause.33, 34 Because of these observations, cross-cohort analyses of genetic variation in cardiovascular disease and reproductive aging are currently underway.35, 36 But studies of both family history of menopause and family history of CVD are also needed. These studies could identify the optimal family and reproductive history variables for inclusion in CVD risk algorithms.
Limitations
Validity and Accuracy of Self-Report of Age at Menopause
Our study has several limitations including the accuracy and precision of self-reported menopause. Given a recent NIH consensus definition of natural menopause as “12 months of amenorrhea following the final menstrual period (FMP)”37, menstrual calendars performed at the time of waning ovarian function are a reasonable gold standard for determining menopause. However, this requires following women beginning early in life. One menstrual calendar study that began during college life found that at ~7 years post-menopause, 76% of women accurately report menopause within 1 year of their final menstrual period. On average, this recalled age is slightly higher the menstrual calendar age at menopause.
Studies of precision of natural menopause recall from the Nurses’ Health Study38 and NHANES39 have found worsening precision over time. Depending on the follow-up interval, 82%-44% of women recalls an age at menopause within 1 year of their initial report. Precision appears to decrease as time since menopause increases, but this decrease is not related to current age or education.39 It may have a regression toward the mean pattern, with women initially reporting early menopause subsequently reporting an older age at menopause.40 These previous studies of menopausal recall suggest that our findings may be biased toward the null.
Women’s accuracy of reporting ovarian removal at hysterectomy appears low with a bias toward underreporting when checked against medical records (sensitivity and PPV, 64% and 100%).41 Women’s precision in recalling age at surgical menopause appears superior to recalling age at natural menopause, with less variance over time.38, 39, 42 Given these data, some participants excluded for reporting simple hysterectomies were likely surgically menopausal; however, it is unlikely that surgically menopausal participants were misclassified. In general, accuracy and precision issues surrounding self-reported surgical menopause should not bias our findings significantly.
Exclusion of Simple Hysterectomy
Bias may be introduced by the exclusion or inclusion of women with simple hysterectomy in studies of the timing of menopause and clinical outcomes.43, 44 Loss of menstrual bleeding due to a waning of ovarian function cannot be observed in hysterectomized women. Age at menopause is thus unknown in this group. Studies including women with simple hysterectomy use varying definitions for age at menopause in women with simple hysterectomy (based on symptoms, surgery, or arbitrary age cutpoints). These varying definitions have been shown by some43 but not other44 researchers to alter conclusions significantly. We excluded women with simple hysterectomy from our analyses and our findings are not applicable to this group.
Survival Bias
MESA participants were CVD-free at baseline at ages 45–84. Women in MESA may represent survivors of early menopause who did not develop CHD or die prior to enrollment. The true point estimate for the relationship between early menopause and CVD may be larger than we observed because of survival bias.
Strengths and Implications
We found that early menopause is a moderate predictor of CHD and stroke, even after adjusting for traditional CVD risk factors in a diverse population of US women. This may suggest that early menopause, if possible, should be avoided and that women with early menopause may be a group to target for aggressive CVD prevention strategies. Prior to the WHI trial findings, physicians recommended oral HT anticipating that this therapy would negate any detrimental cardiovascular effects associated with menopause. Given the lack of cardioprotective benefit and potential harms of menopausal HT16, 17, long term therapy with HT is no longer recommended for postmenopausal women.45 However, the use of HT in women with early menopause until the time of an average age of menopause (~50) remains in guidelines46, 47, although a paucity of data exists regarding the risks and benefits of this approach.
Given the current lack of evidence that peri-menopausal or menopausal HT is cardioprotective, reducing the incidence of early surgical menopause by reducing elective oophorectomy may be an important step to reduce CVD risk. Since the release of the WHI results, the elective oophorectomy rate has decreased.48 In 2008, the American College of Obstetrics and Gynecology suggested that elective oophorectomy should be avoided in premenopausal women.49 However, although women from the WHI observational cohort with hysterectomy have worse CVD risk as compared with women without hysterectomy50, recent data from the WHI suggests that simple hysterectomy and hysterectomy with oophorectomy carry equivalent CVD risks.26 This suggests that CVD risk factors may contribute to problems underlying to the decision to undergo hysterectomy as well as to the development of CVD.
It is possible that CVD risk early in life contributes to both early menopause and CVD through similar mechanisms. Trajectories of risk factors for CVD (such as cholesterol, weight, and blood pressure) measured during the premenopause have been associated with the timing of subsequent natural menopause in one study.51 Smoking is associated with early natural menopause in numerous studies29–31, although there is no available evidence that smoking cessation extends menopausal age. Reducing early menopause by eliminating smoking exposure could provide an additional benefit from smoking cessation.
Conclusion
In summary, our findings demonstrate a moderate association between early menopause and future CHD and stroke. This adds to the body of evidence that early menopause may identify the at-risk woman who may benefit from aggressive CVD primary prevention. Strategies to prevent early menopause, such as avoidance of smoking and oophorectomy, may have significant public health relevance for the prevention of CVD in women.
Supplementary Material
Acknowledgements
Funding/Support: This research was supported by contracts N01-HC-95159 through N01-HC-95169 from the National Institutes of Health (NIH)/National Heart, Lung, and Blood Institute (NHLBI). Drs. Ouyang, Schreiner, Herrington and Vaidya receive grant support from the NIH. Dr. Wellons is supported by a career development award K23-HL-87114 from the NIH/NHLBI. Dr. Wellons and Ouyang receive consulting fees from the Society for Women’s Health Research and the Women’s Health Initiative (NIH), respectively.
The authors thank the other investigators, the staff, and the participants of the MESA study for their valuable contributions. A full list of participating MESA investigators and institutions can be found at http://www.mesa-nhlbi.org.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Financial disclosures/conflicts of interest: None reported.
References
- 1.Leading causes of death in females, United States. [Accessed July 27, 2010];Atlanta:Centers for Disease Control and Prevention (US) 2006 Available at: http://www.cdc.gov/women/lcod/.
- 2.Ridker P, Buring J, Rifai N, Cook N. Development and validation of improved algorithms for the assessment of global cardiovascular risk in women: the Reynolds Risk Score. JAMA. 2007 Feb;297(6):611–619. doi: 10.1001/jama.297.6.611. [DOI] [PubMed] [Google Scholar]
- 3.Expert Panel on Detection E, and Treatment of High Blood Cholesterol in Adults. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III) JAMA. 2001 May;285(19):2486–2497. doi: 10.1001/jama.285.19.2486. [DOI] [PubMed] [Google Scholar]
- 4. [Accessed November 1, 2010];Expert Panel on Detection E, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Framingham 10-year Risk Calculator (online version) 2004 http://hp2010.nhlbihin.net/atpiii/calculator.asp?usertype=prof.
- 5.Gordon T, Kannel W, Hjortland M, McNamara P. Menopause and coronary heart disease. The Framingham Study. Ann Intern Med. 1978 Aug;89(2):157–161. doi: 10.7326/0003-4819-89-2-157. [DOI] [PubMed] [Google Scholar]
- 6.Kannel W, Hjortland M, McNamara P, Gordon T. Menopause and risk of cardiovascular disease: the Framingham study. Ann Intern Med. 1976 Oct;85(4):447–452. doi: 10.7326/0003-4819-85-4-447. [DOI] [PubMed] [Google Scholar]
- 7.Jacobsen B, Nilssen S, Heuch I, Kvåle G. Does age at natural menopause affect mortality from ischemic heart disease? J Clin Epidemiol. 1997 Apr;50(4):475–479. doi: 10.1016/s0895-4356(96)00425-8. [DOI] [PubMed] [Google Scholar]
- 8.Jacobsen B, Knutsen S, Fraser G. Age at natural menopause and total mortality and mortality from ischemic heart disease: the Adventist Health Study. J Clin Epidemiol. 1999 Apr;52(4):303–307. doi: 10.1016/s0895-4356(98)00170-x. [DOI] [PubMed] [Google Scholar]
- 9.Lisabeth L, Beiser A, Brown D, Murabito J, Kelly-Hayes M, Wolf P. Age at natural menopause and risk of ischemic stroke: the Framingham heart study. Stroke. 2009 Apr;40(4):1044–1049. doi: 10.1161/STROKEAHA.108.542993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hu F, Grodstein F, Hennekens C, et al. Age at natural menopause and risk of cardiovascular disease. Arch Intern Med. 1999 May;159(10):1061–1066. doi: 10.1001/archinte.159.10.1061. [DOI] [PubMed] [Google Scholar]
- 11.van der Schouw Y, van der Graaf Y, Steyerberg E, Eijkemans J, Banga J. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996 Mar;347(9003):714–718. doi: 10.1016/s0140-6736(96)90075-6. [DOI] [PubMed] [Google Scholar]
- 12.Snowdon D, Kane R, Beeson W, et al. Is early natural menopause a biologic marker of health and aging? Am J Public Health. 1989 Jun;79(6):709–714. doi: 10.2105/ajph.79.6.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Summary of the second report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel II) JAMA. 1993 Jun;269(23):3015–3023. [PubMed] [Google Scholar]
- 14.Matthews K, Meilahn E, Kuller L, Kelsey S, Caggiula A, Wing R. Menopause and risk factors for coronary heart disease. N Engl J Med. 1989 Sep;321(10):641–646. doi: 10.1056/NEJM198909073211004. [DOI] [PubMed] [Google Scholar]
- 15.Grundy S, Cleeman J, Merz C, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol. 2004 Aug;44(3):720–732. doi: 10.1016/j.jacc.2004.07.001. [DOI] [PubMed] [Google Scholar]
- 16.Rossouw J, Anderson G, Prentice R, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial. JAMA. 2002 Jul;288(3):321–333. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- 17.Anderson G, Limacher M, Assaf A, et al. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women's Health Initiative randomized controlled trial. JAMA. 2004 Apr;291(14):1701–1712. doi: 10.1001/jama.291.14.1701. [DOI] [PubMed] [Google Scholar]
- 18.Bild DE, Bluemke DA, Burke GL, et al. Multi-ethnic study of atherosclerosis: objectives and design. Am J Epidemiol. 2002 Nov;156(9):871–881. doi: 10.1093/aje/kwf113. [DOI] [PubMed] [Google Scholar]
- 19.Mellitus ECotDaCoD. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003 Jan;26(Suppl 1):S5–S20. doi: 10.2337/diacare.26.2007.s5. [DOI] [PubMed] [Google Scholar]
- 20.Golden SH, Dobs AS, Vaidya D, et al. Endogenous sex hormones and glucose tolerance status in postmenopausal women. J Clin Endocrinol Metab. 2007 Apr;92(4):1289–1295. doi: 10.1210/jc.2006-1895. [DOI] [PubMed] [Google Scholar]
- 21.Rayburn WF. Sixth report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure. A summary. J Reprod Med. 1998 May;43(5):444–450. [PubMed] [Google Scholar]
- 22.Hysterectomy. National Women's Health Information Center (2006-07-01) 2009 www.4women.gov/faq/hysterectomy.htm,
- 23.Feinstein M, Liu K, Ning H, Fitchett G, Lloyd-Jones DM. Burden of cardiovascular risk factors, subclinical atherosclerosis, and incident cardiovascular events across dimensions of religiosity: The multi-ethnic study of atherosclerosis. Circulation. 2010 Feb;121(5):659–666. doi: 10.1161/CIRCULATIONAHA.109.879973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Løkkegaard E, Jovanovic Z, Heitmann B, Keiding N, Ottesen B, Pedersen A. The association between early menopause and risk of ischaemic heart disease: influence of Hormone Therapy. Maturitas. 2006 Jan;53(2):226–233. doi: 10.1016/j.maturitas.2005.04.009. [DOI] [PubMed] [Google Scholar]
- 25.Rosenberg L, Hennekens C, Rosner B, Belanger C, Rothman K, Speizer F. Early menopause and the risk of myocardial infarction. Am J Obstet Gynecol. 1981 Jan;139(1):47–51. doi: 10.1016/0002-9378(81)90410-5. [DOI] [PubMed] [Google Scholar]
- 26.Jacoby VL, Grady D, Wactawski-Wende J, et al. Oophorectomy vs Ovarian Conservation With Hysterectomy: Cardiovascular Disease, Hip Fracture, and Cancer in the Women's Health Initiative Observational Study. Arch Intern Med. 2011 Apr;171(8):760–768. doi: 10.1001/archinternmed.2011.121. [DOI] [PubMed] [Google Scholar]
- 27.Baba Y, Ishikawa S, Amagi Y, Kayaba K, Gotoh T, Kajii E. Premature menopause is associated with increased risk of cerebral infarction in Japanese women. Menopause. 2009 Dec; doi: 10.1097/gme.0b013e3181c7dd41. [DOI] [PubMed] [Google Scholar]
- 28.Jacobsen B, Heuch I, Kvåle G. Age at natural menopause and stroke mortality: cohort study with 3561 stroke deaths during 37-year follow-up. Stroke. 2004 Jul;35(7):1548–1551. doi: 10.1161/01.STR.0000131746.49082.5c. [DOI] [PubMed] [Google Scholar]
- 29.Kaufman D, Slone D, Rosenberg L, Miettinen O, Shapiro S. Cigarette smoking and age at natural menopause. Am J Public Health. 1980 Apr;70(4):420–422. doi: 10.2105/ajph.70.4.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Willett W, Stampfer M, Bain C, et al. Cigarette smoking, relative weight, and menopause. Am J Epidemiol. 1983 Jun;117(6):651–658. doi: 10.1093/oxfordjournals.aje.a113598. [DOI] [PubMed] [Google Scholar]
- 31.McKinlay S, Bifano N, McKinlay J. Smoking and age at menopause in women. Ann Intern Med. 1985 Sep;103(3):350–356. doi: 10.7326/0003-4819-103-3-350. [DOI] [PubMed] [Google Scholar]
- 32.Society NAM. Menopause Guidebook. Cleveland: North American Menopause Society; 2006. [Google Scholar]
- 33.Cramer DW, Xu H, Harlow BL. Family history as a predictor of early menopause. Fertil Steril. 1995 Oct;64(4):740–745. doi: 10.1016/s0015-0282(16)57849-2. [DOI] [PubMed] [Google Scholar]
- 34.Torgerson DJ, Thomas RE, Reid DM. Mothers and daughters menopausal ages: is there a link? Eur J Obstet Gynecol Reprod Biol. 1997 Jul;74(1):63–66. doi: 10.1016/s0301-2115(97)00085-7. [DOI] [PubMed] [Google Scholar]
- 35.Musunuru K, Lettre G, Young T, et al. Candidate gene association resource (CARe): design, methods, and proof of concept. Circ Cardiovasc Genet. 2010 Jun;3(3):267–275. doi: 10.1161/CIRCGENETICS.109.882696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Psaty BM, O'Donnell CJ, Gudnason V, et al. Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium: Design of prospective meta-analyses of genome-wide association studies from 5 cohorts. Circ Cardiovasc Genet. 2009 Feb;2(1):73–80. doi: 10.1161/CIRCGENETICS.108.829747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Soules MR, Sherman S, Parrott E, et al. Executive summary: Stages of Reproductive Aging Workshop (STRAW) Fertil Steril. 2001 Nov;76(5):874–878. doi: 10.1016/s0015-0282(01)02909-0. [DOI] [PubMed] [Google Scholar]
- 38.Colditz GA, Stampfer MJ, Willett WC, et al. Reproducibility and validity of self-reported menopausal status in a prospective cohort study. Am J Epidemiol. 1987 Aug;126(2):319–325. doi: 10.1093/aje/126.2.319. [DOI] [PubMed] [Google Scholar]
- 39.Hahn R, Eaker E, Rolka H. Reliability of reported age at menopause. Am J Epidemiol. 1997 Nov;146(9):771–775. doi: 10.1093/oxfordjournals.aje.a009353. [DOI] [PubMed] [Google Scholar]
- 40.Rödström K, Bengtsson C, Lissner L, Björkelund C. Reproducibility of self-reported menopause age at the 24-year follow-up of a population study of women in Göteborg, Sweden. Menopause. 2005 May-Jun;12(3):275–280. doi: 10.1097/01.gme.0000135247.11972.b3. 2005. [DOI] [PubMed] [Google Scholar]
- 41.Phipps AI, Buist DS. Validation of self-reported history of hysterectomy and oophorectomy among women in an integrated group practice setting. Menopause. 2009 May-Jun;16(3):576–581. doi: 10.1097/gme.0b013e31818ffe28. 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.den Tonkelaar I. Validity and reproducibility of self-reported age at menopause in women participating in the DOM-project. Maturitas. 1997 Jun;27(2):117–123. doi: 10.1016/s0378-5122(97)01122-5. [DOI] [PubMed] [Google Scholar]
- 43.Rockhill B, Colditz G, Rosner B. Bias in breast cancer analyses due to error in age at menopause. Am J Epidemiol. 2000 Feb;151(4):404–408. doi: 10.1093/oxfordjournals.aje.a010220. [DOI] [PubMed] [Google Scholar]
- 44.Phipps AI, Ichikawa L, Bowles EJ, et al. Defining menopausal status in epidemiologic studies: A comparison of multiple approaches and their effects on breast cancer rates. Maturitas. 2010 Sep;67(1):60–66. doi: 10.1016/j.maturitas.2010.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Taylor HS, Manson JE. Update in hormone therapy use in menopause. J Clin Endocrinol Metab. 2011 Feb;96(2):255–264. doi: 10.1210/jc.2010-0536. [DOI] [PubMed] [Google Scholar]
- 46.Santen R, Allred D, Ardoin S, et al. Postmenopausal hormone therapy: an Endocrine Society scientific statement. J Clin Endocrinol Metab. 2010 Jul;95(7 Suppl 1):s1–s66. doi: 10.1210/jc.2009-2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Estrogen and progestogen use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010 Mar;17(2):242–255. doi: 10.1097/gme.0b013e3181d0f6b9. [DOI] [PubMed] [Google Scholar]
- 48.Asante A, Whiteman MK, Kulkarni A, Cox S, Marchbanks PA, Jamieson DJ. Elective oophorectomy in the United States: trends and in-hospital complications, 1998–2006. Obstet Gynecol. 2010 Nov;116(5):1088–1095. doi: 10.1097/AOG.0b013e3181f5ec9d. [DOI] [PubMed] [Google Scholar]
- 49.ACOG. ACOG Practice Bulletin No. 89. Elective and risk-reducing salpingo-oophorectomy. Obstet Gynecol. 2008 Jan;111(1):231–241. doi: 10.1097/01.AOG.0000291580.39618.cb. [DOI] [PubMed] [Google Scholar]
- 50.Howard BV, Kuller L, Langer R, et al. Risk of cardiovascular disease by hysterectomy status, with and without oophorectomy: the Women's Health Initiative Observational Study. Circulation. 2005 Mar 29;111(12):1462–1470. doi: 10.1161/01.CIR.0000159344.21672.FD. [DOI] [PubMed] [Google Scholar]
- 51.Kok H, van Asselt K, van der Schouw Y, et al. Heart disease risk determines menopausal age rather than the reverse. J Am Coll Cardiol. 2006 May;47(10):1976–1983. doi: 10.1016/j.jacc.2005.12.066. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.