Abstract

Synthetic efforts toward the chlorinated propellane alkaloid acutumine (1) are described. The key vicinal quaternary centers were constructed by a photochemical [2+2] cycloaddition reaction of a furanyl-tetrahydroindolone. Dihydroxylation of the [2+2] product enabled a tandem retro-aldol/intramolecular ketalization reaction, which revealed the aza-propellane core of 1 while generating an unusual, caged, pentacyclic hemi-ketal product.
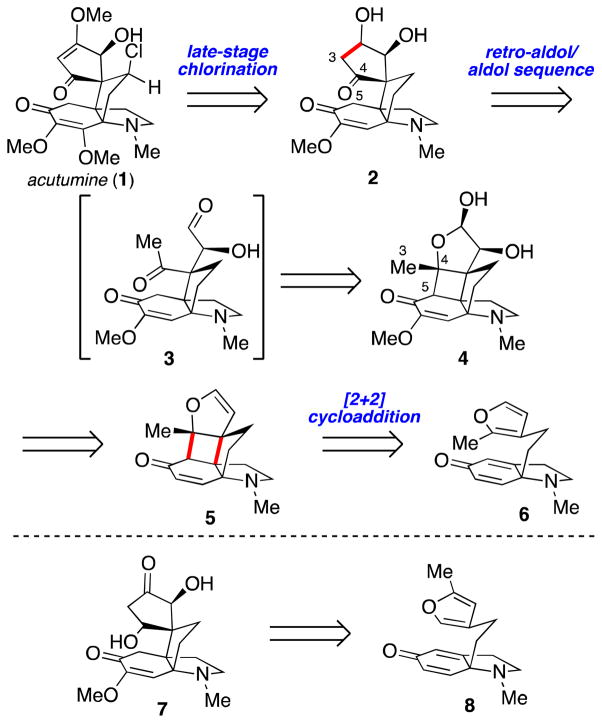
Acutumine (1, Scheme 1) is a chlorinated aza-propellane alkaloid first isolated in 1929 by Goto and Sudzuki from the medicinal herb Sinomenium acutum.1,2 This densely functionalized small molecule exhibits promising biological properties, including selective T-cell cytotoxicity3 and antiamnesic activity.4 The aza-propellane skeleton is adorned with a spirocyclic cyclopentenone moiety, and contains a neopentyl chloride and two all-carbon quaternary centers embedded within five contiguous stereogenic carbons. Although its structural and biological features have attracted attention from the synthetic community,5 only a single enantioselective synthesis of the alkaloid has been reported to date.6
Scheme 1.
Retrosynthetic analysis of acutumine (1).
As a part of our program aimed at developing a unified synthetic strategy toward several structurally distinct aza-propellane alkaloids, we recently reported the preparation of N-tert-butanesulfinimine 9 (see Scheme 2), a compound that undergoes highly diastereoselective 1,2-addition reactions with a variety of organometallic reagents.7,8,9 Based on these findings, we were able to complete concise total syntheses of the hasubanan alkaloid 8-demethoxyrunanine, as well as the structurally related compounds cepharatines A, C and D.7 Herein, we report that addition of furanyl-based nucleophiles to N-tert-butanesulfinimine 9 enables the rapid construction of the aza-propellane core of acutumine by a photochemical [2+2] cycloaddition/retro-aldol sequence.
Scheme 2.
Diastereoselective 1,2-addition of furanyl Grignard nucleophiles.
Retrosynthetically, acutumine (1) was simplified to diol 2, an intermediate envisioned to arise from lactol 4 by a retro-aldol/aldol sequence (Scheme 1). In this key reaction, the strained nature of the cyclobutane embedded within 4 was expected to facilitate cleavage of the C4–C5 bond to give keto-aldehyde 3, which is poised to undergo intramolecular aldol ring-closure. We hypothesized that lactol 4 could be prepared by dihydroxylation of the corresponding dihydrofuran (5), which itself was expected to arise from an intramolecular photochemical [2+2] cycloaddition of furanyl-dihydroindolone 6.
Alternatively, we recognized that isomeric dihydroindolone 8 could also be a viable intermediate en route to acutumine. Indeed, its advancement through the same reaction sequence was anticipated to produce cyclopentanone 7, an isomeric intermediate also amenable to elaboration to 1. We expected both dihydroindolones 6 and 8 to be accessible from 9 by short reaction sequences involving Grignard addition, N-methylation, and pyrrolidine formation.7,8
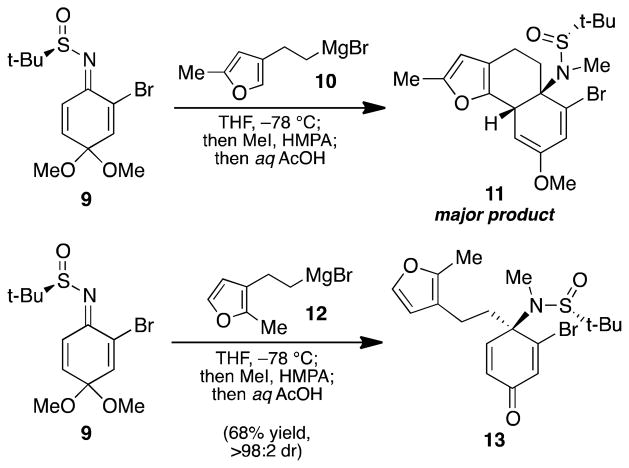
To rapidly assess the feasibility of our [2+2] cycloaddition/retro-aldol/aldol strategy, we began our studies by targeting compounds 6 and 8. To this end, we examined the use of furan-containing Grignard reagents 10 and 12 as nucleophiles for our sulfinimine methodology. Exposure of bromo-sulfinimine 9 to Grignard reagent 10 at −78 °C resulted in the expected 1,2-addition reaction; however, upon purification by flash chromatography, the major product was enol ether 11 (Scheme 2).10 Presumably, the mildly acidic silica gel mediates an intramolecular Friedel–Crafts type conjugate addition. Although it might have been possible to identify purification conditions that allowed for isolation of the 1,2-addition product, the acid sensitivity of this substrate suggested it would not be amenable to pyrrolidine formation using our previously established conditions.7 We hypothesized that in the analogous 2,3-disubstituted furanyl substrate (12), the C2 methyl group should mitigate this type of reactivity and allow for the isolation of the corresponding 1,2-addition product. This hypothesis was validated when, upon treatment of sulfinimine 9 with Grignard reagent 12 followed by in situ methylation, sulfinamide 13 was isolated in 68% yield as a single diastereomer.
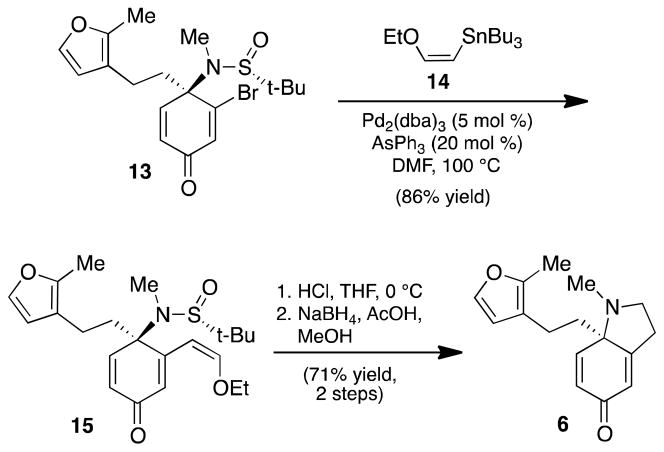
Sulfinamide 13 was elaborated to dihydroindolone 6 following our optimized three-step protocol for pyrrolidine formation. Thus, Pd-catalyzed cross-coupling of sulfinamide 13 with vinyl stannane 14 proceeded smoothly to give enol ether 15 (Scheme 3). Acid-mediated cyclization furnished an enamine intermediate, which was then selectively reduced to afford photocycloaddition substrate 6 in 61% yield over three steps.
Scheme 3.
Preparation of dihydroindolone 6.
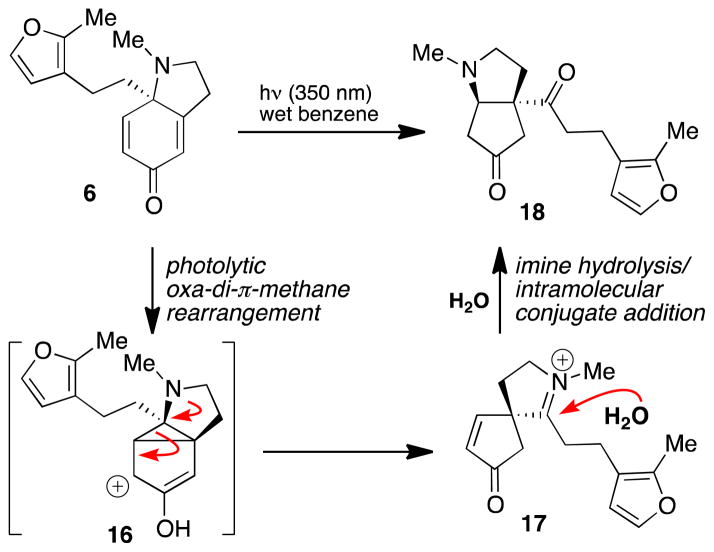
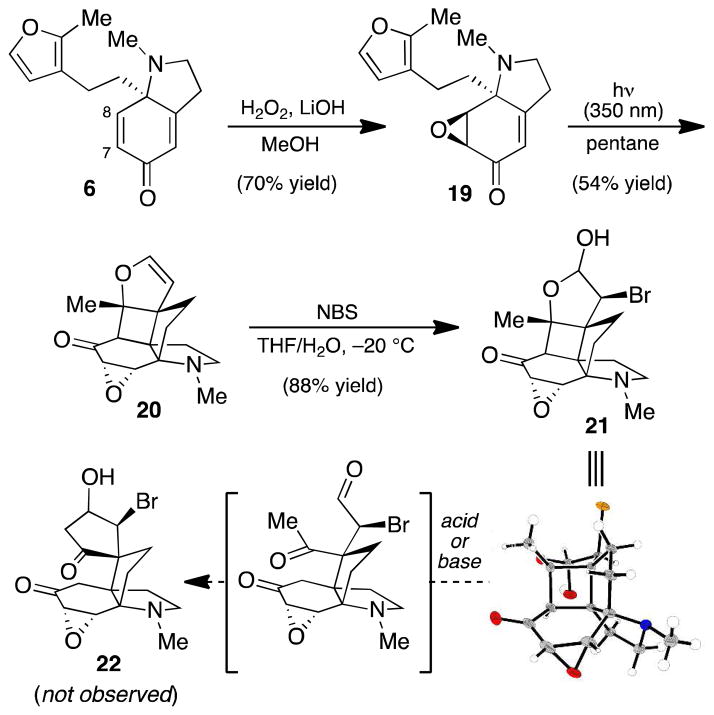
With dihydroindolone 6 in hand, we conducted a screen of photochemical reaction conditions, varying both the solvent and the irradiation wavelength.11 Irradiation of 6 at 350 nm in benzene did afford trace amounts of the desired [2+2] product (as determined by 1H NMR analysis). However, following characterization by NMR and IR spectroscopy and HRMS, the major product of this reaction was assigned as diketone 18 (Scheme 4). Diketone 18 is hypothesized to occur by a photolytic oxa-di-π-methane rearrangement,12 which, following C–C bond fragmentation, generates the unstable iminium ion 17. Nucleophilic addition of adventitious water results in iminium hydrolysis and intramolecular conjugate addition to produce 18. To circumvent this type of undesired reactivity, the C7–C8 enone functionality was masked as its epoxide under nucleophilic epoxidation conditions (Scheme 5). We were pleased to find that photochemical [2+2] cycloaddition of enone 19 proved more fruitful: irradiation of 19 at 350 nm in pentane delivered dihydrofuran 20 in 54% yield. Notably, this reaction exhibits a high degree of regioselectivity (for the linear vs. crossed product) and installs the vicinal all-carbon quaternary centers of acutumine in a single step.
Scheme 4.
[2+2] Photocycloaddition of dihydroindolone 6.
Scheme 5.
[2+2] Photocycloaddition of epoxy enone 19.
Having successfully prepared the propellane core of acutumine (1), we examined the conversion of the dihydrofuran to a viable retro-aldol/aldol substrate. Whereas subjection of 20 to a number of electrophilic epoxidation conditions led primarily to N-oxidation, exposure to N-bromosuccinimide in aqueous THF13 afforded bromohydrin 21 in excellent yield (~3:1 mixture of diastereomers at the lactol carbon). The structural assignment of 21 was confirmed by single-crystal X-ray diffraction. To our dismay, attempts to effect a retro-aldol fragmentation of 21 under mildly acidic or basic conditions led to significant decomposition and failed to produce detectable quantities of 22 or similar fragmentation products.
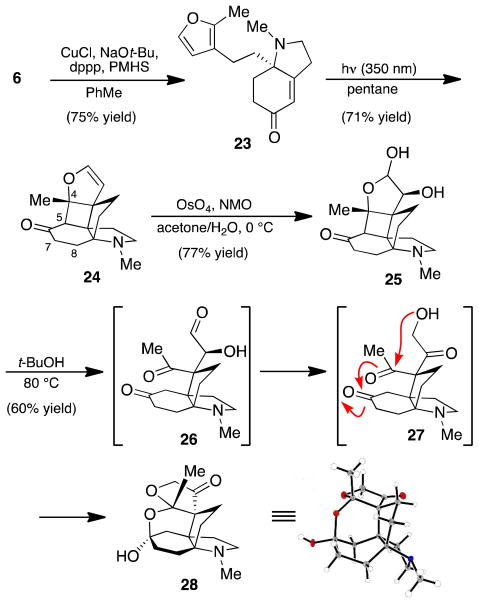
In light of these results, we suspected that the epoxide moiety of 21 might be a source of undesired reactivity under our retro-aldol conditions. We turned our attention to the construction of dihydrofuran 24, an intermediate in which the C7–C8 alkene has been reduced (Scheme 6). To this end, Cu-catalyzed conjugate reduction of 6 furnished enone 23, a substrate that performed well under our optimized photochemical [2+2] reaction conditions to give dihydrofuran 24 in 71% yield. Oxidation of dihydrofuran 24 was achieved under Upjohn dihydroxylation conditions,14 providing diol 25 in good yield.
Scheme 6.
Isolation of ketal rearrangement product 28.
With access to lactol 25, a number of conditions were evaluated for their ability to cleave the C4–C5 bond by a retro-aldol reaction. Unfortunately, as observed with bromohydrin 21, a variety of acidic or basic conditions failed to deliver the desired product. However, we noticed that a benzene stock solution of diol 25 used for TLC analysis slowly developed a single, new spot over the course of several days. To our surprise, isolation and single-crystal X-ray diffraction of this compound identified it as hemiketal 28.
It is proposed that aldehyde 26, the product directly formed by retro-aldol fragmentation of 25, readily tautomerizes to keto-alcohol 27. Formation of this primary alcohol triggers an intramolecular cyclization cascade, leading to hemiketal 28. A brief solvent screen identified t-BuOH as the optimal solvent for this rearrangement, which could be achieved in 60% yield when the reaction mixture was heated to 80 °C. Ketal 28 has thus far proven recalcitrant to our efforts toward further elaboration to 1.
In conclusion, we have enantioselectively prepared hemiketal 28, a compound that contains the aza-propellane core of acutumine, in only eight steps from N-tert-butanesulfinimine 9. The key vicinal quaternary centers were constructed by a photochemical [2+2] cycloaddition reaction of furanyl-tetrahydroindolone 23 to give 24, which was elaborated by dihydroxylation and retro-aldol fragmentation to ketal 28. Although it is unlikely that 28 will serve as an intermediate en route to acutumine, these studies confirm the viability of a [2+2] cycloaddition/retro-aldol sequence to prepare the aza-propellane core. Studies aimed at advancing 25 and more appropriately oxidized intermediates to the natural product are ongoing in our laboratory.
Supplementary Material
Acknowledgments
We thank several of our colleagues at the California Institute of Technology: Dr. Michael Day and Mr. Larry Henling for X-ray crystallographic structural determination, Dr. David VanderVelde for assistance with NMR structural determination, as well as Prof. Brian Stoltz, Dr. Scott Virgil, and the Caltech Center for Catalysis and Chemical Synthesis for access to analytical equipment. Fellowship support was provided by the Gates Millennium Scholars Program (R.N.) and the NIH (R.N., F31GM098025). HRMS and X-ray crystallographic data were obtained on instruments purchased through awards to the California Institute of Technology by the NSF CRIF program (CHE-0639094, CHE-0541745). Financial support from the California Institute of Technology and the NSF (CAREER-1057143) is gratefully acknowledged.
Footnotes
Supporting Information Available: Experimental procedures and spectral data (1H and 13C NMR, IR, and HRMS) for all new compounds. This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Original isolation paper: Goto K, Sudzuki H. Bull Chem Soc Jpn. 1929;4:220.
- 2.Structural assignment: Tomita M, Okamoto Y, Kikuchi T, Osaki K, Nishikawa M, Kamiya K, Sasaki Y, Matoba K, Goto K. Chem Pharm Bull. 1971;19:770.Tomita M, Okamoto Y, Kikuchi T, Osaki K, Nishikawa M, Kamiya K, Sasaki Y, Matoba K, Goto K. Tetrahedron Lett. 1967:2421.Tomita M, Okamoto Y, Kikuchi T, Osaki K, Nishikawa M, Kamiya K, Sasaki Y, Matoba K, Goto K. Tetrahedron Lett. 1967:2425.
- 3.Yu BW, Chen JY, Wang YP, Cheng KF, Li XY, Qin GW. Phytochemistry. 2002;61:439. doi: 10.1016/s0031-9422(02)00162-0. [DOI] [PubMed] [Google Scholar]
- 4.Qin GW, Tang XC, Lestage P, Caignard DH, Renard P. PCT Int Appl WO 2004000815. 2003 [Google Scholar]
- 5.For synthetic studies toward acutumine, see: Nguyen TX. PhD Thesis. University of California; San Diego: 2009. Moreau RJ, Sorensen EJ. Tetrahedron. 2007;63:6446.
- 6.(a) Li F, Tartakoff SS, Castle SL. J Am Chem Soc. 2009;131:6674. doi: 10.1021/ja9024403. [DOI] [PubMed] [Google Scholar]; (b) Li F, Tartakoff SS, Castle SL. J Org Chem. 2009;74:9082. doi: 10.1021/jo902006q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chuang KV, Navarro R, Reisman SE. Angew Chem Int Ed. 2011;50:9447. doi: 10.1002/anie.201104487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chuang KV, Navarro R, Reisman SE. Chem Sci. 2011;2:1086. [Google Scholar]
- 9.For a seminal report regarding N-tert-butanesulfinimines: Ellman JA, Cogan DA. J Am Chem Soc. 1999;121:268.
- 10.Enol ether 11 was isolated in 54% yield as a difficult to separate 9:1 mixture with an unidentified side product. Analytically pure material was obtained by multiple purifications. See Supporting Information.
- 11.For examples of intramolecular [2+2] photocycloaddition between furan and enone systems, see: Fontana G, Savona G, Vivona N, Rodriguez B. Eur J Org Chem. 1999:2011.Crimmins MT, Pace JM, Nantermet PG, Kim-Meade AS, Thomas JB, Watterson SH, Wagman AS. J Am Chem Soc. 1999;121:10249.
- 12.For examples of oxa-di-π-methane rearrangements of cyclohexadienones, see: Pirrung MC, Nunn DS. Tetrahedron. 1996;52:5707.Schuster DI, Brisimitzakis AC. J Org Chem. 1987;52:3644.Zimmerman HE, Swenton JS. J Am Chem Soc. 1967;89:906.Zimmerman HE, Schuster DI. J Am Chem Soc. 1961;83:4486.Barton DHR, de Mayo P, Shafiq M. J Chem Soc. 1958:3314.
- 13.de Cienfuegos LA, Mota AJ, Robles R. Org Lett. 2005;7:2161. doi: 10.1021/ol050496v. [DOI] [PubMed] [Google Scholar]
- 14.VanRheenen V, Kelly RC, Cha DY. Tetrahedron Lett. 1976;17:1973. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.