Abstract
The pathogenesis of viral bronchiolitis is poorly understood. The aim of this study was to analyze interleukin (IL)-15, IL-18 and interferon (IFN)-γ concentrations and the activity of NK cells and CD4+ and CD8+ lymphocytes in 23 children not older than 30 months of age with acute viral bronchiolitis using blood samples drawn within the first 24 h of their hospital admission, in comparison to a healthy group. In children with bronchiolitis, the mean concentrations of IL-15, IL-18 and IFN-γ were 9.39±11.55, 884.03±645.44 and 17.92±27.14 pg/ml, respectively, and were significantly higher than those in the control group [2.34±0.61 pg/ml (p<0.05), 248.69±98.73 pg/ml (p<0.001) and 2.75±1.72 pg/ml (p<0.005), respectively]. In the bronchiolitis group, mean z-scores were −1.15±1.9 for CD4+ cells and −0.9±1.23 for CD8+ cells; these scores were significantly lower than those of the general Polish population (p<0.001 and <0.01, respectively). However, the mean z-score of the ratio of CD4+/CD8+ and the NK cell count in children with bronchiolitis did not differ significantly from those of the controls. In conclusion, cytokines such as IL-15, IL-18 and IFN-γ play a role in the pathogenesis of bronchiolitis in children.
Keywords: bronchiolitis, cytokines, interleukin-15, interleukin-18, interferon-γ, NK cells, children
Introduction
Bronchiolitis in children is a serious self-limiting disease (mortality rate <1%) of respiratory tract infections. However, in high-risk groups such as children with bronchopulmonary dysplasia, congenital heart disease or cystic fibrosis, mortality increases to 5–10%. The leading cause of bronchiolitis are viral infections, with the most common agent being respiratory syncytial virus infection (RSV) (60–80% of cases) (1–4). During RSV infection, the cytokine cascade is activated, leading to the activation of Th1 and Th2 lymphocytes. Thus, an increase in the concentrations of cytokines such as interleukin (IL)-2, -6, -10, -12, -13 and a decrease in interferon (IFN)-γ and IL-4 concentrations are observed (5–12). During non-RSV viral infections, the Th1 type response of the immunologic system with an increase in the IFN-γ concentration is commonly observed (5,8,13,14).
IL-15 and IL-18 are relatively newly discovered cytokines that are produced principally by macrophages during immune response. IL-15 has multiple biological properties, including the induction of the production of other cytokines and the inhibition of T-cell apoptosis (15,16). IL-18 is a pro-inflammatory cytokine with pleiotropic properties and plays a crucial role in the maintenance of Th1-cell response. This cytokine activates NK cells, leads to eosinophilia and increases histamine concentrations (17–20). There are various reports indicating that, during viral infection, IL-15, IL-18 and IFN-γ concentrations are elevated (21). A secondary increase in NK cell activity has also been reported (22). However, the role of IL-15 and IL-18 in viral bronchiolitis in children remains unknown.
The aim of this study was to analyze IL-15, IL-18 and IFN-γ concentrations and the activity of NK cells as well as CD4+ and CD8+ lymphocytes in children with acute viral bronchiolitis.
Patients and methods
Twenty-three children with clinical presentation of viral bronchiolitis aged 3–30 months (median 9) were included in the study as the patient group. The concentrations of cytokines and T lymphocytes were analyzed in blood samples drawn within the first 2 h of hospital admission.
The control group consisted of 15 age-matched children for IL-18, 14 for IL-15 and 30 for IFN-γ. As a cut-off for the normal range of analyzed cytokines, values up to the 95th percentile were allowed.
The concentrations of IL-15, IL-18 and IFN-γ were determined using ELISA: OPT EIA Human IL-15 (Pharmingen), Human ELISA IL-18 (R&D Systems), and the OPT EIA Human IFN Gamma kit (Pharmingen).
CD4+, CD8+ and NK cell activity was analyzed using the Coulter Epics XL 40256 flow cytometer. Monoclonal antibodies against CD3, CD4, CD8, CD16 and CD56 (Dako) were applied. The cells with a CD3−(CD16+CD56)+ phenotype were defined as NK cells. Results were expressed as age- and gender-matched z-scores ± 1 SDS when compared to Polish population reference values (23). The Z-score was calculated using the equation: z-score = X − Xmean/SD, where X = the result and Xmean and SD are the mean value and standard deviation. The normal values were between the 5th and 95th percentile of the calculated z-scores (mean ± 1.645 SD of z-scores).
The etiology of infection was identified with the serological Becton-Dickinson Directigen™ RSV test kit and with the Lencomm Euroimmun Pneumo FIDE M (RTP1) kit, which detect viruses such as RSV, adenovirus, influeza and parainfluenza. In cases where viral agents could not be diagnosed using these methods, the viral etiology of infection was defined according to the following criteria: WBC <12,000/μl with lymphocytosis, C-reactive protein (CRP) <5 mg/dl and procalcitonin (PCT) <0.5 ng/dl. In cases where bacterial infection was suspected based on a physical examination conducted in the pediatric emergency department, a chest X-ray (CXR) examination was additionally performed. Only cases in which the CXR was without inflammatory changes and peripheral oedema or atelectasis were included (17,24,25). Children with confirmed RSV infection were excluded from the study.
The clinical scoring of the severity of the bronchiolitis was determined according to the criteria proposed by Papadopoulos et al (26), with modifications (Table I).
Table I.
Bronchiolitis severity scale.
| Parameter | Score |
||
|---|---|---|---|
| 1 | 2 | 3 | |
| Heart rate (beats/min) | <120 | 120–160 | >160 |
| Breath rate (breaths/min) | <40 | 40–60 | >60 |
| Wheezing | Expiratory | Inspiratory | Heard without auscultation |
| Cyanosis | Normal skin | Mild cyanosis | Moderate and serious cyanosis |
| Feedinga | Readily | Reluctantly | Oral feeding impossible |
| SaO2 (%) | >98 | 94–98 | <94 |
SaO2 with the use of pulsoximeter; anatural or artificial feeding. Score: ≤6, mild; 7–12, moderate; 13–18, serious bronchiolitis.
Statistical analysis was carried out with Statistica PL 6.0 using a Student's t-test and χ2 test with Yates' correction when appropriate. The level of significance was set at p<0.05.
The study was approved by the local ethics committee of the Collegium Medicum in Bydgoszcz, Nicolaus Copernicus University.
Results
In the children with bronchiolitis, the mean concentrations of IL-15, IL-18 and IFN-γ were 9.39±11.55, 884.03±645.44 and 17.92±27.14 pg/ml, respectively, and were significantly higher than in the control group [2.34±0.61 pg/ml (p=0.0295), 248.69±98.73 pg/ml (p=0.0009) and 2.75±1.72 pg/ml (p=0.0035), respectively] (Table II).
Table II.
Concentration of cytokines in children with bronchiolitis.
| IL-15 (pg/ml) | IL-18 (pg/ml) | IFN-γ (pg/ml) | ||||
|---|---|---|---|---|---|---|
|
|
|
|
||||
| Control | ZO | Control | ZO | Control | ZO | |
| No. | 14 | 23 | 15 | 23 | 30 | 23 |
| Mean | 2.34 | 9.39 | 248.69 | 884.03 | 2.75 | 17.92 |
| SD | 0.61 | 11.55 | 98.73 | 645.44 | 1.72 | 27.14 |
| Median | 2.45 | 6.50 | 274.80 | 699.60 | 2.13 | 9.46 |
| Min | 1.12 | 2.72 | 58.00 | 143.32 | 0.30 | 2.27 |
| Max | 3.18 | 59.09 | 358.50 | 2,621.20 | 6.12 | 103.83 |
| P-value | 0.0295 | 0.0009 | 0.0035 | |||
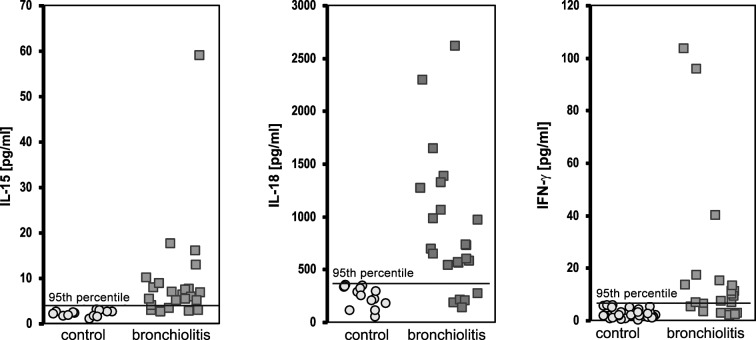
At the time of presentation, the concentrations of cytokines in the children with viral bronchiolitis above the 95th percentile of the control values were elevated for IL-15 in 19/23 (82.6%) cases, for IL-18 in 18/23 (78.3%) cases and for IFN-γ in 17/23 (73.9%) cases, and were significantly higher than in the control group [1/35 (2.8%, p<0.0001) cases, 2/35 (5.7%, p<0.0001) cases and 2/33(6.1%, p<0.0001) cases, respectively] (Fig. 1).
Figure 1.
Concentrations of IL-15, IL-18 and IFN-γ in children with bronchiolitis.
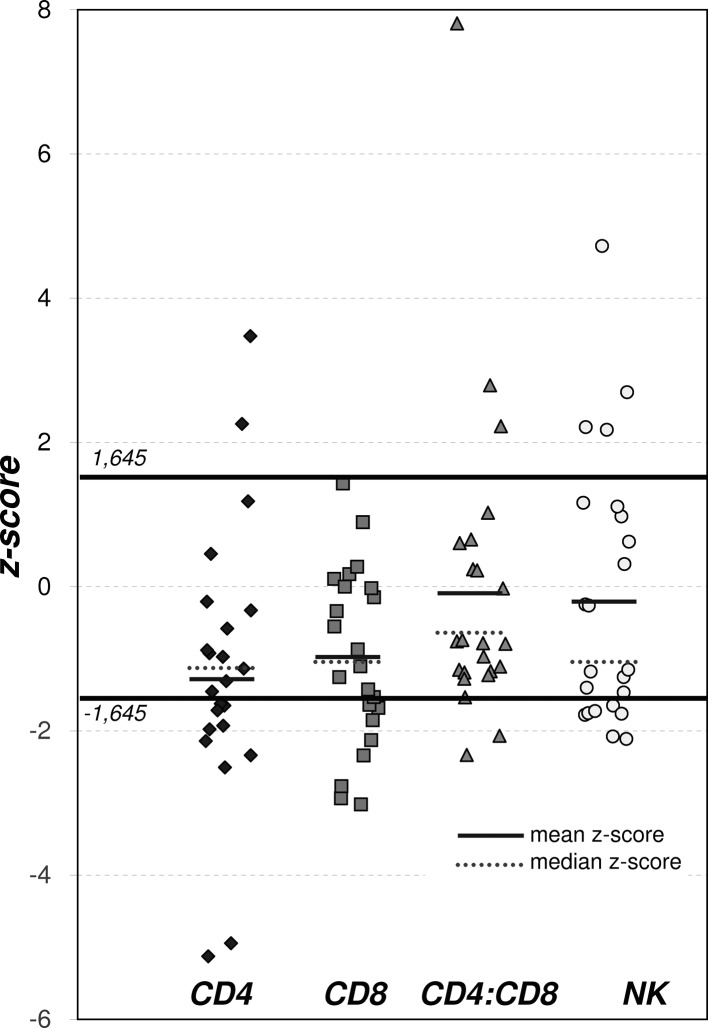
The mean value of z-scores for CD4+ and CD8+ cells in the bronchiolitis group was significantly lower than that of the general Polish population (CD4+, p<0.001 and CD8+, p<0.01) (Table III, Fig. 2). However, neither the mean z-score of the CD4+/CD8+ ratio nor the NK cell count differed significantly between the patients and the controls. At presentation, 12/23 (52.17%) and 7/23 (30.4%) of patients had CD4+, CD8+ SDS values below −1.645 SDS (<5th percentile) (Table III, Fig. 2).
Table III.
z-scores for NK cell activity and CD4+, CD8+ and CD4+/CD8+ ratio.
| No. | z-score |
z-score distribution |
|||||
|---|---|---|---|---|---|---|---|
| Mean ± SD | Median (min-max) | <−1.645 | −1.645 to 1.645 | >1.645 | P-valuea | ||
| CD4 | 23 | −1.15±1.90 | −1.31 (−5.13 to 3.47) | 12 (52.17%) | 10 (43.48%) | 1 (4.35%) | 0.001 |
| CD8 | 23 | −0.90±1.23 | −1.10 (−3.02 to 1.43) | 7 (30.43%) | 15 (65.22%) | 1 (4.35%) | 0.01 |
| CD4:CD8 | 23 | −0.06±2.13 | −0.78 (−2.33 to 7.81) | 2 (8.70%) | 18 (78.26%) | 3 (13.04%) | NS |
| NK cells | 23 | −0.17±1.85 | −1.15 (−2.11 to 4.72) | 6 (26.09%) | 13 (56.52%) | 4 (17.39%) | NS |
Distribution of z-scores in comparison to the normal range for the Polish population [Zeman et al, 1996 (23)].
Figure 2.
z-scores for NK cell activity, CD4+, CD8+ and CD4+/CD8+ ratio.
No relationships were found between cytokine concentrations and parameters such as age at the time of hospitalization, duration of hospitalization, respiratory rate, saturation, bronchiolitis clinical scoring, CRP and PCT. A positive correlation was found between IL-15 and time elapsed between the first symptoms and hospitalization (r=0.4893, p=0.024). IL-15 was also significantly related to IFN-γ (r=0.7776, p=0.0001). There were no significant correlations between CD4+, CD8+, CD4+/CD8+ and age, time elapsed until hospitalization, duration of hospitalization, saturation, clinical scoring or CRP, but a significant correlation was found between the CD4+ count and PCT concentrations (r=0.9234, p=0.0001). No correlations were observed between NK cell count and IL-15, IL-18 and IFN-γ concentrations. A significant negative correlation was found between breath rate and NK cell activity (r=−0.4880, p=0.025).
Discussion
To our knowledge, this is the first study to demonstrate that the cytokines IL-15 and IL-18 along with IFN-γ are involved in the pathogenesis of viral bronchiolitis in children. The mean concentrations of these cytokines, determined within the first 24 h following hospital admission, were significantly higher in children with bronchiolitis than in the control group. The incidence of increased concentrations of cytokines, over the 95th percentile of the normal values, was also observed more frequently in the bronchiolitis group. The activity of CD4+ and CD8+ cells was lower than in the controls, whereas the activity of NK cells did not differ significantly compared to the controls.
Our results are corroborated by the indirect findings of Okamura et al (21) and Mueller et al (22), who found a significant increase in IL-15, IL-18 and NK cell activity during viral infection. Others have reported a predominant increase in IFN-γ concentrations in children with bronchiolitis (5). This increase in IFN-γ concentrations appears to be secondary to the increase in IL-18 concentrations, as this interleukin is one of the most powerful agents stimulating the production and release of IFN-γ (20). The relationships between infection and IL-15 or NK cell activity are similar. IL-15 is a well-known activator of NK cells and T lymphocytes (27). Therefore, in most viral infections, a primary elevation in IL-15 and IL-18 and a secondary increase in NK cell activity and IFN-γ concentration are observed (28,29). This increase is noted in the presence of activation, predominantly of the TH1 type, of the cytokine cascade (30,31). Contrary to these findings, no elevation of CD4+, CD8+ or NK cell activity was noted in the present study, while an unexpected and significant decrease in the subpopulation of T lymphocytes was observed. The activity of NK cells did not differ significantly from that of the controls. However, in the first phase of viral infection, a decrease was observed in NK cell, CD4+ and CD8+ activity (14,32). It is possible that our data reflect these phenomena. Diminished values of NK cell, CD4+ and CD8+ activity may be risk factors for more serious infection, indicating the necessity of hospitalization. An increase in IL-15 concentrations in relation to the time elapsed before hospitalization was observed in our study, suggesting that IL-15 may also be a sensitive marker of the severity of the disease (33–35). Therefore, in future studies we will investigate the concentrations of IL-15, IL-18, IFN-γ, T lymphocytes and NK cells throughout the period of hospitalization for bronchiolitis.
Interpretations of our results have some limitations. We used serological methods to define the etiology of bronchiolitis infection and excluded the RSV pathogen. These methods are of limited value, but at the time of the study molecular methods were not available. In that respect, we could not entirely exclude the RSV etiology, and it is likely that in some of the children more than one virus was identified (36). Moreover, our data were gathered using blood samples; in future, concentrations of the investigated cytokines should be determined from respiratory secretions as well. All of the aforementioned limitations may have impacted the obtained results.
In summary, our results suggest that IL-15, IL-18 and IFN-γ participate in the generation of inflammatory response during bronchiolitis in children. During the initial phase of disease, a significant increase in IL-15, IL-18 and IFN-γ was noted, with a decrease in the activity of CD4+ and CD8+ and NK cells.
References
- 1.Kotagal UR, Robbins JM, Kini HD, Kirschbaum MS. Impact of bronchiolitis guideline – a multisided demonstration project. Chest. 2002;121:1789–1797. doi: 10.1378/chest.121.6.1789. [DOI] [PubMed] [Google Scholar]
- 2.Everard ML. Bronchiolitis. Origins and optimal management. Drugs. 1995;49:885–896. doi: 10.2165/00003495-199549060-00003. [DOI] [PubMed] [Google Scholar]
- 3.Chehab MS, Bafagih HA, Al-Dabbagh MM. Overview of bronchiolitis. Saudi Med J. 2005;26:177–190. [PubMed] [Google Scholar]
- 4.Smyth RI, Openshaw PJM. Bronchiolitis. Lancet. 2006;368:312–322. doi: 10.1016/S0140-6736(06)69077-6. [DOI] [PubMed] [Google Scholar]
- 5.Van Schaik SM, Tristram DA, Nagpal IS, Hintz KM, Welliver RC, II, Welliver RC. Increased production of IFN-gamma and cysteinyl leukotrienes in virus-induced wheezing. J Allergy Clin Immunol. 1999;103:630–636. doi: 10.1016/s0091-6749(99)70235-6. [DOI] [PubMed] [Google Scholar]
- 6.Bont L, Heijnen CJ, Kavelaars A, et al. Peripheral blood cytokine responses and disease severity in respiratory syncytial virus bronchiolitis. Eur Respir J. 1999;14:144–149. doi: 10.1034/j.1399-3003.1999.14a24.x. [DOI] [PubMed] [Google Scholar]
- 7.Aoyagi M, Shimojo N, Sekine K, Nishimuta T, Kohno Y. Respiratory syncytial virus infection suppresses IFN-gamma production of gamma delta T cells. Clin Exp Immunol. 2003;131:312–317. doi: 10.1046/j.1365-2249.2003.02062.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Joshi P, Shaw A, Kakakios A, Isaacs D. Interferon-gamma levels in nasopharyngeal secretions of infants with respiratory syncytial virus and other respiratory viral infections. Clin Exp Immunol. 2003;131:143–147. doi: 10.1046/j.1365-2249.2003.02039.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Semple MG, Cowell A, Dove W, Greensill J, McNamara PS, Halfhide C, Shears P, Smyth RL, Hart CA. Dual infection of infants by human metapneumovirus and human respiratory syncytial virus is strongly associated with severe bronchiolitis. J Infect Dis. 2005;191:382–386. doi: 10.1086/426457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang SZ, Harrod KS. The immunobiology of respiratory syncytial virus infection. Clin Applied Immunol Rev. 2006;6:37–52. [Google Scholar]
- 11.Castro M, Schweiger T, Yin-Declue H, et al. Cytokine response after severe respiratory syncytial virus bronchiolitis in early life. J Allergy Clin Immunol. 2008;122:726–733. doi: 10.1016/j.jaci.2008.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee YM, Miyahara N, Takeda K, Prpich J, Oh A, Balhorn A, Joetham A, Gelfand EW, Dakhama A. IFN-gamma production during initial infection determines the outcome of reinfection with respiratory syncytial virus. Am J Respir Crit Care Med. 2008;177:208–218. doi: 10.1164/rccm.200612-1890OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Konno S, Grindle KA, Lee WM, et al. Interferon-gamma enhances rhinovirus-induced RANTES secretion by airway epithelial cells. Am J Respir Cell Mol Biol. 2002;26:594–601. doi: 10.1165/ajrcmb.26.5.4438. [DOI] [PubMed] [Google Scholar]
- 14.Brooks DG, Teyton L, Oldstone MBA, McGawern DB. Intrinsic functional dysregulation of CD4 T cells occurs rapidly following persistent viral infection. J Virol. 2005;79:10514–10527. doi: 10.1128/JVI.79.16.10514-10527.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park YB, Kim DS, Lee WK, Suh CH, Lee SK. Elevated serum interleukin-15 levels in systemic lupus erythematosus. Yonsei Med J. 1999;40:343–348. doi: 10.3349/ymj.1999.40.4.343. [DOI] [PubMed] [Google Scholar]
- 16.Cassatella MA, McDonald PP. Interleukin-15 and its impact on neutrophil function. Curr Opin Hematol. 2000;7:174–177. doi: 10.1097/00062752-200005000-00008. [DOI] [PubMed] [Google Scholar]
- 17.Gendrel D, Josette R, Coste J, et al. Comparison of procalcitonin with C-reactive protein, interleukin 6 and interferon-alpha for differentiation of bacterial vs. viral infections. Pediatr Infect Disease J. 1999;18:875–881. doi: 10.1097/00006454-199910000-00008. [DOI] [PubMed] [Google Scholar]
- 18.Wang SZ, Bao YX, Rosenberger CL, Tesfaigzi Y, Stark JM, Harrod KS. IL-12p40 and IL-18 modulate inflammatory and immune responses to respiratory syncytial virus infection. J Immunol. 2004;173:4040–4049. doi: 10.4049/jimmunol.173.6.4040. [DOI] [PubMed] [Google Scholar]
- 19.Vankayalapati R, Wisel B, Weis SE, Samten B, Girard WM, Barnes PF. Production of interleukin-18 in human tuberculosis. J Infect Dis. 2000;182:234–239. doi: 10.1086/315656. [DOI] [PubMed] [Google Scholar]
- 20.Ngoumou G, Schaefer D, Mattes J, Kopp MV. Interleukin-18 enhances the production of interferon-gamma (IFN-γ) by allergen-specific and unspecific stimulated cord blood mononuclear cells. Cytokine. 2004;25:172–178. doi: 10.1016/j.cyto.2003.11.013. [DOI] [PubMed] [Google Scholar]
- 21.Okamura H, Tsutsi H, Komatsu T, Yutsudo M, Hakura A, Tanimoto T. Cloning of a new cytokine that induces IFN-gamma production by T cells. Nature. 1995;378:88–91. doi: 10.1038/378088a0. [DOI] [PubMed] [Google Scholar]
- 22.Mueller YM, Petrovas C, Bojczuk PM, et al. Interleukin-15 increases effector memory CD8+ T cells and NK cells in simian immunodeficiency virus-infected macaques. J Virol. 2005;79:4877–4885. doi: 10.1128/JVI.79.8.4877-4885.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zeman K, Fornalczyk-Wachowska E, Pokoca L, et al. Skład odsetkowy podstawowych subpopulacji limfocytów oraz komórek NK we krwi obwodowej populacji polskiej. Central Europ J Immunol. 1996;21:107–113. [Google Scholar]
- 24.Putto A, Ruuskanen O, Meurman O, et al. C reactive protein in the evaluation of febrile illness. Arch Dis Child. 1986;61:24–29. doi: 10.1136/adc.61.1.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Prat C, Dominiquez J, Rodrigo C, et al. Procalcitonin, C-reactive protein and leucocyte count in children with lower respiratory tract infection. Pediatr Infect Dis J. 2003;22:963–968. doi: 10.1097/01.inf.0000095197.72976.4f. [DOI] [PubMed] [Google Scholar]
- 26.Papadopoulos NG, Moustaki M, Tsolia M, et al. Association of rhinovirus infection with increased disease severity in acute bronchiolitis. Am J Resp Crit Care Med. 2002;165:1285–1289. doi: 10.1164/rccm.200112-118BC. [DOI] [PubMed] [Google Scholar]
- 27.Fehniger TA, Caligiuri MA. Interleukin 15: biology and relevance to human disease. Blood. 2001;97:14–32. doi: 10.1182/blood.v97.1.14. [DOI] [PubMed] [Google Scholar]
- 28.Van Benten IJ, van Drunen CM, Koopman LP, et al. RSV-induced bronchiolitis but not upper respiratory tract infection is accompanied by an increased nasal IL-18 response. J Med Virol. 2003;71:290–297. doi: 10.1002/jmv.10482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Domachowske JB, Rosenberg HF. Advances in the treatment and prevention of severe viral bronchiolitis. Pediatr Ann. 2005;34:35–41. doi: 10.3928/0090-4481-20050101-10. [DOI] [PubMed] [Google Scholar]
- 30.Stark JM, Busse WW. Respiratory virus infections and airway hyper-reactivity in children. Pediatr Allergy Immunol. 1991;2:95–110. [Google Scholar]
- 31.Semple MG, Dankert HM, Ebrahimi B, et al. Severe respiratory syncytial virus bronchiolitis in infants is associated with reduced airway interferon gamma and substance P. PLoS ONE. 2007;2:e1038. doi: 10.1371/journal.pone.0001038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Biet F, Locht C, Kremer L. Immunoregulatory functions of interleukin 18 and its role in defense against bacterial pathogens. J Mol Med. 2002;80:147–162. doi: 10.1007/s00109-001-0307-1. [DOI] [PubMed] [Google Scholar]
- 33.Pitrez PM, Machado DC, Jones MH, Andrade F, Camozzato C, Stein RT. Th-1 and Th-2 cytokine production in infants with virus-associated wheezing. Braz J Med Biol Res. 2005;38:51–54. doi: 10.1590/s0100-879x2005000100008. [DOI] [PubMed] [Google Scholar]
- 34.Brooks GD, Buchta KA, Swenson CA, Gern JE, Busse WW. Rhinovirus-induced interferon-gamma and airway responsiveness in asthma. Am J Respir Crit Care Med. 2003;168:1091–1094. doi: 10.1164/rccm.200306-737OC. [DOI] [PubMed] [Google Scholar]
- 35.Chung JY, Han TH, Kim JS, Kim SW, Park CG, Hwang ES. Th1 and Th2 cytokine levels in nasopharyngeal aspirates from children with human bocavirus bronchiolitis. J Clin Virol. 2008;43:223–225. doi: 10.1016/j.jcv.2008.06.008. [DOI] [PubMed] [Google Scholar]
- 36.Henrickson KJ, Hoover S, Kehl KS, Hua W. National disease burden of respiratory viruses detected in children by polymerase chain reaction. Pediatr Infect Dis J. 2004;23(Suppl 1):11–18. doi: 10.1097/01.inf.0000108188.37237.48. [DOI] [PubMed] [Google Scholar]