Abstract
Objective
Beta-thalassemia major is an autosomal recessive disease causing severe and hemolytic anemia, which begins about 2-6 months after birth. Iron overload, which arises from recurrent transfusion and ineffective erythropoiesis, can enhance oxidative stress in thalassemic patients. The aim of this study was to evaluate the serum total antioxidant capacity of patients with ß-Thalassemia major.
Methods
Sixty six Iranian patients with β-thalassemia major and 66 age-gender matched controls were evaluated for serum total antioxidant status (TAS), uric acid (UA), bilirubin and albumin. In addition, serum ferritin and transaminases were recorded in these subjects.
Findings
Significant increases of TAS, UA, and bilirubin were observed in the patient group, compared with the control group (P<0.01). Mean TAS and bilirubin in male patients was higher than in females (P=0.005 and P=0.008, respectively). There was also direct correlation between TAS and albumin (P<0.001), bilirubin (P<0.001) and UA (P=0.002).
Conclusion
Endogenous antioxidants such as ferritin, UA and bilirubin can result in increased level of TAS in the patients with Beta-thalassemia major. Compensatory excess of TAS to oxidative stress could also be the reason for difference between our findings and previous studies.
Keywords: β-Thalassemia Major, Oxidative Stress, Antioxidants, Ferritin, Uric Acid
Introduction
Beta-thalassemia major is an autosomal recessive disease that leads to a severe hemolytic anemia in early infancy[1, 2]. Depletion or impaired synthesis of β-globin chain can result in an imbalanced production of globin chains towards higher production of α-chain, which converts hemoglobin from a normal oxygen transporting function into toxic inclusion bodies, causing peripheral erythrocyte hemolysis [1, 3, 4]. Glutathione decrease is another reason for destabilization of hemoglobin [5, 6].
Several clinical phenotypes with varieties in severity in the patients with β-thalassemia can be seen as a consequence of hemolytic condition and ineffective erythropoiesis. Anemia, growth retardation, pathological fracture of long bones and vertebrae, hepatosplenomegaly, gall stones, leg ulcers, and congestive heart failure are the main clinical characteristics of these patients.
The patients with β-thalassemia major usually suffer from iron overload as a consequence of recurrent transfusion and ineffective erythropoiesis. Iron has a catalytic role to produce powerful reactive oxidant species (ROS) and free radicals, which lead to oxidative damage [1, 2, 7–11].
Antioxidants play an essential role in protection of the cells from oxidative damage. They include several agents such as enzymes (glutathione peroxidase, superoxide dismutase, catalase), large molecules (ferritin, albumin), and small molecules (uric acid, glutathione, bilirubin, ascorbic acid, α tocopherol, and vitamin E). Their defense mechanism in biological system involves chain breaking (SOD) and preventive (Vitamin E) mechanisms [12–15]. Antioxidant defense can be evaluated by measurement of either individual antioxidants levels in cells and plasma or total antioxidant capacity. The latter can be estimated by measuring total reducing activity of body fluids such as serum and plasma [12–17].
The present study was performed to assess the serum total antioxidant in the selected patients with major β-thalassemia. In addition, the results of endogenous antioxidant agents such as serum total bilirubin, albumin and uric acid in the patients were compared to the results in control group.
Subjects and Methods
Sixty six patients with β-thalassemia major (mean age 14.7±7 years), who were consecutively referred to the Children's Medical Center in Tehran, from June 2007 to June 2008, were investigated. The diagnosis of β-thalassemia major was made considering the results of hemoglobin electrophoresis and clinical features of the patients.
Blood transfusions were conducted regularly for all patients with 10-15 ml/kg packed cells, every 21-25 days for maintaining hemoglobin at 10-11g/dl; all the patients were under iron chelation therapy with subcutaneous deferoxamine (DFO) 20-60 mg/kg/d. Moreover 66 age- and sex-matched healthy subjects (mean age 15.2±7 years), without any hematology disorder or systemic disease, were enrolled in the study. The study was approved by the Ethics Committee of Tehran University of Medical Sciences. Informed consents were taken from all subjects or their parents prior to participation in the study. Exclusion criteria included use of vitamins and presence of acute illness.
Two milliliters blood was obtained after 8-12 h fasting from each subject; in thalassemic patients, sampling was done just before the transfusion. The blood samples were centrifuged at 3000 rpm for 10 minutes and sera were kept in -70°C until analysis. Serum total antioxidant, albumin, total bilirubin and uric acid (UA) were measured in serum samples. The levels of ferritin and transaminases had been previously measured in thalassemic patients, which were extracted from their medical records.
Albumin and UA were measured spectrophotometrically (using Bromocresol green and Uricase methods, respectively). Bilirubin level was measured using dichloroaniline method. All spectrophotmetric assays are performed on Hitachi 717 autoanalyser (Hitachi Ltd, Tokyo, Japan).
To determine the serum total antioxidant, Randox kit [Total Antioxidants Status (TAS), RANDOX Laboratories Ltd, UK] was utilized. The method is based on the suppression of the blue green color of 2,2’-azinobis-3-ethylbenzo-thiazoline-6-sulfonic acid (ABTS) radical cation by antioxidants. Calculated total antioxidant status (cTAS), was determined according to the following formula:
[18].
Descriptive results were reported as mean (standard deviation). Independent sample t-tests were used to compare the results between groups. The associations between variables were assessed by Pearson's correlation coefficient. A P-value of <0.05 was considered as statistically significant.
Findings
Demographic and biochemical characteristics of the studied subjects are presented in Table 1. TAS, UA and bilirubin levels in the patient group were significantly higher than in the control group (P<0.001) (Table 1). TAS and bilirubin levels in the male patients were significantly higher than in the females (P=0.005 and P=0.008, respectively) (Table 2). There were no significant differences in other measured parameters between males and females (Table 2). cTAS in the patients was also significantly higher than in controls.
Table 1.
Demographic and biochemical characteristic of patients and controls
| Parameters* | Patients (n=66) | Control (n=66) | P-value |
|---|---|---|---|
| Gender (Male/female) | 34/32 | 34/32 | - |
| Age (year) | 14.73 (6.95) | 15.15 (7.38) | 0.735 |
| TAS (mmol/l) | 1.50 (0.18) | 1.36 (0.20) | <0.001 |
| Uric acid (mg/dl) | 4.11 (1.28) | 3.27 (1.05) | <0.001 |
| Albumin (g/dl) | 4.58 (0.43) | 4.65 (0.41) | 0.328 |
| Bilirubin (mg/dl) | 1.25 (0.83) | 0.67 (0.37) | <0.001 |
| Ferritin (ng/ml) | 2291 (1668) | - | - |
| AST (u/l) | 31.72 (14.88) | - | - |
| ALT (u/l) | 30.37 (21.14) | - | - |
TAS: Total antioxidant status, AST: Aspartate aminotransferase, ALT: Alanine aminotransferase
The data are given based on mean values (Standard Deviation)
Table 2.
Gender differences in level of uric acid, albumin, total bilirubin, antioxidant and ferritin in the patient and control groups
| Variables | Reference range | Patient group | P-value | Control group | P-value | ||
|---|---|---|---|---|---|---|---|
| Male (n=34) | Female (n=32) | Male (n=34) | Female (n=32) | ||||
| Uric acid (mg/dl) | 3–7 | 4.35 (1.33) | 3.86 (1.19) | 0.1 | 3.40 (0.92) | 3.13 (1.18) | 0.3 |
| Albumin (g/dl) | 3.5–5.5 | 4.58 (0.45) | 4.58 (0.41) | 0.9 | 4.69 (0.41) | 4.61 (0.42) | 0.5 |
| Bilirubin (mg/dl) | 0–1 | 1.51 (0.98) | 0.98 (0.54) | 0.008** | 0.64 (0.42) | 0.70 (0.32) | 0.5 |
| TAS (mmol/l) | 1.3–1.77 | 1.56 (0.18) | 1.44 (0.17) | 0.005** | 1.40 (0.22) | 1.31 (0.17) | 0.09 |
| Ferritin (ng/ml) | 10–400 | 2121 (1623) | 2472 (1763) | 0.4 | - | - | - |
| AST (u/l) | <40 | 29.50 (13.85) | 27.58 (17.63) | 0. 2 | - | - | - |
| ALT (u/l) | <40 | 34.09 (15.78) | 33.34 (24.26) | 0. 2 | - | - | - |
TAS: Total antioxidant status, AST: Aspartate aminotransferase, ALT: Alanine aminotransferase
The data are given based on mean values (Standard Deviation)
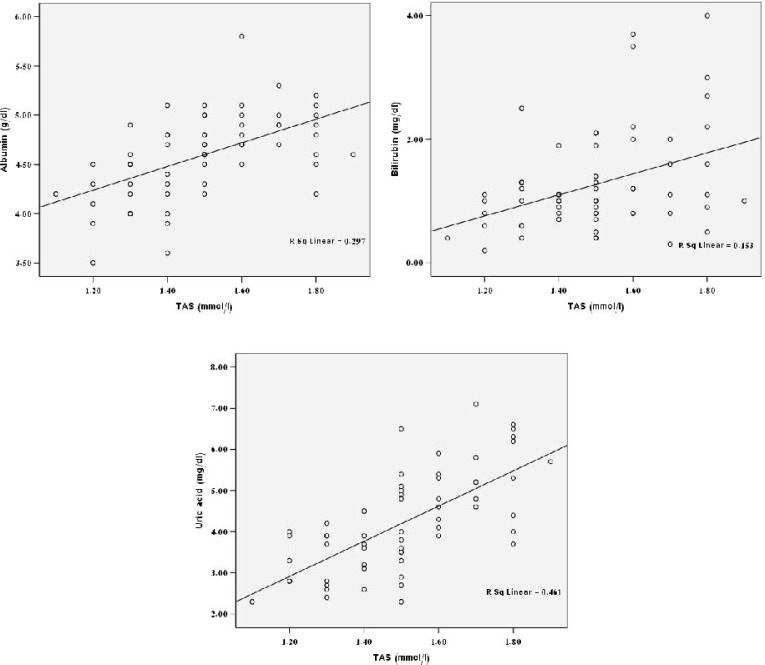
There were direct correlation between TAS and albumin (P<0.001), bilirubin (P<0.001) and UA (P=0.002) (Fig. 1). However, there was no significant correlation between these parameters in the control subjects. The observed reverse correlation between TAS and ferritin in the patients was not statistically significant (r square=0.011, P=0.4). The positive trend between TAS and the times of the transfusion was not significant (r square=0.018, P=0.3) as well. There was no correlation between age and TAS level in the studied populations (r square<0.001, P=0.9).
Fig. 1.
Scatter plots showing significant correlation between TAS and albumin (P<0.001), bilirubin (P<0.001) and UA (P=0.002) in β-thalassemic patients
Discussion
Patients with β-thalassemia are mainly exposed to oxidative stress due to iron overload. Therefore evaluation and maintenance of antioxidant defense can be useful in protecting β-thalassemia patients from more serious complications of the disease [2, 3, 11, 12, 14, 19].
In the present study, the antioxidant defense was evaluated by measuring total antioxidant status (TAS) in serum. The measurement of different antioxidant molecules separately is labor-intensive, time-consuming and costly. Moreover, some investigators suggest that assessment of total antioxidant capacity of plasma may be more useful than measuring antioxidants individually since their synergistic interaction could be determined [13, 15, 17].
Our results revealed significant increased levels of TAS, UA and Bilirubin in thalassemic patients compared to healthy ones. Several studies have been performed to assess antioxidant defense of thalassemic patients. In the majority of these studies, a decreased serum vitamin E was reported[2, 3, 12, 14, 19]; despite contradictory results on levels of erythrocyte superoxide dismotase (SOD) and glutathione peroxidase[2, 3, 5, 7, 12, 14].
There are limited studies about assessment of serum total antioxidant capacity in thalassemic patients. Ghon et al showed the depletion of antioxidants in thalassemic patients[2]. In a study in Italy[20], a significant decrease of Total Antioxidant Capacity (TAC) in thalassemic patients was reported compared to controls. A significantly lower concentration of TAC in thalassemic patient with DFO chelation therapy compared to patient without DFO was also reported in the same study[20]. However, Cakmak et al [21] reported no significant differences in TAC between thalassemic and control groups, in spite of increased level of Oxidant Status (OS) and Oxidative Stress Index (OSI).
There are several hypotheses to explain the enhanced TAS in our patients. The compensation antioxidant response arising from excessive oxidative stress and transfusion dependent elevation in the proportion of younger RBC could be considered as two probable mechanisms.
Another mechanism to justify the elevation of assessed antioxidant capacity may be relevant to chelation therapy by deferoxamine[14, 22, 23].
Increased bilirubin and UA in thalassemic patients, as the antioxidant implicate in TAS, might be a reason for observed TAS elevation[15–18]. Previous studies have shown the effect of UA on total antioxidant capacity in patients with renal dysfunction, which is in agreement with this hypothesis. Therefore, in order to attenuate this effect, we used cTAS[18], which is calculated in accordance with the amount of albumin, UA and bilirubin. However, the gap between measured and calculated TAS, which implies the other unmeasured antioxidant agents, were significantly higher in the patient group compared with controls, which could suggest that elevation of TAS cannot be solely explained as a result of increased UA and bilirubin.
Finally, significant urate level difference between the patients and the controls, as a pitfall, can influence the measured antioxidant by inhibition of ABTS production[24].
The significant increased bilirubin level observed in the patients with β-thalassemia in contrast with the control group could be due to hemolysis that occurs in thalassemic patients. In addition, iron overload could potentially induce hepatic toxicity, and consequently increased bilirubin level, that arises from decrease in activity of cythochrome c oxidase disrupting the mitochondrial respiration. Hepatic damage induced by iron overload could lead to decrease in vitamin E serum level in the absence of transaminase augmentation[3, 8, 25, 26].
In our study, markedly greater bilirubin level was found in males compared to females in the patient group. This gender difference was indicated in the previous studies in normal population with age range of more than 10 years[26, 27], which was justified by the effect of hormonal changes due to puberty on metabolism of bilirubin. Although considering the mean age of our patients (14.7±6.9 years) this difference could be explained by these hormonal alterations, lack of similar results in the control group is puzzling.
The significant observed antioxidant rise in males in comparison with females was previously reported on mice[28]. Another study also revealed that oxidative stress in males is slightly greater than in females, which may be explained by the compensatory antioxidants rise in males in our study. On the other hand, this finding maybe due to the significant rise of bilirubin in male in comparison with female.
However, confirming the association between oxidative stress and compensatory increase in TAS requires their concurrent measurement.
This study had some limitations, for example, liver enzymes and ferritin levels have not been checked in control group. Indeed, although the control group was apparently normal, the possibility of some degrees of metabolic disturbance or dormant disease cannot be excluded.
Conclusion
This study showed an increased level of total antioxidant in the patients with beta-thalassemia major in comparison with the controls. Whereas increased endogenous antioxidants such as ferritin, UA and bilirubin in the patients can also lead to increased level of TAS. Regular monitoring of antioxidant defense and administration of proper antioxidant may protect thalassemic patients from consequences of oxidative damages. Further studies in different regions with more subjects are needed to confirm the results of this study.
Acknowledgment
This research has been approved and supported by a grant of the School of Medicine, Tehran University of Medical Sciences & Health Services. The study was approved by the Ethics Committee of Tehran University of Medical Sciences. We thank all staff of Clinical Laboratory of Children's Medical Center, especially Ms. Heshmat Irani, Ms. Ameneh Mohseni, Ms. Fahimeh Jafari and Mr. Mohsen Siavash for specimen collection and processing.
Conflict of Interest
None
References
- 1.Benz EJ. Disorder of hemoglobin. In: Fauci AS, Braunwald E, Kasper DL, editors. Harrison's Principle of Internal Medicine. USA: McGraw-Hill; 2008. pp. 640–1. [Google Scholar]
- 2.Ghone RA, Kumbar KM, Suryakar AN, et al. Oxidative stress and disturbance in antioxidant balance in beta thalassemia major. Ind J Clini Biochem. 2008;23:337–40. doi: 10.1007/s12291-008-0074-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kassab-Chekir A, Laradi S, Ferchichi S, et al. Oxidant, antioxidant status and metabolic data in patients with beta-thalassemia. Clin Chim Acta. 2003;338:79–86. doi: 10.1016/j.cccn.2003.07.010. [DOI] [PubMed] [Google Scholar]
- 4.Joshi W, Leb L, Piotrowski J, et al. Increased sensitivity of isolated alpha subunits of normal human hemoglobin to oxidative damage and crosslinkage with spectrin. J Lab Clin Med. 1983;102:46–52. [PubMed] [Google Scholar]
- 5.Naithani R, Chandra J, Bhattacharjee J, et al. Peroxidative stress and antioxidant enzymes in children with b-thalassemia major. Pediatr Blood Cancer. 2006;46:780–5. doi: 10.1002/pbc.20669. [DOI] [PubMed] [Google Scholar]
- 6.Scott MD, Eaton JW. Thalassemic erythrocytes: Cellular suicide arising from iron and glutathione-dependent oxidation reactions? Br J Haematol. 1995;91:811–9. doi: 10.1111/j.1365-2141.1995.tb05394.x. [DOI] [PubMed] [Google Scholar]
- 7.Das N, Chowdhury TD, Chattopadhyay A, Datta AG. Attenuation of oxidative stress-induced changes in thalassemic erythrocytes by vitamin E. Pol J Pharmacol. 2004;56:85–96. [PubMed] [Google Scholar]
- 8.Livrea MA, Tesoriere L, Pintaudi AM, et al. Oxidative stress and Antioxidant status in beta thalassemia major: Iron overload and depletion of lipid-soluble antioxidants. Blood. 1996;88:3608–14. [PubMed] [Google Scholar]
- 9.Mulligan M, Althaus B, Linder MC. Non-ferritin, non-heme iron pools in rat tissues. Int J Biochem. 1986;18:791. doi: 10.1016/0020-711x(86)90055-8. [DOI] [PubMed] [Google Scholar]
- 10.Deighton N, Hider RC. Intracellular low molecular weight iron. Biochem Soc Trans. 1989;17:490. [Google Scholar]
- 11.Swaminathan S, Fonseca VA, Alam MG, Shah SV. The role of iron in diabetes and its complications. Diabetes Care. 2007;30(7):1926–33. doi: 10.2337/dc06-2625. [DOI] [PubMed] [Google Scholar]
- 12.Dhawan V, Ratan Kumar K, Marwaha RK, et al. Antioxidant status in children with homozygous β-thalassemia. Ind Pediatr. 2005;42:1141–5. [PubMed] [Google Scholar]
- 13.Van Zoeren-Grobben D, Lindeman JH, Houdkamp E, et al. Markers of oxidative stress and antioxidant activity in plasma and erythrocytes in neonatal respiratory distress syndrome. Acta Paediatr. 1997;86:1356–62. doi: 10.1111/j.1651-2227.1997.tb14913.x. [DOI] [PubMed] [Google Scholar]
- 14.Şimşek F, Öztürk G, Kemahlı S, et al. Oxidant and antioxidant status in beta thalassemia major patients. Ankara Univ Tip Fak Mec. 2005;58:34–8. [Google Scholar]
- 15.Kampa M, Nistikaki A, Tsaousis V, et al. A new automated method for the determination of the Total Antioxidant Capacity (TAC) of human plasma, based on the crocin bleaching assay. BMC Clin Pathol. 2002;2(1):3. doi: 10.1186/1472-6890-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kwak HK, Yoom S. Relation of serum total antioxidant status with metabolic risk factors in Korean adults. Nutr Res Practic. 2007;1(4):335–40. doi: 10.4162/nrp.2007.1.4.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Erel O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem. 2004;37:277–85. doi: 10.1016/j.clinbiochem.2003.11.015. [DOI] [PubMed] [Google Scholar]
- 18.Bonnefont-Rousselot D, Lehmann E, Jaudon MC, et al. Blood oxidative stress and lipoprotein oxidizability in haemodialysis patients: effect of the use of a vitamin E-coated dialysis membrane. Nephrol Dial Transplant. 2000;15:2020–8. doi: 10.1093/ndt/15.12.2020. [DOI] [PubMed] [Google Scholar]
- 19.Laksmitawati DR, Handayani S, Udyaningsih-Freisleben SK, et al. Iron status and oxidative stress in beta-thalassemia patients in Jakarta. Biofactors. 2003;19:53–62. doi: 10.1002/biof.5520190107. [DOI] [PubMed] [Google Scholar]
- 20.Hamed EA, ElMelegy NT. Renal functions in pediatric patients with beta-thalassemia major: relation to chelation therapy: original prospective study. Italian J Pediatr. 2010;36:39. doi: 10.1186/1824-7288-36-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cakmak A, Soker M, Koc A, Aksoy N. Prolidase activity and oxidative status in patients with thalassemia major. J Clin Lab Analysis. 2010;24:6–11. doi: 10.1002/jcla.20361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Martínez-Romero R, Martínez-Lara E, Aguilar-Quesada R, et al. PARP-1 modulates deferoxamine-induced HIF-1α accumulation through the regulation of nitric oxide and oxidative stress. J Cell Biochem. 2008;104:2248–60. doi: 10.1002/jcb.21781. [DOI] [PubMed] [Google Scholar]
- 23.Sangchot P, Sharma S, Chetsawang B, et al. Deferoxamine attenuates Iron-induced oxidative stress and prevents mitochondrial aggregation and α-synuclein translocation in SK-N-SH cells in culture. Dev Neurosci. 2002;24:143–53. doi: 10.1159/000065700. [DOI] [PubMed] [Google Scholar]
- 24.Cao G, Prior RL. Comparison of different analytical methods for assessing total antioxidant capacity of human serum. Clin Chemistr. 1998;44(6):1309–15. [PubMed] [Google Scholar]
- 25.Zucker SD, Horn PS, Sherman KE. Serum bilirubin levels in the U.S. population: gender effect and inverse correlation with colorectal cancer. Hepatology. 2004;40:827–35. doi: 10.1002/hep.20407. [DOI] [PubMed] [Google Scholar]
- 26.Rosenthal P, Pincus M, Fink D. Sex- and age-related differences in bilirubin concentrations in serum. Clin Chem. 1984;30:1380–2. [PubMed] [Google Scholar]
- 27.White GL, Nelson JA, Pedersen DM, Ash KO. Fasting and gender influence reference intervals for serum bilirubin in healthy adults. Clin Chem. 1981;27:1140–2. [PubMed] [Google Scholar]
- 28.Sverko V, Sobocanec S, Balog T, Marotti T. Age and gender differences in antioxidant enzyme activity: potential relationship to liver carcinogenesis in male mice. Biogerontology. 2004;5:235–42. doi: 10.1023/B:BGEN.0000038024.58911.6e. [DOI] [PubMed] [Google Scholar]