Abstract
A better understanding of the aging process is necessary to ensure that the healthcare needs of an aging population are met. With the trend toward increased human life expectancies, identification of candidate genes affecting the regulation of lifespan and its relationship to environmental factors is essential. Through misexpression screening of EP mutant lines, we previously isolated several genes extending lifespan when ubiquitously overexpressed, including the two genes encoding the fatty-acid-binding protein and dodecenoyl-CoA delta-isomerase involved in fatty-acid β-oxidation, which is the main energy resource pathway in eukaryotic cells. In this study, we analyzed flies overexpressing the two main components of fatty-acid β-oxidation, and found that overexpression of fatty-acid-β-oxidation-related genes extended the Drosophila lifespan. Furthermore, we found that the ability of dietary restriction to extend lifespan was reduced by the overexpression of fatty-acid-β-oxidation-related genes. Moreover, the overexpression of fatty-acid-β-oxidation-related genes enhanced stress tolerance to oxidative and starvation stresses and activated the dFOXO signal, indicating translocation to the nucleus and transcriptional activation of the dFOXO target genes. Overall, the results of this study suggest that overexpression of fatty-acid-β-oxidation-related genes extends lifespan in a dietary-restriction-related manner, and that the mechanism of this process may be related to FOXO activation.
1. Introduction
The trend towards increased life expectancy demands a greater understanding of the aging process to ensure that healthcare needs of an aging population are met. This goal requires identification of the so-called “longevity candidate genes,” which are potential genes important to the regulation of lifespan, as well as appropriate understanding of how the effects of these genes are modulated by environmental factors such as diet. Numerous longevity candidate genes have been identified in model systems using extended longevity mutant phenotypes, offering important insights into the mechanisms of aging and lifespan determination [1–5]. Insulin/insulin-like growth factor (IGF) signaling (IIS), a major nutrient-sensing pathway, is a well-characterized age-related pathway. The loss of IIS function by mutations affecting insulin/IGF receptor, phosphatidylinositol-3 kinase (PI3K), Akt, and forkhead box (FOXO) has been found to extend the lifespan of C. elegans, Drosophila, and mammals [3, 6–11]. In addition, energy-sensing pathways such as those associated with sirtuins, target of rapamycin (TOR) and AMP-activated protein kinase (AMPK) signaling are well known to be linked to the aging process [3–5, 12]. As nutrient-sensing pathways are linked to aging, the reduction of dietary intake, namely dietary restriction, also extends the lifespan of various model systems [13–16]. Furthermore, the ecdysteroid hormone pathway is known to modulate organismal lifespan [17, 18].
While investigating longevity candidate genes, we previously conducted misexpression screening of EP lines containing 14 copies of upstream activator sequence (UAS) to which Gal4 binds, allowing conditional overexpression or knockdown of genes of flanking genomic DNA located downstream of the basal promoter dependent on its insertion orientation [19]. In that study, we preliminary selected 40 EP lines to demonstrate the lifespan extension, including the two EP lines (EPCG6783, EPCG13890) targeting fatty-acid-β-oxidation-related genes (CG6783, CG13890), but they were excluded from further investigation since they were induced in the absence of Gal4 driver [19]. It has long been suggested that lipid metabolism plays a central role in regulation of the metazoan lifespan. One of the well-known longevity-candidate genes, AMPK, was reported to regulate fatty-acid synthesis and oxidation through the phosphorylation of acetyl-CoA-carboxylase [20]. In addition, calorie restriction and IIS mutation has been reported to promote fatty-acid β-oxidation [21, 22]. However, there has been no direct evidence of lifespan extension through the modulation of fatty-acid β-oxidation to date, except for our previous study [19], in which we did not investigate the relationship with dietary restriction and its underlying mechanisms.
In the current study, we analyzed EP lines that overexpressed two main components of fatty-acid β-oxidation and found that the overexpression of fatty-acid β-oxidation related genes extended their lifespan in a dietary-restriction-related manner, increased their stress resistance, and activated the FOXO transcription factor.
2. Materials and Methods
2.1. Fly Stocks and Food Preparation
Drosophila melanogaster were cultured and reared at 25°C. Cantonized white (CS10 [23]) was used as wild-type control. The EPCG6783 (GX62810) and EPCG13890 (GX4385) lines, which carry the P-element mediated upstream activator sequence (UAS) on the 5′ untranslated region of the CG6783 or CG13890 genes, respectively, were obtained from GenExel Inc. (KAIST Bio Medical Research Center, Korea). To generate UAS-CG6783 flies, the full open reading frame of fabp-RA from RH46282 (Drosophila Genomics Resource Center, Bloomington, USA) was cloned into pUAST using EcoRI/BglII sites. Standard germline transformation into a w 1118 background was then performed for transgenic lines. Corn meal-sugar-yeast (CSY) media (5.2% cornmeal, 11% sucrose, 2.4% yeast, 0.8% agar, and 0.2% methyl-4-hydroxybenzoate (Sigma-Aldrich, St. Louis, MO, USA)) was used for larval development and routine culture. In the dietary restriction (DR) experiment, the concentration of yeast in the media fed to separate groups of flies was 2, 4, 8, 12, and 16%.
2.2. Lifespan Assays
Newly eclosed F1 generations were collected over 48 hours and the males were randomly assigned to 500 mL demography cages to achieve a final density of 100 male flies per cage. Food vials containing SY diet (10% sucrose, the indicated concentration of yeast, 0.2% methyl-4-hydroxybenzoate, and 0.8% agar) were affixed to separate cages and changed every two days, at which time the dead flies were removed and recorded. Three replicate cages were established for this experiment.
2.3. Stress-Resistance Assay
Oxidative Stress —
Ten-day-old flies (20 males per vial) were fed SY medium supplemented with 18 mM paraquat (methyl viologen dichloride hydrate, Sigma-Aldrich). The flies were transferred into fresh vials containing paraquat solution every six hours, and the dead flies were scored after each transfer. Fifteen replicates were established.
Starvation Stress —
Newly eclosed flies were kept in vials (20 males per vial) containing 1% agar and transferred into fresh vials containing agar every six hours. Dead flies were scored after each transfer. Fifteen replicates were established.
2.4. Immunostaining of the Larval Fat Bodies
Dissected third instar larvae were fixed with 4% paraformaldehyde (USB Corp., Cleveland, OH, USA) for 30 min, washed with phosphate-buffered saline/0.1% Triton X-100/2% bovine serum albumin (PBST-BSA), and then incubated overnight with primary antibodies in PBST-BSA at 4°C. Samples were then washed in PBST-BSA, incubated with Alexa488 (Molecular Probes, Eugene, OR, USA) for 1 hour at 25°C, and washed and mounted with Vectashield (Vector Labs, CA, USA). The resulting images were analyzed using a Confocal Laser Scanning Microscope (LSM510 META, Carl Zeiss Inc., Germany). Anti-dFOXO antibody (a gift from O. Puig) was diluted to 1 : 300 in 2% BSA solution. DAPI was used to counterstain the nuclei.
2.5. Real-Time qPCR
Five-day-old adults were frozen in liquid nitrogen and stored at −80°C until analysis. Total RNA from homogenized whole-body lysates was prepared with RNAiso reagent (TAKARA, Japan). Next, total RNA (5 μg) was reverse-transcribed using PrimeScript RT Reagent Kit (TAKARA) and real-time qPCR was performed on an ABI Prism 7000 Sequence Detection System (Applied Biosystems, USA) using SYBR Premix Ex-Taq II (TAKARA). Mean induction folds were calculated from the values of 3–6 independent experiments and statistically evaluated by a Student t-test.
2.6. Statistical Analysis
Data are presented as the mean ± SEM. Statistical analyses for the demographic data were carried out using standard survival models in the JMP statistical package (SAS, Cary, NC, USA).
3. Results
3.1. Overexpression of Fatty-Acid β-Oxidation Components Extended Lifespan in a Dietary-Restriction-Dependent Manner
In the previous study, we selected long-lived EP lines, which extend lifespan when crossed to da-Gal4 driver [19]. Among them, the two EP lines (GX62810, GX4385) targeting fatty-acid-β-oxidation-related genes (CG6783, CG13890) were of interest, however, they were excluded to further investigation in the previous study because they were induced in the absence of Gal4 driver [19]. CG6783 encodes the fatty-acid-binding protein (FABP), which facilitates the intracellular movement of fatty acids, thus permitting the initiation of fatty-acid oxidation [24], while CG13890 encodes the dodecenoyl-CoA delta-isomerase (DCI) localized in the inner mitochondria where it catalyzes the degradation of long-chain fatty acids during fatty-acid β-oxidation [25, 26] (Figure 1(a)). We confirmed that the EP lines overexpressed CG6783 or CG13890 when crossed with the da-Gal4 driver compared to da/+ flies via real-time qPCR (Figure 1(b)).
Figure 1.
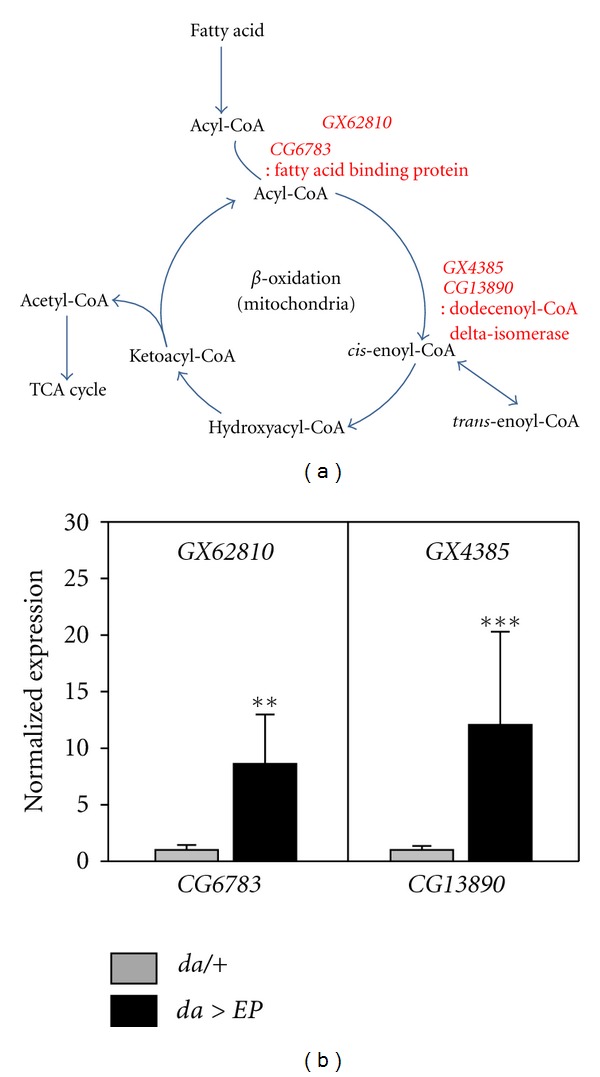
Overexpression of fatty-acid β-oxidation-related genes using EP lines. (a) Schematic representation of fatty-acid β-oxidation. CG6783 encodes the fatty-acid-binding protein, which mediates the transportation of acyl-CoA to cellular organelles. CG13890 encodes dodecenoyl-CoA delta-isomerase, which modifies cis-enoyl-CoA to trans-enoyl-CoA, a common substrate for enoyl-CoA hydratase in the β-oxidation cycle of saturated fatty acyl-CoA esters. (b) Overexpression of CG6783 or CG13890 using EP lines (GX62610, GX4385) and da-Gal4. The mRNA of CG6783 or CG13890 was analyzed in the whole body RNA extract from da > EP (black bars) and da/+ (gray bars) flies. Significance was determined via a t-test (**P < 0.001, ***P < 0.0001).
To further assess the EP lines, they were crossed with da-Gal4 driver or wild-type control stock to produce the da/EP and two controls (EP/+ and da/+) after backcrossing eight times to rule out heterosis. Consistent with previous reports [19], the overexpression of these two fatty-acid β-oxidation-related genes using the da-Gal4 driver increased lifespan. In media containing 16% yeast, the median lifespan of CG6783- or CG13980-overexpressing flies was extended to nearly 58 or 42 days, respectively, from the 32 days observed for the da/+ control flies (Figure 2(a)). In addition, the EP line overexpressing CG6783 also showed extended maximum lifespan (closed-square line in Figure 2(a)), and the two EP lines consistently reduced mortality rate across adult ages (Figure 2(b)). However, it should be noted that the EP/+ control cohorts without the Gal4 driver showed longer lifespans than the parental cohorts (open-square line and open-triangule line, Figure 2(a)), which could be considered to be the side effect of EP insertion to express the target genes under control of the basal promoter possessed in the EP.
Figure 2.
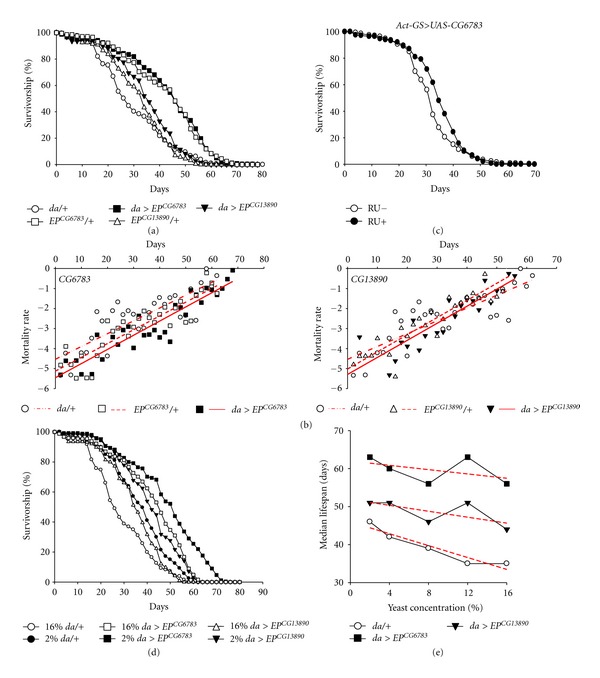
Overexpression of CG6783 or CG13890 throughout the whole body extends lifespan, which is associated with diet restriction. (a) Flies exhibiting overexpression of EP CG6783 (closed-square line) and EP CG13890 (closed-triangule line) fed 16% SY media display increased median and/or maximum lifespan when compared to the da-Gal4 driver alone (open-circular line). Significance was determined via a log-rank test (CG6783, P < 0.001 to da/+, P = 0.6 to EP CG6783/+; CG13890, P < 0.1 to da/+, P < 0.05 to EP CG13890/+). (b) Flies overexpressing CG6783 using UAS-CG6783 and Act-GS-Gal4 showed increased median lifespan in response to feeding with RU486-containing food from day 3 of adulthood (RU+, closed-symbol line) when compared to the uninduced control (RU−, open-symbol line). Significance was determined via a log-rank test (P < 0.0001). (c) Mortality curves of the flies that overexpressed CG6783 or CG13890. The natural log of the mortality rate was plotted using the Gompertz mortality model. Red lines indicate linear regressions for each category. (d) Overexpression of CG6783 or CG13890 throughout the whole body reduced the lifespan extension with dietary restriction. Survival curves of the flies that overexpressed CG6783 (square line) or CG13890 (triangular line) fed 2% or 16% SY media. (e) Dietary restriction in adult Drosophila when CG6783 or CG13890 are overexpressed throughout the whole body. The median lifespan was calculated from Kaplan-Meier survival analysis of the EP lines fed a range of yeast concentrations. Red dashed lines indicate linear regressions for each line.
To further confirm the extension of lifespan by the β-oxidation-related gene, we used the UAS-CG6783 transgenic line, which was generated by standard germ line transformation using a pUAST-CG6783 construct, and analyzed the lifespan when the transgene was driven in adults with the conditional Gene Switch (GS) driver system [27] to produce cohorts of identical genetic background. Female offspring of the Act-GS-Gal4 > UAS-CG6783 genotype showed increased median lifespan compared to uninduced control (Figure 2(c)), indicating that the extension of lifespan by the β-oxidation-related gene occurs independently from the insertion site and the genetic background.
To investigate whether or not the mechanistic basis of dietary restriction has an effect on fatty-acid β-oxidation in association with lifespan extension, flies overexpressing fatty-acid oxidation components were fed an SY diet ranging from 2-to-16% yeast. The median lifespan of the control cohorts increased with decreasing yeast concentration [13, 28]. While control flies showed a 31.4% increase in lifespan upon 2% SY compared to 16% SY conditions (circular lines, Figures 2(d) and 2(e)), the flies expressing CG6783 or CG13890 showed a reduction in the lifespan extension with dietary restriction (12% or 15%, respectively, Figures 2(d) and 2(e)). These results indicated that the promotion of fatty-acid β-oxidation extends lifespan via a mechanism similar to dietary restriction.
3.2. Overexpression of Fatty-Acid β-Oxidation Components Increased Resistance to Oxidative and/or Starvation Stress
A positive relationship between stress tolerance and longevity has been well defined [29, 30], and long-lived organisms tend to be resistant to various forms of environmental stress [31]. Thus, we investigated the effects of overexpressed fatty-acid β-oxidation-related genes on stress resistance. To induce oxidative stress, flies overexpressing fatty-acid-β-oxidation components were subjected to feed dosed with 18 mM paraquat. We found that both of the flies overexpressing each fatty-acid-β-oxidation component showed substantial resistance to oxidative stress (Figures 3(a) and 3(b)). In addition, when subjected to nutrient deprivation, flies overexpressing CG13890 showed more resistance to starvation than the control (Figure 3(d)), whereas the survival rate of the flies overexpressing CG6783 was not significantly altered by starvation (Figure 3(c)). These results indicated that overexpression of fatty-acid β-oxidation-related genes increases lifespan and stress tolerance.
Figure 3.
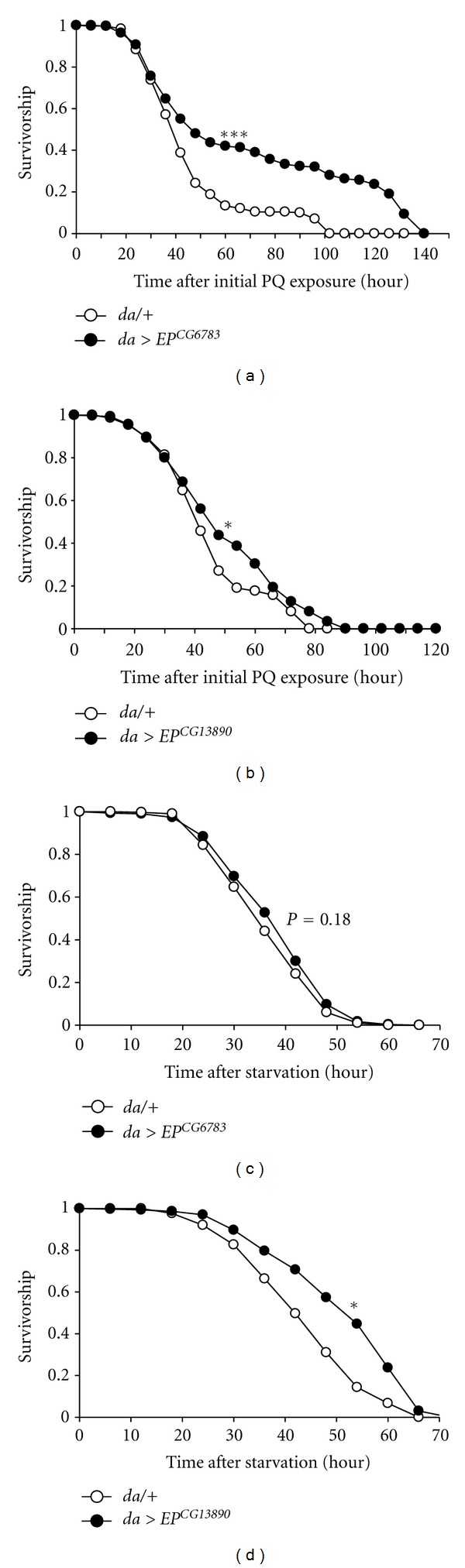
Overexpression of CG6783 or CG13890 increases resistance to stresses. Flies expressing CG6783 or CG13890 exhibited elevated resistance to oxidative stress (a, b) and starvation (c, d) when compared to the control (da/+, open-circule lines). Significance was determined via a log-rank test (*P < 0.01, ***P < 0.0001).
3.3. Overexpression of Fatty-Acid-β-Oxidation Components Activated the dFOXO Signal
Forkhead box (FOXO) is a key mediator of the aging-related pathway that is regulated by signaling pathways including IIS/PI3K/Akt, JNK, AMPK, MST1, CBP, and Sirt1 [12, 32–36]. In addition, dFOXO activation in Drosophila fat body is reportedly associated with physiological traits such as aging, stress resistance, and lipid metabolism [15, 37, 38]. To determine whether lifespan extension and stress resistance produced by the overexpression of fatty-acid-β-oxidation components were associated with dFOXO activation, we immunostained larval fat bodies with anti-dFOXO antibody. In the control fat bodies, an endogenous dFOXO signal was detected in the cytoplasm of all cells and the nuclei of some of the cells (Figure 4(a)). However, the dFOXO signal increased in the nuclei of the fat body as a result of overexpression of the fatty-acid-β-oxidation components (Figure 4(a)).
Figure 4.
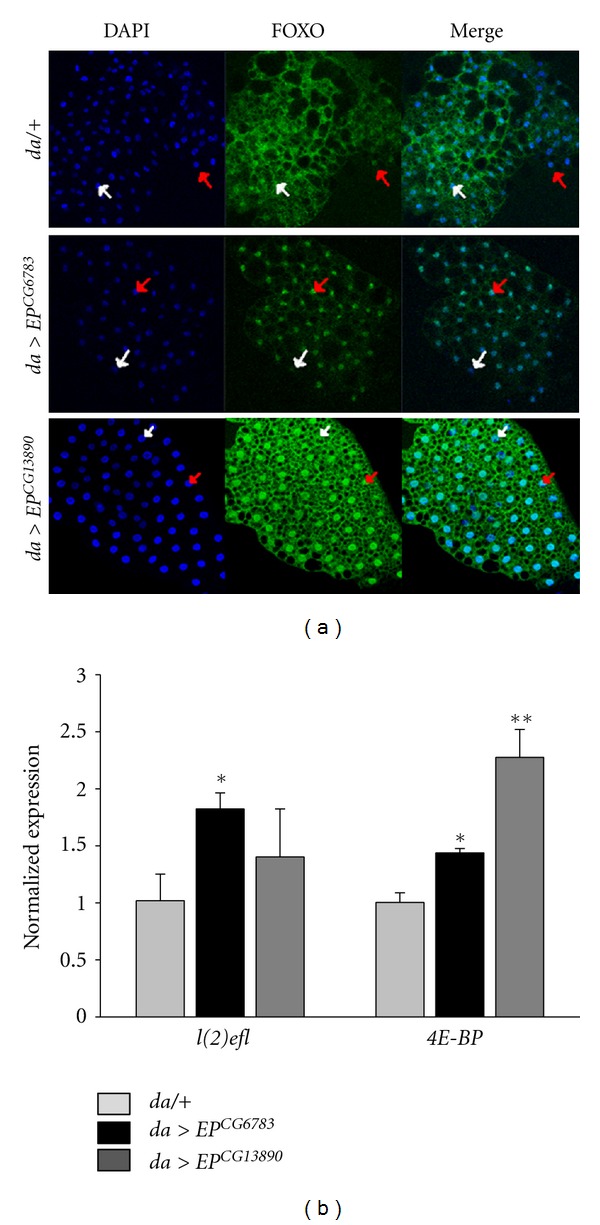
Overexpression of CG6783 or CG13890 activates dFOXO. (a) Overexpression of CG6783 or CG13890 induced the translocation of dFOXO to the nucleus. The fat bodies of the third instar larvae expressing CG6783 or CG13890 under da-Gal4 were stained with anti-dFOXO (green) and DAPI (blue). White arrows indicate dFOXO-negative cells and red arrows indicate dFOXO-positive cells. Original magnification is 200x. (b) Overexpression of CG6783 or CG13890 increased the expression of dFOXO target genes. The mRNA of l(2)efl and 4E-BP was analyzed in the whole body RNA extract from da > EP (black or dark gray bars) and da/+ (gray bars) flies. Significance was determined via a t-test (*P < 0.01, **P < 0.001).
To further assess whether the fatty-acid β-oxidation-related genes activate dFOXO in adults, we analyzed the expression level of the dFOXO transcriptional target gene l(2)efl and 4E-BP in adult whole bodies. The mRNA level of l(2)efl and 4E-BP in the adult whole bodies increased in response to overexpression of the fatty-acid β-oxidation component (Figure 4(b)). These results indicated that increased fatty-acid β-oxidation leads to the activation of FOXO signaling, suggesting that fatty-acid-β-oxidation-induced lifespan extension is linked to FOXO activation.
4. Discussion
In this study, we demonstrated that the overexpression of fatty-acid-β-oxidation-related genes extended median and maximum lifespan and increased stress resistance, suggesting that the level of fatty-acid β-oxidation regulates lifespan. Consistent with our results, many investigations have suggested fatty-acid β-oxidation as a lifespan determinant. One of the well-known longevity-candidate genes, AMPK reportedly regulates fatty-acid synthesis and oxidation [20]. Moreover, calorie restriction and IIS have been reported to promote fatty-acid β-oxidation [21, 22]. In addition, enigma mutant, which exhibits oxidative stress resistance and a longevity phenotype, was found to encode a fatty-acid-β-oxidation related enzyme [39]. A mutant of Withered, which contains the carnitine palmitoyltransferase activity used to import long-chain fatty acids into the mitochondria, was found to be hypersensitive to oxidative and starvation stresses [40]. Furthermore, the mutant fly for mitochondria trifunctional protein containing three kinds of enzyme activities associated with fatty-acid β-oxidation, was recently reported to have a shortened lifespan and decreased locomotion and fecundity [41]. However, the present study is the first to provide direct evidence that the modulation of fatty-acid-β-oxidation components extends lifespan.
Our data showed that lifespan extension by dietary restriction decreased with the overexpression of fatty-acid β-oxidation-related genes, indicating that lifespan extension by fatty-acid-β-oxidation components is associated with dietary restriction. It was previously reported that calorie restriction increased whole-body-fat oxidation [21]. Energy deprivation subsequent to calorie restriction activates AMPK, which subsequently enables the increase of fatty-acid oxidation necessary to utilize the energy resource. These findings suggested that fatty acid oxidation and dietary restriction are related by same underlying mechanisms. However, it should be noted that flies expressing fatty-acid-β-oxidation-related genes still responded to dietary restriction, especially in the lowest-yeast-feeding group. This result suggests that the flies have gained longevity through changes in the fatty-acid-β-oxidation-related genes and also other mechanism(s) unrelated to fatty-acid-β-oxidation in the dietary restriction condition.
Overexpression of the two fatty-acid-β-oxidation components showed similar effects, such as the extension of lifespan, mortality, stress resistance and dFOXO activation, throughout current study. However, flies overexpressing FABP (CG6783) were more resistant to oxidative stress, while DCI (CG13890) expressing flies were more resistant to starvation when compared to each other. FABP facilitates the intracellular movement of fatty acids, thus permitting the translocation of fatty acids to the mitochondria for fatty-acid oxidation and to the nucleus for activation of transcription of the FABP target gene via the fatty-acid nuclear receptors [42]. Therefore, the different effects of the two components on stress resistance may be caused by distinct functions of FABP and DCI.
In this study, we showed that the enhancement of fatty-acid oxidation components activates FOXO transcription factor, suggesting that fatty-acid-β-oxidation-induced lifespan extension is associated with FOXO activation. Fasting DCI homozygous mutants have been found to deposit large amounts of triglycerides in their hepatocytes and accumulated unsaturated fatty acyl groups in their ester lipids [25]. Surprisingly, our data showed that overexpression of FABP and DCI genes led to a mild increase in triglycerides levels (data not shown). This finding was likely the result of activation of FOXO by FABP or DCI overexpression, as it was recently reported that constitutively nuclear FOXO1 in mouse liver produces increased triglyceride accumulation [43]. As a mediator of aging-related signaling pathways, dFOXO is known to be regulated by several factors, including AMPK, JNK, MST1, Sir2, and IIS [12, 32–36]. Thus, further investigations are needed to determine whether the FOXO activation is required for longevity and stress resistance in flies overexpressing fatty acid oxidation-related genes and which signaling pathways are associated with fatty-acid-β-oxidation-related FOXO activation.
Authors' Contribution
S.-H. Lee and S.-K. Lee are contributed equally to the paper.
Acknowledgments
The authors thank Dr. O. Puig for providing antibody and the KAIST Bio Medical Research Center and Bloomington Stock Center for the fly stocks. This work was supported by a grant from Inha University.
References
- 1.Paaby AB, Schmidt PS. Dissecting the genetics of longevity in Drosophila melanogaster . Fly. 2009;3(1):29–38. doi: 10.4161/fly.3.1.7771. [DOI] [PubMed] [Google Scholar]
- 2.Ziv E, Hu D. Genetic variation in insulin/IGF-1 signaling pathways and longevity. Ageing Research Reviews. 2011;10(2):201–204. doi: 10.1016/j.arr.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 3.Kenyon CJ. The genetics of ageing. Nature. 2010;464(7288):504–512. doi: 10.1038/nature08980. [DOI] [PubMed] [Google Scholar]
- 4.Kapahi P, Chen D, Rogers AN, et al. With TOR, less is more: a key role for the conserved nutrient-sensing TOR pathway in aging. Cell Metabolism. 2010;11(6):453–465. doi: 10.1016/j.cmet.2010.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mair W, Morantte I, Rodrigues APC, et al. Lifespan extension induced by AMPK and calcineurin is mediated by CRTC-1 and CREB. Nature. 2011;470(7334):404–408. doi: 10.1038/nature09706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tatar M, Kopelman A, Epstein D, Tu MP, Yin CM, Garofalo RS. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science. 2001;292(5514):107–110. doi: 10.1126/science.1057987. [DOI] [PubMed] [Google Scholar]
- 7.Walker GA, Lithgow GJ. Lifespan extension in C. elegans by a molecular chaperone dependent upon insulin-like signals. Aging Cell. 2003;2(2):131–139. doi: 10.1046/j.1474-9728.2003.00045.x. [DOI] [PubMed] [Google Scholar]
- 8.Carter CS, Ramsey MM, Sonntag WE. A critical analysis of the role of growth hormone and IGF-1 in aging and lifespan. Trends in Genetics. 2002;18(6):295–301. doi: 10.1016/S0168-9525(02)02696-3. [DOI] [PubMed] [Google Scholar]
- 9.Klöting N, Blüher M. Extended longevity and insulin signaling in adipose tissue. Experimental Gerontology. 2005;40(11):878–883. doi: 10.1016/j.exger.2005.07.004. [DOI] [PubMed] [Google Scholar]
- 10.Kaletsky R, Murphy CT. The role of insulin/IGF-like signaling in C. elegans longevity and aging. DMM Disease Models and Mechanisms. 2010;3(7-8):415–419. doi: 10.1242/dmm.001040. [DOI] [PubMed] [Google Scholar]
- 11.Salminen A, Kaarniranta K. Insulin/IGF-1 paradox of aging: regulation via AKT/IKK/NF-κB signaling. Cellular Signalling. 2010;22(4):573–577. doi: 10.1016/j.cellsig.2009.10.006. [DOI] [PubMed] [Google Scholar]
- 12.Rizki G, Iwata TN, Li J, et al. The evolutionarily conserved longevity determinants HCF-1 and SIR-2.1/SIRT1 collaborate to regulate DAF-16/FOXO. PLoS Genetics. 2011;7(9) doi: 10.1371/journal.pgen.1002235.e1002235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tatar M. Diet restriction in Drosophila melanogaster: design and analysis. Interdisciplinary Topics in Gerontology. 2007;35:115–136. doi: 10.1159/000096559. [DOI] [PubMed] [Google Scholar]
- 14.O’Brien DM, Min KJ, Larsen T, Tatar M. Use of stable isotopes to examine how dietary restriction extends Drosophila lifespan. Current Biology. 2008;18(4):R155–R156. doi: 10.1016/j.cub.2008.01.021. [DOI] [PubMed] [Google Scholar]
- 15.Min KJ, Yamamoto R, Buch S, Pankratz M, Tatar M. Drosophila lifespan control by dietary restriction independent of insulin-like signaling. Aging Cell. 2008;7(2):199–206. doi: 10.1111/j.1474-9726.2008.00373.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Smith JV, Heilbronn LK, Ravussin E. Energy restriction and aging. Current Opinion in Clinical Nutrition and Metabolic Care. 2004;7(6):615–622. doi: 10.1097/00075197-200411000-00005. [DOI] [PubMed] [Google Scholar]
- 17.Gáliková M, Klepsatel P, Senti G, Flatt T. Steroid hormone regulation of C. elegans and Drosophila aging and life history. Experimental Gerontology. 2011;46(2-3):141–147. doi: 10.1016/j.exger.2010.08.021. [DOI] [PubMed] [Google Scholar]
- 18.Broué F, Liere P, Kenyon C, Baulieu EE. A steroid hormone that extends the lifespan of Caenorhabditis elegans . Aging Cell. 2007;6(1):87–94. doi: 10.1111/j.1474-9726.2006.00268.x. [DOI] [PubMed] [Google Scholar]
- 19.Paik D, Jang YG, Lee YE, et al. Misexpression screen delineates novel genes controlling Drosophila lifespan. Mechanisms of Ageing and Development. 2012;133(5):234–245. doi: 10.1016/j.mad.2012.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hardie DG, Pan DA. Regulation of fatty acid synthesis and oxidation by the AMP-activated protein kinase. Biochemical Society Transactions. 2002;30(6):1064–1070. doi: 10.1042/bst0301064. [DOI] [PubMed] [Google Scholar]
- 21.Bruss MD, Khambatta CF, Ruby MA, Aggarwal I, Hellerstein MK. Calorie restriction increases fatty acid synthesis and whole body fat oxidation rates. American Journal of Physiology. 2010;298(1):E108–E116. doi: 10.1152/ajpendo.00524.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu X, Gopalacharyulu P, Seppanen- T, et al. Insulin signaling regulates fatty acid catabolism at the level of CoA activation. PLoS Genetics. 2012;8(1) doi: 10.1371/journal.pgen.1002478.e1002478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zimmerman JE, Chan MT, Jackson N, Maislin G, Pack AI. Genetic background has a major impact on differences in sleep resulting from environmental influences in Drosophila . Sleep. 2012;35:545–557. doi: 10.5665/sleep.1744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Veerkamp JH, Maatman RGHJ. Cytoplasmic fatty acid-binding proteins: their structure and genes. Progress in Lipid Research. 1995;34(1):17–52. doi: 10.1016/0163-7827(94)00005-7. [DOI] [PubMed] [Google Scholar]
- 25.Janssen U, Stoffel W. Disruption of mitochondrial β-oxidation of unsaturated fatty acids in the 3,2-trans-enoyl-CoA isomerase-deficient mouse. Journal of Biological Chemistry. 2002;277(22):19579–19584. doi: 10.1074/jbc.M110993200. [DOI] [PubMed] [Google Scholar]
- 26.Houten SM, Wanders RJA. A general introduction to the biochemistry of mitochondrial fatty acid β-oxidation. Journal of Inherited Metabolic Disease. 2010;33(5):469–477. doi: 10.1007/s10545-010-9061-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roman G, Endo K, Zong L, Davis RL. P[switch], a system for spatial and temporal control of gene expression in Drosophila melanogaster . Proceedings of the National Academy of Sciences of the United States of America. 2001;98(22):12602–12607. doi: 10.1073/pnas.221303998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Piper MDW, Bartke A. Diet and aging. Cell Metabolism. 2008;8(2):99–104. doi: 10.1016/j.cmet.2008.06.012. [DOI] [PubMed] [Google Scholar]
- 29.Ristow M, Schmeisser S. Extending life span by increasing oxidative stress. Free Radical Biology and Medicine. 2011;51(2):327–336. doi: 10.1016/j.freeradbiomed.2011.05.010. [DOI] [PubMed] [Google Scholar]
- 30.Yu BP, Chung HY. Stress resistance by caloric restriction for longevity. Annals of the New York Academy of Sciences. 2001;928:39–47. doi: 10.1111/j.1749-6632.2001.tb05633.x. [DOI] [PubMed] [Google Scholar]
- 31.Broughton SJ, Piper MDW, Ikeya T, et al. Longer lifespan, altered metabolism, and stress resistance in Drosophila from ablation of cells making insulin-like ligands. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(8):3105–3110. doi: 10.1073/pnas.0405775102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee KS, Iijima-Ando K, Iijima K, et al. JNK/FOXO-mediated neuronal expression of fly homologue of peroxiredoxin II reduces oxidative stress and extends life span. Journal of Biological Chemistry. 2009;284(43):29454–29461. doi: 10.1074/jbc.M109.028027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Greer EL, Banko MR, Brunet A. AMP-activated protein kinase and FoxO transcription factors in dietary restriction-induced longevity. Annals of the New York Academy of Sciences. 2009;1170:688–692. doi: 10.1111/j.1749-6632.2009.04019.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lehtinen MK, Yuan Z, Boag PR, et al. A conserved MST-FOXO signaling pathway mediates oxidative-stress responses and extends life span. Cell. 2006;125(5):987–1001. doi: 10.1016/j.cell.2006.03.046. [DOI] [PubMed] [Google Scholar]
- 35.Dansen TB, Smits LMM, Van Triest MH, et al. Redox-sensitive cysteines bridge p300/CBP-mediated acetylation and FoxO4 activity. Nature Chemical Biology. 2009;5(9):664–672. doi: 10.1038/nchembio.194. [DOI] [PubMed] [Google Scholar]
- 36.Kim DH, Kim JY, Yu BP, Chung HY. The activation of NF-κB through Akt-induced FOXO1 phosphorylation during aging and its modulation by calorie restriction. Biogerontology. 2008;9(1):33–47. doi: 10.1007/s10522-007-9114-6. [DOI] [PubMed] [Google Scholar]
- 37.Hwangbo DS, Gershman B, Tu MP, Palmer M, Tatar M. Drosophila dFOXO controls lifespan and regulates insulin signalling in brain and fat body. Nature. 2004;429(6991):562–566. doi: 10.1038/nature02549. [DOI] [PubMed] [Google Scholar]
- 38.Giannakou ME, Goss M, Jacobson J, Vinti G, Leevers SJ, Partridge L. Dynamics of the action of dFOXO on adult mortality in Drosophila . Aging Cell. 2007;6(4):429–438. doi: 10.1111/j.1474-9726.2007.00290.x. [DOI] [PubMed] [Google Scholar]
- 39.Mourikis P, Hurlbut GD, Artavanis-Tsakonas S. Enigma, a mitochondrial protein affecting lifespan and oxidative stress response in Drosophila . Proceedings of the National Academy of Sciences of the United States of America. 2006;103(5):1307–1312. doi: 10.1073/pnas.0510564103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Strub BR, Parkes TL, Mukai ST, et al. Mutations of the withered (whd) gene in Drosophila melanogaster confer hypersensitivity to oxidative stress and are lesions of the carnitine palmitoyltransferase I (CPT I) gene. Genome. 2008;51(6):409–420. doi: 10.1139/G08-023. [DOI] [PubMed] [Google Scholar]
- 41.Kishita Y, Tsuda M, Aigaki T. Impaired fatty acid oxidation in a Drosophila model of mitochondrial trifunctional protein (MTP) deficiency. Biochemical and Biophysical Research Communications. 2012;419:344–349. doi: 10.1016/j.bbrc.2012.02.026. [DOI] [PubMed] [Google Scholar]
- 42.Gerstner JR, Vanderheyden WM, Shaw PJ, Landry CF, Yin JC. Cytoplasmic to nuclear localization of fatty-acid binding protein correlates with specific forms of long-term memory in Drosophila . Communicative & Integrative Biology. 2011;4:623–626. doi: 10.4161/cib.4.5.16927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Matsumoto M, Han S, Kitamura T, Accili D. Dual role of transcription factor FoxO1 in controlling hepatic insulin sensitivity and lipid metabolism. Journal of Clinical Investigation. 2006;116(9):2464–2472. doi: 10.1172/JCI27047. [DOI] [PMC free article] [PubMed] [Google Scholar]


