Abstract
OBJECTIVE
There is a dearth of long-term data regarding patient and limb survival in patients with diabetic foot ulcers (DFUs). The purpose of our study was therefore to prospectively investigate the limb and person survival of DFU patients during a follow-up period of more than 10 years.
RESEARCH DESIGN AND METHODS
Two hundred forty-seven patients with DFUs and without previous major amputation consecutively presenting to a single diabetes center between June 1998 and December 1999 were included in this study and followed up until May 2011. Mean patient age was 68.8 ± 10.9 years, 58.7% were male, and 55.5% had peripheral arterial disease (PAD). Times to first major amputation and to death were analyzed with Kaplan-Meier curves and Cox multiple regression.
RESULTS
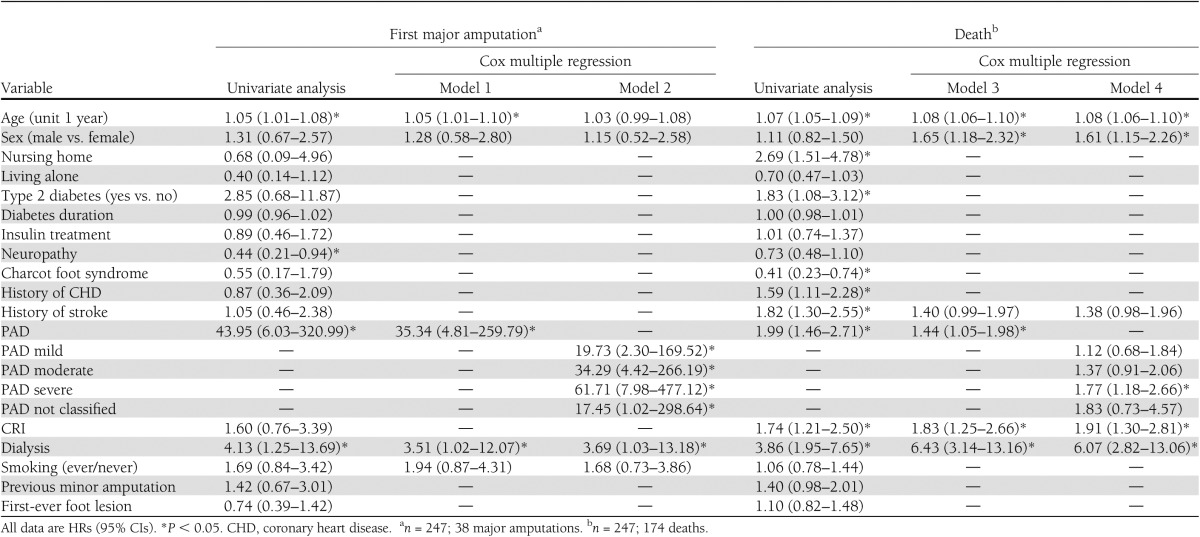
A first major amputation occurred in 38 patients (15.4%) during follow-up. All but one of these patients had evidence of PAD at inclusion in the study, and 51.4% had severe PAD [ankle-brachial pressure index ≤0.4]). Age (hazard ratio [HR] per year, 1.05 [95% CI, 1.01–1.10]), being on dialysis (3.51 [1.02–12.07]), and PAD (35.34 [4.81–259.79]) were significant predictors for first major amputation. Cumulative mortalities at years 1, 3, 5, and 10 were 15.4, 33.1, 45.8, and 70.4%, respectively. Significant predictors for death were age (HR per year, 1.08 [95% CI, 1.06–1.10]), male sex ([1.18–2.32]), chronic renal insufficiency (1.83 [1.25–2.66]), dialysis (6.43 [3.14–13.16]), and PAD (1.44 [1.05–1.98]).
CONCLUSIONS
Although long-term limb salvage in this modern series of diabetic foot patients is favorable, long-term survival remains poor, especially among patients with PAD or renal insufficiency.
Diabetic foot disease is generally associated with high prevalence of advanced microangiopathic and macroangiopathic comorbidities that lead to high morbidity and mortality (1). Although this may indeed be the case, previously published studies have generally confined their outcome analyses to the early phase after initial patient registration, and only a very few have followed up their cohorts for periods of 5 years or longer and reported both amputation and mortality rates (2–5).
Remarkably, we are aware of only two reports in the medical literature that have followed patients with diabetic foot disease for a 10-year period (6–8). One of these studies described cumulative amputation rate and mortality after amputation; the other reported mortality among patients with a history of foot ulceration (without specific description of amputation status). The purpose of our study is therefore to report risk factors associated with first major amputation as well as mortality during a long-term observation period of at least 10 years in a cohort of consecutively presenting patients with diabetic foot ulcers.
RESEARCH DESIGN AND METHODS
Between June 1998 and December 1999, a total of 247 patients with new diabetic foot ulcers and without previous major amputation presented to the study center. They were included in this prospective study and followed up until 31 May 2011, or until death.
Seventy-nine patients (32.0%) were treated by the diabetes team of the study center for the whole observation period or until death, while the remaining patients received foot care in various other institutions. Patients who were cared for at the study center continuously were invited for control at least every 3 months and treated by the same interdisciplinary team both as inpatients and outpatients when they had any new foot lesion. Patients who continued their treatment at external institutions were contacted by the study center at least once yearly.
All patients gave informed consent for participation in the study. They agreed to be contacted personally or allowed the investigators to obtain information on their outcomes from their relatives or their family physicians.
Data assessment
A diabetologist and a diabetic care nurse assessed all patients initially and performed the follow-up visits.
Patient history included data on demographic characteristics, type and duration of diabetes, microvascular and macrovascular comorbidities, and smoking habits. The physical examination included objective evaluation for peripheral neuropathy and peripheral arterial disease (PAD). Protective sensation was assessed separately for each leg with the calibrated Rydel-Seiffer tuning fork and the 5.07 monofilament. Loss of protective sensation as a result of neuropathy was presumed in the presence of insensitivity to the 5.07 monofilament or a vibration perception of 4/8 or below. PAD was defined by an ankle-brachial pressure index (ABI) <0.9 with additional investigation by means of duplex ultrasonography or angiography. Degree of severity of PAD was subdivided according to measured ABI and classified as mild (ABI, 0.7–0.9), moderate (ABI, 0.41–0.69), or severe (ABI, ≤0.4) (9,10). Patients with noncompressible arteries as a result of medial arterial calcification (ABI, ≥1.31) in whom PAD had been diagnosed by imaging techniques were analyzed as having “not classified PAD.” Ischemic heart disease was defined as the presence of a history of angina pectoris or myocardial infarction, any positive cardiac stress test result, or pathological signs on coronary angiography. History of stroke was assumed to be present with any event of neurologic deficiency, whether persistent or resolved. Full definitions and related references have been published elsewhere (11). In accordance with an earlier publication from the same center, we defined chronic renal insufficiency (CRI) by a serum creatinine concentration ≥1.5 mg/dL and dialysis as the continuous need for renal replacement therapy (advanced renal disease) (12). Patients not fulfilling these criteria were considered to have normal renal function. Clinical and demographic data, as well as outcome data (healing, amputation, ulcer recurrence, death), were collected continuously according to a preset standardized protocol and sampled in a study database. The reported data concerned first major amputation and mortality rates among the studied individuals. The cause of death was established from clinical findings before death, from death certificates, or by autopsy.
Statistical analysis
Baseline variables were described depending on their distributions by means, SDs, ranges, or frequency tables. Time from study entry to first major limb amputation and time from study entry to death were evaluated separately as censored event times by Kaplan-Meier curves. Patient data were censored at last observation. In case of the outcome first major amputation, death without major amputation was also counted as a censoring event. Potential risk factors or confounders for both outcomes were analyzed by fitting Cox multiple regression models. The assumption of proportional hazards was assessed graphically by stratified Kaplan-Meier curves (not all shown). The following factors were included in the analysis as possible predictors or confounders: sex, age, smoking, living in a nursing home, living alone, diabetes type, diabetes duration, insulin treatment (yes vs. no), neuropathy, Charcot foot syndrome, history of coronary heart disease, history of stroke, CRI, dialysis, PAD, minor amputation before inclusion, and first-ever foot lesion (yes vs. no). At first, each factor was included as independent variable in an univariate model. With stepwise and backward variable selection (significance level for entry,10%; significance level for remaining, 15%), the most relevant factors were selected. Finally, two resulting main Cox multiple regression models were fitted. Instead of PAD yes or no, the finer classified PAD variable was included in an alternative model. The effect of nonselected covariates from above was estimated in some few cases by including this factor additionally in the final model, and interactions between PAD and CRI were investigated. All statistical tests were performed two-sided at a significance level of 5% if not stated otherwise. Statistical analyses were calculated by the SAS statistical software package (version 9.3; SAS Institute Inc., Cary, NC).
RESULTS
Study population
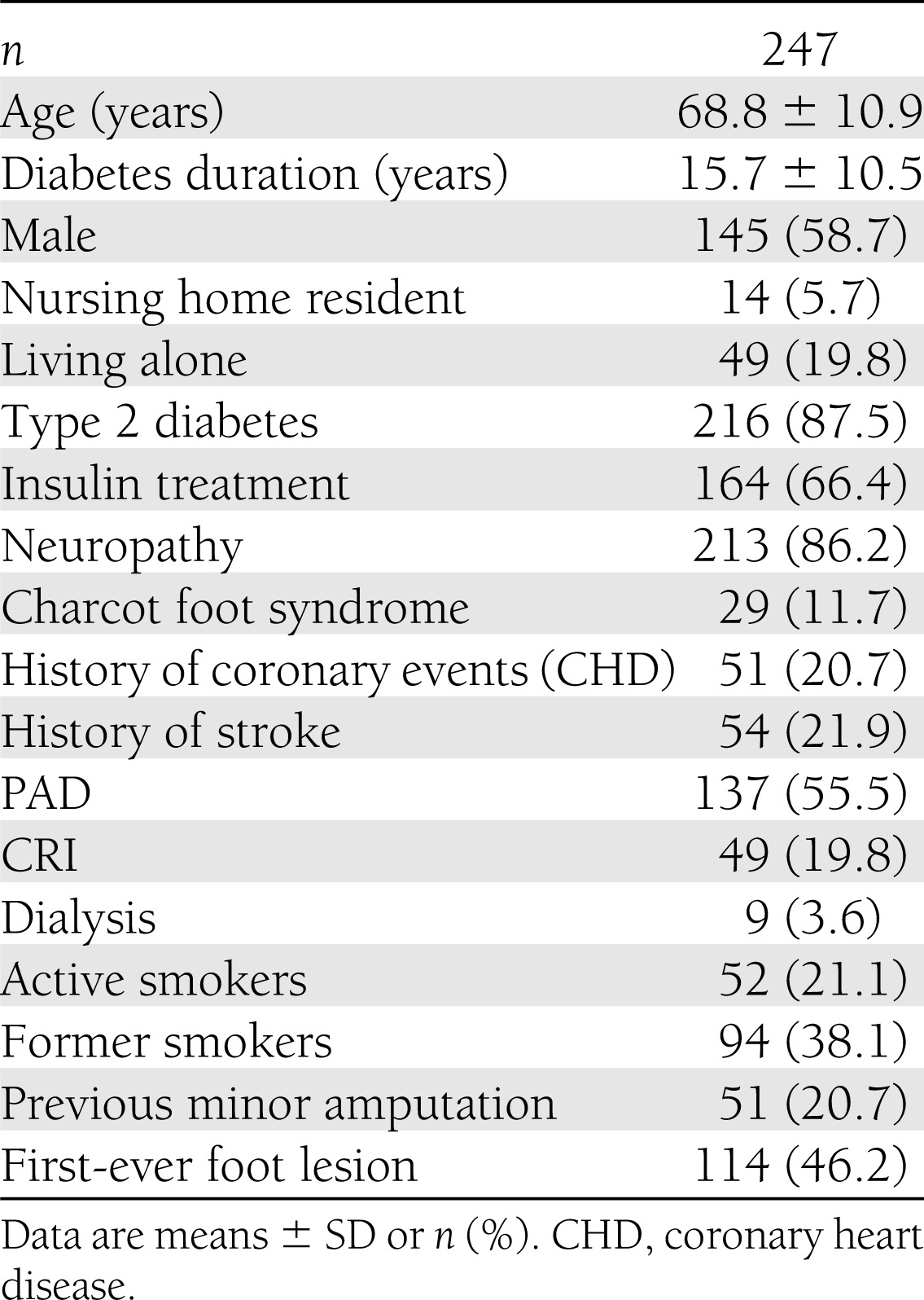
The demographic and clinical patient characteristics are summarized in Table 1. Mean patient age at study inclusion was 68.8 ± 10.9 years (range, 25–91 years), mean diabetes duration was 15.7 ± 10.5 years (range, 0–53 years), 58.7% of the patients were male, and the majority had type 2 diabetes (87.5%). Neuropathy and PAD were present at study initiation in 86.2% and 55.5% of the patients, respectively. The combination of both risk conditions was found in 112 individuals (45.3%). Forty-eight patients with PAD (37.2%) met the definition for a severe stage of the disease in at least one leg. In 40 patients (16.2%), arteries were noncompressible as a result of medial arterial calcification. In eight patients with medial arterial calcification, PAD could not be classified on the basis of ABI. In nine patients (3.6%), there was neither neuropathy nor PAD detected by study criteria. Twenty-nine patients (11.7%) had evidence of active or inactive Charcot foot syndrome at the time of inclusion. Fifty-eight patients (23.5%) fulfilled the criteria for CRI or were receiving renal replacement therapy. Fifty-two patients (21.1%) were active smokers (26.9% of the male patients and 12.8% of the female patients), and 94 patients (38.1%) were former smokers (51.7% of the male patients and 18.6% of the female patients). Histories of a coronary event or stroke were reported by 51 (20.7%) and 54 (21.9%) of the subjects, respectively, without major differences by sex.
Table 1.
Demographic data, risk factors, and comorbidities of the study population
The mean follow-up period was 5.7 ± 4.4 years (range, 0.003–13.2 years), including both survivors and patients who died during the study period. The resulting cumulative observation period was 1,409 person-years.
First major amputation in the study population
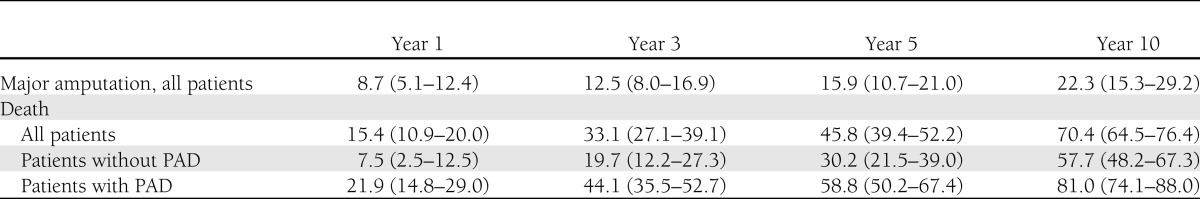
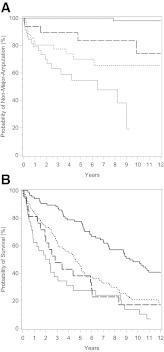
Thirty-eight patients had a first major amputation during the follow-up period. The cumulative probabilities of a first major amputation were 8.7% (5.1–12.4%), 12.5% (8.0–16.9%), 15.9% (10.7–21.0%), and 22.3% (15.3–29.2%) at years 1, 3, 5, and 10, respectively (Table 2). All except one of the affected patients had evidence of PAD at inclusion in the study, and 51.4% had evidence of severe PAD in the concerned extremity. In separate univariate Cox regression models, age, dialysis, and PAD were significant risk increasing factors, and neuropathy was a significant preventive factor (Table 3). After taking PAD into the model as the second variable, however, neuropathy no longer remained significant (hazard ratio [HR], 0.62; P = 0.218). Thus the seemingly protective effect of neuropathy was almost fully explained by the absence of PAD. Age, PAD, dialysis, and smoking were selected by stepwise and backward selection in Cox regression. Age (HR per year, 1.05 [95% CI, 1.01–1.10]; P = 0.023), being on dialysis at baseline (HR, 3.51 [95% CI, 1.02–12.07]; P = 0.046) and PAD at baseline (35.34 [4.81–259.79]; P < 0.001) were the independent predictive variables for a first major limb amputation during follow-up (Table 3, model 1). Presence of mild PAD at baseline increased the risk of a first major amputation 20-fold, moderate PAD 34-fold, and severe PAD 62-fold (Table 3, model 2) relative to no PAD. The HR between severe and mild PAD was 3.13 (1.11–8.79; P = 0.03), and that between severe and moderate PAD was 1.80 (0.84–3.86; P = 0.13). Models including an additional interaction variable between PAD and dialysis did not yield valid results because of the low number of dialysis cases. Kaplan-Meier curves corresponding to cumulative probabilities of the first major amputation stratified by PAD classes present a visual representation of these results (Fig. 1A).
Table 2.
Cumulative probabilities (with 95% CI) of first major limb amputation or death
Table 3.
Univariate analysis and Cox multiple regression models of association between variables and major amputation or death
Figure 1.
A: Relevance of the presence and severity of PAD for the cumulative probability of a first major limb amputation. To avoid complexity, PAD not classified because of medial arterial calcification is not shown (n = 8; only 1 event of first major limb amputation). The highest curve represents no PAD, the second curve represents mild PAD, the third curve represents moderate PAD, and the lowest curve represents severe PAD. B: Relevance of the presence or absence of PAD, advanced renal disease, or both combined for the cumulative probability of death. The highest curve represents no PAD and no renal disease, the second curve represents renal disease and no PAD, the third curve represents PAD and no renal disease, and the lowest curve represents PAD and renal disease.
Patient sex did not significantly influence the risk of a first major amputation.
There was no statistical difference in the risk of a first major amputation during the follow-up period between patients who had undergone a minor amputation before inclusion in the study when this factor was included as an additional covariate (HR, 1.21 [95% CI, 0.56–2.59]; P = 0.630) in model 1 of Table 3.
Mortality in the study population
By 31 May 2011, a total of 174 of the subjects had died, including 83 (47.7%) from cardiac diseases, 17 (9.8%) after stroke, 18 (10.3%) from malignancies, 23 (13.2%) from renal complications, and 24 (13.8%) as a result of septic conditions. Combinations of causes were possible. Septic conditions as cause of death were more prevalent among patients who had CRI or were receiving renal replacement therapy than among patients without obvious renal impairment (21.3 vs. 11.0%).
The cumulative mortalities for the whole cohort at years 1, 3, 5, and 10 were 15.4% (10.9–20.0%), 33.1% (27.1–39.1%), 45.8% (39.4–52.2%), and 70.4% (64.5–76.4%), respectively. For patients with PAD at baseline, the corresponding numbers were 21.9% (14.8–9.0%), 44.1% (35.5–52.7%), 58.8% (50.2–67.4%), and 81.0 (74.1–88.0), respectively (Table 2).
In separate univariate Cox regression models, age, living in a nursing home, type 2 diabetes, history of coronary heart disease, history of stroke, CRI, dialysis, and PAD were significant risk increasing factors, the presence of Charcot foot syndrome a significant preventive factor (Table 3). Age, sex, PAD, CRI, dialysis, and history of stroke were selected by stepwise and backward selection in Cox regression. Living in a nursing home was selected only by backward selection, was not significant (P = 0.116), and was not included in the final model. Independent predictive variables for death were age (HR per year, 1.08 [95% CI 1.06–1.10]; P < 0.001), male sex (1.65 [1.18–2.32]; P = 0.004), CRI (1.83 [1.25–2.66]; P = 0.002), dialysis (6.43 [3.14–13.16]; P < 0.001), and PAD (1.44 [1.05–1.98]; P = 0.023) (Table 3, model 3). When the classified PAD variable was used, severe PAD dominated the significant association of PAD with the risk of death; however, the overall P value for the class variable was not significant (P = 0.065). A significant interaction (P = 0.023) between PAD (yes or no) and CRI was concluded by adding an interaction variable in model 3 of Table 3 (changed HRs [95% CIs] of the main variables, PAD, 1.72 [1.20–1.44]; CRI, 3.25 [1.80–5.85]; and interaction of PAD and CRI, 0.42 [0.20–0.89]). This means higher HRs of the PAD-only and CRI-only patients but lower HR than the corresponding product HR for patients with PAD and CRI (reference, no PAD and no CRI). The interaction between PAD and dialysis was not significant.
Kaplan-Meier survival curves stratified by PAD and renal disease are presented in Fig. 1B.
Patients who had undergone a minor amputation for a previous foot lesion had no significant increase in probability of death during follow-up (HR, 1.25 [95% CI, 0.85–1.83]; P = 0.258) (additional covariable in model 3 of Table 3).
CONCLUSIONS
The results of this study suggest that although long-term limb salvage in a modern series of diabetic foot patients is favorable, long-term patient survival still appears to be poor, especially among patients with PAD, renal insufficiency, or the combination of both. To our knowledge, this is the first report in the medical literature that has examined both risk for major amputation and mortality in a primary data set of patients followed up for more than a decade.
Our cohort was remarkably similar in patient age and high prevalence of PAD to other European cohorts studied for outcomes of diabetic foot disease over shorter (13–15) or longer (≥5 years) (2–5,16) observation periods. In accordance with other studies, we observed a predominance of male patients in our cohort (58.7%); however, it was less pronounced than in some studies (3–5,13,16–18).
Among the patients in our study, 15.4% had a first major amputation during follow-up. Limb loss was observed almost exclusively in patients who had evidence of PAD at study initiation. An independent association between PAD and amputation has been found in other long-term studies as well (3–5); however, all those studies combined minor and major amputations rather than analyzing major amputation exclusively. In a study investigating a large cohort of diabetic foot patients (n = 1,088) treated at centers of excellence in 10 different European countries, the major amputation rate among patients with PAD during a 12-month follow-up was 8%, compared with 2% among patients without PAD (P < 0.001) (13).
More than 50% of the individuals in our study who had a first major amputation had evidence of severe PAD at baseline. The cumulative probability of a first major amputation was strongly linked to the severity of preexisting PAD. None of the other three long-term studies provided clear stratification of severity of PAD. Interestingly, in the report from Ghanassia et al. (5), who followed 84 of 95 hospitalized diabetic foot ulcer patients (95%) for 6.5 years after hospital release, popliteal stenosis as a potential marker for more distal and probably more severe peripheral vascular disease (19) was the only independent predictor of amputation (relative risk, 2.67 [95% CI, 1.34–10.07]; P ≤ 0.01). In two other recent publications, severe PAD (ankle pressure ≤50 mm Hg or toe pressure ≤30 mm Hg) was a predictor of increased major amputation risk in diabetic patients with neuroischemic or ischemic foot ulcers (14,20).
Being on renal replacement therapy at the time of inclusion (HR, 3.51 [95% CI, 1.02–12.07]; P = 0.046) was an independent predictive variable for a first major amputation during follow-up in our study. In the EURODIALE (European Study Group on Diabetes and the Lower Extremity) study, the presence of end-stage renal disease (defined as dependency on hemodialysis or peritoneal dialysis or a previous renal transplant procedure) was an independent predictor of lack of healing (odds ratio, 2.51 [95% CI, 1.41–4.48]; P = 0.002), and that was true for patients with and without PAD (13); however, major amputation in that analysis was not a specifically analyzed end point. In addition, in the work of Gershater et al. (14), uremia was significantly associated with major amputation among patients with neuroischemic and ischemic ulcers (2.43 [1.33–4.45]; P = 0.004) as well as with minor or major amputation among patients with neuropathic ulcers (2.62 [1.39–4.96]; P = 0.003). Both studies, however, had only short observation periods. In the 6.5-year follow-up study from Ghanassia et al. (5) among 62 patients without previous amputation after multivariate analysis, only diabetic nephropathy remained as an independent predictor of first amputation (relative risk, 6.00 [95% CI, 1.62–22.21]; P < 0.01). Authors of the other long-term studies either did not explore renal impairment as an influential factor for major amputation (4) or decided that a possible association with baseline variables would not be significant because of the small numbers of major amputation events in the study (3).
Excess mortality among patients with diabetic foot disease over observation periods of variable duration has been reported repeatedly during recent decades (2–5,7,8,13–17,20,21). The published results are somewhat difficult to compare, however, because some studies describe death without healing of a distinct diabetic foot lesion as the end point (13,14,18), while others report cumulative mortality 1, 3, or 5 years after patient inclusion in the study (2,4,16–18). Death before healing of the initial ulcer occurred in 5.8% of our cases, which is almost identical to the results reported from the EURODIALE study (6.0%) (13). Somewhat higher frequencies were reported from Sweden (14) and from the UK (18). Gershater et al. (14) included patients who died with unhealed stumps after major amputation, however, while our study and the EURODIALE consortium considered the major amputation event to be the definite end point.
Our 1-year mortality was in the same range as the data reported from Nottingham (18) (15.9 and 16.7%, respectively), whereas our 5-year mortality was comparable to the reports of Moulik et al. (4) and Young et al. (17) (45.8% vs. 44 and 48.0%, respectively). Mortality data during a 10-year follow-up period have only been published in two medical reports so far. Izumi et al. (7) studied 277 patients receiving amputations between 1993 and 1997, following them up for as long as 10 years (through 2003). They reported cerebrovascular, cardiovascular, and end-stage renal diseases as being strongly associated with death in those receiving distal amputations. For those receiving high-level amputations, only coronary artery disease was associated with increased mortality risk . Additionally, people with high-level amputations were at greater risk of death than were those receiving low-level amputations. These results confirm the observation of poor survival after major amputation in patients with and without diabetes reported from studies with shorter follow-up periods (22–24); however, the Izumi et al. study (7) exclusively followed up patients who had received a lower limb amputation, not all patients had the opportunity to complete ten years of follow-up, and 10-year mortality was not stated. Iversen et al. (8) used 1995–1997 data from the Nord-Trøndelag Health Study (HUNT 2) to evaluate mortality among people with diabetes who did or did not have a diabetic foot ulcer. People with a diabetic foot ulcer (n = 155) had a 2.3-fold greater risk for death relative to nondiabetic patients during the 10-year follow-up period (49.0% vs. 35.2%), with age, male sex, and smoking as significant covariate factors. The extent of mortality excess equals that observed after 3 years of follow-up in Swedish primary healed diabetic foot ulcer patients (2.35-fold) but is considerably lower than the 3-year value for those with amputation in the same study (3.94-fold) (2). The 10-year cumulative mortality reported from the study of Iversen et al (8) is also substantially lower than the number from our study (70.4%). The difference could be due to a possible underestimation of the mortality risk in HUNT 2 explained by its design. It was stated by the authors that diabetic individuals who did not respond to the questionnaire on foot ulcers reported otherwise more advanced disease. In addition, recruitment procedures in that study made it difficult for housebound or institutionalized individuals to participate, which probably led to a reduced number of elderly people with a history of foot ulcers or amputation in that study relative to other studies (2,21).
In addition to age (HR per year, 1.08), male sex (HR, 1.65 [95% CI, 1.18–2.32]; P = 0.004), CRI (HR, 1.83), being on dialysis (6.43), and PAD (1.44) were independent predictive variables for death in our study. Concerning CRI, this result is in agreement with those of two other long-term studies that analyzed this variable: Ghanassia et al. (5) found renal impairment (relative risk, 4.57 [95% CI, 1.1–19.4]; P < 0.05) to be the only independent predictor of mortality in a multivariate analysis (5), and in the work of Faglia et al. (3) renal impairment was a predictor of death in the univariate analysis (HR, 2.57 [95% CI, 1.22–5.41]; P = 0.013) but was not confirmed in the multivariate analysis. Conversely, Ghanassia et al. (5) did not report a statistical relevance of PAD as a predictor of death, whereas Faglia found the independent association of an ABI ≤0.5 (2.29 [1.29–4.08]; P = 0.005) confirmed by multivariate analysis. This fits nicely with our finding, that severe PAD (ABI ≤0.4) dominates the significant association of PAD with the risk of death when using the classified PAD variables. The increased risk among death of patients with a history of diabetic foot ulcers and its association with a low ABI has recently also been described for an Asian population (25).
In concordance with our own study, male sex was a significant predictor in the 10-year study done by Iversen et al. (8). Although three of the other long-term studies did not report significant sex differences concerning mortality (2,4,5), Faglia et al. (3) found female sex to be independently associated with death (HR, 1.96 [95% CI, 1.08–3.56]; P = 0.027). Because the discrepancy in age between male and female patients in that study was comparable to that among our patients (62.0 vs. 67.2 years and 66.5 vs. 72.0 years, respectively), this conflicting age-adjusted observation remains unexplained for the moment.
More than half of the deaths in our study were from cardio- and cerebrovascular events. This observation is in full agreement with the information given in other European studies with long-term follow-up (2–5,8,16). In contrast, studies on mortality of diabetic foot ulcer patients in developing and newly developed countries report a substantially higher proportion of deaths from septic conditions (26). Neuropathy in and of itself is known to be highly associated with cardiovascular mortality (27–29). Interestingly, in our study neuropathy was not significantly associated with death. This could be explained most plausibly by two factors: 1) nearly all of our patients had clinically significant neuropathy on entry into the cohort, and 2) we did not specifically investigate cardiovascular mortality but rather all-cause mortality.
The main strength of the study is the almost complete follow-up over a long observation period, as long as 13.2 years. Only 20 patients (8.1%) were unavailable for follow-up before death or the end of the observation period. The comparability with other studies investigating the outcome of diabetic foot disease with regard to relevant demographic variables and risk factors may be a further advantage.
A potential negative selection bias always has to be considered when cohorts from specialized diabetic foot care centers are analyzed. It is to be expected that a number of more superficial ulcers of neuropathic origin are treated successfully in primary health care without being seen by a specialized diabetic foot center. Therefore our cohort should be typical for high-risk patients in specialized centers but not for the entire diabetic population. In addition, male predominance was less pronounced in our cohort than in some other reported studies.
In conclusion, data from this long-term study suggest strongly that limb preservation today is the rule rather than the exception, even in high-risk patients with diabetes. Long-term survival remains poor, however, probably because of myriad comorbid conditions for patients whose first presentation to an interdisciplinary clinic is with a diabetic foot ulcer. Efforts to assess and capture these patients earlier in such a clinic may ultimately prove beneficial not only in preventing amputation but also in tangentially prolonging life.
Acknowledgments
No potential conflicts of interest relevant to this article were reported.
S.M. researched patients and wrote the draft of the manuscript. H.F. and H.H. were the study project officers and researched patients. U.G., K.K., G.-T.K., U.K., and T.R. researched patients and contributed to the discussion. A.I. and D.G.A. contributed to the data interpretation and to writing the manuscript. B.H. assisted with writing the manuscript and carried out the statistical analysis. G.R., Z.G.A., and M.B. contributed to the discussion and reviewed and edited the manuscript. S.M. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
The authors thank the patients who participated in this study, their caregivers, and the cooperating medical professionals in the region.
References
- 1.Boulton AJ, Vileikyte L, Ragnarson-Tennvall G, Apelqvist J. The global burden of diabetic foot disease. Lancet 2005;366:1719–1724 [DOI] [PubMed] [Google Scholar]
- 2.Apelqvist J, Larsson J, Agardh CD. Long-term prognosis for diabetic patients with foot ulcers. J Intern Med 1993;233:485–491 [DOI] [PubMed] [Google Scholar]
- 3.Faglia E, Favales F, Morabito A. New ulceration, new major amputation, and survival rates in diabetic subjects hospitalized for foot ulceration from 1990 to 1993: a 6.5-year follow-up. Diabetes Care 2001;24:78–83 [DOI] [PubMed] [Google Scholar]
- 4.Moulik PK, Mtonga R, Gill GV. Amputation and mortality in new-onset diabetic foot ulcers stratified by etiology. Diabetes Care 2003;26:491–494 [DOI] [PubMed] [Google Scholar]
- 5.Ghanassia E, Villon L, Thuan Dit Dieudonné JF, Boegner C, Avignon A, Sultan A. Long-term outcome and disability of diabetic patients hospitalized for diabetic foot ulcers: a 6.5-year follow-up study. Diabetes Care 2008;31:1288–1292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Izumi Y, Satterfield K, Lee S, Harkless LB. Risk of reamputation in diabetic patients stratified by limb and level of amputation: a 10-year observation. Diabetes Care 2006;29:566–570 [DOI] [PubMed] [Google Scholar]
- 7.Izumi Y, Satterfield K, Lee S, Harkless LB, Lavery LA. Mortality of first-time amputees in diabetics: a 10-year observation. Diabetes Res Clin Pract 2009;83:126–131 [DOI] [PubMed] [Google Scholar]
- 8.Iversen MM, Tell GS, Riise T, et al. History of foot ulcer increases mortality among individuals with diabetes: ten-year follow-up of the Nord-Trøndelag Health Study, Norway. Diabetes Care 2009;32:2193–2199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hiatt WR. Medical treatment of peripheral arterial disease and claudication. N Engl J Med 2001;344:1608–1621 [DOI] [PubMed]
- 10.Chantelau E, Lee KM. Ankle/arm pressure index in diabetes mellitus. Diabetologia 1994;37:223. [DOI] [PubMed] [Google Scholar]
- 11.Morbach S, Lutale JK, Viswanathan V, et al. Regional differences in risk factors and clinical presentation of diabetic foot lesions. Diabet Med 2004;21:91–95 [DOI] [PubMed] [Google Scholar]
- 12.Morbach S, Quante C, Ochs HR, Gaschler F, Pallast JM, Knevels U. Increased risk of lower-extremity amputation among Caucasian diabetic patients on dialysis. Diabetes Care 2001;24:1689–1690 [DOI] [PubMed] [Google Scholar]
- 13.Prompers L, Schaper N, Apelqvist J, et al. Prediction of outcome in individuals with diabetic foot ulcers: focus on the differences between individuals with and without peripheral arterial disease. The EURODIALE Study. Diabetologia 2008;51:747–755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gershater MA, Löndahl M, Nyberg P, et al. Complexity of factors related to outcome of neuropathic and neuroischaemic/ischaemic diabetic foot ulcers: a cohort study. Diabetologia 2009;52:398–407 [DOI] [PubMed] [Google Scholar]
- 15.Winkley K, Stahl D, Chalder T, Edmonds ME, Ismail K. Risk factors associated with adverse outcomes in a population-based prospective cohort study of people with their first diabetic foot ulcer. J Diabetes Complications 2007;21:341–349 [DOI] [PubMed] [Google Scholar]
- 16.Winkley K, Sallis H, Kariyawasam D, et al. Five-year follow-up of a cohort of people with their first diabetic foot ulcer: the persistent effect of depression on mortality. Diabetologia 2012;55:303–310 [DOI] [PubMed] [Google Scholar]
- 17.Young MJ, McCardle JE, Randall LE, Barclay JI. Improved survival of diabetic foot ulcer patients 1995-2008: possible impact of aggressive cardiovascular risk management. Diabetes Care 2008;31:2143–2147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jeffcoate WJ, Chipchase SY, Ince P, Game FL. Assessing the outcome of the management of diabetic foot ulcers using ulcer-related and person-related measures. Diabetes Care 2006;29:1784–1787 [DOI] [PubMed] [Google Scholar]
- 19.Graziani L, Silvestro A, Bertone V, et al. Vascular involvement in diabetic subjects with ischemic foot ulcer: a new morphologic categorization of disease severity. Eur J Vasc Endovasc Surg 2007;33:453–460 [DOI] [PubMed] [Google Scholar]
- 20.Apelqvist J, Elgzyri T, Larsson J, Löndahl M, Nyberg P, Thörne J. Factors related to outcome of neuroischemic/ischemic foot ulcer in diabetic patients. J Vasc Surg 2011;53:1582–1588, e2 [DOI] [PubMed] [Google Scholar]
- 21.Boyko EJ, Ahroni JH, Smith DG, Davignon D. Increased mortality associated with diabetic foot ulcer. Diabet Med 1996;13:967–972 [DOI] [PubMed] [Google Scholar]
- 22.Subramaniam B, Pomposelli F, Talmor D, Park KW. Perioperative and long-term morbidity and mortality after above-knee and below-knee amputations in diabetics and nondiabetics. Anesth Analg 2005;100:1241–1247 [DOI] [PubMed] [Google Scholar]
- 23.Tentolouris N, Al-Sabbagh S, Walker MG, Boulton AJ, Jude EB. Mortality in diabetic and nondiabetic patients after amputations performed from 1990 to 1995: a 5-year follow-up study. Diabetes Care 2004;7:1598–1604 [DOI] [PubMed] [Google Scholar]
- 24.Icks A, Scheer M, Morbach S, et al. Time-dependent impact of diabetes on mortality in patients after major lower extremity amputation: survival in apopulation-based 5-year cohort in Germany. Diabetes Care 2011;34:1350–1354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Junrungsee S, Kosachunhanun N, Wongthanee A, Rerkasem K. History of foot ulcers increases mortality among patients with diabetes in Northern Thailand. Diabet Med 2011;28:608–611 [DOI] [PubMed] [Google Scholar]
- 26.Morbach S, Gröblinghoff U, Schulze H, et al. All-cause mortality after diabetes-related amputation in Barbados: a prospective case-control study: response to Hambleton et Al. Diabetes Care 2009;32:e100–; author reply e101. [DOI] [PubMed] [Google Scholar]
- 27.Forsblom CM, Sane T, Groop PH, et al. Risk factors for mortality in Type II (non-insulin-dependent) diabetes: evidence of a role for neuropathy and a protective effect of HLA-DR4. Diabetologia 1998;41:1253–1262 [DOI] [PubMed] [Google Scholar]
- 28.Coppini DV, Bowtell PA, Weng C, Young PJ, Sönksen PH. Showing neuropathy is related to increased mortality in diabetic patients - a survival analysis using an accelerated failure time model. J Clin Epidemiol 2000;53:519–523 [DOI] [PubMed] [Google Scholar]
- 29.Chammas NK, Hill RL, Foster AV, Edmonds ME. Is neuropathic ulceration the key to understanding increased mortality due to ischaemic heart disease in diabetic foot ulcer patients? A population approach using a proportionate model. J Int Med Res 2002;30:553–559 [DOI] [PubMed] [Google Scholar]