Abstract
Background
Although thoracoscopic resection is less invasive than resection via thoracotomy, it does not allow manual palpation of the lung to identify any metastases not visible on the preoperative scan or at the time of resection.
Hypothesis
Preoperative imaging underestimates the number of pulmonary melanoma metastases.
Design
Retrospective review of a prospectively maintained database.
Setting
Tertiary referral center.
Patients and Interventions
All patients who underwent preoperative computed tomography (CT) of the chest, followed within 30 days by thoracotomy for resection of pulmonary metastatic melanoma.
Main Outcome Measures
Number of pathology-confirmed pulmonary metastases detected by preoperative chest CT versus intraoperative manual palpation.
Results
The 170 patients had a mean age of 49.5 years at initial diagnosis of melanoma and 57.1 years at diagnosis of pulmonary metastases; 69% of patients were male. A total of 334 pulmonary metastases were resected; mean lesion size was 2.0 cm (range 0.1 – 14.0). In 49 of 190 pulmonary resections (26%), manual palpation of the subpleural parenchyma revealed lesions not identified during preoperative imaging. Rate of 5-year overall survival was 33%.
Conclusions
Preoperative imaging underestimates the number of pulmonary lesions in patients with metastatic melanoma. Since incomplete resection of metastatic disease is associated with worse outcomes, we recommend caution when considering a minimally invasive approach for the resection of pulmonary metastatic melanoma.
INTRODUCTION
The first planned pulmonary metastasectomy was performed in 1927 by Divis in Europe and then in North America by Barney and Churchhill in 1939.1 Barney and Churchill performed a lobectomy for a pulmonary renal cell metastasis; postoperative disease-free interval exceeded 20 years.2,3 Subsequent studies have reported long-term survival after pulmonary metastasectomy for various malignancies in appropriately selected patients.1,2,4–9
Approximately 30% of patients with cutaneous melanoma develop metastatic disease.1,10 Approximately 40% of patients with distant metastatic melanoma present with isolated pulmonary metastasis.2,7,10 New systemic therapies offered to patients with pulmonary melanoma metastases are usually palliative but very rarely curative. Surgical resection can produce long-term survival but only when all pulmonary metastases are removed.1,4,11,12
Most pulmonary metastases in patients with melanoma are detected by computed tomography (CT) of the chest.6,13 Increased numbers of patients are being referred for minimally invasive techniques to resect lesions detected on CT.13 While these minimally invasive techniques can decrease postoperative pain and hospital stay, they rely heavily on preoperative imaging.14 In contrast, bimanual palpation of the lung parenchyma to detect additional metastatic lesions can only be achieved via a thoracotomy. While the resolution of CT images has improved dramatically over the last few decades, we hypothesize that preoperative CT cannot detect all lesions identified during intraoperative manual palpation of the lung parenchyma.
The purpose of our study was to examine differences between the number of lesions reported on preoperative chest CT and the number of lesions detected by manual palpation and visual inspection of the lung parenchyma during surgery and confirmed by pathology.
METHODS
The melanoma database at the John Wayne Cancer Institute (JWCI) was queried to identify patients who underwent thoracotomy and resection for pulmonary metastatic melanoma from 1994 to 2010. We excluded any patient who had not undergone CT scanning within 30 days before thoracotomy, and any patient whose pathology and radiology reports were not available. At JWCI, selection of patients for pulmonary metastasectomy is based on previously published criteria including complete treatment of the primary melanoma.1,4 Standard preoperative workup involves CT of the chest/abdomen/pelvis and magnetic resonance imaging of the brain. Whole-body positron emission tomography is sometimes included, but our experience confirms previous reports that this technique is more useful for detection of larger (> 5 mm) metastatic lesions.15 Patients with evidence of unresectable extrathoracic disease are referred for systemic therapy instead of metastasectomy.5,12,13 All surgical candidates must demonstrate adequate cardiopulmonary reserve and the ability to tolerate the operative procedure.
The technique used at JWCI for pulmonary metastasectomy has been previously described.1 Briefly, the operative exposure for pulmonary resections is determined by the number and location of metastatic lesions demonstrated on preoperative chest CT. The lung parenchyma is carefully examined by bimanual palpation and visual inspection in both the inflated and deflated conditions. The location of each metastasis is marked with a silk suture. All metastatic lesions are marked prior to resection of any lesion because resection inevitably creates artifacts at the suture line, which can obscure additional lesions. Because most metastatic lesions are located subpleurally, a wedge resection is frequently possible. Resection of pulmonary metastases should remove each lesion with a tumor-negative pathologic margin of 2–3 cm, while preserving the maximum amount of lung parenchyma.1,4
Data regarding patient demographics, primary tumor site, radiographic findings, intraoperative findings, and pathologic analysis were retrieved for analysis. The number of lesions identified during preoperative imaging was compared with the number of lesions identified during metastasectomy for each case, for each year, and for the entire study period. Student t-test or chi-square analysis was used to compare patient-related and tumor-related factors based on radiographic and pathologic findings. A p-value of <0.05 was considered significant. The study was reviewed and determined to be exempt from institutional review board approval.
RESULTS
During the study period, 170 eligible patients underwent pulmonary resections for metastatic melanoma; 117 patients (69%) were men and 53 (31%) were women. Mean age at initial melanoma diagnosis was 49.5 years. Of the 170 primary melanomas, 50 (29%) were on the head and neck, 53 (31%) on the trunk, 17 (10%) on the upper extremity, 20 (12%) on the lower extremity, and 30 (18%) were unknown. The primary melanomas had a mean Breslow thickness of 2.55 mm; 21 (12%) primary melanomas were ulcerated. The median interval between initial diagnosis of melanoma and pulmonary resection was 58 months. The mean age at pulmonary resection was 57.1 years (Table 1). All pulmonary metastasectomies were performed via a thoracotomy incision or median sternotomy in patients with disease in both lungs. The median length of hospital stay after pulmonary metastasectomy was 4 days (range, 2 – 14 days).
Table 1.
Patient demographics and primary melanoma characteristics.
| Variable | Number |
|---|---|
| Sex | |
| Male | 53 (31%) |
| Female | 117 (69%) |
| Mean age at primary diagnosis, in years | 49.5 ± 14.6 |
| Primary Location | |
| Head and Neck | 50 (29%) |
| Trunk | 53 (31%) |
| Upper Extremity | 17 (10%) |
| Lower Extremity | 20 (12%) |
| Unknown | 30 (18%) |
| Mean Breslow thickness, in mm | 2.55 ± 3.35 |
| Ulceration Present | 21 (12%) |
| Mean age at pulmonary resection, in years | 57.1 ±14.8 |
| Median interval from primary diagnosis to pulmonary resection, in months | 58 |
The 170 patients underwent 190 elective pulmonary resections to remove 262 lesions identified during preoperative chest CT (Figure 1A). During 49 of the 190 operative procedures (26%), additional lesions were detected by manual palpation and/or visual inspection of the subpleural parenchyma. The number of additional lesions detected visually versus manually was not recorded in the operative record. Final pathology reports indicated that 334 pulmonary metastatic melanoma lesions were excised during the 190 procedures (Figure 1B).
Figure 1.
A – Number of pulmonary metastatic lesions detected by preoperative imaging. B – Number of pulmonary metastatic lesions detected through visual inspection and manual palpation and confirmed by pathology. C – Percentage of missed lesions by pulmonary lobe.
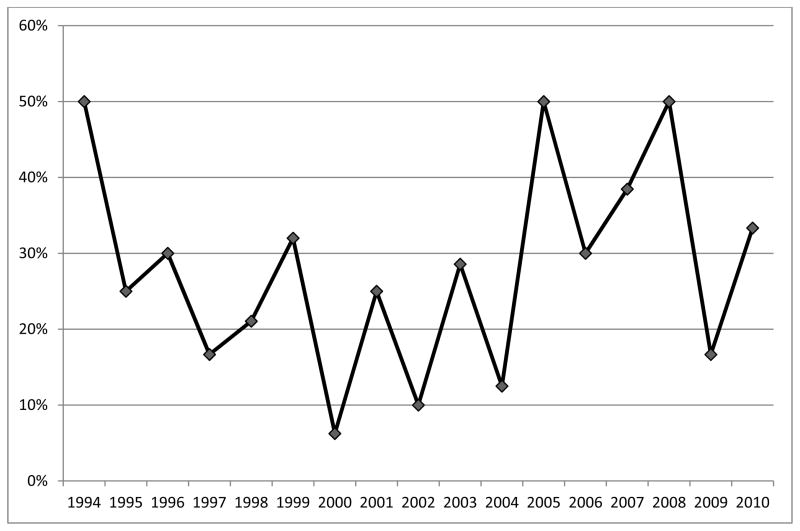
Intraoperative detection of additional lesions was directly correlated with the number of preoperatively imaged lesions (2.0 ± 0.4 for cases with additional lesions versus 1.2 ± 0.5 for cases with no additional lesions, p=0.0007). More surprisingly, intraoperative detection of additional lesions was inversely correlated with the diameter of preoperatively imaged lesions (1.5 cm ± 0.3 for cases with additional lesions versus 2.2 cm ± 0.4 for cases with no additional lesions, p=0.003). The mean diameter of a missed lesion was 0.5 cm. A lesion was detected in a different lobe of lung in 33% of cases in which additional lesions were found. As shown in Figure 2, the rate of missed lesions did not appear to decrease during the study period.
Figure 2.
Percentage of cases per year in which intraoperative palpation and/or visual inspection of lung parenchyma identified pulmonary metastases not detected during preoperative computed tomographic imaging of the chest.
The five-year overall survival was 33%, with a median time from pulmonary resection to death of 17 months.
DISCUSSION
The use of pulmonary metastasectomy in the treatment of selected patients with pulmonary metastatic melanoma, as well as other extrathoracic malignancies, improves overall survival.1–5,7,9,14 These improvements in survival are found when judicious patient selection is combined with complete surgical resection of all disease.1,4,5,12 In addition to appropriate cardiopulmonary reserve, patients should lack evidence of extrathoracic disease, and exhibit a tumor volume doubling time (TVDT) greater than 40–60 days.4,12 We have found that median survival after pulmonary metastasectomy correlates positively with TVDT and that TVDT is important in determining prognosis.1,4,5
Previous groups have compared detection of pulmonary metastases by preoperative chest CT versus bimanual palpation.16,17 Parsons et al18 performed a retrospective review of 34 patients who underwent 41 pulmonary resections for metastases from various cancers. The most common cancer was osteosarcoma. Two patients with metastatic melanoma were included in this study. Of the 41 patients, nine (22%) had additional malignant nodules detected during intraoperative bimanual palpation.18 Similar results were reported by Cerfolio et al19; in their prospective study of 152 patients, 51 (34%) patients had 57 pulmonary nodules that were not imaged preoperatively and 32 of the 57 (56%) nodules were malignant. Advocates for thoracoscopic pulmonary metastasectomy have argued that while this technique does rely heavily on preoperative imaging, lesions not detected on preoperative imaging will also be detected and resected. However, Ellis et al20 performed a retrospective review of patients who underwent metastasectomy via thoracoscopy versus thoracotomy. While the number of lesions detected during preoperative imaging was not significantly different between patients in thoracoscopy and thoracotomy groups, the number of resected lesions was significantly higher in the thoracotomy group. In addition, Mutsaerts et al16 prospectively studied 28 patients enrolled in a trial of thoracoscopic resection followed immediately by confirmatory thoracotomy. Of 17 patients who were able to undergo both procedures, 5 had residual disease demonstrated by confirmatory thoracotomy.
In a follow-up study examining long-term survival, Mutsaerts et al21 compared 19 patients undergoing metastasectomy via thoracotomy and 12 patients undergoing thoracoscopic metastasectomy. Three melanoma patients were included in this study. Rates of complications were higher in the thoracotomy group (5 versus 0, p=0.049). There was no difference in 2-year overall survival between the thoracoscopic and thoracotomy groups (67% versus 70%, p=NS).
Efforts have been made to develop guidelines to determine whether a patient should undergo metastasectomy via a thoracoscopic or thoracotomy approach.21 It has been suggested that patients with a small solitary lesion detected on preoperative imaging should be candidates for thoracoscopic metastasectomy. It is true that we were more likely to find additional metastatic lesions in patients who had a higher mean number of imaged lesions. However 61% (30/49) of the resections undertaken to remove a solitary imaged lesion revealed additional metastatic lesions. With regards to size, patients with a smaller mean tumor diameter based on preoperative imaging were more likely to have additional metastatic lesions identified at the time of metastasectomy. Unfortunately our data did not allow us to determine whether manual palpation or visual inspection was more effective for intraoperative detection of metastases. However, in our experience, most pulmonary metastatic lesions are located in the subpleural space and are detected manually.
Detterbeck et al22 performed a literature review regarding imaging requirements in the practice of pulmonary metastasectomy. The average reported rate of pulmonary metastasis not detected on preoperative imaging was 25%. Interestingly there was no difference in lesion detection with 5-mm helical chest CT versus the high-resolution 3-mm chest CT.
The clinical significance of non-imaged, resected malignant nodules is unknown. The five-year overall survival for our study was 33%, with a median time from pulmonary resection to death of 17 months. Bhargava et al23 compared 31 patients who underwent either VATS or open metastasectomy for metastatic pulmonary melanoma. Median survival was 20.7 months for the VATS group and 26.5 months for the open group (p=0.17).23
In our study, bimanual palpation and visual inspection of the lung parenchyma during pulmonary metastasectomy detected lesions missed by preoperative imaging in 26% of cases. In addition, in 33% of cases the missed lesion was detected in a different lobe of the lung. Although ongoing improvements in imaging quality and technologies may eventually render manual palpation obsolete, as yet we cannot report a trend towards improved preoperative detection of pulmonary metastatic melanoma lesions (Figure 2). We therefore recommend caution when considering a thoracoscopic approach for the resection of pulmonary metastatic melanoma.
Acknowledgments
Funding/Support: This study was supported in part by grant P01 CA12582 from the National Institutes of Health (DL Morton, Principal Investigator), and by funding from the John Wayne Cancer Institute Auxiliary (Santa Monica, CA), Dr. Miriam & Sheldon G. Adelson Medical Research Foundation (Boston, MA), and the Melanoma Research Alliance (Washington, DC). The content is solely the responsibility of the authors and does not necessarily represent the official view of the National Cancer Institute or the National Institutes of Health.
Footnotes
Previous Presentation: Presented at the Pacific Coast Surgical Association’s Annual Meeting in Napa Valley, CA. February 18, 2012
References
- 1.Tafra L, Dale PS, Wanek LA, et al. Resection and adjuvant immunotherapy for melanoma metastatic to the lung and thorax. J Thorac Cardiovasc Surg. 1995;110:119–28. doi: 10.1016/S0022-5223(05)80017-0. discussion 129. [DOI] [PubMed] [Google Scholar]
- 2.Greenlish JPFJ. Secondary Pulmonary Malignancy. Surg Clin North Am. 2000;80:633–657. doi: 10.1016/s0039-6109(05)70204-8. [DOI] [PubMed] [Google Scholar]
- 3.Sellors TH. Treatment of isolated pulmonary metastases. Br Med J. 1970;2:253–6. doi: 10.1136/bmj.2.5704.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ollila DW, Morton DL. Surgical resection as the treatment of choice for melanoma metastatic to the lung. Chest Surg Clin N Am. 1998;8:183–96. [PubMed] [Google Scholar]
- 5.Ollila DW, Stern SL, Morton DL. Tumor doubling time: a selection factor for pulmonary resection of metastatic melanoma. J Surg Oncol. 1998;69:206–11. doi: 10.1002/(sici)1096-9098(199812)69:4<206::aid-jso3>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 6.Sternberg DI, Sonett JR. Surgical therapy of lung metastases. Semin Oncol. 2007;34:186–96. doi: 10.1053/j.seminoncol.2007.03.004. [DOI] [PubMed] [Google Scholar]
- 7.Hornbech K, Ravn J, Steinbruchel DA. Current status of pulmonary metastasectomy. Eur J Cardiothorac Surg. 2011;39:955–62. doi: 10.1016/j.ejcts.2010.10.001. [DOI] [PubMed] [Google Scholar]
- 8.Rusch VW. Pulmonary metastasectomy. Current indications. Chest. 1995;107:322S–331S. doi: 10.1378/chest.107.6_supplement.322s. [DOI] [PubMed] [Google Scholar]
- 9.Leo F, Cagini L, Rocmans P, et al. Lung metastases from melanoma: when is surgical treatment warranted? Br J Cancer. 2000;83:569–72. doi: 10.1054/bjoc.2000.1335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee YT. Malignant melanoma. pattern of metastasis. CA Cancer J Clin. 1980;30:137–42. doi: 10.3322/canjclin.30.3.137. [DOI] [PubMed] [Google Scholar]
- 11.Petersen RP, Hanish SI, Haney JC, et al. Improved survival with pulmonary metastasectomy: an analysis of 1720 patients with pulmonary metastatic melanoma. J Thorac Cardiovasc Surg. 2007;133:104–10. doi: 10.1016/j.jtcvs.2006.08.065. [DOI] [PubMed] [Google Scholar]
- 12.Lee JH, Gulec SA, Kyshtoobayeva A, et al. Biological factors, tumor growth kinetics, and survival after metastasectomy for pulmonary melanoma. Ann Surg Oncol. 2009;16:2834–9. doi: 10.1245/s10434-009-0583-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Joseph WL, Morton DL, Adkins PC. Prognostic significance of tumor doubling time in evaluating operability in pulmonary metastatic disease. J Thorac Cardiovasc Surg. 1971;61:23–32. [PubMed] [Google Scholar]
- 14.Yano T, Shoji F, Maehara Y. Current status of pulmonary metastasectomy from primary epithelial tumors. Surg Today. 2009;39:91–7. doi: 10.1007/s00595-008-3820-9. [DOI] [PubMed] [Google Scholar]
- 15.Reinhardt MJ, Wiethoelter N, Matthies A, et al. PET recognition of pulmonary metastases on PET/CT imaging: impact of attenuation-corrected and non-attenuation-corrected PET images. Eur J Nucl Med Mol Imaging. 2006;33:134–9. doi: 10.1007/s00259-005-1901-1. [DOI] [PubMed] [Google Scholar]
- 16.Mutsaerts EL, Zoetmulder FA, Meijer S, et al. Outcome of thoracoscopic pulmonary metastasectomy evaluated by confirmatory thoracotomy. Ann Thorac Surg. 2001;72:230–3. doi: 10.1016/s0003-4975(01)02629-7. [DOI] [PubMed] [Google Scholar]
- 17.Nakas A, Klimatsidas MN, Entwisle J, et al. Video-assisted versus open pulmonary metastasectomy: the surgeon’s finger or the radiologist’s eye? Eur J Cardiothorac Surg. 2009;36:469–74. doi: 10.1016/j.ejcts.2009.03.050. [DOI] [PubMed] [Google Scholar]
- 18.Parsons AM, Detterbeck FC, Parker LA. Accuracy of helical CT in the detection of pulmonary metastases: is intraoperative palpation still necessary? Ann Thorac Surg. 2004;78:1910–6. doi: 10.1016/j.athoracsur.2004.05.065. discussion 1916–8. [DOI] [PubMed] [Google Scholar]
- 19.Cerfolio RJ, Bryant AS, McCarty TP, et al. A prospective study to determine the incidence of non-imaged malignant pulmonary nodules in patients who undergo metastasectomy by thoracotomy with lung palpation. Ann Thorac Surg. 2011;91:1696–700. doi: 10.1016/j.athoracsur.2011.02.075. discussion 1700–1. [DOI] [PubMed] [Google Scholar]
- 20.Ellis MC, Hessman CJ, Weerasinghe R, et al. Comparison of pulmonary nodule detection rates between preoperative CT imaging and intraoperative lung palpation. Am J Surg. 2011;201:619–22. doi: 10.1016/j.amjsurg.2011.01.005. [DOI] [PubMed] [Google Scholar]
- 21.Mutsaerts EL, Zoetmulder FA, Meijer S, et al. Long term survival of thoracoscopic metastasectomy vs metastasectomy by thoracotomy in patients with a solitary pulmonary lesion. Eur J Surg Oncol. 2002;28:864–8. doi: 10.1053/ejso.2002.1284. [DOI] [PubMed] [Google Scholar]
- 22.Detterbeck FC, Grodzki T, Gleeson F, et al. Imaging requirements in the practice of pulmonary metastasectomy. J Thorac Oncol. 2010;5:S134–9. doi: 10.1097/JTO.0b013e3181dcf64d. [DOI] [PubMed] [Google Scholar]
- 23.Bhargava A, Schuchert MJ, Wilson DO, Landreneau RJ, Luketich JD, Christie NA. Metastasectomy for melanoma in the VATS era. J Clin Oncol. 2009;27(suppl):abstr e20001. [Google Scholar]