Abstract
Although small molecule actin modulators have been widely used as research tools, only one cell permeable small molecule inhibitor of actin depolymerization (jasplakinolide) is commercially available. We report that the natural product cucurbitacin E inhibits actin depolymerization and show that its mechanism of action is different from jasplakinolide. In assays using pure fluorescently labeled actin, cucurbitacin E specifically affected depolymerization without affecting polymerization. It inhibited actin depolymerization at sub-stoichiometric concentrations up to 1:6 cucurbitacin:actin E. Cucurbitacin E specifically binds to filamentous actin (F-actin) forming a covalent bond at residue Cys257, but not to monomeric actin (G-actin). Based on its compatibility with phalloidin staining, we show that cucurbitacin E occupies a different binding site on actin filaments. Using loss of fluorescence after localized photoactivation, we found that cucurbitacin E inhibited actin depolymerization in live cells. Cucurbitacin E is a widely available plant-derived natural product, making it a useful tool to study actin dynamics in cells and actin-based processes such as cytokinesis.
INTRODUCTION
Actin is one of the most important and abundant proteins in the cell. It is a key component of the cytoskeleton and is involved in many cellular functions such as cell morphogenesis, movement, adhesion, polarity and division. In cells, actin is found in monomeric form (G-actin) and in filamentous form (F-actin). Most of actin’s roles are related to its ability to form filaments and networks, which is tightly controlled through numerous actin-modifying proteins (1, 2). These include proteins involved in polymerization and depolymerization of actin filaments, nucleators, and severing proteins as well as proteins involved in organizing filaments such as bundling proteins or filament crosslinkers (3, 4). Despite the wealth of knowledge about actin regulation, there are many outstanding questions about how it participates in the different cellular processes that it controls. Small molecules that target actin have been very useful and successful in understanding the different cellular roles of actin (5, 6). These small molecules are mostly derived from nature, and include compounds that inhibit actin polymerization such as cytochalasin and latrunculin (7), as well as F-actin stabilizers jasplakinolide (Fig. 1A) and phalloidin (8). Some, such as cytochalasin, have been instrumental in our understanding of F-actin polymerization and regulation. Indeed, cytochalasin was used to show that the same protein, actin, was involved in different processes such as cell migration, ruffling and division and revealed that actin polymerization in the lamellipodium occurred at the tip of the leading edge (6, 9). Phalloidin, which is not cell-permeable, selectively binds to F-actin and is a major tool in actin imaging in fixed cells.
Figure 1.
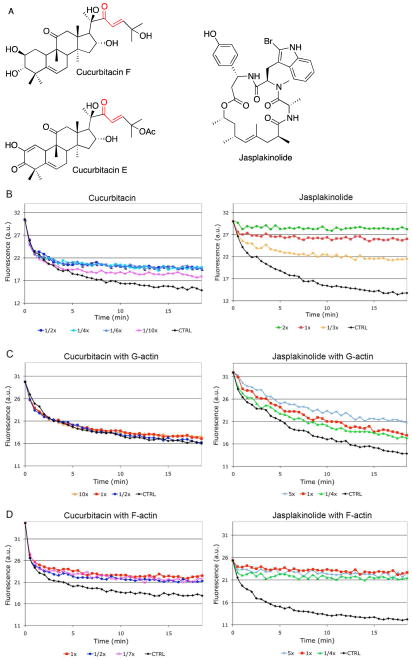
(A) Chemical structures of cucurbitacin F, cucurbitacin E, and jasplakinolide. The Michael acceptor on the cucurbitacins is colored red.
(B) Cucurbitacin E inhibits actin depolymerization at substoichiometric concentrations. The graphs show decrease in F-actin as a function of time as measured by the fluorescence signal emitted by pyrene-actin. (left panel) After a short initial depolymerization phase, cucurbitacin E at 1/2x, 1/4x, and 1/6x the actin concentration (i.e. 1.5μM, 0.75μM, and 0.5μM) inhibits F-actin depolymerization (3μM). At lower concentrations cucurbitacin E has a dose-dependent effect. (right panel) Jasplakinolide inhibits depolymerization at higher actin:compound ratios than cucurbitacin E. Jasplakinolide concentrations at 2x, 1x, and 1/3x the actin concentration are shown. A representative example of ten independent experiments for both drugs is shown.
(C, D) Cucurbitacin E affects F- but not G-actin. The graphs show decrease in F-actin as a function of time as measured by the fluorescence signal emitted by pyrene-actin. (C, left panel) Cucurbitacin E does not inhibit depolymerization of F-actin in a pyrene-actin assay if F-actin was formed from G-actin that has been pre-incubated with cucurbitacin E and then washed prior to F-actin formation. (D, left panel) On the contrary, when cucurbitacin E is incubated with F-actin directly, it inhibits depolymerization. Cucurbitacin E concentrations were 1/7x, 1/2x, 1x, and 10x the actin concentration. (C, right panel) The same experiment repeated with jasplakinolide and G-actin and (D, right panel) jasplakinolide and F-actin. Jasplakinolide concentrations were 1/4x, 1x, and 5x the actin concentration. A representative example of ten independent experiments for both drugs is shown for each panel.
We discovered cucurbitacin F (Fig. 1A), isolated from an extract of the plant Physocarpus capitatus, in a screen for small molecule inhibitors of cell division and observed that it aggregates actin (10, 11). Many plants make cucurbitacins, triterpenoid compounds originally identified as the bitter components of the Cucurbit family (12). All cucurbitacins share the same tetracyclic scaffold, but have varying substituents (cucurbitacin A–T) and are also often glycosylated. Although they are toxic, cucurbitacins have been used as traditional medicines, for example, the juice of the squirting cucumber plant (Ecbalium elaterium) may have anti-inflammatory properties (13). More recently, cucurbitacins have been reported to be antitumor agents and to inhibit the JAK-STAT pathway (14). It has been well documented that different cucurbitacins cause actin aggregation, although the underlying cause for this phenotype is the subject of some debate (15–17). Here, we report that cucurbitacin E stabilizes F-actin filaments and inhibits actin depolymerization in vitro and in cells through a novel mechanism.
RESULTS AND DISCUSSION
To understand the striking effect of cucurbitacins on actin morphology in cells, we first evaluated its effects on actin polymerization and depolymerization. Because the commercially available cucurbitacin E had been reported to have a similar phenotype to the one we observed in cells treated with cucurbitacin F (15), we used cucurbitacin E in the studies described here. The most common way to evaluate the polymerization state of actin and its perturbations by proteins or small molecules is a pyrene-actin assay (18). In this assay, purified actin is conjugated to a pyrene fluorophore, which is quenched by the interaction with solvent molecules while actin is monomeric (G-actin). When actin polymerizes into F-actin, the fluorophore becomes less accessible and begins to fluoresce, resulting in an increase in the fluorescence signal that can be detected. As has been reported (16), cucurbitacin E had no effect on actin polymerization, even at high concentrations, while cytochalasin D decreased polymer formation, and the actin stabilizer jasplakinolide accelerated polymer formation (Fig. S1).
To evaluate cucurbitacin E’s effect on pyrene-actin depolymerization, we pre-formed F-actin as described above, and measured depolymerization as a decrease in pyrene fluorescence. Depolymerization can be induced by dilution or by addition of monomer sequesterers, such as vitamin D binding protein, DNAse1 or latrunculin (19). Addition of monomer sequesterers maintains the actin monomer concentration at zero so that depolymerization alone can be studied. Unlike in dilution experiments, addition of monomer sequesterers to induce depolymerization allows continuous monitoring of fluorescence within the same fluorescence range (i.e. polymerization and depolymerization in the same experiment). We therefore chose addition of vitamin D binding protein as our main assay, and observed similar results with DNase1 and latrunculin. In good agreement with a previous report that used a dilution-based depolymerization assay (16), we found that cucurbitacin E inhibits actin depolymerization. Cucurbitacin E’s mode of inhibition of actin depolymerization differed from other small molecule inhibitors (i.e. phalloidin and jasplakinolide). Both of these compounds completely and immediately inhibit depolymerization. Cucurbitacin E, in contrast, does not affect the rate of depolymerization during the first two minutes, even if the small molecule is pre-incubated with F-actin for several hours (Fig. 1B). This could explain why a study using dilution-induced depolymerization assays with cucurbitacin I did not report inhibition of actin depolymerization (17). Subsequent inhibition of depolymerization is robust and sustained over hours, suggesting that cucurbitacin E stabilizes a subset of F-actin structures.
Sub-stoichiometric amounts of cucurbitacin E relative to actin stabilize actin filaments. Depolymerization is inhibited at a similar rate when actin is incubated with cucurbitacin E up to 1:6 times the actin concentration (i.e. 0.5 μM cucurbitacin E, relative to an actin concentration of 3 μM) (Fig. 1B, left panel). At ratios below 1:6 the effect tapers off dose-dependently, but even at 1:15 relative to actin cucurbitacin E has a stabilizing effect. In contrast, jasplakinolide requires higher compound:actin ratios to inhibit depolymerization. In the pyrene assays, it strongly stabilizes filaments at equal concentrations and has significant, dose-dependent effects at concentrations up to 1:3 (Fig. 1B, right panel), which is consistent with the fact that jasplakinolide binds to the interface of three actin subunits (20). Cucurbitacin E stabilizes actin filaments with different kinetics and at lower ratios than jasplakinolide, suggesting that it has a different mechanism of action.
Because cucurbitacin E inhibits actin depolymerization at substoichiometric concentrations, we hypothesized that it might act on fully formed filaments or a subset of filaments and investigated binding of cucurbitacin E to G- and/or F-actin. We prepared samples of either G- or F-actin and incubated with various concentrations of cucurbitacin E. Excess small molecule was then washed out by spinning repeatedly through a membrane with a molecular weight cut-off of 30kDa (G-actin has a molecular weight of ~42 kDa). When G-actin was pretreated with cucurbitacin E, washed, and then allowed to polymerize, the subsequent depolymerization occurred at the same rate for samples with or without compound, suggesting that cucurbitacin E does not bind to G-actin (Fig. 1C, left panel). On the other hand, the corresponding experiment with F-actin results in inhibition of depolymerization at similar cucurbitacin E:actin ratios as seen in previous experiments (Fig. 1D, left panel). Jasplakinolide at higher concentrations retains some ability to inhibit depolymerization when it is pre-incubated with G-actin (Fig. 1C, right panel), possibly because it can nucleate small clusters of actin trimers that cannot be removed by washing. It also inhibits depolymerization when incubated with F-actin (Fig. 1D, right panel). These data show that cucurbitacin E’s effect on actin is a result of action on actin filaments rather than actin monomers and provides further evidence for a unique mechanism of action.
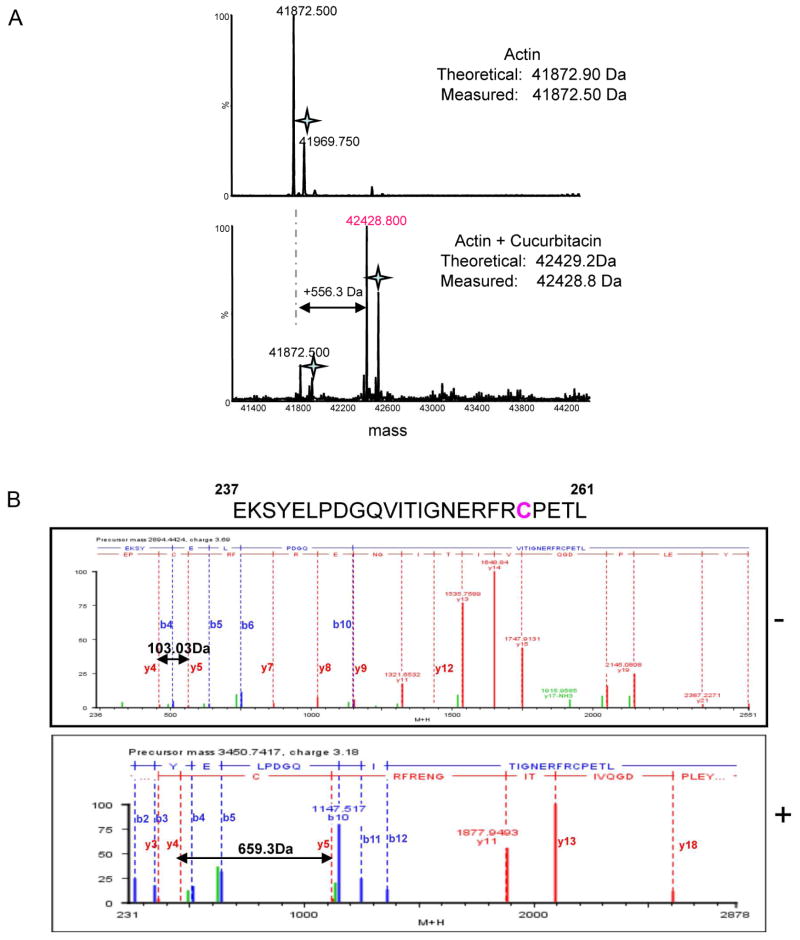
Cucurbitacins include an electrophilic Michael acceptor group (colored red in Fig. 1A), which can form a covalent bond with a nucleophile, for example, a cysteine, on its target protein. Rabbit skeletal muscle actin includes five cysteines, at positions 10, 217, 257, 285, and 374, which are possible candidates for a reaction with cucurbitacin E. In support of such covalent linkage, cucurbitacin E’s effects on cells are irreversible. HeLa cells treated with cucurbitacin E, followed by washout in drug-free media for 20 hrs, do not recover their pre-drug actin phenotype (Fig. 2). We used mass spectrometry to test if we could detect a covalent bond, which would be indicated by a cucurbitacin E-linked residue in actin incubated with cucurbitacin E. To increase the experimental range and to account for variations in commercial preparations, we conducted these experiments with highly related bovine, chicken, and rabbit actins. In all three cases we observed covalent adducts between actin and cucurbitacin E and, after enzymatic digestion with pepsin, we observed peptides where cucurbitacin E was linked to Cys257 (Fig. 3). With chicken actin, we observed an additional peptide where cucurbitacin E was also linked to Cys285 (Fig. S2A). We never saw binding to any other cysteine residue, including Cys374, an exposed residue that is often the first to be modified when actin is reacted with an electrophile, for example, during the synthesis of pyrene-actin (21) (Fig. S2B). We used high concentrations of cucurbitacin E (100x actin) in order to obtain a sufficiently high ratio of peptides with labeled cysteine residues that can be detected by mass spectrometry. This can be explained by cucurbitacin E’s substoichiometric action, suggesting that only a subset of actin monomers within filaments are affected at physiological conditions. However, the interaction between Cys257 and cucurbitacin E is clearly not random because Cys257 was covalently modified while other, more accessible cysteines, were not affected.
Figure 2.
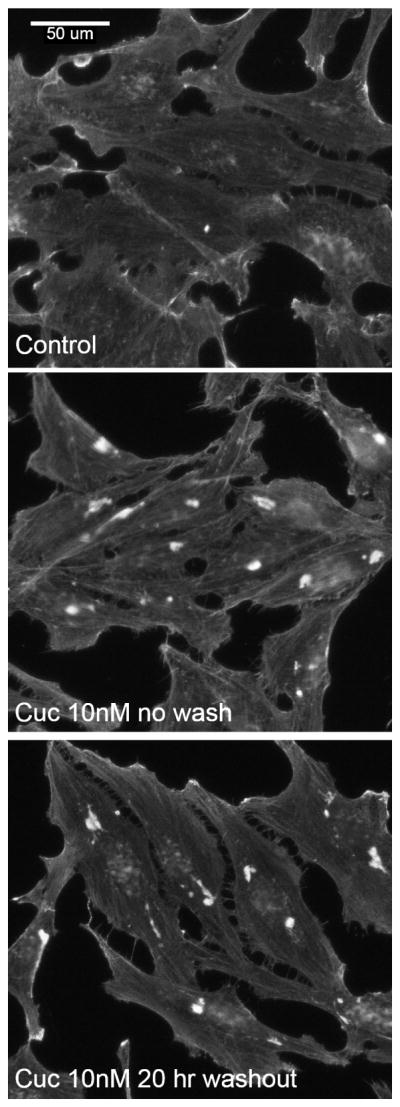
Cucurbitacin E’s effect on actin in cells is irreversible. HeLa cells treated with cucurbitacin E for 4 hrs, followed by subsequent washout of the drug for 20 hrs (bottom panel) exhibit the same phenotype as cells where the drug has not been washed out (middle panel). Scale bar = 50μm
Figure 3.
Mass spectrometric analysis of actin modification by cucurbitacin E shows covalent binding to Cys257. (A) Intact ESI mass spectra of rabbit skeletal muscle actin treated with DMSO (top) or cucurbitacin E (bottom). The transformed mass spectra are shown, and the measured and theoretical molecular weights of unmodified and modified actin are indicated. Addition of one cucurbitacin E molecule increases the molecular weight of the protein by 556.30 Da. Peaks corresponding to a phosphate adduct are indicated with a star. (B) Tandem MS analysis of pepsin digest of modified actin identified Cys257 as the main site of modification, thus verifying covalent bond formation between cucurbitacin E and actin. ESI-MS/MS spectra of the actin peptic peptide 237–261 ([M+H]+ = 2894.4 Da) alone (upper panel) and covalently modified ([M+H]+ = 3450.7 Da) (lower panel). The mass difference between fragment ions y4 and y5 in the untreated actin sample (upper panel, red color) shows the unmodified Cys257. The mass difference between fragment ions y4 and y5 in the modified actin (lower panel, red color) indicates that Cys257 is the site of covalent attachment by cucurbitacin E on actin.
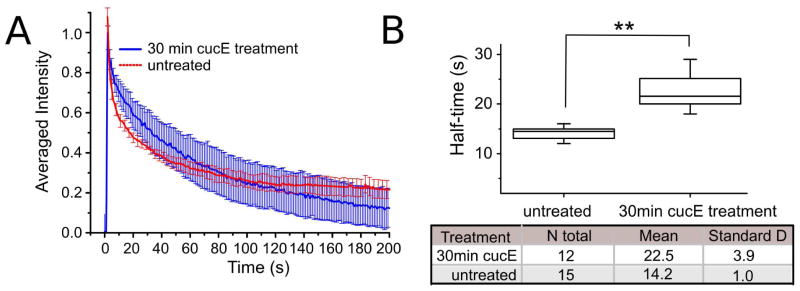
Cucurbitacin E stabilizes actin filaments by a covalent mechanism in vitro. For a small molecule to be broadly useful as a biological probe, its effects on cells need to be correlated to biochemical results. To investigate the impact of cucurbitacin E on depolymerization within the cellular actin network, we examined loss of fluorescence after localized photoactivation of PAGFP-actin. A similar assay has been used successfully to show that jasplakinolide shifts the equilibrium between G- and F-actin towards filaments (22). We transiently transfected HeLa cells with a plasmid that expressed actin tagged with a photo-activatable GFP and RFP (the latter as a background comparison to the varying GFP signal). After photo-activation, we monitored GFP fluorescence loss, which reports on actin depolymerization in the presence and absence of 10 nM cucurbitacin E. In samples treated with cucurbitacin E for 30 min, fluorescence loss was slowed 1.6 fold (halftime increased from 14 ± 1 s in control samples to 22.5 ± 4 s in cucurbitacin E treated samples, N>12 cells for each, p < 0.01, Fig. 4). We conclude that cucurbitacin E not only slows down actin depolymerization dynamics in vitro, but also in cells.
Figure 4.
Cucurbitacin E inhibits actin depolymerization in living cells. (A) Photoactivation experiments show a decrease in fluorescence decay in the presence of 10 nM cucurbitacin E. After 30min cucurbitacin E treatment (blue trace), the graphs show significant decrease of 1.6 fold in actin depolymerization relative to control (red trace).
(B) Cucurbitacin E treatment results in a reduction of the decay halftime from 14 ± 1 s to 22.5 ± 4 s (p < 0.01, N=12).
Our biochemical data suggest that cucurbitacin E and jasplakinolide have different mechanisms of action, likely by binding to different regions of actin filaments. To test this in cells, we analyzed and compared their effects at different concentrations and time points (Figs. S3 and S4). Consistent with different mechanisms of action, jasplakinolide and cucurbitacin E’s effects on cells are not identical. Both small molecules’ phenotypes vary considerably with concentration and length of exposure. At the minimum concentration at which we can observe reproducible effects in HeLa cells (10 nM for both), small star-shaped aggregates are formed after a few minutes (Fig. S3A, top panel). These aggregates remain mostly amorphous after 4 hours of jasplakinolide treatment, but coalesce into large aggregates with cucurbitacin E treatment (Fig. S3A, middle panel). Cucurbitacin E depletes actin from the cortex and from stress fibers more efficiently, especially after longer treatments, where cellular actin structures have been dissolved and we can only visualize actin in cucurbitacin E-induced aggregates (Fig. S3A, lower panel). We used actin antibodies to visualize actin in these experiments because jasplakinolide binds to F-actin competitively with phalloidin, suggesting it has the same or a closely situated binding pocket (8). Therefore phalloidin cannot bind to filaments where its binding sites have been saturated with jasplakinolide (Fig. S4, note the absence of phalloidin staining in the panel on the bottom right). Cucurbitacin E treatment has no effect on phalloidin staining, which shows that cucurbitacin E does not compete with jasplakinolide or phalloidin for the same binding site and therefore has a previously undescribed mechanism of stabilizing actin filaments in cells.
Cucurbitacin E’s binding mode to actin filaments is characterized by two unique features: it functions at substoichiometric concentrations and it inhibits depolymerization of actin filaments only after initial depolymerization occurs. These data both support a model where cucurbitacin E binds to a subset of actin filaments or a specific region within filaments. This can explain why we see an initial burst of depolymerization after cucurbitacin E treatment in pyrene-actin assays (non cucurbitacin-bound regions/filaments depolymerize), followed by an inhibition of depolymerization (cucurbitacin-bound regions/filaments are stabilized).
A high-resolution structure of F-actin has never been solved, so it is not possible to use modeling to predict a potential binding pocket for cucurbitacin E on F-actin, even though we know one of the residues cucurbitacin E interacts with. Several lower resolution structures of F-actin exist, and recent reports suggest that actin filaments may exist in many different conformations (23–26). The multitude of F-actin conformations suggests that cooperative and allosteric properties of actin play an important role for cellular function (27, 28). It has recently been proposed that the extremely high degree of sequence conservation for all actin residues across many different species, including residues that are deeply buried, may be due to the mechanical and conformational properties required for actin filaments’ functions (29). It is possible that cucurbitacin E binds to specific conformations of F-actin.
Several structures of G-actin have been solved, including G-actin bound to different actin binding proteins. Cys257, the residue cucurbitacin E binds to, appears to be deeply buried in all of these structures. There is a large body of historical work on the reactivities of different cysteines in actin, which is mostly focused on Cys10 and Cys374. Cys257 was generally not found to be very reactive, although one study reports that Cys257 is labeled preferentially with 7-dimethylamino-4-methyl-(N-maleimidyl) coumarin (30). One of the ways in which cells regulate actin dynamics is by cycling actin between ATP- and ADP-bound states. It has been suggested that Cys257 may be more reactive in nucleotide-free G-actin (31). Since cucurbitacin E does not bind to G-actin, we cannot test this directly, but it is possible that cucurbitacin E binds to and stabilizes specific regions in F-actin that have distinct nucleotide states. In cells, actin disassembly is accelerated by the concerted effort of several proteins, including members of the ADF/cofilin family, Aip1, and coronin (32). ADF/cofilin proteins, for example, promote disassembly both by severing of the actin polymer and by increasing the off-rate of actin monomers from the filament (33). If cucurbitacin E acts by stabilizing regions within actin filaments, depolymerization of filaments in cells would be affected, but perhaps not severing.
We report that cucurbitacin E binds to and stabilizes F-actin, without affecting actin polymerization or nucleation. Unlike jasplakinolide, it is compatible with phalloidin staining to visualize actin filaments in cells. Also unlike jasplakinolide, a natural product derived from a rare marine sponge in limited supply, cucurbitacin E is a widely available plant-derived natural product. We therefore propose that cucurbitacin E is a very useful research tool to study processes that involve actin dynamics.
Because actin is involved in so many different cellular processes, actin binders have traditionally been thought to be too toxic to be developed as potential therapeutics for anti-cancer chemotherapy. Different cucurbitacin E derivatives, however, have been described as potential anti-cancer drugs (34), suggesting that their mechanism of actin binding may be less toxic to cells and may therefore be worth exploring in a clinical setting.
Supplementary Material
Acknowledgments
We thank the staff at the Nikon Imaging Center at Harvard Medical School for assistance and HY Kueh and T. Mitchison for helpful discussions. Cucurbitacin F was originally isolated from plant extracts by K. Maloney and M. Fujita in J. Clardy’s lab. PS and UE were supported by NIH grant R01 GM082834, the Harvard Chemical Biology PhD program, and the Dana-Farber Cancer Institute. RI and JE acknowledge NIH grant R01 GM086507 and a research collaboration with the Waters Corporation. GC and MF gratefully acknowledge funding from the Human Frontier Science Program through a Young Investigator grant to GC and the UCL Comprehensive Biomedical Research Centre for generous funding of microscopy equipment. GC is a Royal Society University Research Fellow.
Footnotes
This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Siripala AD, Welch MD. SnapShot: actin regulators II. Cell. 2007;128:1014. doi: 10.1016/j.cell.2007.02.021. [DOI] [PubMed] [Google Scholar]
- 2.Siripala AD, Welch MD. SnapShot: actin regulators I. Cell. 2007;128:626. doi: 10.1016/j.cell.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 3.Campellone KG, Welch MD. A nucleator arms race: cellular control of actin assembly. Nat Rev Mol Cell Biol. 11:237–251. doi: 10.1038/nrm2867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Winder SJ. Structural insights into actin-binding, branching and bundling proteins. Curr Opin Cell Biol. 2003;15:14–22. doi: 10.1016/s0955-0674(02)00002-9. [DOI] [PubMed] [Google Scholar]
- 5.Atilla-Gokcumen GE, Castoreno AB, Sasse S, Eggert US. Making the cut: the chemical biology of cytokinesis. ACS Chem Biol. 2010;5:79–90. doi: 10.1021/cb900256m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peterson JR, Mitchison TJ. Small molecules, big impact: a history of chemical inhibitors and the cytoskeleton. Chem Biol. 2002;9:1275–1285. doi: 10.1016/s1074-5521(02)00284-3. [DOI] [PubMed] [Google Scholar]
- 7.Spector I, Shochet NR, Kashman Y, Groweiss A. Latrunculins: novel marine toxins that disrupt microfilament organization in cultured cells. Science. 1983;219:493–495. doi: 10.1126/science.6681676. [DOI] [PubMed] [Google Scholar]
- 8.Bubb MR, Senderowicz AM, Sausville EA, Duncan KL, Korn ED. Jasplakinolide, a cytotoxic natural product, induces actin polymerization and competitively inhibits the binding of phalloidin to F-actin. J Biol Chem. 1994;269:14869–14871. [PubMed] [Google Scholar]
- 9.Forscher P, Smith SJ. Actions of cytochalasins on the organization of actin filaments and microtubules in a neuronal growth cone. The Journal of Cell Biology. 1988;107:1505–1516. doi: 10.1083/jcb.107.4.1505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Eggert US, Kiger AA, Richter C, Perlman ZE, Perrimon N, Mitchison TJ, Field CM. Parallel chemical genetic and genome-wide RNAi screens identify cytokinesis inhibitors and targets. PLoS Biol. 2004;2:e379. doi: 10.1371/journal.pbio.0020379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maloney KN, Fujita M, Eggert US, Schroeder FC, Field CM, Mitchison TJ, Clardy J. Actin-Aggregating Cucurbitacins from Physocarpus capitatus. J Nat Prod. 2008 doi: 10.1021/np8005259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen JC, Chiu MH, Nie RL, Cordell GA, Qiu SX. Cucurbitacins and cucurbitane glycosides: structures and biological activities. Nat Prod Rep. 2005;22:386–399. doi: 10.1039/b418841c. [DOI] [PubMed] [Google Scholar]
- 13.Raikhlin-Eisenkraft B, Bentur Y. Ecbalium elaterium (squirting cucumber)--remedy or poison? J Toxicol Clin Toxicol. 2000;38:305–308. doi: 10.1081/clt-100100936. [DOI] [PubMed] [Google Scholar]
- 14.Graness A, Poli V, Goppelt-Struebe M. STAT3-independent inhibition of lysophosphatidic acid-mediated upregulation of connective tissue growth factor (CTGF) by cucurbitacin I. Biochem Pharmacol. 2006;72:32–41. doi: 10.1016/j.bcp.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 15.Duncan KL, Duncan MD, Alley MC, Sausville EA. Cucurbitacin E-induced disruption of the actin and vimentin cytoskeleton in prostate carcinoma cells. Biochem Pharmacol. 1996;52:1553–1560. doi: 10.1016/s0006-2952(96)00557-6. [DOI] [PubMed] [Google Scholar]
- 16.Momma K, Masuzawa Y, Nakai N, Chujo M, Murakami A, Kioka N, Kiyama Y, Akita T, Nagao M. Direct interaction of Cucurbitacin E isolated from Alsomitra macrocarpa to actin filament. Cytotechnology. 2008;56:33–39. doi: 10.1007/s10616-007-9100-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Knecht DA, LaFleur RA, Kahsai AW, Argueta CE, Beshir AB, Fenteany G. Cucurbitacin I inhibits cell motility by indirectly interfering with actin dynamics. PLoS One. 2010;5:e14039. doi: 10.1371/journal.pone.0014039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cooper JA, Walker SB, Pollard TD. Pyrene actin: documentation of the validity of a sensitive assay for actin polymerization. J Muscle Res Cell Motil. 1983;4:253–262. doi: 10.1007/BF00712034. [DOI] [PubMed] [Google Scholar]
- 19.Lees A, Haddad JG, Lin S. Brevin and vitamin D binding protein: comparison of the effects of two serum proteins on actin assembly and disassembly. Biochemistry. 1984;23:3038–3047. doi: 10.1021/bi00308a030. [DOI] [PubMed] [Google Scholar]
- 20.Heidecker M, Yan-Marriott Y, Marriott G. Proximity relationships and structural dynamics of the phalloidin binding site of actin filaments in solution and on single actin filaments on heavy meromyosin. Biochemistry. 1995;34:11017–11025. doi: 10.1021/bi00035a007. [DOI] [PubMed] [Google Scholar]
- 21.Kouyama T, Mihashi K. Fluorimetry study of N-(1-pyrenyl)iodoacetamide-labelled F-actin. Local structural change of actin protomer both on polymerization and on binding of heavy meromyosin. Eur J Biochem. 1981;114:33–38. [PubMed] [Google Scholar]
- 22.Kiuchi T, Nagai T, Ohashi K, Mizuno K. Measurements of spatiotemporal changes in G-actin concentration reveal its effect on stimulus-induced actin assembly and lamellipodium extension. The Journal of cell biology. 2011;193:365–380. doi: 10.1083/jcb.201101035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Oda T, Iwasa M, Aihara T, Maeda Y, Narita A. The nature of the globular- to fibrous-actin transition. Nature. 2009;457:441–445. doi: 10.1038/nature07685. [DOI] [PubMed] [Google Scholar]
- 24.Oda T, Maeda Y. Multiple Conformations of F-actin. Structure. 2010;18:761–767. doi: 10.1016/j.str.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 25.Galkin VE, Orlova A, Schroder GF, Egelman EH. Structural polymorphism in F-actin. Nat Struct Mol Biol. 2010;17:1318–1323. doi: 10.1038/nsmb.1930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kueh HY, Brieher WM, Mitchison TJ. Dynamic stabilization of actin filaments. Proc Natl Acad Sci U S A. 2008;105:16531–16536. doi: 10.1073/pnas.0807394105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Egelman EH, Orlova A. Allostery, cooperativity, and different structural states in F-actin. J Struct Biol. 1995;115:159–162. doi: 10.1006/jsbi.1995.1040. [DOI] [PubMed] [Google Scholar]
- 28.Papp G, Bugyi B, Ujfalusi Z, Barko S, Hild G, Somogyi B, Nyitrai M. Conformational changes in actin filaments induced by formin binding to the barbed end. Biophys J. 2006;91:2564–2572. doi: 10.1529/biophysj.106.087775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Galkin VE, Orlova A, Egelman EH. Actin filaments as tension sensors. Curr Biol. 2012;22:R96–R101. doi: 10.1016/j.cub.2011.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu DF, Wang D, Stracher A. The accessibility of the thiol groups on G- and F-actin of rabbit muscle. Biochem J. 1990;266:453–459. doi: 10.1042/bj2660453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kabsch W, Vandekerckhove J. Structure and function of actin. Annu Rev Biophys Biomol Struct. 1992;21:49–76. doi: 10.1146/annurev.bb.21.060192.000405. [DOI] [PubMed] [Google Scholar]
- 32.Insall RH, Machesky LM. Actin dynamics at the leading edge: from simple machinery to complex networks. Dev Cell. 2009;17:310–322. doi: 10.1016/j.devcel.2009.08.012. [DOI] [PubMed] [Google Scholar]
- 33.Andrianantoandro E, Pollard TD. Mechanism of actin filament turnover by severing and nucleation at different concentrations of ADF/cofilin. Mol Cell. 2006;24:13–23. doi: 10.1016/j.molcel.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 34.Lee DH, Thoennissen NH, Goff C, Iwanski GB, Forscher C, Doan NB, Said JW, Koeffler HP. Synergistic effect of low-dose cucurbitacin B and low-dose methotrexate for treatment of human osteosarcoma. Cancer Lett. 2011;306:161–170. doi: 10.1016/j.canlet.2011.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.