Abstract
The activity of calcium/calmodulin-dependent protein kinase IV (CaMK4) is increased in T cells from patients with SLE and has been shown to reduce IL-2 production by promoting the effect of the transcriptional repressor cAMP responsive element modulator (CREM)-α on the IL2 promoter. Here we demonstrate that T cells from MRL/lpr mice display increased levels of CaMK4 in the nucleus and that genetic deletion of Camk4 results in improved survival. We demonstrate that absence of CaMK4 restores IL-2 production, curbs increased T cell activation, and augments the number and activity of regulatory T cells. Analogously, silencing of CaMK4 in T cells from patients with SLE increases the expression of FoxP3 upon stimulation in the presence of TGF-β. Our results demonstrate the importance of the serine/threonine kinase CaMK4 in the generation and function of regulatory T cells in patients with SLE and lupus-prone mice and its potential to serve as a therapeutic target.
INTRODUCTION
The hallmark of systemic lupus erythematosus (SLE) is the development of a chronic autoimmune response driven towards ubiquitous, mostly intracellular, auto-antigens (1). Both B and T cells participate in this pathologic response (2) that causes diverse clinical manifestations when activated lymphocytes or their products (e.g. autoantibodies, cytokines, etc.) enter tissues and cause inflammatory organ damage.
T cells from patients with SLE exhibit abnormal signaling upon TCR engagement and have an altered gene expression profile (3). Accordingly, the regulation of several transcription factors is distorted in SLE T cells upon activation. In particular, activation of cAMP response element-binding (CREB) and cAMP responsive element modulator (CREM)-α has been shown to be unbalanced. Increased activation of CREM-α, along with a reciprocal decrease in activated CREB, result in impaired IL-2 production by T cells from patients with SLE (4, 5). Calcium/calmodulin-dependent protein kinase IV (CaMK4), a serine/threonine kinase expressed in T cells (6), regulates the activity of several transcription factors including CREM (7). In vitro experiments have shown that T cells from patients with SLE have increased levels of activated CaMK4 that impairs IL-2 production by inducing augmented DNA binding of CREM-α to the IL2 promoter (8). Thus, T cells from patients with SLE have abnormal activation of CaMK4 that inhibits IL-2 production by facilitating the activity of CREM-α.
To determine whether increased activation of CaMK4 plays a pathogenic role in lupus in vivo, we treated MRL/lpr mice with a small molecule inhibitor of CaMK4 (KN-93). CaMK4 inhibition was able to suppress the development of glomerulonephritis and skin disease in MRL/lpr mice (9). Accordingly, genetic deletion of Camk4 in MRL/lpr mice reduced disease progression (10). Importantly, we showed that mesangial cells from MRL/lpr mice proliferate at abnormally high levels and that Camk4 deficiency corrects this defect, suggesting that in a manner independent of its T cell effects, its activation in resident renal cells plays a role in determining local susceptibility to immune mediated injury (10).
In this communication we demonstrate that T cells from MRL/lpr mice express increased amounts of CaMK4 in the nucleus. Genetic deletion of Camk4 in MRL/lpr mice promotes the production of IL-2 and increases the activity of regulatory T cells accompanied by mitigation of clinical parameters and prolonged survival. Similarly, silencing of CaMK4 in SLE T cells increased the expression of FoxP3 upon stimulation in the presence of TGF-β.
MATERIALS AND METHODS
Mice
Female MRL/MpJ-Tnfrsf6lpr (MRL/lpr), B6.129X1-Camk4tm1Tch/J, MRL/MPJ, C57BL/6J and B6.MRL-Faslpr/J (B6.lpr) mice were purchased from The Jackson Laboratory. B6.lpr.Camk4−/− mice were generated by backcrossing B6.129X1-Camk4tm1Tch/J mice into C57BL6/J; these mice were backcrossed into MRL/lpr mice for 9 generations to obtain MRL/lpr.Camk4−/− mice. Animals were sacrificed at the end of their 8th or 16th week of age. Mice were maintained in a SPF animal facility (Beth Israel Deaconess Medical Center). Experiments were approved by the Institutional Animal Care Committee of Beth Israel Deaconess Medical Center.
Flow cytometry and determination of cell number
Spleen, and lymph nodes were excised from mice, and single-cell suspensions were obtained by teasing the organs through a nylon mesh. Isolated cells were stained for flow cytometry with antibodies against CD3e (17A2, eBioscience), CD4 (GK1.5, BioLegend), CD8a (53-6.7, eBioscience), CD11c (HL3, BD Pharmingen), CD19 (1D3, eBioscience), CD25 (PC61, eBioscience), CD44 (IM7, BD Pharmingen), CD62L (MEL14, BD Pharmingen), or F4/80 (BM8, eBioscience) for 30 min at 4 °C. For intracellular staining of Foxp3 (MF23, BD Biosciences) and IL-2 (JES6-5H4, BioLegend), the Foxp3-Staining Buffer Set (fixation/permeabilization and permeabilization buffers; eBioscience) and BD cytofix/cytoperm plus (with Golgi Plug™) intracellular staining kits (BD Biosciences) were used according to the manufacturer’s protocol. Total cell numbers were determined by counting live cells. Absolute cell numbers were calculated on the basis of the percentage of each population and represented as median.
Cell sorting
Cell sorting was performed in a FACS Aria II cell sorter (BD Biosciences). Splenocytes from MRL/lpr mice were isolated, and a minimum of 1 × 108 total splenocytes were incubated with antibodies against CD3, CD4, CD8, CD19, CD11c and F4/80 for 30 min at 4°C. Cells were sorted into six different populations, CD3+ CD4+ CD8−, CD3+ CD4−CD8+, CD3+ CD4− CD8−, CD3− CD19+, CD3− CD11c+ and CD3− F4/80+. The purity of the sorted cells populations ranged from 94 to >99%.
RNA isolation and real time reverse transcription polymerase chain reaction (real time PCR)
Total mRNA was isolated from spleen cells using the RNeasy Mini Kit (Qiagen). cDNA was produced using random primers from an equal amount of RNA. Real-time PCR for mouse Il2, mouse Camk4, mouse Gapdh, human FOXP3, and human Actb were performed (Light Cycler 480; Roche, Indianapolis, IN) with ABI Taqman Gene Expression assays (Applied Biosystems, Foster City, CA). Gene expression was assessed by comparative CT method.
Polyclonal activation and suppression assay
CD3+CD4+CD25− T cells (responder T cells; Tresp) and CD3+CD4+CD25+ T (regulatory T cells; Tregs) cells were isolated from spleens by FACS sorting. CD4+CD25− T cells were labeled with 0.5 μM CFSE (Invitrogen/Molecular Probes) for 10 min at 37°C. CFSE labeled CD4+CD25− T cells were cultured in 96-well round bottom plates at 2×105 cells/well in the presence of varying amounts of CD4+CD25+ T cells and stimulated with plate-bound goat anti-hamster Abs (MP Biomedicals), soluble anti-CD3 (0.25 μg/ml, clone 145-2C11; BioLegend) and anti-CD28 (0.5 μg/ml, clone 37.51; BioLegend). After 5 days, cells were harvested and stained for surface markers and CFSE signal of gated lymphocytes was analyzed by flow cytometry.
Isolation of Cells and Cell Culture
Naïve CD4 T cells were isolated by magnetic cell sorting (CD4+CD62L+ T cell isolation kit II; Miltenyi Biotec). Postsorting purity was >95%. Cells were stimulated in RPMI 1640 supplemented with 10% FBS, 2 mM glutamine, 50μM β-mercaptoethanol, 50 units/mL penicillin G, and 50 μg/mL streptomycin sulfate with plate-bound goat anti-hamster Abs, soluble anti-CD3 (0.25 μg/ml, clone 145-2C11) and anti-CD28 (0.5μg/ml, clone 37.51). In vitro Treg differentiation assays were performed by stimulating cells with plate-bound anti-CD3 (2.5μg/mL), soluble anti-CD28 (5 μg/mL), TGF-β1 (5 ng/ml, R&D Systems), anti-IFN-γ, anti-IL4 (both at 10 μg/mL, Biolegend). Additionally, human IL-2 (100 IU/mL) or anti-murine IL-2 (10 μg/mL, BioLegend) was added to some wells.
ELISA
Splenic naïve CD4+CD62+ T cells (5 × 105 cells/sample) were stimulated as described above. After 24h, IL-2 was measured in supernatants by ELISA (R&D Systems). Serum anti-dsDNA antibody concentration was detected by mouse anti-dsDNA IgG ELISA kit (Alpha Diagnostic).
Western blotting
Splenocytes of MRL/lpr or MRL/lpr.Camk4−/− mice were lysed in RIPA buffer at 4°C for 30 min. After centrifugation (14,000 rpm; 30 min; 4°C) supernatants were collected and nuclei were isolated using the Nuclear Extract Kit (Active Motif, Carlsbad, CA). An identical amount of protein from each lysate (5μg/well) was separated on NuPAGE 4–12% Bis-Tris Gel (Invitrogen). Proteins were transferred to a nitrocellulose or PVDF membrane, which was subsequently blocked for 1h using 2% bovine serum albumin (BSA) in phosphate buffered saline (PBS) and incubated at room temperature with anti-CaMK4 (Cell signaling), anti-CREM (IMGEX) anti-phospho-Ser271, anti-phospho-Ser274 CREM (Novus) and anti-actin (Sigma). The membrane was washed with TBS-T and incubated with a 1:3000 dilution of goat anti-rabbit IgG or donkey anti-goat IgG coupled with horseradish peroxidase (HRP; Jackson Immunoresearch, West Grove, PA, USA). The enhanced chemiluminescence (ECL) system (Amersham, Buckinghamshire, UK) was used for detection.
Human SLE T cells
T cells were obtained from the peripheral blood of patients with SLE as described previously under an Institutional Review Board-approved protocol and transfected with CAMK4-specific or control siRNA as described previously (8). After 24 hours, cells were stimulated with plate-bound goat anti-mouse Abs (Chemicon Millipore), soluble anti-CD3 (5μg/ml, clone OKT3; BioXcell), anti-CD28 (2.5μg/ml, clone 28.2; BioLegend) and TGF-β1 (5 ng/ml, R&D Systems) and cultured for 96 hr.
Statistical analyses
Student two-tailed t tests and Mann Whitney tests were used. Mouse survival was analyzed by the Kaplan-Meier method and the log rank test. Statistical analyses were performed in GraphPad Prism 5.01 software. A level of p<0.05 was considered significant.
RESULTS
CaMK4 is found in the nuclei of MRL/lpr cells
Activity of the serine/threonine kinase CaMK4 is abnormally increased in T cells from patients with SLE (8). Activation of CaMK4 induces its migration into the cell nucleus (7). Accordingly, higher levels of CaMK4 are found in the nuclei of T cells from patients with SLE than in healthy controls. Increased CaMK4 activity has been linked to a number of phenotypic abnormalities in SLE T cells, including increased activation of the transcriptional factor CREM-α (8) and decreased IL-2 production (8). Because IL-2 production by CD4 T cells is similarly decreased in MRL/lpr mice (11) that develop a lupus-like disease, we decided to investigate whether the expression and cellular localization of CaMK4 was abnormal in these mice, to determine if they represent a useful system to study the consequences of increased CaMK4 activity in systemic autoimmunity.
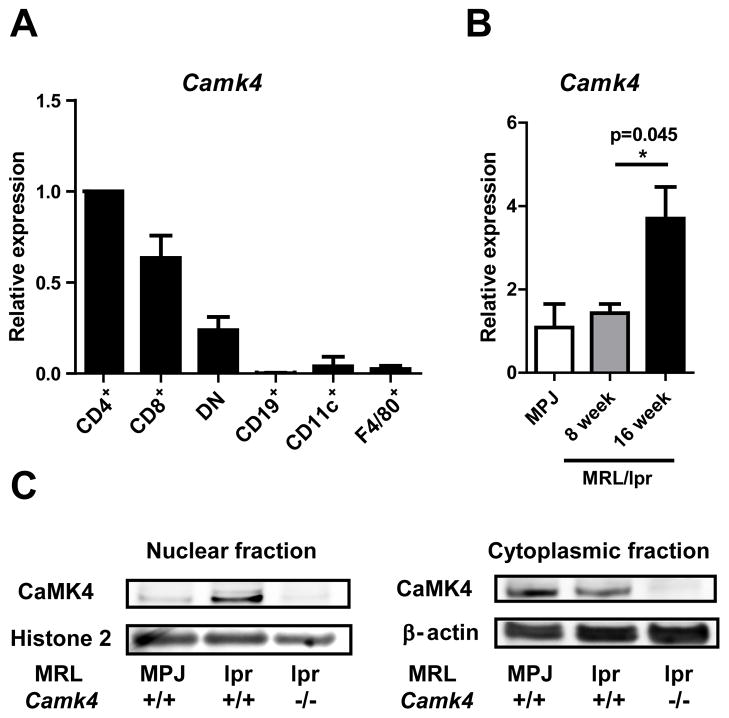
As shown in Figure 1A, high levels of Camk4-encoding transcripts were found in T lymphocytes, particularly CD4+ T cells, of MRL/lpr mice. In contrast, levels of Camk4 mRNA were scarce in spleen-derived B cells, dendritic cells, and macrophages. We compared the mRNA levels of Camk4 in spleen cells from 8 week old (before disease onset) and 16 week old (established disease) MRL/lpr mice. Camk4 expression in 8 week old MRL/lpr mice did not differ from that of the control strain MRL/MPJ. In contrast, 16 week old MRL/lpr mice had significantly higher levels of Camk4 measured by quantitative PCR (Figure 1B). Since activated CaMK4 migrates to the cell nucleus, we investigated its abundance in the cytosolic and nuclear fractions of spleen cells isolated from MRL/lpr, MRL/MPJ, and MRL/lpr.Camk4−/− mice. High levels of CaMK4 were detected in the nuclei of MRL/lpr spleen cells. This contrasted sharply with the cellular distribution of CaMK4 in spleen cells of the Fas-intact control strain MRL/MPJ where virtually all CaMK4 was found in the cytoplasmic fraction of the cell lysate (Figure 1C).
Figure 1. CaMK4 activity is increased in T cells from MRL/lpr mice.
A, Camk4 expression was quantified using real time PCR in RNA extracts from T cell subsets (CD4+, CD8+, CD4−CD8−), B cells (CD19+), dendritic cells (CD11c+), and macrophages (F4/80+) isolated by FACS sorting from 16 week old MRL/lpr mice. B, Camk4 expression was quantified by real time PCR in spleen cells from MRL/MPJ control mice and from 8 and 16 week old MRL/lpr mice. C, CaMK4 was detected by western blot in the nuclear and cytoplasmic fractions of spleen cell lysates from MRL/MPJ, MRL/lpr, and MRL/lpr.Camk4−/− mice. Data are representative of two independent experiments, each using four mice per group.
These results indicate that CaMK4 expression in the spleen is mainly restricted to T cells and demonstrate that, as in patients with SLE (8), CaMK4 levels and activity are increased in diseased MRL/lpr mice.
CaMK4 deficiency ameliorates disease and reduces mortality in MRL/lpr mice
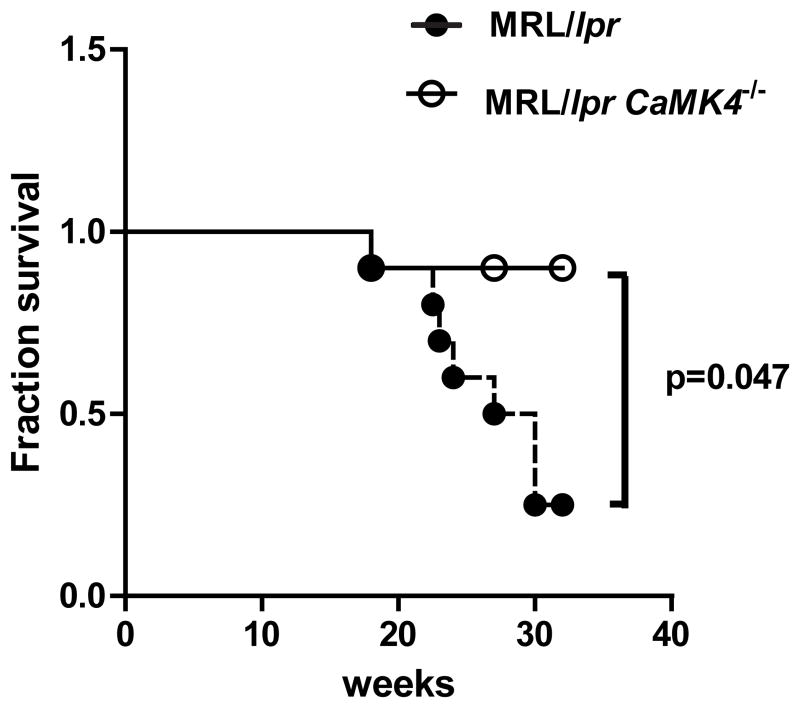
Pharmacological inhibition or genetic deletion of CaMK4 reduces autoantibody production and reduces progression of kidney disease in MRL/lpr mice (9, 10). We confirmed those results and found that the effect of Camk4 deficiency reduces the mortality of MRL/lpr mice in a statistically significant manner. The survival rate of MRL/lpr.Camk4−/− mice was 89% at 32 weeks of age compared to only 25% in the MRL/lpr group (P<0.05; Figure 2). As previously shown, the severity of the glomerulonephritis, as well as the levels of C3 deposits in the kidney and the extent of skin injury were notably decreased in MRL/lpr.Camk4−/− mice (Supplemental Figure 1).
Figure 2. CaMK4 deficiency prolongs survival of MRL/lpr mice.
Survival of MRL/lpr and MRL/lpr.Camk4−/− are depicted. Mice were observed until 32 weeks of age (n=10 mice per group).
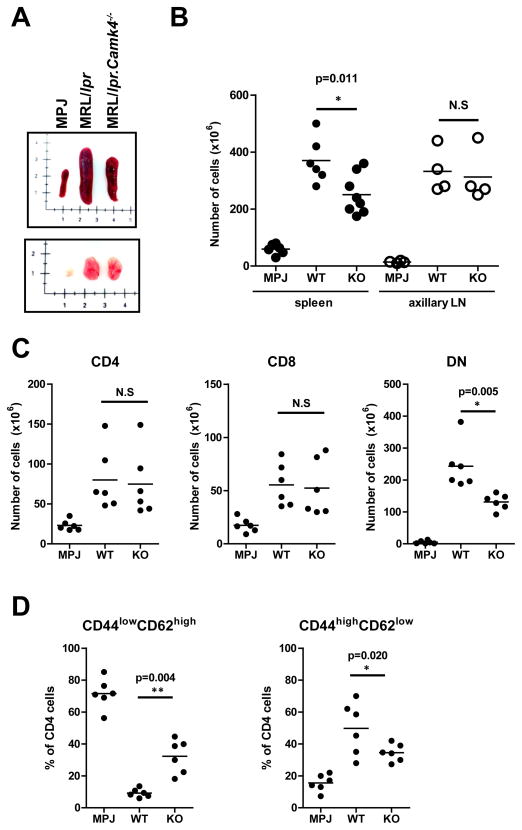
T and B cell activation and proliferation are prominently increased in MRL/lpr mice (12) and occur simultaneously to the development of autoantibodies and organ damage. In a previous work, we demonstrated that CaMK4 facilitates the proliferation of mesangial cells in the renal glomeruli of MRL/lpr mice (10). To determine if CaMK4 is also involved in the augmented lymphoid activation and proliferation of MRL/lpr mice, we analyzed the phenotype of different T and B cell subsets using multicolor flow cytometry. CaMK4 deficiency was associated with a significant decrease in spleen size and cellularity (P=0.01) in 16 week-old mice. In contrast, absence of CaMK4 did not affect in a statistically significant manner the size and total cell content of the axillary lymph nodes (Figure 3A and B). Importantly, the abundance of CD4−CD8− (double negative) T cells and activated CD4+ (CD44+ CD62Llo) T cells was significantly reduced in the absence of CaMK4 (Figure 3C and D). A reciprocal increase in naïve CD4+ (CD44− CD62Lhigh) T cell was likewise observed (Fig. 3D).
Figure 3. CaMK4 deficiency limits lymphoid cell activation and proliferation.
A, Representative photograph of the spleens and axillary lymph nodes from 16 week old MRL/MPJ, MRL/lpr and MRL/lpr.CaMK4−/− mice. B, Absolute numbers of spleen and axillary lymph node cells are shown. C, Absolute numbers of splenic CD4+, CD8+, and double negative (CD4−CD8−) T cells. D, Abundance of naïve (CD44lowCD62Lhigh) and activated (CD44highCD62Llow) CD4 T cells (shown as percentage of CD4+ T cells). All experiments were performed in 16 week old female mice. Each dot represents an individual mouse. Horizontal bars indicate mean for each group. Data are representative of 3 independent experiments with 2–5 mice per group.
These results suggest that CaMK4 is involved in the activation and proliferation of lymphoid cells in the MRL/lpr mice, because its deficiency decreases the numbers of activated cells.
CaMK4 deficiency restores IL-2 production by MRL/lpr CD4 T cells
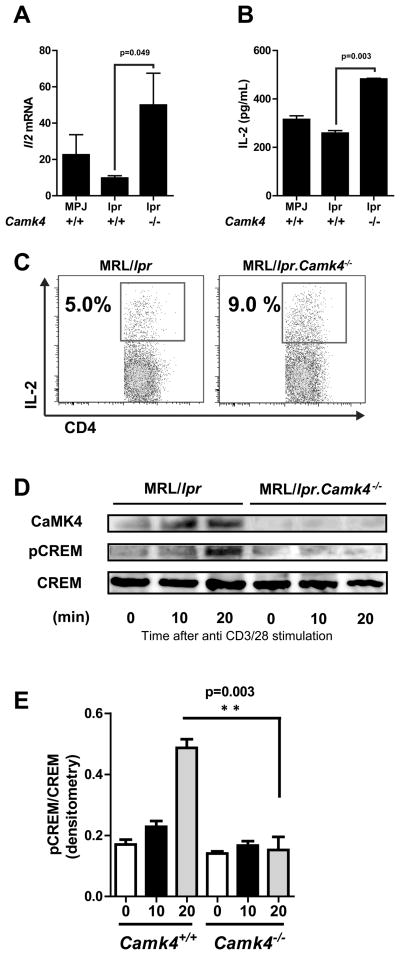
Transcription of Il2 upon T cell stimulation is abnormally decreased in patients with SLE (13, 14) and in MRL/lpr mice (15, 16). Although several molecular alterations have been linked to this phenotype (17), an imbalance between the repressor CREM and the enhancer CREB contributes to decreased IL2 transcription (4). Increased binding of CREM to the IL2 promoter has been shown to impair IL-2 production in T cells from patients with SLE in an analogous manner to its effects in anergic cells (4, 8). Since CaMK4 modulates the activity of CREM (4, 18), we hypothesized that Camk4 deficiency would affect the production of IL-2 by naïve CD4 T cells. As shown in Figure 4, absence of CaMK4 completely restored the production of IL-2 in MRL/lpr mice. This was evident at the mRNA and protein levels (ELISA and intracellular flow cytometry).
Figure 4. CaMK4 deficiency restores IL-2 production.
A, Naive CD4+ CD62L+ T cells were isolated from 16 week old MRL/MPJ, MRL/lpr and MRL/lpr.Camk4−/− mice and stimulated with anti CD3 and anti-CD28 during 24 hours. Il2 mRNA expression levels were measured by real time PCR. B, IL-2 was quantified by ELISA in supernatants of cells stimulated as in A. C, IL-2 production was measured in CD4+ T cells at the single-cell level in cells stimulated as in A by flow cytometry. D, Expression of CaMK4 and phosphorylated CREM detected by western blot analyses after stimulation of spleen cells with anti-CD3 and anti-CD28 during the indicated time points. Data are representative of 2 independent experiments with 4–5 mice/group *, P < 0.05.
To determine whether the effect on IL-2 production was exerted through CREM activation, we analyzed the phosphorylation of CREM in MRL/lpr and MRL/lpr.Camk4−/− mice following T cell activation. As shown in Figure 4D, phosphorylated CREM was detected in MRL/lpr cells after 20 minutes of activation with anti-CD3 and anti-CD28. In sharp contrast, no phosphorylated CREM could be detected in Camk4 null cells.
CaMK4 controls regulatory T cell differentiation and function
IL-2 is intimately linked to the maintenance and function of regulatory T cells (Treg) (19). In fact, mice deficient in IL-2 or in critical components of its receptor develop a lethal autoimmune disease caused by a lack of Tregs (20). Numerical and functional Treg deficiencies have also been described in patients with SLE (21) and Tregs from MRL/lpr mice have been reported to be functionally abnormal (22). We reasoned that the decrease in spontaneous T cell activation observed in the Camk4-deficient MRL/lpr mice could be associated with restored Treg function, perhaps facilitated by the rescue of the IL-2 production capacity.
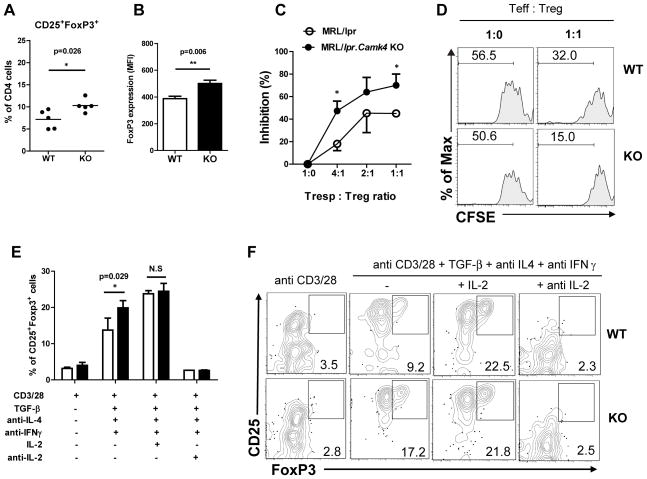
To address this, we quantified the abundance of Tregs in the spleens of MRL/lpr and MRL/lpr.Camk4−/− mice. Tregs (CD4+CD25+FoxP3+) were significantly increased in Camk4−/− mice when compared to Camk4-sufficient mice (10.3 ± 0.6% vs. 7.2 ± 0.9%, p<0.05) (Figure 5A). Moreover, expression of FoxP3 at the protein level was increased in Tregs from MRL/lpr.Camk4−/− mice compared to Tregs from MRL/lpr mice (Figure 5B). To determine if CaMK4 modulates the differentiation of Tregs from naïve CD4 T cells, we quantified induction of Tregs under different in vitro conditions. To this end, CD4+CD25− T cells isolated by FACS sorting were stimulated with anti-CD3 and anti-CD28 in the presence of TGF-β, anti-IFN-γ, and anti-IL-4 for 5 days. Cells activated in the absence of TGF-β served as controls. As shown in Figure 5E, Treg induction was significantly increased in cells isolated from MRL/lpr.Camk4−/− mice (Camk4+/+ 13.7% vs. Camk4−/− 19.9%, p<0.05). This effect was probably mediated through IL-2 since the addition of exogenous IL-2 to the culture was able to increase the yield of CD25+FoxP3+ T cells generated from Camk4-sufficient mice to the same level observed in cells from Camk4-deficient animals (Figure 5E).
Figure 5. CaMK4 regulates the generation and function of Tregs.
A, Tregs (CD25+ FoxP3+) were quantified in spleens of 16 week old MRL/lpr and MRL/lpr.Camk4−/−mice using flow cytometry. Results are expressed as percentage of CD4+ T cells. B, FoxP3 expression was analyzed as MFI in CD4+CD25+ cells from MRL/lpr and MRL/lpr.Camk4−/−. C, CD4+CD25+ T cells and CD4+CD25− T cells were isolated by FACS sorting. CFSE-stained CD4+CD25− T cells (Tresp, 1×105) were cultured with varying amounts of Tregs (CD4+CD25+) in the presence of anti-CD3 and anti-CD28 for 4 days. Proliferation was measured as percentage of cells that diluted CFSE. Results are expressed as the inhibition of proliferation relative to CD4+CD25− T cells stimulated in the absence of Tregs. C, Representative CFSE histograms are shown. D, CD4+CD25− T cells were stimulated with anti-CD3 and anti-CD28 in the presence of TGF-β (5 ng/ml), anti-IFN-γ, and anti-IL-4. IL-2 (100 iu/mL) or anti-IL-2 (10 μg/ml) was added to some wells. After 5 days the percentage of Tregs (CD25+FoxP3+ cells) was quantified by flow cytometry. Data is presented as mean + SD of 2 independent experiments, each with 3 mice per group. E, Representative dot plots are shown. Data are representative of 2 independent experiments with 3 mice per group *, P < 0.05
The suppressive capacity of Camk4-deficient and -sufficient Tregs was evaluated in in vitro co-culture assays. As shown in Figure 5C, Tregs from MRL/lpr.Camk4−/− mice suppressed more efficiently the proliferation of CD4+CD25− Camk4-sufficient T cells than Tregs from wild type mice.
Taken together, our results suggest that by rescuing IL-2 production, CaMK4 deficiency restores Treg numbers and function that curb spontaneous lymphoid activation.
Silencing of CaMK4 results in overexpression of Treg cells in patients with SLE
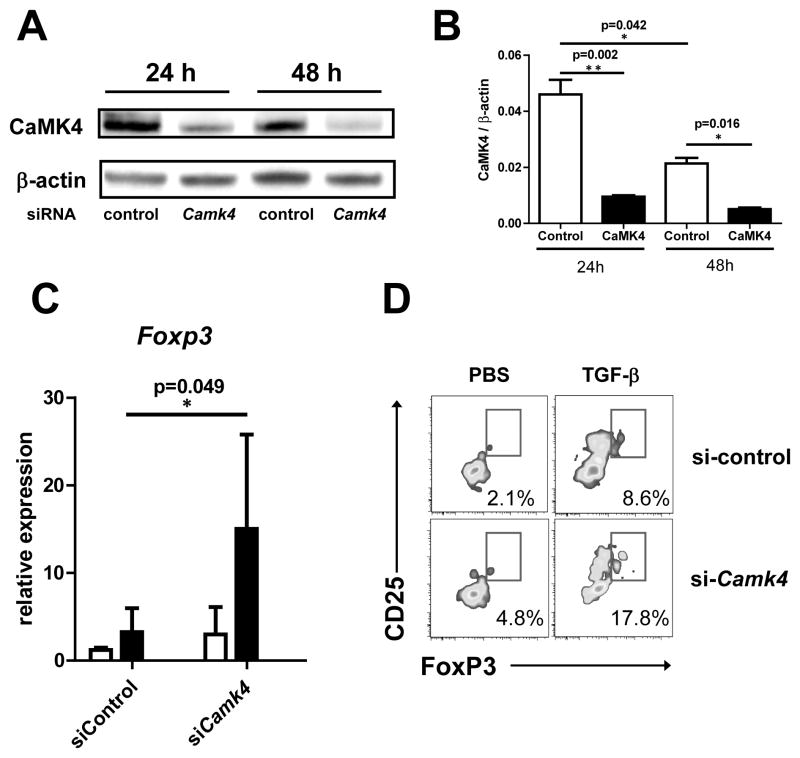
To determine the relevance of our findings to human SLE, we analyzed the effect of CaMK4 inhibition in T cells from patients with SLE. By using RNA interference we successfully knocked-down CaMK4 expression in T cells (Fig. 6Aand B). To determine the effect of CaMK4 inhibition in FoxP3 expression, we stimulated cells transfected with Camk4-specific of with control siRNA with anti-CD3, anti-CD28, and TGF-β. As shown in Figure 6, CaMK4 inhibition caused a significant increase in the expression of Foxp3 gene as determined by real time PCR (Fig. 6C). Accordingly, CaMK4 inhibition significantly augmented the percentage of CD25+FoxP3+ T cells (Fig. 6D). These results indicate that, as in MRL/lpr mice, CaMK4 is a negative regulator of Tregs in patients with SLE.
Figure 6. CaMK4 regulates FoxP3 in T cells from SLE patients.
A, T cells from 4 patients with SLE were transfected with either control siRNA or CAMK4-specific siRNA. A representative image of CaMK4 expression (western blot) at 24 and 48 hr after transfection is shown in the left panel. The right panel shows cumulative data from 4 experiments. B, 24 hr after transfection cells were either left unstimulated (white bars) or stimulated with anti-CD3 and anti-CD28 plus TGF-β (5 ng/mL; black bars). After 96 hr, FoxP3 expression was measured by real time PCR (left panel) or flow cytometry (right panel).
DISCUSSION
Decreased IL-2 production upon T cell activation represents a significant cytokine aberration in SLE (13, 14) that has been recognized in MRL/lpr mice (15, 16). Our work identifies CaMK4 as a key molecule involved in the downregulation of IL-2 in MRL/lpr mice and proves that rescue of IL-2 production has beneficial immunological and clinical consequences. Summarily, CaMK4 promotes CREM-α-mediated suppression of IL-2 production and Treg function associated with increased presence of activated T cells and lupus-related pathology.
Complete absence of IL-2 or key elements of its receptor and associated signaling molecules cause lethal autoimmune disease (20, 23, 24). This syndrome is caused by lack of Tregs that depend on IL-2 signaling for their maintenance (20). Reduced Treg numbers (21, 25) and function (26) have been reported in patients with SLE and these defects have been associated with increased lupus disease activity (21). Although reduced IL-2 secretion has been proposed to underlie the SLE Treg defect (27), other causes, in particular chronic inflammation are also thought to contribute (28). Moreover, because the magnitude of Treg deficiency is variable and not absolute, its contribution to SLE has been debated. Administration of IL-2 through an IL-2-encoding vaccinia virus was able to decrease disease severity in MRL/lpr mice (29). However, the mechanism underlying disease amelioration was not sought in this early report (29). Two recent clinical trials performed in patients with immune-mediated diseases (hepatitis C virus-induced vasculitis and chronic graft vs. host disease) demonstrated that administration of exogenous IL-2 can indeed have an immune modulating effect associated with a rise in the numbers of Treg (30, 31). By studying the effects of the deficiency of Camk4 in mice with a chronic inflammatory autoimmune disease, we have produced data that indicates that CaMK4 is a central link between inflammation and decreased IL-2 production and that by eliminating this single kinase, IL-2 production is completely restored. Importantly, restoration of IL-2 has vast consequences in the immune system. It restricts the spontaneous activation of T cells reducing the fraction of CD44highCD62Llow and CD4−CD8− T cells and it increases the abundance of FoxP3+ regulatory T cells. The relevance of these changes is reflected in a significant increase in survival. Thus, our results support the concept that reduced IL-2 secretion is directly associated with SLE pathogenesis and that its restoration could be beneficial through effects in Treg number, Treg function and decreased T cell activation.
The fact that young MRL/lpr mice have normal levels of Camk4 suggests that this defect is not associated with the lack of Fas or with the genetic background that causes susceptibility to autoimmunity in this particular strain. Rather, increased CaMK4 levels and activation in diseased MRL/lpr mice may result from the autoimmune and/or inflammatory changes that occur in these mice when disease develops. We have previously reported that autoantibodies in sera from patients with SLE can induce CaMK4 activation and alter IL-2 production (8). Thus, CaMK4 activation in MRL/lpr mice could be caused in a similar fashion. The fact that CaMK4 deficiency abrogates lupus-associated pathology indicates that although high CaMK4 activity is not a primary defect it certainly contributes to disease development. Our data indicates that absence of CaMK4 increases the numbers and function of regulatory T cells. This effect is probably secondary to the restoration of IL-2 production since we were able to mimic it by adding exogenous IL-2 in the in vitro Treg differentiation assays. However, we cannot rule out if CaMK4 exerts a direct suppressive effect on FoxP3 expression that could account for higher FoxP3 levels in regulatory T cells of Camk4 deficient mice. We should point out that for the functional assays we used regulatory T cells sorted by the expression of CD25, a population that may also include effector CD4+ T cells.
We have previously reported biochemical evidence that shows that CaMK4 can directly inhibit IL-2 production by activating the repressor activity of CREM-α (8). The results presented in this manuscript suggest that at the in vivo level Camk4 deficiency may also rescue IL-2 production by decreasing the number of activated T cells and increasing the pool of naïve T cells that are more efficient IL-2 producers (32).
In this work, as well as in a previous report from our group (8, 10), we have identified CaMK4 as a kinase that becomes activated in the setting of chronic inflammation. CaMK4 activity was linked to high mesangial cell proliferation rates in MRL/lpr mice and its ablation protected the kidney from glomerulonephritis (10). Here, we have shown that CaMK4 also facilitates T cell activation and proliferation in MRL/lpr mice and in its absence T cell activation is reduced. Human T cells incubated with sera from patients with SLE show enhanced CaMK4 activity (8). The identification of the elements that regulate CaMK4 activation and that explain its link to inflammation will be sought in future work.
In conclusion, we have provided evidence that CaMK4 plays a central role in the development of autoimmunity in the MRL/lpr mouse. Its increased activation promotes T cell activation and organ damage. These effects are mediated by the suppression of IL-2 production and by reducing the numbers and function of Tregs. Our data indicate that IL-2 deficient production may represent a key molecular defect in the setting of SLE amenable of being corrected by the inhibition of CaMK4.
Supplementary Material
Footnotes
This work was supported by National Institute of Health Grant R01 A1068787 (GCT).
References
- 1.Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011;365:2110–2121. doi: 10.1056/NEJMra1100359. [DOI] [PubMed] [Google Scholar]
- 2.Shlomchik MJ, Craft JE, Mamula MJ. From T to B and back again: positive feedback in systemic autoimmune disease. Nat Rev Immunol. 2001;1:147–153. doi: 10.1038/35100573. [DOI] [PubMed] [Google Scholar]
- 3.Crispin JC, Liossis SN, Kis-Toth K, Lieberman LA, Kyttaris VC, Juang YT, Tsokos GC. Pathogenesis of human systemic lupus erythematosus: recent advances. Trends in molecular medicine. 2010;16:47–57. doi: 10.1016/j.molmed.2009.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Solomou EE, Juang YT, Gourley MF, Kammer GM, Tsokos GC. Molecular basis of deficient IL-2 production in T cells from patients with systemic lupus erythematosus. J Immunol. 2001;166:4216–4222. doi: 10.4049/jimmunol.166.6.4216. [DOI] [PubMed] [Google Scholar]
- 5.Tenbrock K, Juang YT, Tolnay M, Tsokos GC. The cyclic adenosine 5′-monophosphate response element modulator suppresses IL-2 production in stimulated T cells by a chromatin-dependent mechanism. J Immunol. 2003;170:2971–2976. doi: 10.4049/jimmunol.170.6.2971. [DOI] [PubMed] [Google Scholar]
- 6.Hanissian SH, Frangakis M, Bland MM, Jawahar S, Chatila TA. Expression of a Ca2+/calmodulin-dependent protein kinase, CaM kinase-Gr, in human T lymphocytes. Regulation of kinase activity by T cell receptor signaling. J Biol Chem. 1993;268:20055–20063. [PubMed] [Google Scholar]
- 7.Racioppi L, Means AR. Calcium/calmodulin-dependent kinase IV in immune and inflammatory responses: novel routes for an ancient traveller. Trends Immunol. 2008;29:600–607. doi: 10.1016/j.it.2008.08.005. [DOI] [PubMed] [Google Scholar]
- 8.Juang YT, Wang Y, Solomou EE, Li Y, Mawrin C, Tenbrock K, Kyttaris VC, Tsokos GC. Systemic lupus erythematosus serum IgG increases CREM binding to the IL-2 promoter and suppresses IL-2 production through CaMKIV. J Clin Invest. 2005;115:996–1005. doi: 10.1172/JCI200522854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ichinose K, Juang YT, Crispin JC, Kis-Toth K, Tsokos GC. Suppression of autoimmunity and organ pathology in lupus-prone mice upon inhibition of calcium/calmodulin-dependent protein kinase type IV. Arthritis Rheum. 2011;63:523–529. doi: 10.1002/art.30085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ichinose K, Rauen T, Juang YT, Kis-Toth K, Mizui M, Koga T, Tsokos GC. Cutting edge: Calcium/Calmodulin-dependent protein kinase type IV is essential for mesangial cell proliferation and lupus nephritis. J Immunol. 2011;187:5500–5504. doi: 10.4049/jimmunol.1102357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wofsy D, Murphy ED, Roths JB, Dauphinee MJ, Kipper SB, Talal N. Deficient interleukin 2 activity in MRL/Mp and C57BL/6J mice bearing the lpr gene. J Exp Med. 1981;154:1671–1680. doi: 10.1084/jem.154.5.1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cohen PL, Eisenberg RA. Lpr and gld: single gene models of systemic autoimmunity and lymphoproliferative disease. Annu Rev Immunol. 1991;9:243–269. doi: 10.1146/annurev.iy.09.040191.001331. [DOI] [PubMed] [Google Scholar]
- 13.Linker-Israeli M, Bakke AC, Kitridou RC, Gendler S, Gillis S, Horwitz DA. Defective production of interleukin 1 and interleukin 2 in patients with systemic lupus erythematosus (SLE) J Immunol. 1983;130:2651–2655. [PubMed] [Google Scholar]
- 14.Alcocer-Varela J, Alarcon-Segovia D. Decreased production of and response to interleukin-2 by cultured lymphocytes from patients with systemic lupus erythematosus. J Clin Invest. 1982;69:1388–1392. doi: 10.1172/JCI110579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Altman A, Theofilopoulos AN, Weiner R, Katz DH, Dixon FJ. Analysis of T cell function in autoimmune murine strains. Defects in production and responsiveness to interleukin 2. J Exp Med. 1981;154:791–808. doi: 10.1084/jem.154.3.791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Theofilopoulos AN, Dixon FJ. Murine models of systemic lupus erythematosus. Adv Immunol. 1985;37:269–390. doi: 10.1016/s0065-2776(08)60342-9. [DOI] [PubMed] [Google Scholar]
- 17.Lieberman LA, Tsokos GC. The IL-2 defect in systemic lupus erythematosus disease has an expansive effect on host immunity. J Biomed Biotechnol. 2010;2010:740619. doi: 10.1155/2010/740619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Means AR, Ribar TJ, Kane CD, Hook SS, Anderson KA. Regulation and properties of the rat Ca2+/calmodulin-dependent protein kinase IV gene and its protein products. Recent progress in hormone research. 1997;52:389–406. discussion 406–387. [PubMed] [Google Scholar]
- 19.Almeida AR, Legrand N, Papiernik M, Freitas AA. Homeostasis of peripheral CD4+ T cells: IL-2R alpha and IL-2 shape a population of regulatory cells that controls CD4+ T cell numbers. J Immunol. 2002;169:4850–4860. doi: 10.4049/jimmunol.169.9.4850. [DOI] [PubMed] [Google Scholar]
- 20.Setoguchi R, Hori S, Takahashi T, Sakaguchi S. Homeostatic maintenance of natural Foxp3(+) CD25(+) CD4(+) regulatory T cells by interleukin (IL)-2 and induction of autoimmune disease by IL-2 neutralization. J Exp Med. 2005;201:723–735. doi: 10.1084/jem.20041982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Valencia X, Yarboro C, Illei G, Lipsky PE. Deficient CD4+CD25high T regulatory cell function in patients with active systemic lupus erythematosus. J Immunol. 2007;178:2579–2588. doi: 10.4049/jimmunol.178.4.2579. [DOI] [PubMed] [Google Scholar]
- 22.Parietti V, Monneaux F, Decossas M, Muller S. Function of CD4+,CD25+ Treg cells in MRL/lpr mice is compromised by intrinsic defects in antigen-presenting cells and effector T cells. Arthritis Rheum. 2008;58:1751–1761. doi: 10.1002/art.23464. [DOI] [PubMed] [Google Scholar]
- 23.Campbell DJ, Koch MA. Phenotypical and functional specialization of FOXP3+ regulatory T cells. Nat Rev Immunol. 2011;11:119–130. doi: 10.1038/nri2916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Malek TR, Yu A, Vincek V, Scibelli P, Kong L. CD4 regulatory T cells prevent lethal autoimmunity in IL-2Rbeta-deficient mice. Implications for the nonredundant function of IL-2. Immunity. 2002;17:167–178. doi: 10.1016/s1074-7613(02)00367-9. [DOI] [PubMed] [Google Scholar]
- 25.Barath S, Aleksza M, Tarr T, Sipka S, Szegedi G, Kiss E. Measurement of natural (CD4+CD25high) and inducible (CD4+IL−10+) regulatory T cells in patients with systemic lupus erythematosus. Lupus. 2007;16:489–496. doi: 10.1177/0961203307080226. [DOI] [PubMed] [Google Scholar]
- 26.Yan B, Ye S, Chen G, Kuang M, Shen N, Chen S. Dysfunctional CD4+,CD25+ regulatory T cells in untreated active systemic lupus erythematosus secondary to interferon-alpha-producing antigen-presenting cells. Arthritis Rheum. 2008;58:801–812. doi: 10.1002/art.23268. [DOI] [PubMed] [Google Scholar]
- 27.Gomez-Martin D, Diaz-Zamudio M, Crispin JC, Alcocer-Varela J. Interleukin 2 and systemic lupus erythematosus: beyond the transcriptional regulatory net abnormalities. Autoimmunity reviews. 2009;9:34–39. doi: 10.1016/j.autrev.2009.02.035. [DOI] [PubMed] [Google Scholar]
- 28.Kyttaris VC, Juang YT, Tsokos GC. Immune cells and cytokines in systemic lupus erythematosus: an update. Current opinion in rheumatology. 2005;17:518–522. doi: 10.1097/01.bor.0000170479.01451.ab. [DOI] [PubMed] [Google Scholar]
- 29.Gutierrez-Ramos JC, Andreu JL, Revilla Y, Vinuela E, Martinez C. Recovery from autoimmunity of MRL/lpr mice after infection with an interleukin-2/vaccinia recombinant virus. Nature. 1990;346:271–274. doi: 10.1038/346271a0. [DOI] [PubMed] [Google Scholar]
- 30.Koreth J, Matsuoka K, Kim HT, McDonough SM, Bindra B, Alyea EP, 3rd, Armand P, Cutler C, Ho VT, Treister NS, Bienfang DC, Prasad S, Tzachanis D, Joyce RM, Avigan DE, Antin JH, Ritz J, Soiffer RJ. Interleukin-2 and regulatory T cells in graft-versus-host disease. N Engl J Med. 2011;365:2055–2066. doi: 10.1056/NEJMoa1108188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Saadoun D, Rosenzwajg M, Joly F, Six A, Carrat F, Thibault V, Sene D, Cacoub P, Klatzmann D. Regulatory T-cell responses to low-dose interleukin-2 in HCV-induced vasculitis. N Engl J Med. 2011;365:2067–2077. doi: 10.1056/NEJMoa1105143. [DOI] [PubMed] [Google Scholar]
- 32.Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999;401:708–12. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.