Abstract
Purpose
Leptin has been considered a link between metabolic state and reproductive activity. Defective reproductive function can occur in leptin-deficient and leptin-excessive conditions. The aim of this study was to examine the effects of centrally injected leptin on the hypothalamic KiSS-1 system in relation to gonadotropin-releasing hormone (GnRH) action in the initial stage of puberty.
Methods
Leptin (1 µg) was injected directly into the ventricle of pubertal female mice. The resultant gene expressions of hypothalamic GnRH and KiSS-1 and pituitary LH, 2 and 4 hours after injection, were compared with those of saline-injected control mice. The changes in the gene expressions after blocking the GnRH action were also analyzed.
Results
The basal expression levels of KiSS-1, GnRH, and LH were significantly higher in the pubertal mice than in the prepubertal mice. The 1-µg leptin dose significantly decreased the mRNA expression levels of KiSS-1, GnRH, and LH in the pubertal mice. A GnRH antagonist significantly increased the KiSS-1 and GnRH mRNA expression levels, and the additional leptin injection decreased the gene expression levels compared with those in the control group.
Conclusion
The excess leptin might have suppressed the central reproductive axis in the pubertal mice by inhibiting the KiSS-1 expression, and this mechanism is independent of the GnRH-LH-estradiol feedback loop.
Keywords: Leptin, KiSS-1, Gonadotropin-releasing hormone, Luteinizing hormone, Puberty
Introduction
Leptin produced by adipose tissue controls the hypothalamic regulation of appetite and energy consumption1) and is also believed to affect reproductive function. This latter idea is based on the observations that activation of the hypothalamic-pituitary-gonadal (HPG) axis depends on sufficient body energy stores2) and that prolonged negative energy balance can delay or prevent pubertal onset and cause reproductive dysfunction2,3). The most definitive evidence for the link between leptin and the reproductive axis is that ob/ob mice and db/db mice are infertile1,4), and that exogenous leptin restores fertility in ob/ob mice.
Although an appropriate leptin level is important for pubertal onset and maintenance of reproduction, high levels of leptin may downregulate GnRH release. One in vitro study showed that exposure of hypothalamic tissues to high leptin concentration inhibits and GnRH secretion5). Female transgenic skinny mice overexpressing leptin have late-onset hypothalamic hypogonadism6). Leptin also has inhibitory effects on estradiol production in the ovary7,8), but studies investigating the role of excessive leptin on the HPG axis are limited.
Reports that GnRH neurons do not physiologically express the leptin receptors suggest the existence of an intermediate pathway between leptin and GnRH9-11). Kisspeptins, products of the KiSS-1 gene, are regarded as key suspects. The KiSS-1 gene is expressed mainly by the arcuate nucleus and anteroventral periventricular nucleus (AVPV) of the hypothalamus, and their G-protein-coupled receptor KiSS-1R has been identified in the hypothalamus and pituitary gland12,13). Hypothalamic KiSS-1 mRNA and KiSS-1R mRNA expression increase with the initiation of puberty in both male and female rats14). Intraventricular injection of kisspeptin to adult rats increases plasma LH level, and this response is blocked by a GnRH antagonist, suggesting that kisspeptin acts on the gonadotropic axis via GnRH15). KiSS-1 expression is known to be controlled by metabolic status. Leptin is suspected to be a metabolic control factor of the KiSS-1 system.
The present experiments were performed to determine the effect of centrally injected leptin on the gene expression of hypothalamic KiSS-1 and GnRH and pituitary LH in the pubertal state and to determine whether leptin controls KiSS-1 or GnRH gene independently. To address these issues, we analyzed the effect of centrally injected leptin with or without a GnRH antagonist on KiSS-1 and GnRH gene expression.
Materials and methods
1. Animals and drugs
Female Imprinting Control Region mice in the 4th and 5th postnatal weeks were obtained from Dae Han Biolink Co. (Seoul, Korea). The mice were housed five per cage in a room maintained at 22±2℃ with an alternating 12/12-hour light/dark cycle. Food and water were available ad libitum. The experimental procedures were approved by the Institutional Animal Care and Use Committee of Kangwon National University. Recombinant mouse leptin (>98% pure) was purchased from Biodesign International, Meridian Life Science, Inc. (Memphis, TN, USA). We used cetrorelix acetate (Merck Serono S.A., Geneva, Switzerland) as the GnRH antagonist.
2. Intracerebroventricular (ICV) injection
Leptin was injected into the lateral ventricle according to a procedure described previously by Laursen and Belknap16). The ICV injection volume was 5 µL, and control mice were administered the same volume of saline.
3. Experimental design
We defined the pubertal sign in female mice as the existence of a vaginal opening. Pubertal mice (postnatal day 35 to 37) were assigned to a leptin or a control group. We injected 1 µg of leptin suspended in 5 µL of saline into the lateral ventricle of mice at 1:00 p.m. and the same volume of saline in the control group.
To block the action of GnRH, the GnRH antagonist cetrorelix (10 µg) was injected intraperitoneal 48, 24, and 1 hour before the ICV injection of either leptin or saline. Cetrorelix dose and injection interval were modified from a previous report17).
The responses were observed 2 and 4 hours after the ICV leptin or saline injection. Mice were anesthetized with pentobarbital and killed by decapitation. The brains were removed and placed in 0.05 M phosphate buffer treated with diethylpyrocarbonate. Pituitary glands were collected, and whole hypothalamic tissues including preoptic area were taken from 3-mm coronal sections caudal to the bregma using a Vibratome R brain matrix device (Ted Pella, Redding, CA, USA). The hypothalamic section size was determined based on the histology atlas of Paxinos and Franklin18).
We compared the mRNA expression of KiSS-1, GnRH, and LH between four groups (n=7 per subgroup): 1) ICV saline, 2) ICV leptin, 3) cetrorelix+ICV saline, and 4) cetrorelix+ICV leptin.
4. Analysis of mRNA expression by real-time polymerase chain reaction (PCR)
Total RNA was isolated from the hypothalamic and pituitary samples using the Easy-Spin Total RNA Extraction kit (INTRON Biotechnology Inc., Seongnam, Korea), and cDNA was synthesized from 2 µg of total RNA using OmniScript Reverse Transcriptase (QIAGEN Inc., Valencia, CA, USA). Hypothalamic expression of KiSS-1 and GnRH mRNA, and pituitary expression of LH mRNA were assessed by real-time PCR using the ABI Prism 7900 System (Applied Biosystems Inc., Foster City, CA, USA). The synthesized cDNAs were amplified further in triplicate by PCR using SYBR green I as the fluorescent dye.
To verify the changes in gene expression, real-time PCR was performed on the experimental samples using the Sequence Detection System ver. 2.2 (Applied Biosystems Inc.). The CT for each sample was calculated using the Applied Biosystems Sequence Detection System ver. 2.2 software. Primer sets for mouse KiSS-1, GNRH1, and Lhb used in this study are provided in Table 1.
Table 1.
Mouse-Specific Primer Sets for Amplification of Transcripts of KiSS-1, GNRH1, and Lhb Genes, Used for Real-Time Polymerase Chain Reaction
Relative expression levels of the target mRNAs were calculated using the standard curve method. The amount of mRNA was obtained from the standard curve and then normalized to the expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH), which is a house-keeping gene used as an internal control. The expression ratio of the target gene relative to the control was obtained by dividing the experimental sample value corrected for GAPDH by the control sample value corrected for GAPDH.
5. Statistical analysis
All values are expressed as mean±standard error. Statistical analysis was performed using IBM SPSS ver. 18.0 (IBM Co., Armonk, NY, USA). Changes in the expression of the KiSS-1, GnRH, and LH genes at puberty and the effects of leptin or the GnRH antagonist on the expression of these genes were assessed by Mann-Whitney U-test. P<0.05 was considered significant.
Results
1. Comparison of gene expression between pubertal and prepubertal mice
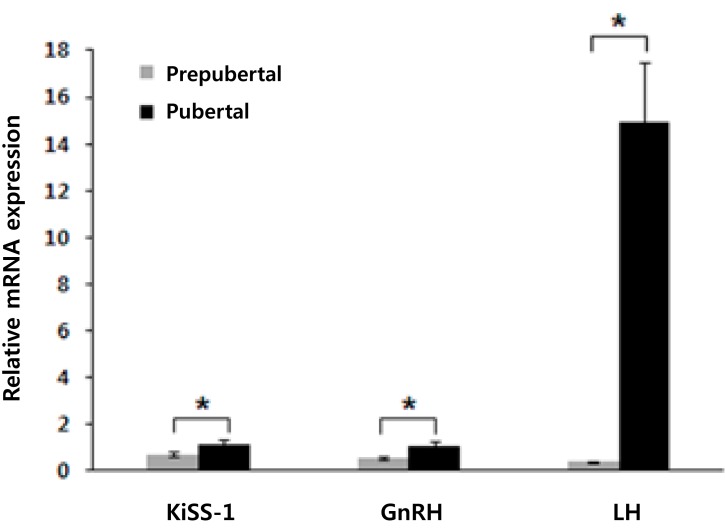
The gene expression levels of KiSS-1, GnRH, and LH were significantly higher in pubertal mice than in prepubertal mice in the basal state. Compared with the prepubertal mice, pubertal mice showed the following elevations in relative gene expression: 1.61-fold of KiSS-1 (pubertal/prepubertal, 1.14/0.71; P=0.007), 1.98-fold of GnRH (pubertal/prepubertal, 1.07/0.54; P=0.000), and 38.21-fold of LH (pubertal/prepubertal, 14.90/0.39; P=0.000) (Fig. 1).
Fig. 1.
Differences in gene expression levels between the prepubertal (gray columns) and pubertal mice (black columns) not treated with a specific drug. The values are means±standard error. *P<0.01 versus prepubertal mice.
2. Gene expression response to leptin
The experiments were performed with pubertal mice, which have an activated HPG axis and elevated target gene expression. Because the expression of each gene peaks at a different time, the responses were observed 2 and 4 hours after the ICV injection. The expression of the genes at each time was compared with that in the control at the same time to avoid the effects caused by diurnal variations.
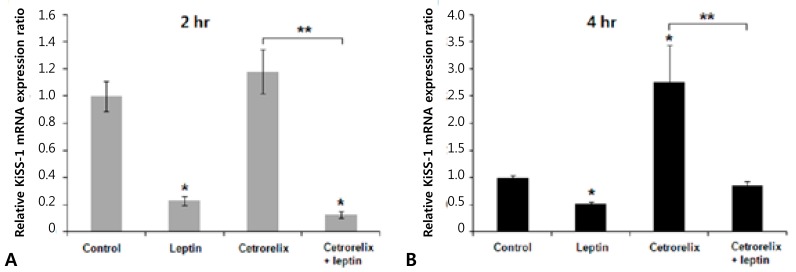
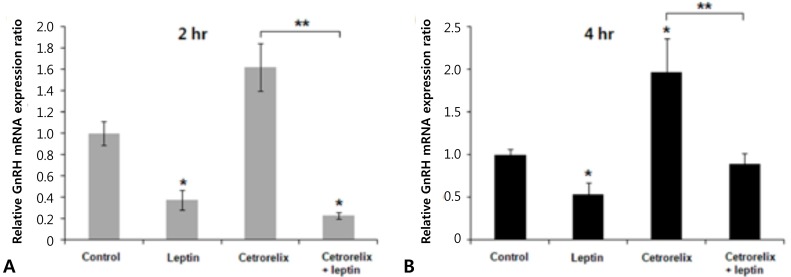
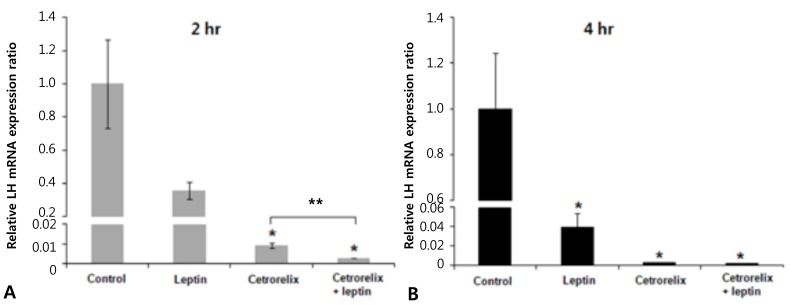
Leptin-injected mice had significantly reduced expression of KiSS-1, GnRH, and LH mRNA compared with those of the control. The ratios of the KiSS-1 mRNA expression in the leptin group relative to that in the control group were 0.23 at 2 hours (P=0.000) (Fig. 2A) and 0.52 at 4 hours (P=0.000) (Fig. 2B). The corresponding ratios for the GnRH gene were 0.37 at 2 hours (P=0.000) (Fig. 3A) and 0.53 at 4 hours (P=0.000) (Fig. 3B). The corresponding ratios for the LH gene were 0.36 at 2 hours (P=0.274) (Fig. 4A) and 0.04 at 4 hours (P=0.000) (Fig. 4B).
Fig. 2.
Effects of leptin and the GnRH antagonist (cetrorelix) on the hypothalamic KiSS-1 gene expression. (A) Gene expression levels 2 hours after intracerebroventricular (ICV) injection. (B) Gene expression levels 4 hours after ICV injection. The KiSS-1 mRNA expression level significantly decreased in the leptin group and increased in the cetrorelix group. The KiSS-1 mRNA expression level decreased significantly in the cetrorelix+leptin group as compared with the cetrorelix and control groups. The values are the means±standard error. *P<0.01 versus the control group. **P<0.01 versus the cetrorelix group.
Fig. 3.
Effects of leptin and cetrorelix on hypothalamic GnRH gene expression. (A) The gene expression levels 2 hours after the intracerebroventricular (ICV) injection. (B) The gene expression levels 4 hours after ICV injection. The GnRH mRNA expression level significantly decreased in the leptin group and increased in the cetrorelix group. The GnRH mRNA expression level significantly decreased in the cetrorelix+leptin group compared with the cetrorelix and control groups. The values are means±standard error. *P<0.01 versus the control group. **P<0.01 versus the cetrorelix group.
Fig. 4.
Effects of leptin and cetrorelix on pituitary LH gene expression. (A) Gene expression levels 2 hours after intracerebroventricular (ICV) injection. (B) Gene expression levels 4 hours after ICV injection. LH mRNA expression decreased significantly in the leptin group and in the cetrorelix group. LH mRNA expression decreased further in the cetrorelix+leptin group compared with the cetrorelix group. Values are means±standard error. *P<0.01 vs. control group. **P<0.01 vs. the cetrorelix group.
3. Gene expression response to the GnRH antagonist
Mice administered the GnRH antagonist had significantly increased KiSS-1 and GnRH mRNA expression compared with those of the control. The ratios of the KiSS-1 mRNA expression in the cetrorelix group relative to that in the control group were 1.18 at 2 hours (P=0.940) (Fig. 2A) and 2.76 at 4 hours (P=0.000) (Fig. 2B). The corresponding ratios for the GnRH gene were 1.62 at 2 hours (P=0.092) (Fig. 3A) and 1.97 at 4 hours (P=0.005) (Fig. 3B). The GnRH antagonist significantly blocked LH mRNA expression. The corresponding ratios for the LH gene were 0.0095 at 2 hours (P=0.000) (Fig. 4A) and 0.0025 at 4 hours (P=0.000) (Fig. 4B).
4. Gene expression response to the GnRH antagonist plus leptin
KiSS-1 and GnRH mRNA expression decreased significantly in the cetrorelix plus leptin group compared with that in either the cetrorelix group or the control. The ratios of the KiSS-1 mRNA expression in the cetrorelix plus leptin group relative to that in the cetrorelix group were 0.11 at 2 hours (0.13/1.18, P=0.000) (Fig. 2A) and 0.30 at 4 hours (0.85/2.76, P=0.000) (Fig. 2B). The corresponding ratios for the GnRH gene were 0.14 at 2 hours (0.23/1.62, P=0.000) (Fig. 3A) and 0.45 at 4 hours (0.89/1.97, P=0.007) (Fig. 3B). The LH mRNA expression decreased further in the cetrorelix plus leptin group, and the corresponding ratios were 0.31 at 2 hours (0.0029/0.0095, P=0.000) (Fig. 4A) and 0.80 at 4 hours (0.0020/0.0025, P=0.359) (Fig. 4B).
Compared with the control group, the ratios were 0.13 at 2 hours (P=0.000) (Fig. 2A) and 0.85 at 4 hours (P=0.176) (Fig. 2B) for the KiSS-1 gene, 0.23 at 2 hours (P=0.000) (Fig. 3A) and 0.89 at 4 hours (P=0.159) (Fig. 3B) for the GnRH gene, and 0.0029 at 2 hours (P=0.000) (Fig. 4A) and 0.0020 at 4 hours (P=0.000) (Fig. 4B) for the LH gene.
Discussion
Our results show that central injection of 1 µg leptin has a suppressive effect on KiSS-1, GnRH, and LH gene expression related to reproductive axis in pubertal female mice. We were interested in the direct effect of leptin on the expression of these genes, so leptin was administered via a central route. The acute administration of leptin directly into the ventricle might expose the brain to higher concentrations than are physiologically normal, and high level of leptin might suppress the expression of hypothalamic KiSS-1 and GnRH mRNAs and pituitary LH mRNA in normal pubertal mice.
Previous studies have shown that leptin is a permissive factor for puberty and plays a role in maintaining reproductive function. It is believed that there is a threshold level of circulating leptin to enter the puberty and leptin threshold of -3 ng/mL is necessary for normal reproductive function in human19). Leptin-deficient conditions are associated with reproductive dysfunction. Although the concept that leptin stimulates the hypothalamic-pituitary reproductive axis is prevalent, some reports have suggested that leptin also has a suppressive effect on the reproductive axis under conditions of leptin excess5,6,20,21).
When hypothalamic and pituitary tissues were exposed to various concentrations of leptin in vitro, the increase in LH secretion that is observed at lower leptin concentrations (10-12 to 10-9 M) was not observed at higher concentrations (10-7 to 10-5 M), and GnRH secretion decreased below the control level at the highest leptin concentration (10-6 M)5). Female transgenic skinny mice that overexpress leptin exhibit accelerated puberty and late-onset hypothalamic hypogonadism, characterized by prolonged menstrual cycles, atrophied ovaries, reduced hypothalamic GnRH content, and an impaired spontaneous LH pulse or a weak LH response to exogenous GnRH6). Although the mechanism of the suppressive effect of excessive leptin on reproductive axis has not be studied yet, these results suggest that excessive leptin downregulates the signal transmission in the hypothalamus and suppresses the central reproductive axis. Recent evidence suggests that leptin acts upstream of KiSS-1 neurons and this effect may be indirect22,23). We also suggest that the suppressive action of excessive leptin is upstream of KiSS-1 and GnRH neurons, but the mechanism of inhibitory signal should be further investigated.
Clinical conditions associated with hyperleptinemia are obesity, renal failure, and the cases using supraphysiological dose of leptin for therapeutic purposes. Women with obesity have low serum gonadotropin and estrogen levels and decreased LH secretion in response to exogenous GnRH20). Female DBA/2J mice with diet-induced obesity showed hyperleptinemia, and the pregnancy rate decreased by more than 60% but was normalized after stimulation with exogenous gonadotropin21). The prevalent view is that the dysfunction of the central reproductive axis in obesity is caused by leptin resistance. However, the direct effect of excessive leptin in obesity on the HPG axis needs to be investigated in further studies. Leptin has inhibitory effects on estradiol production in the ovary7,8).
Recent studies demonstrating that treatment of leptin enhances LH secretion and KiSS-1 mRNA expression were conducted mainly in adult mice or rats with hypoleptinemia and defective reproductive function. Leptin administered into the ventricles of adult male rats fasted for 5 days increased the blood LH level compared with the level in a control group24). In streptozotocin-induced diabetic rats, central infusion of leptin significantly increased blood LH level and hypothalamic KiSS-1 mRNA expression25). Peripheral administration of leptin into ob/ob mice recovers the defective KiSS-1 mRNA expression to some degree26). We carried out the experiment for female mice which weighed 19 to 22 g on normal diets and showed vaginal opening in time, so they are suspected to have normal physiology of leptin. The ICV injection of leptin to normal pubertal mice could be regarded as the condition of leptin excess.
Yu et al.5) also conducted an in vivo experiment for normal adult Sprague-Dawley rat and showed that the administration of 10 µg of human leptin into the 3rd ventricle increased plasma LH level in 10 to 50 minutes. Our study is similar to this study in experimental design, but the results are opposite. The different points are as follows. There is timing difference in tissue or blood sampling. We checked the gene expression in 2 and 4 hours observe a persisting effect, for we could not get the consistent results for immediate response (within 30 minutes) after leptin administration in preliminary experiments. Considering that we used mouse leptin and the cerebrospinal fluid volume of a mouse is 10- to 20-fold less than that of a rat an, 1 µg of mouse leptin in a pubertal mouse might act on the brain tissue more strongly than 10 µg of human leptin in a adult rat.
Becuase GnRH neurons do not express the leptin receptor under physiological conditions, GnRH neurons are regulated by leptin indirectly9-11). Recent evidence suggests that leptin may mediate the reproductive axis by regulation of kisspeptins. In our study, the decreased GnRH expression caused by leptin administration might have resulted from the suppression of KiSS-1 gene expression.
KiSS-1 neurons express the estrogen receptor-α (ERα), which is involved in feedback control27). Estradiol can inhibit the KiSS-1 neurons located in the arcuate nucleus28,29), but estradiol activates KiSS-1 in AVPV28). We administered a GnRH antagonist to observe the feedback effect of low estradiol concentration and to eliminate the effect of estradiol induced by GnRH/LH. As expected, the GnRH antagonist effectively blocked LH mRNA expression. The GnRH antagonist itself increased KiSS-1 and GnRH mRNA expression. Because GnRH neurons lack the ERα29), the increase in GnRH mRNA level is probably caused by both an indirect effect that is dependent of KiSS-1 and an ultrashort-loop autocrine feedback mechanism30).
The observation that the stimulation of KiSS-1 and GnRH mRNA expression by the GnRH antagonist was suppressed by leptin suggests that leptin inhibits the of KiSS-1 expression regardless of feedback control. LH mRNA expression blocked by a GnRH antagonist was further reduced by leptin. This suggests that there was an indirect regulation or an effect secondary to changes in hypothalamic gene expression.
In summary, we demonstrated that supraphysiologic level of leptin in normal pubertal female mice suppresses the hypothalamicpituitary reproductive axis by inhibiting the expression of the KiSS-1 gene, and that this action is independent of the GnRH-LH-estradiol feedback loop.
This study has several limitations. First, since the results show the acute and short-term effects of a single injection of leptin, it is difficult to link with the chronic or long-term effect. Second, the results can be confined to pubertal mice in which the HPG axis is initially activated. Third, if hormonal level would be checked at the same time of tissue collection, it would give more information about the clinical correlation. Fourth, a dose response curve for leptin or at least an examination of a lower leptin dose would strengthen the report.
In addition, we should consider more complex neuronal interactions among the nuclei in hypothalamus. Further studies are required not only to reveal the connection between leptin excess and reproductive dysfunction but also to discover underlying mechanisms. The present study is the experimental evidence for another aspect of leptin and represents a step forward in understanding the control of reproductive function.
Acknowledgment
This work was supported by a Korea Research Foundation Grant funded by the Korean Government (MOEHRD, Basic Research Promotion Fund) (KRF-2008-331-E00153).
References
- 1.Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994;372:425–432. doi: 10.1038/372425a0. [DOI] [PubMed] [Google Scholar]
- 2.Parent AS, Teilmann G, Juul A, Skakkebaek NE, Toppari J, Bourguignon JP. The timing of normal puberty and the age limits of sexual precocity: variations around the world, secular trends, and changes after migration. Endocr Rev. 2003;24:668–693. doi: 10.1210/er.2002-0019. [DOI] [PubMed] [Google Scholar]
- 3.Cunningham MJ, Clifton DK, Steiner RA. Leptin's actions on the reproductive axis: perspectives and mechanisms. Biol Reprod. 1999;60:216–222. doi: 10.1095/biolreprod60.2.216. [DOI] [PubMed] [Google Scholar]
- 4.Chen H, Charlat O, Tartaglia LA, Woolf EA, Weng X, Ellis SJ, et al. Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell. 1996;84:491–495. doi: 10.1016/s0092-8674(00)81294-5. [DOI] [PubMed] [Google Scholar]
- 5.Yu WH, Kimura M, Walczewska A, Karanth S, McCann SM. Role of leptin in hypothalamic-pituitary function. Proc Natl Acad Sci U S A. 1997;94:1023–1028. doi: 10.1073/pnas.94.3.1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yura S, Ogawa Y, Sagawa N, Masuzaki H, Itoh H, Ebihara K, et al. Accelerated puberty and late-onset hypothalamic hypogonadism in female transgenic skinny mice overexpressing leptin. J Clin Invest. 2000;105:749–755. doi: 10.1172/JCI8353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Agarwal SK, Vogel K, Weitsman SR, Magoffin DA. Leptin antagonizes the insulin-like growth factor-I augmentation of steroidogenesis in granulosa and theca cells of the human ovary. J Clin Endocrinol Metab. 1999;84:1072–1076. doi: 10.1210/jcem.84.3.5543. [DOI] [PubMed] [Google Scholar]
- 8.Zachow RJ, Weitsman SR, Magoffin DA. Leptin impairs the synergistic stimulation by transforming growth factor-beta of follicle-stimulating hormone-dependent aromatase activity and messenger ribonucleic acid expression in rat ovarian granulosa cells. Biol Reprod. 1999;61:1104–1109. doi: 10.1095/biolreprod61.4.1104. [DOI] [PubMed] [Google Scholar]
- 9.Burcelin R, Thorens B, Glauser M, Gaillard RC, Pralong FP. Gonadotropin-releasing hormone secretion from hypothalamic neurons: stimulation by insulin and potentiation by leptin. Endocrinology. 2003;144:4484–4491. doi: 10.1210/en.2003-0457. [DOI] [PubMed] [Google Scholar]
- 10.Finn PD, Cunningham MJ, Pau KY, Spies HG, Clifton DK, Steiner RA. The stimulatory effect of leptin on the neuroendocrine reproductive axis of the monkey. Endocrinology. 1998;139:4652–4662. doi: 10.1210/endo.139.11.6297. [DOI] [PubMed] [Google Scholar]
- 11.Hakansson ML, Brown H, Ghilardi N, Skoda RC, Meister B. Leptin receptor immunoreactivity in chemically defined target neurons of the hypothalamus. J Neurosci. 1998;18:559–572. doi: 10.1523/JNEUROSCI.18-01-00559.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kotani M, Detheux M, Vandenbogaerde A, Communi D, Vanderwinden JM, Le Poul E, et al. The metastasis suppressor gene KiSS-1 encodes kisspeptins, the natural ligands of the orphan G protein-coupled receptor GPR54. J Biol Chem. 2001;276:34631–34636. doi: 10.1074/jbc.M104847200. [DOI] [PubMed] [Google Scholar]
- 13.Ohtaki T, Shintani Y, Honda S, Matsumoto H, Hori A, Kanehashi K, et al. Metastasis suppressor gene KiSS-1 encodes peptide ligand of a G-protein-coupled receptor. Nature. 2001;411:613–617. doi: 10.1038/35079135. [DOI] [PubMed] [Google Scholar]
- 14.Navarro VM, Castellano JM, Fernández-Fernandez R, Barreiro ML, Roa J, Sanchez-Criado JE, et al. Developmental and hormonally regulated messenger ribonucleic acid expression of KiSS-1 and its putative receptor, GPR54, in rat hypothalamus and potent luteinizing hormone-releasing activity of KiSS-1 peptide. Endocrinology. 2004;145:4565–4574. doi: 10.1210/en.2004-0413. [DOI] [PubMed] [Google Scholar]
- 15.Irwig MS, Fraley GS, Smith JT, Acohido BV, Popa SM, Cunningham MJ, et al. Kisspeptin activation of gonadotropin releasing hormone neurons and regulation of KiSS-1 mRNA in the male rat. Neuroendocrinology. 2004;80:264–272. doi: 10.1159/000083140. [DOI] [PubMed] [Google Scholar]
- 16.Laursen SE, Belknap JK. Intracerebroventricular injections in mice. Some methodological refinements. J Pharmacol Methods. 1986;16:355–357. doi: 10.1016/0160-5402(86)90038-0. [DOI] [PubMed] [Google Scholar]
- 17.Meirow D, Assad G, Dor J, Rabinovici J. The GnRH antagonist cetrorelix reduces cyclophosphamide-induced ovarian follicular destruction in mice. Hum Reprod. 2004;19:1294–1299. doi: 10.1093/humrep/deh257. [DOI] [PubMed] [Google Scholar]
- 18.Paxinos G, Franklin KB. The mouse brain in stereotaxic coordinates. Compact 3rd ed. Boston: Elsevier Academic Press; 2008. [Google Scholar]
- 19.Dardeno TA, Chou SH, Moon HS, Chamberland JP, Fiorenza CG, Mantzoros CS. Leptin in human physiology and therapeutics. Front Neuroendocrinol. 2010;31:377–393. doi: 10.1016/j.yfrne.2010.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Santoro N, Lasley B, McConnell D, Allsworth J, Crawford S, Gold EB, et al. Body size and ethnicity are associated with menstrual cycle alterations in women in the early menopausal transition: The Study of Women's Health across the Nation (SWAN) Daily Hormone Study. J Clin Endocrinol Metab. 2004;89:2622–2631. doi: 10.1210/jc.2003-031578. [DOI] [PubMed] [Google Scholar]
- 21.Tortoriello DV, McMinn J, Chua SC. Dietary-induced obesity and hypothalamic infertility in female DBA/2J mice. Endocrinology. 2004;145:1238–1247. doi: 10.1210/en.2003-1406. [DOI] [PubMed] [Google Scholar]
- 22.Quennell JH, Howell CS, Roa J, Augustine RA, Grattan DR, Anderson GM. Leptin deficiency and diet-induced obesity reduce hypothalamic kisspeptin expression in mice. Endocrinology. 2011;152:1541–1550. doi: 10.1210/en.2010-1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cravo RM, Margatho LO, Osborne-Lawrence S, Donato J, Jr, Atkin S, Bookout AL, et al. Characterization of Kiss1 neurons using transgenic mouse models. Neuroscience. 2011;173:37–56. doi: 10.1016/j.neuroscience.2010.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hwa JJ, Ghibaudi L, Compton D, Fawzi AB, Strader CD. Intracerebroventricular injection of leptin increases thermogenesis and mobilizes fat metabolism in ob/ob mice. Horm Metab Res. 1996;28:659–663. doi: 10.1055/s-2007-979873. [DOI] [PubMed] [Google Scholar]
- 25.Castellano JM, Navarro VM, Fernandez-Fernandez R, Roa J, Vigo E, Pineda R, et al. Expression of hypothalamic KiSS-1 system and rescue of defective gonadotropic responses by kisspeptin in streptozotocin-induced diabetic male rats. Diabetes. 2006;55:2602–2610. doi: 10.2337/db05-1584. [DOI] [PubMed] [Google Scholar]
- 26.Smith JT, Acohido BV, Clifton DK, Steiner RA. KiSS-1 neurones are direct targets for leptin in the ob/ob mouse. J Neuroendocrinol. 2006;18:298–303. doi: 10.1111/j.1365-2826.2006.01417.x. [DOI] [PubMed] [Google Scholar]
- 27.Smith JT, Cunningham MJ, Rissman EF, Clifton DK, Steiner RA. Regulation of Kiss1 gene expression in the brain of the female mouse. Endocrinology. 2005;146:3686–3692. doi: 10.1210/en.2005-0488. [DOI] [PubMed] [Google Scholar]
- 28.Smith JT, Popa SM, Clifton DK, Hoffman GE, Steiner RA. Kiss1 neurons in the forebrain as central processors for generating the preovulatory luteinizing hormone surge. J Neurosci. 2006;26:6687–6694. doi: 10.1523/JNEUROSCI.1618-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dorling AA, Todman MG, Korach KS, Herbison AE. Critical role for estrogen receptor alpha in negative feedback regulation of gonadotropin-releasing hormone mRNA expression in the female mouse. Neuroendocrinology. 2003;78:204–209. doi: 10.1159/000073703. [DOI] [PubMed] [Google Scholar]
- 30.Woller MJ, Meyer S, Ada-Nguema A, Waechter-Brulla D. Dissecting autocrine effects on pulsatile release of gonadotropin-releasing hormone in cultured rat hypothalamic tissue. Exp Biol Med (Maywood) 2004;229:56–64. doi: 10.1177/153537020422900107. [DOI] [PubMed] [Google Scholar]