Abstract
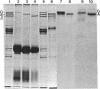
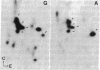
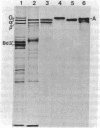
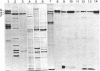
Goblin , a high molecular weight (Mr, 260,000) polypeptide of avian erythrocyte plasma membranes characterized by hormone-dependent phosphorylation, is shown by a variety of criteria to be the avian equivalent of ankyrin, the membrane attachment protein for spectrin; a polyclonal monospecific goblin antiserum reacts specifically with ankyrin from mammalian erythrocyte ghosts; goblin and ankyrin have highly homologous, although distinct, two-dimensional peptide maps; and, in reconstitution experiments, goblin binds to spectrin and band 3 in approximately the same molar ratio as ankyrin. Immunoautoradiography and immunofluorescence with goblin antiserum reveal that a serologically related polypeptide (Mr, 235,000) is present in highly purified membrane fractions of mammalian myocardium and in whole extracts of adult chicken cardiac and skeletal muscle-nonerythroid tissues which express predominantly the erythroid (alpha beta-) spectrin phenotype. Erythroid spectrin and goblin (ankyrin) are codistributed in skeletal muscle at the sarcolemma as discrete foci adjacent to the Z lines and, in pectoral muscle, also at the periphery of the Z discs. These spatial relationships indicate that goblin and spectrin in muscle cells form a structural framework that serves as the attachment site for the myofiber at the level of the Z line on the sarcolemma.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alper S. L., Beam K. G., Greengard P. Hormonal control of Na+-K+ co-transport in turkey erythrocytes. Multiple site phosphorylation of goblin, a high molecular weight protein of the plasma membrane. J Biol Chem. 1980 May 25;255(10):4864–4871. [PubMed] [Google Scholar]
- Alper S. L., Palfrey H. C., DeRiemer S. A., Greengard P. Hormonal control of protein phosphorylation in turkey erythrocytes. Phosphorylation by cAMP-dependent and Ca2+-dependent protein kinases of distinct sites in goblin, a high molecular weight protein of the plasma membrane. J Biol Chem. 1980 Nov 25;255(22):11029–11039. [PubMed] [Google Scholar]
- Beam K. G., Alper S. L., Palade G. E., Greengard P. Hormonally regulated phosphoprotein of turkey erythrocytes: localization to plasma membrane. J Cell Biol. 1979 Oct;83(1):1–15. doi: 10.1083/jcb.83.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett V., Branton D. Selective association of spectrin with the cytoplasmic surface of human erythrocyte plasma membranes. Quantitative determination with purified (32P)spectrin. J Biol Chem. 1977 Apr 25;252(8):2753–2763. [PubMed] [Google Scholar]
- Bennett V., Davis J. Erythrocyte ankyrin: immunoreactive analogues are associated with mitotic structures in cultured cells and with microtubules in brain. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7550–7554. doi: 10.1073/pnas.78.12.7550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett V. Immunoreactive forms of human erythrocyte ankyrin are present in diverse cells and tissues. Nature. 1979 Oct 18;281(5732):597–599. doi: 10.1038/281597a0. [DOI] [PubMed] [Google Scholar]
- Bennett V., Stenbuck P. J. Human erythrocyte ankyrin. Purification and properties. J Biol Chem. 1980 Mar 25;255(6):2540–2548. [PubMed] [Google Scholar]
- Bennett V., Stenbuck P. J. Identification and partial purification of ankyrin, the high affinity membrane attachment site for human erythrocyte spectrin. J Biol Chem. 1979 Apr 10;254(7):2533–2541. [PubMed] [Google Scholar]
- Bennett V., Stenbuck P. J. The membrane attachment protein for spectrin is associated with band 3 in human erythrocyte membranes. Nature. 1979 Aug 9;280(5722):468–473. doi: 10.1038/280468a0. [DOI] [PubMed] [Google Scholar]
- Bennett V. The molecular basis for membrane - cytoskeleton association in human erythrocytes. J Cell Biochem. 1982;18(1):49–65. doi: 10.1002/jcb.1982.240180106. [DOI] [PubMed] [Google Scholar]
- Blikstad I., Nelson W. J., Moon R. T., Lazarides E. Synthesis and assembly of spectrin during avian erythropoiesis: stoichiometric assembly but unequal synthesis of alpha and beta spectrin. Cell. 1983 Apr;32(4):1081–1091. doi: 10.1016/0092-8674(83)90292-1. [DOI] [PubMed] [Google Scholar]
- Branton D., Cohen C. M., Tyler J. Interaction of cytoskeletal proteins on the human erythrocyte membrane. Cell. 1981 Apr;24(1):24–32. doi: 10.1016/0092-8674(81)90497-9. [DOI] [PubMed] [Google Scholar]
- Calvert R., Bennett P., Gratzer W. Properties and structural role of the subunits of human spectrin. Eur J Biochem. 1980 Jun;107(2):355–361. doi: 10.1111/j.1432-1033.1980.tb06036.x. [DOI] [PubMed] [Google Scholar]
- Chiesi M., Ho M. M., Inesi G., Somlyo A. V., Somlyo A. P. Primary role of sarcoplasmic reticulum in phasic contractile activation of cardiac myocytes with shunted myolemma. J Cell Biol. 1981 Dec;91(3 Pt 1):728–742. doi: 10.1083/jcb.91.3.728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder J. H., Pickett R. A., 2nd, Hampton J., Lerner R. A. Radioiodination of proteins in single polyacrylamide gel slices. Tryptic peptide analysis of all the major members of complex multicomponent systems using microgram quantities of total protein. J Biol Chem. 1977 Sep 25;252(18):6510–6515. [PubMed] [Google Scholar]
- Glenney J. R., Jr, Glenney P. Fodrin is the general spectrin-like protein found in most cells whereas spectrin and the TW protein have a restricted distribution. Cell. 1983 Sep;34(2):503–512. doi: 10.1016/0092-8674(83)90383-5. [DOI] [PubMed] [Google Scholar]
- Granger B. L., Lazarides E. Desmin and vimentin coexist at the periphery of the myofibril Z disc. Cell. 1979 Dec;18(4):1053–1063. doi: 10.1016/0092-8674(79)90218-6. [DOI] [PubMed] [Google Scholar]
- Granger B. L., Lazarides E. Synemin: a new high molecular weight protein associated with desmin and vimentin filaments in muscle. Cell. 1980 Dec;22(3):727–738. doi: 10.1016/0092-8674(80)90549-8. [DOI] [PubMed] [Google Scholar]
- Granger B. L., Lazarides E. The existence of an insoluble Z disc scaffold in chicken skeletal muscle. Cell. 1978 Dec;15(4):1253–1268. doi: 10.1016/0092-8674(78)90051-x. [DOI] [PubMed] [Google Scholar]
- Granger B. L., Repasky E. A., Lazarides E. Synemin and vimentin are components of intermediate filaments in avian erythrocytes. J Cell Biol. 1982 Feb;92(2):299–312. doi: 10.1083/jcb.92.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hargreaves W. R., Giedd K. N., Verkleij A., Branton D. Reassociation of ankyrin with band 3 in erythrocyte membranes and in lipid vesicles. J Biol Chem. 1980 Dec 25;255(24):11965–11972. [PubMed] [Google Scholar]
- Hubbard B. D., Lazarides E. Copurification of actin and desmin from chicken smooth muscle and their copolymerization in vitro to intermediate filaments. J Cell Biol. 1979 Jan;80(1):166–182. doi: 10.1083/jcb.80.1.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones L. R., Besch H. R., Jr, Fleming J. W., McConnaughey M. M., Watanabe A. M. Separation of vesicles of cardiac sarcolemma from vesicles of cardiac sarcoplasmic reticulum. Comparative biochemical analysis of component activities. J Biol Chem. 1979 Jan 25;254(2):530–539. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Luna E. J., Kidd G. H., Branton D. Identification by peptide analysis of the spectrin-binding protein in human erythrocytes. J Biol Chem. 1979 Apr 10;254(7):2526–2532. [PubMed] [Google Scholar]
- Morrow J. S., Speicher D. W., Knowles W. J., Hsu C. J., Marchesi V. T. Identification of functional domains of human erythrocyte spectrin. Proc Natl Acad Sci U S A. 1980 Nov;77(11):6592–6596. doi: 10.1073/pnas.77.11.6592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson W. J., Granger B. L., Lazarides E. Avian lens spectrin: subunit composition compared with erythrocyte and brain spectrin. J Cell Biol. 1983 Oct;97(4):1271–1276. doi: 10.1083/jcb.97.4.1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson W. J., Lazarides E. Expression of the beta subunit of spectrin in nonerythroid cells. Proc Natl Acad Sci U S A. 1983 Jan;80(2):363–367. doi: 10.1073/pnas.80.2.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson W. J., Lazarides E. Switching of subunit composition of muscle spectrin during myogenesis in vitro. 1983 Jul 28-Aug 3Nature. 304(5924):364–368. doi: 10.1038/304364a0. [DOI] [PubMed] [Google Scholar]
- Olmsted J. B. Affinity purification of antibodies from diazotized paper blots of heterogeneous protein samples. J Biol Chem. 1981 Dec 10;256(23):11955–11957. [PubMed] [Google Scholar]
- Palfrey H. C., Schiebler W., Greengard P. A major calmodulin-binding protein common to various vertebrate tissues. Proc Natl Acad Sci U S A. 1982 Jun;79(12):3780–3784. doi: 10.1073/pnas.79.12.3780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardo J. V., Siliciano J. D., Craig S. W. A vinculin-containing cortical lattice in skeletal muscle: transverse lattice elements ("costameres") mark sites of attachment between myofibrils and sarcolemma. Proc Natl Acad Sci U S A. 1983 Feb;80(4):1008–1012. doi: 10.1073/pnas.80.4.1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Repasky E. A., Granger B. L., Lazarides E. Widespread occurrence of avian spectrin in nonerythroid cells. Cell. 1982 Jul;29(3):821–833. doi: 10.1016/0092-8674(82)90444-5. [DOI] [PubMed] [Google Scholar]
- Street S. F. Lateral transmission of tension in frog myofibers: a myofibrillar network and transverse cytoskeletal connections are possible transmitters. J Cell Physiol. 1983 Mar;114(3):346–364. doi: 10.1002/jcp.1041140314. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler J. M., Hargreaves W. R., Branton D. Purification of two spectrin-binding proteins: biochemical and electron microscopic evidence for site-specific reassociation between spectrin and bands 2.1 and 4.1. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5192–5196. doi: 10.1073/pnas.76.10.5192. [DOI] [PMC free article] [PubMed] [Google Scholar]