Abstract
The molecular defect in a patient with a moderately severe form of osteogenesis imperfecta was characterized by nuclease S1 mapping. Single-stranded 5' and 3' end-labeled DNA probes coding for 80% of the carboxyl-propeptide of the pro alpha 2(I) collagen gene were hybridized to mRNA isolated from cultured fibroblasts of the patient and his parents. Nuclease S1 digestion revealed a homozygous mutation in the patient and a heterozygous pattern in the consanguineous parents. As a result of the defect in the gene, none of the pro alpha 2(I) chains synthesized by the patient's fibroblasts were incorporated into a type I procollagen heterotrimer consisting of two pro alpha 1(I) chains and one pro alpha 2(I) chain. Cultured skin fibroblasts from the patient have previously been shown to secrete only pro alpha 1(I) trimers. As shown here, fibroblasts from both parents, who do not have osteogenesis imperfecta, secrete both pro alpha 1(I) trimers and normal type I procollagen. A further observation was that synthesis of pro alpha 2(I) chains was decreased in fibroblasts from the patient and his parents. The decrease in the synthesis of pro alpha 2(I) chains is not caused by decreased transcription of the pro alpha 2(I) collagen alleles, since the pro alpha 1(I)/pro alpha 2(I) mRNA ratios were normal in the patient and his parents.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barsh G. S., Byers P. H. Reduced secretion of structurally abnormal type I procollagen in a form of osteogenesis imperfecta. Proc Natl Acad Sci U S A. 1981 Aug;78(8):5142–5146. doi: 10.1073/pnas.78.8.5142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barsh G. S., David K. E., Byers P. H. Type I osteogenesis imperfecta: a nonfunctional allele for pro alpha 1 (I) chains of type I procollagen. Proc Natl Acad Sci U S A. 1982 Jun;79(12):3838–3842. doi: 10.1073/pnas.79.12.3838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berk A. J., Sharp P. A. Spliced early mRNAs of simian virus 40. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1274–1278. doi: 10.1073/pnas.75.3.1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnett W., Rosenbloom J. Isolation and translation of elastin mRNA from chick aorta. Biochem Biophys Res Commun. 1979 Feb 14;86(3):478–484. doi: 10.1016/0006-291x(79)91739-x. [DOI] [PubMed] [Google Scholar]
- Byers P. H., Shapiro J. R., Rowe D. W., David K. E., Holbrook K. A. Abnormal alpha 2-chain in type I collagen from a patient with a form of osteogenesis imperfecta. J Clin Invest. 1983 Mar;71(3):689–697. doi: 10.1172/JCI110815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bächinger H. P., Bruckner P., Timpl R., Prockop D. J., Engel J. Folding mechanism of the triple helix in type-III collagen and type-III pN-collagen. Role of disulfide bridges and peptide bond isomerization. Eur J Biochem. 1980 May;106(2):619–632. doi: 10.1111/j.1432-1033.1980.tb04610.x. [DOI] [PubMed] [Google Scholar]
- Bächinger H. P., Fessler L. I., Timpl R., Fessler J. H. Chain assembly intermediate in the biosynthesis of type III procollagen in chick embryo blood vessels. J Biol Chem. 1981 Dec 25;256(24):13193–13199. [PubMed] [Google Scholar]
- Chu M. L., Myers J. C., Bernard M. P., Ding J. F., Ramirez F. Cloning and characterization of five overlapping cDNAs specific for the human pro alpha 1(I) collagen chain. Nucleic Acids Res. 1982 Oct 11;10(19):5925–5934. doi: 10.1093/nar/10.19.5925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu M. L., Williams C. J., Pepe G., Hirsch J. L., Prockop D. J., Ramirez F. Internal deletion in a collagen gene in a perinatal lethal form of osteogenesis imperfecta. Nature. 1983 Jul 7;304(5921):78–80. doi: 10.1038/304078a0. [DOI] [PubMed] [Google Scholar]
- Deak S. B., Nicholls A., Pope F. M., Prockop D. J. The molecular defect in a nonlethal variant of osteogenesis imperfecta. Synthesis of pro-alpha 2(I) chains which are not incorporated into trimers of type I procollagen. J Biol Chem. 1983 Dec 25;258(24):15192–15197. [PubMed] [Google Scholar]
- Goossens M., Lee K. Y., Liebhaber S. A., Kan Y. W. Globin structural mutant alpha 125Leu leads to Pro is a novel cause of alpha-thalassaemia. Nature. 1982 Apr 29;296(5860):864–865. doi: 10.1038/296864a0. [DOI] [PubMed] [Google Scholar]
- Hollister D. W., Byers P. H., Holbrook K. A. Genetic disorders of collagen metabolism. Adv Hum Genet. 1982;12:1–87. doi: 10.1007/978-1-4615-8315-8_1. [DOI] [PubMed] [Google Scholar]
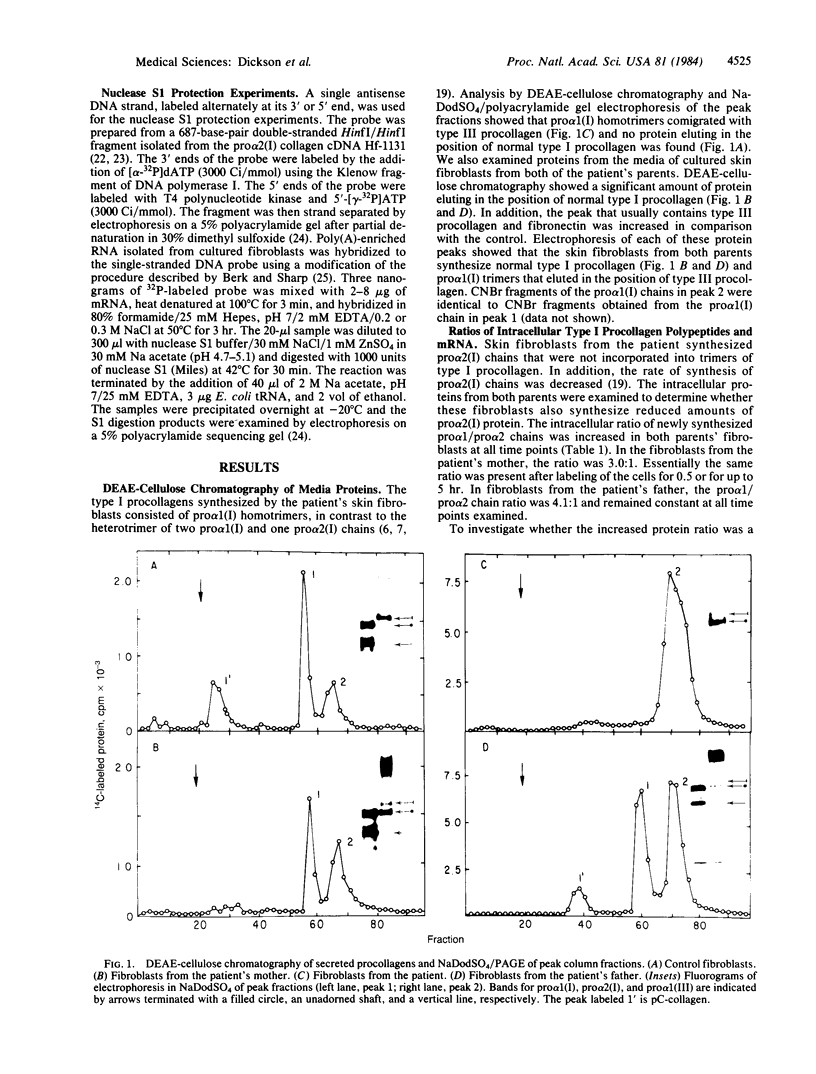
- Kao W. W., Prockop D. J., Berg R. A. Kinetics for the secretion of nonhelical procollagen by freshly isolated tendon cells. J Biol Chem. 1979 Apr 10;254(7):2234–2243. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Myers J. C., Chu M. L., Faro S. H., Clark W. J., Prockop D. J., Ramirez F. Cloning a cDNA for the pro-alpha 2 chain of human type I collagen. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3516–3520. doi: 10.1073/pnas.78.6.3516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers J. C., Dickson L. A., de Wet W. J., Bernard M. P., Chu M. L., Di Liberto M., Pepe G., Sangiorgi F. O., Ramirez F. Analysis of the 3' end of the human pro-alpha 2(I) collagen gene. Utilization of multiple polyadenylation sites in cultured fibroblasts. J Biol Chem. 1983 Aug 25;258(16):10128–10135. [PubMed] [Google Scholar]
- Nicholls A. C., Pope F. M., Schloon H. Biochemical heterogeneity of osteogenesis imperfecta: New variant. Lancet. 1979 Jun 2;1(8127):1193–1193. doi: 10.1016/s0140-6736(79)91872-5. [DOI] [PubMed] [Google Scholar]
- Peltonen L., Palotie A., Prockop D. J. A defect in the structure of type I procollagen in a patient who had osteogenesis imperfecta: excess mannose in the COOH-terminal propeptide. Proc Natl Acad Sci U S A. 1980 Oct;77(10):6179–6183. doi: 10.1073/pnas.77.10.6179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penttinen R. P., Lichtenstein J. R., Martin G. R., McKusick V. A. Abnormal collagen metabolism in cultured cells in osteogenesis imperfecta. Proc Natl Acad Sci U S A. 1975 Feb;72(2):586–589. doi: 10.1073/pnas.72.2.586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenbloom J., Endo R., Harsch M. Termination of procollagen chain synthesis by puromycin. Evidence that assembly and secretion require a COOH-terminal extension. J Biol Chem. 1976 Apr 10;251(7):2070–2076. [PubMed] [Google Scholar]
- Steinmann B., Tuderman L., Peltonen L., Martin G. R., McKusick V. A., Prockop D. J. Evidence for a structural mutation of procollagen type I in a patient with the Ehlers-Danlos syndrome type VII. J Biol Chem. 1980 Sep 25;255(18):8887–8893. [PubMed] [Google Scholar]
- Vogt V. M. Purification and further properties of single-strand-specific nuclease from Aspergillus oryzae. Eur J Biochem. 1973 Feb 15;33(1):192–200. doi: 10.1111/j.1432-1033.1973.tb02669.x. [DOI] [PubMed] [Google Scholar]
- Williams C. J., Prockop D. J. Synthesis and processing of a type I procollagen containing shortened pro-alpha 1(I) chains by fibroblasts from a patient with osteogenesis imperfecta. J Biol Chem. 1983 May 10;258(9):5915–5921. [PubMed] [Google Scholar]
- de Wet W. J., Pihlajaniemi T., Myers J., Kelly T. E., Prockop D. J. Synthesis of a shortened pro-alpha 2(I) chain and decreased synthesis of pro-alpha 2(I) chains in a proband with osteogenesis imperfecta. J Biol Chem. 1983 Jun 25;258(12):7721–7728. [PubMed] [Google Scholar]