Abstract
Pathological cardiac hypertrophy is a key risk factor for heart failure. It is associated with increased interstitial fibrosis, cell death and cardiac dysfunction. The progression of pathological cardiac hypertrophy has long been considered as irreversible. However, recent clinical observations and experimental studies have produced evidence showing the reversal of pathological cardiac hypertrophy. Left ventricle assist devices used in heart failure patients for bridging to transplantation not only improve peripheral circulation but also often cause reverse remodeling of the geometry and recovery of the function of the heart. Dietary supplementation with physiologically relevant levels of copper can reverse pathological cardiac hypertrophy in mice. Angiogenesis is essential and vascular endothelial growth factor (VEGF) is a constitutive factor for the regression. The action of VEGF is mediated by VEGF receptor-1, whose activation is linked to cyclic GMP-dependent protein kinase-1 (PKG-1) signaling pathways, and inhibition of cyclic GMP degradation leads to regression of pathological cardiac hypertrophy. Most of these pathways are regulated by hypoxia-inducible factor. Potential therapeutic targets for promoting the regression include: promotion of angiogenesis, selective enhancement of VEGF receptor-1 signaling pathways, stimulation of PKG-1 pathways, and sustention of hypoxia-inducible factor transcriptional activity. More exciting insights into the regression of pathological cardiac hypertrophy are emerging. The time of translating the concept of regression of pathological cardiac hypertrophy to clinical practice is coming.
Keywords: Angiogenesis, HIF-1, hypertrophy, LVADs, PKG-1, regression, VEGF, VEGF Receptors
1. Introduction
The simple definition of cardiac hypertrophy is the enlargement of the heart. This enlargement results from the growth of the adult heart in response to an increase in load. Physical exercise and pregnancy can lead to cardiac hypertrophy along with normal or enhanced contractile function (Weeks & McMullen, 2011), which is defined as physiological cardiac hypertrophy and is reversible. In response to chronic pressure or volume overload under certain disease conditions such as hypertension, valvular heart disease, and coronary artery disease, the heart becomes hypertrophic (Opie et al., 2006; Weeks & McMullen, 2011). But this hypertrophy is associated with further development to cardiac dysfunction or heart failure, so that it is defined as pathological cardiac hypertrophy, which has been considered to be irreversible.
The traditional view of the irreversibility of pathological cardiac hypertrophy has recently been challenged. Clinical observation of left ventricular assist devices (LVADs) for bridging to transplantation has shown that some pathological changes in the heart can be reversed after a certain period of LVADs support. In some cases, the pathological cardiac hypertrophy can be reversed and the contractility of the heart can be recovered (Cai et al., 2009; Li et al., 2008; Moens et al., 2008). These observations provide a novel therapeutic option for heart failure patients, namely LVADs therapy for heart failure (Baughman & Jarcho, 2007; Birks et al., 2011; Maybaum et al., 2007; Strueber et al., 2011). Experimental studies have produced strong evidence that shows improvement of angiogenesis in hypertrophic heart can lead to regression of pathological cardiac hypertrophy and the prevention of heart failure (Jiang et al., 2007; Zhou et al., 2008, 2009a). With regard to this, the role of the trace element copper in the regulation of hypoxia-inducible factor-1 (HIF-1) transcriptional activity and the subsequent effects on the expression of genes involved in angiogenesis has become an interesting field of cardiac research (Borkow et al., 2010; Feng et al., 2009; Xie & Kang, 2009; Xie & Collins, 2011). Experimental studies have shown that supplementation with physiologically relevant levels of copper can reverse heart hypertrophy and improve cardiac function in a mouse model of pressure overload-induced pathological cardiac hypertrophy (Jiang et al., 2007).
How does the regression of pathological cardiac hypertrophy occur under the aforementioned clinical and experimental conditions? Comprehensive understanding of this novel aspect of cardiac research and medicine has not been available, but recent studies have produced some interesting insights. A critical pathway involving cyclic guanosine monophosphate (cGMP)-dependent protein kinase-1 (PKG-1) has been shown to be the major player in the regression of pathological cardiac hypertrophy (Takimoto et al., 2005). It has also been extensively demonstrated that vascular endothelial growth factor (VEGF) plays an essential role in triggering the regression pathway. This pathway is different from that of VEGF-stimulated cardiac hypertrophy in which VEGF is also essential. The regression pathway is mediated by VEGF receptor-1, but the hypertrophic pathway is mediated by VEGF receptor-2. Copper is a critical element in switching the VEGF pathways from VEGF receptor-2-dependent to receptor-1-dependent in the regression of pathological cardiac hypertrophy (Zhou et al., 2009a)).
Regression of pathological cardiac hypertrophy opens a new avenue of cardiac medicine. Currently clinical approaches basically focus on prevention of cardiac hypertrophy by blocking the hypertrophic signaling pathways. However, in most cases, pathological cardiac hypertrophy is a secondary development derived from primary cardiac diseases such as hypertension, valvular heart disease, and coronary artery disease. Exploring molecular targets that are involved in the regression of pathological cardiac hypertrophy would generate alternative and probably more effective remedies for heart failure patients.
In this review, a brief summary of biochemical and functional changes in pathological cardiac hypertrophy will be presented to outline the distinct features of this pathological process. This will be followed by discussion of clinical observations and experimental studies demonstrating the use of LVADs in the regression of pathological cardiac hypertrophy. Then, the discussion will be focused on the role of modulation of angiogenesis in the process of the regression, signaling pathways associated with the promotion of the regression, and identification of potential therapeutic targets to promote the regression. Hopefully, this review will provide a timely understanding of the new direction of cardiac research and medicine.
2. Distinct features of pathological cardiac hypertrophy
The distinction between physiological and pathological cardiac hypertrophy is difficult to define from anatomic features; both types of cardiac hypertrophy can be observed in the form of either concentric or eccentric; the former is characterized by an increase in wall thickness with no change or slight reduction in chamber volume and the latter is an increase in chamber volume with no or small change in wall thickness (Mihl et al., 2008). At the cellular level, concentric hypertrophy results from an increase in myocardial cell width due to a parallel addition of sarcomeres in the cell (Libonati, 2011). Eccentric hypertrophy results from an increase in myocardial cell length due to a series addition of sarcomeres in the cell (Libonati, 2011). Furthermore, increased mitochondrial dynamics in hypertrophic cardiomyocytes is described as an important feature for both types of cardiac hypertrophy. However, there are distinguishable changes between the two types of cardiac hypertrophy. The foremost difference is defined by the functional alteration; physiological hypertrophy is characterized by normal or enhanced cardiac contractile function whereas pathological hypertrophy shows decreased contractility often associated with arrhythmia. The cardiac dysfunction reflects the distinct pathogenesis of pathological cardiac hypertrophy. Experimental approaches have focused on the signaling pathways by which various stimuli can trigger physiological and pathological cardiac hypertrophy. There are many interesting studies and review articles on this topic (Abel & Doenst, 2011; Backs et al., 2009; Heineke et al., 2010; Luo et al., 2012; Suckau et al., 2009; van Berlo et al., 2010; Yang et al., 2012; Zhong et al., 2010), which will not be covered in this review. Some of the key features of physiological versus pathological cardiac hypertrophy are summarized in Table 1.
Table 1.
Characteristics of pathological cardiac hypertrophy in comparison with physiological cardiac hypertrophy.
| Characteristics | Pathological cardiac hypertrophy |
Physiological cardiac hypertrophy |
|---|---|---|
| Morphological changes | Increase in wall-thickness Increase in organ mass |
Increase in wall-thickness Increase in organ mass |
| Addition of new sarcomeres | Addition of new sarcomeres | |
| Cardiomyocyte dynamics | Apoptosis | - |
| Necrosis Increased efferocytosis |
- - |
|
| Increased autophagy | Increased autophagy | |
| ECM alterations | Increased collagen I deposition |
- |
| Increased collagen III deposition Interstitial fibrosis |
- - |
|
| Increased MMPs | - | |
| Increased TIMPs | - | |
| Myocardial metabolism | Decreased FA oxidation | Increased FA oxidation |
| Increased glucose oxidation | Increased glucose oxidation | |
| Decreased PCr | - | |
| Fetal gene expression | Up-regulation of ANP, BNP, β-MHC, and α–skeletal actin |
- - |
| Genes related to contractility functional alterations |
Down-regulation of SERCA2a, and α-MHC Decreased contractility |
- - Normal or enhanced contractility |
2.1. Interstitial fibrosis in pathological cardiac hypertrophy
The extracellular matrix (ECM), mainly fibrillar collagen, provides structural integrity to adjoining cardiomyocytes. This not only functions as a structural support for the myocardial tissue, but also facilitates myocyte shortening, which translates into efficient cardiac pump function (Baicu et al., 2003; Cleutjens & Creemers, 2002; Pelouch et al., 1993). The proportional distribution or deposition of fibrillar collagen in the heart is critical for cardiac contractile function. One of the unique features of pathological cardiac hypertrophy is increased interstitial fibrosis, or excessive deposition of fibrillar collagen. This excessive collagen deposition is not found in physiological cardiac hypertrophy. Most studies have demonstrated that in the hypertrophic heart of hypertension patients, there is increased diffuse fibrosis in which types I and III collagens are predominant (Brilla & Maisch, 1994; Diez et al., 2001; Gonzalez et al., 2002; Harada et al., 2007). The excessive collagen deposition is related to changes in the activity of fibroblasts under the condition of pathological stimulations.
Fibroblasts synthesize pro-collagen that is secreted into the interstitial space where it is split by pro-collagen N- and C-proteinases (PCP) in the end-terminal pro-peptide sequences to enable collagen fiber formation (Weber, 1997). Myocardial inflammation is an early response to pathological stimulation. Cytokines and chemokines produced under the inflammatory condition further augment the inflammatory effects as well as stimulate counteracting responses. Under the inflammatory condition, cell loss due to apoptosis and necrosis triggers compensatory responses. All of these together significantly increase the activity of fibroblasts, leading to excessive deposition of fibrillar collagens. Many studies have demonstrated the role of transforming growth factor-β (TGF-β) and tumor necrosis factor-α (TNF-α) in the initiation and progression of cardiac interstitial fibrosis (Bujak & Frangogiannis, 2007; Khan & Sheppard, 2006; Schultz Jel et al., 2002; Sun et al., 2007). Recent studies have shown microRNAs also participate in the regulation of cardiac fibrogenesis. Myocardial infarction leads to a down-regulation of the miR-29 family microRNAs. The miR-29 family targets mRNAs that encode proteins involved in cardiac fibrosis, including collagen production and deposition, dynamics of fibrillins, and activities of elastin. Therefore, down-regulation of miR-29 relieves the repression of these mRNAs, leading to enhanced cardiac fibrosis (Khan & Sheppard, 2006; van Rooij et al., 2008).
Collagen turnover is mainly regulated by matrix metalloproteinases (MMPs) and the expression of these proteins is significantly up-regulated in pathological cardiac hypertrophy. Although the production of MMPs is increased, the net deposition of fibrillar collagens remains predominant. This is due to the production of tissue inhibitors of metalloproteinases (TIMPs) accompanying the increase in MMPs. These inhibitors function in preventing the degradation of fibrillar collagens by MMPs, ensuring the formation of interstitial fibrosis. Therefore, the ratio of MMPs/TIMPs maintains the equilibrium of collagen deposition and degradation within the myocardium (Cleutjens, 1996).
2.2. Myocardial cell loss in pathological cardiac hypertrophy
Prolonged pressure or volume overload in the setting of cardiovascular disease leads to myocardial cell death in the modes of both apoptosis and necrosis. The myocardial cells that survive the insults become adaptive both in their structure and function. The initial adaptation includes hypertrophy, which is considered to be compensatory in the early phase but transitions to pathological in the constant presence of the insults. In the pathogenesis of pathological cardiac hypertrophy, apoptosis was found to be critically involved (Gottlieb et al., 1994), which has been demonstrated in heart failure patients (Olivetti et al., 1997).
Many in vivo studies have shown that only a very small percentage of myocardial cell populations undergo apoptosis under pathological conditions. For example, less than 0.5% of cells appeared apoptotic in myocardial tissue in the hypertrophic heart of a mouse model (Kang et al., 2000). At first glance, this number seems to be too insignificant to account for myocardial pathogenesis. In a carefully designed time–course study (Kajstura et al., 1996), it was estimated that cardiomyocyte apoptosis is completed in less than 20 hours in rats. Myocytes that undergo apoptosis are lost and may not be replaced under pathological conditions. Although the possibility of myocardial regeneration has been identified (Anversa et al., 2006; Beltrami et al., 2003; Leri et al., 2005; Nadal-Ginard et al., 2003), inhibition of regeneration or degenerative activities often become predominant under myocardial disease conditions (Bicknell et al., 2007; Buja & Vela, 2008).
Myocardial cell death is a critical event in myocardial infarction, which had been considered as a consequence of necrosis (Eliot et al., 1977). It is now recognized that apoptosis contributes significantly to myocardial infarction (Yaoita et al., 2000). Apoptosis and necrosis were originally described as two distinct modes of cell death that can be clearly distinguished (Wyllie, 1994). However, the processes of apoptosis and necrosis are exchangeable. There is a critical control point for a cell to undergo apoptotic pathway. If the apoptotic program is aborted before this control point and the trigger event is severe, cell death may occur by necrosis (Leist et al., 1997). Therefore, the triggering events can be common, but a downstream controller determines the pathway of cell death, which is related to the intensity and duration of insults.
The removal of apoptotic cells is an important process of the cell death program. Efferocytosis is the process by which apoptotic cells are removed by phagocytic cells, which can be regarded as the burying of dead cells (deCathelineau & Henson, 2003). During this process, the cell membrane of phagocytic cells engulfs the apoptotic cell to form a large fluid-filled vesicle containing the dead cell. This vesicle is called an efferosome. Efferocytosis removes dead cells before their membrane integrity is completely lost and the membrane ruptured, so that the contents of apoptotic cells will not leak into the surrounding tissue. This prevents toxic and autoimmune responses due to the exposure to the contents of apoptotic cells (Vandivier et al., 2006). There are “eat me” signals on the membrane of apoptotic cells, such as phosphatidyl serine and calreticulin (Gardai et al., 2005). These signals distinguish apoptotic cells from living cells and guide the process of efferocytosis.
Cell death leads to myocardial cell loss in pathological cardiac hypertrophy. It cannot be excluded that cell death also occurs in physiological cardiac hypertrophy. However, the capacity of myocardial regeneration remains in physiological cardiac hypertrophy so that the replacement of lost myocardial cells may take place. This capacity in pathological cardiac hypertrophy is inhibited and the replacement does not occur.
2.3. Autophagy in pathological cardiac hypertrophy
Autophagy is a “housekeeping” process whereby damaged and dysfunctional cellular organelles and protein aggregates are phagocytosed to produce energy during metabolic stress (Levine & Klionsky, 2004). Autophagy occurs in all eukaryotic cells under starvation, hypoxia, and toxic insults, as well as in response to hormones and developmental signals. There are two forms of autophagy, called selective autophagy and nonselective autophagy. Selective autophagy of mitochondria is termed mitophagy, which is triggered by opening of the mitochondrial permeability transition pore and loss of mitochondrial membrane potential (Tolkovsky, 2009). In cardiomyocytes, mitophagy is a continuous process of mitochondrial turnover, but the rate of this turnover is influenced by stress and makes a critical contribution to myocardial pathogenesis. Nonselective autophagy has been observed in response to nutrient starvation; the degradation of cytosolic components including mitochondria provides amino acids and lipid substrates for intermediary metabolism (Tolkovsky, 2009).
Autophagy is a highly coordinated process, during which dysfunctional organelles and damaged proteins are encircled in a double-membrane vesicle called autophagosome for the delivery to lysosomes, and the contents are hydrolyzed to produce amino acids, fatty acids, and substrates for ATP generation (Klionsky & Emr, 2000; Z. Xie & Klionsky, 2007; Yoshimori, 2004). Autophagy can be divided into 4 steps: (1) initiation of autophagy in response to triggers; (2) formation of autophagosomes; (3) fusion of autophagosomes with lysosomes; and (4) degradation of autophagic contents (Klionsky & Emr, 2000; Xie & Klionsky, 2007). There are multiple proteins involved in the molecular machinery and signaling pathways in autophagy (Shintani & Klionsky, 2004; Wang & Klionsky, 2003). These proteins are found in cardiomyocytes (Gustafsson & Gottlieb, 2009). It has been shown that a basal, constitutive level of autophagy is essential for normal cardiac structural integrity and biological function (Nakai et al., 2007), and up-regulation of autophagy takes place in response to cardiac pressure overload (Halestrap et al., 2004; Paillard et al., 2009) and ischemia-reperfusion (Hamacher-Brady et al., 2006; Kyosola, 1981; Yan et al., 2005).
The up-regulation of autophagy in the setting of cardiac disease conditions is related to (1) nutritional deprivation and depletion of high energy phosphate stores (Inoki et al., 2005); (2) increases in intracellular Ca2+ concentrations (Brady et al., 2007; Hoyer-Hansen et al., 2007; Yitzhaki et al., 2009); (3) generation of reactive oxygen species (Becker et al., 1999; Chen et al., 2007, 2009; Scherz-Shouval et al., 2007a; Scherz-Shouval et al., 2007b; Vanden Hoek et al., 1997; Yang et al., 2008); (4) opening of the mitochondrial permeability transition pore (Brady et al., 2006; Gustafsson & Gottlieb, 2009; Halestrap et al., 2004); and (5) activation of a hypoxia-responsive protein Bnip3 (Bcl-2/adenovirus E1B 19-kilodalton interacting protein) (Kubasiak et al., 2002; Regula et al., 2002). In addition, ER stress, accumulation of damaged or misfolded proteins, and increases in dysfunctional organelles further enhance autophagy (Minamino & Kitakaze, 2010; Qi et al., 2007; Severino et al., 2007; Thuerauf et al., 2006). While these triggers of autophagy in the heart are well documented, the critical issue is whether up-regulation of autophagy is protective or detrimental.
The role of autophagy in cardiac pathogenesis is complex. At present, it is commonly accepted that whether autophagy is beneficial or detrimental depends on the context and amplitude of the induction (Kuma et al., 2004; Yue et al., 2003). For instance, when the cell is starved of energy due to ischemia, activation of autophagy is protective (Gottlieb et al., 2009). However, activation of autophagy during post-ischemic reperfusion is maladaptive (Gottlieb et al., 2009). In the load-stressed heart, activation of autophagy is detrimental (Halestrap et al., 2004). Regardless of being beneficial or detrimental, the increase in autophagy in response to various stresses is a natural process of the heart. However, overt enhancement of autophagy contributes to pathogenesis of pathological cardiac hypertrophy.
2.4. Metabolic disorder in pathological cardiac hypertrophy
ATP is the most important molecule in myocardial metabolism. In the heart, the primary ATP-utilizing reactions are catalyzed by actomyosin ATPase in the myofibril, the Ca2+-ATPase in the SR, and the Na+,K+-ATPase in the sarcolemma (Ingwall & Weiss, 2004). ATP is also needed for molecular synthesis and degradation in the heart as the same as in other organ systems. ATP synthesis by oxidative phosphorylation in the mitochondria is usually sufficient to support the normal needs of the heart, even when the work output of the heart increases three- to five-fold (Ingwall & Weiss, 2004). In addition, the glycolytic pathway and the tricarboxylic acid cycle also make small contributions to ATP synthesis. The concentration of ATP does not define the energetic state of the heart. The amount of ATP made and used at any given time is many times greater than the size of the measurable ATP pool (Ingwall & Weiss, 2004). Thus, cardiac myocytes contain high concentrations of mitochondria, which ensure that ATP remains constant through oxidation of a variety of carbon-based fuels for ATP synthesis under different conditions.
A unique feature in energy metabolism of the heart is the use of energy reserve systems, such as phosphocreatine (PCr), to maintain a high phosphorylation potential to drive ATPase reactions under highly demanding conditions (Ingwall et al., 1985). PCr exists in the heart at twice the ATP concentration (Bittl & Ingwall, 1985). The enzyme creatine kinase (CK) transfers the phosphoryl group between ATP and PCr at a rate about 10 times faster than the rate of ATP synthesis by oxidative phosphorylation. The reaction catalyzed by CK is: PCr + ADP + H+ ↔ creatine + ATP. Under the conditions when ATP demand exceeds ATP supply, the use of PCr is a major pathway to maintain a constant supply of ATP. The CK reaction is also important to maintain low ADP and Pi concentrations, thereby retaining high phosphorylation potential (Saupe et al., 2000). Creatine is not made in the heart but accumulates against a large concentration gradient facilitated by a creatine transporter. In the normal heart, about two-thirds of the total creatine pool is phosphorylated through the CK reaction to form PCr (Neubauer et al., 1999; Wallimann et al., 1998).
The continuous synthesis of ATP via mitochondrial oxidative phosphorylation is mandatory for the work of the heart (Huss & Kelly, 2005). Under normal conditions, the oxidation of fatty acid (FA) is the major pathway, providing about 70% of the total energy demand. In contrast, the oxidation of glucose provides about 30% of the total energy demand (Shipp, 1961; Wisneski et al., 1987). In physiological cardiac hypertrophy, both FA and glucose oxidation are enhanced. However, in pathological cardiac hypertrophy, there is a metabolic shift from FA- to glucose-dependent energy supply. Thus, decreased FA oxidation and increased glucose utilization in association with depressed FA deposition and increased glucose uptake are observed in pathological hypertrophic and failing hearts (van Bilsen et al., 2004). This shift enhances the glycolytic pathway, and thus increases anaerobic metabolism. It remains uncertain whether this metabolic shift to the so-called “fetal phenotype” is adaptive or maladaptive. It is important to note that the “shift” is only partial, and even when the proportion of ATP synthesized from glucose increases many fold, aerobic metabolism still remains dominant (van Bilsen et al., 2004).
2.5. Fetal gene expression in pathological cardiac hypertrophy
Fetal gene expression in pathological cardiac hypertrophy has been extensively investigated for many years (Park et al., 2011; Sheehy et al., 2009; Tanno et al., 2011; Zhao et al., 2004). The most studied and well-characterized fetal genes whose expression is significantly up-regulated in myocardial pathogenesis include atrial natriuretic peptide (ANP), B-type natriuretic peptide (BNP), and α-skeletal actin (Holtwick et al., 2003; McKie et al., 2010; Stilli et al., 2006). With the increase in the expression of these fetal genes, a simultaneous down-regulation of genes involved in critical function of cardiomyocytes takes place. For instance, the calcium-handling protein sarco(endo)plasmic reticulum Ca2+-ATPase 2A (SERCA2a) is among the depressed proteins found in pathological cardiac hypertrophy (Song et al., 2008). In addition, alterations in cardiac contractile proteins, such as decreased α-myosin heavy chain (α-MHC) and increased β-myosin heavy chain (β-MHC) proteins, are also found in pathological cardiac hypertrophy.
The progression of these distinct biochemical and molecular changes in pathological cardiac hypertrophy discussed above leads to myocardial dysfunction and eventual heart failure. This progression involves myocardial remodeling that has been believed to propagate forwardly, resulting in irreversible pathogenesis. However, recent clinical observations and experimental studies have accumulated evidence showing that reverse remodeling occurs and the pathological cardiac hypertrophy is reversible. In this context, the use of left ventricular assist devices has generated strong evidence as discussed in the next section.
3. Left ventricular assist devices (LVADs) in the regression of pathological cardiac hypertrophy
Myocardial remodeling has been a major topic in pathological cardiac hypertrophy. Left ventricular remodeling is the process involving alterations in ventricular size, shape, protein content and composition, and function resulting from the action of mechanical, neurohormonal, and genetic factors (Gao et al., 2005; Huang et al., 2004b; Sedmera et al., 2005). The progression of this remodeling leads to the transition from pathological cardiac hypertrophy to heart failure. Many efforts have been devoted to understand the mechanisms of heart failure. In this context, the hemodynamic model of heart failure explains the effect of an altered load on the failing left ventricle, which results in use of vasodilators and inotropic agents (Berenji et al., 2005; Carroll & Tyagi, 2005; Nishikimi et al., 2005; Wang et al., 2004). The neurohormonal model investigates the importance of the activation of the renin-angiotensin-aldosterone axis and the sympathetic nervous system in the progression of cardiac dysfunction, which leads to the use of angiotensin converting enzyme (ACE) inhibitors and angiotensin-receptor antagonists (Pasini et al., 2004; Sethi et al., 2004; Somogyi et al., 2005; Xu et al., 2004) for the treatment of heart failure. Molecular mechanistic understanding of heart failure has recognized an autocrine and paracrine function of the neurohormonal factors (Chatterjee, 2005; Davila et al., 2005; Kamath et al., 2005; Kaye & Esler, 2005; Opgaard & Wang, 2005; Vonder Muhll et al., 2004) dictating an effect on myocardium itself in addition to circulation. The use of left ventricular assist devices (LVADs) has provided alternative insights into regression of heart failure.
3.1. Left ventricular assist devices (LVADs)
End-stage heart failure is currently viewed as an irreversible condition for which current medical therapy offers only temporary symptomatic relief. Patients with end-stage heart failure rely on transplantation to reduce long-term morbidity and to improve survival (Herrington & Tsirka, 2004; Meine & Russell, 2005; Spann & Van Meter, 1998). Due to the limitation of donors and a long waiting list, left ventricular assist devices (LVADs) have been used as a bridge to transplantation (Acker, 2004; Haddad et al., 2005; Kherani & Oz, 2004; Nemeh & Smedira, 2003; Westaby, 2004). This truly novel engineering concept has been further advanced in response to the need for new heart failure therapy. In particular, the HeartMate® (Thoratec Inc) left ventricular assist system (LVAS) has been approved by the U.S. Food and Drug Administration in 2002 as a destination therapy for patients ineligible for cardiac transplantation.
Serendipitously, the clinical experience with LVADs has suggested that the use of LVADs not only has become a reliable means of sustaining medically refractory patients with heart failure awaiting cardiac transplantation, but also implicated the potential as a bridge to recovery (Bick et al., 2005; Dandel et al., 2005; Entwistle, 2004; Wohlschlaeger et al., 2005; Zhang & Narula, 2004). An early report in 1994 documented the improvement of cardiac parameters in a transplant candidate living in a hospital with the support of a LVAD (Frazier, 1994). The patient lived for 505 days on LVAD support. Since the first report, numerous case studies have demonstrated a consistent improvement in survival and improved quality of life in patients supported by several different LVADs (Hon & Yacoub, 2003; Massie, 2002; Rose et al., 2001; Salzberg et al., 2003). In a retrospective analysis of a large cohort of patients who were supported by LVADs as a bridge to transplantation, 5% of the patients were weaned successfully so that a transplant was no longer needed (Mancini et al., 1998). This is a surprising discovery because the patients who were selected to receive LVADs support were based on the criteria that define LVADs as a bridge to transplantation, not to recovery. Many studies thus have focused on the phenomenon of LVAD-assisted improvement in cardiac structural and functional parameters. “Reverse remodeling” of the heart with LVAD support has been observed, as described in the following sections.
3.2. Reversal of myocardial hypertrophy by LVAD support
In a study specifically addressing the effect of LVAD support on human left ventricle hypertrophy, Zafeirides et al. (1998) observed that in 6 patients for whom echocardiographs were obtained before and after LVAD support for an average of 75 days, left ventricular mass decreased almost 45% and left ventricular end-diastolic diameter decreased by more than 25%. Other clinical studies using echocardiograph also showed a decrease in left ventricular end-diastolic dimension and left ventricular mass (Heerdt et al., 2000; Ogletree-Hughes et al., 2001). LVADs appear to offer long-term survival with an acceptable quality of life to the LVAD-implanted patients (Frazier et al., 1996). In some LVAD patients who required removal of the device because of device failure, native cardiac function could be improved sufficiently to avoid the second device replacement or transplantation (Zafeiridis et al., 1998). Histopathologic analysis also supported the reversal of cardiac hypertrophy in the LVAD-support patients (Barbone et al., 2001; Dipla et al., 1998).
Studies using isolated cardiac myocytes to evaluate changes in myocyte size, shape, and heterogeneity after sustained LVAD support demonstrated that the average volume of failing human myocytes was nearly twice as large as that observed in nonfailing myocytes. LVAD support caused a 60% reduction in the size of hypertrophied cardiomyocytes, a 62% regression of the increase in average myocyte length, and a decrease in the heterogeneity of myocyte length associated with advanced heart failure (Zafeiridis et al., 1998). This observation was followed by another report demonstrating that the decrease in myocyte size during LVAD support was not observed in the minimally unloaded right ventricle (Barbone et al., 2001). Other studies also showed that long-term LVAD support resulted in a 28% reduction in cardiomyocyte volume, 20% reduction in cell length, 20% reduction in cell width, and 32% reduction in cell length-to-thickness ratio. These cellular changes were associated with reductions in left ventricular dilation and left ventricular mass measured by echocardiograph (Hon & Yacoub, 2003; Salzberg et al., 2003).
There are more studies that provided evidence showing that LVADs induce the reversion of cardiomyocyte hypertrophy, a process described as “reverse remodeling” (Popovic et al., 2005). However, there were also studies reporting that the LVAD unloading did not cause cardiomyocyte atrophy, but resulted in increased microvascular density (Massie, 2002). Given that human myocyte hypertrophy is associated with nuclear polyploidization, Rivello et al. examined myocyte diameter, nuclear size and DNA content with static cytomorphometry on tissue sections of the myocardium before and after the implantation of a LVAD, and found that the use of LVADs was associated with a reduction in cardiomyocyte diameter and a marked decline in nuclear DNA content (Ogletree-Hughes et al., 2001).
3.3. Reversal of myocardial contractile dysfunction by LVAD support
Several studies have demonstrated that myocardial contractile function can be significantly improved after a sustained LVAD support. Using isolated cardiomyocytes, Dipla et al. (1998) observed a significant improvement in the fractional shortening and in the rate of shortening and relaxation after 1 to 6 months of LVAD support. Another study using isolated cardiac muscle strips reported a 38% improvement in developed force after LVAD support (Heerdt et al., 2000). The latter study was conducted using a paired tissue analysis in which the muscle strips were obtained from left ventricular apical core before LVAD support and the post-LVAD tissue strips were obtained from the left ventricular free wall. However, there were also studies that demonstrated neither alterations in the developed tension nor changes in contractile parameters under basal conditions after LVAD support, but improved adrenergic responses were observed (Ogletree-Hughes et al., 2001). Several studies also examined the effect of LVAD support on left ventricle contractile function in vivo. One study performed by Frazier et al. showed an improvement of the left ventricle ejection fraction from 11 ± 5% to 22 ± 17% after LVAD support (Frazier et al., 1996). Other studies also demonstrated improved left ventricle ejection fraction after LVAD support (Loebe et al., 1999; Muller et al., 1997).
Impaired calcium handling is an important mechanism for decreased contractility of cardiomyocytes. Thus, several studies have explored the possibility of improvement in myocardial calcium handling after LVAD support. Dipla et al. using isolated myocytes identified changes in the shape of the calcium transient in association with changes in myocyte shortening after LVAD support (Dipla et al., 1998). Other investigators have shown that calcium uptake rate and binding in isolated sarcoplasmic reticulum membranes were improved after LVAD support (Frazier et al., 1996; Heerdt et al., 2000). These results were accompanied by the finding that LVAD support increased mRNA abundance for sarcoplasmic reticulum ATPase, ryanodine receptor, and sodium and calcium exchanger (Nishikimi et al., 2005). These molecules are important for calcium handling and their defects have been recognized in heart failure. Therefore, LVAD-assisted improvement in calcium handling may result from multiple effects on the regulation of expression and function of key proteins.
3.4. Reversal of impaired beta-adrenergic responsiveness by LVAD support
Impaired myocardial adrenergic responsiveness is a characteristic feature of heart failure and is responsible for poor exercise tolerance. While examining the effect of LVAD support on basal contractile function, several studies also investigated the effects of LVAD on myocardial adrenergic responsiveness. Using isolated myocytes, Wisneski et al. showed that LVAD support significantly enhanced the shortening response to isoproterenol stimulation (Wisneski et al., 1987). In the study described above using cardiac muscle strips, Ogletree-Hughes et al. have reported that in response to isoproterenol a marked improvement in peak developed tension, +dP/dt, and −dP/dt, was observed from the LVAD supported failing muscle strips relative to before LVAD support (Ogletree-Hughes et al., 2001). Furthermore, the reversal of impaired adrenergic responsiveness by LVAD support was suggested by the fact that the improved response to isoproterenol observed in the LVAD-supported failing heart muscle strips was essentially the same as that observed in human cardiac trabeculae from non-failing hearts (Ogletree-Hughes et al., 2001).
It has been observed that gradual normalization of the increased circulating epinephrine and norepinephrine levels in heart failure patients takes place during the first 1 to 2 months after the initiation of LVAD support (Loebe et al., 1999; Muller et al., 1997). LVAD support also resulted in normalization of myocardial beta-receptor density in failing human hearts, which was decreased in failing hearts before LVAD support in comparison with non-failing hearts (Ogletree-Hughes et al., 2001). Because these factors are critically involved in the initiation and progression of heart failure, their normalization would make a significant contribution to the reversal of impaired adrenergic responsiveness.
3.5. Reversal of abnormal cardiac electrophysiology by LVAD support
Heart failure is associated with a prolongation of QT interval observed from electrocardiograph, which results from prolongation of the action potential duration of cardiomyocytes. Harding et al. examined electrophysiological alterations after the mechanical circulatory support from LVAD in patients with advanced heart failure (Harding et al., 2001). It was observed that there were two phases of LVAD effects on QT intervals. Cardiac unloading caused an immediate increase in the heart rate-adjusted QT interval despite an immediate decrease in QRS duration. However, there was a secondary decrease in QT interval during sustained LVAD support, which paralleled decreases in the action potential duration measured from isolated human ventricular myocytes. This secondary decrease in QT interval by sustained LVAD support suggests a reversal of the electrophysiological abnormality associated with heart failure.
3.6. Reverse or forward remodeling of myocardial extracellular matrix (ECM)
The results obtained from studies examining the effect of LVAD support on myocardial fibrosis in failing hearts are controversial, ranging from no effect (Kinoshita et al., 1996; Li et al., 2001) to increase in fibrosis (Frazier et al., 1996; Madigan et al., 2001; McCarthy et al., 1995; Scheinin et al., 1992) and to reversal of fibrosis (Bruckner et al., 2001). However, there are critical issues that need to be addressed before these data can be compared. Among these issues are sampling procedures, different measurement techniques, duration of LVAD support, etiology of heart failure, and adjuvant therapies. Bruckner et al. examined paired myocardial samples of 18 patients with end-stage cardiomyopathy obtained at the time of LVAD implantation and removal and found that total collagen content was reduced by 72%, collagen I content decreased by 66% and collagen III content decreased by 62% (Bruckner et al., 2001). In this study, the mean age of patents was 46 ± 3 years (range 24 – 64) and the mean length of LVAD support was 159 ± 25 days (range 18 – 420).
Li et al. reported that after 2 months of LVAD support, myocardial collagen content was unchanged (Li et al., 2001), but the degree of ECM cross-linking was increased in association with decreases in MMP-1 and –9 and increases in TIMP-1 and –3, but no changes in MMP-2 and –3 and TIMP-2 and –4. A time course analysis of LVAD effect on myocardial fibrosis showed that relative collagen content was significantly increased in failing hearts supported with LVAD for more than 40 days (Madigan et al., 2001). Other studies also showed increased myocardial fibrosis after a relative sustained support by LVAD (Barbone et al., 2001; Scheinin et al., 1992).
3.7. Reversal or alteration of signaling pathways and inhibition of apoptosis by LVAD support
Inhibition of myocardial apoptosis is a common observation in the studies of LVAD-assisted regression of cardiomyopathy. There are a few studies that have elucidated signaling transduction pathways that may lead to myocardial phenotype changes, including inhibition of apoptosis in failing hearts supported by LVAD. In this context, mitogen-activated protein kinase (MAPK) pathways have been studied (Bartling et al., 1999; Flesch et al., 2001). Flesch et al. showed that LVAD support significantly reduced the activity of p44/42 and JNK1/2 but significantly increased p38 activity. These differential changes in MAPKs were associated with significant reductions in cardiac myocyte size and myocyte apoptosis after LVAD support relative to non-LVAD support (Flesch et al., 2001). Bartling et al. showed that the activity of ERK and Akt in failing hearts dramatically decreased after LVAD support, but GSK-3 increased. At the same time, cardiac myocyte apoptosis and myocyte size also significantly decreased after LVAD support (Bartling et al., 1999). However, the link between the changes in the kinase activities and the reductions in myocyte size and apoptosis remains elusive.
3.8. LVADs as a bridge to recovery in heart failure patients
Based upon the studies described above, many efforts have focused on the demonstration of using LVADs for the treatment of end-stage heart failure as a “bridge to recovery” (Boyle et al., 2011; Smedira et al., 2010). Furthermore, long-term circulatory support “destination therapy” has become an increasingly realistic alternative to cardiac transplantation for most of the heart failure patients who are ineligible for transplantation because of associated co-morbidity (Pamboukian et al., 2011). Because the mechanical unloading by LVADs can decrease oxygen consumption and reduce excess load of the left ventricle in heart failure (Baughman & Jarcho, 2007; Blume et al., 2006; Klotz et al., 2008), the use of LVADs improves survival (Kugler et al., 2011; Radovancevic et al., 2007) and cardiac function (Kukucka et al., 2011) for some of the patients who were thought to have irreversible heart failure. While the “destination therapy” is available to more heart failure patients, the bridge to recovery by LVADs is an attractive concept in cardiac medicine.
Reversal of pathological cardiac hypertrophy is associated with improvement in coronary circulation. Recent studies have demonstrated that while cardiac hypertrophic growth is angiogenesis-dependent, regression of cardiac hypertrophy also relies on angiogenesis. The next section will specifically discuss angiogenesis in the regression of pathological cardiac hypertrophy.
4. Angiogenesis and regression of pathological cardiac hypertrophy
It is widely recognized that cardiac hypertrophy is accompanied by the change of coronary angiogenesis. Physiological cardiac hypertrophy is associated with normal or increased density of myocardial capillaries (Hudlicka et al., 1992). Pathological hypertrophy is accompanied by enhanced coronary angiogenesis at the initial stage of cardiac hypertrophy (Huo et al., 2007; Kassab et al., 1993, 2000). However, the capillary density obviously decreases in the late phase of pathological cardiac hypertrophy (Anversa et al., 1986; Flanagan et al., 1991; Hamasaki et al., 2000; Hudlicka et al., 1992; Karch et al., 2005; Shimamatsu & Toshima, 1987). Reduced coronary reserve, defined as the difference between basal flow and maximal flow during infusion of agents such as adenosine, occurs in pathological cardiac hypertrophy (Shimamatsu & Toshima, 1987). Hypertension with left ventricular hypertrophy is associated with both coronary vascular remodeling and attenuated endothelial and non-endothelial coronary flow reserve (Hamasaki et al., 2000).
Inhibition of angiogenesis by suppressing the vascular endothelial growth factor (VEGF) signaling pathway can completely block the enlargement of the heart under hypertrophic stimulation in animal models (Higashikuni et al., 2011; Jiang et al., 2007; Makino et al., 2009; Shiojima et al., 2005). While angiogenesis is essential for the enlargement of the heart, it is quite interesting that the regression of cardiac hypertrophy is also angiogenesis-dependent (Jiang et al., 2007), and VEGF is required for the reduction in the size of hypertrophic cardiomyocytes (Zhou et al., 2008, 2009a; Zuo et al., 2010).
4.1. Disrupted coordination between angiogenesis and cardiac hypertrophy in heart failure
The match of the increase in the density of myocardial capillaries to the hypertrophic growth of the heart muscle is critical to keep cardiac function normal or ensure the functional adaptation to the increase in cardiac load. A study using a conditional transgenic mouse model in which Akt1 gene expression can be specifically activated in the heart showed that Akt activation led to cardiomyocyte hypertrophy (Taniyama et al., 2005). In this system, both physiological and pathological cardiac hypertrophy could be developed through a sequential manner of induction of Akt1 gene expression. Physiological cardiac hypertrophy was induced by a short-term Akt activation and displayed preserved contractile function with enhanced coronary angiogenesis. However, a long-term activation of Akt in the same mouse model resulted in pathological cardiac hypertrophy with reduced contractility and impaired coronary angiogenesis (Taniyama et al., 2005). Therefore, the coordination between coronary angiogenesis and the enlargement of the heart ensures that the hypertrophy is physiological and disruption of this coordination results in the pathogenesis of pathological cardiac hypertrophy and its transition to heart failure.
In the conditional Akt1 transgenic system in mice, coronary angiogenesis is related to mammalian target of rapamycin (mTOR)-dependent expression of VEGF-A and angiopoietin-1 (Taniyama et al., 2005). Detailed analysis has revealed that under physiological hypertrophic conditions, the expression of VEGF-A and angiopoietin-1 is up-regulated and under pathological hypertrophy, the expression of these genes is down-regulated (Taniyama et al., 2005). However, even in the acute induction of Akt expression, which led to physiological cardiac hypertrophy, the inhibition of coronary angiogenesis by a VEGF decoy receptor switched the development of physiological cardiac hypertrophy to the pathogenesis of heart failure, characterized by reduced capillary density along with contractile dysfunction and impaired cardiac growth. Thus, VEGF signaling plays a critical role in coronary angiogenesis, which in turn is essential in the sustention of normal cardiac function under hypertrophic stimulation.
4.2. Angiogenesis is essential for regression of pathological cardiac hypertrophy
Promotion of coronary angiogenesis can lead to reversal of pathological cardiac hypertrophy. In a mouse model of pathological cardiac hypertrophy induced by pressure overload through ascending aortic constriction it was found that the density of myocardial capillaries significantly decreased (Jiang et al., 2007). The decrease in capillary density was associated with suppression of VEGF and a concomitant decrease in the concentration of myocardial copper (Jiang et al., 2007). The expression of VEGF is regulated by hypoxia-inducible factor-1 (HIF-1) and copper is essential for VEGF gene expression because copper is required for HIF-1 transcriptional activity (Feng et al., 2009; Jiang et al., 2007). Importantly, dietary copper supplementation promoted VEGF production and relieved the suppression of coronary angiogenesis leading to regression of pathological cardiac hypertrophy. Moreover, anti-VEGF antibody completely blunted the copper-induced coronary angiogenesis and the subsequent regression of pathological cardiac hypertrophy (Jiang et al., 2007).
In another mouse model of pressure overload-induced pathological cardiac hypertrophy, Takimoto et al. showed that chronic inhibition of cyclic GMP phosphodiesterase 5A (PDE5A) by sildenafil (Viagra) reversed the pre-established pathological cardiac hypertrophy induced by transverse aortic constriction (Takimoto et al., 2005). It was found that the inhibition of PDE5A deactivated multiple hypertrophic signaling pathways triggered by pressure overload, including the calcineurin/NFAT, phosphoinositide-3 kinase/Akt, and ERK1/2 signaling pathway (Takimoto et al., 2005). The inhibition of the hypertrophic signaling pathways would make perfect sense for the inhibition of progression of cardiac hypertrophy, for which it had been shown that PDE5A inhibition indeed prevented pressure overload-induced cardiac hypertrophy (Takimoto et al., 2005). How does PDE5A inhibition relate to regression of cardiac hypertrophy? Subsequent analyses showed that PDE5A inhibition stimulates VEGF and angiopoietin-1 production and promotes coronary angiogenesis (Koneru et al., 2008; Vidavalur et al., 2006). In addition, PDE5A inhibition also activates the cGMP-dependent protein kinase-1 (PKG-1) signaling pathway in cardiomyocytes (Zhang et al., 2010), which will be discussed in next section. Acting together, these effects result in the regression of pathological cardiac hypertrophy.
4.3. Pharmaceutical agents affecting myocardial angiogenesis
Angiotensin-converting enzyme (ACE) inhibitors are pharmaceutical drugs primarily used for the treatment of hypertension and congestive heart failure. Frequently prescribed ACE inhibitors include captopril, enalapril, lisinopril, and ramipril. ACE inhibitors have been shown to be effective for not only patients with hypertension, but also patients with normal blood pressure. Use of a maximum dose of ACE inhibitors for prevention of diabetic nephropathy, congestive heart failure, and prophylaxis of cardiovascular events is justified because it improves clinical outcomes, independent of the blood pressure-lowering effect of ACE inhibitors. Several studies have demonstrated that ACE inhibitors can attenuate the progression of cardiac hypertrophy in experimental models of heart failure by affecting the coronary capillary density and function (Chiba et al., 1994; Ruzicka et al., 1995; Weinberg et al., 1994).
Dipyridamole is a drug that inhibits thrombus formation when given chronically and causes vasodilation when given at high doses over a short period of time. Treatment with dipyridamole has been shown to increase endomyocardial capillary density in cardiac hypertrophy (Torry et al., 1992). It increases myocardial perfusion and left ventricular function in patients with ischemic cardiomyopathy.
NO can modify the structure of vascular tissues by inhibiting the growth of vascular smooth muscle cells. There is an increase in the basal activity of NO (without a significant change in endothelial NO synthase expression) in early compensated left ventricular hypertrophy, followed by a decrease in both endothelial NO synthase expression and NO bioactivity during the transition to heart failure (Grieve et al., 2001). Releasing NO by bradykinin-NO pathway can inhibit the progression of cardiac hypertrophy (Balligand et al., 1993; Brady et al., 1993; Ishigai et al., 1997). These preventing effects on cardiac hypertrophy and on the transition to heart failure are all due to the promotion by NO of myocardial angiogenesis (Janssens et al., 2004; Murohara et al., 1998).
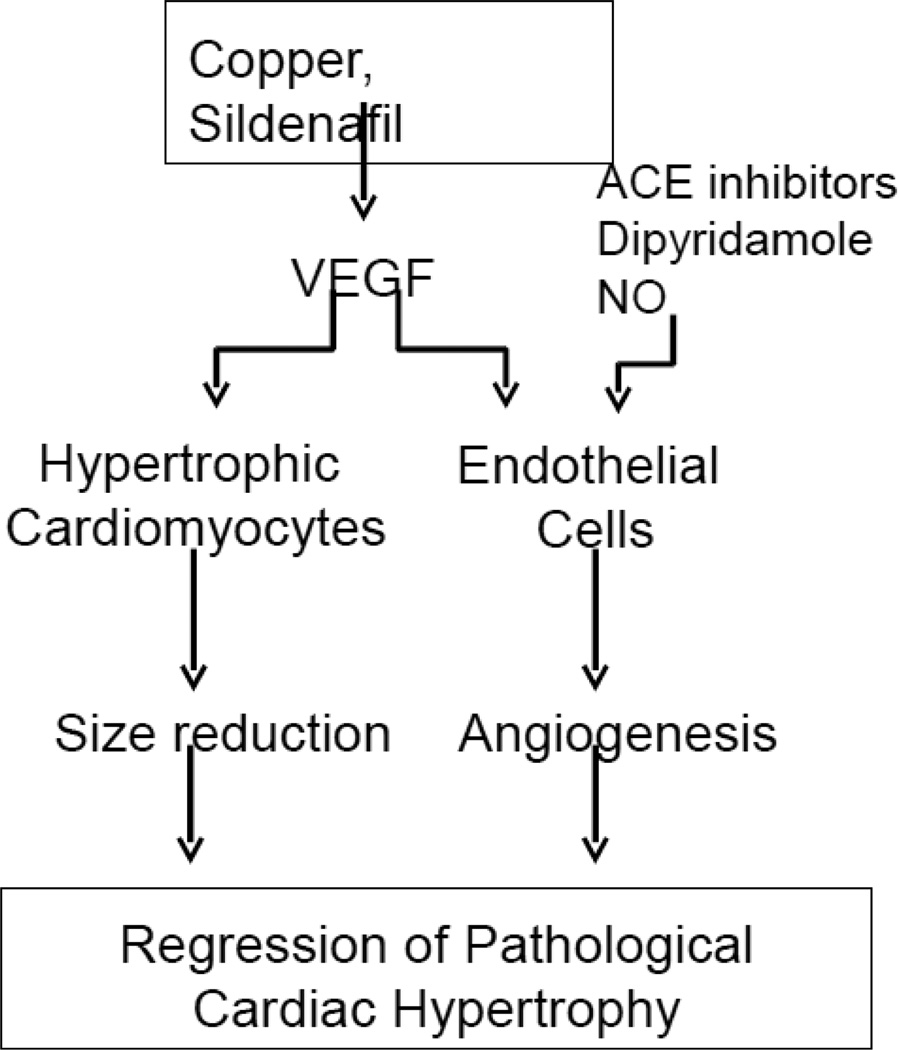
While the essentiality of myocardial angiogenesis has been sufficiently demonstrated, the molecular event leading to reversal of cardiomyocyte hypertrophy is another important topic. Angiogenesis improves the microenvironment of hypertrophic cardiomyocytes, the regression of myocyte hypertrophy is the process occurring inside of the cells. However, both angiogenesis and reversal of cardiomyocyte hypertrophy are VEGF-dependent, as elucidated in Figure 1. The understanding of signaling pathways existing in cardiomyocytes and involved in the reversal of myocyte hypertrophy, independent of signaling pathways leading to angiogenesis, is important for the development of pharmaceutical agents in the regression of cardiac hypertrophy.
Figure 1. VEGF-dependent angiogenesis and reversal of cardiomyocyte hypertrophy.
Both copper and sildenafil cause up-regulation of VEGF expression, which is also affected by ACE inhibitors, dipyridamole, and NO. There are VEGF receptors on both endothelial cells and cardiac myocytes. Activation of the endothelial cells leads to angiogenesis and the response of hypertrophic cardiomyocytes to VEGF results in reduction in cell size, all together promoting the regression of pathological cardiac hypertrophy.
5. Signaling pathways leading to regression of pathological cardiac hypertrophy
The signaling pathways involved in cardiac hypertrophy have been extensively studied and some of the molecules involved in the critical pathways have been the targets of pharmaceutical development (Belmonte & Blaxall, 2011; Care et al., 2007; Gaspar-Pereira et al., 2011; McKinsey & Kass, 2007; Wang et al., 2010; Zhang & Mende, 2011). However, the signaling pathways involved in the regression of cardiac hypertrophy are a relatively new field of study to which investigation has just begun. There are several exciting clues and experimentally-tested signaling molecules that have been identified in the regression of pathological cardiac hypertrophy. Among these are the VEGF-VEGF receptor-1 signaling pathway, activation of PKG-1 pathway, and regulation of HIF-1 transcriptional activity. This review will cover only the current mainstays of the experimentally-tested signaling pathways, although there are other molecules that are also suggested to function in the regression of pathological cardiac hypertrophy. It should be noted that these currently, extensively studied signaling pathways are still far from being comprehensively understood. There are many missing components that need to be identified for the full elucidation of the signaling pathways.
5.1. VEGF-VEGF receptor-1 signaling pathway
VEGF triggers cellular responses through its receptors on the cell membrane. Binding of VEGF promotes the receptors to dimerize and be activated through autophosphorylation, leading to signaling transduction cascades (Hoeben et al., 2004). There are three VEGF receptors (VEGFRs) and each receptor functions differently and coordinately with each other. VEGFR-2 is a dominant component in VEGF-triggered cell growth and differentiation signaling pathways. Activation of VEGFR-2 by VEGF in cells devoid of VEGFR-1 results in a mitogenic response, while the activation of VEGFR-1 in cells lacking VEGFR-2 does not induce cell proliferation (Kliche & Waltenberger, 2001; Nishi et al., 2008; Roberts et al., 2004). VEGFR-2 activates mitogen-activated protein kinase (MAPK) signaling pathway whereas VEGFR-1 cannot activate this pathway (Haddad et al., 2005). Extensive studies performed in endothelial cells suggest that VEGFR-2 mediates most of the known cellular responses to VEGF such as embryonic vasculogenesis and tumor angiogenesis (Sakurai et al., 2005; Takahashi, 2011; Yang & Cepko, 1996). It is traditionally believed that VEGFR-1 regulates VEGFR-2 signaling, either negatively or positively (Clark et al., 1998; Zeng et al., 2001).
VEGF also stimulates cell growth in myocardial tissue (Sano et al., 2007; Shimojo et al., 2007; Shyu et al., 2005; Song et al., 2007; Zisa et al., 2009). A VEGFR-2 antagonist blocks cardiac growth induced by Akt1 activation (Izumiya et al., 2006; Shiojima et al., 2005), indicating a link between the VEGFR-2 and the Akt1 signaling pathway. However, in the hypertrophic myocardium or hypertrophic cardiomyocytes in cultures, VEGF causes regression of hypertrophy (Jiang et al., 2007; Shiojima et al., 2005; Zhou et al., 2008). This suggests that VEGF has a dual function in cardiomyocytes; stimulating cell growth under physiological or stress conditions and reducing the size of cardiomyocytes under hypertrophic conditions.
Expression of the dual function of VEGF needs to be mediated by VEGFRs. The link of VEGFR-2 to the growth stimulation pathway suggests that other receptors would link to the regression pathway. It has been shown that an increase in the ratio of VEGFR-1 to VEGFR-2 in the regression of hypertrophy occurs in cultured cardiomyocytes (Zhou et al., 2009). The cause-effect relationship between the increase in the ratio of VEGFR-1 to VEGFR-2 and the reversal of cardiomyocyte hypertrophy has been demonstrated using siRNA targeting of VEGFR-2 to elevate the ratio of VEGFR-1 to VEGFR-2. This increase in the ratio of VEGFR-1 to VEGFR-2 in hypertrophic cardiomyocytes led to the regression of the hypertrophy (Zhou et al., 2009). This regression was blocked when anti-VEGF antibody was applied to the cells. Furthermore, gene silencing of VEGFR-1 blocked the reversal of cardiomyocyte hypertrophy, further demonstrating the role of VEGF-VEGFR-1 signaling pathway in the regression of pathological cardiac hypertrophy.
VEGFR-1 is associated with PKG-1 activation. The link of VEGFR-1 to the PKG-1 pathway in the regression of heart hypertrophy has been demonstrated in hypertrophic cardiomyocytes (Zhou et al., 2009). The PKG-1 signaling pathway is the most studied and critically important pathway in the regression of heart hypertrophy, as will be discussed in next section. It was observed that the suppression of VEGFR-1 reduced the activity of PKG-1, indicating the activation of PKG-1 is at least in part VEGFR-1 dependent (Zhou et al., 2009). Conversely, a PKG-1 inhibitor, 8-(4-chlorophenylthio)guanosine-3’,5’-cyclic monophosphorothioate, Rp-isomer (Rp-8-pCPT-cGMPS) effectively blocked the reversal of cardiomyocyte hypertrophy induced by the increase in the ratio of VEGFR-1 to VEGFR-2. Therefore, the down-stream players of VEGFR-1 signaling pathway involve PKG-1 in hypertrophic cardiomyocytes. However, it is unknown how VEGFR-1 and PKG-1 interact to ignite this signaling transduction.
The aforementioned inhibition of PDE5A by sildenafil leading to regression of pathological cardiac hypertrophy is related to VEGF up-regulation. In a rat model of myocardial ischemia and reperfusion, it was observed that treatment with sildenafil before occlusion of the left anterior descending artery (ischemia) followed by reperfusion significantly increased capillary and arteriolar density along with increased blood flow. The improvement in angiogenesis was accompanied by the increase in gene expression of VEGF and angiopoietin-1. These changes resulted in a significant reduction in myocardial infarct size, and reduced apoptosis of cardiomyocytes and endothelial cells. These changes were also associated with a significant improvement in cardiac function including an increase in fractional shortening and ejection fraction, and increased contractile reserve (Koneru et al., 2008). Therefore, it was concluded that up-regulation of VEGF and angiopoietin-1 by sildenafil most likely stimulates a cascade of events leading to neovascularization and conferring myocardial protection against injuries by ischemia and reperfusion, although further insights into mechanisms by which sildenafil activates these pathways need to obtained.
Pressure overload-induced cardiac hypertrophy in a mouse model is associated with a loss of myocardial copper concentrations. Dietary supplementation with physiologically relevant levels of copper not only replenishes cardiac copper levels but also reverses cardiac hypertrophy. In searching for potential mechanisms leading to copper repletion-induced regression of cardiac hypertrophy, it was found that copper deficiency suppressed the expression of VEGF in the heart and copper repletion restored VEGF expression (Jiang et al., 2007). The increased VEGF expression was accompanied by increased myocardial capillary density. It was also found that administration of anti-VEGF completely blocked copper supplementation-enhanced angiogenesis and suppressed the reversal of cardiac hypertrophy. Therefore, it was concluded that recovery of VEGF expression and the subsequent angiogenesis contribute to copper supplementation-induced regression of pathological cardiac hypertrophy. Further analysis revealed that copper functions in switching the VEGFR2-dependent to the VEGFR1-dependent signaling pathway in the hypertrophic cardiomyocytes (Zhou et al., 2009).
5.2. Activation of PKG-1 pathway
It has been known that activation of PKG-1 leads to inhibition of myocardial growth (Fiedler et al., 2002; Wollert et al., 2002), and further studies have determined that PKG-1 is involved in the regression of cardiac hypertrophy (Takimoto et al., 2005). Activation of PKG-1 has been observed in pressure overload-induced heart hypertrophy (Takimoto et al., 2005). In a mouse model of cardiac pressure overload induced by transverse aorta constriction (TAC), heart hypertrophy was observed after TAC for 3 weeks. PKG-1 activity was significantly elevated in the hypertrophic myocardium (Takimoto et al., 2005). This activation was considered as a counteracting action to heart hypertrophy induced by pressure overload. However, the PKG-1 activation was not sufficient to prevent the development of heart hypertrophy (Takimoto et al., 2005). However, treatment with an oral PDE5A inhibitor, sildenafil, blocked the intrinsic catabolism of cGMP leading to sustained activation of PKG-1 with an accompanying regression of heart hypertrophy.
In the study using cardiomyocytes in cultures (Zhou et al., 2009), it has been demonstrated that phenylepherine (PE) increased the size of cardiomyocytes, which was associated with enhanced PKG-1 activity. Addition of copper reduced the size of hypertrophic cardiomyocytes. Copper did not affect PE-induced activation of PKG-1. However, copper inhibited VEGFR-2, which is linked to the Akt-1 signaling pathway. The inhibition of VEGFR-2 was associated with the increase in the ratio of VEGFR-1 to VEGFR-2, thus activating PKG-1 due to the link between VEGFR-1 and PKG-1. Therefore, under the VEGFR-1-PKG-1 dominant condition, the signaling pathway switches from VEGFR-2 mediated hypertrophic stimulation to regression of cardiac hypertrophy, as shown in Figure 2.
Figure 2. VEGFR1-dependent regression pathways in hypertrophic cardiomyocytes.
GPCRs (guanine nucleotide binding-protein coupled receptors) recognize a variety of ligands and stimuli causing cardiac hypertrophy. These receptors affect the generation of small molecules that act as intracellular mediators or second messengers leading to hypertrophic signaling cascades. Among these pathways is the activation of Gq (G-protein subunit q), leading to activation of its downstream signaling transductions. Ca2+-dependent signaling pathways are critically involved in cardiac hypertrophy, including calcineurin/nuclear factor of activated T cells (NFAT), calmodulin-dependent kinases (CaMK) and mitogen-activated protein kinase (MAPK) pathways. VEGF binding to VEGFR1 leads to PKG-1 activation, which in turn activates regulator of G-protein signaling 2 (RGS2) that inhibits Gq, and phosphorylates vasodilator-stimulated phosphoprotein (VASP) that suppresses Ca2+ signaling pathways. PKG-1 also inhibits L-type calcium channels and transient receptor potential cation channel, subfamily 95 C, member 6 (TRPC6). These are counteractions against GPCR-mediated hypertrophic responses. Atrial natriuretic peptide (ANP) binds to natriuretic peptide receptors (NPRs), also called guanylyl cyclases (GCs), to convert GTP to cGMP and activate PKG-1. Inside the cells, L-arginine is converted to NO by endothelial nitric oxide synthase (eNOS), which in turn activates soluble guanylyl cyclase (sGC) and increases the levels of cGMP. Sildenafil inhibits phosphodiesterase type 5A (PDE5A) to block the conversion from cGMP to GMP, leading to sustained accumulation of cGMP and activation of PKG-1.
The link between VEGFR-1 and PKG-1 is a recent finding in the PKG-1 signaling pathway field. Factors upstream of PKG-1 activation have long been known to be related to natriuretic peptides (Kotera et al., 2003). For instance, cardiac atrial natriuretic peptide (ANP) binds to the transmembrane guanylyl cyclase (GC) receptor, GC-A, to exert its diverse functions, including regulating arterial blood pressure, modulating cardiomyocyte growth, and stimulating angiogenesis. The GC-A receptor is coupled to the c-GMP-dependent or PKG-1 signaling pathway, preventing pathological increases in intracellular Ca2+ and counteracting the hypertrophic signaling pathway. However, in chronic cardiac hypertrophy, ANP levels are markedly increased and the GC-A receptor is desensitized, leading to blunted responsiveness of the cGMP-dependent signaling pathway (Kotera et al., 2003). The newly-identified link between VEGFR-1 and PKG-1 would thus reactivate the cGMP-dependent signaling pathway, compensating for the desensitized effect of GC-A receptor.
How does PKG-1 activation lead to regression of cardiac hypertrophy? There are several downstream pathways that are linked to PKG-1, including both positive and negative regulators. PKG-1 is a crucial component in the NO-dependent signaling pathway, in which NO activates sGC leading to the release of cGMP and the activation of PKG-1. In general, PKG-1 causes down-regulation of hypertrophic pathways through its interactions with some critical molecules.
The transient receptor potential canonical (TRPC) 6 is a receptor-operated Ca2+ channel known to positively regulate the signaling pathway of calcineurin-nuclear factor of activated T cells (NFAT), which plays a pivotal role in cardiac hypertrophy. TRPC6 expression increased in the heart of mice subjected to pressure overload, and in neonatal and adult rat cardiomyocytes treated with angiotensin II or endothelin-1 (Koitabashi et al., 2010). A study using cardiac myocytes showed that the activation of PKG-1 by ANP induced phosphorylation of TRPC6 at threonine 69, leading to a significant inhibition of agonist-evoked NFAT activation and Ca2+ influx, and attenuation of cardiac hypertrophy (Kinoshita et al., 2010). These inhibitory effects were abolished by specific PKG-1 inhibitors or by substituting an alanine for threonine 69 in TRPC6. When the activation of PKG-1 was blocked by the ANP receptor GC-A deletion, the calcineurin-NFAT pathway was constitutively activated. A selective TRPC channel blocker, BTP2 (N-{4-[3,5-bis(Trifluoromethyl)-1H-pyrazol-1-yl]phenyl}-4-methyl-1,2,3-thiadiazole-5-carboxamide), significantly attenuated agonist-induced cardiac hypertrophy (Kinoshita et al., 2010). Therefore, it was concluded that TRPC6 is a critical target for PKG-1 activation, and its blockade contributes to the regression of cardiac hypertrophy.
Vasodilator-stimulated phosphoprotein (VASP) is a molecule that is suggested to be involved in the regulation of cardiac hypertrophy. Pressure overload induced by ascending aortic constriction in mice resulted in a significant increase in the expression and phosphorylation of VASP. The beta-adrenergic agonist isoproterenol activates the cAMP-dependent signaling pathway and promotes the rapid and reversible phosphorylation of VASP at Ser157 and Ser239. The inhibition of the cAMP-dependent protein kinase blocked the phosphorylation of VASP (Sartoretto et al., 2009). The cAMP-dependent phosphorylation of VASP is associated with increases in intracellular Ca2+ and cardiac hypertrophy. However, the activation of the cGMP-dependent signaling pathway by ANP leads to phosphorylation of VASP only at Ser239 (Sartoretto et al., 2009). This phosphorylation is associated with attenuated cellular Ca2+ entry, and altered growth and proliferation (O'Neil, 2007). Therefore, VASP as a target of PKG-1 plays an alternative role in the regulation of cardiac hypertrophy, whereas it promotes cardiac hypertrophy when activated by the cAMP-dependent signaling pathway.
The regulator of G-protein signaling 2 (RGS2) is a target of PKG-1. PKG-1 binds to, phosphorylates, and activates RGS2, which in turn disrupts the activation of intracellular calcium signaling via its inhibitory effect on Gq-coupled signaling pathway (Tang et al., 2003). PKG-1, through this calcium signaling inhibitory effect, causes multiple suppressive actions on calcium-dependent regulatory proteins, such as the L-type calcium channel, the ryanodine receptor calcium release channel, and troponin I (Saucerman & McCulloch, 2004). In addition, PKG-1 also directly interacts with several calcium-dependent regulatory proteins (Gray et al., 1999).
5.3. Regulation of HIF-1 transcriptional activity
HIF-1 plays a critical role in the regulation of cellular metabolism, homeostasis, and responses to stresses (Hirsila et al., 2005; Maxwell & Salnikow, 2004; Semenza, 2000). Therefore, the regulation of HIF-1 transcriptional activity has been a major focus in understanding the fundamental process of HIF-1 activation and its therapeutic significance. HIF-1 is composed of HIF-1α and HIF-1β or ARNT (aryl hydrocarbon receptor nuclear translocator). The cellular stability of HIF-1α determines the ultimate activation of HIF-1. The expression level of HIF-1α is undetectable in most cell types under normoxic conditions due to its degradation by the ubiquitin-proteasome pathway. In this process, one or both of the two conserved proline residues (Pro402 and Pro564) in HIF-1α is recognized by members of the prolyl hydroxylase domain-containing proteins (PHDs), which catalyze the reaction of proline hydroxylation (Huang et al., 1998; Ivan et al., 2001; Jaakkola et al., 2001). The hydroxylated HIF-1α is recognized by the von Hippel-Lindau protein (pVHL), which is a constituent of a ubiquitin ligase complex, targeting the HIF-1α subunit for degradation by the proteasome in the cytoplasm (Masson et al., 2001; Maxwell et al., 1999; Ohh et al., 2000; Tanimoto et al., 2000). Under hypoxic conditions, HIF-1α escapes from the degradation pathway, accumulates in the cytoplasm, and translocates to the nucleus, where it dimerizes with HIF-1β and interacts with cofactors to assemble the HIF-1 transcriptional complex, leading to transcriptional activation.
Several transition metals are capable of increasing the accumulation of HIF-1α in the cell. For instance, cobalt inhibits PHDs (Hirsila et al., 2005; Ke et al., 2005; Maxwell & Salnikow, 2004; Yuan et al., 2003), leading to the stabilization of HIF-1α and its accumulation in the cell. Other metals, such as nickel and copper, share the same inhibitory action on PHDs with cobalt (Hirsila et al., 2005; Ke et al., 2005; Martin et al., 2005; Maxwell & Salnikow, 2004; van Heerden et al., 2004; Yuan et al., 2003). However, recent study has demonstrated that copper is required for HIF-1 transcriptional activity under hypoxic conditions besides its inhibitory action on PHDs (Feng et al., 2009).
The mechanism of action of copper on HIF-1 transcriptional activity involves its role in the interaction between HIF-1 and the hypoxia-responsive element (HRE) sequence of target genes and the formation of the HIF-1 transcriptional complex (Feng et al., 2009). Because these processes take place in the nucleus, copper has to be transported into the nucleus to effect activation of the transcriptional activity of HIF-1. The copper chaperone for Cu, Zn-SOD (CCS) most likely functions as a copper chaperone for its nuclear translocation. It has been shown that gene silencing of CCS inhibited HIF-1 transcriptional activity under hypoxic conditions (Feng et al., 2009). The inhibitory effect of CCS deletion on HIF-1 transcriptional activity cannot be relieved by excess copper, although the inhibitory effect of copper restriction by copper chelation on HIF-1 transcriptional activity can be reversed by excess copper (Feng et al., 2009), indicating that CCS is essential in this action.
The most important relevance of copper regulation of HIF-1 transcriptional activity in the regression of pathological cardiac hypertrophy is that during the pathogenesis of cardiac hypertrophy, copper levels in the heart significantly decrease (Jiang et al., 2007). Under ischemic or hypoxic conditions, HIF-1α is accumulated in the heart, which has been observed in biopsy tissue samples of patients and in animal models (Kakinuma et al., 2001; Lee et al., 2000). However, it has been known that long-term ischemia or hypoxia leads to inhibition of VEGF expression and suppression of angiogenesis (Wang et al., 2010). It is thus interesting to know why the increased HIF-1α levels are associated with decreased expression of genes regulated by HIF-1 in the ischemic or hypertrophic heart. The loss of copper in ischemic or hypertrophic heart leading to suppression of HIF-1 transcriptional activity provides at least a partial explanation (He & Kang, 2012).
The association of copper deficiency with ischemic heart disease and cardiac hypertrophy has been recognized for a long time (Davydenko et al., 1995; Klevay, 1989; Shliakhover et al., 1990), but its clinical significance has not been fully explored. An early study using hearts from people who died from ischemic heart disease showed that copper concentrations were decreased in the uninjured tissues of the infarcted hearts relative to normal heart tissues (Wester, 1965). Another study showed that copper concentrations decreased in the hearts of people who died from acute ischemic infarction (Zama & Towns, 1986). The death rate from ischemic heart disease in soft water areas was found to be increased and copper concentrations were lower in the hearts of people who died from chronic ischemic heart disease (Anderson et al., 1975; Chipperfield & Chipperfield, 1978). Copper deficiency leads to congestive heart failure (Elsherif et al., 2003). The most important aspect of copper deficiency-induced cardiac hypertrophy is that upon copper repletion through dietary supplementation, the pre-established cardiac hypertrophy can be reversed (Elsherif et al., 2004a, 2004b; Zhou et al., 2009b). A study in a small population of chronic heart failure patients showed that dietary supplementation with micronutrients for 9 months increases left ventricle ejection and decreases left ventricle volume along with improvement of quality of life (Witte et al., 2005). Copper (1.2 mg/day) was among the formulated micronutrients in that study.
Further studies have shown that pressure overload-induced cardiac hypertrophy is also reversible by dietary copper supplementation. In a mouse model of pressure overload-induced cardiac hypertrophy and dysfunction, it has been found that pressure overload is associated with copper loss in the heart (Jiang et al., 2007). Dietary supplementation with physiologically relevant levels of copper replenishes cardiac copper levels and reverses cardiac hypertrophy and dysfunction even in the presence of chronic pressure overload. The cardiac effect of dietary copper supplementation is associated with recovery of normal VEGF expression and enhanced angiogenesis. In the presence of anti-VEGF antibody, dietary copper supplementation failed to reverse the pre-established heart hypertrophy and dysfunction. As discussed above, this effect results from copper activation of HIF-1 transcriptional activity (Feng et al., 2009). It is important to note that VEGF and VEGFR-1 are regulated by HIF-1, and VEGFR-2 is not. This would provide another explanation for copper-induced switching from VEGFR-2-dependent to VEGFR-1-dependent signaling pathways.
These signaling pathways and transcriptional regulation of critical molecules involved in regression of pathological cardiac hypertrophy discussed above become important targets for therapeutic intervention. Understanding these targets will provide insightful suggestions for the consideration for pharmaceutical development.
6. Potential therapeutic targets for promoting regression of pathological cardiac hypertrophy
Regression of pathological cardiac hypertrophy is a relatively new concept. Clinical and experimental observations have noted the regression phenomenon, but the significance has not been fully appreciated. There are multiple obstacles that need to be overcome in order to make the regression of pathological cardiac hypertrophy a realistic medical approach: (1) It is still a mainstay belief that pathological cardiac hypertrophy is not reversible, although there is accumulating evidence that shows it is possible. (2) Experimental approaches to understand the mechanisms by which regression of pathological cardiac hypertrophy occur must focus on blocking the pathogenesis pathways leading to cardiac hypertrophy. This approach would help develop interventions to inhibit the progression, but would hardly provide critical insights into the regression because the regression would not be a simple reversal of the forward process. (3) Experimental models focusing on regression of pathological cardiac hypertrophy, not prevention of cardiac hypertrophy, are not appreciably available. Making such experimental models is timeand effort-consuming, which add more difficulties to the existing complexity. (4) Pharmaceutical approaches take time to be established and a long-term vision needs to be created in order to guide drug development.
There are some clues that implicate potential therapeutic targets that would play important roles in the regression of pathological cardiac hypertrophy. It again needs to be pointed out that this journey is at its beginning and validation of any of these targets has not been done.
6.1. Promotion of myocardial angiogenesis
Stimulation of vascular growth in normal adult mouse hearts leads to a physiological cardiac hypertrophy (Karpanen et al., 2008; Tirziu et al., 2007; Villari et al., 1992). In pathological cardiac hypertrophy, the enhancement of angiogenesis improves myocardial function and inhibits the progression of cardiac hypertrophy (Jiang et al., 2007; Suzuki et al., 2005). Increasing capillary density in the heart is, therefore, one of the potential therapeutic strategies to promote the regression of pathological cardiac hypertrophy (Izumiya et al., 2006; Mukherjee, 2004; Tamirisa & Mukherjee, 2002). In a mouse model of pathological cardiac hypertrophy induced by pressure overload, this concept has been tested. Dietary supplementation with physiologically relevant levels of copper leads to promotion of angiogenesis and reversal of cardiac hypertrophy, which can be blocked by anti-VEGF antibody (Jiang et al., 2007).
Although VEGF is necessary for myocardial angiogenesis, it is not, however, sufficient to promote the maturation of angiogenesis. The efficacy of VEGF therapy for ischemic cardiomyopathy has been studied and the results have suggested the effectiveness of VEGF therapy for improving disease conditions (Hughes et al., 2004; Losordo et al., 1998; Pearlman et al., 1995; Zhou et al., 2005). However, the long-term expression of VEGF as a means of therapy has been shown to cause circulatory leakage (Melgar-Lesmes et al., 2009; Weis & Cheresh, 2005). It has been shown that the complete angiogenesis effect requires coordinated interactions between VEGF and other factors such as angiopoietin 1 and 2 (Su et al., 2009; Tao et al., 2011; Zhou et al., 2005). Therefore, to promote myocardial angiogenesis it is important to consider a cocktail approach and to ensure coordination of multiple factors in the process.
6.2. Selective stimulation of VEGFR-1 signaling pathway
The distinct actions of VEGF are mediated by receptor tyrosine kinases or VEGFRs (de Vries et al., 1992; Millauer et al., 1993; Terman et al., 1992). As discussed above, copper-induced regression of pathological cardiac hypertrophy is mediated by the increase in VEGF production (Jiang et al., 2007). In primary cultures of neonatal rat cardiomyocytes it was found that PE-induced cardiomyocyte hypertrophy was also reversible by copper addition to the cultures and this reversal was also VEGF-dependent (Zhou et al., 2008). However, the critical determining factor for VEGF functioning in the regression of pathological cardiac hypertrophy is the increase in the ratio of VEGFR-1/VEGFR-2 (Madonna & De Caterina, 2009; Zhou et al., 2009a; Zuo et al., 2010). Gene silencing of VEGFR-1 blocked copper-induced reversal of cardiomyocyte hypertrophy, demonstrating the role of VEGFR-1 in the regression process (Madonna & De Caterina, 2009; Zhou et al., 2009a; Zuo et al., 2010).
The link of VEGFR-1 to the PKG-1 signaling pathway in the regression of pathological cardiac hypertrophy provides an explanation for the VEGFR-1-dependent reversal of cardiomyocyte hypertrophy (Madonna & De Caterina, 2009; Zhou et al., 2009a; Zuo et al., 2010). However, there would be multiple pathways that are triggered by VEGFR-1 activation. The pathways other than PKG-1 need to be identified in order to provide a comprehensive understanding of VEGFR-1 signaling mechanisms in the regression of pathological cardiac hypertrophy. On the other hand, pharmaceutical development other than copper supplementation for selectively stimulating VEGFR-1 would be an effective, alternative approach in the reversal of pathological cardiac hypertrophy, as summarized in Figure 3.
Figure 3. Copper-dependent switching from VEGFR2-dependent to VEGFR1-dependent signaling pathways.
VEGFR2 is involved in myocardial mitogenic responses as described for Figure 2 and in the text. VEGFR1 is involved in the regression of cardiac hypertrophy as described in Figure 2. Copper is required for the activation of HIF-1 transcriptional activity, which activates the expression of VEGF and VEGFR1, and not VEGFR2. Supplementation with copper leads to the switching from VEGFR2-dependent to VEGFR1-dependent signaling pathways. Pharmaceuticals that have the potential for causing the same switching effect as copper would facilitate the regression of pathological cardiac hypertrophy.
6.3. Activation of PKG-1 pathway
The activation of PKG-1 is related to inhibition of hypertrophic growth of the heart and regression of cardiomyocyte hypertrophy. The former has been known for a long time, and the latter has been observed in recent studies (Koitabashi et al., 2010; Zhou et al., 2009a). It is important to recognize that activation of PKG-1 is a natural process in response to cardiac hypertrophy. Strategies need to be undertaken not only to increase PKG-1 activity, but also to sustain the elevation. Previous approaches have used the PDE5A inhibitor, sildenafil, to suppress the degradation process of cGMP, leading to regression of pathological cardiac hypertrophy induced by pressure overload in mice (Nagayama et al., 2009; Takimoto et al., 2005). This inhibition increases as well as sustains the activity of PKG-1.
Another consideration is that activation of the PKG-1 pathway involved in the regression of pathological cardiac hypertrophy would be more important than the increase in the PKG-1 activity per se. As mentioned above, the PKG-1 activity has been shown to be elevated in hypertrophic cardiomyocytes, but the enlargement of the heart remains predominant. In the study using cultured cardiomyocytes it was found that PE treatment enhanced PKG-1 activity, and copper addition did not affect the PE-induced activation of PKG-1 (Zhou et al., 2009a)). However, the regression of cardiomyocyte hypertrophy does not take place until the addition of copper to the cultures. The critical determining factor is the switching by copper from VEGFR-2-dependent to VEGFR-1-dependent signal transduction, because the latter is linked to the PKG-1-mediated regression pathway (Zhou et al., 2009a)).
6.4. Activation of HIF-1 transcriptional activity
HIF-1 regulates many relevant genes that are not only essential for the initiation, but also critical for the maturation of the newly formed vessels. A study using conditionally cardiac-specific HIF-1α-knockout mouse model has demonstrated that the depression of HIF-1α resulted in impaired expression of genes VEGF, phosphoglycerate kinase, glucose transport-1, lactate dehydrogenase, and sarcoplasmic reticulum calcium pump SERCA2 (Huang et al., 2004a)). Consequentially, these defects lead to diminished vascularization, reduced cellular phosphocreatine, ATP and lactate levels, altered calcium flux, and suppressed contractility (Huang et al., 2004a)).
Cobalt is capable of stabilizing HIF-1α, stimulating the transcriptional activity of HIF-1. For this reason, cobalt has been studied for its potential as a drug to stimulate cellular function regulated by HIF-1 (Endoh et al., 2000; Ohtomo et al., 2008). Cobalt increases the cytosolic accumulation of HIF-1α, but has not been shown to be required for the activity of HIF-1 in the nucleus. In a recent study, it was found that cobalt-induced HIF-1 transcriptional activity is copper-dependent because copper is required for HIF-1 transcriptional activation in the nucleus (Qiu et al., 2012). This is significantly related to the development of cobalt as a therapeutic agent. Under ischemic conditions, copper levels in the tissues are significantly decreased and this decrease has been shown to be severe enough to result in depressed HIF-1 transcriptional activity although the level of HIF-1α is increased, as shown in Figure 4. Therefore, it is critical to consider the activity of HIF-1 rather than only the level of HIF-1α in manipulation of the role of this transcription factor in the regression of pathological cardiac hypertrophy.
Figure 4. Effects of copper efflux on HIF-1 transactivity in ischemic myocardium.
Myocardial ischemia inhibits PHDs leading to HIF-1α accumulation. HIF-1α translocates into nucleus and dimerizes with HIF-1β (ARNT). The heterodimer then recruits cofactors such as 96 CBP, p300 and SRC-1 to form transcriptional complex for the transcriptional activation. On the other hand, myocardial ischemia results in copper efflux, leading to copper deficiency in the nucleus. This in turn suppresses copper-binding proteins functioning in HIF-1 transcriptional complex formation and HIF-1 interaction with HRE sequence in the target genes, leading to suppressed expression of genes including VEGF and VEGFR1 regulated by HIF-1.
7. Summary and perspectives
The distinction between physiological and pathological cardiac hypertrophy is not only identified in biochemical and functional alterations in the development of the two forms of heart enlargement, but also in their reversibility; the former is reversible and the latter was believed to be irreversible. However, the traditional belief of the irreversibility of pathological cardiac hypertrophy has been challenged by recent clinical observations and experimental studies that have presented evidence showing the regression of pathological cardiac hypertrophy. This regression was unexpectedly observed in LVAD-supported heart failure patients. The key factor for the regression to occur is the promotion of myocardial angiogenesis. Careful analysis of the effects generated by pharmaceutical agents affecting angiogenesis revealed the accompanied regression of pathological cardiac hypertrophy. While myocardial angiogenesis provides an improved microenvironment for the regression to take place, molecular events occurring in the hypertrophic cardiomyocytes undergoing the process of regression are of significant clinical relevance. Some novel signaling pathways by which the regression of pathological cardiac hypertrophy can be initiated and propagated have been identified, among which are the VEGF-dependent and VEGFR-1-activated PKG-1 signaling pathways. These pathways are not the reversed process of the forward signaling pathways leading to cardiac hypertrophy, but are distinct molecular events. Based on current understanding, therapeutic targets for promoting the regression of pathological cardiac hypertrophy can be identified by mainly focusing on improvement of myocardial angiogenesis and promotion of reversal signaling pathways in hypertrophic cardiomyocytes. Such efforts as selective stimulation of VEGFR-1 and PKG-1 signaling pathways and ensuring HIF-1 transcriptional activity, would promote the regression of pathological cardiac hypertrophy. More comprehensive understanding of this new field of cardiac research and medicine is anticipated. The translation of this new concept of reversal of pathological cardiac hypertrophy to clinical practice would greatly benefit heart failure patients.
Acknowledgement
The authors would like to express grateful attitude to Yuping Xie, Xiao Zuo, Yonglin Chen, and Rui Li for assistance in collecting literature information for the preparation of this review article. The studies from the authors’ laboratory cited in this article were supported in part by NIH grant HL63760 to YJK.
Abbreviations
- ACE
angiotensin converting enzyme
- ANP
atrial natriuretic peptide
- BNP
B-type natriuretic peptide
- CCS
copper chaperone for superoxide dismutase
- CK
creatine kinase
- Cu, Zn-SOD
Cu, Zn-superoxide dismutase
- ECM
extracellular matrix
- ERK
extracellular regulated protein kinases
- FA
fatty acid
- GC
guanylyl cyclase
- GC-A
guanylyl cyclase-A
- GMP
guanosine monophosphate
- GSK-3
Glycogen synthase kinase 3
- HIF-1
hypoxia inducible factor-1
- HRE
hypoxia-responsive element
- LVADs
left ventricular assist devices
- MAPK
mitogen-activated protein kinase
- MMPs
matrix metalloproteinases
- mTOR
mammalian target of rapamycin
- NFAT
nuclear factor of activated T cells
- PCP
pro-collagen N- and C-proteinases
- PCr
phosphocreatine
- PDE5A
phosphodiesterase 5A
- PE
phenylephrine
- PKG-1
cyclic GMP-dependent protein kinase-1
- MRA
magnetic resonance angiography
- pVHL
von Hippel-Lindau protein
- RGS2
regulator of G protein signaling 2
- SERCA2a
sarco(endo)plasmic reticulum Ca2+-ATPase 2A
- TAC
transverse aorta constriction
- TGF-β
transforming growth factor-β
- TIMP
tissue inhibitors of metalloproteinase
- TNF-α
tumor Necrosis Factor-α
- TRPC
transient receptor potential canonical
- VASP
Vasodilator-stimulated phosphoprotein
- VEGF
Vascular endothelial growth factor
- VEGFR-1
VEGF receptor-1
- VEGFR-2
VEGF receptor-2
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of Interests Statement
The authors declare that there are no conflicts of interest.
References
- Abel ED, Doenst T. Mitochondrial adaptations to physiological vs. pathological cardiac hypertrophy. Cardiovasc Res. 2011;90:234–242. doi: 10.1093/cvr/cvr015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acker MA. Surgical therapies for heart failure. J Card Fail. 2004;10:S220–S224. doi: 10.1016/j.cardfail.2004.09.005. [DOI] [PubMed] [Google Scholar]
- Anderson TW, Neri LC, Schreiber GB, Talbot FD, Zdrojewski A. Letter: Ischemic heart disease, water hardness and myocardial magnesium. Can Med Assoc J. 1975;113:199–203. [PMC free article] [PubMed] [Google Scholar]
- Anversa P, Beghi C, Kikkawa Y, Olivetti G. Myocardial infarction in rats. Infarct size, myocyte hypertrophy, and capillary growth. Circ Res. 1986;58:26–37. doi: 10.1161/01.res.58.1.26. [DOI] [PubMed] [Google Scholar]
- Anversa P, Leri A, Kajstura J. Cardiac regeneration. J Am Coll Cardiol. 2006;47:1769–1776. doi: 10.1016/j.jacc.2006.02.003. [DOI] [PubMed] [Google Scholar]
- Backs J, Backs T, Neef S, Kreusser MM, Lehmann LH, Patrick DM, Grueter CE, Qi X, Richardson JA, Hill JA, Katus HA, Bassel-Duby R, Maier LS, Olson EN. The delta isoform of CaM kinase II is required for pathological cardiac hypertrophy and remodeling after pressure overload. Proc Natl Acad Sci U S A. 2009;106:2342–2347. doi: 10.1073/pnas.0813013106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baicu CF, Stroud JD, Livesay VA, Hapke E, Holder J, Spinale FG, Zile MR. Changes in extracellular collagen matrix alter myocardial systolic performance. Am J Physiol Heart Circ Physiol. 2003;284:H122–H132. doi: 10.1152/ajpheart.00233.2002. [DOI] [PubMed] [Google Scholar]
- Balligand JL, Ungureanu D, Kelly RA, Kobzik L, Pimental D, Michel T, Smith TW. Abnormal contractile function due to induction of nitric oxide synthesis in rat cardiac myocytes follows exposure to activated macrophage-conditioned medium. J Clin Invest. 1993;91:2314–2319. doi: 10.1172/JCI116461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbone A, Holmes JW, Heerdt PM, The AH, Naka Y, Joshi N, Daines M, Marks AR, Oz MC, Burkhoff D. Comparison of right and left ventricular responses to left ventricular assist device support in patients with severe heart failure: a primary role of mechanical unloading underlying reverse remodeling. Circulation. 2001;104:670–675. doi: 10.1161/hc3101.093903. [DOI] [PubMed] [Google Scholar]
- Bartling B, Milting H, Schumann H, Darmer D, Arusoglu L, Koerner MM, El-Banayosy A, Koerfer R, Holtz J, Zerkowski HR. Myocardial gene expression of regulators of myocyte apoptosis and myocyte calcium homeostasis during hemodynamic unloading by ventricular assist devices in patients with end-stage heart failure. Circulation. 1999;100:II216–II223. doi: 10.1161/01.cir.100.suppl_2.ii-216. [DOI] [PubMed] [Google Scholar]
- Baughman KL, Jarcho JA. Bridge to life--cardiac mechanical support. N Engl J Med. 2007;357:846–849. doi: 10.1056/NEJMp078131. [DOI] [PubMed] [Google Scholar]
- Becker LB, vanden Hoek TL, Shao ZH, Li CQ, Schumacker PT. Generation of superoxide in cardiomyocytes during ischemia before reperfusion. Am J Physiol. 1999;277:H2240–H2246. doi: 10.1152/ajpheart.1999.277.6.H2240. [DOI] [PubMed] [Google Scholar]
- Belmonte SL, Blaxall BC. G protein coupled receptor kinases as therapeutic targets in cardiovascular disease. Circ Res. 2011;109:309–319. doi: 10.1161/CIRCRESAHA.110.231233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, Kasahara H, Rota M, Musso E, Urbanek K, Leri A, Kajstura J, Nadal-Ginard B, Anversa P. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell. 2003;114:763–776. doi: 10.1016/s0092-8674(03)00687-1. [DOI] [PubMed] [Google Scholar]
- Berenji K, Drazner MH, Rothermel BA, Hill JA. Does load-induced ventricular hypertrophy progress to systolic heart failure? Am J Physiol Heart Circ Physiol. 2005;289:H8–H16. doi: 10.1152/ajpheart.01303.2004. [DOI] [PubMed] [Google Scholar]
- Bick RJ, Grigore AM, Poindexter BJ, Schnee PM, Nussmeier NA, Gregoric ID, Shah NA, Myers TJ, Buja LM, Frazier OH. Left Ventricular Unloading with an Assist Device Results in Receptor Relocalization as well as Increased Beta-Adrenergic Receptor Numbers: Are These Changes Indications for Outcome? Journal of Cardiac Surgery. 2005;20:332–336. doi: 10.1111/j.1540-8191.2005.2004105.x. [DOI] [PubMed] [Google Scholar]
- Bicknell KA, Coxon CH, Brooks G. Can the cardiomyocyte cell cycle be reprogrammed? J Mol Cell Cardiol. 2007;42:706–721. doi: 10.1016/j.yjmcc.2007.01.006. [DOI] [PubMed] [Google Scholar]
- Birks EJ, George RS, Hedger M, Bahrami T, Wilton P, Bowles CT, Webb C, Bougard R, Amrani M, Yacoub MH, Dreyfus G, Khaghani A. Reversal of severe heart failure with a continuous-flow left ventricular assist device and pharmacological therapy: a prospective study. Circulation. 2011;123:381–390. doi: 10.1161/CIRCULATIONAHA.109.933960. [DOI] [PubMed] [Google Scholar]
- Bittl JA, Ingwall JS. Reaction rates of creatine kinase and ATP synthesis in the isolated rat heart. A 31P NMR magnetization transfer study. J Biol Chem. 1985;260:3512–3517. [PubMed] [Google Scholar]
- Blume ED, Naftel DC, Bastardi HJ, Duncan BW, Kirklin JK, Webber SA. Outcomes of children bridged to heart transplantation with ventricular assist devices: a multi-institutional study. Circulation. 2006;113:2313–2319. doi: 10.1161/CIRCULATIONAHA.105.577601. [DOI] [PubMed] [Google Scholar]
- Borkow G, Gabbay J, Dardik R, Eidelman AI, Lavie Y, Grunfeld Y, Ikher S, Huszar M, Zatcoff RC, Marikovsky M. Molecular mechanisms of enhanced wound healing by copper oxide-impregnated dressings. Wound Repair Regen. 2010;18:266–275. doi: 10.1111/j.1524-475X.2010.00573.x. [DOI] [PubMed] [Google Scholar]
- Boyle AJ, Ascheim DD, Russo MJ, Kormos RL, John R, Naka Y, Gelijns AC, Hong KN, Teuteberg JJ. Clinical outcomes for continuous-flow left ventricular assist device patients stratified by pre-operative INTERMACS classification. J Heart Lung Transplant. 2011;30:402–407. doi: 10.1016/j.healun.2010.10.016. [DOI] [PubMed] [Google Scholar]
- Brady AJ, Warren JB, Poole-Wilson PA, Williams TJ, Harding SE. Nitric oxide attenuates cardiac myocyte contraction. Am J Physiol. 1993;265:H176–H182. doi: 10.1152/ajpheart.1993.265.1.H176. [DOI] [PubMed] [Google Scholar]
- Brady NR, Hamacher-Brady A, Gottlieb RA. Proapoptotic BCL-2 family members and mitochondrial dysfunction during ischemia/reperfusion injury, a study employing cardiac HL-1 cells and GFP biosensors. Biochim Biophys Acta. 2006;1757:667–678. doi: 10.1016/j.bbabio.2006.04.011. [DOI] [PubMed] [Google Scholar]
- Brady NR, Hamacher-Brady A, Yuan H, Gottlieb RA. The autophagic response to nutrient deprivation in the hl-1 cardiac myocyte is modulated by Bcl-2 and sarco/endoplasmic reticulum calcium stores. FEBS J. 2007;274:3184–3197. doi: 10.1111/j.1742-4658.2007.05849.x. [DOI] [PubMed] [Google Scholar]
- Brilla CG, Maisch B. Regulation of the structural remodelling of the myocardium: from hypertrophy to heart failure. Eur Heart J. 1994;15(Suppl D):45–52. doi: 10.1093/eurheartj/15.suppl_d.45. [DOI] [PubMed] [Google Scholar]
- Bruckner BA, Stetson SJ, Perez-Verdia A, Youker KA, Radovancevic B, Connelly JH, Koerner MM, Entman ME, Frazier OH, Noon GP, Torre-Amione G. Regression of fibrosis and hypertrophy in failing myocardium following mechanical circulatory support. J Heart Lung Transplant. 2001;20:457–464. doi: 10.1016/s1053-2498(00)00321-1. [DOI] [PubMed] [Google Scholar]
- Buja LM, Vela D. Cardiomyocyte death and renewal in the normal and diseased heart. Cardiovasc Pathol. 2008;17:349–374. doi: 10.1016/j.carpath.2008.02.004. [DOI] [PubMed] [Google Scholar]
- Bujak M, Frangogiannis NG. The role of TGF-beta signaling in myocardial infarction and cardiac remodeling. Cardiovasc Res. 2007;74:184–195. doi: 10.1016/j.cardiores.2006.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai J, Yi FF, Bian ZY, Shen DF, Yang L, Yan L, Tang QZ, Yang XC, Li H. Crocetin protects against cardiac hypertrophy by blocking MEK-ERK1/2 signalling pathway. J Cell Mol Med. 2009;13:909–925. doi: 10.1111/j.1582-4934.2008.00620.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Care A, Catalucci D, Felicetti F, Bonci D, Addario A, Gallo P, Bang ML, Segnalini P, Gu Y, Dalton ND, Elia L, Latronico MV, Hoydal M, Autore C, Russo MA, Dorn GW, 2nd, Ellingsen O, Ruiz-Lozano P, Peterson KL, Croce CM, Peschle C, Condorelli G. MicroRNA-133 controls cardiac hypertrophy. Nat Med. 2007;13:613–618. doi: 10.1038/nm1582. [DOI] [PubMed] [Google Scholar]
- Carroll JF, Tyagi SC. Extracellular matrix remodeling in the heart of the homocysteinemic obese rabbit. Am J Hypertens. 2005;18:692–698. doi: 10.1016/j.amjhyper.2004.11.035. [DOI] [PubMed] [Google Scholar]
- Chatterjee K. Neurohormonal activation in congestive heart failure and the role of vasopressin. Am J Cardiol. 2005;95:8B–13B. doi: 10.1016/j.amjcard.2005.03.003. [DOI] [PubMed] [Google Scholar]
- Chen Y, Azad MB, Gibson SB. Superoxide is the major reactive oxygen species regulating autophagy. Cell Death Differ. 2009;16:1040–1052. doi: 10.1038/cdd.2009.49. [DOI] [PubMed] [Google Scholar]
- Chen Y, McMillan-Ward E, Kong J, Israels SJ, Gibson SB. Mitochondrial electron-transport-chain inhibitors of complexes I and II induce autophagic cell death mediated by reactive oxygen species. J Cell Sci. 2007;120:4155–4166. doi: 10.1242/jcs.011163. [DOI] [PubMed] [Google Scholar]
- Chiba K, Moriyama S, Ishigai Y, Fukuzawa A, Irie K, Shibano T. Lack of correlation of hypotensive effects with prevention of cardiac hypertrophy by perindopril after ligation of rat coronary artery. Br J Pharmacol. 1994;112:837–842. doi: 10.1111/j.1476-5381.1994.tb13155.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chipperfield B, Chipperfield JR. Differences in metal content of the heart muscle in death from ischemic heart disease. Am Heart J. 1978;95:732–737. doi: 10.1016/0002-8703(78)90503-3. [DOI] [PubMed] [Google Scholar]
- Clark DE, Smith SK, He Y, Day KA, Licence DR, Corps AN, Lammoglia R, Charnock-Jones DS. A vascular endothelial growth factor antagonist is produced by the human placenta and released into the maternal circulation. Biol Reprod. 1998;59:1540–1548. doi: 10.1095/biolreprod59.6.1540. [DOI] [PubMed] [Google Scholar]
- Cleutjens JP. The role of matrix metalloproteinases in heart disease. Cardiovasc Res. 1996;32:816–821. [PubMed] [Google Scholar]
- Cleutjens JP, Creemers EE. Integration of concepts: cardiac extracellular matrix remodeling after myocardial infarction. J Card Fail. 2002;8:S344–S348. doi: 10.1054/jcaf.2002.129261. [DOI] [PubMed] [Google Scholar]
- Dandel M, Weng Y, Siniawski H, Potapov E, Lehmkuhl HB, Hetzer R. Long-term results in patients with idiopathic dilated cardiomyopathy after weaning from left ventricular assist devices. Circulation. 2005;112:I37–I45. doi: 10.1161/CIRCULATIONAHA.104.525352. [DOI] [PubMed] [Google Scholar]
- Davila DF, Nunez TJ, Odreman R, de Davila CA. Mechanisms of neurohormonal activation in chronic congestive heart failure: pathophysiology and therapeutic implications. Int J Cardiol. 2005;101:343–346. doi: 10.1016/j.ijcard.2004.08.023. [DOI] [PubMed] [Google Scholar]
- Davydenko NV, Smirnova IP, Kvasha EA, Gorbas IM. [The relationship between the copper and zinc intake with food and the prevalence of ischemic heart disease and its risk factors] Lik Sprava. 1995:73–77. [PubMed] [Google Scholar]
- de Vries C, Escobedo JA, Ueno H, Houck K, Ferrara N, Williams LT. The fms-like tyrosine kinase, a receptor for vascular endothelial growth factor. Science. 1992;255:989–991. doi: 10.1126/science.1312256. [DOI] [PubMed] [Google Scholar]
- deCathelineau AM, Henson PM. The final step in programmed cell death: phagocytes carry apoptotic cells to the grave. Essays Biochem. 2003;39:105–117. doi: 10.1042/bse0390105. [DOI] [PubMed] [Google Scholar]
- Diez J, Lopez B, Gonzalez A, Querejeta R. Clinical aspects of hypertensive myocardial fibrosis. Curr Opin Cardiol. 2001;16:328–335. doi: 10.1097/00001573-200111000-00003. [DOI] [PubMed] [Google Scholar]
- Dipla K, Mattiello JA, Jeevanandam V, Houser SR, Margulies KB. Myocyte recovery after mechanical circulatory support in humans with end-stage heart failure. Circulation. 1998;97:2316–2322. doi: 10.1161/01.cir.97.23.2316. [DOI] [PubMed] [Google Scholar]
- Eliot RS, Clayton FC, Pieper GM, Todd GL. Influence of environmental stress on pathogenesis of sudden cardiac death. Fed Proc. 1977;36:1719–1724. [PubMed] [Google Scholar]
- Elsherif L, Jiang Y, Saari JT, Kang YJ. Dietary copper restriction-induced changes in myocardial gene expression and the effect of copper repletion. Exp Biol Med (Maywood) 2004a;229:616–622. [PubMed] [Google Scholar]
- Elsherif L, Ortines RV, Saari JT, Kang YJ. Congestive heart failure in copper-deficient mice. Exp Biol Med (Maywood) 2003;228:811–817. doi: 10.1177/15353702-0322807-06. [DOI] [PubMed] [Google Scholar]
- Elsherif L, Wang L, Saari JT, Kang YJ. Regression of dietary copper restriction-induced cardiomyopathy by copper repletion in mice. J Nutr. 2004b;134:855–860. doi: 10.1093/jn/134.4.855. [DOI] [PubMed] [Google Scholar]
- Endoh H, Kaneko T, Nakamura H, Doi K, Takahashi E. Improved cardiac contractile functions in hypoxia-reoxygenation in rats treated with low concentration Co(2+) Am J Physiol Heart Circ Physiol. 2000;279:H2713–H2719. doi: 10.1152/ajpheart.2000.279.6.H2713. [DOI] [PubMed] [Google Scholar]
- Entwistle JW., 3rd Short- and long-term mechanical ventricular assistance towards myocardial recovery. Surg Clin North Am. 2004;84:201–221. doi: 10.1016/S0039-6109(03)00213-5. [DOI] [PubMed] [Google Scholar]
- Feng W, Ye F, Xue W, Zhou Z, Kang YJ. Copper regulation of hypoxia-inducible factor-1 activity. Mol Pharmacol. 2009;75:174–182. doi: 10.1124/mol.108.051516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiedler B, Lohmann SM, Smolenski A, Linnemuller S, Pieske B, Schroder F, Molkentin JD, Drexler H, Wollert KC. Inhibition of calcineurin-NFAT hypertrophy signaling by cGMP-dependent protein kinase type I in cardiac myocytes. Proc Natl Acad Sci U S A. 2002;99:11363–11368. doi: 10.1073/pnas.162100799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flanagan MF, Fujii AM, Colan SD, Flanagan RG, Lock JE. Myocardial angiogenesis and coronary perfusion in left ventricular pressure-overload hypertrophy in the young lamb. Evidence for inhibition with chronic protamine administration. Circ Res. 1991;68:1458–1470. doi: 10.1161/01.res.68.5.1458. [DOI] [PubMed] [Google Scholar]
- Flesch M, Margulies KB, Mochmann HC, Engel D, Sivasubramanian N, Mann DL. Differential regulation of mitogen-activated protein kinases in the failing human heart in response to mechanical unloading. Circulation. 2001;104:2273–2276. doi: 10.1161/hc4401.099449. [DOI] [PubMed] [Google Scholar]
- Frazier OH. First use of an untethered, vented electric left ventricular assist device for long-term support. Circulation. 1994;89:2908–2914. doi: 10.1161/01.cir.89.6.2908. [DOI] [PubMed] [Google Scholar]
- Frazier OH, Benedict CR, Radovancevic B, Bick RJ, Capek P, Springer WE, Macris MP, Delgado R, Buja LM. Improved left ventricular function after chronic left ventricular unloading. Ann Thorac Surg. 1996;62:675–681. doi: 10.1016/s0003-4975(96)00437-7. discussion 681–672. [DOI] [PubMed] [Google Scholar]
- Gao XM, Kiriazis H, Moore XL, Feng XH, Sheppard K, Dart A, Du XJ. Regression of pressure overload-induced left ventricular hypertrophy in mice. Am J Physiol Heart Circ Physiol. 2005;288:H2702–H2707. doi: 10.1152/ajpheart.00836.2004. [DOI] [PubMed] [Google Scholar]
- Gardai SJ, McPhillips KA, Frasch SC, Janssen WJ, Starefeldt A, Murphy-Ullrich JE, Bratton DL, Oldenborg PA, Michalak M, Henson PM. Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell. 2005;123:321–334. doi: 10.1016/j.cell.2005.08.032. [DOI] [PubMed] [Google Scholar]
- Gaspar-Pereira S, Fullard N, Townsend PA, Banks PS, Ellis EL, Fox C, Maxwell AG, Murphy LB, Kirk A, Bauer R, Caamano JH, Figg N, Foo RS, Mann J, Mann DA, Oakley F. The NF-kappaB Subunit c-Rel Stimulates Cardiac Hypertrophy and Fibrosis. Am J Pathol. 2011 doi: 10.1016/j.ajpath.2011.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez A, Lopez B, Querejeta R, Diez J. Regulation of myocardial fibrillar collagen by angiotensin II. A role in hypertensive heart disease? J Mol Cell Cardiol. 2002;34:1585–1593. doi: 10.1006/jmcc.2002.2081. [DOI] [PubMed] [Google Scholar]
- Gottlieb RA, Burleson KO, Kloner RA, Babior BM, Engler RL. Reperfusion injury induces apoptosis in rabbit cardiomyocytes. J Clin Invest. 1994;94:1621–1628. doi: 10.1172/JCI117504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gottlieb RA, Finley KD, Mentzer RM., Jr. Cardioprotection requires taking out the trash. Basic Res Cardiol. 2009;104:169–180. doi: 10.1007/s00395-009-0011-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray DB, Polo-Parada L, Pilar GR, Eang P, Metzger RR, Klann E, Meriney SD. A nitric oxide/cyclic GMP-dependent protein kinase pathway alters transmitter release and inhibition by somatostatin at a site downstream of calcium entry. J Neurochem. 1999;72:1981–1990. doi: 10.1046/j.1471-4159.1999.0721981.x. [DOI] [PubMed] [Google Scholar]
- Grieve DJ, MacCarthy PA, Gall NP, Cave AC, Shah AM. Divergent biological actions of coronary endothelial nitric oxide during progression of cardiac hypertrophy. Hypertension. 2001;38:267–273. doi: 10.1161/01.hyp.38.2.267. [DOI] [PubMed] [Google Scholar]
- Gustafsson AB, Gottlieb RA. Autophagy in ischemic heart disease. Circ Res. 2009;104:150–158. doi: 10.1161/CIRCRESAHA.108.187427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haddad H, Elabbassi W, Moustafa S, Davies R, Mesana T, Hendry P, Masters R, Mussivand T. Left Ventricular Assist Devices as Bridge to Heart Transplantation in Congestive Heart Failure with Pulmonary Hypertension. ASAIO Journal. 2005;51:456–460. doi: 10.1097/01.mat.0000169125.21268.d7. [DOI] [PubMed] [Google Scholar]
- Halestrap AP, Clarke SJ, Javadov SA. Mitochondrial permeability transition pore opening during myocardial reperfusion--a target for cardioprotection. Cardiovasc Res. 2004;61:372–385. doi: 10.1016/S0008-6363(03)00533-9. [DOI] [PubMed] [Google Scholar]
- Hamacher-Brady A, Brady NR, Gottlieb RA. Enhancing macroautophagy protects against ischemia/reperfusion injury in cardiac myocytes. J Biol Chem. 2006;281:29776–29787. doi: 10.1074/jbc.M603783200. [DOI] [PubMed] [Google Scholar]
- Hamasaki S, Al Suwaidi J, Higano ST, Miyauchi K, Holmes DR, Jr., Lerman A. Attenuated coronary flow reserve and vascular remodeling in patients with hypertension and left ventricular hypertrophy. J Am Coll Cardiol. 2000;35:1654–1660. doi: 10.1016/s0735-1097(00)00594-5. [DOI] [PubMed] [Google Scholar]
- Harada M, Takeishi Y, Arimoto T, Niizeki T, Kitahara T, Goto K, Walsh RA, Kubota I. Diacylglycerol kinase zeta attenuates pressure overload-induced cardiac hypertrophy. Circ J. 2007;71:276–282. doi: 10.1253/circj.71.276. [DOI] [PubMed] [Google Scholar]
- Harding JD, Piacentino V, 3rd, Gaughan JP, Houser SR, Margulies KB. Electrophysiological alterations after mechanical circulatory support in patients with advanced cardiac failure. Circulation. 2001;104:1241–1247. doi: 10.1161/hc3601.095718. [DOI] [PubMed] [Google Scholar]
- He W, Kang YJ. Ischemia-induced Copper Loss and Suppression of Angiogenesis in the Pathogenesis of Myocardial Infarction. Cardiovasc Toxicol. 2012 doi: 10.1007/s12012-012-9174-y. [DOI] [PubMed] [Google Scholar]
- Heerdt PM, Holmes JW, Cai B, Barbone A, Madigan JD, Reiken S, Lee DL, Oz MC, Marks AR, Burkhoff D. Chronic unloading by left ventricular assist device reverses contractile dysfunction and alters gene expression in end-stage heart failure. Circulation. 2000;102:2713–2719. doi: 10.1161/01.cir.102.22.2713. [DOI] [PubMed] [Google Scholar]
- Heineke J, Auger-Messier M, Correll RN, Xu J, Benard MJ, Yuan W, Drexler H, Parise LV, Molkentin JD. CIB1 is a regulator of pathological cardiac hypertrophy. Nat Med. 2010;16:872–879. doi: 10.1038/nm.2181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrington CS, Tsirka AE. Pediatric cardiac transplantation. Semin Thorac Cardiovasc Surg. 2004;16:404–409. doi: 10.1053/j.semtcvs.2004.09.008. [DOI] [PubMed] [Google Scholar]
- Higashikuni Y, Sainz J, Nakamura K, Takaoka M, Enomoto S, Iwata H, Tanaka K, Sahara M, Hirata Y, Nagai R, Sata M. The ATP-Binding Cassette Transporter ATP-Binding Cassette Transporter Subfamily G Member 2 Protects Against Pressure Overload-Induced Cardiac Hypertrophy and Heart Failure by Promoting Angiogenesis and Antioxidant Response. Arterioscler Thromb Vasc Biol. 2011 doi: 10.1161/ATVBAHA.111.240341. [DOI] [PubMed] [Google Scholar]
- Hirsila M, Koivunen P, Xu L, Seeley T, Kivirikko KI, Myllyharju J. Effect of desferrioxamine and metals on the hydroxylases in the oxygen sensing pathway. FASEB J. 2005;19:1308–1310. doi: 10.1096/fj.04-3399fje. [DOI] [PubMed] [Google Scholar]
- Hoeben A, Landuyt B, Highley MS, Wildiers H, Van Oosterom AT, De Bruijn EA. Vascular endothelial growth factor and angiogenesis. Pharmacol Rev. 2004;56:549–580. doi: 10.1124/pr.56.4.3. [DOI] [PubMed] [Google Scholar]
- Holtwick R, van Eickels M, Skryabin BV, Baba HA, Bubikat A, Begrow F, Schneider MD, Garbers DL, Kuhn M. Pressure-independent cardiac hypertrophy in mice with cardiomyocyte-restricted inactivation of the atrial natriuretic peptide receptor guanylyl cyclase-A. J Clin Invest. 2003;111:1399–1407. doi: 10.1172/JCI17061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hon JKF, Yacoub MH. Bridge to recovery with the use of left ventricular assist device and clenbuterol. Ann Thorac Surg. 2003;75:S36–S41. doi: 10.1016/s0003-4975(03)00460-0. [DOI] [PubMed] [Google Scholar]
- Hoyer-Hansen M, Bastholm L, Szyniarowski P, Campanella M, Szabadkai G, Farkas T, Bianchi K, Fehrenbacher N, Elling F, Rizzuto R, Mathiasen IS, Jaattela M. Control of macroautophagy by calcium, calmodulin-dependent kinase kinase-beta, and Bcl-2. Mol Cell. 2007;25:193–205. doi: 10.1016/j.molcel.2006.12.009. [DOI] [PubMed] [Google Scholar]
- Huang LE, Gu J, Schau M, Bunn HF. Regulation of hypoxia-inducible factor 1alpha is mediated by an O2-dependent degradation domain via the ubiquitin-proteasome pathway. Proc Natl Acad Sci U S A. 1998;95:7987–7992. doi: 10.1073/pnas.95.14.7987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Y, Hickey RP, Yeh JL, Liu D, Dadak A, Young LH, Johnson RS, Giordano FJ. Cardiac myocyte-specific HIF-1alpha deletion alters vascularization, energy availability, calcium flux, and contractility in the normoxic heart. FASEB J. 2004a;18:1138–1140. doi: 10.1096/fj.04-1510fje. [DOI] [PubMed] [Google Scholar]
- Huang Y, Hunyor SN, Jiang L, Kawaguchi O, Shirota K, Ikeda Y, Yuasa T, Gallagher G, Zeng B, Zheng X. Remodeling of the chronic severely failing ischemic sheep heart after coronary microembolization: functional, energetic, structural, and cellular responses. Am J Physiol Heart Circ Physiol. 2004b;286:H2141–H2150. doi: 10.1152/ajpheart.00829.2003. [DOI] [PubMed] [Google Scholar]
- Hudlicka O, Brown M, Egginton S. Angiogenesis in skeletal and cardiac muscle. Physiol Rev. 1992;72:369–417. doi: 10.1152/physrev.1992.72.2.369. [DOI] [PubMed] [Google Scholar]
- Hughes GC, Biswas SS, Yin B, Coleman RE, DeGrado TR, Landolfo CK, Lowe JE, Annex BH, Landolfo KP. Therapeutic angiogenesis in chronically ischemic porcine myocardium: comparative effects of bFGF and VEGF. Ann Thorac Surg. 2004;77:812–818. doi: 10.1016/j.athoracsur.2003.09.060. [DOI] [PubMed] [Google Scholar]
- Huo Y, Linares CO, Kassab GS. Capillary perfusion and wall shear stress are restored in the coronary circulation of hypertrophic right ventricle. Circ Res. 2007;100:273–283. doi: 10.1161/01.RES.0000257777.83431.13. [DOI] [PubMed] [Google Scholar]
- Huss JM, Kelly DP. Mitochondrial energy metabolism in heart failure: a question of balance. J Clin Invest. 2005;115:547–555. doi: 10.1172/JCI200524405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingwall JS, Kramer MF, Fifer MA, Lorell BH, Shemin R, Grossman W, Allen PD. The creatine kinase system in normal and diseased human myocardium. N Engl J Med. 1985;313:1050–1054. doi: 10.1056/NEJM198510243131704. [DOI] [PubMed] [Google Scholar]
- Ingwall JS, Weiss RG. Is the failing heart energy starved? On using chemical energy to support cardiac function. Circ Res. 2004;95:135–145. doi: 10.1161/01.RES.0000137170.41939.d9. [DOI] [PubMed] [Google Scholar]
- Inoki K, Corradetti MN, Guan KL. Dysregulation of the TSC-mTOR pathway in human disease. Nat Genet. 2005;37:19–24. doi: 10.1038/ng1494. [DOI] [PubMed] [Google Scholar]
- Ishigai Y, Mori T, Ikeda T, Fukuzawa A, Shibano T. Role of bradykinin-NO pathway in prevention of cardiac hypertrophy by ACE inhibitor in rat cardiomyocytes. Am J Physiol. 1997;273:H2659–H2663. doi: 10.1152/ajpheart.1997.273.6.H2659. [DOI] [PubMed] [Google Scholar]
- Ivan M, Kondo K, Yang H, Kim W, Valiando J, Ohh M, Salic A, Asara JM, Lane WS, Kaelin WG., Jr. HIFalpha targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. Science. 2001;292:464–468. doi: 10.1126/science.1059817. [DOI] [PubMed] [Google Scholar]
- Izumiya Y, Shiojima I, Sato K, Sawyer DB, Colucci WS, Walsh K. Vascular endothelial growth factor blockade promotes the transition from compensatory cardiac hypertrophy to failure in response to pressure overload. Hypertension. 2006;47:887–893. doi: 10.1161/01.HYP.0000215207.54689.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ, Kriegsheim A, Hebestreit HF, Mukherji M, Schofield CJ, Maxwell PH, Pugh CW, Ratcliffe PJ. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science. 2001;292:468–472. doi: 10.1126/science.1059796. [DOI] [PubMed] [Google Scholar]
- Janssens S, Pokreisz P, Schoonjans L, Pellens M, Vermeersch P, Tjwa M, Jans P, Scherrer-Crosbie M, Picard MH, Szelid Z, Gillijns H, Van de Werf F, Collen D, Bloch KD. Cardiomyocyte-specific overexpression of nitric oxide synthase 3 improves left ventricular performance and reduces compensatory hypertrophy after myocardial infarction. Circ Res. 2004;94:1256–1262. doi: 10.1161/01.RES.0000126497.38281.23. [DOI] [PubMed] [Google Scholar]
- Jiang Y, Reynolds C, Xiao C, Feng W, Zhou Z, Rodriguez W, Tyagi SC, Eaton JW, Saari JT, Kang YJ. Dietary copper supplementation reverses hypertrophic cardiomyopathy induced by chronic pressure overload in mice. J Exp Med. 2007;204:657–666. doi: 10.1084/jem.20061943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajstura J, Cheng W, Reiss K, Clark WA, Sonnenblick EH, Krajewski S, Reed JC, Olivetti G, Anversa P. Apoptotic and necrotic myocyte cell deaths are independent contributing variables of infarct size in rats. Lab Invest. 1996;74:86–107. [PubMed] [Google Scholar]
- Kakinuma Y, Miyauchi T, Yuki K, Murakoshi N, Goto K, Yamaguchi I. Novel molecular mechanism of increased myocardial endothelin-1 expression in the failing heart involving the transcriptional factor hypoxia-inducible factor-1alpha induced for impaired myocardial energy metabolism. Circulation. 2001;103:2387–2394. doi: 10.1161/01.cir.103.19.2387. [DOI] [PubMed] [Google Scholar]
- Kamath SA, Laskar SR, Yancy CW. Novel therapies for heart failure: vasopressin and selective aldosterone antagonists. Congest Heart Fail. 2005;11:21–29. doi: 10.1111/j.1527-5299.2005.03912.x. [DOI] [PubMed] [Google Scholar]
- Kang YJ, Zhou ZX, Wu H, Wang GW, Saari JT, Klein JB. Metallothionein inhibits myocardial apoptosis in copper-deficient mice: role of atrial natriuretic peptide. Lab Invest. 2000;80:745–757. doi: 10.1038/labinvest.3780078. [DOI] [PubMed] [Google Scholar]
- Karch R, Neumann F, Ullrich R, Neumuller J, Podesser BK, Neumann M, Schreiner W. The spatial pattern of coronary capillaries in patients with dilated, ischemic, or inflammatory cardiomyopathy. Cardiovasc Pathol. 2005;14:135–144. doi: 10.1016/j.carpath.2005.03.003. [DOI] [PubMed] [Google Scholar]
- Karpanen T, Bry M, Ollila HM, Seppanen-Laakso T, Liimatta E, Leskinen H, Kivela R, Helkamaa T, Merentie M, Jeltsch M, Paavonen K, Andersson LC, Mervaala E, Hassinen IE, Yla-Herttuala S, Oresic M, Alitalo K. Overexpression of vascular endothelial growth factor-B in mouse heart alters cardiac lipid metabolism and induces myocardial hypertrophy. Circ Res. 2008;103:1018–1026. doi: 10.1161/CIRCRESAHA.108.178459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassab GS, Imoto K, White FC, Rider CA, Fung YC, Bloor CM. Coronary arterial tree remodeling in right ventricular hypertrophy. Am J Physiol. 1993;265:H366–H375. doi: 10.1152/ajpheart.1993.265.1.H366. [DOI] [PubMed] [Google Scholar]
- Kassab GS, Schatz A, Imoto K, Fung YC. Remodeling of the bifurcation asymmetry of right coronary ventricular branches in hypertrophy. Ann Biomed Eng. 2000;28:424–430. doi: 10.1114/1.280. [DOI] [PubMed] [Google Scholar]
- Kaye D, Esler M. Sympathetic neuronal regulation of the heart in aging and heart failure. Cardiovasc Res. 2005;66:256–264. doi: 10.1016/j.cardiores.2005.02.012. [DOI] [PubMed] [Google Scholar]
- Ke Q, Kluz T, Costa M. Down-regulation of the expression of the FIH-1 and ARD-1 genes at the transcriptional level by nickel and cobalt in the human lung adenocarcinoma A549 cell line. Int J Environ Res Public Health. 2005;2:10–13. doi: 10.3390/ijerph2005010010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan R, Sheppard R. Fibrosis in heart disease: understanding the role of transforming growth factor-beta in cardiomyopathy, valvular disease and arrhythmia. Immunology. 2006;118:10–24. doi: 10.1111/j.1365-2567.2006.02336.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kherani AR, Oz MC. Ventricular assistance to bridge to transplantation. Surg Clin North Am. 2004;84:75–89. viii–ix. doi: 10.1016/S0039-6109(03)00221-4. [DOI] [PubMed] [Google Scholar]
- Kinoshita H, Kuwahara K, Nishida M, Jian Z, Rong X, Kiyonaka S, Kuwabara Y, Kurose H, Inoue R, Mori Y, Li Y, Nakagawa Y, Usami S, Fujiwara M, Yamada Y, Minami T, Ueshima K, Nakao K. Inhibition of TRPC6 channel activity contributes to the antihypertrophic effects of natriuretic peptides-guanylyl cyclase-A signaling in the heart. Circ Res. 2010;106:1849–1860. doi: 10.1161/CIRCRESAHA.109.208314. [DOI] [PubMed] [Google Scholar]
- Kinoshita M, Takano H, Takaichi S, Taenaka Y, Nakatani T. Influence of prolonged ventricular assistance on myocardial histopathology in intact heart. Ann Thorac Surg. 1996;61:640–645. doi: 10.1016/0003-4975(95)01087-4. [DOI] [PubMed] [Google Scholar]
- Klevay LM. Ischemic heart disease as copper deficiency. Adv Exp Med Biol. 1989;258:197–208. doi: 10.1007/978-1-4613-0537-8_17. [DOI] [PubMed] [Google Scholar]
- Kliche S, Waltenberger J. VEGF receptor signaling and endothelial function. IUBMB Life. 2001;52:61–66. doi: 10.1080/15216540252774784. [DOI] [PubMed] [Google Scholar]
- Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290:1717–1721. doi: 10.1126/science.290.5497.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klotz S, Jan Danser AH, Burkhoff D. Impact of left ventricular assist device (LVAD) support on the cardiac reverse remodeling process. Prog Biophys Mol Biol. 2008;97:479–496. doi: 10.1016/j.pbiomolbio.2008.02.002. [DOI] [PubMed] [Google Scholar]
- Koitabashi N, Aiba T, Hesketh GG, Rowell J, Zhang M, Takimoto E, Tomaselli GF, Kass DA. Cyclic GMP/PKG-dependent inhibition of TRPC6 channel activity and expression negatively regulates cardiomyocyte NFAT activation Novel mechanism of cardiac stress modulation by PDE5 inhibition. J Mol Cell Cardiol. 2010;48:713–724. doi: 10.1016/j.yjmcc.2009.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koneru S, Varma Penumathsa S, Thirunavukkarasu M, Vidavalur R, Zhan L, Singal PK, Engelman RM, Das DK, Maulik N. Sildenafil-mediated neovascularization and protection against myocardial ischaemia reperfusion injury in rats: role of VEGF/angiopoietin-1. J Cell Mol Med. 2008;12:2651–2664. doi: 10.1111/j.1582-4934.2008.00319.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kotera J, Grimes KA, Corbin JD, Francis SH. cGMP-dependent protein kinase protects cGMP from hydrolysis by phosphodiesterase-5. Biochem. J. 2003;372:419–426. doi: 10.1042/BJ20030107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubasiak LA, Hernandez OM, Bishopric NH, Webster KA. Hypoxia and acidosis activate cardiac myocyte death through the Bcl-2 family protein BNIP3. Proc Natl Acad Sci U S A. 2002;99:12825–12830. doi: 10.1073/pnas.202474099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kugler C, Malehsa D, Tegtbur U, Guetzlaff E, Meyer AL, Bara C, Haverich A, Strueber M. Health-related quality of life and exercise tolerance in recipients of heart transplants and left ventricular assist devices: a prospective, comparative study. J Heart Lung Transplant. 2011;30:204–210. doi: 10.1016/j.healun.2010.08.030. [DOI] [PubMed] [Google Scholar]
- Kukucka M, Potapov E, Stepanenko A, Weller K, Mladenow A, Kuppe H, Habazettl H. Acute impact of left ventricular unloading by left ventricular assist device on the right ventricle geometry and function: effect of nitric oxide inhalation. J Thorac Cardiovasc Surg. 2011;141:1009–1014. doi: 10.1016/j.jtcvs.2010.08.010. [DOI] [PubMed] [Google Scholar]
- Kuma A, Hatano M, Matsui M, Yamamoto A, Nakaya H, Yoshimori T, Ohsumi Y, Tokuhisa T, Mizushima N. The role of autophagy during the early neonatal starvation period. Nature. 2004;432:1032–1036. doi: 10.1038/nature03029. [DOI] [PubMed] [Google Scholar]
- Kyosola K. Mitochondrial injury and autophagocytosis within the myocardial cell after the cold ischaemic anoxic asystole. Scand J Thorac Cardiovasc Surg. 1981;15:83–85. doi: 10.3109/14017438109101028. [DOI] [PubMed] [Google Scholar]
- Lee SH, Wolf PL, Escudero R, Deutsch R, Jamieson SW, Thistlethwaite PA. Early expression of angiogenesis factors in acute myocardial ischemia and infarction. N Engl J Med. 2000;342:626–633. doi: 10.1056/NEJM200003023420904. [DOI] [PubMed] [Google Scholar]
- Leist M, Single B, Castoldi AF, Kuhnle S, Nicotera P. Intracellular adenosine triphosphate (ATP) concentration: a switch in the decision between apoptosis and necrosis. J Exp Med. 1997;185:1481–1486. doi: 10.1084/jem.185.8.1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leri A, Kajstura J, Anversa P. Cardiac stem cells and mechanisms of myocardial regeneration. Physiol Rev. 2005;85:1373–1416. doi: 10.1152/physrev.00013.2005. [DOI] [PubMed] [Google Scholar]
- Levine B, Klionsky DJ. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell. 2004;6:463–477. doi: 10.1016/s1534-5807(04)00099-1. [DOI] [PubMed] [Google Scholar]
- Li HL, Liu C, de Couto G, Ouzounian M, Sun M, Wang AB, Huang Y, He CW, Shi Y, Chen X, Nghiem MP, Liu Y, Chen M, Dawood F, Fukuoka M, Maekawa Y, Zhang L, Leask A, Ghosh AK, Kirshenbaum LA, Liu PP. Curcumin prevents and reverses murine cardiac hypertrophy. J Clin Invest. 2008;118:879–893. doi: 10.1172/JCI32865. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Li YY, Feng Y, McTiernan CF, Pei W, Moravec CS, Wang P, Rosenblum W, Kormos RL, Feldman AM. Downregulation of matrix metalloproteinases and reduction in collagen damage in the failing human heart after support with left ventricular assist devices. Circulation. 2001;104:1147–1152. doi: 10.1161/hc3501.095215. [DOI] [PubMed] [Google Scholar]
- Libonati JR. Cardiac remodeling and function following exercise and angiotensin II receptor antagonism. Eur J Appl Physiol. 2011 doi: 10.1007/s00421-011-2263-y. [DOI] [PubMed] [Google Scholar]
- Loebe M, Muller J, Hetzer R. Ventricular assistance for recovery of cardiac failure. Curr Opin Cardiol. 1999;14:234–248. doi: 10.1097/00001573-199905000-00008. [DOI] [PubMed] [Google Scholar]
- Losordo DW, Vale PR, Symes JF, Dunnington CH, Esakof DD, Maysky M, Ashare AB, Lathi K, Isner JM. Gene therapy for myocardial angiogenesis: initial clinical results with direct myocardial injection of phVEGF165 as sole therapy for myocardial ischemia. Circulation. 1998;98:2800–2804. doi: 10.1161/01.cir.98.25.2800. [DOI] [PubMed] [Google Scholar]
- Luo X, Hojayev B, Jiang N, Wang ZV, Tandan S, Rakalin A, Rothermel BA, Gillette TG, Hill JA. STIM1-dependent store-operated Ca2+ entry is required for pathological cardiac hypertrophy. J Mol Cell Cardiol. 2012;52:136–147. doi: 10.1016/j.yjmcc.2011.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madigan JD, Barbone A, Choudhri AF, Morales DL, Cai B, Oz MC, Burkhoff D. Time course of reverse remodeling of the left ventricle during support with a left ventricular assist device. J Thorac Cardiovasc Surg. 2001;121:902–908. doi: 10.1067/mtc.2001.112632. [DOI] [PubMed] [Google Scholar]
- Madonna R, De Caterina R. VEGF receptor switching in heart development and disease. Cardiovasc Res. 2009;84:4–6. doi: 10.1093/cvr/cvp270. [DOI] [PubMed] [Google Scholar]
- Makino A, Suarez J, Wang H, Belke DD, Scott BT, Dillmann WH. Thyroid hormone receptor-beta is associated with coronary angiogenesis during pathological cardiac hypertrophy. Endocrinology. 2009;150:2008–2015. doi: 10.1210/en.2008-0634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mancini DM, Beniaminovitz A, Levin H, Catanese K, Flannery M, DiTullio M, Savin S, Cordisco ME, Rose E, Oz M. Low incidence of myocardial recovery after left ventricular assist device implantation in patients with chronic heart failure. Circulation. 1998;98:2383–2389. doi: 10.1161/01.cir.98.22.2383. [DOI] [PubMed] [Google Scholar]
- Martin F, Linden T, Katschinski DM, Oehme F, Flamme I, Mukhopadhyay CK, Eckhardt K, Troger J, Barth S, Camenisch G, Wenger RH. Copper-dependent activation of hypoxia-inducible factor (HIF)-1: implications for ceruloplasmin regulation. Blood. 2005;105:4613–4619. doi: 10.1182/blood-2004-10-3980. [DOI] [PubMed] [Google Scholar]
- Massie BM. LVADs as long-term cardiac replacement: Success but on what terms and at what cost? J Card Fail. 2002;8:61–62. doi: 10.1054/jcaf.2002.33167. [DOI] [PubMed] [Google Scholar]
- Masson N, Willam C, Maxwell PH, Pugh CW, Ratcliffe PJ. Independent function of two destruction domains in hypoxia-inducible factor-alpha chains activated by prolyl hydroxylation. EMBO J. 2001;20:5197–5206. doi: 10.1093/emboj/20.18.5197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxwell P, Salnikow K. HIF-1: an oxygen and metal responsive transcription factor. Cancer Biol Ther. 2004;3:29–35. doi: 10.4161/cbt.3.1.547. [DOI] [PubMed] [Google Scholar]
- Maxwell PH, Wiesener MS, Chang GW, Clifford SC, Vaux EC, Cockman ME, Wykoff CC, Pugh CW, Maher ER, Ratcliffe PJ. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399:271–275. doi: 10.1038/20459. [DOI] [PubMed] [Google Scholar]
- Maybaum S, Mancini D, Xydas S, Starling RC, Aaronson K, Pagani FD, Miller LW, Margulies K, McRee S, Frazier OH, Torre-Amione G. Cardiac improvement during mechanical circulatory support: a prospective multicenter study of the LVAD Working Group. Circulation. 2007;115:2497–2505. doi: 10.1161/CIRCULATIONAHA.106.633180. [DOI] [PubMed] [Google Scholar]
- McCarthy PM, Nakatani S, Vargo R, Kottke-Marchant K, Harasaki H, James KB, Savage RM, Thomas JD. Structural and left ventricular histologic changes after implantable LVAD insertion. Ann Thorac Surg. 1995;59:609–613. doi: 10.1016/0003-4975(94)00953-8. [DOI] [PubMed] [Google Scholar]
- McKie PM, Cataliotti A, Lahr BD, Martin FL, Redfield MM, Bailey KR, Rodeheffer RJ, Burnett JC., Jr. The prognostic value of N-terminal pro-B-type natriuretic peptide for death and cardiovascular events in healthy normal and stage A/B heart failure subjects. J Am Coll Cardiol. 2010;55:2140–2147. doi: 10.1016/j.jacc.2010.01.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKinsey TA, Kass DA. Small-molecule therapies for cardiac hypertrophy: moving beneath the cell surface. Nat Rev Drug Discov. 2007;6:617–635. doi: 10.1038/nrd2193. [DOI] [PubMed] [Google Scholar]
- Meine TJ, Russell SD. A history of orthotopic heart transplantation. Cardiol Rev. 2005;13:190–196. doi: 10.1097/01.crd.0000134861.15430.a6. [DOI] [PubMed] [Google Scholar]
- Melgar-Lesmes P, Tugues S, Ros J, Fernandez-Varo G, Morales-Ruiz M, Rodes J, Jimenez W. Vascular endothelial growth factor and angiopoietin-2 play a major role in the pathogenesis of vascular leakage in cirrhotic rats. Gut. 2009;58:285–292. doi: 10.1136/gut.2008.155028. [DOI] [PubMed] [Google Scholar]
- Mihl C, Dassen WR, Kuipers H. Cardiac remodelling: concentric versus eccentric hypertrophy in strength and endurance athletes. Neth Heart J. 2008;16:129–133. doi: 10.1007/BF03086131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millauer B, Wizigmann-Voos S, Schnurch H, Martinez R, Moller NP, Risau W, Ullrich A. High affinity VEGF binding and developmental expression suggest Flk-1 as a major regulator of vasculogenesis and angiogenesis. Cell. 1993;72:835–846. doi: 10.1016/0092-8674(93)90573-9. [DOI] [PubMed] [Google Scholar]
- Minamino T, Kitakaze M. ER stress in cardiovascular disease. J Mol Cell Cardiol. 2010;48:1105–1110. doi: 10.1016/j.yjmcc.2009.10.026. [DOI] [PubMed] [Google Scholar]
- Moens AL, Takimoto E, Tocchetti CG, Chakir K, Bedja D, Cormaci G, Ketner EA, Majmudar M, Gabrielson K, Halushka MK, Mitchell JB, Biswal S, Channon KM, Wolin MS, Alp NJ, Paolocci N, Champion HC, Kass DA. Reversal of cardiac hypertrophy and fibrosis from pressure overload by tetrahydrobiopterin: efficacy of recoupling nitric oxide synthase as a therapeutic strategy. Circulation. 2008;117:2626–2636. doi: 10.1161/CIRCULATIONAHA.107.737031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukherjee D. Current clinical perspectives on myocardial angiogenesis. Mol Cell Biochem. 2004;264:157–167. doi: 10.1023/b:mcbi.0000044385.55012.8f. [DOI] [PubMed] [Google Scholar]
- Muller J, Wallukat G, Weng YG, Dandel M, Spiegelsberger S, Semrau S, Brandes K, Theodoridis V, Loebe M, Meyer R, Hetzer R. Weaning from mechanical cardiac support in patients with idiopathic dilated cardiomyopathy. Circulation. 1997;96:542–549. doi: 10.1161/01.cir.96.2.542. [DOI] [PubMed] [Google Scholar]
- Murohara T, Asahara T, Silver M, Bauters C, Masuda H, Kalka C, Kearney M, Chen D, Symes JF, Fishman MC, Huang PL, Isner JM. Nitric oxide synthase modulates angiogenesis in response to tissue ischemia. J Clin Invest. 1998;101:2567–2578. doi: 10.1172/JCI1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadal-Ginard B, Kajstura J, Anversa P, Leri A. A matter of life and death: cardiac myocyte apoptosis and regeneration. J Clin Invest. 2003;111:1457–1459. doi: 10.1172/JCI18611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagayama T, Hsu S, Zhang M, Koitabashi N, Bedja D, Gabrielson KL, Takimoto E, Kass DA. Sildenafil stops progressive chamber, cellular, and molecular remodeling and improves calcium handling and function in hearts with pre-existing advanced hypertrophy caused by pressure overload. J Am Coll Cardiol. 2009;53:207–215. doi: 10.1016/j.jacc.2008.08.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakai A, Yamaguchi O, Takeda T, Higuchi Y, Hikoso S, Taniike M, Omiya S, Mizote I, Matsumura Y, Asahi M, Nishida K, Hori M, Mizushima N, Otsu K. The role of autophagy in cardiomyocytes in the basal state and in response to hemodynamic stress. Nat Med. 2007;13:619–624. doi: 10.1038/nm1574. [DOI] [PubMed] [Google Scholar]
- Nemeh HW, Smedira NG. Mechanical treatment of heart failure: the growing role of LVADs and artificial hearts. Cleve Clin J Med. 2003;70:223–226. 229–233. doi: 10.3949/ccjm.70.3.223. [DOI] [PubMed] [Google Scholar]
- Neubauer S, Remkes H, Spindler M, Horn M, Wiesmann F, Prestle J, Walzel B, Ertl G, Hasenfuss G, Wallimann T. Downregulation of the Na(+)-creatine cotransporter in failing human myocardium and in experimental heart failure. Circulation. 1999;100:1847–1850. doi: 10.1161/01.cir.100.18.1847. [DOI] [PubMed] [Google Scholar]
- Nishi J, Minamino T, Miyauchi H, Nojima A, Tateno K, Okada S, Orimo M, Moriya J, Fong GH, Sunagawa K, Shibuya M, Komuro I. Vascular endothelial growth factor receptor-1 regulates postnatal angiogenesis through inhibition of the excessive activation of Akt. Circ Res. 2008;103:261–268. doi: 10.1161/CIRCRESAHA.108.174128. [DOI] [PubMed] [Google Scholar]
- Nishikimi T, Hagaman JR, Takahashi N, Kim HS, Matsuoka H, Smithies O, Maeda N. Increased susceptibility to heart failure in response to volume overload in mice lacking natriuretic peptide receptor-A gene. Cardiovasc Res. 2005;66:94–103. doi: 10.1016/j.cardiores.2004.12.014. [DOI] [PubMed] [Google Scholar]
- O'Neil RG. VASP: a TRPC4-associated phosphoprotein that mediates PKG-induced inhibition of store-operated calcium influx. Am J Physiol Renal Physiol. 2007;293:F1766–F1767. doi: 10.1152/ajprenal.00482.2007. [DOI] [PubMed] [Google Scholar]
- Ogletree-Hughes ML, Stull LB, Sweet WE, Smedira NG, McCarthy PM, Moravec CS. Mechanical unloading restores beta-adrenergic responsiveness and reverses receptor downregulation in the failing human heart. Circulation. 2001;104:881–886. doi: 10.1161/hc3301.094911. [DOI] [PubMed] [Google Scholar]
- Ohh M, Park CW, Ivan M, Hoffman MA, Kim TY, Huang LE, Pavletich N, Chau V, Kaelin WG. Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel-Lindau protein. Nat Cell Biol. 2000;2:423–427. doi: 10.1038/35017054. [DOI] [PubMed] [Google Scholar]
- Ohtomo S, Nangaku M, Izuhara Y, Takizawa S, Strihou CY, Miyata T. Cobalt ameliorates renal injury in an obese, hypertensive type 2 diabetes rat model. Nephrol Dial Transplant. 2008;23:1166–1172. doi: 10.1093/ndt/gfm715. [DOI] [PubMed] [Google Scholar]
- Olivetti G, Abbi R, Quaini F, Kajstura J, Cheng W, Nitahara JA, Quaini E, Di Loreto C, Beltrami CA, Krajewski S, Reed JC, Anversa P. Apoptosis in the failing human heart. N Engl J Med. 1997;336:1131–1141. doi: 10.1056/NEJM199704173361603. [DOI] [PubMed] [Google Scholar]
- Opgaard OS, Wang PH. IGF-I is a matter of heart. Growth Hormone & IGF Research. 2005;15:89–94. doi: 10.1016/j.ghir.2005.02.002. [DOI] [PubMed] [Google Scholar]
- Opie LH, Commerford PJ, Gersh BJ, Pfeffer MA. Controversies in ventricular remodelling. Lancet. 2006;367:356–367. doi: 10.1016/S0140-6736(06)68074-4. [DOI] [PubMed] [Google Scholar]
- Paillard M, Gomez L, Augeul L, Loufouat J, Lesnefsky EJ, Ovize M. Postconditioning inhibits mPTP opening independent of oxidative phosphorylation and membrane potential. J Mol Cell Cardiol. 2009;46:902–909. doi: 10.1016/j.yjmcc.2009.02.017. [DOI] [PubMed] [Google Scholar]
- Pamboukian SV, Tallaj JA, Brown RN, Holman WL, Blood M, George JF, Costanzo MR, Cadeiras M, Smallfield MC, McGiffin DC, Kirklin JK. Improvement in 2-year survival for ventricular assist device patients after implementation of an intensive surveillance protocol. J Heart Lung Transplant. 2011;30:879–887. doi: 10.1016/j.healun.2011.03.001. [DOI] [PubMed] [Google Scholar]
- Park JY, Li W, Zheng D, Zhai P, Zhao Y, Matsuda T, Vatner SF, Sadoshima J, Tian B. Comparative analysis of mRNA isoform expression in cardiac hypertrophy and development reveals multiple post-transcriptional regulatory modules. PLoS One. 2011;6:e22391. doi: 10.1371/journal.pone.0022391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasini E, Cargnioni A, Pastore F, Razzetti R, Bongrani S, Gitti GL, Ferrari R. Effect of nolomirole on monocrotaline-induced heart failure. Pharmacol Res. 2004;49:1–5. doi: 10.1016/s1043-6618(03)00246-9. [DOI] [PubMed] [Google Scholar]
- Pearlman JD, Hibberd MG, Chuang ML, Harada K, Lopez JJ, Gladstone SR, Friedman M, Sellke FW, Simons M. Magnetic resonance mapping demonstrates benefits of VEGF-induced myocardial angiogenesis. Nat Med. 1995;1:1085–1089. doi: 10.1038/nm1095-1085. [DOI] [PubMed] [Google Scholar]
- Pelouch V, Dixon IM, Golfman L, Beamish RE, Dhalla NS. Role of extracellular matrix proteins in heart function. Mol Cell Biochem. 1993;129:101–120. doi: 10.1007/BF00926359. [DOI] [PubMed] [Google Scholar]
- Popovic ZB, Khot UN, Novaro GM, Casas F, Greenberg NL, Garcia MJ, Francis GS, Thomas JD. Effects of sodium nitroprusside in aortic stenosis associated with severe heart failure: pressure-volume loop analysis using a numerical model. Am J Physiol Heart Circ Physiol. 2005;288:H416–H423. doi: 10.1152/ajpheart.00615.2004. [DOI] [PubMed] [Google Scholar]
- Qi X, Vallentin A, Churchill E, Mochly-Rosen D. deltaPKC participates in the endoplasmic reticulum stress-induced response in cultured cardiac myocytes and ischemic heart. J Mol Cell Cardiol. 2007;43:420–428. doi: 10.1016/j.yjmcc.2007.07.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu L, Ding X, Zhang Z, Kang YJ. Copper is required for cobalt-induced transcriptional activity of hypoxia-inducible factor-1. J Pharmacol Exp Ther. 2012 doi: 10.1124/jpet.112.194662. [DOI] [PubMed] [Google Scholar]
- Radovancevic B, Vrtovec B, de Kort E, Radovancevic R, Gregoric ID, Frazier OH. End-organ function in patients on long-term circulatory support with continuous- or pulsatile-flow assist devices. J Heart Lung Transplant. 2007;26:815–818. doi: 10.1016/j.healun.2007.05.012. [DOI] [PubMed] [Google Scholar]
- Regula KM, Ens K, Kirshenbaum LA. Inducible expression of BNIP3 provokes mitochondrial defects and hypoxia-mediated cell death of ventricular myocytes. Circ Res. 2002;91:226–231. doi: 10.1161/01.res.0000029232.42227.16. [DOI] [PubMed] [Google Scholar]
- Roberts DM, Kearney JB, Johnson JH, Rosenberg MP, Kumar R, Bautch VL. The vascular endothelial growth factor (VEGF) receptor Flt-1 (VEGFR-1) modulates Flk-1 (VEGFR-2) signaling during blood vessel formation. Am J Pathol. 2004;164:1531–1535. doi: 10.1016/S0002-9440(10)63711-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose EA, Gelijns AC, Moskowitz AJ, Heitjan DF, Stevenson LW, Dembitsky W, Long JW, Ascheim DD, Tierney AR, Levitan RG, Watson JT, Ronan NS, Shapiro PA, Lazar RM, Miller LW, Gupta L, Frazier OH, Desvigne-Nickens P, Oz MC, Poirier VL, Meier P. Long-Term Use of a Left Ventricular Assist Device for End-Stage Heart Failure. New England Journal of Medicine. 2001;345:1435–1443. doi: 10.1056/NEJMoa012175. [DOI] [PubMed] [Google Scholar]
- Ruzicka M, Skarda V, Leenen FH. Effects of ACE inhibitors on circulating versus cardiac angiotensin II in volume overload-induced cardiac hypertrophy in rats. Circulation. 1995;92:3568–3573. doi: 10.1161/01.cir.92.12.3568. [DOI] [PubMed] [Google Scholar]
- Sakurai Y, Ohgimoto K, Kataoka Y, Yoshida N, Shibuya M. Essential role of Flk-1 (VEGF receptor 2) tyrosine residue 1173 in vasculogenesis in mice. Proc Natl Acad Sci U S A. 2005;102:1076–1081. doi: 10.1073/pnas.0404984102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salzberg S, Lachat M, Zund G, Oechslin E, Schmid ER, DeBakey M, Turina M. Left ventricular assist device as bridge to heart transplantation--lessons learned with the MicroMed DeBakey axial blood flow pump. Eur J Cardiothorac Surg. 2003;24:113–118. doi: 10.1016/s1010-7940(03)00179-9. [DOI] [PubMed] [Google Scholar]
- Sano M, Minamino T, Toko H, Miyauchi H, Orimo M, Qin Y, Akazawa H, Tateno K, Kayama Y, Harada M, Shimizu I, Asahara T, Hamada H, Tomita S, Molkentin JD, Zou Y, Komuro I. p53-induced inhibition of Hif-1 causes cardiac dysfunction during pressure overload. Nature. 2007;446:444–448. doi: 10.1038/nature05602. [DOI] [PubMed] [Google Scholar]
- Sartoretto JL, Jin BY, Bauer M, Gertler FB, Liao R, Michel T. Regulation of VASP phosphorylation in cardiac myocytes: differential regulation by cyclic nucleotides and modulation of protein expression in diabetic and hypertrophic heart. Am J Physiol Heart Circ Physiol. 2009;297:H1697–H1710. doi: 10.1152/ajpheart.00595.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saucerman JJ, McCulloch AD. Mechanistic systems models of cell signaling networks: a case study of myocyte adrenergic regulation. Prog Biophys Mol Biol. 2004;85:261–278. doi: 10.1016/j.pbiomolbio.2004.01.005. [DOI] [PubMed] [Google Scholar]
- Saupe KW, Spindler M, Hopkins JC, Shen W, Ingwall JS. Kinetic, thermodynamic, and developmental consequences of deleting creatine kinase isoenzymes from the heart. Reaction kinetics of the creatine kinase isoenzymes in the intact heart. J Biol Chem. 2000;275:19742–19746. doi: 10.1074/jbc.M001932200. [DOI] [PubMed] [Google Scholar]
- Scheinin SA, Capek P, Radovancevic B, Duncan JM, McAllister HA, Jr., Frazier OH. The effect of prolonged left ventricular support on myocardial histopathology in patients with end-stage cardiomyopathy. ASAIO J. 1992;38:M271–M274. doi: 10.1097/00002480-199207000-00035. [DOI] [PubMed] [Google Scholar]
- Scherz-Shouval R, Shvets E, Elazar Z. Oxidation as a post-translational modification that regulates autophagy. Autophagy. 2007a;3:371–373. doi: 10.4161/auto.4214. [DOI] [PubMed] [Google Scholar]
- Scherz-Shouval R, Shvets E, Fass E, Shorer H, Gil L, Elazar Z. Reactive oxygen species are essential for autophagy and specifically regulate the activity of Atg4. EMBO J. 2007b;26:1749–1760. doi: 10.1038/sj.emboj.7601623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz Jel J, Witt SA, Glascock BJ, Nieman ML, Reiser PJ, Nix SL, Kimball TR, Doetschman T. TGF-beta1 mediates the hypertrophic cardiomyocyte growth induced by angiotensin II. J Clin Invest. 2002;109:787–796. doi: 10.1172/JCI14190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sedmera D, Cook AC, Shirali G, McQuinn TC. Current issues and perspectives in hypoplasia of the left heart. Cardiol Young. 2005;15:56–72. doi: 10.1017/S1047951105000132. [DOI] [PubMed] [Google Scholar]
- Semenza GL. HIF-1 and human disease: one highly involved factor. Genes Dev. 2000;14:1983–1991. [PubMed] [Google Scholar]
- Sethi R, Shao Q, Ren B, Saini HK, Takeda N, Dhalla NS. Changes in beta-adrenoceptors in heart failure due to myocardial infarction are attenuated by blockade of renin-angiotensin system. Mol Cell Biochem. 2004;263:11–20. [PubMed] [Google Scholar]
- Severino A, Campioni M, Straino S, Salloum FN, Schmidt N, Herbrand U, Frede S, Toietta G, Di Rocco G, Bussani R, Silvestri F, Piro M, Liuzzo G, Biasucci LM, Mellone P, Feroce F, Capogrossi M, Baldi F, Fandrey J, Ehrmann M, Crea F, Abbate A, Baldi A. Identification of protein disulfide isomerase as a cardiomyocyte survival factor in ischemic cardiomyopathy. J Am Coll Cardiol. 2007;50:1029–1037. doi: 10.1016/j.jacc.2007.06.006. [DOI] [PubMed] [Google Scholar]
- Sheehy SP, Huang S, Parker KK. Time-warped comparison of gene expression in adaptive and maladaptive cardiac hypertrophy. Circ Cardiovasc Genet. 2009;2:116–124. doi: 10.1161/CIRCGENETICS.108.806935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimamatsu M, Toshima H. Impaired coronary vasodilatory capacity after dipyridamole administration in hypertrophic cardiomyopathy. Jpn Heart J. 1987;28:387–401. doi: 10.1536/ihj.28.387. [DOI] [PubMed] [Google Scholar]
- Shimojo N, Jesmin S, Zaedi S, Otsuki T, Maeda S, Yamaguchi N, Aonuma K, Hattori Y, Miyauchi T. Contributory role of VEGF overexpression in endothelin-1-induced cardiomyocyte hypertrophy. Am J Physiol Heart Circ Physiol. 2007;293:H474–H481. doi: 10.1152/ajpheart.00922.2006. [DOI] [PubMed] [Google Scholar]
- Shintani T, Klionsky DJ. Autophagy in health and disease: a double-edged sword. Science. 2004;306:990–995. doi: 10.1126/science.1099993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiojima I, Sato K, Izumiya Y, Schiekofer S, Ito M, Liao R, Colucci WS, Walsh K. Disruption of coordinated cardiac hypertrophy and angiogenesis contributes to the transition to heart failure. J Clin Invest. 2005;115:2108–2118. doi: 10.1172/JCI24682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipp JC. Glucose regulation: role of alpha and beta cells of pancreatic islets. J Med Assoc State Ala. 1961;31:115–118. [PubMed] [Google Scholar]
- Shliakhover VE, Zinkovich II, Trankovskii AM, Karakash SA. [The copper content of the myocardium in the acute form of ischemic heart disease] Vrach Delo. 1990:11–13. [PubMed] [Google Scholar]
- Shyu KG, Liou JY, Wang BW, Fang WJ, Chang H. Carvedilol prevents cardiac hypertrophy and overexpression of hypoxia-inducible factor-1alpha and vascular endothelial growth factor in pressure-overloaded rat heart. J Biomed Sci. 2005;12:409–420. doi: 10.1007/s11373-005-3008-x. [DOI] [PubMed] [Google Scholar]
- Smedira NG, Hoercher KJ, Yoon DY, Rajeswaran J, Klingman L, Starling RC, Blackstone EH. Bridge to transplant experience: factors influencing survival to and after cardiac transplant. J Thorac Cardiovasc Surg. 2010;139:1295–1305. 1305, e1291–e1294. doi: 10.1016/j.jtcvs.2009.12.006. [DOI] [PubMed] [Google Scholar]
- Somogyi A, Ruzicska E, Blazovics A, Ver A, Rosta K, Toth M. Insulin treatment decreases the antioxidant defense mechanism in experimental diabetes. Med Sci Monit. 2005;11:BR206–BR211. [PubMed] [Google Scholar]
- Song LJ, Wang GL, Liu J, Qiu QY, Ou JH, Guan YY. Cellular mechanisms of reduced sarcoplasmic reticulum Ca2+ content in L-thyroxin induced rat ventricular hypertrophy. Acta Pharmacol Sin. 2008;29:430–436. doi: 10.1111/j.1745-7254.2008.00763.x. [DOI] [PubMed] [Google Scholar]
- Song YH, Gehmert S, Sadat S, Pinkernell K, Bai X, Matthias N, Alt E. VEGF is critical for spontaneous differentiation of stem cells into cardiomyocytes. Biochem Biophys Res Commun. 2007;354:999–1003. doi: 10.1016/j.bbrc.2007.01.095. [DOI] [PubMed] [Google Scholar]
- Spann JC, Van Meter C. Cardiac transplantation. Surg Clin North Am. 1998;78:679–690. doi: 10.1016/s0039-6109(05)70344-3. [DOI] [PubMed] [Google Scholar]
- Stilli D, Bocchi L, Berni R, Zaniboni M, Cacciani F, Chaponnier C, Musso E, Gabbiani G, Clement S. Correlation of alpha-skeletal actin expression, ventricular fibrosis and heart function with the degree of pressure overload cardiac hypertrophy in rats. Exp Physiol. 2006;91:571–580. doi: 10.1113/expphysiol.2005.032607. [DOI] [PubMed] [Google Scholar]
- Strueber M, O'Driscoll G, Jansz P, Khaghani A, Levy WC, Wieselthaler GM. Multicenter evaluation of an intrapericardial left ventricular assist system. J Am Coll Cardiol. 2011;57:1375–1382. doi: 10.1016/j.jacc.2010.10.040. [DOI] [PubMed] [Google Scholar]
- Su H, Takagawa J, Huang Y, Arakawa-Hoyt J, Pons J, Grossman W, Kan YW. Additive effect of AAV-mediated angiopoietin-1 and VEGF expression on the therapy of infarcted heart. Int J Cardiol. 2009;133:191–197. doi: 10.1016/j.ijcard.2007.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suckau L, Fechner H, Chemaly E, Krohn S, Hadri L, Kockskamper J, Westermann D, Bisping E, Ly H, Wang X, Kawase Y, Chen J, Liang L, Sipo I, Vetter R, Weger S, Kurreck J, Erdmann V, Tschope C, Pieske B, Lebeche D, Schultheiss HP, Hajjar RJ, Poller WC. Long-term cardiac-targeted RNA interference for the treatment of heart failure restores cardiac function and reduces pathological hypertrophy. Circulation. 2009;119:1241–1252. doi: 10.1161/CIRCULATIONAHA.108.783852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun M, Chen M, Dawood F, Zurawska U, Li JY, Parker T, Kassiri Z, Kirshenbaum LA, Arnold M, Khokha R, Liu PP. Tumor necrosis factor-alpha mediates cardiac remodeling and ventricular dysfunction after pressure overload state. Circulation. 2007;115:1398–1407. doi: 10.1161/CIRCULATIONAHA.106.643585. [DOI] [PubMed] [Google Scholar]
- Suzuki G, Lee TC, Fallavollita JA, Canty JM., Jr. Adenoviral gene transfer of FGF-5 to hibernating myocardium improves function and stimulates myocytes to hypertrophy and reenter the cell cycle. Circ Res. 2005;96:767–775. doi: 10.1161/01.RES.0000162099.01268.d1. [DOI] [PubMed] [Google Scholar]
- Takahashi S. Vascular endothelial growth factor (VEGF), VEGF receptors and their inhibitors for antiangiogenic tumor therapy. Biol Pharm Bull. 2011;34:1785–1788. doi: 10.1248/bpb.34.1785. [DOI] [PubMed] [Google Scholar]
- Takimoto E, Champion HC, Li M, Belardi D, Ren S, Rodriguez ER, Bedja D, Gabrielson KL, Wang Y, Kass DA. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nat Med. 2005;11:214–222. doi: 10.1038/nm1175. [DOI] [PubMed] [Google Scholar]
- Tamirisa KP, Mukherjee D. Gene therapy in cardiovascular diseases. Curr Gene Ther. 2002;2:427–435. doi: 10.2174/1566523023347643. [DOI] [PubMed] [Google Scholar]
- Tang KM, Wang GR, Lu P, Karas RH, Aronovitz M, Heximer SP, Kaltenbronn KM, Blumer KJ, Siderovski DP, Zhu Y, Mendelsohn ME. Regulator of G-protein signaling-2 mediates vascular smooth muscle relaxation and blood pressure. Nat Med. 2003;9:1506–1512. doi: 10.1038/nm958. [DOI] [PubMed] [Google Scholar]
- Tanimoto K, Makino Y, Pereira T, Poellinger L. Mechanism of regulation of the hypoxia-inducible factor-1 alpha by the von Hippel-Lindau tumor suppressor protein. EMBO J. 2000;19:4298–4309. doi: 10.1093/emboj/19.16.4298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniyama Y, Ito M, Sato K, Kuester C, Veit K, Tremp G, Liao R, Colucci WS, Ivashchenko Y, Walsh K, Shiojima I. Akt3 overexpression in the heart results in progression from adaptive to maladaptive hypertrophy. J Mol Cell Cardiol. 2005;38:375–385. doi: 10.1016/j.yjmcc.2004.12.002. [DOI] [PubMed] [Google Scholar]
- Tanno AP, das Neves VJ, Rosa KT, Cunha TS, Giordano FC, Calil CM, Guzzoni V, Fernandes T, de Oliveira EM, Novaes PD, Irigoyen MC, Moura MJ, Marcondes FK. Nandrolone and resistance training induce heart remodeling: role of fetal genes and implications for cardiac pathophysiology. Life Sci. 2011;89:631–637. doi: 10.1016/j.lfs.2011.08.004. [DOI] [PubMed] [Google Scholar]
- Tao Z, Chen B, Tan X, Zhao Y, Wang L, Zhu T, Cao K, Yang Z, Kan YW, Su H. Coexpression of VEGF and angiopoietin-1 promotes angiogenesis and cardiomyocyte proliferation reduces apoptosis in porcine myocardial infarction (MI) heart. Proc Natl Acad Sci U S A. 2011;108:2064–2069. doi: 10.1073/pnas.1018925108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terman BI, Dougher-Vermazen M, Carrion ME, Dimitrov D, Armellino DC, Gospodarowicz D, Bohlen P. Identification of the KDR tyrosine kinase as a receptor for vascular endothelial cell growth factor. Biochem Biophys Res Commun. 1992;187:1579–1586. doi: 10.1016/0006-291x(92)90483-2. [DOI] [PubMed] [Google Scholar]
- Thuerauf DJ, Marcinko M, Gude N, Rubio M, Sussman MA, Glembotski CC. Activation of the unfolded protein response in infarcted mouse heart and hypoxic cultured cardiac myocytes. Circ Res. 2006;99:275–282. doi: 10.1161/01.RES.0000233317.70421.03. [DOI] [PubMed] [Google Scholar]
- Tirziu D, Chorianopoulos E, Moodie KL, Palac RT, Zhuang ZW, Tjwa M, Roncal C, Eriksson U, Fu Q, Elfenbein A, Hall AE, Carmeliet P, Moons L, Simons M. Myocardial hypertrophy in the absence of external stimuli is induced by angiogenesis in mice. J Clin Invest. 2007;117:3188–3197. doi: 10.1172/JCI32024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tolkovsky AM. Mitophagy. Biochim Biophys Acta. 2009;1793:1508–1515. doi: 10.1016/j.bbamcr.2009.03.002. [DOI] [PubMed] [Google Scholar]
- Torry RJ, O'Brien DM, Connell PM, Tomanek RJ. Dipyridamole-induced capillary growth in normal and hypertrophic hearts. Am J Physiol. 1992;262:H980–H986. doi: 10.1152/ajpheart.1992.262.4.H980. [DOI] [PubMed] [Google Scholar]
- van Berlo JH, Elrod JW, van den Hoogenhof MM, York AJ, Aronow BJ, Duncan SA, Molkentin JD. The transcription factor GATA-6 regulates pathological cardiac hypertrophy. Circ Res. 2010;107:1032–1040. doi: 10.1161/CIRCRESAHA.110.220764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Bilsen M, Smeets PJ, Gilde AJ, van der Vusse GJ. Metabolic remodelling of the failing heart: the cardiac burn-out syndrome? Cardiovasc Res. 2004;61:218–226. doi: 10.1016/j.cardiores.2003.11.014. [DOI] [PubMed] [Google Scholar]
- van Heerden D, Vosloo A, Nikinmaa M. Effects of short-term copper exposure on gill structure, metallothionein and hypoxia-inducible factor-1alpha (HIF-1alpha) levels in rainbow trout (Oncorhynchus mykiss) Aquat Toxicol. 2004;69:271–280. doi: 10.1016/j.aquatox.2004.06.002. [DOI] [PubMed] [Google Scholar]
- van Rooij E, Sutherland LB, Thatcher JE, DiMaio JM, Naseem RH, Marshall WS, Hill JA, Olson EN. Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci U S A. 2008;105:13027–13032. doi: 10.1073/pnas.0805038105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanden Hoek TL, Li C, Shao Z, Schumacker PT, Becker LB. Significant levels of oxidants are generated by isolated cardiomyocytes during ischemia prior to reperfusion. J Mol Cell Cardiol. 1997;29:2571–2583. doi: 10.1006/jmcc.1997.0497. [DOI] [PubMed] [Google Scholar]
- Vandivier RW, Henson PM, Douglas IS. Burying the dead: the impact of failed apoptotic cell removal (efferocytosis) on chronic inflammatory lung disease. Chest. 2006;129:1673–1682. doi: 10.1378/chest.129.6.1673. [DOI] [PubMed] [Google Scholar]
- Vidavalur R, Penumathsa SV, Zhan L, Thirunavukkarasu M, Maulik N. Sildenafil induces angiogenic response in human coronary arteriolar endothelial cells through the expression of thioredoxin, hemeoxygenase and vascular endothelial growth factor. Vascul Pharmacol. 2006;45:91–95. doi: 10.1016/j.vph.2006.03.010. [DOI] [PubMed] [Google Scholar]
- Villari B, Hess OM, Moccetti D, Vassalli G, Krayenbuehl HP. Effect of progression of left ventricular hypertrophy on coronary artery dimensions in aortic valve disease. J Am Coll Cardiol. 1992;20:1073–1079. doi: 10.1016/0735-1097(92)90360-y. [DOI] [PubMed] [Google Scholar]
- Vonder Muhll I, Liu P, Webb G. Applying standard therapies to new targets: the use of ACE inhibitors and B-blockers for heart failure in adults with congenital heart disease. Int J Cardiol. 2004;97:25–33. doi: 10.1016/j.ijcard.2004.08.006. [DOI] [PubMed] [Google Scholar]
- Wallimann T, Dolder M, Schlattner U, Eder M, Hornemann T, O'Gorman E, Ruck A, Brdiczka D. Some new aspects of creatine kinase (CK): compartmentation, structure, function and regulation for cellular and mitochondrial bioenergetics and physiology. Biofactors. 1998;8:229–234. doi: 10.1002/biof.5520080310. [DOI] [PubMed] [Google Scholar]
- Wang CW, Klionsky DJ. The molecular mechanism of autophagy. Mol Med. 2003;9:65–76. [PMC free article] [PubMed] [Google Scholar]
- Wang L, Seidman JG, Seidman CE. Narrative review: harnessing molecular genetics for the diagnosis and management of hypertrophic cardiomyopathy. Ann Intern Med. 2010;152:513–520. W181. doi: 10.1059/0003-4819-152-8-201004200-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang QD, Bohlooly YM, Sjoquist PO. Murine models for the study of congestive heart failure: Implications for understanding molecular mechanisms and for drug discovery. J Pharmacol Toxicol Methods. 2004;50:163–174. doi: 10.1016/j.vascn.2004.05.005. [DOI] [PubMed] [Google Scholar]
- Weber KT. Monitoring tissue repair and fibrosis from a distance. Circulation. 1997;96:2488–2492. [PubMed] [Google Scholar]
- Weeks KL, McMullen JR. The athlete's heart vs. the failing heart: can signaling explain the two distinct outcomes? Physiology (Bethesda) 2011;26:97–105. doi: 10.1152/physiol.00043.2010. [DOI] [PubMed] [Google Scholar]
- Weinberg EO, Schoen FJ, George D, Kagaya Y, Douglas PS, Litwin SE, Schunkert H, Benedict CR, Lorell BH. Angiotensin-converting enzyme inhibition prolongs survival and modifies the transition to heart failure in rats with pressure overload hypertrophy due to ascending aortic stenosis. Circulation. 1994;90:1410–1422. doi: 10.1161/01.cir.90.3.1410. [DOI] [PubMed] [Google Scholar]
- Weis SM, Cheresh DA. Pathophysiological consequences of VEGF-induced vascular permeability. Nature. 2005;437:497–504. doi: 10.1038/nature03987. [DOI] [PubMed] [Google Scholar]
- Westaby S. Ventricular assist devices as destination therapy. Surg Clin North Am. 2004;84:91–123. doi: 10.1016/j.suc.2003.12.010. [DOI] [PubMed] [Google Scholar]
- Wester PO. Trace elements in human myocardial infarction determined by neutron activation analysis. Acta Med Scand. 1965;178:765–788. doi: 10.1111/j.0954-6820.1965.tb04329.x. [DOI] [PubMed] [Google Scholar]
- Wisneski JA, Gertz EW, Neese RA, Mayr M. Myocardial metabolism of free fatty acids. Studies with 14C-labeled substrates in humans. J Clin Invest. 1987;79:359–366. doi: 10.1172/JCI112820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witte KK, Nikitin NP, Parker AC, von Haehling S, Volk HD, Anker SD, Clark AL, Cleland JG. The effect of micronutrient supplementation on quality-of-life and left ventricular function in elderly patients with chronic heart failure. Eur Heart J. 2005;26:2238–2244. doi: 10.1093/eurheartj/ehi442. [DOI] [PubMed] [Google Scholar]
- Wohlschlaeger J, Schmitz KJ, Schmid C, Schmid KW, Keul P, Takeda A, Weis S, Levkau B, Baba HA. Reverse remodeling following insertion of left ventricular assist devices (LVAD): a review of the morphological and molecular changes. Cardiovasc Res. 2005;68:376–386. doi: 10.1016/j.cardiores.2005.06.030. [DOI] [PubMed] [Google Scholar]
- Wollert KC, Fiedler B, Gambaryan S, Smolenski A, Heineke J, Butt E, Trautwein C, Lohmann SM, Drexler H. Gene transfer of cGMP-dependent protein kinase I enhances the antihypertrophic effects of nitric oxide in cardiomyocytes. Hypertension. 2002;39:87–92. doi: 10.1161/hy1201.097292. [DOI] [PubMed] [Google Scholar]
- Wyllie AH. Death from inside out: an overview. Philos Trans R Soc Lond B Biol Sci. 1994;345:237–241. doi: 10.1098/rstb.1994.0099. [DOI] [PubMed] [Google Scholar]
- Xie H, Kang YJ. Role of copper in angiogenesis and its medicinal implications. Curr Med Chem. 2009;16:1304–1314. doi: 10.2174/092986709787846622. [DOI] [PubMed] [Google Scholar]
- Xie L, Collins JF. Transcriptional regulation of the Menkes copper ATPase (Atp7a) gene by hypoxia-inducible factor (HIF2{alpha}) in intestinal epithelial cells. Am J Physiol Cell Physiol. 2011;300:C1298–C1305. doi: 10.1152/ajpcell.00023.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie Z, Klionsky DJ. Autophagosome formation: core machinery and adaptations. Nat Cell Biol. 2007;9:1102–1109. doi: 10.1038/ncb1007-1102. [DOI] [PubMed] [Google Scholar]
- Xu J, Carretero OA, Liu YH, Yang F, Shesely EG, Oja-Tebbe N, Yang XP. Dual inhibition of ACE and NEP provides greater cardioprotection in mice with heart failure. J Card Fail. 2004;10:83–89. [PubMed] [Google Scholar]
- Yan L, Vatner DE, Kim SJ, Ge H, Masurekar M, Massover WH, Yang G, Matsui Y, Sadoshima J, Vatner SF. Autophagy in chronically ischemic myocardium. Proc Natl Acad Sci U S A. 2005;102:13807–13812. doi: 10.1073/pnas.0506843102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, Wu LJ, Tashino S, Onodera S, Ikejima T. Reactive oxygen species and nitric oxide regulate mitochondria-dependent apoptosis and autophagy in evodiamine-treated human cervix carcinoma HeLa cells. Free Radic Res. 2008;42:492–504. doi: 10.1080/10715760802112791. [DOI] [PubMed] [Google Scholar]
- Yang KC, Jay PY, McMullen JR, Nerbonne JM. Enhanced cardiac PI3Kalpha signalling mitigates arrhythmogenic electrical remodelling in pathological hypertrophy and heart failure. Cardiovasc Res. 2012;93:252–262. doi: 10.1093/cvr/cvr283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X, Cepko CL. Flk-1, a receptor for vascular endothelial growth factor (VEGF), is expressed by retinal progenitor cells. J Neurosci. 1996;16:6089–6099. doi: 10.1523/JNEUROSCI.16-19-06089.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaoita H, Ogawa K, Maehara K, Maruyama Y. Apoptosis in relevant clinical situations: contribution of apoptosis in myocardial infarction. Cardiovasc Res. 2000;45:630–641. doi: 10.1016/s0008-6363(99)00349-1. [DOI] [PubMed] [Google Scholar]
- Yitzhaki S, Huang C, Liu W, Lee Y, Gustafsson AB, Mentzer RM, Jr., Gottlieb RA. Autophagy is required for preconditioning by the adenosine A1 receptor-selective agonist CCPA. Basic Res Cardiol. 2009;104:157–167. doi: 10.1007/s00395-009-0006-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimori T. Autophagy: a regulated bulk degradation process inside cells. Biochem Biophys Res Commun. 2004;313:453–458. doi: 10.1016/j.bbrc.2003.07.023. [DOI] [PubMed] [Google Scholar]
- Yuan Y, Hilliard G, Ferguson T, Millhorn DE. Cobalt inhibits the interaction between hypoxia-inducible factor-alpha and von Hippel-Lindau protein by direct binding to hypoxia-inducible factor-alpha. J Biol Chem. 2003;278:15911–15916. doi: 10.1074/jbc.M300463200. [DOI] [PubMed] [Google Scholar]
- Yue Z, Jin S, Yang C, Levine AJ, Heintz N. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci U S A. 2003;100:15077–15082. doi: 10.1073/pnas.2436255100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zafeiridis A, Jeevanandam V, Houser SR, Margulies KB. Regression of cellular hypertrophy after left ventricular assist device support. Circulation. 1998;98:656–662. doi: 10.1161/01.cir.98.7.656. [DOI] [PubMed] [Google Scholar]
- Zama N, Towns R. Cardiac copper, magnesium, and zinc in recent and old myocardial infarction. Biological Trace Element Research. 1986;10:201–208. doi: 10.1007/BF02795618. [DOI] [PubMed] [Google Scholar]
- Zeng H, Dvorak HF, Mukhopadhyay D. Vascular permeability factor (VPF)/vascular endothelial growth factor (VEGF) peceptor-1 down-modulates VPF/VEGF receptor-2-mediated endothelial cell proliferation, but not migration, through phosphatidylinositol 3-kinase-dependent pathways. J Biol Chem. 2001;276:26969–26979. doi: 10.1074/jbc.M103213200. [DOI] [PubMed] [Google Scholar]
- Zhang J, Narula J. Molecular biology of myocardial recovery. Surg Clin North Am. 2004;84:223–242. doi: 10.1016/S0039-6109(03)00222-6. [DOI] [PubMed] [Google Scholar]
- Zhang M, Takimoto E, Hsu S, Lee DI, Nagayama T, Danner T, Koitabashi N, Barth AS, Bedja D, Gabrielson KL, Wang Y, Kass DA. Myocardial remodeling is controlled by myocyte-targeted gene regulation of phosphodiesterase type 5. J Am Coll Cardiol. 2010;56:2021–2030. doi: 10.1016/j.jacc.2010.08.612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang P, Mende U. Regulators of G-protein signaling in the heart and their potential as therapeutic targets. Circ Res. 2011;109:320–333. doi: 10.1161/CIRCRESAHA.110.231423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao M, Chow A, Powers J, Fajardo G, Bernstein D. Microarray analysis of gene expression after transverse aortic constriction in mice. Physiol Genomics. 2004;19:93–105. doi: 10.1152/physiolgenomics.00040.2004. [DOI] [PubMed] [Google Scholar]
- Zhong J, Basu R, Guo D, Chow FL, Byrns S, Schuster M, Loibner H, Wang XH, Penninger JM, Kassiri Z, Oudit GY. Angiotensin-converting enzyme 2 suppresses pathological hypertrophy, myocardial fibrosis, and cardiac dysfunction. Circulation. 2010;122:717–728. doi: 10.1161/CIRCULATIONAHA.110.955369. 718 p following 728. [DOI] [PubMed] [Google Scholar]
- Zhou L, Ma W, Yang Z, Zhang F, Lu L, Ding Z, Ding B, Ha T, Gao X, Li C. VEGF165 and angiopoietin-1 decreased myocardium infarct size through phosphatidylinositol-3 kinase and Bcl-2 pathways. Gene Ther. 2005;12:196–202. doi: 10.1038/sj.gt.3302416. [DOI] [PubMed] [Google Scholar]
- Zhou Y, Bourcy K, Kang YJ. Copper-induced regression of cardiomyocyte hypertrophy is associated with enhanced vascular endothelial growth factor receptor-1 signalling pathway. Cardiovasc Res. 2009a;84:54–63. doi: 10.1093/cvr/cvp178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y, Jiang Y, Kang YJ. Copper reverses cardiomyocyte hypertrophy through vascular endothelial growth factor-mediated reduction in the cell size. J Mol Cell Cardiol. 2008;45:106–117. doi: 10.1016/j.yjmcc.2008.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z, Johnson WT, Kang YJ. Regression of copper-deficient heart hypertrophy: reduction in the size of hypertrophic cardiomyocytes. J Nutr Biochem. 2009b;20:621–628. doi: 10.1016/j.jnutbio.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zisa D, Shabbir A, Mastri M, Suzuki G, Lee T. Intramuscular VEGF repairs the failing heart: role of host-derived growth factors and mobilization of progenitor cells. Am J Physiol Regul Integr Comp Physiol. 2009;297:R1503–R1515. doi: 10.1152/ajpregu.00227.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo X, Xie H, Dong D, Jiang N, Zhu H, Kang YJ. Cytochrome c oxidase is essential for copper-induced regression of cardiomyocyte hypertrophy. Cardiovasc Toxicol. 2010;10:208–215. doi: 10.1007/s12012-010-9080-0. [DOI] [PubMed] [Google Scholar]