Abstract
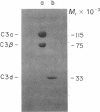
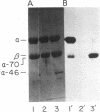
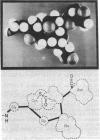
Human complement protein C3 was inactivated by using methylamine and thereby generating a SH group from the internal thiol ester. The protein was coupled via this SH group to activated thiol-Sepharose and digested with elastase. Fragment C3d remained attached to the thiol-Sepharose and was subsequently eluted with L-cysteine. Concomitantly, the original SH group was regenerated, and it was then labeled with iodo[2-3H]acetic acid. Partial sequence analysis of the radiolabeled C3d fragment showed that both components of the thiol ester are located close to the amino terminus (residues 23 and 26). Specific chemical cleavage of the α-chain was achieved after S-cyanylation of the thiol. The two fragments obtained corresponded to the amino-terminal section (Mr ≈46,000) and the carboxy-terminal section (Mr ≈70,000). These results together indicate that fragment C3d occupies approximately positions 345-610 of the α-chain. The partial sequence of C3d was extended by completion of the sequence of a previously described tryptic peptide. Comparison of residues 1-49 of C3d with a peptide from α2-macroglobulin [Swenson, R. P. & Howard, J. B. (1980) J. Biol. Chem. 255, 8087-8091] shows a previously recognized identity of seven residues around the thiol ester site and a second region of identity around a known glycosylation site of α2-macroglobulin. The relationships among these proteins and protein C4 are discussed. An overall outline of the structure of C3 is presented, showing the locations of various fragments and cleavage sites. The thiol ester group places constraints on the local folding of the peptide chain; a possible conformation is suggested and discussed in relation to the mechanism of activation.
Keywords: active site, acyl group transfer, fragment C3d, γ-glutamylmethylamide, S-cyanocysteine
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bokisch V. A., Dierich M. P., Mūller-Eberhard H. J. Third component of complement (C3): structural properties in relation to functions. Proc Natl Acad Sci U S A. 1975 Jun;72(6):1989–1993. doi: 10.1073/pnas.72.6.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brade V., Hall R. E., Colten H. R. Biosynthesis of pro-C3, a precursor of the third component of complement. J Exp Med. 1977 Sep 1;146(3):759–765. doi: 10.1084/jem.146.3.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell R. D., Gagnon J., Porter R. R. Amino acid sequence around the proposed thiolester bond of human complement component C4 and comparison with the corresponding sequences from C3 and alpha 2-macroglobulin. Biosci Rep. 1981 May;1(5):423–429. doi: 10.1007/BF01116192. [DOI] [PubMed] [Google Scholar]
- Crossley L. G., Porter R. R. Purification of the human complement control protein C3b inactivator. Biochem J. 1980 Oct 1;191(1):173–182. doi: 10.1042/bj1910173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DALMASSO A. P., MUELLER-EBERHARD H. J. INTERACTION OF AUTOLOGOUS COMPLEMENT WITH RED CELLS IN THE ABSENCE OF ANTIBODY. Proc Soc Exp Biol Med. 1964 Dec;117:643–650. doi: 10.3181/00379727-117-29658. [DOI] [PubMed] [Google Scholar]
- Davies S. G., Sim R. B. Intramolecular general acid catalysis in the binding reactions of alpha 2-macroglobulin and complement components C3 and C4. Biosci Rep. 1981 Jun;1(6):461–468. doi: 10.1007/BF01121579. [DOI] [PubMed] [Google Scholar]
- Degani Y., Neumann H., Patchornik A. Selective cyanylation of sulfhydryl groups. J Am Chem Soc. 1970 Nov 18;92(23):6969–6971. doi: 10.1021/ja00726a043. [DOI] [PubMed] [Google Scholar]
- Degani Y., Patchornik A. Cyanylation of sulfhydryl groups by 2-nitro-5-thiocyanobenzoic acid. High-yield modification and cleavage of peptides at cysteine residues. Biochemistry. 1974 Jan 1;13(1):1–11. doi: 10.1021/bi00698a001. [DOI] [PubMed] [Google Scholar]
- Ghebrehiwet B., Müller-Eberhard H. J. C3e: an acidic fragment of human C3 with leukocytosis-inducing activity. J Immunol. 1979 Aug;123(2):616–621. [PubMed] [Google Scholar]
- Goldberger G., Abraham G. N., Williams J., Colten H. R. NH2-terminal sequence analysis of pro-C4, the precursor of the fourth component of guinea pig complement. J Biol Chem. 1980 Aug 10;255(15):7071–7074. [PubMed] [Google Scholar]
- Goldberger G., Thomas M. L., Tack B. F., Williams J., Colten H. R., Abraham G. N. NH2-terminal structure and cleavage of guinea pig pro-C3, the precursor of the third complement component. J Biol Chem. 1981 Dec 25;256(24):12617–12619. [PubMed] [Google Scholar]
- Gorski J. P., Howard J. B. Effect of methylamine on the structure and function of the fourth component of human complement, C4. J Biol Chem. 1980 Nov 10;255(21):10025–10028. [PubMed] [Google Scholar]
- Hammer C. H., Wirtz G. H., Renfer L., Gresham H. D., Tack B. F. Large scale isolation of functionally active components of the human complement system. J Biol Chem. 1981 Apr 25;256(8):3995–4006. [PubMed] [Google Scholar]
- Harpel P. C., Hayes M. B., Hugli T. E. Heat-induced fragmentation of human alpha 2-macroglobulin. J Biol Chem. 1979 Sep 10;254(17):8669–8678. [PubMed] [Google Scholar]
- Harpel P. C. Studies on human plasma alpha 2-macroglobulin-enzyme interactions. Evidence for proteolytic modification of the subunit chain structure. J Exp Med. 1973 Sep 1;138(3):508–521. doi: 10.1084/jem.138.3.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison R. A., Lachmann P. J. Novel cleavage products of the third component of human complement. Mol Immunol. 1980 Feb;17(2):219–228. doi: 10.1016/0161-5890(80)90074-7. [DOI] [PubMed] [Google Scholar]
- Harrison R. A., Thomas M. L., Tack B. F. Sequence determination of the thiolester site of the fourth component of human complement. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7388–7392. doi: 10.1073/pnas.78.12.7388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard J. B. Reactive site in human alpha 2-macroglobulin: circumstantial evidence for a thiolester. Proc Natl Acad Sci U S A. 1981 Apr;78(4):2235–2239. doi: 10.1073/pnas.78.4.2235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard J. B., Vermeulen M., Swenson R. P. The temperature-sensitive bond in human alpha 2-macroglobulin is the alkylamine-reactive site. J Biol Chem. 1980 May 10;255(9):3820–3823. [PubMed] [Google Scholar]
- Janatova J., Lorenz P. E., Schechter A. N., Prahl J. W., Tack B. F. Third component of human complement: appearance of a sulfhydryl group following chemical or enzymatic inactivation. Biochemistry. 1980 Sep 16;19(19):4471–4478. [PubMed] [Google Scholar]
- Janatova J., Tack B. F. Fourth component of human complement: studies of an amine-sensitive site comprised of a thiol component. Biochemistry. 1981 Apr 28;20(9):2394–2402. doi: 10.1021/bi00512a005. [DOI] [PubMed] [Google Scholar]
- Janatova J., Tack B. F., Prahl J. W. Third component of human complement: structural requirements for its function. Biochemistry. 1980 Sep 16;19(19):4479–4485. doi: 10.1021/bi00560a015. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lambris J. D., Dobson N. J., Ross G. D. Isolation of lymphocyte membrane complement receptor type two (the C3d receptor) and preparation of receptor-specific antibody. Proc Natl Acad Sci U S A. 1981 Mar;78(3):1828–1832. doi: 10.1073/pnas.78.3.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law S. K., Fearon D. T., Levine R. P. Action of the C3b-inactivator on the cell-bound C3b. J Immunol. 1979 Mar;122(3):759–765. [PubMed] [Google Scholar]
- Law S. K., Levine R. P. Interaction between the third complement protein and cell surface macromolecules. Proc Natl Acad Sci U S A. 1977 Jul;74(7):2701–2705. doi: 10.1073/pnas.74.7.2701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law S. K., Lichtenberg N. A., Holcombe F. H., Levine R. P. Interaction between the labile binding sites of the fourth (C4) and fifth (C5) human complement proteins and erythrocyte cell membranes. J Immunol. 1980 Aug;125(2):634–639. [PubMed] [Google Scholar]
- Law S. K., Lichtenberg N. A., Levine R. P. Covalent binding and hemolytic activity of complement proteins. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7194–7198. doi: 10.1073/pnas.77.12.7194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law S. K., Lichtenberg N. A., Levine R. P. Evidence for an ester linkage between the labile binding site of C3b and receptive surfaces. J Immunol. 1979 Sep;123(3):1388–1394. [PubMed] [Google Scholar]
- Lundwall A., Malmheden I., Hellman U., Sjöquist J. Interaction between the labile binding site of human C4 and methylamine. Scand J Immunol. 1981;13(2):199–203. doi: 10.1111/j.1365-3083.1981.tb00125.x. [DOI] [PubMed] [Google Scholar]
- Müller-Eberhard H. J., Götze O. C3 proactivator convertase and its mode of action. J Exp Med. 1972 Apr 1;135(4):1003–1008. doi: 10.1084/jem.135.4.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller-Eberhard H. J., Schreiber R. D. Molecular biology and chemistry of the alternative pathway of complement. Adv Immunol. 1980;29:1–53. doi: 10.1016/s0065-2776(08)60042-5. [DOI] [PubMed] [Google Scholar]
- Nagasawa S., Stroud R. M. Mechanism of action of the C3b inactivator: requirement for a high molecular weight cofactor (C3b-C4bINA cofactor) and production of a new C3b derivative (C3b'). Immunochemistry. 1977 Nov-Dec;14(11-12):749–756. doi: 10.1016/0019-2791(77)90345-7. [DOI] [PubMed] [Google Scholar]
- Narayanan A. S., Anwar R. A. The specificity of purified porcine pancreatic elastase. Biochem J. 1969 Aug;114(1):11–17. doi: 10.1042/bj1140011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pangburn M. K., Müller-Eberhard H. J. Relation of putative thioester bond in C3 to activation of the alternative pathway and the binding of C3b to biological targets of complement. J Exp Med. 1980 Oct 1;152(4):1102–1114. doi: 10.1084/jem.152.4.1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pangburn M. K., Schreiber R. D., Müller-Eberhard H. J. Human complement C3b inactivator: isolation, characterization, and demonstration of an absolute requirement for the serum protein beta1H for cleavage of C3b and C4b in solution. J Exp Med. 1977 Jul 1;146(1):257–270. doi: 10.1084/jem.146.1.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porter R. R., Reid K. B. Activation of the complement system by antibody-antigen complexes: the classical pathway. Adv Protein Chem. 1979;33:1–71. doi: 10.1016/s0065-3233(08)60458-1. [DOI] [PubMed] [Google Scholar]
- Reboul A., Thielens N., Villiers M. B., Colomb M. G. Structural changes in C4 produced by cleavage with C1-s. FEBS Lett. 1980 Jun 16;115(1):118–122. doi: 10.1016/0014-5793(80)80739-3. [DOI] [PubMed] [Google Scholar]
- Reid K. B., Porter R. R. The proteolytic activation systems of complement. Annu Rev Biochem. 1981;50:433–464. doi: 10.1146/annurev.bi.50.070181.002245. [DOI] [PubMed] [Google Scholar]
- Ross G. D., Polley M. J. Specificity of human lymphocyte complement receptors. J Exp Med. 1975 May 1;141(5):1163–1180. doi: 10.1084/jem.141.5.1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salvesen G. S., Sayers C. A., Barrett A. J. Further characterization of the covalent linking reaction of alpha 2-macroglobulin. Biochem J. 1981 May 1;195(2):453–461. doi: 10.1042/bj1950453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seya T., Nagasawa S. Limited proteolysis of the third component of human complement, C3, by heat treatment. J Biochem. 1981 Feb;89(2):659–664. doi: 10.1093/oxfordjournals.jbchem.a133243. [DOI] [PubMed] [Google Scholar]
- Sim R. B., Sim E. Autolytic fragmentation of complement components C3 and C4 under denaturing conditions, a property shared with alpha 2-macroglobulin. Biochem J. 1981 Jan 1;193(1):129–141. doi: 10.1042/bj1930129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sim R. B., Twose T. M., Paterson D. S., Sim E. The covalent-binding reaction of complement component C3. Biochem J. 1981 Jan 1;193(1):115–127. doi: 10.1042/bj1930115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sottrup-Jensen L., Hansen H. F., Mortensen S. B., Petersen T. E., Magnusson S. Sequence location of the reactive thiol ester in human alpha 2-macroglobulin. FEBS Lett. 1981 Jan 12;123(1):145–148. doi: 10.1016/0014-5793(81)80039-7. [DOI] [PubMed] [Google Scholar]
- Sottrup-Jensen L., Petersen T. E., Magnusson S. A thiol-ester in alpha 2-macroglobulin cleaved during proteinase complex formation. FEBS Lett. 1980 Dec 1;121(2):275–279. doi: 10.1016/0014-5793(80)80361-9. [DOI] [PubMed] [Google Scholar]
- Swenson R. P., Howard J. B. Amino acid sequence of the tryptic peptide containing the alkylamine-reactive site from human alpha 2-macroglobulin. Identification of gamma-glutamylmethylamide. J Biol Chem. 1980 Sep 10;255(17):8087–8091. [PubMed] [Google Scholar]
- Swenson R. P., Howard J. B. Characterization of alkylamine-sensitive site in alpha 2-macroglobulin. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4313–4316. doi: 10.1073/pnas.76.9.4313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tack B. F., Harrison R. A., Janatova J., Thomas M. L., Prahl J. W. Evidence for presence of an internal thiolester bond in third component of human complement. Proc Natl Acad Sci U S A. 1980 Oct;77(10):5764–5768. doi: 10.1073/pnas.77.10.5764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor J. C., Crawford I. P., Hugli T. E. Limited degradation of the third component (C3) of human complement by human leukocyte elastase (HLE): partial characterization of C3 fragments. Biochemistry. 1977 Jul 26;16(15):3390–3396. doi: 10.1021/bi00634a016. [DOI] [PubMed] [Google Scholar]
- Vanaman T. C., Stark G. R. A study of the sulfhydryl groups of the catalytic subunit of Escherichia coli aspartate transcarbamylase. The use of enzyme--5-thio-2-nitrobenzoate mixed disulfides as intermediates in modifying enzyme sulfhydryl groups. J Biol Chem. 1970 Jul 25;245(14):3565–3573. [PubMed] [Google Scholar]
- West C., Davis N. C., Forristal J., Herbst J., Spitzer R. Antigenic determinants of human beta-1c and beta-1g-globulins. J Immunol. 1966 Apr;96(4):650–658. [PubMed] [Google Scholar]
- Whaley K., Ruddy S. Modulation of C3b hemolytic activity by a plasma protein distinct from C3b inactivator. Science. 1976 Sep 10;193(4257):1011–1013. doi: 10.1126/science.948757. [DOI] [PubMed] [Google Scholar]
- von Zabern I., Nolte R., Vogt W. Treatment of human complement components C4 and C3 with amines or chaotropic ions. Evidence of a functional and structural change that provides uncleaved C4 and C3 with properties of their soluble activated froms, C4b and C3b. Scand J Immunol. 1981;13(5):413–431. doi: 10.1111/j.1365-3083.1981.tb00152.x. [DOI] [PubMed] [Google Scholar]