Abstract
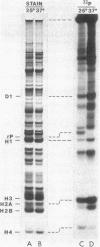
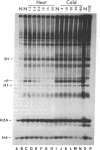
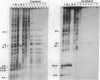
Ribosomes isolated from Drosophila melanogaster tissue culture cells labeled in vivo with 32Pi contain a single, heavily phosphorylated, ribosomal protein. As much as 40% of this protein is phosphorylated in cells cultured at 25 degrees C. The molecular weight and other characteristics of this protein suggest possible homology with ribosomal protein S6. Following a shift-up to 37 degrees C, the protein is specifically and quantitatively dephosphorylated. The kinetics of this dephosphorylation are rapid with a half-time on the order of a few minutes. These kinetics closely parallel the heat shock-induced breakdown of the preexisting polysome population.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashburner M., Bonner J. J. The induction of gene activity in drosophilia by heat shock. Cell. 1979 Jun;17(2):241–254. doi: 10.1016/0092-8674(79)90150-8. [DOI] [PubMed] [Google Scholar]
- Echalier G., Ohanessian A. In vitro culture of Drosophila melanogaster embryonic cells. In Vitro. 1970 Nov-Dec;6(3):162–172. doi: 10.1007/BF02617759. [DOI] [PubMed] [Google Scholar]
- Glover C. V., Vavra K. J., Guttman S. D., Gorovsky M. A. Heat shock and deciliation induce phosphorylation of histone H1 in T. pyriformis. Cell. 1981 Jan;23(1):73–77. doi: 10.1016/0092-8674(81)90271-3. [DOI] [PubMed] [Google Scholar]
- Howard G. A., Smith R. L., Gordon J. Ribosomal proteins: simplification of the methods to prepare them for gel electrophoresis. Anal Biochem. 1975 Jul;67(1):110–114. doi: 10.1016/0003-2697(75)90277-8. [DOI] [PubMed] [Google Scholar]
- Krüger C., Benecke B. J. In vitro translation of Drosophila heat-shock and non--heat-shock mRNAs in heterologous and homologous cell-free systems. Cell. 1981 Feb;23(2):595–603. doi: 10.1016/0092-8674(81)90155-0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lastick S. M., Nielsen P. J., McConkey E. H. Phosphorylation of ribosomal protein S6 in suspension cultured HeLa cells. Mol Gen Genet. 1977 Apr 29;152(3):223–230. doi: 10.1007/BF00693074. [DOI] [PubMed] [Google Scholar]
- Lindquist S. Translational efficiency of heat-induced messages in Drosophila melanogaster cells. J Mol Biol. 1980 Feb 25;137(2):151–158. doi: 10.1016/0022-2836(80)90322-8. [DOI] [PubMed] [Google Scholar]
- McConkey E. H. Composition of mammalian ribosomal subunits: a re-evaluation. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1379–1383. doi: 10.1073/pnas.71.4.1379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKenzie S. L., Henikoff S., Meselson M. Localization of RNA from heat-induced polysomes at puff sites in Drosophila melanogaster. Proc Natl Acad Sci U S A. 1975 Mar;72(3):1117–1121. doi: 10.1073/pnas.72.3.1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirault M. E., Goldschmidt-Clermont M., Moran L., Arrigo A. P., Tissières A. The effect of heat shock on gene expression in Drosophila melanogaster. Cold Spring Harb Symp Quant Biol. 1978;42(Pt 2):819–827. doi: 10.1101/sqb.1978.042.01.082. [DOI] [PubMed] [Google Scholar]
- Oliver D., Chalkley R. An electrophoretic analysis of Drosophila histones. I. Isolation and identification. Exp Cell Res. 1972 Aug;73(2):295–302. doi: 10.1016/0014-4827(72)90051-1. [DOI] [PubMed] [Google Scholar]
- Panyim S., Chalkley R. High resolution acrylamide gel electrophoresis of histones. Arch Biochem Biophys. 1969 Mar;130(1):337–346. doi: 10.1016/0003-9861(69)90042-3. [DOI] [PubMed] [Google Scholar]
- Rodriguez Alfageme C., Rudkin G. T., Cohen L. H. Isolation, properties and cellular distribution of D1, a chromosomal protein of Drosophila. Chromosoma. 1980;78(1):1–31. doi: 10.1007/BF00291907. [DOI] [PubMed] [Google Scholar]
- Rubin G. M., Hogness D. S. Effect of heat shock on the synthesis of low molecular weight RNAs in drosophilia: accumulation of a novel form of 5S RNA. Cell. 1975 Oct;6(2):207–213. doi: 10.1016/0092-8674(75)90011-2. [DOI] [PubMed] [Google Scholar]
- Scott M. P., Pardue M. L. Translational control in lysates of Drosophila melanogaster cells. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3353–3357. doi: 10.1073/pnas.78.6.3353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott M. P., Storti R. V., Pardue M. L., Rich A. Cell-free protein synthesis in lysates of Drosophila melanogaster cells. Biochemistry. 1979 Apr 17;18(8):1588–1594. doi: 10.1021/bi00575a032. [DOI] [PubMed] [Google Scholar]
- Storti R. V., Scott M. P., Rich A., Pardue M. L. Translational control of protein synthesis in response to heat shock in D. melanogaster cells. Cell. 1980 Dec;22(3):825–834. doi: 10.1016/0092-8674(80)90559-0. [DOI] [PubMed] [Google Scholar]
- Wool I. G. The structure and function of eukaryotic ribosomes. Annu Rev Biochem. 1979;48:719–754. doi: 10.1146/annurev.bi.48.070179.003443. [DOI] [PubMed] [Google Scholar]