Abstract
Purpose
We describe two patients with squamous cell papilloma of the conjunctiva due to human papilloma virus (HPV) and review the literature.
Patients and methods
Two patients with conjunctival tumors were examined and treated in the University Eye Clinic and diagnosed in the University Pathology Department, University Hospital of Ioannina, Greece. The first patient was a 48-year-old man presenting with an extended papillomatous lesion in bulbar conjunctiva covering part of the cornea of his right eye. The second patient was a 24-year-old man presenting with a polypoidal papillomatous lesion on the caruncle of his right eye. The two lesions were removed surgically, cryotherapy was applied to the adjacent conjunctiva, and topical mitomycin-C was used. The amniotic membrane was used to restore the conjunctival defect in the first patient. The two removed lesions were sent to the Pathology Department for histopathological examination. Immunohistochemistry, DNA in situ hybridization, and polymerase chain reaction (PCR) analysis were performed.
Results
In the first patient, histopathology showed the presence of a benign squamous papilloma with koilocytosis. DNA in situ hybridization with broad-spectrum probes showed that this patient was positive for HPV DNA. In the second patient, histopathology showed the presence of a squamous papilloma with mild dysplasia and koilocytosis. Immunohistochemical analysis was positive for HPV protein and p16 protein. DNA in situ hybridization with broad-spectrum probes showed that the patient was positive for HPV DNA. PCR analysis showed the presence of HPV 6. According to morphological and molecular findings, both patients were diagnosed with squamous cell papilloma due to HPV.
Conclusion
HPV can infect the ocular surface. According to clinical results, the ophthalmologist in cooperation with the pathologist can recommend appropriate laboratory examinations to confirm the diagnosis and successfully treat conjunctival papillomas.
Keywords: conjunctiva, papilloma, human papillomavirus, koilocytosis, in situ hybridization, polymerase chain reaction
Introduction
Human papillomaviruses (HPVs) are DNA viruses that have a marked tropism for squamous epithelium. More than 100 different mucosal types of HPV have been described, and HPV is estimated to be the most common sexually transmitted disease. 1 Genital HPV types are classified according to their association with cervical carcinogenesis as low- (LR) and high-risk (HR) types. LR types, such as HPV 6 and HPV 11, which are responsible for >90% of condyloma acuminata (genital warts), can also cause low-grade intraepithelial lesions of the uterine cervix as well as rare recurrent respiratory papillomatosis.2 HPV 6 and HPV 11 are also found in the majority of conjunctival papillomas. HR HPV types are responsible for virtually all cases of cervical cancer (99%). HR HPV 16 and HPV 18 are found in more than 70% of cervical cancers. HR HPV types are also responsible for a significant proportion of carcinomas in other sites, including the vulva, vagina, penis, anus, larynx, and tonsils.3–5 HPV 16 and HPV 18 types have also been found in conjunctival papillomas.
Patients and methods
In this article, we present two cases of conjunctival tumors that were examined and treated in the Ocular Inflammation Department of the University Eye Clinic and diagnosed in the University Pathology Department, University Hospital of Ioannina, Greece. We present the diagnostic and therapeutic approach and review the literature.
The first patient was a 48-year-old man presenting with an extended papillomatous lesion in bulbar conjunctiva covering part of the cornea of his right eye. The second patient was a 24-year-old man presenting with a polypoidal papillomatous lesion on the conjunctival caruncle of his right eye. The lesions were removed surgically and sent for histopathological and immunohistochemical examination and were also analyzed using DNA in situ hybridization and PCR analysis.
Hematoxylin and eosin staining
Formalin fixed, paraffin-embedded sections of each lesion were stained with hematoxylin and eosin and examined. Since in the histopathological examination, the presence of a papillomatous growth pattern along with koilocytosis and mild epithelial dysplastic changes were noted, further laboratory investigation to confirm the clinical diagnosis of HPV infection was performed.
Immunohistochemical staining
Formalin-fixed and paraffin-embedded tissue sections from each case were used for immunohistochemical staining of HPV and p16 protein. HPV antibody (HPV Ab-3, Clone: K1H8, MW: 57 kDa, Epitope: nonconfirmational internal linear epitope of major capsid protein) was purchased from Thermo Scientific (Waltham, MA). This antiserum is reactive with papillomavirus genus-specific (common) structural antigens of the following genotypes: 6, 11, 16, 18, 31, 33, 42, 51, 52, 56, and 58. The Ab was diluted 1:20, and staining of formalin-fixed tissues requires boiling of tissue sections in 10 mM citrate buffer pH 6.0 for 10–20 min followed by cooling at room temperature for 20 min. The CINtec® p16INK4a antibody was purchased from Ventana (Tucson, AZ) and was used without dilution. The p16 INK4a (p16) is a cyclin- dependent kinase inhibitor which has been demonstrated to show marked overexpression in cancerous and precancerous cervical lesions caused by persistent infections with HR HPV types. Immunohistochemical staining was performed using the avidin–biotin complex method.6
In situ hybridization
Paraffin-embedded sections from both cases were placed on silane-coated slides and examined with two broad-spectrum HPV probes, INFORMTM HPV II Family 6 Probe (B) and INFORMTM HPV III Family 16 Probe (B), which were also purchased from Ventana. INFORMTM HPV II Family 6 Probe (B) contains a cocktail of labeled HPV genomic probes. The intended targets include the common HPV genotypes not associated with cervical cancer. The probe cocktail has demonstrated positive hybridization to the low-risk genotypes 6 and 11. INFORMTM HPV II Family 6 Probe (B) is used in conjunction with the ISH iVIEWTM Blue Plus Detection Kit to stain paraffin-embedded tissue on the BenchMarkR Series automated slide stainers. Each HPV Family 6 System Control Slide (B) contains one formalin-fixed, paraffin-embedded condyloma tissue section. HPV Family 6 System Control Slides (B) are designed to allow the user to check ISH staining function on a Ventana automated slide stainer. INFORMTM HPV III Family 16 Probe (B) contains a cocktail of labeled HPV genomic probes. The intended targets include the common genital HPV genotypes found to be associated with cervical cancer. The probe cocktail has been demonstrated to show positive hybridization to the following HR genotypes: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 66.7 INFORMTM HPV III Family 16 Probe (B) is used in conjunction with the ISH iVIEWTM Blue Plus Detection Kit to stain paraffin-embedded tissue on the BenchMarkR Series automated slide stainers. HPV 3-in-1 System Control Slides are used to monitor the performance of automated HPV in situ hybridization staining assays. Established controls, such as the HPV 3-in-1 System Control Slides, are processed and run with all clinical specimens. The CaSki section is a cell line that reportedly contains approximately 200–400 copies of HPV type 16 per cell and the HeLa section is a cell line that reportedly contains approximately 10–50 copies of HPV type 18 per cell. The C-33 A negative section is a cell line, which is an HPV-negative human cervical carcinoma.
PCR
For polymerase chain reaction (PCR) analysis, 5 μm-thick paraffin-embedded sections were placed in a classical 2 mL tube (Array Tube-AT; Alere Technologies GmbH; Jena, Germany) biochip (3 × 3 mm) fixed with a low-density microarray (Clinical Array Technology; Genomica, Madrid, Spain) and examined using the CLART HPV 2 kit purchased from Genomica. This kit detects up to 35 (6, 11, 16, 18, 26, 31, 33, 35, 39, 40, 42, 43, 44, 45, 51, 52, 53, 54, 56, 58, 59, 61, 62, 66, 68, 70, 71, 72, 73, 81, 82, 83, 84, 85, and 89) of the most clinically relevant HPV types. Detection of different HPV genotypes is achieved by PCR amplification of a 450-base pair fragment within the highly conserved L1 region of the virus. This highly conserved sequence presents slight variations among each individual HPV type that allows its genomic identification by recognition of the viral DNA using specific probes.8 This slight variation guarantees detection specificity. CLART HPV 2 allows detection of infections and coinfections of up to 35 different HPV genotypes using the combination of a highly specific and sensitive PCR with a new technological platform based on low-density arrays, such as CLART (Clinical Array Technology; Genomica). The platform is based on a low-density microarray fixed at the bottom of a classical 2-mL tube (Array Tube-AT), thus rendering a very efficient and easy to use system. This technology allows the simultaneous detection of multiple molecular markers for diagnostic use while providing the controls needed to ensure the reliability of the results. Moreover, this system considerably simplifies the processes of hybridization and visualization when compared with classic microarray systems.
Every reaction (amplification) tube of the kit contains the following primers:
A pair of primers that amplifies a fragment of the human gene CFTR (genomic DNA or DNA from the patient). This is used as genomic DNA control.
A pair of primers that amplifies a modified plasmid that is included in the tube and which is used as an amplification reaction control.
HPV primers.
The reaction tube has been designed to favor the amplification of the HPV against the two controls. Among these controls, the genomic DNA will be preferentially amplified compared to the amplification reaction control. Hybridization of the amplified PCR product is detected by generation of an insoluble precipitate at the sites of the microarray where the amplified products have been captured by the probes. This is achieved by marking the amplified products with biotin during the PCR procedure. Biotinylated products hybridize to their specific probes attached to the microarray surface and become immobilized. These immobilized biotinylated products are recognized by the streptavidin of a streptavidin-peroxidase conjugate, thus providing peroxidase activity to the hybridized products. Peroxidase activity will then metabolize γ-dianisidine and produce an insoluble product, which will precipitate in places where hybridization occurred. The sensitivity obtained by combining genomic amplification and this visualization method in the microarrays is sufficiently high such that nested amplifications are not required and the contamination associated with such procedures is therefore avoided.
Results
The clinicopathological data and the results of the immunohistochemical staining, DNA in situ hybridization, and PCR analysis for the two patients are summarized in Table 1.
Table 1.
Clinicopathological data and results of HPV analysis in our patients
| Age, sex | Site | Histology | Koilocytosis | Immunohistochemistry | ISH | PCR |
|---|---|---|---|---|---|---|
| 48-year-old male | Bulbar conj and cornea | Squamous papilloma | + | − | 6/11 | − |
| 24-year-old male | Caruncle | Squamous papilloma with mild dysplasia | + | + | 6/11 | HPV 6 |
Abbreviations: conj, conjunctiva; HPV, human papillomavirus; ISH, in situ hybridization; PCR, polymerase chain reaction.
Case 1
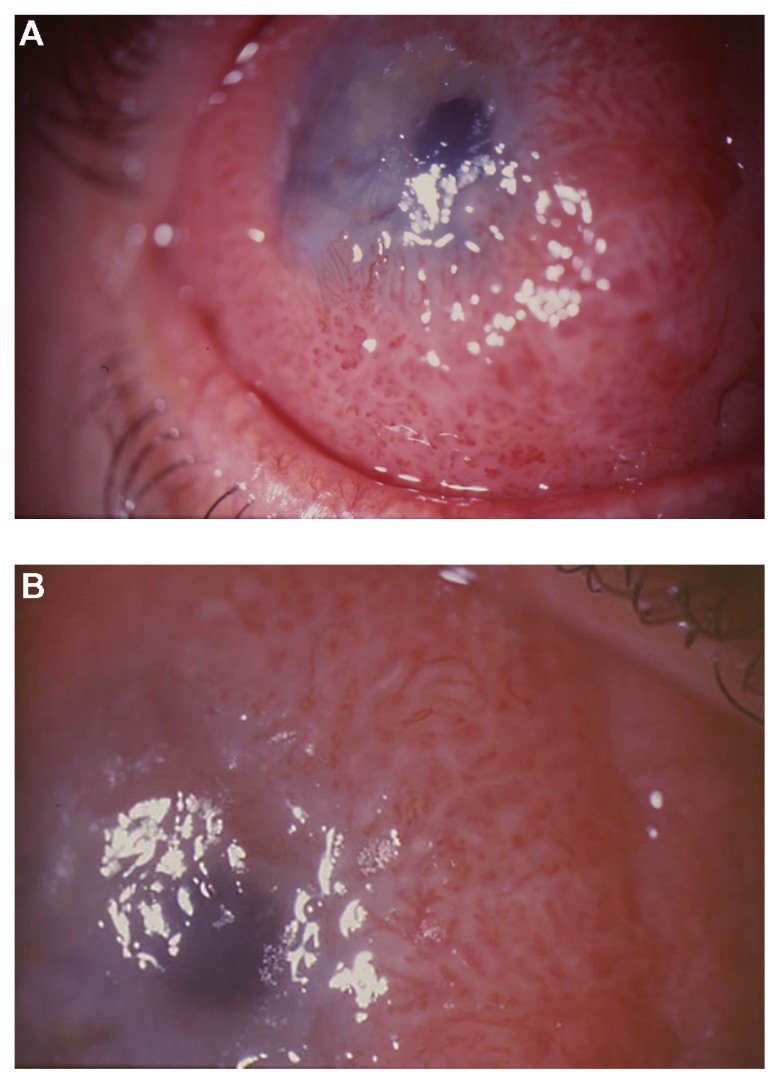
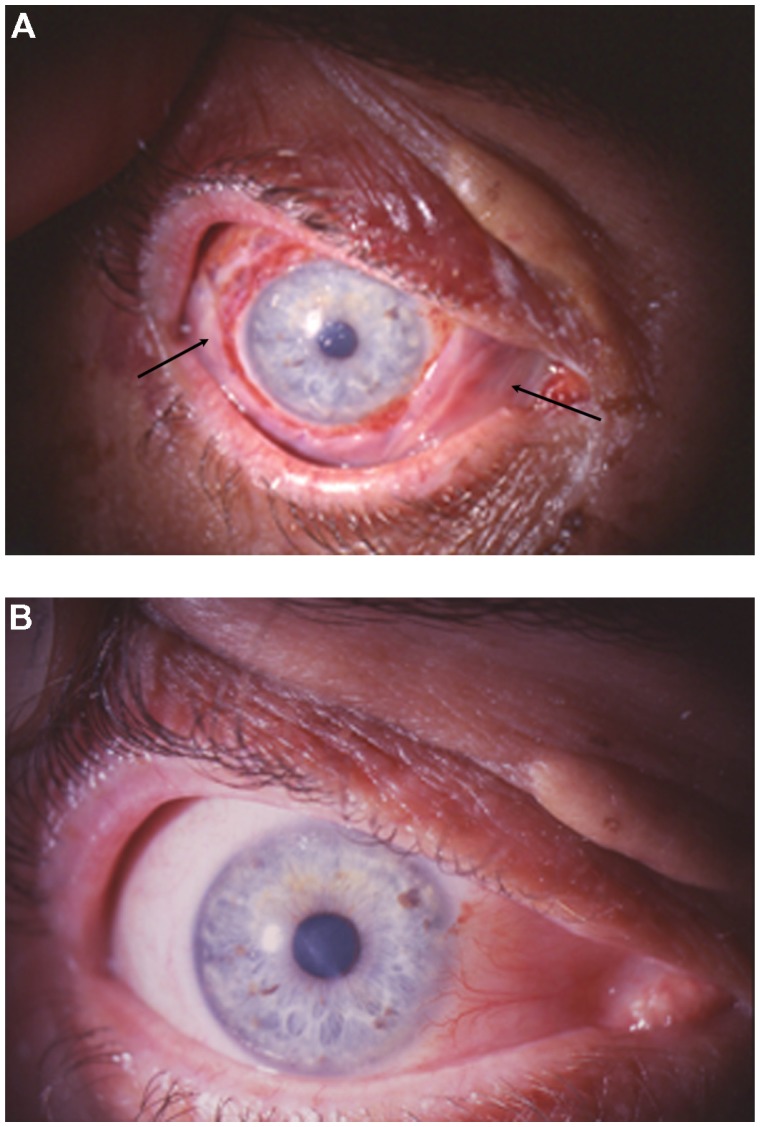
A 48-year-old man with redness in the right eye for 3 years and gradual visual impairment the last year (visual acuity was 0.2 in the right eye) presented a diffuse papillomatous lesion in bulbar conjunctiva covering part of the cornea (Figure 1A and B). The major clinical indication was repeating fibrovascular cores with a geometrically arranged set of red dots. The lesion was removed surgically. After surgical excision, cryotherapy was applied to the adjacent conjunctiva, and amniotic membrane was used to restore the conjunctival defect. Topical mitomycin-C (MMC) was also used intraoperatively on a sponge to prevent recurrences. The concentration of MMC was 0.2 mg/mL and the application time was 2 minutes. The final visual acuity in the right eye was 1.0. No recurrences were observed during a follow-up period of 9 years.
Figure 1.
(A) Diffuse papillomatous lesion in bulbar conjunctiva covering part of the cornea of patient 1 and (B) In the papillomatous lesion of patient 1, repeating fibrovascular cores with geometrically arranged set of red dots are observed.
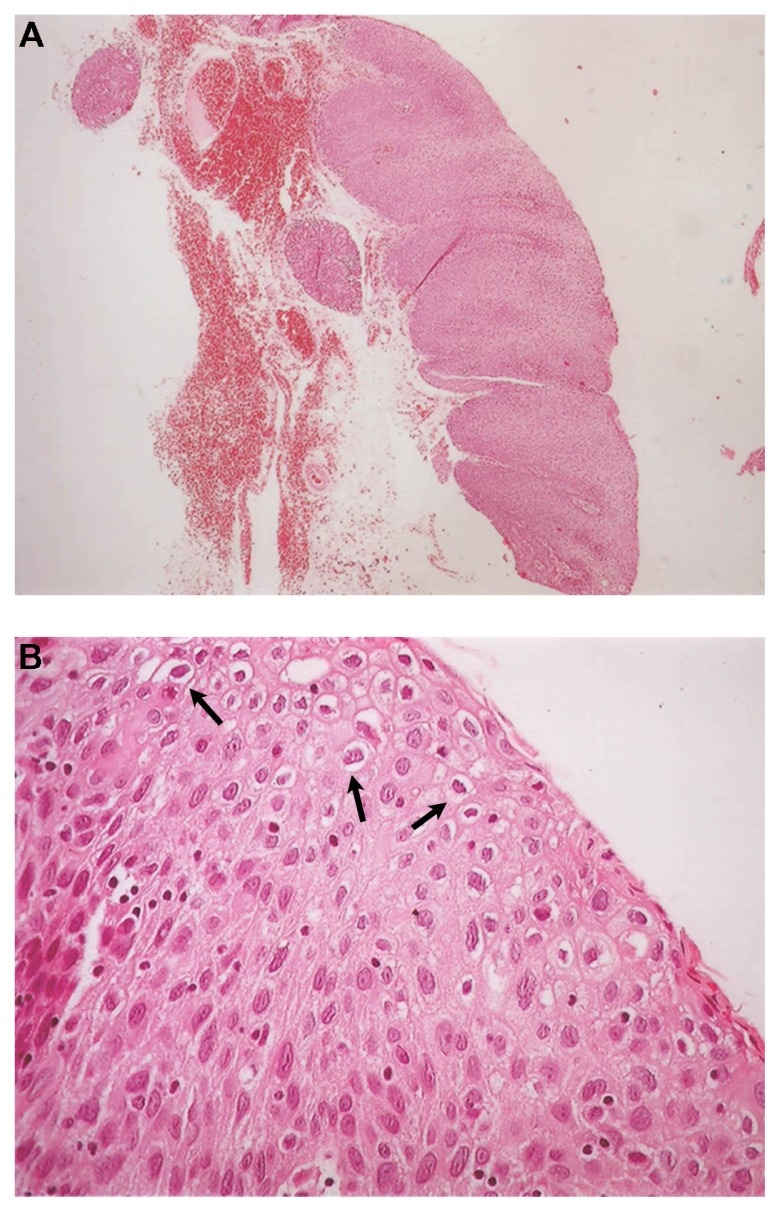
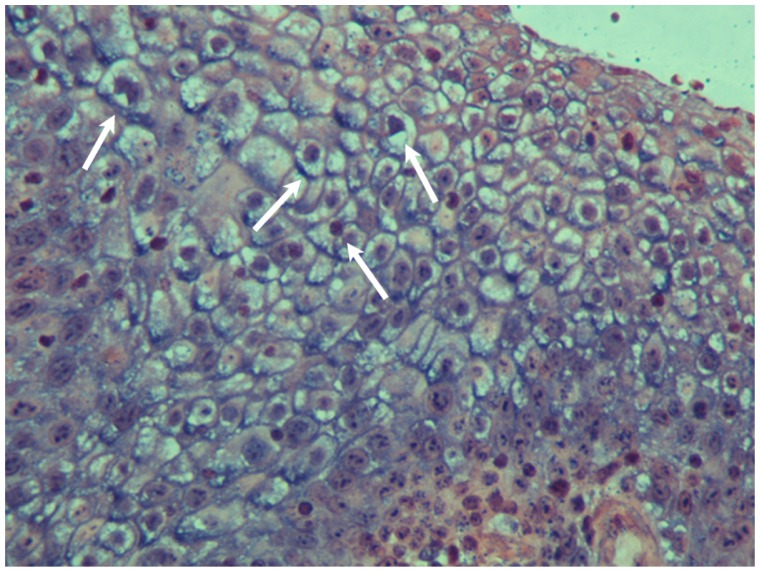
Histopathology showed the presence of a benign squamous cell papilloma with a characteristic papillary growth pattern. Extensive hyperplastic epithelium with papillomatosis, acanthosis, and koilocytosis with mild nuclear atypia were observed (Figure 2A and B). Immunohistochemical analysis for HPV and p16 proteins was negative. Analysis with DNA in situ hybridization was positive for HPV DNA using low risk probe 6/11 (Figure 3). PCR analysis was negative, but it is known that formalin and paraffin wax contain large amounts of PCR inhibitors and that formalin fixation can cause formalin-induced DNA degradation.9
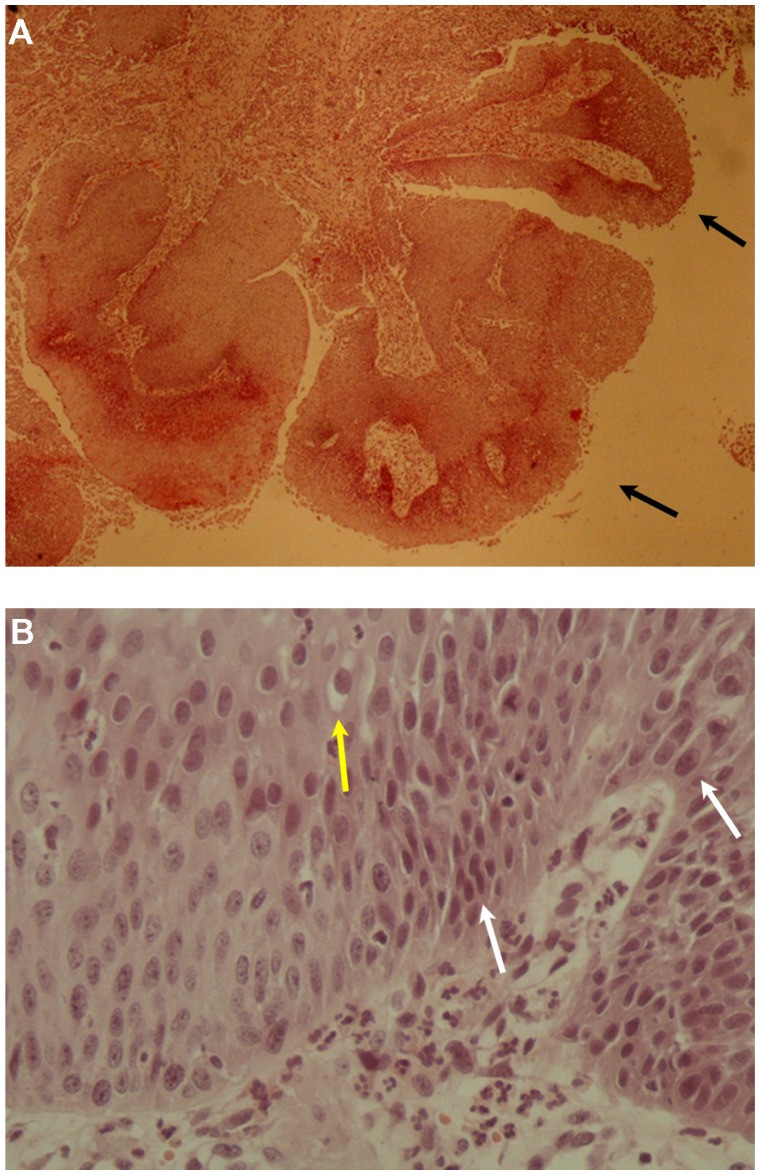
Figure 2.
(A) Benign squamous papilloma of patient 1. The papillary growth pattern is apparent. Extensive hyperplastic epithelium with papillomatosis and acanthosis is observed (hematoxylin and eosin staining, ×40) and (B) Koilocytosis (arrows) with mild nuclear atypia is observed in the benign squamous papilloma of patient 1 (hematoxylin and eosin staining, ×400).
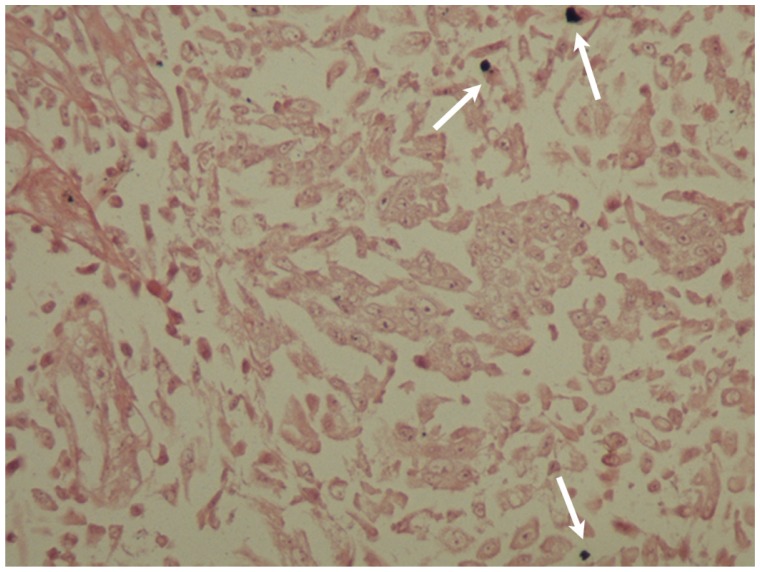
Figure 3.
In situ hybridization of the benign squamous papilloma of patient 1. The nuclei of many epithelial cells are positive (arrows) for HPV DNA with broad spectrum probes (×400).
Abbreviation: HPV, human papillomavirus.
Case 2
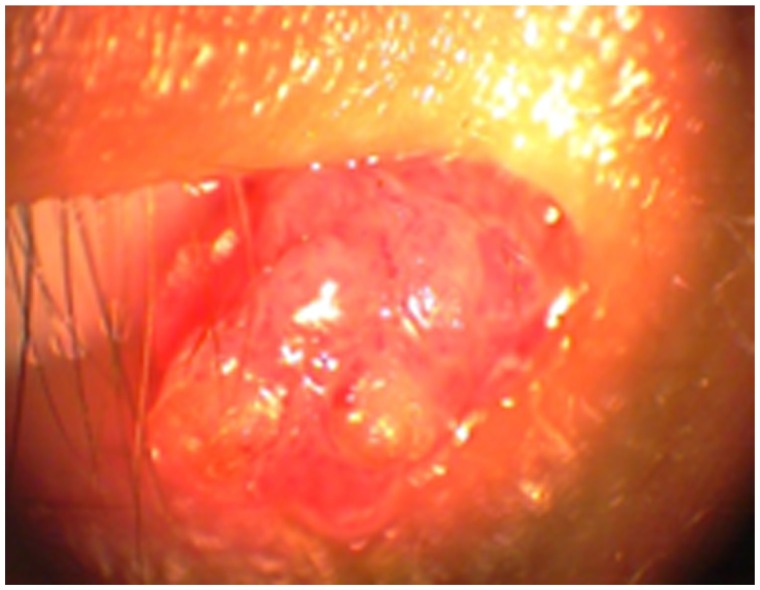
A 24-year-old man presented a polypoidal papillomatous lesion on the conjunctival caruncle of the right eye without any particular symptoms (Figure 4). The lesion was easily removed surgically due to the pedunculated nature of the lesion. After surgical excision, cryotherapy was applied to the adjacent conjunctiva. Topical MMC was also used intraoperatively on a sponge to prevent recurrences. The concentration of MMC was 0.2 mg/mL and the application time was 2 minutes. No recurrences were observed during a follow-up period of 3 years.
Figure 4.
Polypoidal papillomatous lesion on the caruncle of patient 2.
Histopathology showed the presence of a squamous cell papilloma with mild dysplasia. A characteristic papillary growth pattern was noted as well as the presence of koilocytosis (Figure 5A and B). Immunohistochemical analysis was positive for HPV proteins. However, only a few dysplastic epithelial cells showed positive nuclear and cytoplasmic staining for p16 protein (Figure 6A and B). Analysis with DNA in situ hybridization was positive for HPV DNA using LR probe 6/11 (Figure 7). PCR analysis showed the presence of HPV 6.
Figure 5.
(A) Squamous papilloma of patient 2. The papillary growth pattern (arrows) is apparent (hematoxylin and eosin staining, ×40) and (B) Koilocytosis (yellow arrow) and mild dysplasia (white arrows) are apparent in the squamous papilloma of patient 2 (hematoxylin and eosin staining, ×400).
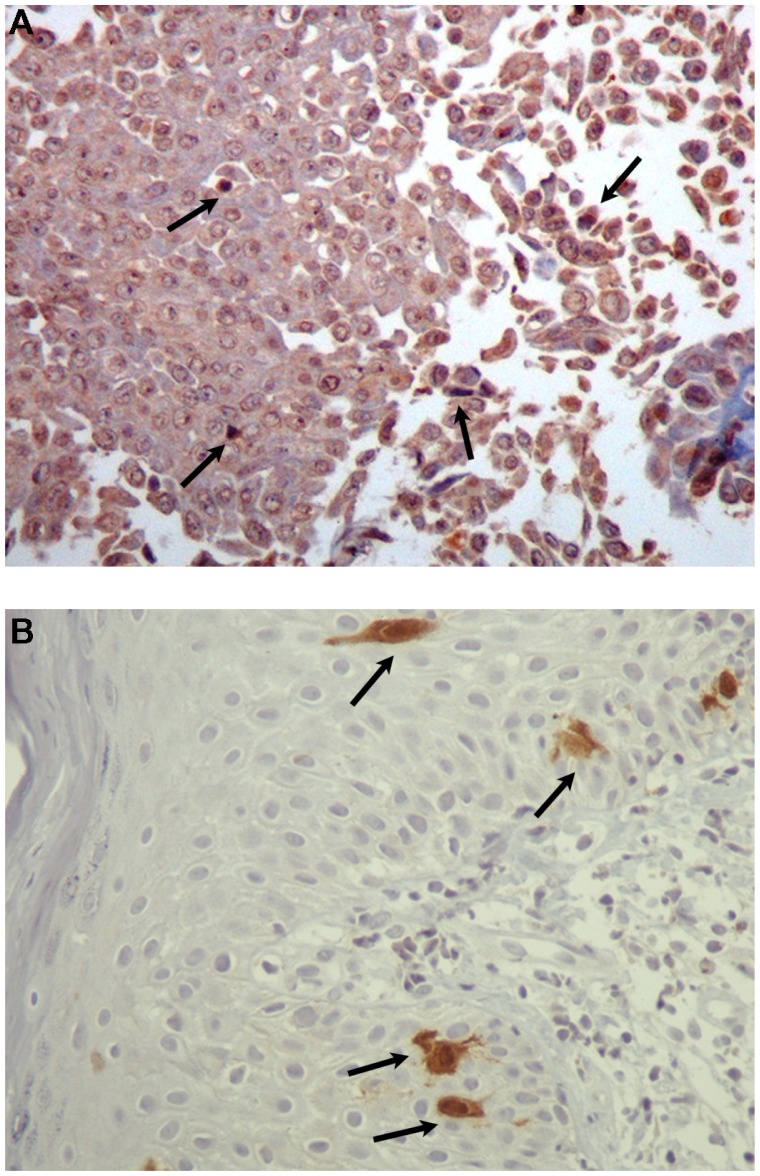
Figure 6.
(A) Immunohistochemical analysis of the squamous papilloma with mild dysplasia of patient 2. The nuclei of some epithelial cells show positive staining (arrows) with antibodies to HPV proteins (×400) and (B) Immunohistochemical analysis of the squamous papilloma with mild dysplasia of patient 2. Only a few dysplastic epithelial cells were positive nuclear and cytoplasmic staining (arrows) for p16 protein (×400).
Abbreviation: HPV, human papillomavirus.
Figure 7.
In situ hybridization of the squamous papilloma with mild dysplasia of patient 2. The nuclei of some epithelial cells are positive (arrows) for HPV DNA with broad spectrum probes (×400).
Abbreviation: HPV, human papillomavirus.
Discussion
Squamous cell papillomas of the conjunctiva are common benign lesions, and HPV is strongly associated with their occurrence.10 Investigations of conjunctival papilloma biopsies for the presence of HPV using PCR analysis have identified HPV DNA in 44%–92% of the lesions.10–15 The HPV types found in conjunctival papillomas are most commonly HPV 6 and HPV 11 (Table 2).
Table 2.
HPV DNA in conjunctival papillomas; comparison of published data
| Study | HPV types | HPV frequency | Cases |
|---|---|---|---|
| Sjö et al15 | 6, 11, 45 | 81% | 165 |
| Eng et al12 | 6/11 | 58% | 24 |
| Sjö et al10 | 6/11, 16 | 92% | 55 |
| Nakamura et al11 | 6 | 44% | 9 |
| Saegusa et al13 | 16 | 75% | 16 |
| McDonnell et al14 | 6 | 65% | 23 |
Abbreviation: HPV, human papillomavirus.
Sjö et al investigated 20 normal conjunctiva biopsy specimens for the presence of HPV and found that all were HPV negative. They concluded that conjunctiva is not an HPV reservoir.10 In contrast, Karcioglu and Issa investigated 19 normal conjunctiva biopsy specimens for the presence of HPV and found HPV 16 and HPV 18 in six patients (32%).16
Conjunctival papillomas typically show an exophytic growth pattern that can be sessile or pedunculated and repeating fibrovascular cores with a geometrically arranged set of red dots are typical findings in the slit lamp examination. Papillomas may also have a mixed or, rarely, an inverted growth pattern.17 Most papillomas are located medially and inferiorly in the conjunctiva. The localization of conjunctival papillomas according to the study of Sjö et al was on the palpebrae 38%, fornices 11%, bulbus 24%, limbus 0%, and caruncle 27%.17 Dysplasia may be present in the lesions, but carcinoma rarely develops in conjunctival papillomas.17
The differential diagnosis of conjunctival papillomas includes a variety of tumors: (1) benign tumors of surface epithelium as keratoacanthoma, epithelial inclusion cyst, benign hyperplasia of epithelium, and actinic keratosis, (2) malignant lesions of surface epithelium as conjunctival intraepithelial neoplasia and squamous cell carcinoma, (3) vascular tumors as pyogenic granuloma, lymphangioma and capillary hemangioma, (4) conjunctival lymphomas, (5) nonpigmented conjunctival melanomas, and (6) secondary tumors. The medical history, slit-lamp examination, and the specific clinical and histopathological features of each tumor are used for accurate diagnosis.
Sjö et al reviewed the files of 219 patients with 245 conjunctival papillomas. They found that conjunctival papillomas are more frequent in men 60.3% (95% confidence interval [CI]: 53.7%–66.9%). The highest incidence of conjunctival papillomas was found among patients aged 20–29 years.17 This is in accordance with the study of Dunne et al who found the highest prevalence of cervicovaginal HPV infection among women aged 20–24 years18 and in accordance also with the study of Insinga et al who found the highest prevalence of genital warts in women aged 20–24 years and in men aged 25–29 years.19
The mode of transmission of HPV infection of the conjunctiva in adults is considered to be autoinoculation from contaminated fingers to the conjunctiva. Sonnex et al have documented hand carriage of HPV types in patients with genital warts.20 Conjunctival papillomas have also been reported in infants. The transmission is considered to happen during birth from the affected genital tract of women to the conjunctiva of the newborn.21
Koilocytosis (nuclear pyknosis and cytoplasmic clearing) is a morphological hallmark of HPV infection. Koilocytosis was present in both of our cases.
Most commonly, LR HPV 6 and HPV 11 are found in most conjunctival papillomas. The same types are responsible for more than 90% of condyloma acuminata (genital warts). HR HPV 16 and HPV 18 have also been found in conjunctival papillomas. These types are strongly associated with the occurrence of high-grade uterine cervical intra-epithelial neoplasia and cervical cancer. HPV 16 and HPV 18 are responsible for more than 70% of cervical cancers followed by other HR HPV types. Other types found in conjunctival papillomas in case reports are HPV 33, HPV 45, and HPV 13.15,22,23
Conjunctival papillomas often show spontaneous regression and cure. It must also be kept in mind that recurrences are frequent (6%–27%).14,17,24 There is a variety of treatment choices for treating large and symptomatic conjunctival papillomas that may cause also cosmetic defects. Surgical excision of the lesion and cryotherapy to the adjacent conjunctiva is considered to be the treatment of choice.25 The amniotic membrane can be used to cover large epithelial defects after the excision of the affected conjunctiva. In addition, topical MMC and topical interferon alpha 2b have been used to prevent recurrences and also to treat recurrent papillomas.26,27 Another agent that has been used for the treatment of conjunctival papillomas is oral cimetidine.28
In our two patients, the treatment of the conjunctival papillomas included surgical excision of the lesion and cryotherapy to the adjacent conjunctiva. Topical MMC was used to prevent recurrences. In the patient with the diffuse papillomatous lesion in bulbar conjunctiva, after the excision, the extended epithelial defect was covered with amniotic membrane with an excellent outcome (Figure 8A and B). Recurrences were not observed in either patient during the long follow-up period.
Figure 8.
(A) Patient 1: 4th postoperative day with the amniotic membrane (arrows) covering the defect of the excised pathological conjunctiva and (B) 6 months after surgery; excellent ocular surface without signs of tumor recurrence.
The oncogenic properties of HPVs are attributed mainly to two viral oncoproteins, E6 and E7. These oncoproteins are necessary for the induction and maintenance of the transformed phenotype. The E6 oncoprotein of the HR HPV types alters numerous cellular pathways, including inactivating p53, blocking apoptosis, activating telomerase, disrupting cell adhesion, polarity, and epithelial differentiation, altering transcription, and reducing immune recognition.29 The E7 oncoprotein of the HR HPV types also alters numerous cellular pathways, including activation of E2F-dependent DNA transcription through degradation of the retinoblastoma oncosuppressor protein (pRB), dysregulation of the G1/S checkpoint of the cell cycle, dysregulation of the epigenetic reprogramming, causing subversion of p53 function, blocking apoptosis, modulating the cytostatic signaling (TGF-β resistance, TNF-α resistance, compromission of IFN signaling, interference with IGF signaling), altering the cellular metabolism, and inducing chromosomal instability.30 In contrast, the E6 and E7 oncoproteins of the LR HPV types have greatly decreased transforming and immortalizing activities in comparison to HR E6 and E7 oncoproteins.30,31
HR HPV types can cause cervical, anal, and other genital cancers. The association between HPV and squamous cell carcinoma of the conjunctiva has been examined. Squamous cell carcinoma of the conjunctiva (SCCC) is a rare tumor that has been strongly associated with UV radiation and immunosuppression (particularly in HIV patients).32,33 The role of HPV infection in the etiology of SCCC remains unclear. HPV16 or HPV 18 DNA and mRNA corresponding to the E6 region have been detected in conjunctival intraepithelial neoplasia.34 Conjunctival intraepithelial neoplasia is a precursor of SCCC. A few other studies have identified the presence of HPV 16 and HPV 18 in severe dysplastic lesions and carcinomas of the conjunctiva.35 HPV 6 and HPV 11 have also been found in severe dysplasias and carcinomas of the conjunctiva.36 In addition, cutaneous HPV types, most commonly HPV 5 and HPV 8, have been found in SCCC, especially in HIV positive patients.37 In contrast, multiple other studies have failed to find a strong association of HPV with SCCC.12,38 According to the 2007 International Agency for Research on Cancer on the carcinogenicity of HPV in humans, limited evidence was available for the carcinogenicity of HPV in the conjunctiva.39
The development of an HPV vaccine and immunization against HPV is expected to have a strong impact on the incidence of HPV-related diseases. There are two presently available prophylactic vaccines (quadrivalent and bivalent). They were based on the production of L1 recombinant proteins of HPV, produced in the form of pentamers, that form virus-like particles, which are “empty shells” that do not contain infectious HPV DNA.40,41 The quadrivalent vaccine includes virus-like particles of the HPV types 16, 18, 6, and 11. The bivalent vaccine includes virus-like particles of the HPV types 16 and 18. The two vaccines are most efficient if used at preadolescent age, before the beginning of sexual activity. HR HPV types 16 and 18 are responsible for more than 70% of the cases of cervical cancer. LR HPV types 6 and 11 are responsible for more than 90% of genital warts. The HPV types 6 and 11 are found in the majority of conjunctival papillomas. Muñoz et al investigated the impact of the quadrivalent prophylactic vaccine against HPV types 6, 11, 16, and 18 on all HPV-associated genital disease in a population of sexually naïve women (HPV-unexposed). They concluded that prophylactic vaccination was 95%–100% effective in reducing HPV 16/18-related high-grade cervical, vulvar, and vaginal lesions and 97% effective in reducing HPV 6/11 related genital warts.42 Thus, for ophthalmology, the quadrivalent vaccine is expected to decrease the incidence of conjunctival papillomas due to HPV infection. In contrast, the HPV vaccination is not expected to prevent SCCC because HPV is not the main oncogenic agent in SCCC. Ultraviolet radiation and immunosuppression (particularly in HIV patients) appear to play a much greater role in the etiology of SCCC. Finally, it should be mentioned that the HPV vaccine can offer a certain degree of cross-protection against other HPV types that are genetically and antigenically closely related to vaccine types. For instance, HPV 31 and HPV 35 are strongly related to HPV 16 and HPV 45 is strongly related to HPV 18.
In conclusion, HPV infection is considered the most common sexually transmitted disease and can also infect the ocular surface. Squamous cell papillomas of the conjunctiva are common benign epithelial tumors, and HPV is strongly associated with their occurrence. Based on the clinical picture, the ophthalmologist, in cooperation with the pathologist, can recommend the appropriate laboratory examinations to confirm the diagnosis and treat the conjunctival papillomas to achieve the best outcome and prevent recurrences.
Footnotes
Disclosure
The authors report no conflicts of interest in this work
References
- 1.Weinstock H, Berman S, Cates W., Jr Sexually transmitted diseases among American youth: incidence and prevalence estimates, 2000. Perspect Sex Reprod Health. 2004;36(1):6–10. doi: 10.1363/psrh.36.6.04. [DOI] [PubMed] [Google Scholar]
- 2.Garland SM, Steben M, Sings HL, et al. Natural history of genital warts: analysis of the placebo arm of 2 randomized phase III trials of a quadrivalent human papillomavirus (types 6, 11, 16, and 18) vaccine. J Infect Dis. 2009;199(6):805–814. doi: 10.1086/597071. [DOI] [PubMed] [Google Scholar]
- 3.Walboomers JM, Jacobs MV, Manos MM, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol. 1999;189(1):12–19. doi: 10.1002/(SICI)1096-9896(199909)189:1<12::AID-PATH431>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 4.Bosch FX, de Sanjosé S. Chapter 1: Human papillomavirus and cervical cancer – burden and assessment of causality. J Natl Cancer Inst Monogr. 2003;31:3–13. doi: 10.1093/oxfordjournals.jncimonographs.a003479. [DOI] [PubMed] [Google Scholar]
- 5.Clifford GM, Smith JS, Plummer M, et al. Human papillomavirus types in invasive cervical cancer worldwide: a meta-analysis. Br J Cancer. 2003;88(1):63–73. doi: 10.1038/sj.bjc.6600688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hsu SM, Raine L, Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem. 1981;29(4):577–580. doi: 10.1177/29.4.6166661. [DOI] [PubMed] [Google Scholar]
- 7.Nakamura Y, Mashima Y, Kameyama K. Human papilloma virus DNA detected in case of inverted squamous papilloma of the lacrimal sac. Br J Ophthalmol. 1995;79(4):392–393. doi: 10.1136/bjo.79.4.392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Coates PJ, d’Ardenne AJ, Khan G, Kangro HO, Slavin G. Simplified procedures for applying the polymerase chain reaction to routinely fixed paraffin wax sections. J Clin Pathol. 1991;44(2):115–118. doi: 10.1136/jcp.44.2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Legrand B, Mazancourt P, Durigon, Khalifat V, Crainic K. DNA genotyping of unbuffered formalin fixed paraffin embedded tissues. Forensic Sci Int. 2002;125(2–3):205–211. doi: 10.1016/s0379-0738(01)00641-7. [DOI] [PubMed] [Google Scholar]
- 10.Sjö NC, Heegaard S, Prause JU, von Buchwald C, Lindeberg H. Human papillomavirus in conjunctival papilloma. Br J Ophthalmol. 2001;85(7):785–787. doi: 10.1136/bjo.85.7.785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nakamura Y, Mashima Y, Kameyama K, Mukai M, Oguchi Y. Detection of human papillomavirus infection in squamous tumours of the conjunctiva and lacrimal sac by immunohistochemistry, in situ hybridisation, and polymerase chain reaction. Br J Ophthalmol. 1997;81(4):308–313. doi: 10.1136/bjo.81.4.308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eng HL, Lin TM, Chen SY, Wu SM, Chen WJ. Failure to detect human papillomavirus DNA in malignant epithelial neoplasms of conjunctiva by polymerase chain reaction. Am J Clin Pathol. 2002;117(3):429–436. doi: 10.1309/RVUP-QMU3-5X6W-3CQ1. [DOI] [PubMed] [Google Scholar]
- 13.Saegusa M, Takano Y, Hashimura M, Okayasu I, Shiga J. HPV type 16 in conjunctival and junctional papilloma, dysplasia, and squamous cell carcinoma. J Clin Pathol. 1995;48(12):1106–1110. doi: 10.1136/jcp.48.12.1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McDonnell PJ, McDonnell JM, Kessis T, Green WR, Shah KV. Detection of human papillomavirus type 6/11 DNA in conjunctival papillomas by in situ hybridization with radioactive probes. Hum Pathol. 1987;18(11):1115–1119. doi: 10.1016/s0046-8177(87)80378-7. [DOI] [PubMed] [Google Scholar]
- 15.Sjö NC, von Buchwald C, Cassonnet P, et al. Human papillomavirus in normal conjunctival tissue and in conjunctival papilloma: types and frequencies in a large series. Br J Ophthalmol. 2007;91(8):1014–1015. doi: 10.1136/bjo.2006.108811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Karcioglu ZA, Issa TM. Human papilloma virus in neoplastic and non-neoplastic conditions of the external eye. Br J Ophthalmol. 1997;81(7):595–598. doi: 10.1136/bjo.81.7.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sjö N, Heegaard S, Prause JU. Conjunctival papilloma. A histopathologically based retrospective study. Acta Ophthalmol Scand. 2000;78(6):663–666. doi: 10.1034/j.1600-0420.2000.078006663.x. [DOI] [PubMed] [Google Scholar]
- 18.Dunne EF, Unger ER, Sternberg M, et al. Prevalence of HPV infection among females in the United States. JAMA. 2007;297(8):813–819. doi: 10.1001/jama.297.8.813. [DOI] [PubMed] [Google Scholar]
- 19.Insinga RP, Dasbach EJ, Myers ER. The health and economic burden of genital warts in a set of private health plans in the United States. Clin Infect Dis. 2003;36(11):1397–1403. doi: 10.1086/375074. [DOI] [PubMed] [Google Scholar]
- 20.Sonnex C, Strauss S, Gray JJ. Detection of human papillomavirus DNA on the fingers of patients with genital warts. Sex Transm Infect. 1999;75(5):317–319. doi: 10.1136/sti.75.5.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Egbert JE, Kersten RC. Female genital tract papillomavirus in conjunctival papillomas of infancy. Am J Ophthalmol. 1997;123(4):551–552. doi: 10.1016/s0002-9394(14)70184-9. [DOI] [PubMed] [Google Scholar]
- 22.Buggage RR, Smith JA, Shen D, Chan CC. Conjunctival papillomas caused by human papillomavirus type 33. Arch Ophthalmol. 2002;120(2):202–204. doi: 10.1001/archopht.120.2.202. [DOI] [PubMed] [Google Scholar]
- 23.Benevides dos Santos PJ, Borborema dos Santos CM, Rufino Mendonça R, Vieira do Carmo MA, Astofi-Filho S. Human papillomavirus type 13 infecting the conjunctiva. Diagn Microbiol Infect Dis. 2005;53(1):71–73. doi: 10.1016/j.diagmicrobio.2005.03.009. [DOI] [PubMed] [Google Scholar]
- 24.Ash JE. Epibulbar tumors. Am J Ophthalmol. 1950;33(8):1203–1219. doi: 10.1016/0002-9394(50)90990-1. [DOI] [PubMed] [Google Scholar]
- 25.Shields CL, Shields JA. Tumors of the conjunctiva and cornea. Surv Ophthalmol. 2004;49(1):3–24. doi: 10.1016/j.survophthal.2003.10.008. [DOI] [PubMed] [Google Scholar]
- 26.de Keizer RJ, de Wolff-Rouendaal D. Topical alpha-interferon in recurrent conjunctival papilloma. Acta Ophthalmol Scand. 2003;81(2):193–196. doi: 10.1034/j.1600-0420.2003.00043.x. [DOI] [PubMed] [Google Scholar]
- 27.Yuen HK, Yeung EF, Chan NR, Chi SC, Lam DS. The use of postoperative topical mitomycin C in the treatment of recurrent conjunctival papilloma. Cornea. 2002;21(8):838–839. doi: 10.1097/00003226-200211000-00023. [DOI] [PubMed] [Google Scholar]
- 28.Shields CL, Lally MR, Singh AD, Shields JA, Nowinski T. Oral cimetidine (Tagamet) for recalcitrant, diffuse conjunctival papillomatosis. Am J Ophthalmol. 1999;128(3):362–364. doi: 10.1016/s0002-9394(99)00265-2. [DOI] [PubMed] [Google Scholar]
- 29.Howie HL, Katzenellenbogen RA, Galloway DA. Papillomavirus E6 proteins. Virology. 2009;384(2):324–334. doi: 10.1016/j.virol.2008.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McLaughlin-Drubin ME, Münger K. The human papillomavirus E7 oncoprotein. Virology. 2009;384(2):335–344. doi: 10.1016/j.virol.2008.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Halbert CL, Demers GW, Galloway DA. The E6 and E7 genes of human papillomavirus type 6 have weak immortalizing activity in human epithelial cells. J Virol. 1992;66(4):2125–2134. doi: 10.1128/jvi.66.4.2125-2134.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Newton R, Ziegler J, Ateenyi-Agaba C, et al. The epidemiology of conjunctival squamous cell carcinoma in Uganda. Br J Cancer. 2002;87(3):301–308. doi: 10.1038/sj.bjc.6600451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ateenyi-Agaba C. Conjunctival squamous-cell carcinoma associated with HIV infection in Kampala, Uganda. Lancet. 1995;345(8951):695–696. doi: 10.1016/s0140-6736(95)90870-6. [DOI] [PubMed] [Google Scholar]
- 34.Scott IU, Karp CL, Nuovo GJ. Human papillomavirus 16 and 18 expression in conjunctival intraepithelial neoplasia. Ophthalmology. 2002;109(3):542–547. doi: 10.1016/s0161-6420(01)00991-5. [DOI] [PubMed] [Google Scholar]
- 35.McDonnell JM, McDonnell PJ, Sun YY. Human papillomavirus DNA in tissues and ocular surface swabs of patients with conjunctival epithelial neoplasia. Invest Ophthalmol Vis Sci. 1992;33(1):184–189. [PubMed] [Google Scholar]
- 36.Tabrizi SN, McCurrach FE, Drewe RH, Borg AJ, Garland SM, Taylor HR. Human papillomavirus in corneal and conjunctival carcinoma. Aust N Z J Ophthalmol. 1997;25(3):211–215. doi: 10.1111/j.1442-9071.1997.tb01394.x. [DOI] [PubMed] [Google Scholar]
- 37.Ateenyi-Agaba C, Franceschi S, Wabwire-Mangen F, et al. Human papillomavirus infection and squamous cell carcinoma of the conjunctiva. Br J Cancer. 2010;102(2):262–267. doi: 10.1038/sj.bjc.6605466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tuppurainen K, Raninen A, Kosunen O, et al. Squamous cell carcinoma of the conjunctiva. Failure to demonstrate HPV DNA by in situ hybridization and polymerase chain reaction. Acta Ophthalmol (Copenh) 1992;70(2):248–254. doi: 10.1111/j.1755-3768.1992.tb04132.x. [DOI] [PubMed] [Google Scholar]
- 39.IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol. 90. Lyon, France: World Health Organization International Agency for Research on Cancer; 2007. Human papillomaviruses. [Google Scholar]
- 40.Bonanni P, Boccalini S, Bechini A. Efficacy, duration of immunity and cross protection after HPV vaccination: a review of the evidence. Vaccine. 2009;27(Suppl 1):A46–A53. doi: 10.1016/j.vaccine.2008.10.085. [DOI] [PubMed] [Google Scholar]
- 41.Kyrgiou M, Shafi M. HPV vaccine. Obstet Gynecol Reprod Med. 2009;19(1):26–28. [Google Scholar]
- 42.Muñoz N, Kjaer SK, Sigurdsson K, et al. Impact of human papilloma-virus (HPV)-6/11/16/18 vaccine on all HPV-associated genital diseases in young women. J Natl Cancer Inst. 2010;102(5):325–339. doi: 10.1093/jnci/djp534. [DOI] [PubMed] [Google Scholar]