Abstract
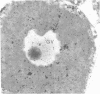
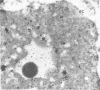
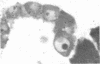
The sperm receptors of the ascidian oocyte are located at the outer surface of the vitelline coat (formerly called the chorion). The fucose residues are the receptor's most important components for sperm recognition and binding. We asked whether the fucosyl-containing glycoproteins of the vitelline coat are a product of the oocyte, the follicle cells, or the test cells. Ovaries of Ciona intestinalis were injected with L-[3H]fucose and the progress of its incorporation was followed by using autoradiography and sodium dodecyl sulfate/polyacrylamide gel electrophoresis of the injected gonads and of the isolated vitelline coats. We found that incorporation of fucose begins within the vitellogenic oocytes, and fucose slowly accumulates in the differentiating vitelline coat. At no time could fucose incorporation be detected in the follicle cells or in the test cells. Sodium dodecyl sulfate/polyacrylamide gel electrophoresis of vitelline coats prepared from the injected ovaries showed fucose incorporation into the same three glycoproteins present in vitelline coats from mature oocytes and identified by their affinity for 125I-labeled fucose-binding protein [Pinto, M. R., De Santis, R., D'Alessio, G. & Rosati, F. (1981) Exp. Cell Res. 132, 289-295]. A radioactive band not found in the mature oocyte was also present.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aketa K., Tsuzuki H., Onitake K. Characterization of the sperm-binding protein from sea urchin egg surface. Exp Cell Res. 1968 Jun;50(3):676–679. doi: 10.1016/0014-4827(68)90436-9. [DOI] [PubMed] [Google Scholar]
- Bleil J. D., Wassarman P. M. Mammalian sperm-egg interaction: identification of a glycoprotein in mouse egg zonae pellucidae possessing receptor activity for sperm. Cell. 1980 Jul;20(3):873–882. doi: 10.1016/0092-8674(80)90334-7. [DOI] [PubMed] [Google Scholar]
- Bleil J. D., Wassarman P. M. Structure and function of the zona pellucida: identification and characterization of the proteins of the mouse oocyte's zona pellucida. Dev Biol. 1980 Apr;76(1):185–202. doi: 10.1016/0012-1606(80)90371-1. [DOI] [PubMed] [Google Scholar]
- Bleil J. D., Wassarman P. M. Synthesis of zona pellucida proteins by denuded and follicle-enclosed mouse oocytes during culture in vitro. Proc Natl Acad Sci U S A. 1980 Feb;77(2):1029–1033. doi: 10.1073/pnas.77.2.1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- De Santis R., Jamunno G., Rosati F. A study of the chorion and the follicle cells in relation to the sperm-egg interaction in the ascidian, Ciona intestinalis. Dev Biol. 1980 Feb;74(2):490–499. doi: 10.1016/0012-1606(80)90449-2. [DOI] [PubMed] [Google Scholar]
- Glabe C. G., Vacquier V. D. Isolation and characterization of the vitelline layer of sea urchin eggs. J Cell Biol. 1977 Nov;75(2 Pt 1):410–421. doi: 10.1083/jcb.75.2.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartmann J. F., Gwatkin R. B., Hutchison C. F. Early contact interactions between mammalian gametes in vitro: evidence that the vitellus influences adherence between sperm and zona pellucida. Proc Natl Acad Sci U S A. 1972 Oct;69(10):2767–2769. doi: 10.1073/pnas.69.10.2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hylander B. L., Summers R. G. An ultrastructural analysis of the gametes and early fertilization in two bivalve molluscs, Chama macerophylla and Spisula solidissima with special reference to gamete binding. Cell Tissue Res. 1977 Sep 5;182(4):469–489. doi: 10.1007/BF00219830. [DOI] [PubMed] [Google Scholar]
- Moy G. W., Vacquier V. D. Immunoperoxidase localization of bindin during the adhesion of sperm to sea urchin eggs. Curr Top Dev Biol. 1979;13(Pt 1):31–44. doi: 10.1016/s0070-2153(08)60688-2. [DOI] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Pinto M. R., De Santis R., D'Alessio G., Rosati F. Studies on fertilization in the ascidians. Fucosyl sites on vitelline coat of Ciona intestinalis. Exp Cell Res. 1981 Apr;132(2):289–295. doi: 10.1016/0014-4827(81)90105-1. [DOI] [PubMed] [Google Scholar]
- Rosati F., De Santis R., Monroy A. Studies on fertilization in the ascidians. II. Lectin binding to the gametes of Ciona intestinalis. Exp Cell Res. 1978 Oct 15;116(2):419–427. doi: 10.1016/0014-4827(78)90465-2. [DOI] [PubMed] [Google Scholar]
- Rosati F., De Santis R. Role of the surface carbohydrates in sperm-egg interaction in Ciona intestinalis. Nature. 1980 Feb 21;283(5749):762–764. doi: 10.1038/283762a0. [DOI] [PubMed] [Google Scholar]
- Rosati F., Monroy A., De Prisco P. Fine structural study of fertilization in the ascidian, Ciona intestinalis. J Ultrastruct Res. 1977 Mar;58(3):261–270. doi: 10.1016/s0022-5320(77)90018-1. [DOI] [PubMed] [Google Scholar]
- Rosati F., de Santis R. Studies on fertilization in the Ascidans. I. Self-sterility and specific recognition between gametes of Ciona intestinalis. Exp Cell Res. 1978 Mar 1;112(1):111–119. doi: 10.1016/0014-4827(78)90531-1. [DOI] [PubMed] [Google Scholar]
- Summers R. G., Hylander B. L. Primary gamete binding. Quantitative determination of its specificity in echinoid fertilization. Exp Cell Res. 1976 Jun;100(1):190–194. doi: 10.1016/0014-4827(76)90341-4. [DOI] [PubMed] [Google Scholar]