Abstract
We describe the performance, in one surgical session, of bilateral pulmonary endarterectomy and a button-technique Bentall operation in a 68-year-old man. The patient had chronic thromboembolic pulmonary hypertension and an ascending aortic aneurysm with moderate aortic regurgitation. The procedures were concurrently completed during short periods of systemic circulatory arrest, with antegrade cerebral perfusion maintained through the brachiocephalic artery at a flow rate of 10 mL/min/kg. The patient's cerebral perfusion was monitored with use of near-infrared spectroscopy, to prevent symmetric bilateral values from falling below 20% of the base value. The patient experienced no multiorgan failure or neurologic sequelae and, by the 6th postoperative day, improved from New York Heart Association functional class IV to class I.
The reliable maintenance of continuous antegrade cerebral perfusion made the lengthy combined operation feasible, with low risk. The use of near-infrared spectroscopy enabled real-time monitoring of the patient's cerebral blood flow. Our experience shows the possibility of safely performing lengthy or multiple procedures in one surgical session.
Key words: Brain ischemia/prevention & control; cardiac surgical procedures; endarterectomy/adverse effects/methods; hypertension, pulmonary/physiopathology/surgery; perfusion/methods; pulmonary artery/surgery; recovery of function; treatment outcome
Pulmonary endarterectomy requires moderate to deep hypothermia associated with several short periods of total circulatory arrest to prevent postoperative neurologic and multiorgan dysfunction.1–4 When antegrade cerebral perfusion is ensured,5,6 it is possible to perform such lengthy interventions in one surgical session rather than in multiple sessions, as is usual. We describe the case of a man who underwent bilateral pulmonary endarterectomy and a Bentall procedure concurrently.
Case Report
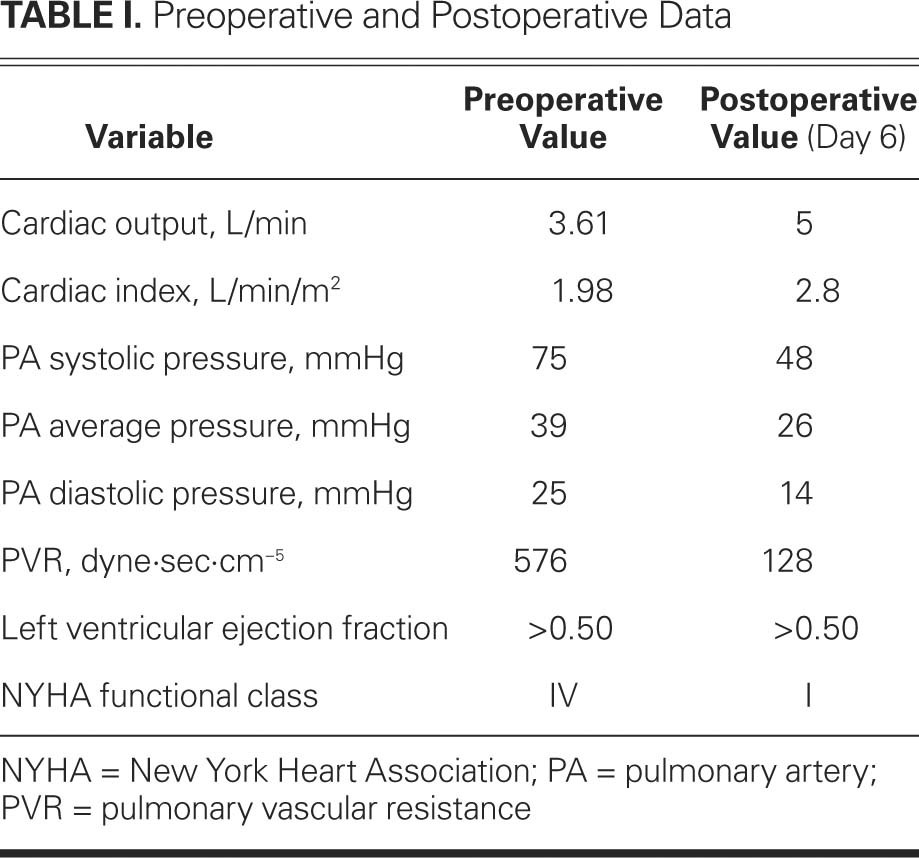
In July 2005, a 68-year-old man was diagnosed with chronic thromboembolic pulmonary hypertension, moderate aortic valve regurgitation, and an ascending aortic aneurysm. His medical therapy consisted of an angiotensin-converting enzyme inhibitor and warfarin. Preoperative findings (Table I) included pulmonary hypertension: pulmonary vascular resistance was 576 dyne·sec·cm−5 with an average pressure of 39 mmHg. Pulmonary angiography showed type 2 disease of the pulmonary arteries (PAs),7,8 with lack of filling due to organized thrombus and intimal thickening mainly in the lower-lobe branches on both sides and in the middle lobe of the right lung. Computed tomography showed dilation of the ascending aorta (5.5 cm), and echocardiography showed moderate aortic valve regurgitation.
TABLE I. Preoperative and Postoperative Data
Upon hospitalization, the patient was in New York Heart Association (NYHA) functional class IV. He was an active smoker, was taking medication for hypertension, and had chronic peripheral obliterative arteriopathy.
Median sternotomy was performed. The right axillary artery was cannulated with interposition of a Hemashield® prosthesis (Boston Scientific Corporation; Natick, Mass) for cardiopulmonary bypass (CPB). Venous drainage was established through bicaval cannulation. The patient's core body temperature was cooled to 26 °C, and the myocardium was protected with ice cooling and the infusion of one dose of Custodiol® Histidine-Tryptophan-Ketoglutarate-HTK cold cardioplegic solution (Essential Pharmaceuticals, LLC; Newtown, Pa). Cerebral perfusion was established through the brachiocephalic artery only. The aortic aneurysm and sinuses of Valsalva were excised, and the PAs were isolated. Arteriotomy of both PAs was performed. A dissection plane was raised within the tunica media of each of the main PAs and was developed to the takeoff of the lower lobar vessels.9 Pulmonary endarterectomy was completed during short periods of systemic circulatory arrest (total, 29 min), with cerebral perfusion maintained at a flow rate of 10 mL/min/kg.
After the bilateral endarterectomy was completed, a button-technique Bentall operation was performed in standard fashion. A 25/28-mm St. Jude valved conduit (St. Jude Medical, Inc.; St. Paul, Minn) was used. An iatrogenic rupture of the base of the left subclavian artery occurred during the isolation and snaring of the brachiocephalic and left carotid arteries, so a Hemashield prosthesis was interposed to restore the flow through it. The open distal anastomosis of the Bentall procedure and repair of the left subclavian artery were completed with the patient under systemic circulatory arrest (total, 49 min) and receiving antegrade cerebral perfusion that was maintained at the flow rate of 10 mL/min/kg.
We performed all procedures while monitoring cerebral perfusion with the use of bifrontal near-infrared spectroscopy, with the goal of ensuring a symmetric bilateral value no lower than 20% below the base value. The total CPB time was 290 min, and the total aortic cross-clamp time was 240 min. The patient was weaned from CPB with a low dose of norepinephrine (maximum, 0.2 μg/kg/min) and dobutamine (maximum, 5 μg/kg/min). Nitric oxide (40 ppm), also used for weaning, was discontinued 70 hours postoperatively. The norepinephrine was discontinued on postoperative day 2 and the dobutamine on day 5. Table I shows the patient's postoperative hemodynamic values, obtained on day 6 before his discharge from the intensive care unit (ICU).
The patient's postoperative course was uneventful. He was supported on mechanical ventilation for 96 hours and was in the ICU for 144 hours. During his ICU stay, he was given 150 mg of sildenafil twice daily. The lengthy ventilation time was required to achieve controlled, slow weaning from the nitric oxide, to administer an intravenous sodium nitroprusside infusion (days 2–10), and to begin sildenafil therapy. The patient was kept in the ICU until day 6 to ensure constant care in the absence of a sub-intensive care unit, and for follow-up of pulmonary pressure trends by means of direct Swan-Ganz catheter monitoring.
Neither renal nor hepatic failure occurred. The patient's neurologic status improved from a slight initial impairment to complete recovery, with no damage. He was discharged from the hospital on postoperative day 19, in NYHA functional class I, on a regimen of warfarin, furosemide, and doxofylline. As of August 2012, he remained in NYHA class I and had not sustained any neurologic event.
Discussion
Pulmonary endarterectomy enables the extensive removal of chronic thromboembolic material from the PA. This procedure takes a long time even when performed by experienced surgeons, and several short periods of total circulatory arrest are usually required. In the event of combined operations, the total time under circulatory arrest can be quite long, with a high risk of postoperative sequelae. Moreover, the need to correct other cardiac diseases, such as in our patient, makes these interventions hazardous and often prohibitive. The possibility of maintaining continuous antegrade cerebral perfusion, even during pulmonary endarterectomy alone, makes such a lengthy and intricate procedure feasible, with considerably lower risk of neurologic damage and a better chance of rapid postoperative recovery. In addition, the use of near-infrared spectroscopy enables real-time monitoring of cerebral blood flow—contributing to the safety of the operation. Our experience shows that it is possible—in only one surgical session—to perform procedures that are usually scheduled for multiple sessions.
Footnotes
Address for reprints: Davide Ricci, MD, Division of Cardiac Surgery, San Giovanni Battista Hospital Molinette, University of Torino, 10126 Torino, Italy
E-mail: dricci@molinette.piemonte.it
References
- 1.Corsico AG, D'Armini AM, Cerveri I, Klersy C, Ansaldo E, Niniano R, et al. Long-term outcome after pulmonary endarterectomy. Am J Respir Crit Care Med 2008;178(4):419–24. [DOI] [PubMed]
- 2.Dartevelle P, Fadel E, Mussot S, Cerrina J, Leroy Ladurie F, Lehouerou D, et al. Surgical treatment of chronic thromboembolic pulmonary hypertension [in French]. Presse Med 2005;34(19 Pt 2):1475–86. [DOI] [PubMed]
- 3.Jamieson SW, Kapelanski DP, Sakakibara N, Manecke GR, Thistlethwaite PA, Kerr KM, et al. Pulmonary endarterectomy: experience and lessons learned in 1,500 cases. Ann Thorac Surg 2003;76(5):1457–64. [DOI] [PubMed]
- 4.Thistlethwaite PA, Kaneko K, Madani MM, Jamieson SW. Technique and outcomes of pulmonary endarterectomy surgery. Ann Thorac Cardiovasc Surg 2008;14(5):274–82. [PubMed]
- 5.Kazui T, Washiyama N, Muhammad BA, Terada H, Yamashita K, Takinami M, Tamiya Y. Total arch replacement using aortic arch branched grafts with the aid of antegrade selective cerebral perfusion. Ann Thorac Surg 2000;70(1):3–9. [DOI] [PubMed]
- 6.Kazui T, Yamashita K, Washiyama N, Terada H, Bashar AH, Suzuki T, Ohkura K. Usefulness of antegrade selective cerebral perfusion during aortic arch operations. Ann Thorac Surg 2002;74(5):S1806–9. [DOI] [PubMed]
- 7.Jamieson SW, Kapelanski DP. Pulmonary endarterectomy. Curr Probl Surg 2000;37(3):165–252. [DOI] [PubMed]
- 8.Thistlethwaite PA, Mo M, Madani MM, Deutsch R, Blanchard D, Kapelanski DP, Jamieson SW. Operative classification of thromboembolic disease determines outcome after pulmonary endarterectomy. J Thorac Cardiovasc Surg 2002; 124(6):1203–11. [DOI] [PubMed]
- 9.Jamieson SW, Auger WR, Fedullo PF, Channick RN, Kriett JM, Tarazi RY, Moser KM. Experience and results with 150 pulmonary thromboendarterectomy operations over a 29-month period. J Thorac Cardiovasc Surg 1993;106(1):116–27. [PubMed]


