Abstract
Context:
Tight glucose control (TGC) to normal-for-age fasting blood glucose levels reduced morbidity and mortality in surgical adult and pediatric intensive care unit (ICU) patients. In adults, TGC did not affect the illness-induced alterations in thyroid hormones. With better feeding in children than in adult patients, we hypothesized that TGC in pediatric ICU patients reactivates the thyroid axis.
Objective:
The aim of this study was to assess the impact of TGC on the thyroid axis in pediatric ICU patients and to investigate how these changes affect the TGC outcome benefit.
Design and Patients:
We conducted a preplanned analysis of all patients not treated with thyroid hormone, dopamine, or corticosteroids who were included in a randomized controlled trial on TGC (n = 700).
Main Outcome Measures:
Serum TSH, T4, T3, and rT3 were measured upon admission and on ICU day 3 or the last ICU day for patients discharged earlier. Changes from baseline were compared for the TGC and usual care groups. The impact on the outcome benefit of TGC was assessed with multivariable Cox proportional hazard analysis, correcting for baseline risk factors.
Results:
TGC further lowered the T3/rT3 ratio (P = 0.03), whereas TSH (P = 0.09) and T4 (P = 0.3) were unaltered. With TGC, the likelihood of earlier live discharge from the ICU was 19% higher at any time (hazard ratio, 1.190; 95% confidence interval, 1.010–1.407; P = 0.03). This benefit was statistically explained by the further reduction of T3/rT3 with TGC because an increase in T3/rT3 was strongly associated with a lower likelihood for earlier live discharge (hazard ratio per unit increase, 0.863; 95% confidence interval, 0.806–0.927; P <0.0001).
Conclusions:
TGC further accentuated the peripheral inactivation of thyroid hormone. This effect, mimicking a fasting response, statistically explained part of the clinical outcome benefit of TGC.
The thyroid axis plays a key role in the endocrine regulation of metabolic homeostasis. For example, with fasting, a rapid inactivation of thyroid hormone, evidenced by a decrease in the circulating levels of the active thyroid hormone T3 and a rise in the inactive rT3, is considered to reduce energy expenditure when nutrients are unavailable (1, 2). Critical illness evokes similar alterations in the thyroid axis, even when nutrients are provided. Typically, a lowering of T3 and an increase in rT3 occur in the acute phase of critical illness (Fig. 1), accompanied by a lowered TSH and T4 in the more chronic phase of illness (3). The acute changes are assumed to be mediated by cytokine-induced reduced activity of the type 1 deiodinase (D1) in liver and kidney and a reactivation of the oncofetal protein type 3 deiodinase (D3) in liver and muscle (4–6). Reduced type 2 deiodinase (D2) activity in muscle did not appear to play a role in the lowering of T3 levels in critical illness (7). Suppression of hypothalamic TRH gene expression likely explains the lowered TSH and, with time, also the lowered T4 during illness (8). Together, these changes are referred to as the “nonthyroidal illness” syndrome, the severity of which is associated with risk of morbidity and mortality (9). Although recovery of critical illness is preceded by a rise in TSH (10), it remains unclear whether these alterations in the thyroid axis during the course of critical illness reflect a beneficial adaptation or a maladaptive harmful response.
Fig. 1.
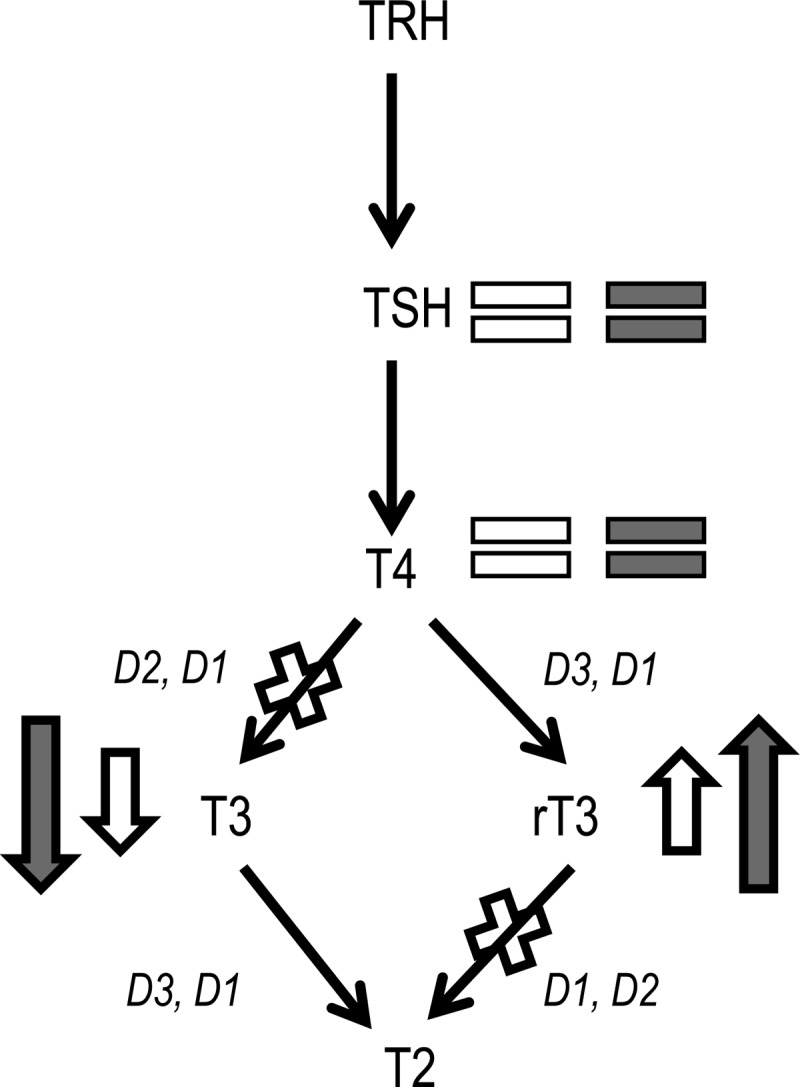
Simplified overview of the thyroid hormone axis. Major changes within the thyroid axis during the acute phase of critical illness (open signs). Changes within the thyroid axis observed with TGC in critically ill infants and children are depicted with the filled signs. (See Results for details.)
A nonthyroidal illness-like syndrome is also present in patients diagnosed with type 1 diabetes mellitus, after a period of uncontrolled hyperglycemia (11, 12). Low T3 levels and increased rT3 levels return to normal in such patients after initiation of insulin therapy and normalization of blood glucose levels. A recent randomized controlled trial (RCT) has shown that prevention of hyperglycemia during critical illness by insulin-titrated tight glucose control (TGC) to age-normal fasting blood glucose levels reduced morbidity and mortality in pediatric intensive care unit (PICU) patients not suffering from diabetes mellitus (13). In critically ill adults, TGC during critical illness did not normalize the illness-induced alterations in thyroid hormones (9), but this could be attributed to relatively inadequate nutrition, which may have precluded an anabolic response and restoration of the thyroid abnormalities. Considering the more aggressive feeding strategies generally applied for children in the intensive care unit (ICU) (14), we hypothesized that a reactivation of the thyroid axis might occur with insulin treatment in this young patient population. Data on nonthyroidal illness in critically ill children are scarce and are limited to small subpopulations (15–20), and the link with outcome is therefore less well documented.
Here, we aimed to investigate the impact of insulin-titrated TGC on thyroid hormone parameters in a large PICU population in the context of a RCT (n = 700) and how any potential changes in thyroid hormone parameters affect the outcome benefit of TGC.
Patients and Methods
Patients
This study was a preplanned part of a large, prospective RCT on TGC in PICU patients (13) (NCT00214916). Institutional Review Board approval (ML2586) was obtained. All infants and children (aged 0–16 yr) admitted to the PICU were eligible for inclusion after written informed consent from the patients' next of kin. For exclusion criteria and the consort flow diagram, we refer to the original report of the study (13).
Patients (n = 700) were randomly assigned to TGC with intensive insulin therapy (n = 349) or to usual care (UC; n = 351). Insulin infusions were prepared with Actrapid HM (Novo Nordisk, Bagsværd, Denmark). In the UC group, insulin infusion was started only when the blood glucose level exceeded 215 mg/dl (11.9 mmol/liter) twice, and the dose was adjusted by the PICU nurses to keep blood glucose between 180 and 215 mg/dl (10–11.9 mmol/liter). When the blood glucose level fell below 180 mg/dl (10 mmol/liter), insulin infusion was tapered down and stopped.
In the TGC group, insulin was infused to control blood glucose levels within the age-adjusted normal fasting range: 50–80 mg/dl (2.8–4.4 mmol/liter) for infants (age <1 yr); and 70–100 mg/dl (3.9–5.5 mmol/liter) for children (ages 1–16 yr) (21). Insulin infusion was started when the blood glucose level exceeded the upper normal limit, and the dose was adjusted to maintain levels within the defined targets. For infants, insulin infusions were stopped when blood glucose fell below 50 mg/dl (2.8 mmol/liter). When blood glucose fell below 30 mg/dl (<1.7 mmol/liter), 1 ml/kg of a 50% dextrose solution was given. For children, insulin infusions were stopped when blood glucose fell below 70 mg/dl (3.9 mmol/liter). When blood glucose fell below 40 mg/dl (2.2 mmol/liter), 1 ml/kg of a 50% dextrose solution was given. Rebound hyperglycemia after hypoglycemia was carefully avoided (13). At discharge from the PICU, the study intervention ended.
Whole blood glucose concentration was systematically measured in arterial blood at 1- to 4-h intervals using the ABL700 analyzer (Radiometer Medical A/S, Copenhagen, Denmark). Hypoglycemia in infants and children was defined, as in adults, as a blood glucose below 40 mg/dl (2.2 mmol/liter) (22). Blood was systematically sampled upon admission and then daily at 0600 h until PICU discharge or death. After clotting and centrifugation, serum was kept frozen at −80 C until the assays were done. Samples were analyzed in batch, with the samples obtained from the same patient analyzed in the same assay run.
Thyroid hormone parameters were measured in all patients originally included in the RCT, who had not received treatment with thyroid hormone, dopamine, or corticosteroids, to avoid confounding iatrogenic effects (23, 24). The number of patients excluded for this reason was similar in both the TGC [51 patients (15%)] and UC groups [61 patients (17%)] (P = 0.3). Baseline characteristics of the studied patients in both groups were comparable for gender, age, and type and severity of illness—the latter reflected by the pediatric logistic organ dysfunction (PELOD) score (25), the need for mechanical ventilation, and the need for extracorporeal membrane oxygenation or other assist devices (Table 1).
Table 1.
Characteristics of the patients
| UC | TGC | P value | |
|---|---|---|---|
| n | 290 | 298 | |
| Patient characteristics | |||
| Infants (age <1 yr) | 130 (44.8) | 135 (45.3) | 0.9 |
| Age (yr), median (IQR) | 1.3 (0.3–4.4) | 1.4 (0.3–5.1) | 0.5 |
| Weight (kg), median (IQR) | 8.5 (4.9–17.0) | 9.3 (5.0–17.0) | 0.5 |
| BMI (kg/m2), median (IQR) | 14.9 (13.5–16.3) | 14.9 (13.6–16.3) | 0.5 |
| Gender, n (% male) | 167 (57.6) | 170 (57.0) | 0.9 |
| Malignancy | 4 (1.4) | 12 (4.0) | 0.05 |
| Diabetes | 3 (1.0) | 3 (1.0) | >0.9 |
| Reason for ICU admission | 0.9 | ||
| Cardiac surgery for congenital heart defects | 235 (81.0) | 241 (80.9) | |
| Complicated/high risk surgery or trauma | 28 (9.7) | 25 (8.4) | |
| Neurological medical disorders | 6 (2.1) | 10 (3.4) | |
| Infectious medical diseases | 11 (3.8) | 10 (3.4) | |
| Other medical disorders | 10 (3.4) | 12 (4.0) | |
| Mechanical ventilation | 280 (96.6) | 289 (97.0) | 0.8 |
| ECMO or other assist device | 6 (2.1) | 8 (2.7) | 0.6 |
| PELOD first 24 h in PICU (median, IQR) | 11 (2–12) | 11 (2–12) | 0.9 |
| Length of stay (d), median (IQR) | 3 (2–6) | 2 (2–5) | 0.02 |
| Short stay (<3 d) | 119 (41.0) | 156 (52.3) | 0.006 |
| Death in ICU | 8 (2.8) | 6 (2.0) | 0.6 |
| Glucose management | |||
| Randomized to intensive insulin therapy | 0 (0) | 298 (100) | NR |
| Treated with insulin | 132 (45.5) | 294 (98.7) | <0.0001 |
| Mean daily dose of insulin (IU/kg/d), median (IQR) | 0.0 (0.0–0.6) | 1.3 (0.9–1.8) | <0.0001 |
| Mean morning blood glucose (mg/dl), median (IQR) | 126.8 (108.2–149) | 88.0 (75.7–99.5) | <0.0001 |
| Patients with hypoglycemia | 4 (1.4) | 72 (24.2) | <0.0001 |
| Lowest blood glucose level during ICU stay (mg/dl) (mean ± sd) | 89.6 ± 26.5 | 53.4 ± 20.1 | <0.0001 |
| Total (kcal/kg) on d 3, median (IQR) | 45.5 (34.1–56.4) | 43.5 (35.5–55.3) | 0.8 |
| Glucose infusion (mg/kg/min) on d 3, median (IQR) | 5.6 (4.4–7.1) | 5.6 (4.4–6.9) | 0.5 |
Data are expressed as number (percentage), unless specified otherwise. Characteristics of the patients in the study after exclusion of those patients who received dopamine, corticosteroids, or thyroid hormone in the ICU prior to the day of sampling. BMI, Body mass index; ECMO, extracorporeal membrane oxygenation; NR, not relevant. To convert glucose to mmol/liter multiply by 0.0555.
Serum analyses
Serum TSH levels were quantified with a commercially available TSH immunoradiometric assay (TSH IRMA kit; Immunotech, Beckman Coulter, Prague, Czech Republic). Serum T4, T3, and rT3 were quantified with commercially available RIA kits (total T4 RIA kit and total T3 RIA kit; Immunotech, Beckman Coulter; rT3 RIA, Immunodiagnostic Systems S.A., Liège, Belgium).
Statistical analysis
Differences between treatment groups were assessed by the χ2 test for proportions, Student's t test for normally distributed data (presented as mean ± sd), and Mann-Whitney U test for not-normally distributed data [presented as median and interquartile range (IQR)], unless specified otherwise.
The predictive value of baseline levels of thyroid hormones for outcome from critical illness was analyzed by multivariable Cox proportional hazard analysis of the time to alive discharge from ICU, correcting for baseline risk factors (upon-admission diagnosis, age, and PELOD severity of illness score). To deal with competing risk between ICU stay and mortality, data from patients who died were censored at a time point beyond that of the longest staying surviving patient (26). Significance was analyzed with the Effect Likelihood Ratio Test.
To assess the impact of TGC on thyroid hormone parameters, we quantified the change in thyroid hormones from the value measured in the upon-admission prerandomization sample to the value on ICU day 3, which is the median duration of ICU stay, or the last ICU day for patients discharged earlier. Differences in these changes in levels of thyroid hormones between TGC and UC groups were analyzed by non-parametrical tests (Mann-Whitney U test and Van der Waerden test). Data are presented as median (IQR) or means with se values. Observed differences with univariable analysis were again analyzed after correction for baseline risk factors (upon-admission diagnosis, age, and PELOD severity of illness score) using linear regression control to confirm these differences.
To assess the impact of any of the observed differences in thyroid hormone changes from baseline with TGC on the outcome benefit with TGC, a multivariable Cox proportional hazard analysis of the time to live discharge from ICU was performed. Risk factors entered into the model, again, were upon-admission diagnosis, age, and PELOD severity of illness score. In this analysis, we started from the initial model that included the randomized treatments groups and the baseline risk factors. In the next step, the TGC-affected changes from baseline in the thyroid hormone parameters were added, with the assumption that if these changes reduced or annihilated the significant effect of TGC, they statistically explained, to a certain extent, the impact of TGC on outcome. Alternatively, if adding these changes in thyroid hormone parameters to the model increased the significant effect of TGC, they could be interpreted as deleterious side effects of TGC. Significance was analyzed with the Effect Likelihood Ratio Test.
All statistical analyses were performed with JMP for Mac version 9.0.0 (SAS Institute Inc., Cary, NC). Statistical significance was set at P < 0.05.
Results
Predictive value for outcome of the baseline thyroid hormone levels
Upon admission, serum levels of TSH, T4, and T3 were low, and rT3 levels were high as compared with healthy reference values for the median age of the study population (Table 2) (27). Corrected for other baseline risk factors, a higher baseline serum T4 level [hazard ratio (HR) per nmol/liter, 1.004; 95% confidence interval (CI), 1.000–1.008; P = 0.02] and a higher T3/rT3 ratio (HR per unit, 1.144; 95% CI, 1.067–1.227; P = 0.0002) was independently associated with an earlier live discharge from ICU (Table 3). TSH was not associated with an earlier live discharge from ICU in this multivariable model.
Table 2.
Baseline values
| Total | Age-matched healthy reference range | UC | TGC | P value | |
|---|---|---|---|---|---|
| n | 588 | 290 | 298 | ||
| TSH (mIU/liter) | 1.49 (0.86–2.34) | 0.6–6.3 | 1.52 (0.86–2.40) | 1.47 (0.87–2.24) | 0.6 |
| T4 (nmol/liter) | 72.3 (57.1–90.6) | 94.0–193.1 | 73.6 (57.6–94.2) | 71.2 (56.1–88.1) | 0.4 |
| T3 (nmol/liter) | 1.05 (0.74–1.45) | 1.62–4.14 | 1.05 (0.72–1.45) | 1.04 (0.77–1.44) | 0.9 |
| rT3 (nmol/liter) | 0.72 (0.51–1.19) | 0.23–1.09 | 0.74 (0.51–1.21) | 0.72 (0.51–1.18) | 0.8 |
| T3/rT3 | 1.45 (0.71–2.43) | 1.5–18 | 1.45 (0.69–2.34) | 1.47 (0.71–2.53) | 0.7 |
Data are expressed as median (IQR). To convert T4 from nmol/liter to μg/dl, multiply by 0.078; to convert T3 from nmol/liter to ng/dl, multiply by 65; to convert rT3 from nmol/liter to ng/dl, multiply by 65. For healthy reference ranges for TSH, T4, T3, and rT3, see Ref. 27. Reference range for T3/rT3 was calculated with the reference values for T3 and rT3.
Table 3.
Independent predictive value of baseline thyroid hormone parameters for recovery from critical illness
| Likelihood for earlier live discharge from ICU | P value | |
|---|---|---|
| Randomization to TGC vs. UC | 1.19 (1.00–1.41) | 0.04 |
| Upon-admission diagnosis (as compared with complicated/high risk surgery or trauma) | 0.001 | |
| Cardiac surgery for congenital heart defects | 1.16 (0.86–1.61) | 0.3 |
| Neurological medical disorders | 0.79 (0.43–1.38) | 0.4 |
| Infectious medical disorders | 0.61 (0.35–1.03) | 0.06 |
| Other medical disorders | 0.51 (0.28–0.87) | 0.01 |
| Age: child vs. infant | 1.26 (1.03–1.53) | 0.02 |
| PELOD score d 1 (per unit added) | 0.95 (0.93–0.96) | <0.0001 |
| TSH adm (per mIU/liter added) | 1.01 (0.97–1.05) | 0.6 |
| T4 adm (per nmol/liter added) | 1.00 (1.00–1.01) | 0.02 |
| T3/rT3 adm (per unit added) | 1.14 (1.07–1.23) | 0.0002 |
Data are expressed as HR (95% CI). adm, Admission value.
Effect of TGC on thyroid hormone levels
Upon-admission serum levels of TSH, T4, T3, and rT3 did not differ between TGC and UC patients (Table 2).
As compared with UC, TGC further lowered serum TSH between admission and ICU day 3 or the last ICU day in patients discharged earlier in univariable analysis (P = 0.007). In multivariable analysis, correcting for baseline risk factors, this difference became nonsignificant (P = 0.09) (Fig. 2). TGC did not affect the change from baseline in serum T4 levels (univariable P = 0.3), whereas it further reduced serum T3 levels (univariable P = 0.04; multivariable P = 0.008), further increased serum rT3 (univariable P = 0.04; multivariable P = 0.04), and hence further lowered the T3/rT3 ratio (univariable P = 0.04; multivariable P = 0.03). These changes were not explained by hypoglycemia because they were also observed in the patients who did not experience hypoglycemia [change in T3/rT3 ratio (mean ± sem), −1.14 ± 0.09 with TGC and −0.83 ± 0.09 with UC; P = 0.006].
Fig. 2.
Effect of TGC on the thyrotropic axis. The line charts (means ± sem) depict the baseline (adm) and day 3 (d3), or last day when patients were discharged earlier (LD), values for serum TSH, T4, T3, rT3, and T3/rT3 of patients randomized to TGC (filled ovals) or UC (open ovals). Patients who received dopamine, corticosteroids, or thyroid hormone in the ICU before the day of sampling were excluded for this analysis. P denotes the P value for the difference in the changes from baseline between TGC and UC groups, obtained after correction for baseline risk factors. adm, Admission.
Impact of the changes in thyroid hormones induced by TGC on the clinical benefit of TGC
Without the changes in thyroid hormones added to the multivariable Cox proportional hazard model, the likelihood of an earlier live discharge from ICU was 19% higher at any time in the TGC group (HR, 1.190; 95% CI, 1.010–1.407; P = 0.03) as compared with the UC group (Table 4). This benefit was at least in part statistically explained by the further reduction of T3/rT3 evoked by TGC. Indeed, adding to the model the change in T3/rT3 revealed that an increase from baseline in T3/rT3 was significantly associated with a lower likelihood for earlier live discharge (HR per unit increase, 0.863; 95% CI, 0.806–0.927; P < 0.0001), which replaced the impact of TGC. Adding the changes in TSH and T4 to the model, which were unaffected by TGC, did not alter this result. This model also revealed that, in contrast to a rise in T3/rT3 within the first 3 d of illness, which was associated with a lower likelihood for earlier live discharge, a rise in T4 was significantly associated with a higher likelihood for earlier live discharge (HR per nmol/liter increase, 1.009; 95% CI, 1.006–1.013; P < 0.0001).
Table 4.
Contribution of the TGC-induced changes in thyroid parameters in explaining the benefit of TGC on outcome
| Likelihood for earlier live discharge from ICU | P value | |
|---|---|---|
| Model 1 | ||
| Randomization to TGC vs. UC | 1.19 (1.01–1.41) | 0.03 |
| Upon-admission diagnosis (as compared with complicated/high risk surgery or trauma) | <0.0001 | |
| Cardiac surgery for congenital heart defects | 1.12 (0.84–1.53) | 0.4 |
| Neurological medical disorders | 0.85 (0.47–1.46) | 0.5 |
| Infectious medical disorders | 0.50 (0.29–0.83) | 0.006 |
| Other medical disorders | 0.46 (0.26–0.77) | 0.002 |
| Age: child vs. infant | 1.45 (1.21–1.74) | <0.0001 |
| PELOD d 1 (per unit added) | 0.94 (0.93–0.95) | <0.0001 |
| Model 2 | ||
| Randomization to TGC vs. UC | 1.14 (0.95–1.36) | 0.1 |
| Upon-admission diagnosis (as compared with complicated/high risk surgery or trauma) | 0.05 | |
| Cardiac surgery for congenital heart defects | 1.15 (0.84–1.60) | 0.3 |
| Neurological medical disorders | 1.00 (0.54–1.74) | 0.9 |
| Infectious medical disorders | 0.81 (0.44–1.40) | 0.4 |
| Other medical disorders | 0.54 (0.30–0.94) | 0.02 |
| Age: child vs. infant | 1.25 (1.02–1.51) | 0.02 |
| PELOD d 1 (per unit added) | 0.94 (0.92–0.95) | <0.0001 |
| T3/rT3 (change from baseline, per unit added) | 0.86 (0.81–0.93) | <0.0001 |
| Model 3 | ||
| Randomization to TGC vs. UC | 1.16 (0.97–1.38) | 0.1 |
| Upon-admission diagnosis (as compared with complicated/high risk surgery or trauma) | 0.002 | |
| Cardiac surgery for congenital heart defects | 1.22 (0.89–1.71) | 0.2 |
| Neurological medical disorders | 0.99 (0.54–1.73) | 0.9 |
| Infectious medical disorders | 0.76 (0.42–1.31) | 0.3 |
| Other medical disorders | 0.51 (0.28–0.89) | 0.01 |
| Age: child vs. infant | 1.27 (1.04–1.55) | 0.01 |
| PELOD d 1 (per unit added) | 0.94 (0.93–0.95) | <0.0001 |
| T3/rT3 (change from baseline, per unit added) | 0.85 (0.79–0.91) | <0.0001 |
| TSH (change from baseline, per mIU/liter added) | 0.96 (0.93–0.99) | 0.01 |
| T4 (change from baseline, per nmol/liter added) | 1.01 (1.01–1.01) | <0.0001 |
Data are expressed as HR (95% CI). Multivariable Cox proportional hazard analysis: Model 1—initial model which includes the randomized treatment groups and the baseline risk factors. Model 2—the TGC affected changes from baseline in the thyroid hormone parameters are added. The change in T3/rT3 annihilates the significant effect of TGC. Model 3—Adding the changes in TSH and T4 to the model, which were unaffected by TGC, does not alter this result.
Discussion
Critically ill children with a lower T4 serum concentration and a lower T3/rT3 ratio upon admission were found to have a lower likelihood of earlier live discharge from ICU. Such associations could either be adaptive or maladaptive for recovery. We previously reported that TGC targeting blood glucose levels strictly to age-normal fasting levels decreased morbidity and mortality of critically ill children, despite an increased incidence of hypoglycemia (13). The current analysis of the impact of this intervention on the thyroid parameters revealed that TGC did not affect TSH and T4 in critically ill children. TGC, rather than elevating T3/rT3 as occurs in insulin-treated diabetes, further lowered T3/rT3. This decrease from baseline in T3/rT3 evoked by TGC statistically explained at least part of its beneficial impact on outcome.
The association between severity of thyroid hormone abnormalities—a low TSH, T4, or T3 and a high rT3—upon admission to the ICU and a subsequent adverse outcome has been described earlier in children, although often in small and selected populations (15–20). However, as in adults, it remained unclear whether this association reflects a beneficial adaptation proportionately to the severity of illness, or whether it reflects a causal relationship that would imply a necessity for treatment.
We here documented that TGC, an intervention that improved survival and reduced morbidity of critically ill children, did not alter TSH and T4 but further decreased T3 and further elevated rT3, thereby lowering the T3/rT3 ratio, during the first days of critical illness. This observation suggests that TGC suppressed the peripheral activity of D1 and D2 and/or stimulated D3 (Fig. 1). In adult patients (human and rabbit) suffering from critical illness, low tissue D1 activity and elevated tissue D3 activity have been documented in relation to the suppressed T3/rT3 ratio (5, 28). Also in fasting healthy animals, such changes in the deiodinase enzymes have been reported (29–31). In the adult ICU patient study on the effect of TGC, insulin treatment did not affect the illness-associated changes in thyroid hormones (9), which contrasts with what is observed with initiation of insulin treatment in newly diagnosed diabetes mellitus (11, 12). This could possibly be explained by selection of long-stay patients only in the adult study on TGC (9). Alternatively, relatively poor feeding during acute critical illness in these adult patients may have prevented any improvements in these thyroid hormone changes with intensive insulin therapy. In the PICU setting, nutritional management is more aggressive than in the adult ICU setting, which made us hypothesize that we would see normalization of thyroid hormones with insulin treatment in critically ill children. This, however, did not happen. In contrast, we found a further aggravation of the peripheral thyroid hormone inactivation with insulin treatment, an effect that also occurs with fasting. The fact that targeting age-adjusted normoglycemia with insulin induced such a “fasting response” in the thyroid hormone parameters of critically ill children, despite the fact that these children were in the fed state, was striking and requires some explanation. One possibility is that the age-adjusted fasting target for blood glucose may have lowered blood glucose to the extent that it signaled through the fasting pathways, which are catabolic. This contrasts with the intuitive thinking that TGC with intensive insulin therapy mainly exerts its effects through anabolic insulin signaling. However, due to hepatic insulin resistance (32), anabolic insulin signaling may be hampered, and the effects of lowering blood glucose levels may have gained importance. Catabolic pathways that could be interesting candidates for further investigation are nutrient sensors such as constitutive androstane receptor, AMP-activated protein kinase, and the bile acid-activated receptors (33–35).
The further lowering of the T3/rT3 ratio by TGC statistically explained part of the benefit of TGC on outcome. The association of a beneficial outcome with a further lowering of the T3/rT3 ratio by TGC was independent of the association between a rising T4 and a better outcome. We could infer from our data that changing the peripheral conversion of T4 from metabolically active T3 to inactive rT3 during the first days of critical illness is adaptive and beneficial for recovery. The association at baseline of a low T3/rT3 ratio with adverse outcome is not in contradiction with our findings. Indeed, our data provide evidence that the inactivation of thyroid hormone upon admission to ICU may already reflect a beneficial adaptive response. Because these changes in thyroid hormone parameters indeed resemble a fasting response, these data are in line with the recently observed benefit of early nutrient restriction on recovery from critical illness in adult ICU patients (36). Hence, our findings may also argue against the use of exogenous T3 in the early phase of critical illness, confirming the results of a study in burn injury patients (37). Also in other studies, beneficial effects of T3 administration on cardiac function in adults and children after cardiac surgery did not translate into shortening of the duration of stay in the ICU or hospital (38, 39). In contrast, our data showed that, independent of the T3/rT3 changes, an early rise in T4 was associated with better chances of recovery. Because T4 was not affected by TGC, this finding remains associative, and further intervention studies are required to interpret this as causal or casual. Also, the absence of a significant effect of TGC on TSH, in the face of unaltered T4, suggests that TGC did not affect the central component of the hypothalamic-anterior pituitary-thyroid axis in the acute phase of critical illness. However, interpretation of TSH levels has to be done with caution because single sample measurements do not allow us to assess the impact on pulsatile TSH secretion (3). Taken together, our findings support the concept that the acute-peripheral-inactivation of thyroid hormone is likely beneficial.
The suppressive effect of TGC on T3/rT3 documented here, which statistically explained at least some of the benefit of TGC, together with the recent evidence in adult patients that nutrient restriction early in the course of critical illness exerts benefits on outcome (36), could be reconciled by the concept that early catabolism may exert benefits for recovery from critical illness. This concept could also be reconciled with GH resistance early during critical illness (40–42), which was shown to be further pronounced by TGC in both the adult and pediatric studies on TGC (43, 44). The potential benefit of catabolic pathways early in the course of critical illness could be explained by improved clearance of damaged organelles and damaged protein aggregates by autophagy, a cellular housekeeping system that is tightly regulated by nutrients and growth factors (45, 46). A long-term follow-up of growth, weight gain, and neurocognitive development of the pediatric patients in both treatment groups is needed to exclude late harm of this TGC-reinforced early catabolism.
Our study has some weaknesses that should be highlighted. First, although we studied a randomized intervention that affected T3 and rT3, the changes in T3 and rT3 were not directly brought about. Further studies in genetically modified animals, underexpressing D1/D2 and/or overexpressing D3, are needed to specifically address the role of deiodinases in critical illness. Second, for obvious ethical reasons, tissue biopsies were not available and hence the interpretation of altered deiodinase activities remains indirect. Finally, to avoid selection bias, we focused on the early changes in thyroid hormones. This precludes any conclusion about the chronic phase of critical illness in these children.
In conclusion, TGC in critically ill infants and children did not reactivate the thyrotropic axis but further accentuated the peripheral inactivation of thyroid hormone. This effect, which mimicked a fasting response probably due to the lower blood glucose levels achieved with TGC, statistically explained the clinical outcome benefit of TGC. These data provide evidence for the concept that peripheral inactivation of thyroid hormone early in the course of critical illness could be a beneficial adaptation.
Acknowledgments
We thank the clinical and nursing staff of the PICU for excellent patient care, protocol compliance, and sample handling. We are also indebted to Erik Van Herck, Willy Coopmans, Inge Derese, and Sarah Vander Perre for valuable technical assistance.
This work was supported by the Applied Biomedical Research program of the Agency for Innovation by Science and Technology–Flanders, Belgium (IWT-TBM 070695), the Research Foundation–Flanders (FWO project G.0578.09), a Ph.D. Fellowship of the Research Foundation–Flanders (FWO) (to M.G.), and a Senior Clinical Investigator Fellowship of the Research Foundation–Flanders (FWO) (to D.M.). G.V.d.B., by the University of Leuven receives structural research financing via the Methusalem program, funded by the Flemish Government.
Clinical trial registration no. NCT00214916 (www.ClinicalTrials.gov).
Disclosure Summary: The authors have no conflicts of interest to report.
Footnotes
- CI
- Confidence interval
- D1
- -2, -3, type 1, 2, 3 deiodinase
- HR
- hazard ratio
- ICU
- intensive care unit
- IQR
- interquartile range
- PELOD
- pediatric logistic organ dysfunction
- PICU
- pediatric ICU
- RCT
- randomized controlled trial
- TGC
- tight glucose control
- UC
- usual care.
References
- 1. Gardner DF, Kaplan MM, Stanley CA, Utiger RD. 1979. Effect of tri-iodothyronine replacement on the metabolic and pituitary responses to starvation. N Engl J Med 300:579–584 [DOI] [PubMed] [Google Scholar]
- 2. Hugues JN, Burger AG, Pekary AE, Hershman JM. 1984. Rapid adaptations of serum thyrotrophin, triiodothyronine and reverse triiodothyronine levels to short-term starvation and refeeding. Acta Endocrinol (Copenh) 105:194–199 [DOI] [PubMed] [Google Scholar]
- 3. Van den Berghe G, de Zegher F, Bouillon R. 1998. Clinical review 95: acute and prolonged critical illness as different neuroendocrine paradigms. J Clin Endocrinol Metab 83:1827–1834 [DOI] [PubMed] [Google Scholar]
- 4. van der Poll T, Romijn JA, Wiersinga WM, Sauerwein HP. 1990. Tumor necrosis factor: a putative mediator of the sick euthyroid syndrome in man. J Clin Endocrinol Metab 71:1567–1572 [DOI] [PubMed] [Google Scholar]
- 5. Peeters RP, Wouters PJ, Kaptein E, van Toor H, Visser TJ, Van den Berghe G. 2003. Reduced activation and increased inactivation of thyroid hormone in tissues of critically ill patients. J Clin Endocrinol Metab 88:3202–3211 [DOI] [PubMed] [Google Scholar]
- 6. Debaveye Y, Ellger B, Mebis L, Darras VM, Van den Berghe G. 2008. Regulation of tissue iodothyronine deiodinase activity in a model of prolonged critical illness. Thyroid 18:551–560 [DOI] [PubMed] [Google Scholar]
- 7. Mebis L, Langouche L, Visser TJ, Van den Berghe G. 2007. The type II iodothyronine deiodinase is up-regulated in skeletal muscle during prolonged critical illness. J Clin Endocrinol Metab 92:3330–3333 [DOI] [PubMed] [Google Scholar]
- 8. Van den Berghe G, de Zegher F, Baxter RC, Veldhuis JD, Wouters P, Schetz M, Verwaest C, Van der Vorst E, Lauwers P, Bouillon R, Bowers CY. 1998. Neuroendocrinology of prolonged critical illness: effects of exogenous thyrotropin-releasing hormone and its combination with growth hormone secretagogues. J Clin Endocrinol Metab 83:309–319 [DOI] [PubMed] [Google Scholar]
- 9. Peeters RP, Wouters PJ, van Toor H, Kaptein E, Visser TJ, Van den Berghe G. 2005. Serum 3,3′,5′-triiodothyronine (rT3) and 3,5,3′-triiodothyronine/rT3 are prognostic markers in critically ill patients and are associated with postmortem tissue deiodinase activities. J Clin Endocrinol Metab 90:4559–4565 [DOI] [PubMed] [Google Scholar]
- 10. Bacci V, Schussler GC, Kaplan TB. 1982. The relationship between serum triiodothyronine and thyrotropin during systemic illness. J Clin Endocrinol Metab 54:1229–1235 [DOI] [PubMed] [Google Scholar]
- 11. Gilani BB, MacGillivray MH, Voorhess ML, Mills BJ, Riley WJ, MacLaren NK. 1984. Thyroid hormone abnormalities at diagnosis of insulin-dependent diabetes mellitus in children. J Pediatr 105:218–222 [DOI] [PubMed] [Google Scholar]
- 12. Joseph J, Saroha V, Payne H, Paul P, Didi M, Isherwood D, Blair J. 2011. Thyroid function at diagnosis of type I diabetes. Arch Dis Child 96:777–779 [DOI] [PubMed] [Google Scholar]
- 13. Vlasselaers D, Milants I, Desmet L, Wouters PJ, Vanhorebeek I, van den Heuvel I, Mesotten D, Casaer MP, Meyfroidt G, Ingels C, Muller J, Van Cromphaut S, Schetz M, Van den Berghe G. 2009. Intensive insulin therapy for patients in paediatric intensive care: a prospective, randomised controlled study. Lancet 373:547–556 [DOI] [PubMed] [Google Scholar]
- 14. Shann F, Henning R, Shekerdemian L. 2008. Paediatric intensive care guidelines. Intensive Care Unit Royal Children's Hospital, Parkville, Victoria, Australia; 94 [Google Scholar]
- 15. Uzel N, Neyzi O. 1986. Thyroid function in critically ill infants with infections. Pediatr Infect Dis 5:516–519 [DOI] [PubMed] [Google Scholar]
- 16. Allen DB, Dietrich KA, Zimmerman JJ. 1989. Thyroid hormone metabolism and level of illness severity in pediatric cardiac surgery patients. J Pediatr 114:59–62 [DOI] [PubMed] [Google Scholar]
- 17. Bettendorf M, Schmidt KG, Tiefenbacher U, Grulich-Henn J, Heinrich UE, Schönberg DK. 1997. Transient secondary hypothyroidism in children after cardiac surgery. Pediatr Res 41:375–379 [DOI] [PubMed] [Google Scholar]
- 18. den Brinker M, Dumas B, Visser TJ, Hop WC, Hazelzet JA, Festen DA, Hokken-Koelega AC, Joosten KF. 2005. Thyroid function and outcome in children who survived meningococcal septic shock. Intensive Care Med 31:970–976 [DOI] [PubMed] [Google Scholar]
- 19. Marks SD, Haines C, Rebeyka IM, Couch RM. 2009. Hypothalamic-pituitary-thyroid axis changes in children after cardiac surgery. J Clin Endocrinol Metab 94:2781–2786 [DOI] [PubMed] [Google Scholar]
- 20. Angelousi AG, Karageorgopoulos DE, Kapaskelis AM, Falagas ME. 2011. Association between thyroid function tests at baseline and the outcome of patients with sepsis or septic shock: a systematic review. Eur J Endocrinol 164:147–155 [DOI] [PubMed] [Google Scholar]
- 21. Brown TC, Connelly JF, Dunlop ME, McDougall PN, Tibballs J. 1980. Fasting plasma glucose in children. Aust Paediatr J 16:28–29 [DOI] [PubMed] [Google Scholar]
- 22. Preissig CM, Rigby MR. 2010. A disparity between physician attitudes and practice regarding hyperglycemia in pediatric intensive care units in the United States: a survey on actual practice habits. Crit Care 14:R11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Van den Berghe G, de Zegher F, Lauwers P. 1994. Dopamine suppresses pituitary function in infants and children. Crit Care Med 22:1747–1753 [PubMed] [Google Scholar]
- 24. Faglia G, Ferrari C, Beck-Peccoz P, Spada A, Travaglini P, Ambrosi B. 1973. Reduced plasma thyrotropin response to thyrotropin releasing hormone after dexamethasone administration in normal subjects. Horm Metab Res 5:289–292 [DOI] [PubMed] [Google Scholar]
- 25. Leteurtre S, Martinot A, Duhamel A, Proulx F, Grandbastien B, Cotting J, Gottesman R, Joffe A, Pfenninger J, Hubert P, Lacroix J, Leclerc F. 2003. Validation of the paediatric logistic organ dysfunction (PELOD) score: prospective, observational, multicentre study. Lancet 362:192–197 [DOI] [PubMed] [Google Scholar]
- 26. Andersen PK, Abildstrom SZ, Rosthøj S. 2002. Competing risks as a multi-state model. Stat Methods Med Res 11:203–215 [DOI] [PubMed] [Google Scholar]
- 27. Fisher DA, Grueters A. 2008. Thyroid disorders in childhood and adolescence. In: Sperling MA, ed. Pediatric endocrinology. 3rd ed Philadelphia: Saunders, Elsevier; 227–253 [Google Scholar]
- 28. Debaveye Y, Ellger B, Mebis L, Van Herck E, Coopmans W, Darras V, Van den Berghe G. 2005. Tissue deiodinase activity during prolonged critical illness: effects of exogenous thyrotropin-releasing hormone and its combination with growth hormone-releasing peptide-2. Endocrinology 146:5604–5611 [DOI] [PubMed] [Google Scholar]
- 29. O'Mara BA, Dittrich W, Lauterio TJ, St Germain DL. 1993. Pretranslational regulation of type I 5′-deiodinase by thyroid hormones and in fasted and diabetic rats. Endocrinology 133:1715–1723 [DOI] [PubMed] [Google Scholar]
- 30. Boelen A, Platvoet-ter Schiphorst MC, van Rooijen N, Wiersinga WM. 1996. Selective macrophage depletion in the liver does not prevent the development of the sick euthyroid syndrome in the mouse. Eur J Endocrinol 134:513–518 [DOI] [PubMed] [Google Scholar]
- 31. Van der Geyten S, Van Rompaey E, Sanders JP, Visser TJ, Kühn ER, Darras VM. 1999. Regulation of thyroid hormone metabolism during fasting and refeeding in chicken. Gen Comp Endocrinol 116:272–280 [DOI] [PubMed] [Google Scholar]
- 32. Mesotten D, Delhanty PJ, Vanderhoydonc F, Hardman KV, Weekers F, Baxter RC, Van Den Berghe G. 2002. Regulation of insulin-like growth factor binding protein-1 during protracted critical illness. J Clin Endocrinol Metab 87:5516–5523 [DOI] [PubMed] [Google Scholar]
- 33. Maglich JM, Watson J, McMillen PJ, Goodwin B, Willson TM, Moore JT. 2004. The nuclear receptor CAR is a regulator of thyroid hormone metabolism during caloric restriction. J Biol Chem 279:19832–19838 [DOI] [PubMed] [Google Scholar]
- 34. Minokoshi Y, Alquier T, Furukawa N, Kim YB, Lee A, Xue B, Mu J, Foufelle F, Ferré P, Birnbaum MJ, Stuck BJ, Kahn BB. 2004. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428:569–574 [DOI] [PubMed] [Google Scholar]
- 35. Fiorucci S, Mencarelli A, Palladino G, Cipriani S. 2009. Bile-acid-activated receptors: targeting TGR5 and farnesoid-X-receptor in lipid and glucose disorders. Trends Pharmacol Sci 30:570–580 [DOI] [PubMed] [Google Scholar]
- 36. Casaer MP, Mesotten D, Hermans G, Wouters PJ, Schetz M, Meyfroidt G, Van Cromphaut S, Ingels C, Meersseman P, Muller J, Vlasselaers D, Debaveye Y, Desmet L, Dubois J, Van Assche A, Vanderheyden S, Wilmer A, Van den Berghe G. 2011. Early versus late parenteral nutrition in critically ill adults. N Engl J Med 365:506–517 [DOI] [PubMed] [Google Scholar]
- 37. Becker RA, Vaughan GM, Ziegler MG, Seraile LG, Goldfarb IW, Mansour EH, McManus WF, Pruitt BA, Jr, Mason AD., Jr 1982. Hypermetabolic low triiodothyronine syndrome of burn injury. Crit Care Med 10:870–875 [DOI] [PubMed] [Google Scholar]
- 38. Bettendorf M, Schmidt KG, Grulich-Henn J, Ulmer HE, Heinrich UE. 2000. Tri-iodothyronine treatment in children after cardiac surgery: a double-blind, randomised, placebo-controlled study. Lancet 356:529–534 [DOI] [PubMed] [Google Scholar]
- 39. Kaptein EM, Sanchez A, Beale E, Chan LS. 2010. Clinical review: thyroid hormone therapy for postoperative nonthyroidal illnesses: a systematic review and synthesis. J Clin Endocrinol Metab 95:4526–4534 [DOI] [PubMed] [Google Scholar]
- 40. Ross R, Miell J, Freeman E, Jones J, Matthews D, Preece M, Buchanan C. 1991. Critically ill patients have high basal growth hormone levels with attenuated oscillatory activity associated with low levels of insulin-like growth factor-I. Clin Endocrinol (Oxf) 35:47–54 [DOI] [PubMed] [Google Scholar]
- 41. Gardelis JG, Hatzis TD, Stamogiannou LN, Dona AA, Fotinou AD, Brestas PS, Constantopoulos AG. 2005. Activity of the growth hormone/insulin-like growth factor-I axis in critically ill children. J Pediatr Endocrinol Metab 18:363–372 [DOI] [PubMed] [Google Scholar]
- 42. Mesotten D, Van den Berghe G. 2006. Changes within the growth hormone/insulin-like growth factor I/IGF binding protein axis during critical illness. Endocrinol Metab Clin North Am 35:793–805, ix–x [DOI] [PubMed] [Google Scholar]
- 43. Mesotten D, Wouters PJ, Peeters RP, Hardman KV, Holly JM, Baxter RC, Van den Berghe G. 2004. Regulation of the somatotropic axis by intensive insulin therapy during protracted critical illness. J Clin Endocrinol Metab 89:3105–3113 [DOI] [PubMed] [Google Scholar]
- 44. Gielen M, Mesotten D, Brugts M, Coopmans W, Van Herck E, Vanhorebeek I, Baxter R, Lamberts S, Janssen JA, Van den Berghe G. 2011. Effect of intensive insulin therapy on the somatotropic axis of critically ill children. J Clin Endocrinol Metab 96:2558–2566 [DOI] [PubMed] [Google Scholar]
- 45. Levine B, Kroemer G. 2008. Autophagy in the pathogenesis of disease. Cell 132:27–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Vanhorebeek I, Gunst J, Derde S, Derese I, Boussemaere M, Güiza F, Martinet W, Timmermans JP, D'Hoore A, Wouters PJ, Van den Berghe G. 2011. Insufficient activation of autophagy allows cellular damage to accumulate in critically ill patients. J Clin Endocrinol Metab 96:E633–E645 [DOI] [PubMed] [Google Scholar]



