Abstract
Infrared (IR) spectroscopy has been widely utilized for the study of protein folding, unfolding and misfolding processes. We have previously developed a theoretical method for calculating IR spectra of proteins in the amide I region. In this work, we apply this method, in combination with replica-exchange molecular dynamics simulations, to study the equilibrium thermal unfolding transition of the villin headpiece subdomain (HP36). Temperature-dependent IR spectra and spectral densities are calculated. The spectral densities correctly reflect the unfolding conformational changes in the simulation. With the help of isotope labeling, we are able to capture the feature that helix 2 of HP36 loses its secondary structure before global unfolding occurs, in agreement with experiment.
I. INTRODUCTION
The folding of a protein into its unique three-dimensional native structure is essential to its biological function. Many native proteins are only marginally stable, in the sense that small changes in the cellular environment such as temperature and pH may lead to unfolding. Unfolding usually causes a severe reduction in biological activity, and can also lead to misfolding, aggregation, and disease.1–5 Although considerable advances have been made in the study of the structure and dynamics of proteins, unraveling the pathways and mechanisms of protein folding, unfolding and misfolding processes remains a challenge.6–18 Such processes involve the rearrangement of a complex network of molecular interactions. Therefore, monitoring these processes in situ poses substantial challenges for experiment as it requires techniques that have sufficient structural and time resolution.12,14,17 Computer simulations complement experiments by providing atomistic details of the structure and dynamics of these processes, although of course they are limited by factors such as force-field accuracy and simulation time-scales.10,13–16
Infrared (IR) spectroscopy is a highly effective method for probing protein folding, unfolding and misfolding due to its unique structural and temporal sensitivity.19–31 IR experiments typically involve the amide I vibrational mode, which is primarily associated with the peptide bond carbonyl stretch32 and is highly sensitive to protein secondary structure.19–21 Recent development of time-resolved two-dimensional IR (2DIR) spectroscopy provides sub-picosecond time resolution.22,24–28,33–42 Moreover, residue-specific information can be obtained by using isotope labeling. For example, a 13C=18O isotope label lowers the amide I frequency by about 70 cm−1.31,43–55 Carbonyls with 13C=18O labels are essentially shifted out of the main amide I band and can be resolved individually.
The villin headpiece subdomain, HP36, is a single-domain protein that folds cooperatively. Its small size, simple topology and fast folding dynamics make it a favorite for experimental and theoretical studies.56–82 HP36 is part of the villin protein, which spans residues 42 to 76 in the full-length villin headpiece.56 It has the sequence MLSDED-FKAVFGMTRSAFANLPLWKQQNLKKEKGLF,56 with the second residue being L42. An additional Met is added to the N-terminus as in the previous experiments and is designated M41.56,69,72 The C-terminal of F76 is amidated. The native state of HP36 contains three helical segments that pack to form a hydrophobic core, as shown in Figure 1(a).56 Using IR spectroscopy, Brewer et al. studied the thermal unfolding transition of HP36 by monitoring its spectral changes in the temperature range from 7 to 86 °C, with both equilibrium and temperature-jump IR experiments.69,72 By isotope labeling A57, which is in helix 2, with 13C=18O as highlighted in the above sequence (in bold) and in Figure 1(a), they were able to identify an unfolding of helix 2 before the melting of the entire protein.72 Their experiments demonstrate the power of IR spectroscopy, in conjunction with isotope labeling, to probe protein folding and unfolding with residue-specific resolution.
FIG. 1.
(a) NMR structure of HP36 (Protein Date Bank ID 1VII). Representative structures of HP36 at (b) 7 °C, (c) 45 °C, (d) 77 °C, (e) 116 °C, and (f) 146 °C obtained from cluster analysis of the REMD simulations. α-helices are shown in blue and the unstructured parts are shown in gray. A57 is in helix 2 and is highlighted by the stick and ball representation.
Detailed interpretation of experimental IR spectra is facilitated by theoretical calculations. We have recently developed amide I frequency “maps” that allow the calculation of instantaneous frequencies directly from molecular dynamics (MD) simulations.83 These maps relate amide I frequencies to the local electric fields on the amide groups, and were developed for both protein backbone and Asn and Gln side chains. We have combined the frequency maps with established nearest-neighbor frequency shift (NNFS)84,85 and coupling schemes,84,86 and utilized a mixed quantum/classical approach to calculate protein IR spectra in the amide I region based on MD trajectories.83 This method provides a means to bridge spectroscopic experiments and molecular simulations and has the potential to interpret complex experimental spectra at the molecular level. It has been validated by applying to IR spectra of model peptides with various secondary structures,83 and has been shown to reproduce successfully the experimental linear and 2DIR spectra of islet amyloid polypeptide (IAPP),37,83,87,88 whose misfolding and aggregation are closely related to type 2 diabetes. We have also previously examined83 the temperature-dependent IR spectra of N-methylacetamide, a commonly used model compound to mimic the protein backbone peptide linkage.89–94 In the present work we focus on HP36 and study its equilibrium thermal unfolding transition using replica-exchange molecular dynamics (REMD) simulations and isotope-edited IR spectral calculations.
The paper is organized as follows. In Section II we describe the details of the REMD simulations and the IR calculations. In Section III, temperature-dependent IR spectra and spectral densities as well as the thermal unfolding curves of HP36 are presented and com- pared with experiments. Finally, conclusions are drawn in Section IV.
II. THEORETICAL METHODS FOR SPECTRAL CALCULATIONS
REMD simulations have been extensively used in the literature to enhance the conformational sampling of proteins.95–98 REMD simulations were performed using the GROMACS simulation package.99–103 56 replicas were used spanning the temperature range 7 – 171 °C. The temperatures were chosen to ensure reasonable exchange rates between replicas and were generated using the webserver at http://folding.bmc.uu.se/remd. The initial configuration of HP36 was taken from an NMR structure (Protein Data Bank ID 1VII),56 as shown in Figure 1(a). The structure was modified to include a C-terminal -NH2 cap, consistent with the IR experiments.69,72 HP36 was put in a cubic box and solvated with 4487 H2O molecules to ensure a distance of at least 1 nm from all box edges. The protein was modeled using the GROMOS96 53a6 force field104–106 and the simple point charge (SPC) model was adopted for water.107 The protein has a net charge of +3, and chloride ions were added to neutralize it. At each temperature the system was energy minimized, equilibrated in the NVT ensemble for 100 ps and then in the NPT ensemble for 10 ns. REMD simulations with constant temperature and a pressure of 1 bar were performed for 10 ns in each replica. Exchange moves between adjacent replicas were attempted every 2 ps. The Nosé-Hoover thermostat108,109 and the Parrinello-Rahman barostat110 were used for the temperature and pressure couplings. The LINCS algorithm was used to constrain all bonds, and particle-mesh Ewald sum was used to treat long-range electrostatic interactions.111,112 Configurations from the trajectories of the REMD simulations were saved every 10 fs for spectral calculations.
The theoretical protocol for calculating protein IR spectra in the amide I region has been described elsewhere.83 Briefly, within the mixed quantum/classical approach, the line shape I(ω) is given by113–115
| (1) |
where i and j index the amide I vibrational chromophores. The vibrational Hamiltonian (over ℏ) in the amide I region is κ(t), whose time propagation is described by the matrix F(t):
| (2) |
with Fij(0) = δij·. mi(t) is the projection of the transition dipole moment m⃗i(t) on the polarization unit vector of the excitation light ε̂, i.e. mi(t) = ε̂·m⃗i(t). Since in experiment the protein is not oriented, we average over all possible orientations by averaging over the three polarizations ε̂ = î, ĵ, k̂. The angular brackets in Eq. (1) indicate a classical equilibrium statistical mechanical average. T1 is the lifetime of the first excited state of an isolated amide I vibration, and the term e−t/2T1 is added phenomenologically to incorporate lifetime broadening. T1 is taken to be 600 fs.47
HP36 contains 36 chromophores in the backbone and 4 chromophores in the Asn and Gln side chains. In this case, κ(t) is a 40×40 matrix, whose diagonal elements are the local frequencies of each chromophore and the off-diagonal elements are the couplings between chromophores. The diagonal elements are obtained by applying the recently developed amide I backbone and side-chain frequency maps,83 with the 36 backbone chromophores corrected by NNFS.84,85 Note that since the C-terminal is amidated, the chromophore’s frequency for F76 is determined by the side-chain map. The off-diagonal terms are calculated using the nearest-neighbor coupling map84 or the transition dipole coupling (TDC) method86 according to the relative positions of the two chromophores.83 As κ(t) is real and symmetric, at any instant in time it can be diagonalized by an orthogonal transformation:
| (3) |
The diagonal matrix Λ contains the eigenvalues while P is composed of the corresponding eigenvectors. For a specific eigenvalue Λkk, its corresponding transition dipole is
| (4) |
In the inhomogeneous limit where the dynamics is sufficiently slow, we can simply average over an ensemble of static configurations.113 If we neglect lifetime broadening, Eq. (1) becomes113
| (5) |
which is called the spectral density (the frequency distribution weighted by the square of the transition dipole). In a variety of isolated-chromophore systems spectral densities have been demonstrated to capture the essential features of spectra.113,116–119 We will show that for HP36 the spectral density closely resembles the spectrum, and as it is much simpler to calculate, most analysis in this work is based on the spectral density instead of the spectrum. The A57-labeled IR spectrum and spectral density are calculated by shifting the local frequency of A57 by −70 cm−137,43–55,83 and keeping all other parameters the same as in the unlabeled calculations.
III. RESULTS AND DISCUSSIONS
REMD simulations were run for 10 ns at each of the 56 temperatures, giving a total simulation length of 560 ns. The average exchange probability per attempt is approximately 29%. Representative configurations of HP36 are extracted from the REMD simulations using cluster analysis. Figures 1(b) – (f) show the configurations most frequently visited at 5 different temperatures. It is clear that HP36 gradually unfolds as temperature increases. The REMD trajectories were used for spectral calculations at 15 temperatures. Note that at high temperature, the unfolded ensemble of HP36 has residual α-helical structure near the N-terminus and is to some extent compact, keeping part of the tertiary contact, in agreement with previous theoretical and experimental results.67,70,120 These partially folded configurations are sampled in the REMD simulations, and are included in the spectral calculations. However, for the purpose of probing the residual secondary and tertiary structures, IR absorption spectroscopy is not ideal as these structures are mostly disordered, and techniques with higher structural sensitivity are required.
Theoretical IR spectra of the unlabeled and A57-labeled HP36 at 7 °C are shown in Figure 2. As expected, incorporating a single label slightly decreases the intensity of the main peak, and increases the intensity roughly 70 cm−1 to the red. The main peaks in both spectra are at about 1636 cm−1, close to the experimental value of 1645 cm−1,69,72 which is consistent with the primarily α-helical structure of HP36.23 The A57 isotope peak is at 1570 cm−1, in good agreement with the experimental value of 1572 cm−1.72 Although the theoretical spectra appear to be less symmetric compared to the experimental amide I bands of HP36 (private communication72) and its structural analog HP35 (which is HP36 without the N-terminal Met),121 they correctly capture the spectral feature around 1670 cm−1. Spectral analysis reveals that the main peak around 1636 cm−1 is due to the unlabeled residues in the three helices, and the shoulder around 1670 cm−1 is mainly due to the rest of the unlabeled residues that are unstructured. Residues M53 and L61 are shielded from the surrounding water molecules by the nearby hydrophobic side chains, and as a result they have very high local frequencies and lead to a minor peak around 1700 cm−1.
FIG. 2.
Calculated IR spectra of unlabeled and A57-labeled HP36 at 7 °C
The spectral density of the A57-labeled HP36 at 7 °C is calculated and compared with the theoretical IR spectrum in Figure 3. The spectral density is a little blue shifted and broader, but overall it captures the essential features in the spectrum very well, and will be used in the following analysis. As the spectral density is the spectrum in the inhomogeneous limit, the good agreement between them indicates that for HP36 at 7 °C, the IR spectral features are mainly due to static structural distributions rather than dynamics.
FIG. 3.
Calculated IR spectrum and spectral density of A57-labeled HP36 at 7 °C. The spectrum and the spectral density are normalized by peak height.
HP36 contains aspartic acid (D) and glutamic acid (E) residues, whose side-chain COO− asymmetric vibrations absorb near 1600 cm−1 and overlap somewhat with the labeled region.74 We have not included these contributions in our calculations. Note that the D and E peaks do not change much with temperature.69 In this work we focus on difference spectra, either between unlabeled and A57-labeled HP36 or between different temperatures, in which cases we think the neglect of the D and E contributions is acceptable.
The difference spectrum calculated by subtracting the A57-labeled spectral density from the unlabeled one is shown in Figure 4. The calculation is based on the REMD trajectory at 27 °C, the simulation temperature closest to the experimental condition.72 As discussed above, isotope labeling A57 leads to an increase in intensity in the isotope region (1550 – 1600 cm−1) centered at around 1570 cm−1, due to the 13C=18O stretching mode, and a decrease in intensity in the main peak region (1620 – 1660 cm−1) centered at 1648 cm−1. Our results agree well with experiment as shown in Figure 4.72 Note that in experiment the A57-labeled and unlabeled spectra were measured at slightly different pH values, leading to two minor peaks at 1594 and 1704 cm−1.72 The comparison between theory and experiment verifies that −70 cm−1 is a reasonable isotope shift.
FIG. 4.
The difference spectrum generated by subtracting the A57-labeled spectrum from the unlabeled spectrum. The theory result is for the spectral density at 27 °C, and experiment is at 25 °C.72 The theoretical and experimental difference spectra are normalized by their values at the peak near 1650 cm−1.
Temperature-dependent difference spectra of A57-labeled HP36 are calculated by subtracting the spectral density at 7 °C from the higher temperature ones. Figure 5(b) shows the results up to 146 °C, which are in qualitative agreement with the experimental data (Figure 5(a)).72 From Figures 1(b) – (f) we saw that the thermal unfolding process of HP36 involves a conformational change from a folded state containing three α-helices to an unfolded, mainly unstructured state. It is well known that IR spectra of random coils peak at higher frequencies than those of α-helices.50 Moreover, it has been shown that increasing temperature leads to the weakening of hydrogen bonds, which consequently leads to a peak shift to higher frequencies in the amide I region.122 One would therefore expect a blue shift in the spectra as thermal unfolding occurs. Indeed, two pairs of changes are most prominent in Figure 5(b). The first pair occurs in the main peak region: a negative feature in the area from 1600 – 1650 cm−1 and a positive feature in the area from 1650 – 1700 cm−1. These are the spectral manifestations of the loss of secondary and tertiary structure and the increase of disordered regions of the whole protein, respectively. Similarly, the other pair appears in the isotope region: a negative feature from 1550 – 1580 cm−1 and a positive feature from 1580 – 1600 cm−1. The isotope region probes the A57 behavior and indicates that the part of HP36 that involves A57 loses secondary and tertiary structure and is more disordered in the unfolding process. All features become more prominent with increasing temperatures. In Figure 5(a) multiple overlapping bands appear in the 1600 – 1700 regions, probably arising from chromophores in different solvation conditions.69,72 These features are not visible in Figure 5(b), partly due to the fact that spectral densities tend to show averaged features compared to IR spectra. In the IR spectra, which include the motional narrowing effect, we observe a hint of multiple peaks (data not shown), but our theoretical modeling cannot fully reproduce the experimental features. We will leave this problem to future studies.
FIG. 5.
Temperature-dependent difference spectra of A57-labeled HP36. Difference spectrum are calculated by subtracting the spectrum at 7 °C from the higher-temperature spectra. (a) Experimental spectra from 7 °C to 86 °C in ~ 10 °C increments (reprinted with permission from Biochemistry 46, 3279 (2007); copyright 2007 American Chemical Society).72 (b) Theoretical data for the spectral density from 7 °C to 146 °C in ~ 20 °C increment. The absolute intensities in experiment and theory are in arbitrary units, and therefore are not directly comparable.
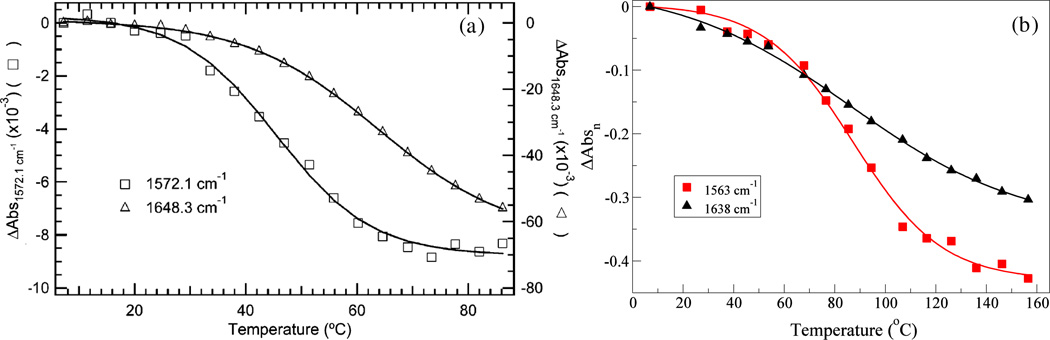
In Figure 5(b), the negative peaks in the labeled and main regions have frequencies close to 1563 and 1638 cm−1, respectively. The former frequency represents the local behavior of A57, and the latter reflects the whole protein. Choosing these two frequencies, we monitor the intensities ΔAbs as a function of temperature to obtain their melting profiles. Since only one of the 40 chromophores is labeled, the intensity of the isotope peak is about 1/40 that of the main peak. We have normalized ΔAbs at frequencies 1563 and 1638 cm−1, respectively, by their absolute intensities at 7 °C to put them on the same scale for better comparison. The normalized intensity is denoted as ΔAbsn. As depicted in Figure 6(b), ΔAbsn decreases sigmoidally with temperature. We fit the temperature-dependent ΔAbsn data to a sigmoidal function subjected to the constraint that :
| (6) |
where T is in Celsius and a1, a2 and a3 are fitting parameters. ΔAbsn has the asymptote . The fitted curves are also shown in Figure 6(b). The unfolding curve at frequency 1563 cm−1 tends to level off at high temperature, while the curve for frequency 1638 cm−1 keeps decreasing in the whole temperature range we study. To quantify the unfolding transition we have calculated the midpoint of the thermal unfolding transition Tm, which is defined as the temperature at which ΔAbsn reaches 0.5*ΔAbsn∞. Tm for the two frequencies 1563 and 1638 cm−1 are 87 and 93 °C, respectively. Experimentally, frequencies of 1572 and 1648 cm−1 were used to represent the local and global unfolding transitions of HP36 (Figure 6(a)).72 The experimental Tm for frequencies 1572 and 1648 cm−1 are about 46 and 64 °C, respectively.72 Although our melting temperatures are higher than those in experiment, the theoretical unfolding curves are qualitatively in agreement with experiment. We attribute the discrepancies in melting temperature to the inaccuracy of the force field that we are using. The spectra calculations rely on the configurations generated from MD simulations, which are closely related to the force field that defines molecular interactions. As force fields are usually developed for simulations at room temperature, they might predict a different protein unfolding temperature compared to experiment. For example, for the unfolding of the entire HP36 protein, the IR experiment shows a Tm of 64 °C, while the GROMOS96 53a6 (used in this work) and the ECEPP/2 force fields predict values of 93 °C and 227 °C,123 respectively. A previous study on HP35 used the AMBER FF03 force field and obtained a Tm of 66 °C, in better agreement with experiment.124
FIG. 6.
Thermal unfolding transition of A57-labeled HP36 (a) experimentally72 (reprinted with permission from Biochemistry 46, 3279 (2007); copyright 2007 American Chemical Society) and (b) theoretically. The theoretical results are normalized by the absolute peak intensities at 1563 and 1638 cm−1 at 7 °C, respectively. Solid lines are the sigmoidal fit curves.
The Tm values in theory and experiment imply that helix 2, in which A57 resides, loses its helical structure before the global unfolding occurs. For the purpose of quantifying the unfolding of the three helices, we calculate the temperature-dependent ΔAbsn for every backbone chromophore, assuming that it is 13C=18O labeled. Helices 1, 2 and 3 are composed of residues 44 – 48, 55 – 58 and 62 – 71, respectively. Residues that belong to the same helix have similar unfolding behavior, and thus for each helix we average over ΔAbsn for all its residues, and fit the resulting data to the sigmoidal function in Eq. (6). We define the percentage decay as ΔAbsn/ΔAbsn∞, and plot it as a function of temperature for the three helices in Figure 7. Tm for helices 1, 2 and 3 are 99, 86 and 92 °C, respectively. From the percentage decay analysis, the thermal unfolding of helix 2 occurs prior to the other two helices. The similarity in Tm’s indicates that the unfolding of helix 2 is well represented by A57, and that the decrease of the main peak reflects the average behavior of the protein.
FIG. 7.
Theoretical thermal unfolding curves for the three helices in HP36. Data for each helix are obtained by averaging over residues belonging to the helix, and are normalized to obtain the percentage decay ΔAbsn /ΔAbsn∞ values. Solid lines are the sigmoidal fit curves.
IV. CONCLUSIONS
In this paper, we presented a combined REMD simulation and theoretical IR spectral study of the equilibrium thermal unfolding processes of HP36. The theoretical IR spectra and spectral densities agree quite well with experiments, providing further support for our calculation protocol.83 With the help of isotope labeling, the thermal unfolding transition of HP36 is followed globally and locally by the decrease of ΔAbs for the main peak and the A57 isotope peak. Theoretical spectral analysis demonstrates that helix 2 loses its secondary structure prior to the global unfolding transition, in agreement with experiment.72 Although the simulation melting temperatures are not in quantitative agreement with those from experiment,72 and the difference in the melting temperatures deduced from the labeled and unlabeled peaks in theory (6 °C) is smaller than in experiment (18 °C), the theoretical spectra do correctly reflect the unfolding conformational changes in the simulation. That is to say, when the helices unfold the spectra shift correspondingly. Discrepancies between theory and experiment are most likely primarily due to inaccuracies of the force field, which is designed to work best close to room temperature (and was applied here at somewhat elevated temperatures), although we cannot rule out contributions from deficiencies in our spectroscopic maps.83
This work provides one example of utilizing the theoretical strategy we have developed for protein IR calculations for the study of protein unfolding processes. Linear and 2DIR spectroscopy experiments, in combination with the isotope-labeling technique, have the potential to probe protein folding, unfolding and misfolding processes in situ with residue-specific resolution. In a complementary vein, computer simulations, with their increasing power, will help elucidate the structural and dynamical details of such processes. Ours and other theoretical frameworks36,94,125–127 that allow for the calculation of isotope-edited IR spectra from MD trajectories bridge simulations and experiments, and will aid in unraveling the pathways and mechanisms of protein folding, unfolding and misfolding.
ACKNOWLEDGMENTS
The authors are grateful to Dr. Scott H. Brewer for kindly providing the experimental data. This work was supported in part by a grant from the NSF (CHE-0832584) to JLS. JLS also thanks NSF for support of this work through grant CHE-1058752, and NIH, through 1R01DK088184-01A1. This research was also supported in part by the National Science Foundation through XSEDE resources provided by the XSEDE Science Gateways program.
References
- 1.Nelson DL, Cox MM. Lehninger Principles of Biochemistry. New York: W. H. Freeman and Company; 2005. [Google Scholar]
- 2.Dobson CM. Trends. Biochem. Sci. 1999;24:329–332. doi: 10.1016/s0968-0004(99)01445-0. [DOI] [PubMed] [Google Scholar]
- 3.Dobson CM. Nature. 2003;426:884–890. doi: 10.1038/nature02261. [DOI] [PubMed] [Google Scholar]
- 4.Chiti F, Dobson CM. Annu. Rev. Biochem. 2006;75:333–366. doi: 10.1146/annurev.biochem.75.101304.123901. [DOI] [PubMed] [Google Scholar]
- 5.Thirumalai D, Reddy G, Straub JE. Acc. Chem. Res. 2012;45:83–92. doi: 10.1021/ar2000869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dill KA. Biochemistry. 1990;29:7133–7155. doi: 10.1021/bi00483a001. [DOI] [PubMed] [Google Scholar]
- 7.Straub JE, Thirumalai D. Proc. Natl. Acad. Sci. USA. 1993;90:809–813. doi: 10.1073/pnas.90.3.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Šali A, Shakhnovich E, Karplus M. Nature. 1994;369:248–251. doi: 10.1038/369248a0. [DOI] [PubMed] [Google Scholar]
- 9.Onuchic JN, Luthey-Schulten Z, Wolynes PG. Annu. Rev. Phys. Chem. 1997;48:545–600. doi: 10.1146/annurev.physchem.48.1.545. [DOI] [PubMed] [Google Scholar]
- 10.Hummer G, Garde S, García AE, Paulaitis ME, Pratt LR. J. Phys. Chem. B. 1998;102:10469–10482. [Google Scholar]
- 11.Shea J-E, Brooks CL. Annu. Rev. Phys. Chem. 2001;52:499–535. doi: 10.1146/annurev.physchem.52.1.499. [DOI] [PubMed] [Google Scholar]
- 12.Dyson HJ, Wright PE. Chem. Rev. 2004;104:3607–3622. doi: 10.1021/cr030403s. [DOI] [PubMed] [Google Scholar]
- 13.Freddolino PL, Harrison CB, Liu Y, Schulten K. Nature Physics. 2010;6:751–758. doi: 10.1038/nphys1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Buchner GS, Murphy RD, Buchete N-V, Kubelka J. Biochim. Biophys. Acta. 2011;1814:1001–1020. doi: 10.1016/j.bbapap.2010.09.013. [DOI] [PubMed] [Google Scholar]
- 15.Bowman GR, Voelz VA, Pande VS. Curr. Opin. Struct. Biol. 2011;21:4–11. doi: 10.1016/j.sbi.2010.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.England JL, Haran G. Annu. Rev. Phys. Chem. 2011;62:257–277. doi: 10.1146/annurev-physchem-032210-103531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Serrano AL, Waegele MM, Gai F. Protein Sci. 2012;21:157–170. doi: 10.1002/pro.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Culik RM, Jo H, DeGrado WF, Gai FJ. Am. Chem. Soc. 2012;134:8026–8029. doi: 10.1021/ja301681v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Susi H, Byler DM. Methods Enzymol. 1986;130:290–311. doi: 10.1016/0076-6879(86)30015-6. [DOI] [PubMed] [Google Scholar]
- 20.Haris PI, Chapman D. Trends. Biochem. Sci. 1992;17:328–333. doi: 10.1016/0968-0004(92)90305-s. [DOI] [PubMed] [Google Scholar]
- 21.Surewicz WK, Mantsch HH, Chapman D. Biochemistry. 1993;32:389–394. doi: 10.1021/bi00053a001. [DOI] [PubMed] [Google Scholar]
- 22.Zanni MT, Hochstrasser RM. Curr. Opin. Struct. Biol. 2001;11:516–522. doi: 10.1016/s0959-440x(00)00243-8. [DOI] [PubMed] [Google Scholar]
- 23.Barth A, Zscherp C. Quart. Rev. Biophys. 2002;35:369–430. doi: 10.1017/s0033583502003815. [DOI] [PubMed] [Google Scholar]
- 24.Hochstrasser RM. Proc. Natl. Acad. Sci. USA. 2007;104:14190–14196. doi: 10.1073/pnas.0704079104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Park S, Kwak K, Fayer MD. Laser Phys. Lett. 2007;4:704–718. [Google Scholar]
- 26.Cho M. Chem. Rev. 2008;108:1331–1418. doi: 10.1021/cr078377b. [DOI] [PubMed] [Google Scholar]
- 27.Ganim Z, Chung HS, Smith AW, DeFlores LP, Jones KC, Tokmakoff A. Acc. Chem. Res. 2008;41:432–441. doi: 10.1021/ar700188n. [DOI] [PubMed] [Google Scholar]
- 28.Strasfeld DB, Ling YL, Shim S-H, Zanni MT. J. Am. Chem. Soc. 2008;130:6698–6699. doi: 10.1021/ja801483n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sagle LB, Zimmermann J, Dawson PE, Romesberg FE. J. Am. Chem. Soc. 2006;128:14232–14233. doi: 10.1021/ja065179d. [DOI] [PubMed] [Google Scholar]
- 30.Zimmermann J, Thielges MC, Yu W, Dawson PE, Romesberg FE. J. Phys. Chem. Lett. 2011;2:412–416. [Google Scholar]
- 31.Nagarajan S, Taskent-Sezgin H, Parul D, Carrico I, Raleigh DP, Dyer RB. J. Am. Chem. Soc. 2011;133:20335–20340. doi: 10.1021/ja2071362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Krimm S, Bandekar J. Adv. Protein Chem. 1986;38:181–364. doi: 10.1016/s0065-3233(08)60528-8. [DOI] [PubMed] [Google Scholar]
- 33.Middleton CT, Buchanan LE, Dunkelberger EB, Zanni MT. J. Phys. Chem. Lett. 2011;2:2357–2361. doi: 10.1021/jz201024m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hamm P, Zanni M. Concepts and methods of 2D infrared spectroscopy. Cambridge, UK: Cambridge University Press; 2011. [Google Scholar]
- 35.Jones KC, Ganim Z, Peng CS, Tokmakoff AJ. Opt. Soc. Am. B. 2012;29:118–129. [Google Scholar]
- 36.Zhuang W, Cui RZ, Silva D-A, Huang XJ. Phys. Chem. B. 2011;115:5415–5424. doi: 10.1021/jp109592b. [DOI] [PubMed] [Google Scholar]
- 37.Wang L, Middleton CT, Singh S, Reddy AS, Woys AM, Strasfeld DB, Marek P, Raleigh DP, de Pablo JJ, Zanni MT, Skinner JL. J. Am. Chem. Soc. 2011;133:16062–16071. doi: 10.1021/ja204035k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chung JK, Thielges MC, Bowman SEJ, Bren KL, Fayer MD. J. Am. Chem. Soc. 2011;133:6681–6691. doi: 10.1021/ja111009s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lessing J, Roy S, Reppert M, Baer M, Marx D, Jansen TLC, Knoester J, Tokmakoff A. J. Am. Chem. Soc. 2012;134:5032–5035. doi: 10.1021/ja2114135. [DOI] [PubMed] [Google Scholar]
- 40.Falvo C, Zhuang W, Kim YS, Axelsen PH, Hochstrasser RM. J. Phys. Chem. B. 2012;116:3322–3330. doi: 10.1021/jp2096423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Moran SD, Woys AM, Buchanan LE, Bixby E, Decatur SM, Zanni MT. Proc. Natl. Acad. Sci. USA. 2012;109:3329–3334. doi: 10.1073/pnas.1117704109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Middleton CT, Marek P, Cao P, Chiu C, Singh S, Woys AM, de Pablo JJ, Raleigh DP, Zanni MT. Nature Chemistry. 2012;4:355–360. doi: 10.1038/nchem.1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Torres J, Adams PD, Arkin IT. J. Mol. Biol. 2000;300:677–685. doi: 10.1006/jmbi.2000.3885. [DOI] [PubMed] [Google Scholar]
- 44.Torres J, Kukol A, Goodman JM, Arkin IT. Biopolymers. 2001;59:396–401. doi: 10.1002/1097-0282(200111)59:6<396::AID-BIP1044>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 45.Fang C, Wang J, Charnley AK, Barber-Armstrong W, Smith AB, Decatur SM, Hochstrasser RM. Chem. Phys. Lett. 2003;382:586–592. [Google Scholar]
- 46.Fang C, Wang J, Kim YS, Charnley AK, Barber-Armstrong W, Smith AB, Decatur SM, Hochstrasser RM. J. Phys. Chem. B. 2004;108:10415–10427. [Google Scholar]
- 47.Mukherjee P, Krummel AT, Fulmer EC, Kass I, Arkin IT, Zanni MT. J. Chem. Phys. 2004;120:10215–10224. doi: 10.1063/1.1718332. [DOI] [PubMed] [Google Scholar]
- 48.Fang C, Hochstrasser RM. J. Phys. Chem. B. 2005;109:18652–18663. doi: 10.1021/jp052525p. [DOI] [PubMed] [Google Scholar]
- 49.Arkin IT. Curr. Opin. Chem. Biol. 2006;10:394–401. doi: 10.1016/j.cbpa.2006.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Decatur SM. Acc. Chem. Res. 2006;39:169–175. doi: 10.1021/ar050135f. [DOI] [PubMed] [Google Scholar]
- 51.Mukherjee P, Kass I, Arkin I, Zanni MT. Proc. Natl. Acad. Sci. USA. 2006;103:3528–3533. doi: 10.1073/pnas.0508833103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mukherjee P, Kass I, Arkin IT, Zanni MT. J. Phys. Chem. B. 2006;110:24740–24749. doi: 10.1021/jp0640530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Shim S-H, Gupta R, Ling YL, Strasfeld DB, Raleigh DP, Zanni MT. Proc. Natl. Acad. Sci. USA. 2009;106:6614–6619. doi: 10.1073/pnas.0805957106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Manor J, Mukherjee P, Lin Y-S, Leonov H, Skinner JL, Zanni MT, Arkin IT. Structure. 2009;17:247–254. doi: 10.1016/j.str.2008.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Woys AM, Lin Y-S, Reddy AS, Xiong W, de Pablo JJ, Skinner JL, Zanni MT. J. Am. Chem. Soc. 2010;132:2832–2838. doi: 10.1021/ja9101776. [DOI] [PubMed] [Google Scholar]
- 56.McKnight CJ, Matsudaira PT, Kim PS. Nat. Struct. Biol. 1997;4:180–184. doi: 10.1038/nsb0397-180. [DOI] [PubMed] [Google Scholar]
- 57.Duan Y, Wang L, Kollman PA. Proc. Natl. Acad. Sci. USA. 1998;95:9897–9902. doi: 10.1073/pnas.95.17.9897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Duan Y, Kollman PA. Science. 1998;282:740–744. doi: 10.1126/science.282.5389.740. [DOI] [PubMed] [Google Scholar]
- 59.Vugmeyster L, Trott O, McKnight CJ, Raleigh DP, Palmer AG. J. Mol. Biol. 2002;320:841–854. doi: 10.1016/S0022-2836(02)00537-5. [DOI] [PubMed] [Google Scholar]
- 60.Zagrovic B, Snow CD, Khaliq S, Shirts MR, Pande VS. J. Mol. Biol. 2002;323:153–164. doi: 10.1016/s0022-2836(02)00888-4. [DOI] [PubMed] [Google Scholar]
- 61.van der Spoel D, Lindahl E. J. Phys. Chem. B. 2003;107:11178–11187. [Google Scholar]
- 62.Lin C-Y, Hu C-K, Hansmann UHE. Proteins. 2003;52:436–445. doi: 10.1002/prot.10351. [DOI] [PubMed] [Google Scholar]
- 63.Wang M, Tang Y, Sato S, Vugmeyster L, McKnight CJ, Raleigh DP. J. Am. Chem. Soc. 2003;125:6032–6033. doi: 10.1021/ja028752b. [DOI] [PubMed] [Google Scholar]
- 64.Kubelka J, Eaton WA, Hofrichter J. J. Mol. Biol. 2003;329:625–630. doi: 10.1016/s0022-2836(03)00519-9. [DOI] [PubMed] [Google Scholar]
- 65.Jang S, Kim E, Shin S, Pak Y. J. Am. Chem. Soc. 2003;125:14841–14846. doi: 10.1021/ja034701i. [DOI] [PubMed] [Google Scholar]
- 66.Ripoll DR, Vila JA, Scheraga HA. J. Mol. Biol. 2004;339:915–925. doi: 10.1016/j.jmb.2004.04.002. [DOI] [PubMed] [Google Scholar]
- 67.Tang Y, Rigotti DJ, Fairman R, Raleigh DP. Biochemistry. 2004;43:3264–3272. doi: 10.1021/bi035652p. [DOI] [PubMed] [Google Scholar]
- 68.Chiu TK, Kubelka J, Herbst-Irmer R, Eaton WA, Hofrichter J, Davies DR. Proc. Natl. Acad. Sci. USA. 2005;102:7517–7522. doi: 10.1073/pnas.0502495102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Brewer SH, Vu DM, Tang Y, Li Y, Franzen S, Raleigh DP, Dyer RB. Proc. Natl. Acad. Sci. USA. 2005;102:16662–16667. doi: 10.1073/pnas.0505432102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tang Y, Goger MJ, Raleigh DP. Biochemistry. 2006;45:6940–6946. doi: 10.1021/bi052484n. [DOI] [PubMed] [Google Scholar]
- 71.Tang Y, Grey MJ, McKnight J, Palmer AG, Raleigh DP. J. Mol. Biol. 2006;355:1066–1077. doi: 10.1016/j.jmb.2005.10.066. [DOI] [PubMed] [Google Scholar]
- 72.Brewer SH, Song B, Raleigh DP, Dyer RB. Biochemistry. 2007;46:3279–3285. doi: 10.1021/bi602372y. [DOI] [PubMed] [Google Scholar]
- 73.Bi Y, Cho J-H, Kim E-Y, Shan B, Schindelin H, Raleigh DP. Biochemistry. 2007;46:7497–7505. doi: 10.1021/bi6026314. [DOI] [PubMed] [Google Scholar]
- 74.Bagchi S, Falvo C, Mukamel S, Hochstrasser RM. J. Phys. Chem. B. 2009;113:11260–11273. doi: 10.1021/jp900245s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Meng W, Shan B, Tang Y, Raleigh DP. Protein Sci. 2009;18:1692–1701. doi: 10.1002/pro.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Xiao S, Bi Y, Shan B, Raleigh DP. Biochemistry. 2009;48:4607–4616. doi: 10.1021/bi8021763. [DOI] [PubMed] [Google Scholar]
- 77.Wei H, Yang L, Gao YQ. J. Phys. Chem. B. 2010;114:11820–11826. doi: 10.1021/jp103770y. [DOI] [PubMed] [Google Scholar]
- 78.Chung JK, Thielges MC, Fayer MD. Proc. Natl. Acad. Sci. USA. 2011;108:3578–3583. doi: 10.1073/pnas.1100587108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Beauchamp KA, Ensign DL, Das R, Pande VS. Proc. Natl. Acad. Sci. USA. 2011;108:12734–12739. doi: 10.1073/pnas.1010880108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Urbanek DC, Vorobyev DY, Serrano AL, Gai F, Hochstrasser RM. J. Phys. Chem. Lett. 2010;1:3311–3315. doi: 10.1021/jz101367d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Roy S, Bagchi B. J. Phys. Chem. B. 2012;116:2958–2968. doi: 10.1021/jp209437j. [DOI] [PubMed] [Google Scholar]
- 82.Lv C, Tan C, Qin M, Zou D, Cao Y, Wang W. Biophys. J. 2012;102:1944–1951. doi: 10.1016/j.bpj.2012.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Wang L, Middleton CT, Zanni MT, Skinner JL. J. Phys. Chem. B. 2011;115:3713–3724. doi: 10.1021/jp200745r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Jansen TLC, Dijkstra AG, Watson TM, Hirst JD, Knoester JJ. Chem. Phys. 2006;125 doi: 10.1063/1.2218516. 044312. [DOI] [PubMed] [Google Scholar]
- 85.Jansen TLC. J. Chem. Phys. 2012;136:209901. [Google Scholar]
- 86.Torii H, Tasumi M. J. Raman Spectrosc. 1998;29:81–86. [Google Scholar]
- 87.Reddy AS, Wang L, Lin Y-S, Ling Y, Chopra M, Zanni MT, Skinner JL, de Pablo JJ. Biophys. J. 2010;98:443–451. doi: 10.1016/j.bpj.2009.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Reddy AS, Wang L, Singh S, Ling YL, Buchanan L, Zanni MT, Skinner JL, de Pablo JJ. Biophys. J. 2010;99:2208–2216. doi: 10.1016/j.bpj.2010.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ham S, Kim J-H, Lee H, Cho M. J. Chem. Phys. 2003;118:3491–3498. [Google Scholar]
- 90.Schmidt JR, Corcelli SA, Skinner JL. J. Chem. Phys. 2004;121:8887–8896. doi: 10.1063/1.1791632. [DOI] [PubMed] [Google Scholar]
- 91.Hayashi T, Zhuang W, Mukamel S. J. Phys. Chem. A. 2005;109:9747–9759. doi: 10.1021/jp052324l. [DOI] [PubMed] [Google Scholar]
- 92.Jansen TLC, Knoester J. J. Chem. Phys. 2006;124 044502. [Google Scholar]
- 93.Lin Y-S, Shorb JM, Mukherjee P, Zanni MT, Skinner JL. J. Phys. Chem. B. 2009;113:592–602. doi: 10.1021/jp807528q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Maekawa H, Ge N-H. J. Phys. Chem. B. 2010;114:1434–1446. doi: 10.1021/jp908695g. [DOI] [PubMed] [Google Scholar]
- 95.Sugita Y, Okamoto Y. Chem. Phys. Lett. 1999;314:141–151. [Google Scholar]
- 96.Sanbonmatsu KY, García AE. Proteins. 2002;46:225–234. doi: 10.1002/prot.1167. [DOI] [PubMed] [Google Scholar]
- 97.Rathore N, Chopra M, de Pablo JJ. J. Chem. Phys. 2005;122 doi: 10.1063/1.1831273. 024111. [DOI] [PubMed] [Google Scholar]
- 98.Chebaro Y, Dong X, Laghaei R, Derreumaux P, Mousseau N. J. Phys. Chem. B. 2009;113:267–274. doi: 10.1021/jp805309e. [DOI] [PubMed] [Google Scholar]
- 99.Bekker H, Berendsen HJC, Dijkstra EJ, Achterop S, van Drunen R, van der Spoel D, Sijbers A, Keegstra H, Reitsma B, Renardus MKR. In: Physics Computing 92. Groot RA, Nadrchal J, editors. Singapore: World Scientific; 1993. [Google Scholar]
- 100.Berendsen HJC, van der Spoel D, van Drunen R. Comput. Phys. Commun. 1995;91:43–56. [Google Scholar]
- 101.Lindahl E, Hess B, van der Spoel D. J. Mol. Model. 2001;7:306–317. [Google Scholar]
- 102.van der Spoel D, Lindahl E, Hess B, van Buuren AR, Apol E, Meulenhoff PJ, Tieleman DP, Sijbers ALTM, Feenstra KA, van Drunen R, Berendsen HJC. Gromacs User Manual version 3.3. 2005 www.gromacs.org. [Google Scholar]
- 103.van der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJC. J. Comput. Chem. 2005;26:1701–1718. doi: 10.1002/jcc.20291. [DOI] [PubMed] [Google Scholar]
- 104.van Gunsteren WF, Billeter SR, Eising AA, Hünenberger PH, Krüger P, Mark AE, Scott WRP, Tironi IG. Biomolecular Simulation: The GROMOS96 manual and user guide. Zürich, Switzerland: Hochschuleverlag AG an der ETH Zürich; 1996. [Google Scholar]
- 105.Scott WRP, Hünenberger PH, Tironi IG, Mark AE, Billeter SR, Fennen J, Torda AE, Huber T, Krüger P, van Gunsteren WF. J. Phys. Chem. A. 1999;103:3596–3607. [Google Scholar]
- 106.Oostenbrink C, Villa A, Mark AE, van Gunsteren WF. J. Comput. Chem. 2004;25:1656–1676. doi: 10.1002/jcc.20090. [DOI] [PubMed] [Google Scholar]
- 107.Berendsen HJC, Postma JPM, van Gunsteren WF, Hermans J. In: Intermolecular Forces. Pullman B, editor. Dordrecht: Reidel; 1981. [Google Scholar]
- 108.Nosé S. J. Chem. Phys. 1984;81:511–519. [Google Scholar]
- 109.Hoover WG. Phys. Rev. A. 1985;31:1695–1697. doi: 10.1103/physreva.31.1695. [DOI] [PubMed] [Google Scholar]
- 110.Parrinello M, Rahman A. J. Appl. Phys. 1981;52:7182–7190. [Google Scholar]
- 111.Darden T, York D, Pedersen L. J. Chem. Phys. 1993;98:10089–10092. [Google Scholar]
- 112.Essmann U, Perera L, Berkowitz ML, Darden T, Lee H, Pedersen LG. J. Chem. Phys. 1995;103:8577–8593. [Google Scholar]
- 113.Auer BM, Skinner JL. J. Chem. Phys. 2007;127:104105. doi: 10.1063/1.2766943. [DOI] [PubMed] [Google Scholar]
- 114.Auer BM, Skinner JL. J. Chem. Phys. 2008;128:224511. doi: 10.1063/1.2925258. [DOI] [PubMed] [Google Scholar]
- 115.Mukamel S. Principles of Nonlinear Optical Spectroscopy. Oxford: New York; 1995. [Google Scholar]
- 116.Auer BM, Skinner JL. J. Chem. Phys. 2008;129:214705. doi: 10.1063/1.3012568. [DOI] [PubMed] [Google Scholar]
- 117.Pieniazek PA, Tainter CJ, Skinner JL. J. Am. Chem. Soc. 2011;133:10360–10363. doi: 10.1021/ja2026695. [DOI] [PubMed] [Google Scholar]
- 118.Pieniazek PA, Tainter CJ, Skinner JL. J. Chem. Phys. 2011;135 doi: 10.1063/1.3613623. 044701. [DOI] [PubMed] [Google Scholar]
- 119.Skinner JL, Pieniazek PA, Gruenbaum SM. Acc. Chem. Res. 2012;45:93–100. doi: 10.1021/ar200122a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wickstrom L, Okur A, Song K, Hornak V, Raleigh DP, Simmerling CL. J. Mol. Biol. 2006;360:1094–1107. doi: 10.1016/j.jmb.2006.04.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bunagan MR, Gao J, Kelly JW, Gai F. J. Am. Chem. Soc. 2009;131:7470–7476. doi: 10.1021/ja901860f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mukherjee S, Chowdhury P, Gai F. J. Phys. Chem. B. 2007;111:4596–4602. doi: 10.1021/jp0689060. [DOI] [PubMed] [Google Scholar]
- 123.Trebst S, Troyer M, Hansmann UHE. J. Chem. Phys. 2006;124:174903. doi: 10.1063/1.2186639. [DOI] [PubMed] [Google Scholar]
- 124.Lei H, Wu C, Liu H, Duan Y. Proc. Natl. Acad. Sci. USA. 2007;104:4925–4930. doi: 10.1073/pnas.0608432104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zhuang W, Abramavicius D, Hayashi T, Mukamel S. J. Phys. Chem. B. 2006;110:3362–3374. doi: 10.1021/jp055813u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zhuang W, Hayashi T, Mukamel S. Angew. Chem. Int. Ed. Engl. 2009;48:3750–3781. doi: 10.1002/anie.200802644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Smith AW, Lessing J, Ganim Z, Peng CS, Tokmakoff A, Roy S, Jansen TLC, Knoester J. J. Phys. Chem. B. 2010;114:10913–10924. doi: 10.1021/jp104017h. [DOI] [PubMed] [Google Scholar]