Abstract
Mammalian brain development is initiated in utero and internal and external environmental signals can affect this process all the way until adulthood. Recent observations suggest that one such external cue is the indigenous microbiota which has been shown to affect developmental programming of the brain. This may have consequences for brain maturation and function that impact on cognitive functions later in life. This review discusses these recent findings from a developmental perspective.
Keywords: behavior, brain development, gut microbiota, microbiota-gut-brain axis, probiotics
Introduction
Gut Microbiota
The gut contains an immense and complex microbial ecosystem, comprising a myriad of bacteria, of which most are strict anaerobes.1 The term ‘gut microbiome’ is used to describe the constituents of the microbiota, ranging from its bacterial genes to their proteins and metabolites.2 While the gut microbiome is assumed to be similar between family members,3 recent data suggest that the human microbiome may be subgrouped into ‘enterotypes’ depending on bacterial prevalence.4 Gut microbiota serve the host by protecting against pathogens, participating in the intake of dietary nutrients, metabolizing certain drugs and carcinogens, and influencing the absorption and distribution of fat.5 The influence of the microbiota extends beyond the gastrointestinal (GI) tract; contributing to, for example, pain perception in the skin6 and fat deposition in the liver.7,8 Disruption of the symbiotic relationship between the microbiota and the GI tract9 perturbs host functions and, in some cases, will contribute to a “leaky” gut-immune barrier10 leading to malfunction and illness such as inflammatory bowel disease (IBD) and Clostridium difficile colitis.11
Although it is known that a bi-directional communication between the GI tract and the central nervous system (CNS) occurs, less is known on how microbiota can influence this gut-to-brain communication. Recent data, however, have begun to shed some light on the ability of microbiota to influence behavior in mice.12-15 The mechanisms are essentially unknown but are currently subject to intense investigation.
Gut-brain axis
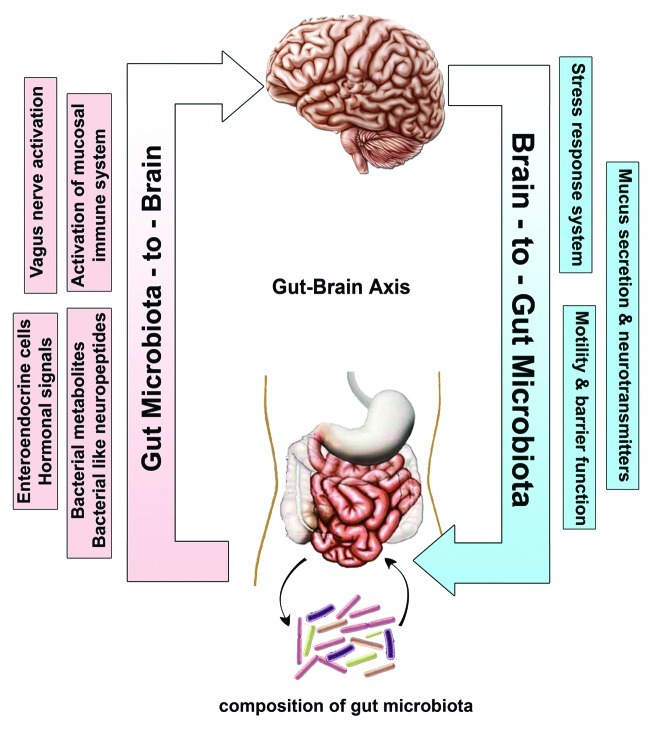
The brain and the gut are highly integrated organs, with communication occurring in a bidirectional manner via passive and active mechanisms including neural, immunological and endocrine pathways as well as through hypothalamic-pituitary-adrenal (HPA) stress axis16-18 (Fig. 1). Research on gut-brain axis has focused on top-down communication on how the brain affects GI motility, blood flow and secretion, and on the bottom-up effects of intestinal activity on visceral perception and CNS activity. The neuroanatomy of the gut-brain axis has been reviewed elsewhere.19,20
Figure 1. Bidirectional communication between the gut microbiota and the central nervous system (CNS). The composition of gut microbiota could modulate the function of CNS through various communication means including neural (vagus nerve activation), hormonal (enteroendocrine cells and bacterial neuropeptides), humeral (bacterial metabolites) and immunological (activation of mucosal immune system). The Brain-to-Gut microbiota axis is mediated via stress factors, alteration in intestinal permeability and motility and through release of neurotransmitters and mucus.
Gut microbiota-to-brain
Over the years, the ability of microbiota to impact on brain function has been subject to an intense debate following the observation that administration of oral antibiotics and laxatives resulted in a dramatic improvement in patients with hepatic encephalopathy.21,22 The observation that common categories of GI diseases (functional and inflammatory) often display correlation with psychiatric comorbidity that includes depression and anxiety in up to 80% of patients, supports the possibility that alteration of microbiota can affect CNS function.23,24 These clinical findings are supported by results from animal studies showing that certain pathogenic enteric bacteria, during the initial phase of infection, can induce an anxiety-like behavior.25 Furthermore, microbiota has also been shown to modulate the levels of adreno-corticotrophic hormone (ACTH) in mice.13 Hence, the term: ‘gut feelings’ and its role in intuitive decision making has perhaps some bearing and the Asian understanding of the gut being the ‘location of the soul and the center of spiritual and physical strength’ and not only a simple digestive system are in support of these findings.26,27 These findings were recently corroborated by us28 and others14,29 in which it was shown that germ free (GF) mice display (1) increased motor activity and reduced anxiety (2) decreased N-methyl-D-aspartate receptor subunit NR2B mRNA expression in the amygdale, (3) decreased 5HT-1A receptor in the hippocampus and (4) increased expression of brain-derived neurotrophic factor (BDNF) in the hippocampus.14,28-30
Modification of microbiota by a combination of probiotics (Lactobacillus helveticus and Bifidobacterium longum) has been shown to reduce anxiety in animals and bring beneficial psychological effects with a decrease in serum cortisol in patients.31 Furthermore, Bravo et al. showed that long lasting treatment of mice with the probiotic bacterium Lactobacillus rhamnosus appears to impact on emotional behavior and the expression of the neurotransmitter GABA (γ-aminobutyric acid) in the CNS in a region-dependent manner.32 Likewise, Lactobacillus rhamnosus treatment may be associated with a decrease in corticosterone levels as well as anxiety- and stress-related behavior. These alterations were not observed in vagotomized mice, indicating the vagus nerve may be part of the communication pathways that allow communication between the gut and the brain.32 By analogy, Li et al., have observed a temporal association between diet-induced alterations in intestinal microbiota diversity and changes in working and reference memories.15 Consistent with Li’s findings, Gareau et al., showed that memory dysfunction in Citrobacter rodentium infected mice was prevented by daily treatment with probiotics.33 The study also showed a deficit in non-spatial and working memory in GF mice even in the absence of stress. While several mechanisms have been proposed, including the release of bacterial metabolites, neuropeptides or directly by activating vagal pathways at the level,33 the precise pathways remain to be identified (Fig. 1).
Brain-to-gut microbiota
Whereas the focus of current research is mainly on studying the influence of gut microbiota on the brain, there are also data to suggest that descending signals from CNS can alter the composition and function of the gut microbiota in rodents34,35 and primates.36,37 For example, while acute stress increased colonic paracellular permeability involving overproduction of IFN-gamma and a decrease in mRNA expression of ZO-2 and occludin,38 stress induced secretion of norepinephrine favored overgrowth of non-pathogenic isolates of Escherichia coli as well as the pathogenic E. coli 0157:H7.39,40 Other mechanisms such as changes in mucus secretion41 and in GI motility42 have also been shown to affect gut microbiome composition (Fig. 1).
Gut Microbiota and Brain Development (Prenatal and Postnatal)
The prenatal and postnatal periods in mammalian development are characterized by rapid changes in neuronal organization, thus providing a critical window of opportunity during which environmental factors could have long-term influences on brain and behavior. During the last few years, a number of studies pointed to an ability of microbiota to influence the fine maturation of the brain which have long lasting effects on functions of the brain.13,28
Prenatal
A rapidly growing body of empirical evidence supports the Barker hypothesis regarding the influence of the intrauterine environment on fetal development and susceptibility to a wide range of metabolic, neurodevelopmental and psychiatric diseases later in life.43 The developing brain has been shown to be susceptible to both internal and external environmental cues during prenatal life. Epidemiological data have indicated a possible common link between neurodevelopmental disorders, such as autism and schizophrenia, and microbial pathogen infections during the prenatal period.44-46 Experimental studies in rodents further support these findings, demonstrating that exposure to microbial pathogens during similar developmental periods results in behavioral abnormalities, including anxiety-like behavior and impaired cognitive function.47-49 The role of gut microbiota in modulating mood and behavior has been the focus of many research studies.14,28,29 As mentioned above, GF mice display increased motor activity and decreased anxiety compared with conventionally raised mice. Notably, colonizing adult GF mice with a whole microbiota failed to normalize the behavior of GF mice. On the other hand, colonizing the mother for at least 30 d before mating was successful in reverting the behavioral phenotype in the offspring.28 This interesting finding hints to a crucial role of the microbiota on the mother during pregnancy that might have programming effects later in life. The fetus lives in an almost sterile environemt50-53and communicates with the mother through the placenta54 (Fig. 2). It is possible that maternal microbial metabolites could reach the growing fetus through the placenta and affect fetal brain development. Several recent studies indicate an important role of the placenta in shaping the fetus development. The placenta, nicknamed the Fetal Armor,55 has been shown to protect the fetus from damage when the mother is deprived of food by breaking down its own tissue (placental autophagy) to nourish energy demanding organs like the fetal brain.56 The placenta appears also to provide the hormone serotonin essential for fetal forebrain development.57 Using a novel ex vivo preparation, the authors demonstrated that the placenta could convert maternal tryptophan into the neurotransmitter serotonin (5-hydroxytryptophan; 5-HT), providing the primary source of 5-HT for the developing mouse forebrain at midgestation.57 The hormonal interaction between the placenta and the fetal hypothalamus-pituitary adrenal axis (HPA) was shown to be involved in regulating fetal brain development especially during stress. In rodents, prenatal stress had programming effects that extended to adulthood. These effects included elevation of stress-induced HPA axis activity,58,59 increased anxiety and behavioral reactivity/fearfulnes,60-62 decreased hippocampal glucocorticoid binding capacity58,63 and a decline in cognitive performance.64 Several studies pointed to the effect of microbiota in modulating the activity of HPA axis. Prenatally, maternal probiotic supplementation during pregnancy and lactation normalized the high corticosterone concentrations and restored corticotrophin-releasing hormone seen in maternally separated neonates.65 The mechanism whereby microbiota influences brain prenatally is yet to be determined, but the placenta is one of several possibilities.
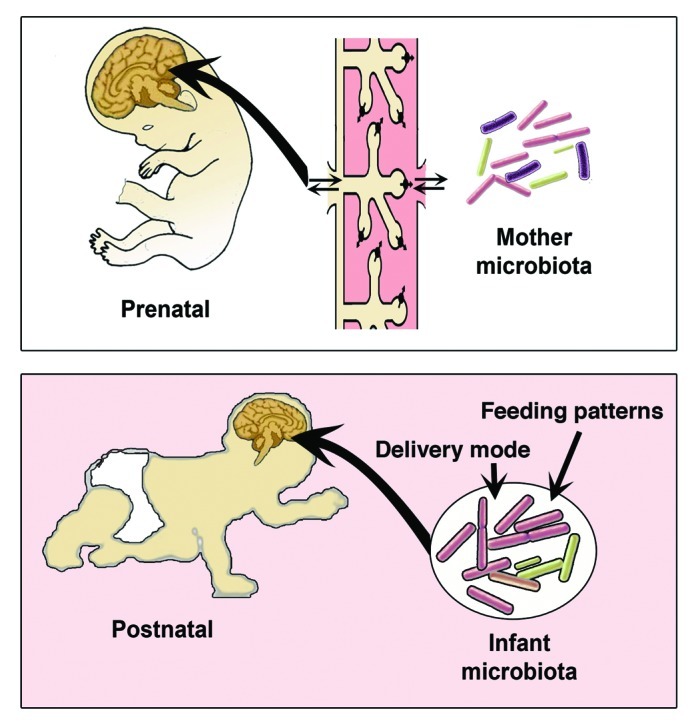
Figure 2. Gut microbiota-to-Brain communications during prenatal and postnatal development.
Postnatal
The postnatal period is another critical period for brain development. For most vertebrates, the majority of organs and tissue development occurs during embryogenesis, and postnatal changes are primarily concerned with growth. However, the CNS is different in that a considerable amount of morphological development, cell differentiation and acquisition of function, takes place during postnatal development.66 Colonization of the GI tract with microbiota begins postnatally at birth, overlapping with this critical period of brain development. Microbiota colonization is actually influenced to a great degree by mode of delivery67 and feeding patterns68 (Fig. 2). Clinical studies have shown that breast-fed infants have better neurodevelopment outcomes and higher scores on intelligence tests.69,70 The microbiota of breast-fed infants appear to be more diverse and heterogeneous than the formula-fed according to a recent metagenomic study.71 However, the effect of breastfeeding on the composition of the infant gut microbiota, more specifically on the predominance of bifidobacteria is still controversial. Several reports72,73 found no difference for bifidobacteria between breast-and formula-fed infants. Interestingly the strains of bifidobacteria that colonize the infant intestine are different from adult predominating mainly with Bifidobacterium longum and Bifidobacterium bifidum. These two strains are then replaced with Bifidobacterium adolescentis and by other strains of the same species.74 Using bifidobacteria “of human infant intestinal origin” in probiotics may have beneficial effects on depression in rats exposed to maternal separation stress in early life.12 Recently, Stanton and coworkers showed that feeding mice with two forms of bifidobacteria isolated from infant feces could influence brain fatty acid composition.75 These studies show the ability of infant microbiota to reset and influence neurophysiological parameters.
Gut microbiota also synthesize vitamins that are essential for human survival such as vitamins K2 and B12. Vitamin B12 or cobalamin is produced mainly by Lactobacillus reuteri76 and is important for development of the nervous system.77,78
Further evidence for the role of microbiota on postnatal brain development came from animal studies on GF mice. The activity of the HPA axis13 and more recently the hippocampal serotonergic system79 are altered in GF mice with elevation in corticosterone and serotonin levels. Colonisation of GF mice post weaning was insufficient to reverse the CNS neurochemical consequences79 and had no effect on the HPA axis,13 introducing the notion of a critical time window very early in life. These finding gives support to the ‘microflora hypothesis80- an extension of the hygiene hypothesis which suggests that insufficient exposure to microbiota early in life may affect the composition and maturation of the adult microbiota that, in turn, skews the immune development in a way that increases susceptibility to contract allergic, and immune related diseases. The micorflora hypothesis may also be considered in the etiology of autism81—a neurodevelopment disorder of unknown etiology that starts to develop in the late postnatal period. Numerous reports have pointed to a possible role of microbiota in the development of autism: (1) most children with autism often experience a range of GI disorders, (2) onset of the disease usually follows antimicrobial therapy, (3) a significant percentage of children with late-onset autism (18–24 mo of age) have a history of extensive antibiotic use and (4) oral vancomycin treatment showed a decrease in autistic symptoms, while relapse occurs following cessation of treatment.45,82 Real time qPCR83 and culture-based microbiota45 profiling techniques support the possibility that alteration in microbiota may contribute to disease phenotype. For example, a 10-fold increase in certain clusters of Clostridium spp. in stool samples from autistic children compared with healthy controls has been observed. The authors further speculated on the possibility that exposure to trimethoprim/sulfamethoxazole antibiotics were more likely to precede diagnosis of late-onset autism than exposure to any other antibiotic regimen because trimethoprim/sulfamethoxazole are not effective against Clostridium spp., while oral vancomycin specifically targets Gram positive organisms which include Clostridium spp.45 It has also been suggested that Clostridia spores could be one reason for the high rates of autism seen among siblings.84 Additional mechanisms are of course equally possible and great caution needs to be exercised in the interpretation of these data on autism and it could very well be a chicken-and-egg situation whereby symptoms observed in autistic children were the result of their autism rather than the cause.
Putative Mechanisms Underlying Microbiota Brain Axis
Bacterial metabolites and bacterial neuro-like peptides
Dietary carbohydrates can be digested in colon by gut microbiota into short chain fatty acids (SCFA) such as butyrate, acetate and proprionate.85 SCFA are sensed by G protein coupled receptors (GPRs), GPR41 and GPR43, which induce colonic motility, regulate appetite86 and suppress colon cancer.87 Butyrate can be directly used by colonocytes as an energy source, whereas propionate and acetate are taken up directly to the blood stream and transported to various organs.88,89 Butyrate as well as propionate are strong inhibitors of histone deacetylases (HDACs), whereas acetate is not able to block HDAC functions.90 Oral administration of acetate91 or butyrate92 was able to ameliorate the disease phenotype in DSS-induced colitis model in mice. SCFAs can interact with nerve cells by stimulating sympathetic nervous system93 and butyrate in particular has been suggested to influence memory and learning processes via HDAC inhibition.94,95 Moreover systemic injection of butyrate exerted a potential anti-depressant effect by inducing histone hyperacetylation in mice that had increased BDNF transcripts in the frontal cortex.96 On the same treatment regime another SCFA, propionate, has been reported not only to accelerate an autism-like behavior in rodents97 but also to impair specific object recognition and social behavior in rats exposed to propionate compared with controls.98
Modulation of transmitters (e.g., serotonin, melatonin, gamma-aminobutyric acid, histamines and acetylcholine) within the gut is yet another possible mechanism of action that could mediate the effects of the gut microbiota. For example, metabolic profiling of GF and conventionalized mice have revealed that conventionalization of GF mice by gut microbiota results in a 2.8-fold increase in plasma serotonin levels.99 In line with this study, administration of the commensal bacteria, Bifidobacteria infantis, increased the plasma concentration of tryptophan, suggesting that the normal microbiota can influence the precursor pool for serotonin (5-HT).12 It has been reported that microbiota may express and secrete neuropeptide like molecules proposed to influence behavior and emotion.100
Stimulation of afferent system to CNS
Another possible mechanism mediating the gut-brain communication may be via established neuronal circuits. Microbiota can elicit signals via the vagal nerve to the brain and vice versa.101,102 Several reports showed or supported a direct link between microbiota and the ENS. Oral ingestion of Lactobacillus rhamnosus has been shown to reduce stress-induced corticosterone and anxiety- and depression-related behavior in mice via the vagal nerve.32 Moreover, using another Lactobacillus strain, Lactobacillus reuteri, Kunze and colleagues observed activation in calcium-dependent potassium channels in a specific subset of enteric neurons in the colonic mysenteric plexus of Sprague Dawley rats,103 thus pointing to a direct link between microbiota and the ENS. Another study showed that treatment of Trichur muris infected mice with a specific probiotic Bifidobacterium longum was effective in normalizing anxiety-like behavior exhibited by the infected mice. This effect of Bifidobacterium longum required an intact vagus nerve and was not accompanied by changes in gut immune functions.104
Activation of the mucosal immune system
The gut is patrolled by a variety of immune cells such as T-regulatory cells and antigen presenting cells (APC) which could traffic from the gut associated immune cells (GALT) to other peripheral lymphoid sites including the CNS. Recent studies established that GALT is shaped by components of the gut microbiota, some supporting the differentiation of interleukin (IL)-17-producing TH17 cells,105-107 while others the generation of regulatory T lymphocytes (Treg).108 Immune cell populations induced within the gut could cross the blood-brain barrier and within the CNS be reactivated by the appropriate resident APC.109
Endocrine mechanisms
Enteroendocrine cells (EEC) form the basis of the largest endocrine system in the body. They secrete gut hormones such as cholecystokinin (CCK), glucagon-like peptide-1 (GLP-1), oxyntomodulin, gut peptide YY (PYY) which control food intake and energy homeostasis. The brain receives hormonal signals from the EEC via two main mechanisms either by a paracrine effect on proximate cells (notably vagal afferent fibers), or by a classical endocrine fashion through the circulation acting mainly on the hypothalamus where the blood–brain barrier is leaky or absent. Gut microbiota have been reported to regulate EEC and influence the release of gut hormones.110 Furthermore, the presence of gut microbiota is necessary for the differentiation of EEC in zebrafish.111 Cani et al. in a series of studies showed a connection between gut microbiota and levels of two gut hormones GLP-1 and GLP-2. GLP-1 promotes satiety and weight loss112 while GLP-2 stimulates intestinal glucose transport and reduces gut permeability.113 In rats, oligofructose treatment, which increases the proportion of Bifidobacteria has been associated with an increase in GLP-1 secreting EEC in the colon.110 Ob/ob mice treated with a prebiotic had an altered gut microbiome and higher levels of GLP-1 and GLP-2114. Another evidence for the role of gut microbiota in modulating gut hormones comes from studies after gastric bypass surgery which showed a large shift in the bacterial population of the gut. Firmicutes which were dominant in normal weight and obese individuals, decreased significantly in individuals after gastric bypass surgery.115 As we know, gut hormones are able to reduce appetite and weight loss after gastric bypass surgery.116,117 Interestingly, administration of probiotics after surgery induced an accelerated weight loss,118 thus supporting the notion that gut microbiota could indeed modulate gut hormones.
Summary/Conclusion
Considerable progress has already been made in understanding the bi-directional crosstalk governing the gut-brain axis, however we are only just beginning to realize the physiological impact of the microbota on this process. The finding that microbiota can modulate behavior raises the prospect of its involvement in developmental programming. How does microbiota-to-brain signaling affect brain development? How beneficial it is to supply mothers during pregnancy with probiotics or prebiotics to shape neural development of the fetus? One of the great advantages in addressing these questions is the fact that we have the tools and techniques to study the Microbiota-Gut-Brain Axis. This include mechanistic studies in rodents and in piglets which can be translated into the human situation. Germ free animals such as rodents and piglets are a tremendous tool to study such interactions. Stool samples for metagenomic sequencing and gut tissue that can be easily obtained by endoscopic biopsies for analysis of signaling mechanisms. With the rapid expansion of non-invasive techniques to monitor brain structure, function and signaling, we are quite hopeful that we will see a rapid and significant progress in unlocking the mysterious black box of Gut. Thus, by modulating microbiota, you may modulate the mind.
Acknowledgments
We would like to thank Qatar University for the PhD funding of M.A. The authors and their works are supported by grants from VR, Cancerfonden, EU-project TORNADO and Singapore Millenium Foundation. We are also grateful for valuable comments from Velmurugesan Arulampalam.
Footnotes
Previously published online: www.landesbioscience.com/journals/gutmicrobes/article/21287
References
- 1.Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, et al. Diversity of the human intestinal microbial flora. Science. 2005;308:1635–8. doi: 10.1126/science.1110591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Korecka A, Arulampalam V. The gut microbiome: scourge, sentinel or spectator? J Oral Microbiol. 2012;4:9367. doi: 10.3402/jom.v4i0.9367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Reyes A, Haynes M, Hanson N, Angly FE, Heath AC, Rohwer F, et al. Viruses in the faecal microbiota of monozygotic twins and their mothers. Nature. 2010;466:334–8. doi: 10.1038/nature09199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Arumugam M, Raes J, Pelletier E, Le Paslier D, Yamada T, Mende DR, et al. Enterotypes of the human gut microbiome. Nature. 2011;473:174. doi: 10.1038/nature09944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hooper LV, Gordon JI. Commensal host-bacterial relationships in the gut. Science. 2001;292:1115–8. doi: 10.1126/science.1058709. [DOI] [PubMed] [Google Scholar]
- 6.Amaral FA, Sachs D, Costa VV, Fagundes CT, Cisalpino D, Cunha TM, et al. Commensal microbiota is fundamental for the development of inflammatory pain. Proc Natl Acad Sci USA. 2008;105:2193–7. doi: 10.1073/pnas.0711891105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dumas ME, Barton RH, Toye A, Cloarec O, Blancher C, Rothwell A, et al. Metabolic profiling reveals a contribution of gut microbiota to fatty liver phenotype in insulin-resistant mice. Proc Natl Acad Sci USA. 2006;103:12511–6. doi: 10.1073/pnas.0601056103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aronsson L, Huang Y, Parini P, Korach-André M, Håkansson J, Gustafsson JA, et al. Decreased fat storage by Lactobacillus paracasei is associated with increased levels of angiopoietin-like 4 protein (ANGPTL4) PLoS ONE. 2010;5:5. doi: 10.1371/journal.pone.0013087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hawrelak JA, Myers SP. The causes of intestinal dysbiosis: a review. Altern Med Rev. 2004;9:180–97. [PubMed] [Google Scholar]
- 10.Daneman R, Rescigno M. The gut immune barrier and the blood-brain barrier: are they so different? Immunity. 2009;31:722–35. doi: 10.1016/j.immuni.2009.09.012. [DOI] [PubMed] [Google Scholar]
- 11.Lepage P, Colombet J, Marteau P, Sime-Ngando T, Doré J, Leclerc M. Dysbiosis in inflammatory bowel disease: a role for bacteriophages? Gut. 2008;57:424–5. doi: 10.1136/gut.2007.134668. [DOI] [PubMed] [Google Scholar]
- 12.Desbonnet L, Garrett L, Clarke G, Bienenstock J, Dinan TG. The probiotic Bifidobacteria infantis: An assessment of potential antidepressant properties in the rat. J Psychiatr Res. 2008;43:164–74. doi: 10.1016/j.jpsychires.2008.03.009. [DOI] [PubMed] [Google Scholar]
- 13.Sudo N, Chida Y, Aiba Y, Sonoda J, Oyama N, Yu XN, et al. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J Physiol. 2004;558:263–75. doi: 10.1113/jphysiol.2004.063388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Neufeld KM, Kang N, Bienenstock J, Foster JA. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol Motil. 2011;23:255–64, e119. doi: 10.1111/j.1365-2982.2010.01620.x. [DOI] [PubMed] [Google Scholar]
- 15.Li W, Dowd SE, Scurlock B, Acosta-Martinez V, Lyte M. Memory and learning behavior in mice is temporally associated with diet-induced alterations in gut bacteria. Physiol Behav. 2009;96:557–67. doi: 10.1016/j.physbeh.2008.12.004. [DOI] [PubMed] [Google Scholar]
- 16.Berthoud HR. Vagal and hormonal gut-brain communication: from satiation to satisfaction. Neurogastroenterol Motil. 2008;20(Suppl 1):64–72. doi: 10.1111/j.1365-2982.2008.01104.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Drossman DA. What does the future hold for irritable bowel syndrome and the functional gastrointestinal disorders? J Clin Gastroenterol. 2005;39(Suppl 3):S251–6. doi: 10.1097/01.mcg.0000156107.13247.69. [DOI] [PubMed] [Google Scholar]
- 18.Shanahan F. Brain-gut axis and mucosal immunity: a perspective on mucosal psychoneuroimmunology. Semin Gastrointest Dis. 1999;10:8–13. [PubMed] [Google Scholar]
- 19.Mayer EA. Gut feelings: the emerging biology of gut-brain communication. Nat Rev Neurosci. 2011;12:453–66. doi: 10.1038/nrn3071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Van Oudenhove L, Demyttenaere K, Tack J, Aziz Q. Central nervous system involvement in functional gastrointestinal disorders. Best Pract Res Clin Gastroenterol. 2004;18:663–80. doi: 10.1016/j.bpg.2004.04.010. [DOI] [PubMed] [Google Scholar]
- 21.Morgan MY. The treatment of chronic hepatic encephalopathy. Hepatogastroenterology. 1991;38:377–87. [PubMed] [Google Scholar]
- 22.Morgan MY, Blei A, Grüngreiff K, Jalan R, Kircheis G, Marchesini G, et al. The treatment of hepatic encephalopathy. Metab Brain Dis. 2007;22:389–405. doi: 10.1007/s11011-007-9060-7. [DOI] [PubMed] [Google Scholar]
- 23.Whitehead WE, Palsson O, Jones KR. Systematic review of the comorbidity of irritable bowel syndrome with other disorders: what are the causes and implications? Gastroenterology. 2002;122:1140–56. doi: 10.1053/gast.2002.32392. [DOI] [PubMed] [Google Scholar]
- 24.Garakani A, Win T, Virk S, Gupta S, Kaplan D, Masand PS. Comorbidity of irritable bowel syndrome in psychiatric patients: a review. Am J Ther. 2003;10:61–7. doi: 10.1097/00045391-200301000-00014. [DOI] [PubMed] [Google Scholar]
- 25.Lyte M, Li W, Opitz N, Gaykema RP, Goehler LE. Induction of anxiety-like behavior in mice during the initial stages of infection with the agent of murine colonic hyperplasia Citrobacter rodentium. Physiol Behav. 2006;89:350–7. doi: 10.1016/j.physbeh.2006.06.019. [DOI] [PubMed] [Google Scholar]
- 26.Yu F, Takahashi T, Moriya J, Kawaura K, Yamakawa J, Kusaka K, et al. Traditional Chinese medicine and Kampo: a review from the distant past for the future. J Int Med Res. 2006;34:231–9. doi: 10.1177/147323000603400301. [DOI] [PubMed] [Google Scholar]
- 27.Bischoff SC. 'Gut health': a new objective in medicine? BMC Med. 2011:9. doi: 10.1186/1741-7015-9-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Heijtz RD, Wang S, Anuar F, Qian Y, Björkholm B, Samuelsson A, et al. Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci USA. 2011;108:3047–52. doi: 10.1073/pnas.1010529108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bercik P, Denou E, Collins J, Jackson W, Lu J, Jury J, et al. The intestinal microbiota affect central levels of brain-derived neurotropic factor and behavior in mice. Gastroenterology. 2011;141:599–609. doi: 10.1053/j.gastro.2011.04.052. [DOI] [PubMed] [Google Scholar]
- 30.Collins S. Intestinal microbiota and the brain-gut axis. in Finlay, B (ed), Microbiota: Agents for Health and Disease, The Biomedical & Life Sciences Collection. London: Henry Stewart Talks Ltd, 2011. [Google Scholar]
- 31.Messaoudi M, Lalonde R, Violle N, Javelot H, Desor D, Nejdi A, et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br J Nutr. 2011;105:755–64. doi: 10.1017/S0007114510004319. [DOI] [PubMed] [Google Scholar]
- 32.Bravo JA, Forsythe P, Chew MV, Escaravage E, Savignac HM, Dinan TG, et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc Natl Acad Sci USA. 2011;108:16050–5. doi: 10.1073/pnas.1102999108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gareau MG, Wine E, Rodrigues DM, Cho JH, Whary MT, Philpott DJ, et al. Bacterial infection causes stress-induced memory dysfunction in mice. Gut. 2011;60:307–17. doi: 10.1136/gut.2009.202515. [DOI] [PubMed] [Google Scholar]
- 34.Tannock GW, Savage DC. Influences of dietary and environmental stress on microbial populations in the murine gastrointestinal tract. Infect Immun. 1974;9:591–8. doi: 10.1128/iai.9.3.591-598.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Suzuki K, Harasawa R, Yoshitake Y, Mitsuoka T. Effects of crowding and heat stress on intestinal flora, body weight gain, and feed efficiency of growing rats and chicks. Nihon Juigaku Zasshi. 1983;45:331–8. doi: 10.1292/jvms1939.45.331. [DOI] [PubMed] [Google Scholar]
- 36.Holdeman LV, Good IJ, Moore WEC. Human fecal flora: variation in bacterial composition within individuals and a possible effect of emotional stress. Appl Environ Microbiol. 1976;31:359–75. doi: 10.1128/aem.31.3.359-375.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bailey MT, Coe CL. Maternal separation disrupts the integrity of the intestinal microflora in infant rhesus monkeys. Dev Psychobiol. 1999;35:146–55. doi: 10.1002/(SICI)1098-2302(199909)35:2<146::AID-DEV7>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 38.Demaude J, Salvador-Cartier C, Fioramonti J, Ferrier L, Bueno L. Phenotypic changes in colonocytes following acute stress or activation of mast cells in mice: implications for delayed epithelial barrier dysfunction. Gut. 2006;55:655–61. doi: 10.1136/gut.2005.078675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Freestone PP, Williams PH, Haigh RD, Maggs AF, Neal CP, Lyte M. Growth stimulation of intestinal commensal Escherichia coli by catecholamines: a possible contributory factor in trauma-induced sepsis. Shock. 2002;18:465–70. doi: 10.1097/00024382-200211000-00014. [DOI] [PubMed] [Google Scholar]
- 40.Freestone PP, Haigh RD, Williams PH, Lyte M. Involvement of enterobactin in norepinephrine-mediated iron supply from transferrin to enterohaemorrhagic Escherichia coli. FEMS Microbiol Lett. 2003;222:39–43. doi: 10.1016/S0378-1097(03)00243-X. [DOI] [PubMed] [Google Scholar]
- 41.Rubio CA, Huang CB. Quantification of the sulphomucin-producing cell population of the colonic mucosa during protracted stress in rats. In Vivo. 1992;6:81–4. [PubMed] [Google Scholar]
- 42.Collins SM, Bercik P. The relationship between intestinal microbiota and the central nervous system in normal gastrointestinal function and disease. Gastroenterology. 2009;136:2003–14. doi: 10.1053/j.gastro.2009.01.075. [DOI] [PubMed] [Google Scholar]
- 43.Barker DJP. Maternal nutrition, fetal nutrition, and disease in later life. Nutrition. 1997;13:807–13. doi: 10.1016/S0899-9007(97)00193-7. [DOI] [PubMed] [Google Scholar]
- 44.Bale TL, Baram TZ, Brown AS, Goldstein JM, Insel TR, McCarthy MM, et al. Early life programming and neurodevelopmental disorders. Biol Psychiatry. 2010;68:314–9. doi: 10.1016/j.biopsych.2010.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Finegold SM, Molitoris D, Song Y, Liu C, Vaisanen ML, Bolte E, et al. Gastrointestinal microflora studies in late-onset autism. Clin Infect Dis. 2002;35(Suppl 1):S6–16. doi: 10.1086/341914. [DOI] [PubMed] [Google Scholar]
- 46.Mittal VA, Ellman LM, Cannon TD. Gene-environment interaction and covariation in schizophrenia: the role of obstetric complications. Schizophr Bull. 2008;34:1083–94. doi: 10.1093/schbul/sbn080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Goehler LE, Park SM, Opitz N, Lyte M, Gaykema RP. Campylobacter jejuni infection increases anxiety-like behavior in the holeboard: possible anatomical substrates for viscerosensory modulation of exploratory behavior. Brain Behav Immun. 2008;22:354–66. doi: 10.1016/j.bbi.2007.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bilbo SD, Levkoff LH, Mahoney JH, Watkins LR, Rudy JW, Maier SF. Neonatal infection induces memory impairments following an immune challenge in adulthood. Behav Neurosci. 2005;119:293–301. doi: 10.1037/0735-7044.119.1.293. [DOI] [PubMed] [Google Scholar]
- 49.Sullivan R, Wilson DA, Feldon J, Yee BK, Meyer U, Richter-Levin G, et al. The International Society for Developmental Psychobiology annual meeting symposium: Impact of early life experiences on brain and behavioral development. Dev Psychobiol. 2006;48:583–602. doi: 10.1002/dev.20170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jiménez E, Fernández L, Marín ML, Martín R, Odriozola JM, Nueno-Palop C, et al. Isolation of commensal bacteria from umbilical cord blood of healthy neonates born by cesarean section. Curr Microbiol. 2005;51:270–4. doi: 10.1007/s00284-005-0020-3. [DOI] [PubMed] [Google Scholar]
- 51.Jiménez E, Marín ML, Martín R, Odriozola JM, Olivares M, Xaus J, et al. Is meconium from healthy newborns actually sterile? Res Microbiol. 2008;159:187–93. doi: 10.1016/j.resmic.2007.12.007. [DOI] [PubMed] [Google Scholar]
- 52.Satokari R, Grönroos T, Laitinen K, Salminen S, Isolauri E. Bifidobacterium and Lactobacillus DNA in the human placenta. Lett Appl Microbiol. 2009;48:8–12. doi: 10.1111/j.1472-765X.2008.02475.x. [DOI] [PubMed] [Google Scholar]
- 53.Steel JH, Malatos S, Kennea N, Edwards AD, Miles L, Duggan P, et al. Bacteria and inflammatory cells in fetal membranes do not always cause preterm labor. Pediatr Res. 2005;57:404–11. doi: 10.1203/01.PDR.0000153869.96337.90. [DOI] [PubMed] [Google Scholar]
- 54.Dancis J. The Role of the Placenta in Fetal Survival. Pediatr Clin North Am. 1965;12:477–92. doi: 10.1016/s0031-3955(16)31728-x. [DOI] [PubMed] [Google Scholar]
- 55.Kalb C. Fetal armor. Sci Am. 2012;306:72–3. doi: 10.1038/scientificamerican0212-72. [DOI] [PubMed] [Google Scholar]
- 56.Broad KD, Keverne EB. Placental protection of the fetal brain during short-term food deprivation. Proc Natl Acad Sci USA. 2011;108:15237–41. doi: 10.1073/pnas.1106022108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bonnin A, Goeden N, Chen K, Wilson ML, King J, Shih JC, et al. A transient placental source of serotonin for the fetal forebrain. Nature. 2011;472:347–50. doi: 10.1038/nature09972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Henry C, Kabbaj M, Simon H, Le Moal M, Maccari S. Prenatal stress increases the hypothalamo-pituitary-adrenal axis response in young and adult rats. J Neuroendocrinol. 1994;6:341–5. doi: 10.1111/j.1365-2826.1994.tb00591.x. [DOI] [PubMed] [Google Scholar]
- 59.McCormick CM, Smythe JW, Sharma S, Meaney MJ. Sex-specific effects of prenatal stress on hypothalamic-pituitary-adrenal responses to stress and brain glucocorticoid receptor density in adult rats. Brain Res Dev Brain Res. 1995;84:55–61. doi: 10.1016/0165-3806(94)00153-Q. [DOI] [PubMed] [Google Scholar]
- 60.Alonso J, Castellano MA, Rodriguez M. Behavioral lateralization in rats: prenatal stress effects on sex differences. Brain Res. 1991;539:45–50. doi: 10.1016/0006-8993(91)90684-N. [DOI] [PubMed] [Google Scholar]
- 61.Vallée M, Mayo W, Dellu F, Le Moal M, Simon H, Maccari S. Prenatal stress induces high anxiety and postnatal handling induces low anxiety in adult offspring: correlation with stress-induced corticosterone secretion. J Neurosci. 1997;17:2626–36. doi: 10.1523/JNEUROSCI.17-07-02626.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Caldji C, Tannenbaum B, Sharma S, Francis D, Plotsky PM, Meaney MJ. Maternal care during infancy regulates the development of neural systems mediating the expression of fearfulness in the rat. Proc Natl Acad Sci USA. 1998;95:5335–40. doi: 10.1073/pnas.95.9.5335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Maccari S, Piazza PV, Kabbaj M, Barbazanges A, Simon H, Le Moal M. Adoption reverses the long-term impairment in glucocorticoid feedback induced by prenatal stress. J Neurosci. 1995;15:110–6. doi: 10.1523/JNEUROSCI.15-01-00110.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vallée M, MacCari S, Dellu F, Simon H, Le Moal M, Mayo W. Long-term effects of prenatal stress and postnatal handling on age-related glucocorticoid secretion and cognitive performance: a longitudinal study in the rat. Eur J Neurosci. 1999;11:2906–16. doi: 10.1046/j.1460-9568.1999.00705.x. [DOI] [PubMed] [Google Scholar]
- 65.Barouei J, Hodgson DM. Prophylactic Potential of Maternal Probiotic Supplementation Against Intestinal Dysfunctions Induced by Early Life Stress. J Dev Orig Health Dis. 2011;2:S77–S. [Google Scholar]
- 66.Noback CRSNL, Demarest RJ. The Human Nervous System. Philadelphia: Williams and Wilkins, 1995. [Google Scholar]
- 67.Dominguez-Bello MG, Costello EK, Contreras M, Magris M, Hidalgo G, Fierer N, et al. Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci USA. 2010;107:11971–5. doi: 10.1073/pnas.1002601107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Pop M. We are what we eat: how the diet of infants affects their gut microbiome. Genome Biol. 2012;13:152. doi: 10.1186/gb-2012-13-4-152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kramer MS, Aboud F, Mironova E, Vanilovich I, Platt RW, Matush L, et al. Promotion of Breastfeeding Intervention Trial (PROBIT) Study Group Breastfeeding and child cognitive development: new evidence from a large randomized trial. Arch Gen Psychiatry. 2008;65:578–84. doi: 10.1001/archpsyc.65.5.578. [DOI] [PubMed] [Google Scholar]
- 70.Guxens M, Mendez MA, Moltó-Puigmartí C, Julvez J, García-Esteban R, Forns J, et al. Breastfeeding, long-chain polyunsaturated fatty acids in colostrum, and infant mental development. Pediatrics. 2011;128:e880–9. doi: 10.1542/peds.2010-1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schwartz S, Friedberg I, Ivanov IV, Davidson LA, Goldsby JS, Dahl DB, et al. A metagenomic study of diet-dependent interaction between gut microbiota and host in infants reveals differences in immune response. Genome Biol. 2012;13:r32. doi: 10.1186/gb-2012-13-4-r32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Palmer C, Bik EM, DiGiulio DB, Relman DA, Brown PO. Development of the human infant intestinal microbiota. PLoS Biol. 2007;5:e177. doi: 10.1371/journal.pbio.0050177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Adlerberth I, Wold AE. Establishment of the gut microbiota in Western infants. Acta Paediatr. 2009;98:229–38. doi: 10.1111/j.1651-2227.2008.01060.x. [DOI] [PubMed] [Google Scholar]
- 74.Shkoporov AN, Khokhlova EV, Kulagina EV, Smeianov VV, Kafarskaia LI, Efimov BA. Application of several molecular techniques to study numerically predominant Bifidobacterium spp. and Bacteroidales order strains in the feces of healthy children. Biosci Biotechnol Biochem. 2008;72:742–8. doi: 10.1271/bbb.70628. [DOI] [PubMed] [Google Scholar]
- 75.Wall R, Marques TM, O’Sullivan O, Ross RP, Shanahan F, Quigley EM, et al. Contrasting effects of Bifidobacterium breve NCIMB 702258 and Bifidobacterium breve DPC 6330 on the composition of murine brain fatty acids and gut microbiota. Am J Clin Nutr. 2012;95:1278–87. doi: 10.3945/ajcn.111.026435. [DOI] [PubMed] [Google Scholar]
- 76.Santos F, Wegkamp A, de Vos WM, Smid EJ, Hugenholtz J. High-Level folate production in fermented foods by the B12 producer Lactobacillus reuteri JCM1112. Appl Environ Microbiol. 2008;74:3291–4. doi: 10.1128/AEM.02719-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Marques TM, Wall R, Ross RP, Fitzgerald GF, Ryan CA, Stanton C. Programming infant gut microbiota: influence of dietary and environmental factors. Curr Opin Biotechnol. 2010;21:149–56. doi: 10.1016/j.copbio.2010.03.020. [DOI] [PubMed] [Google Scholar]
- 78.Dror DK, Allen LH. Effect of vitamin B12 deficiency on neurodevelopment in infants: current knowledge and possible mechanisms. Nutr Rev. 2008;66:250–5. doi: 10.1111/j.1753-4887.2008.00031.x. [DOI] [PubMed] [Google Scholar]
- 79.Clarke G, Grenham S, Scully P, Fitzgerald P, Moloney RD, Shanahan F, et al. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry. 2012 doi: 10.1038/mp.2012.77. [DOI] [PubMed] [Google Scholar]
- 80.Noverr MC, Huffnagle GB. The ‘microflora hypothesis’ of allergic diseases. Clin Exp Allergy. 2005;35:1511–20. doi: 10.1111/j.1365-2222.2005.02379.x. [DOI] [PubMed] [Google Scholar]
- 81.Becker KG. Autism, asthma, inflammation, and the hygiene hypothesis. Med Hypotheses. 2007;69:731–40. doi: 10.1016/j.mehy.2007.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Sandler RH, Finegold SM, Bolte ER, Buchanan CP, Maxwell AP, Väisänen ML, et al. Short-term benefit from oral vancomycin treatment of regressive-onset autism. J Child Neurol. 2000;15:429–35. doi: 10.1177/088307380001500701. [DOI] [PubMed] [Google Scholar]
- 83.Song Y, Liu C, Finegold SM. Real-time PCR quantitation of clostridia in feces of autistic children. Appl Environ Microbiol. 2004;70:6459–65. doi: 10.1128/AEM.70.11.6459-6465.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Finegold SM. Therapy and epidemiology of autism--clostridial spores as key elements. Med Hypotheses. 2008;70:508–11. doi: 10.1016/j.mehy.2007.07.019. [DOI] [PubMed] [Google Scholar]
- 85.Roy CC, Kien CL, Bouthillier L, Levy E. Short-chain fatty acids: ready for prime time? Nutr Clin Pract. 2006;21:351–66. doi: 10.1177/0115426506021004351. [DOI] [PubMed] [Google Scholar]
- 86.Sleeth ML, Thompson EL, Ford HE, Zac-Varghese SE, Frost G. Free fatty acid receptor 2 and nutrient sensing: a proposed role for fibre, fermentable carbohydrates and short-chain fatty acids in appetite regulation. Nutr Res Rev. 2010;23:135–45. doi: 10.1017/S0954422410000089. [DOI] [PubMed] [Google Scholar]
- 87.Tang Y, Chen Y, Jiang H, Robbins GT, Nie D. G-protein-coupled receptor for short-chain fatty acids suppresses colon cancer. Int J Cancer. 2011;128:847–56. doi: 10.1002/ijc.25638. [DOI] [PubMed] [Google Scholar]
- 88.Wolever TMS, Spadafora P, Eshuis H. Interaction between colonic acetate and propionate in humans. Am J Clin Nutr. 1991;53:681–7. doi: 10.1093/ajcn/53.3.681. [DOI] [PubMed] [Google Scholar]
- 89.Scheppach W. Effects of short chain fatty acids on gut morphology and function. Gut. 1994;35(Suppl):S35–8. doi: 10.1136/gut.35.1_Suppl.S35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Waldecker M, Kautenburger T, Daumann H, Busch C, Schrenk D. Inhibition of histone-deacetylase activity by short-chain fatty acids and some polyphenol metabolites formed in the colon. J Nutr Biochem. 2008;19:587–93. doi: 10.1016/j.jnutbio.2007.08.002. [DOI] [PubMed] [Google Scholar]
- 91.Maslowski KM, Vieira AT, Ng A, Kranich J, Sierro F, Yu D, et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature. 2009;461:1282–6. doi: 10.1038/nature08530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Vieira EL, Leonel AJ, Sad AP, Beltrao NR, Costa TF, Ferreira TM, et al. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J Nutr Biochem. 2011 doi: 10.1016/j.jnutbio.2011.01.007. [DOI] [PubMed] [Google Scholar]
- 93.Kimura I, Inoue D, Maeda T, Hara T, Ichimura A, Miyauchi S, et al. Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41) Proc Natl Acad Sci USA. 2011;108:8030–5. doi: 10.1073/pnas.1016088108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Vecsey CG, Hawk JD, Lattal KM, Stein JM, Fabian SA, Attner MA, et al. Histone deacetylase inhibitors enhance memory and synaptic plasticity via CREB:CBP-dependent transcriptional activation. J Neurosci. 2007;27:6128–40. doi: 10.1523/JNEUROSCI.0296-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Stefanko DP, Barrett RM, Ly AR, Reolon GK, Wood MA. Modulation of long-term memory for object recognition via HDAC inhibition. Proc Natl Acad Sci USA. 2009;106:9447–52. doi: 10.1073/pnas.0903964106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Schroeder FA, Lin CL, Crusio WE, Akbarian S. Antidepressant-like effects of the histone deacetylase inhibitor, sodium butyrate, in the mouse. Biol Psychiatry. 2007;62:55–64. doi: 10.1016/j.biopsych.2006.06.036. [DOI] [PubMed] [Google Scholar]
- 97.MacFabe DF, Cain DP, Rodriguez-Capote K, Franklin AE, Hoffman JE, Boon F, et al. Neurobiological effects of intraventricular propionic acid in rats: possible role of short chain fatty acids on the pathogenesis and characteristics of autism spectrum disorders. Behav Brain Res. 2007;176:149–69. doi: 10.1016/j.bbr.2006.07.025. [DOI] [PubMed] [Google Scholar]
- 98.MacFabe DF, Cain NE, Boon F, Ossenkopp KP, Cain DP. Effects of the enteric bacterial metabolic product propionic acid on object-directed behavior, social behavior, cognition, and neuroinflammation in adolescent rats: Relevance to autism spectrum disorder. Behav Brain Res. 2011;217:47–54. doi: 10.1016/j.bbr.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 99.Wikoff WR, Anfora AT, Liu J, Schultz PG, Lesley SA, Peters EC, et al. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc Natl Acad Sci USA. 2009;106:3698–703. doi: 10.1073/pnas.0812874106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Fetissov SO, Déchelotte P. The new link between gut-brain axis and neuropsychiatric disorders. Curr Opin Clin Nutr Metab Care. 2011;14:477–82. doi: 10.1097/MCO.0b013e32834936e7. [DOI] [PubMed] [Google Scholar]
- 101.Borovikova LV, Ivanova S, Zhang MH, Yang H, Botchkina GI, Watkins LR, et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature. 2000;405:458–62. doi: 10.1038/35013070. [DOI] [PubMed] [Google Scholar]
- 102.Wang X, Wang BR, Zhang XJ, Xu Z, Ding YQ, Ju G. Evidences for vagus nerve in maintenance of immune balance and transmission of immune information from gut to brain in STM-infected rats. World J Gastroenterol. 2002;8:540–5. doi: 10.3748/wjg.v8.i3.540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kunze WA, Mao YK, Wang B, Huizinga JD, Ma X, Forsythe P, et al. Lactobacillus reuteri enhances excitability of colonic AH neurons by inhibiting calcium-dependent potassium channel opening. J Cell Mol Med. 2009;13(8B):2261–70. doi: 10.1111/j.1582-4934.2009.00686.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Bercik P, Verdu EF, Foster JA, Macri J, Potter M, Huang XX, et al. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. . Gastroenterology. 2010;139:2102–12. doi: 10.1053/j.gastro.2010.06.063. [DOI] [PubMed] [Google Scholar]
- 105.Ivanov II, Frutos Rde L, Manel N, Yoshinaga K, Rifkin DB, Sartor RB, et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe. 2008;4:337–49. doi: 10.1016/j.chom.2008.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Gaboriau-Routhiau V, Rakotobe S, Lécuyer E, Mulder I, Lan A, Bridonneau C, et al. The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity. 2009;31:677–89. doi: 10.1016/j.immuni.2009.08.020. [DOI] [PubMed] [Google Scholar]
- 107.Ivanov II, Atarashi K, Manel N, Brodie EL, Shima T, Karaoz U, et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139:485–98. doi: 10.1016/j.cell.2009.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hall JA, Bouladoux N, Sun CM, Wohlfert EA, Blank RB, Zhu Q, et al. Commensal DNA limits regulatory T cell conversion and is a natural adjuvant of intestinal immune responses. Immunity. 2008;29:637–49. doi: 10.1016/j.immuni.2008.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Diamond B, Huerta PT, Tracey K, Volpe BT. It takes guts to grow a brain: Increasing evidence of the important role of the intestinal microflora in neuro- and immune-modulatory functions during development and adulthood. Bioessays. 2011;33:588–91. doi: 10.1002/bies.201100042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Uribe A, Alam M, Johansson O, Midtvedt T, Theodorsson E. Microflora modulates endocrine cells in the gastrointestinal mucosa of the rat. Gastroenterology. 1994;107:1259–69. doi: 10.1016/0016-5085(94)90526-6. [DOI] [PubMed] [Google Scholar]
- 111.Bates JM, Mittge E, Kuhlman J, Baden KN, Cheesman SE, Guillemin K. Distinct signals from the microbiota promote different aspects of zebrafish gut differentiation. Dev Biol. 2006;297:374–86. doi: 10.1016/j.ydbio.2006.05.006. [DOI] [PubMed] [Google Scholar]
- 112.Reinhardt C, Reigstad CS, Bäckhed F. Intestinal microbiota during infancy and its implications for obesity. J Pediatr Gastroenterol Nutr. 2009;48:249–56. doi: 10.1097/MPG.0b013e318183187c. [DOI] [PubMed] [Google Scholar]
- 113.Drucker DJ. Glucagon-like Peptide 2. Trends Endocrinol Metab. 1999;10:153–6. doi: 10.1016/S1043-2760(98)00136-2. [DOI] [PubMed] [Google Scholar]
- 114.Cani PD, Possemiers S, Van de Wiele T, Guiot Y, Everard A, Rottier O, et al. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut. 2009;58:1091–103. doi: 10.1136/gut.2008.165886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhang H, DiBaise JK, Zuccolo A, Kudrna D, Braidotti M, Yu Y, et al. Human gut microbiota in obesity and after gastric bypass. Proc Natl Acad Sci USA. 2009;106:2365–70. doi: 10.1073/pnas.0812600106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.le Roux CW, Welbourn R, Werling M, Osborne A, Kokkinos A, Laurenius A, et al. Gut hormones as mediators of appetite and weight loss after Roux-en-Y gastric bypass. Ann Surg. 2007;246:780–5. doi: 10.1097/SLA.0b013e3180caa3e3. [DOI] [PubMed] [Google Scholar]
- 117.Korner J, Inabnet W, Febres G, Conwell IM, McMahon DJ, Salas R, et al. Prospective study of gut hormone and metabolic changes after adjustable gastric banding and Roux-en-Y gastric bypass. Int J Obes (Lond) 2009;33:786–95. doi: 10.1038/ijo.2009.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Woodard GA, Encarnacion B, Downey JR, Peraza J, Chong K, Hernandez-Boussard T, et al. Probiotics improve outcomes after Roux-en-Y gastric bypass surgery: a prospective randomized trial. J Gastrointest Surg. 2009;13:1198–204. doi: 10.1007/s11605-009-0891-x. [DOI] [PubMed] [Google Scholar]