Abstract
Purpose
Metastatic involvement of the sacrum is rare and there is a paucity of studies which deal with the management of these tumours since most papers refer to primary sacral tumours. This study aims to review the available literature in the management of sacral metastatic tumours as reflected in the current literature.
Methods
A systematic review of the English language literature was undertaken for relevant articles published over the last 11 years (1999–2010). The PubMed electronic database and reference lists of key articles were searched to identify relevant studies using the terms “sacral metastases” and “metastatic sacral tumours”. Studies involving primary sacral tumours only were excluded. For the assessment of the level of evidence quality, the CEBM (Oxford Centre of Evidence Based Medicine) grading system was utilised.
Results
The initial search revealed 479 articles. After screening, 16 articles identified meeting our inclusion criteria [1 prospective cohort study on radiosurgery (level II); 2 case series (level III); 4 retrospective case series (level IV) and 9 case reports (level IV)].
Conclusion
The mainstay of management for sacral metastatic tumours is palliation. Preoperative angioembolisation is shown to be of value in cases of highly vascularised tumours. Radiotherapy is used as the primary treatment in cases of inoperable tumours without spinal instability where pain relief and neurological improvement are attainable. Minimal invasive procedures such as sacroplasties were shown to offer immediate pain relief and improvement with ambulation, whereas more aggressive surgery, involving decompression and sacral reconstruction, is utilised mainly for the treatment of local advanced tumours which compromise the stability of the spine or threaten neurological status. Adjuvant cryosurgery and radiosurgery have demonstrated promising results (if no neurological compromise or instability) with local disease control.
Keyword: Sacral metastases
Introduction
The skeleton is the third most common site of metastasis after the lungs and liver [1], with the spinal column most frequently involved [1]. Spinal metastases develop in 5–10 % of all cancer patients during the course of their illness [2] with sacral deposits representing the minority of spinal secondaries [3]. Spinal metastatic infiltrations predominantly occur in the thoracic region, followed by the lumbar spine and the lumbosacral junction [4]. Breast, lung, renal, thyroid and prostate tumours form the predominant primary sources, less common primary lesions include lymphoma, melanoma and tumours of unknown origin [4–8]. Spread is mainly by haematogenic dissemination, although direct invasion through locally recurrent pelvic tumours is not uncommon [2]. Sacral metastases are relatively rare pathologies and their management constitutes a complex medical problem. They have become more prevalent in recent years, as the rate of survival improved for many common cancers. Unfortunately, sacral metastases are usually diagnosed in advanced stages when they have already extended beyond the bony margins and around the sacral nerves and other surrounding organs [9]. The clinical presentation depends largely on the structures the tumour erodes or compresses, and a thorough understanding of the anatomy is essential in evaluating structural and functional disorders of the sacrum.
Investigations
Imaging
Plain radiographs are often the first imaging modality performed. However, these may remain inadequate because of the difficulty in evaluating the sacrum on plain X-ray films [10]. The accuracy of the initial radiologic evaluation in patients with proven sacral pathological entities has been demonstrated to be as low as 17 % in one study. Attention should be paid to try to identify loss of the sacral articulate lines, as this can strongly correlate with the presence of metastatic disease [11].
More accurate visualisation of the tumour may be obtained by means of magnetic resonance imaging (MRI), which remains the optimum method for assessing spinal metastatic disease [12–15]. This is used to visualise the relationship between the tumour, bone and soft tissues. The combination of T1- and T2-weighted imaging is suitable for evaluating nerve root, muscle and visceral structure involvement.
Computerised tomography (CT) is essential for defining the osseous anatomy and is invaluable for planning reconstructive surgical procedures. CT scan demonstrates the osseous relations in detail and can also show the presence of intratumoural calcification. In addition to the above, CT scan offers the advantage of a three-dimensional reconstruction of the image and also improves the accuracy of needle biopsy (CT-guided needle biopsy) increasing significantly the diagnostic value of the procedure [16]. Both investigations combined represent the most effective way of evaluating patients with spinal metastases. The site and extent of metastatic tumours together with the extent of involvement of the sacroiliac joint are invaluable factors for preoperative planning, because they influence the surgical strategy and may determine the final surgical outcome.
Nuclear bone scan (scintigraphy) is used mainly to determine if the lesion is monostotic or polyostotic. It is considered as a first-line imaging technique when there is strong clinical suspicion for bone abnormality [17, 18]. However, although it has strong sensitivity for identifying bone lesions, bone scintigraphy lacks specificity in identifying the nature of the abnormality. Therefore, this investigation is frequently used as part of a larger workup for systemic disease than as a stand-alone diagnostic tool. Generally, the presence of multifocal disease is suggestive of bone metastases, multiple myeloma, Paget’s disease, infection and vascular tumours [12].
Single-photon emission tomography (SPECT) is an advanced form of scintigraphy which has demonstrated an increased ability in identifying lesions of any type that may not be detectable on CT, MR or plain X-rays [19, 20]. In addition, SPECT can differentiate metastatic from benign lesions [19, 21, 22].
Preoperative pelvic angiography is performed to assess hypervascular lesions or very large tumours with anticipated complex surgical planes. The diagnostic value of angiography generally is not very important [23], but the definition of the tumour vasculature facilitates the embolisation of the feeding vessels in the preoperative setting, potentially reducing the risk of intraoperative blood loss [12, 24, 25].
Biopsy
Obtaining a sample of tumour tissue is necessary for establishing the diagnosis and planning further management. Histological examination clarifies the diagnosis and may differentiate a metastasis from rare tumour-like conditions such as osteomyelitis [26] or tuberculosis [27]. Sacral biopsies can be performed as open incisional procedures, percutaneously (Tru-cut needle) or with CT guidance.
In addition, other authors suggest that percutaneous CT-guided biopsy provides the least risk and is the method of choice in establishing a diagnosis in the case of metastatic sacral tumours [28]. Nevertheless, in cases when biopsy specimens are inadequate or indeterminate [29], open biopsy would remain the method of choice. Generally, the surgeon who performs the definitive tumour resection should perform or direct the biopsy procedure. Poorly planned incisional biopsies or incomplete debulking operations performed prior to referral to a spinal oncology centre have been shown to increase the risk of local recurrence and metastasis [29–31].
Materials and methods
A systematic review of the available English-language literature on sacral metastases over the last 11 years was performed to investigate the current management and outcome of sacral metastatic tumours.
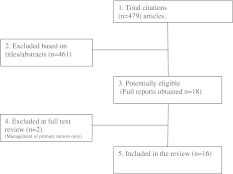
For the purpose of this review, the key words “sacral metastases” and “metastatic sacral tumours” were entered in the search engine of the “PubMed” electronic database. Reference lists of key articles were also included in the study, whereas articles involving primary sacral tumours only were excluded. Sixteen articles were identified as meeting the inclusion criteria (Fig. 1).
Fig. 1.
Flow diagram showing inclusion/exclusion criteria for studies
In this study, special attention was given to the management and outcome of sacral metastases.
Results
The initial search identified 479 papers. After screening, 16 studies were found to be eligible for inclusion (1 prospective cohort study on radiosurgery (level II) [32]; 2 case series (level III) [28, 33]; 4 retrospective case series (level IV) [2, 10, 34, 35] and 9 case reports (level IV) [36–44]; Table 1).
Table 1.
Levels of evidence of all included studies and demographic data
| References | Type | Level of evidence | No. of patients | Sex | Age | Primary tumour | Comorbidities | |
|---|---|---|---|---|---|---|---|---|
| 1 | Gerszten et al. [32] | Prospective cohort study | II | 393 pt/500 lesions, 103 sacral lesions | 251F, 142M | 56 (18–85) | RCC 93, breast 83, lung 80, melanoma 38, colon 32, sarcoma 26, prostate 24, MM 18, unknown 14, squamous cell 12, thyroid 11, other 69 | N/A |
| 2 | Takayuki Akasu et al. [33] | Prospective case series | III | 44 | 35M, 9F | 55 (32–73) | Recurrent rectal ca | N/A |
| 3 | Nader et al. [28] | Literature review/case series | III | 19 | N/A | N/A | RCC 13, breast ca 2, melanoma 1, M. myeloma 1, colon ca 1, liposarcoma 1 | N/A |
| 4 | Kollender et al. [2] | Retrospective case series | IV | 14 pt/5 pt sacral metastases | 7M, 7F | 42 (14–74) | 2 RCC, 2 MC, 1 colon ca | N/A |
| 5 | Ozdemir et al. [10] | Retrospective case series | IV | 65 pt, (34 pt sacral mets) | 25F, 21M | 49 (12–83) | 9 breast, 7 MM, 4 prostate, 3 lung, 3 colon, 2 leiomyosarcoma, 2 lymphoma, 1 ovary, 1RCC, 1 thyroid, 1 larynx | N/A |
| 6 | Dozois et al. [34] | Retrospective case series | IV | 9 | 7M, 2F | 63 (38–78) | Recurrent rectal ca | All had previous resection surgery |
| 7 | Fujibayashi et al. [35] | Retrospective case series | IV | 5 | 4M, 1F | 47, 8 (29–77) | 2 lung, 2 renal, 1 paraganglioma | N/A |
| 8 | Zhang et al. [36] | Case report | IV | 2 | 1M, 1F | 62M, 38F | 62M lymphoma, 38F lung ca | 62M skeletal mets, 38F None |
| 9 | Lee et al. [37] | Case report | IV | 1 | F | 51 | Meningioma | Tumour excision from R frontal lobe |
| 10 | Toro et al. [38] | Case report | IV | 1 | M | 65 | HCC | Hep C(+) segmentectomy child-pugh st A |
| 11 | Uemura et al. [39] | Case report | IV | 1 | M | 76 | HCC | N/A |
| 12 | Kakutani et al. [40] | Case report | IV | 1 | M | 52 | Melanoma | Lung mets |
| 13 | Nebreda et al. [41] | Case report | IV | 1 | F | 48 | Small cell lung ca | N/A |
| 14 | Menegaz et al. [42] | Case report | IV | 1 | F | 45 | Choriocarcinoma | Previous uterine curettage for hydatidiform mole |
| 15 | Turgut et al. [43] | Case report | IV | 1 | M | 63 | Mercel cell ca (skin) | NO |
| 16 | Albareda et al. [44] | Case report | IV | 1 | F | 62 | Endometrial adeno ca | Surgery for endometrial ca |
HCC hepatocellular ca, RCC renal cell ca, MM multiple myeloma, PNET primitive neuroectodermal tumour, MC metastatic carcinoma
Clinical presentation
Metastatic tumours represent the most common malignancy to occur in the sacrum and can signify advanced disease. Sacral neoplasms generally grow insidiously causing ambiguous symptoms in the early stages, which may result in delayed diagnosis [9]. This has been reported in numerous studies [9, 10] despite the fact that metastatic lesions are generally more aggressive in terms of growth and local invasion. Ozdemir et al. [10], in a series of 34 cases with sacral metastases, reported distal organ involvement and widespread spinal metastases in 61 and 43 %, respectively, at the time of diagnosis. Nader et al. [28] in his study of 19 cases with sacral metastases reported 68 % of extraspinal metastases and 53 % involvement of multiple spinal levels at the time of diagnosis.
This late diagnosis may be explained by the ability of the spacious sacral canal to permit asymptomatic tumour expansion, the difficulty in evaluating the sacrum on plane X-ray films which is often the first imaging modality performed and finally the non-specific nature of the presenting symptoms. In most studies, the initial symptom in the clinical presentation of the patients is pain (Table 2). Local pain may present due to the periosteal stretching from the tumour growth and/or the local inflammatory process or mechanically as a result of instability. Radicular pain from irritation of a nerve root [9, 10, 29–31] occurs due to nerve root compression or even tumour infiltration. Radicular pain may radiate uni- or bilaterally into buttocks, posterior thigh or leg, external genitalia and perineum [45–47]. The natural neurological history of an expanding sacral lesion is usually characterised by sensory, multiradicular deficit which progresses to motor deficit and eventually causes bladder, bowel and/or sexual, dysfunction [47]. These symptoms may evolve together or separately in any possible order and combination [48]. The involvement of lumbosacral nerve roots leads to specific motor and sensory deficits [46–48] which correlate with the affected dermatome and/or myotome (Table 2).
Table 2.
Clinical presentation of all patients from studies
| References | Level of metastases | Clinical presentation | Preoperative mobility | Preoperative continence | Preoperative Angiotherapy Radiotherapy Chemotherapy |
|
|---|---|---|---|---|---|---|
| 1 | Gerszten et al. [32] | Sacral (103 levels) | Pain 336, progressive neurology 32 | N/A | N/A | 344/500 radiotherapy |
| 2 | Takayuki Akasu et al. [33] | 24 solitary pelvic, 12 pelvic mets, 8 distant mets | 15 no pain, 17 perineum pain, 7 buttock pain, 3 thigh pain, 2 leg pain | N/A | N/A | 13 pt radiotherapy |
| 3 | Nader et al. [28] | 18 S1,13 S2, 3 S3, 2 S4, L/S 4, SI 4, rest of the spine 10, extraspinal 13 | Motor 3 pt, sensory 8 pt, pain 18 pt | 16 mobile (2 independently) | 3 urine incontinence | N/A |
| 4 | Kollender et al. [2] | 2 mid sacrum, 2 left sacrum, 1 whole sacrum | Severe BP groin and leg pain, 9(64 %) bowel bladder dysfunction | N/A | 9 pt bowel bladder problems | All radiotherapy, 2 angio-embolisation. |
| 5 | Ozdemir et al. [10] | N/A | Pain 58.7 %, neurology 34.8 % | N/A | N/A | N/A |
| 6 | Dozois et al. [34] | 6 S2, 2 S1, 1 L–S | N/A | N/A | N/A | 7 radio, 1 chemo |
| 7 | Fujibayashi et al. [35] | LS joint, sacrum | All had neurology and 4 severe BP, 1 mild BP | 3 Bedridden, 1 wheelchair, 1 independently | N/A | N/A |
| 8 | Zhang et al. [36] | M S1 and T12, L1, L2, L3, F S1 | M No neurology/severe back pain, F no neurology/severe back pain | Normal | Normal | M chemotherapy and radiotherapy, F nothing |
| 9 | Lee et al. [37] | Left sacrum, ilium | Left S1 reduced sensation/power (4/5), low back pain and left sciatica | N/A | N/A | No |
| 10 | Toro et al. [38] | Right S1–S5, left S1, S2 | N/A/severe back pain | Difficulty in walking, pain | N/A | No |
| 11 | Uemura et al. [39] | 1st left sacrum, 2nd right sacrum, (3 months after 1st procedure) | N/A/persisting severe back pain | N/A | N/A | Left angioembolisation and radiotherapy |
| 12 | Kakutani et al. [40] | Left S1, S2, S3, S4 left ilium | Back pain, left S1 radicular pain, weakness, dysuria, left thigh, calf dysaesthesia | N/A | Dysuria | No |
| 13 | Nebreda et al. [41] | Left S1, S2 left SIJ | Back pain and left thigh pain | Normal | Normal | Chemotherapy and cyberknife |
| 14 | Menegaz et al. [42] | L2–S1 | BP, right leg pain weakness and numbness, urinary incontinence, Cauda equina | Difficulty in walking | Urine incontinence | No |
| 15 | Turgut et al. [43] | L5–S1 | Weakness, hypaesthesia left L5–S1 | Difficulty in walking | Normal | No |
| 16 | Albareda et al. [44] | Lower sacrum (S4) | Asymptomatic | Normal | Normal | No |
Treatment
For the sacral metastatic lesions, the treatment is typically palliative, aiming at pain control and salvage of neurologic function [4] (Table 3). Currently, there are two traditional methods for localised intervention in the case of sacral metastasis—namely, radiotherapy and surgery.
Table 3.
Treatment, complications and survival of all patients
| References | Procedure | Postoperative chemotherapy/radiotherapy |
Complications | Clinical outcome | Postoperative mobility |
Postoperative continence |
Survival | |
|---|---|---|---|---|---|---|---|---|
| 1 | Gerszten et al. [32] | Radiosurgery | No | None | 86 % pain relief, 84 % neur. impr. | N/A | N/A | 21 months (3–53) |
| 2 | Takayuki Akasu et al. [33] | Abdominal sacral resection, levels of transection, S2 in 6, S2–3 in 19, S3 in 5, S3–4 in 11, S4 in 1, S4–5 in 2 | N/A | 27 patients morbidity (61 %), 10 (23 %) major complications, 17 (38 %) had minor complications | N/A | N/A | N/A | 2.3 years, (1–15.8) |
| 3 | Nader et al. [28] | 18 laminectomies, 13 vertebrectomies, 12 instrumentation | N/A | Major 16 %, minor 16 % | 8 pt improved, 3 pt worse neurology, 11/16 pain relief, 3/16 no pain | 16 pt, (7 independently) | N/A | 21, 8 months, (5–38.5) |
| 4 | Kollender et al. [2] | Post decompression cryosurgery | N/A | 1 CSF leakage | N/A | N/A | 1 urine incontinence | 4 died, (6–36 months), 1 alive |
| 5 | Ozdemir et al. [10] | 13 pt, curettage or resection and cementation | 21 radio/chemo or combination | In 10/46 cases | Pain improvement | N/A | N/A | 36 months, (30–120) |
| 6 | Dozois et al. [34] | High sacrectomy | 8 chemo | 1 minor, 4 major | N/A | 8 independently | N/A | 5 year 30 % |
| 7 | Fujibayashi et al. [35] | Post instrumentation, T5-Ilium, L2-Ilium, T9-Ilium, L3-Ilium × 2 | N/A | 1 infection, 1 wound breakdown, 1 rod breakdown | Both improved | All independently | N/A | 28, 4, (3 months–6 years) |
| 8 | Zhang et al. [36] | S1 percutaneous sacroplasty | No | M none, F cement leak in needle tract | No neurology pain relief | Both independently | Normal | N/A, M last F up 3/12, F last F up 20/52 |
| 9 | Lee et al. [37] | Total excision in 2 stages, AP approach | Radiation | Mild constipation and urine retention | Left saddle parasthesia, Pain N/A | Independently mobile (left S2–S5 and right S3–S5 sacrificed) | Mild constipation/urinary retention | N/A, last follow up 16/12 |
| 10 | Toro et al. [38] | Sacroplasty at all lesions | No | No | Pain free | Improvement in walking | N/A | N/A last follow up 8/12 |
| 11 | Uemura et al. [39] | Perc. sacroplasty, 1st on left (twice), 2nd on right | Radiation | (1st bleeding intraop), transient numbness, (24 h) R leg | Pain free | Improved (walker) | N/A | 72 days |
| 12 | Kakutani et al. [40] | No surgery chemo and radiotherapy | No | N/A | Both improved | Independently mobile | N/A | 9 months |
| 13 | Nebreda et al. [41] | 1st S1, S1, 2nd SIJ cementation (2 months following the 1st) | No | No | 1st transient pain relief, 2nd pain free | Independently mobile | Normal | 40 days |
| 14 | Menegaz et al. [42] | Chemotherapy and radiotherapy | No | N/A | Both improved | Independently mobile | N/A | 7 months |
| 15 | Turgut et al. [43] | L5 laminectomy and subtotal resection | Chemotherapy | N/A | N/A | N/A | N/A | 2 months |
| 16 | Albareda et al. [44] | Sacrectomy en block resection | Radiotherapy | No | No | Normal | Normal | Last follow up 26/12 |
Radiotherapy may be chosen as the initial therapy for radiosensitive sacral metastases in patients without spinal instability or acute neurological deterioration where significant pain reduction and neurologic improvement are attainable [49, 50]. Radiosensitivity varies among primary tumour types. In general, prostate and lymphoid tumours are radiosensitive, breast cancer is 70 % sensitive and 30 % resistant, and gastrointestinal and renal cell tumours, like melanomas, are radioresistant. Indications for radiation therapy include the presence of radiosensitive tumours such as lymphoma, myeloma and small cell carcinoma of the lung, no neurological impairment or spinal instability and no mechanical pain or significant bony compromise of the spinal canal [49, 50].
Despite the fact that older publications demonstrated that radiotherapy alone could be as effective as a combination of decompressive surgery with adjuvant radiotherapy [51–53], more recent studies have shown that decompressive surgery with/out reconstruction and stabilisation followed by postoperative radiotherapy was superior than radiotherapy alone [54, 55].
Spinal stereotactic radiosurgery (SRS) is an emerging form of radiotherapy, which allows more precise radiation delivery and high-dose hypofractionation.
These characteristics allow the administration of tumouricidal radiation dose even for radioresistant tumours, with minimal exposure of the surrounding normal tissues. Current commercial spinal SRS systems include the Cyber Knife (Accuray Incorporated, Sunnyvale, California) and Novalis (BrainLAB, Heinstetten, Germany) according to a recent prospective study of 500 cases with spinal metastases (including 103 cases of sacral metastatic lesions; Table 3); radiosurgery is safe and provides incremental benefit over conventional radiotherapy with a more durable symptomatic response and local control independent of the histology [32]. In addition, other major benefits of radiosurgery include the relatively short treatment time, which can be in an outpatient setting and the minimal or complete absence of side effects [56]. Currently, spinal radiosurgery is often employed as a salvage technique for those cases in which further conventional irradiation or surgery is not appropriate and the most frequent indication is persisting pain.
However, radiosurgery is not indicated in cases of neurologic deficit caused from bony compression to neural structures. In addition, it does not address spinal instability issues and has a limited ability to deliver radiation to larger lesions [56, 57].
Preoperative angioembolisation of hypervascular metastatic lesions reduces intraoperative blood loss and improves the surgeon’s ability to resect the tumour. In addition, some studies have shown that embolisation may cause tumour growth arrest and subsequently pain alleviation and shorter hospital stay [58–60]. The timing of the preoperative embolisation is also important. Generally, it is recommended that embolisation should be performed as close as possible to the time of surgery. Typically, best results are achieved when surgery is performed within 24–48 h after embolisation [58, 61–63].
Indications for surgical intervention include progressive neurologic dysfunction or persistent pain that is unresponsive to radiation therapy, the need for a diagnostic biopsy and pathologic instability [10, 64]. The use of chemotherapy for the management of spinal tumours is limited. Except from tumours such as Ewing’s sarcoma and neuroblastoma which are chemosensitive, anti-tumour drugs have a limited role in the treatment of spinal metastases in general. Chemotherapeutic agents can be classified into anti-tumour drugs and those which minimise the secondary effects of the tumour [65]. However, drugs which prevent or ameliorate the effects of spinal tumours, such as corticosteroids, bisphosphonates and analgesics, are widely used.
Sacroplasty is gaining favour in cases of metastatic disease without instability or neurologic compromise and represents a minimally invasive alternative to open procedures [66]. The available data on this are based primarily on level IV studies [36, 38, 39, 41] and all cases treated with sacroplasty have shown immediate improvement in mobility and significant pain relief (Table 3).
Surgical intervention in the sacrum represents a complex problem, which, depending on the surgical procedure, may have a high morbidity including massive bleeding, infection, CSF leakage, severe neurological deficits, bowel/bladder dysfunction and spinal or pelvic instability [5]. Six studies report on their results of surgery ranging from posterior decompression with(out) cementation and instrumentation [2, 28, 35, 43] to abdominal sacral resection [33] and en bloc sacrectomy [34, 44].
Such is the heterogeneity of these data that a comprehensive analysis is not possible. What is clear is that optimal management of patients with sacral metastatic disease necessitates a multidisciplinary approach involving specialists in oncology, general surgery, histopathology, spinal surgery and radiology. The treatment decision is based on specific criteria such as the health status of the patient, anatomical characteristics specific to patient and lesion, and the biology of the tumour [67]. The final decision depends on the clinical presentation and the preference of the physician/surgeon and the patient [51].
Various systems using a range of prognostic factors have been devised and correlated with the clinical outcome to predict survival. They allow the recognition of patients who are unlikely to do well after surgery and the choice of suitable management. Recognised systems include that of Tomita et al. [68] and the revised scheme of Tokuhashi et al. [69].
Clinical outcome and complications
The functional outcome of sacral tumours depends greatly on the level of neurologic deficit. In general, patients with root involvement distal to S3 have limited deficits, preservation of sphincter and motor function, but possible sexual dysfunction. On the contrary, great variability is seen when there is involvement of the S2–S3 roots.
Middle resections involving the S2–3 nerve roots rarely cause motor disjunction, but saddle anaesthesia and sphincter dysfunction are common [70]. Stener and Gutenberg [71] observed that continent bowel and bladder function was possible when ≥1 S-2 root was preserved. Lower limb muscle weakness can be observed when S1 nerve roots are sacrificed; however, patients with intact L5 nerves, generally, are able to walk without external support.
Recurrence and survival rates are multifactorial and vary between different studies in the literature (Table 3). Kollender et al. [2] in a study involving 14 sacral tumours (5 of which were metastatic), surgically resected with the use of adjuvant cryotherapy, reported a 6–36 months survival rate in the metastatic group and no local recurrences. Osdemir et al. [10] reviewed the outcome in a series of 34 patients who underwent surgical treatment of malignant sacral tumours. In the group with sacral metastases, they reported 19 % recurrence and 23 % complication rate (16 % characterised as major) following surgery.
Nader et al. [28] in a more recent retrospective study of 19 patients who underwent resection of sacral metastatic lesions reported improved neurology on discharge in eight patients (42 %) and only three cases with deterioration (16 %).
From the 16 patients who reported pain scores based on a visual analogue scale (VAS), 11 reported significant improvement (69 %) and 5 no pain at all (31 %) on discharge. In addition, mobility improved postoperatively and seven patients (37 %) were able to mobilise without assistance postoperatively in comparison with only two (11 %) preoperatively. In this study, both major and minor complications occurred in 16 % of patients, respectively, and the mean survival rate was estimated to be 21.8 months following the first surgery.
Finally, systemic reviews are only as good as the research articles available. With sacral metastases, the studies are very inhomogeneous with differing methods of treatment and uncontrolled cohorts with limited data. It therefore becomes even more necessary to highlight the importance of high-quality multicentric prospective studies and to follow a comprehensive algorithm where the final surgical technique (if applicable) is also well planned and prospectively selected.
Discussion
Metastatic lesions of the sacrum are rare, but pose a complex problem for surgical management. The clinical pattern of presentation depends on the anatomical location of the tumour and whether it invades or compresses neighbouring structures. Early diagnosis is often difficult because symptoms of bladder, bowel, epigastric and sacral plexus compression become evident late on in the presentation. Tumours of breast, lung, renal, thyroid and prostate form the predominant primary sources that metastasis to the sacrum. Evaluation and treatment of sacral metastatic tumours require a multidisciplinary approach, ideally at specialist centres with comprehensive care and experience.
Therapeutic approaches are still a matter of debate with few studies of levels II–IV evidence. However, technical advances which allow a more aggressive and effective surgical procedure to be used have led to improved final outcomes. Angioembolisation can be used effectively as an adjuvant treatment for the management of hypervascular metastatic sacral tumours. Radiation therapy is effective for the treatment of radiosensitive tumours in the absence of neurological impairment or spinal instability, mechanical pain or significant bony compromise of the spinal canal. Radiosurgery is a promising new therapy and sacroplasty allows percutaneous stabilisation with good pain relief. However, in cases of progressive neurological deterioration, open surgical management is the more effective treatment.
Acknowledgments
We would like to thank Professor Peter Varga for proof reading this manuscript.
Conflict of interest
None.
References
- 1.Aaron AD. The management of cancer metastatic to bone. JAMA. 1994;272(15):1206–1209. doi: 10.1001/jama.1994.03520150074040. [DOI] [PubMed] [Google Scholar]
- 2.Kollender Y, Meller I, Bickets J, et al. Role of adjuvant cryosurgery in intralesional treatment of sacral tumors. Cancer. 2003;97:2830–2838. doi: 10.1002/cncr.11383. [DOI] [PubMed] [Google Scholar]
- 3.Cummings BJ, Hobson DI, Bush RS. Chordoma :the results of megavoltage radiation therapy. Int J Radiat Oncol Biol Phys. 1983;9:633–642. doi: 10.1016/0360-3016(83)90228-6. [DOI] [PubMed] [Google Scholar]
- 4.Raque GH, Jr, Vitaz TW, Shields CB. Treatment of neoplastic diseases of the sacrum. J Surg Oncol. 2001;76:301–307. doi: 10.1002/jso.1050. [DOI] [PubMed] [Google Scholar]
- 5.Wuisman P, Lieshout O, Sugihara S, Dijk M. Total sacrectomy and reconstruction: oncologic and functional outcome. Clin Orthop Relat Res. 2000;381:192–203. doi: 10.1097/00003086-200012000-00023. [DOI] [PubMed] [Google Scholar]
- 6.Diel J, Ortiz O, Losada RA, Price DB, Hayt MW, Katz DS. The sacrum: pathologic spectrum, multimodality imaging, and subspecialty approach. Radiographics. 2001;21(1):83–104. doi: 10.1148/radiographics.21.1.g01ja0883. [DOI] [PubMed] [Google Scholar]
- 7.Llauger J, Palmer J, Amores S, Bague S, Camins A. Primary tumors of the sacrum: diagnostic imaging. AJR Am J Roentgenol. 2000;174(2):417–424. doi: 10.2214/ajr.174.2.1740417. [DOI] [PubMed] [Google Scholar]
- 8.Disler DG, Miklic D. Imaging findings in tumors of the sacrum. AJR Am Roentgenol. 1999;173:1699–1706. doi: 10.2214/ajr.173.6.10584822. [DOI] [PubMed] [Google Scholar]
- 9.Feldenzer JA, McGauley JL, McGillicuddy JE. Sacral and presacral tumors: problems in diagnosis and management. Neurosurgery. 1989;25(6):884–891. doi: 10.1227/00006123-198912000-00006. [DOI] [PubMed] [Google Scholar]
- 10.Ozdemir MH, Gurkan I, Yildiz Y, Yilmaz C, Saglik Y. Surgical treatment of malignant tumours of the sacrum. Eur J Surg Oncol. 1999;25(1):44–49. doi: 10.1053/ejso.1998.0598. [DOI] [PubMed] [Google Scholar]
- 11.Amorosa JK, Weintraub S, Amorosa LF, Safer JN, Rafii M. Sacral destruction: foraminal lines revisited. AJR Am J Roentgenol. 1985;145:773–775. doi: 10.2214/ajr.145.4.773. [DOI] [PubMed] [Google Scholar]
- 12.Gerber S, Ollivier L, Leclere J, et al. Imaging of sacral tumours. Skeletal Radiol. 2008;37(4):277–289. doi: 10.1007/s00256-007-0413-4. [DOI] [PubMed] [Google Scholar]
- 13.Yuh WT, Zachar CK, Barloon TJ, et al. Vertebral compression fractures: distinction between benign and malignant causes with MR imaging. Radiology. 1989;172:215–218. doi: 10.1148/radiology.172.1.2740506. [DOI] [PubMed] [Google Scholar]
- 14.Baur A, Stäbler A, Arbogast S, et al. Acute osteoporotic and neoplastic vertebral compression fractures: fluid sign at MR imaging. Radiology. 2002;225:730–735. doi: 10.1148/radiol.2253011413. [DOI] [PubMed] [Google Scholar]
- 15.Jung HS, Jee WH, McCauley TR, Ha KY, Choi KH. Discrimination of metastatic from acute osteoporotic compression fractures with MR imaging. Radiographics. 2003;23:179–187. doi: 10.1148/rg.231025043. [DOI] [PubMed] [Google Scholar]
- 16.Fourney DR, Gokaslan ZL. Sacral tumors: primary and metastatic. In: Dickman CA, Fehlings MG, Gokaslan ZL, editors. Spinal cord and spinal column tumors: principles and practice. New York: Thieme; 2006. pp. 404–419. [Google Scholar]
- 17.Algra PR, Bloem JL, Tissing H, Falke TH, Arndt JW, Verboom LJ. Detection of vertebral metastases: comparison between MR imaging and bone scintigraphy. Radiographics. 1991;11:219–232. doi: 10.1148/radiographics.11.2.2028061. [DOI] [PubMed] [Google Scholar]
- 18.Maeseneer M, Lenchik L, Everaert H, Marcelis S, Bossuyt A, Osteaux M, et al. Evaluation of lower back pain with bone scintigraphy and SPECT. Radiographics. 1999;19:901–904. doi: 10.1148/radiographics.19.4.g99jl03901. [DOI] [PubMed] [Google Scholar]
- 19.Savelli G, Chiti A, Grasselli G, Maccauro M, Rodari M, Bombardieri E. The role of bone SPECT study in diagnosis of single vertebral metastases. Anticancer Res. 2000;20:1115–1120. [PubMed] [Google Scholar]
- 20.Savelli G, Maffioli L, Maccauro M, Deckere E, Bombardieri E. Bone scintigraphy and the added value of SPECT (single photon emission tomography) in detecting skeletal lesions. Q J Nucl Med. 2001;45:27–37. [PubMed] [Google Scholar]
- 21.Bushnell DL, Kahn D, Huston B, Bevering CG. Utility of SPECT imaging for determination of vertebral metastases in patients with known primary tumors. Skeletal Radiol. 1995;24:13–16. doi: 10.1007/BF02425938. [DOI] [PubMed] [Google Scholar]
- 22.Even-Sapir E, Martin RH, Barnes DC, Pringle CR, Iles SE, Mitchell MJ. Role of SPECT in differentiating malignant from benign lesions in the lower thoracic and lumbar vertebrae. Radiology. 1993;187:193–198. doi: 10.1148/radiology.187.1.8451412. [DOI] [PubMed] [Google Scholar]
- 23.Luken MG, III, Michelsen WJ, Whelan MA, Andrews DL. The diagnosis of sacral lesions. Surg Neurol. 1981;15:377–383. doi: 10.1016/0090-3019(81)90176-2. [DOI] [PubMed] [Google Scholar]
- 24.Cristofaro R, Biagini R, Boriani S, Ricci S, Ruggieri P, Rossi G, et al. Selective arterial embolization in the treatment of aneurysmal bone cyst and angioma of bone. Skeletal Radiol. 1992;21:523–527. doi: 10.1007/BF00195235. [DOI] [PubMed] [Google Scholar]
- 25.Peh WC, Koh WL, Kwek JW, Htoo MM, Tan PH. Imaging of painful solitary lesions of the sacrum. Australas Radiol. 2007;51(6):507–515. doi: 10.1111/j.1440-1673.2007.01861.x. [DOI] [PubMed] [Google Scholar]
- 26.Nasir N, Aquilina K, Ryder DQ, et al. Garre’s chronic diffuse sclerosing osteomyelitis of the sacrum : a rare condition mimicking malignancy. Br J Neurosurg. 2006;20(6):415–419. doi: 10.1080/02688690601046819. [DOI] [PubMed] [Google Scholar]
- 27.Kumar A, Varshney MK, Trikha V. Unusual presentation of isolated sacral tuberculosis. Joint bone spine. 2006;73(6):751–752. doi: 10.1016/j.jbspin.2006.01.016. [DOI] [PubMed] [Google Scholar]
- 28.Nader R, Rhines LD, Mendel E. Metastatic sacral tumors. Neurosurg Clin N Am. 2004;15(4):453–457. doi: 10.1016/j.nec.2004.04.009. [DOI] [PubMed] [Google Scholar]
- 29.Babu NV, Titus VT, Chittaranjan S, Abraham G, Prem H, Korula RJ. Computed guided biopsy of the spine. Spine. 1994;19:2436–2442. doi: 10.1097/00007632-199411000-00013. [DOI] [PubMed] [Google Scholar]
- 30.Settle WJ, Ebraheim NA, Coombs R, Saunders RC, Jackson WT. CT-guided biopsy of metastatic sacral tumors. Orthopedics. 1990;13:753–758. doi: 10.3928/0147-7447-19900701-11. [DOI] [PubMed] [Google Scholar]
- 31.Bergh P, Kindblom LG, Gunterberg B, et al. Prognostic factors in chordoma of the sacrum and mobile spine: a study of 39 patients. Cancer. 2000;88:2122–2134. doi: 10.1002/(SICI)1097-0142(20000501)88:9<2122::AID-CNCR19>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 32.Gerszten PC, Burton SA, Ozhasoglu C, Welch WC. Radiosurgery for spinal metastases: clinical experience in 500 cases from a single institution. Spine (Phila Pa 1976) 2007;32(2):193–199. doi: 10.1097/01.brs.0000251863.76595.a2. [DOI] [PubMed] [Google Scholar]
- 33.Akasu T, Yamaguchi T, Fujimoto Y, Ishiguro S, Yamamoto S, Fujita S, Moriya Y. Abdominal sacral resection for posterior pelvic recurrence of rectal carcinoma: analyses of prognostic factors and recurrence patterns. Ann Surg Oncol. 2007;14(1):74–83. doi: 10.1245/s10434-006-9082-0. [DOI] [PubMed] [Google Scholar]
- 34.Dozois EJ, Privitera A, Holubar SD, Aldrete JF, Sim FH, Rose PS, Walsh MF, Bower TC, Leibovich BC, Nelson H, Larson DW. High sacrectomy for locally recurrent rectal cancer: can long-term survival be achieved? J Surg Oncol. 2011;103(2):105–109. doi: 10.1002/jso.21774. [DOI] [PubMed] [Google Scholar]
- 35.Fujibayashi S, Neo M, Nakamura TJ. Palliative dual iliac screw fixation for lumbosacral metastasis. Technical note. Neurosurg Spine. 2007;7(1):99–102. doi: 10.3171/SPI-07/07/099. [DOI] [PubMed] [Google Scholar]
- 36.Zhang J, Wu CG, Gu YF, Li MH. Percutaneous sacroplasty for sacral metastatic tumors under fluoroscopic guidance only. Korean J Radiol. 2008;9(6):572–576. doi: 10.3348/kjr.2008.9.6.572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lee YY, Wen-Wei Hsu R, Huang TJ, Hsueh S, Wang JY. Metastatic meningioma in the sacrum: a case report. Spine (Phila Pa 1976) 2002;27(4):E100–E103. doi: 10.1097/00007632-200202150-00022. [DOI] [PubMed] [Google Scholar]
- 38.Toro A, Pulvirenti E, Manfrè L, Carlo I. Sacroplasty in a patient with bone metastases from hepatocellular carcinoma. A case report. Tumori. 2010;96(1):172–174. doi: 10.1177/030089161009600130. [DOI] [PubMed] [Google Scholar]
- 39.Uemura A, Matsusako M, Numaguchi Y, Oka M, Kobayashi N, Niinami C, Kawasaki T, Suzuki K. Percutaneous sacroplasty for hemorrhagic metastases from hepatocellular carcinoma. AJNR Am J Neuroradiol. 2005;26(3):493–495. [PMC free article] [PubMed] [Google Scholar]
- 40.Kakutani K, Doita M, Nishida K, Miyamoto H, Kurosaka M (2008) Radiculopathy due to malignant melanoma in the sacrum with unknown primary site. Eur Spine J. 17 Suppl 2:S271–S274 (Epub 2007 Dec 13) [DOI] [PMC free article] [PubMed]
- 41.Nebreda C, Vallejo R, Aliaga L, Benyamin R. Percutaneous sacroplasty and sacroiliac joint cementation under fluoroscopic guidance for lower back pain related to sacral metastatic tumors with sacroiliac joint invasion. Pain Pract. 2011;11(6):564–569. doi: 10.1111/j.1533-2500.2010.00439.x. [DOI] [PubMed] [Google Scholar]
- 42.Menegaz RA, Resende AD, Silva CS, Barcelos AC, Murta EF. Metastasis of choriocarcinoma to lumbar and sacral column. Eur J Obstet Gynecol Reprod Biol. 2004;113(1):110–113. doi: 10.1016/j.ejogrb.2003.09.029. [DOI] [PubMed] [Google Scholar]
- 43.Turgut M, Gökpinar D, Barutça S, Erkuş M. Lumbosacral metastatic extradural Merkel cell carcinoma causing nerve root compression—case report. Neurol Med Chir (Tokyo) 2002;42(2):78–80. doi: 10.2176/nmc.42.78. [DOI] [PubMed] [Google Scholar]
- 44.Albareda J, Herrera M, Lopez Salva A, Garcia Donas J, Gonzalez R (2008) Sacral metastasis in a patient with endometrial cancer: case report and review of the literature. Gynecol Oncol.111(3):583–588 (Epub 2008 May 22) [DOI] [PubMed]
- 45.Sciubba DM, Gokaslan ZL. Diagnosis and management of metastatic spine disease. Surg Oncol. 2006;15:141–151. doi: 10.1016/j.suronc.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 46.Goldner J. Pain: extremities and spine-evaluation and differential diagnosis. In: Omer G, Spinner M, editors. Management of peripheral nerve problems. Philadelphia: WB Saunders; 1980. p. 169. [Google Scholar]
- 47.Miralbell R, Louis DN, O’Keefe D, et al. Metastatic ependymoma of the sacrum. Cancer. 1990;65:2353–2355. doi: 10.1002/1097-0142(19900515)65:10<2353::AID-CNCR2820651032>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 48.Wilson SAK. Neurology, vol 2. London: Edward Arnold; 1940. pp. 1285–1321. [Google Scholar]
- 49.Marazano E, Trippa F, Chirico L, et al. Management of metastatic spinal cord compression. Tumori. 2003;89(5):469–475. doi: 10.1177/030089160308900502. [DOI] [PubMed] [Google Scholar]
- 50.Loblaw DA, Laperrier NJ. Emergency treatment of malignant extradural spinal cord compression: an evidence based guideline. J Clin Oncol. 1998;16(4):1613–1624. doi: 10.1200/JCO.1998.16.4.1613. [DOI] [PubMed] [Google Scholar]
- 51.Gilbert RW, Kim JH, Posner JB. Epidural spinal cord compression from metastatic tumor: diagnosis and treatment. Ann Neurol. 1978;3:40–51. doi: 10.1002/ana.410030107. [DOI] [PubMed] [Google Scholar]
- 52.Vieth RG, Odom GL. Extradural spinal metastases and their neurosurgical treatment. J Neurosurg. 1965;23:501–508. doi: 10.3171/jns.1965.23.5.0501. [DOI] [PubMed] [Google Scholar]
- 53.Young RF, Post EM, King GA. Treatment of spinal epidural metastases. Randomized prospective comparison of laminectomy and radiotherapy. J Neurosurg. 1980;53:741–748. doi: 10.3171/jns.1980.53.6.0741. [DOI] [PubMed] [Google Scholar]
- 54.Patchell RA, Tibbs PA, Regine WF, Payne R, Saris S, Kryscio RJ, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366:643–648. doi: 10.1016/S0140-6736(05)66954-1. [DOI] [PubMed] [Google Scholar]
- 55.Klimo P, Jr, Schmidt MH. Surgical management of spinal metastases. Oncologist. 2004;9:188–196. doi: 10.1634/theoncologist.9-2-188. [DOI] [PubMed] [Google Scholar]
- 56.Gerszten PC, Burton SA, Ozhasoglu C, Welch WC. Radiosurgery for spinal metastases: clinical experience of 500 cases from a single institution. Spine (Phila Pa 1976) 2007;32(2):193–199. doi: 10.1097/01.brs.0000251863.76595.a2. [DOI] [PubMed] [Google Scholar]
- 57.Witham TF, Khavkin YA, Gallia GL, Wolinsky JP, Gokaslan ZL. Surgery insight: current management of epidural spinal cord compression from metastatic spine disease. Nat Clin Pract Neurol. 2006;2:87–94. doi: 10.1038/ncpneuro0116. [DOI] [PubMed] [Google Scholar]
- 58.Hess T, Kramann B, Schmidt E, et al. Use of preoperative vascular embolisation in spinal metastasis resection. Arch Orthop Trauma Surg. 1997;116(279):282. doi: 10.1007/BF00390053. [DOI] [PubMed] [Google Scholar]
- 59.Prabhu VC, Bilsky MH, Jambhekar K, et al. Results of preoperative embolization for metastatic spinal neoplasms. J Neurosurg (Spine 2) 2003;98:156–164. doi: 10.3171/spi.2003.98.2.0156. [DOI] [PubMed] [Google Scholar]
- 60.Sundaresan N, Choi IS, Hughes JE, et al. Treatment of spinal metastases from kidney cancer by presurgical embolization and resection. J Neurosurg. 1990;73:548–554. doi: 10.3171/jns.1990.73.4.0548. [DOI] [PubMed] [Google Scholar]
- 61.Gellad FE, Sadato N, Numaguchi Y, et al. Vascular metastatic lesions of the spine: preoperative embolization. Radiology. 1990;176:683–686. doi: 10.1148/radiology.176.3.2389026. [DOI] [PubMed] [Google Scholar]
- 62.Chiras J, Cognard C, Rose M, et al. Percutaneous injection of an alcoholic embolizing emulsion as an alternative preoperative embolization for spine tumor. AJNR Am J Neuroradiol. 1993;14(5):1113–1117. [PMC free article] [PubMed] [Google Scholar]
- 63.Smith J, Wixon D, Watson RC. Giant-cell tumor of the sacrum: clinical and radiologic features in 13 patients. J Can Assoc Radiol. 1979;30:34–39. [PubMed] [Google Scholar]
- 64.Quraishi NA, Gokaslan ZL, Boriani S. Current concepts in the surgical management of metastatic epidural spinal cord compression. J Bone Joint Surg. 2010;92B(8):1054–1060. doi: 10.1302/0301-620X.92B8.22296. [DOI] [PubMed] [Google Scholar]
- 65.Yu MK, Buys SS. Medical management of skeletal metastasis. Neurosurg Clin N Am. 2004;15:529–536. doi: 10.1016/j.nec.2004.04.015. [DOI] [PubMed] [Google Scholar]
- 66.Masala S, Konda D, Massari F, et al. Sacroplasty and iliac osteoplasty under combined CT and fluoroscopic guidance. Spine. 2006;31(18):E667–E669. doi: 10.1097/01.brs.0000231962.04739.ac. [DOI] [PubMed] [Google Scholar]
- 67.Polly DW, Jr, Chou D, Sembrano JN, Ledonio CG, Tomita K. An analysis of decision making and treatment in thoracolumbar metastases. Spine. 2009;34(Suppl):118–127. doi: 10.1097/BRS.0b013e3181ba6d02. [DOI] [PubMed] [Google Scholar]
- 68.Tomita K, Kawahara N, Kobayashi T, et al. Surgical strategy for spinal metastases. Spine. 2001;26:298–306. doi: 10.1097/00007632-200102010-00016. [DOI] [PubMed] [Google Scholar]
- 69.Tokuhashi Y, Matsuzaki H, Oda H, Oshima M, Ryu J. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine. 2005;30:2186–2191. doi: 10.1097/01.brs.0000180401.06919.a5. [DOI] [PubMed] [Google Scholar]
- 70.Biagini R, Ruggieri P, Mercuri M, Capanna R, Briccoli A, Perin S, et al. Neurologic deficit after resection of the sacrum. Chir Organi Mov. 1997;82:357–372. [PubMed] [Google Scholar]
- 71.Stener B, Gunterberg B. High amputation of the sacrum for extirpation of tumors. Principles and technique. Spine. 1978;3:351–366. doi: 10.1097/00007632-197812000-00009. [DOI] [PubMed] [Google Scholar]