Abstract
It was recently proposed that the improved oral bioavailability of genistein aglycone and conjugates in Bcrp1(−/−) mice is mainly due to increased intestinal absorption of aglycone and subsequent elevated exposure to conjugation enzymes. Here we tested this proposed mechanism and found that intestinal absorption of genistein aglycone did not increase in Bcrp1(−/−) mice compared with wild-type mice using an in situ mouse intestinal perfusion model and that inhibition of breast cancer resistance protein (BCRP) in Caco-2 cells also did not significantly increase permeability or intracellular concentration of aglycone. Separately, we showed that 5- to 10-fold increases in exposures of conjugates and somewhat lower fold increases (<2-fold) in exposures of aglycone were apparent after both oral and intraperitoneal administration in Bcrp1(−/−) mice. In contrast, the intestinal and biliary excretion of genistein conjugates significantly decreased in Bcrp1(−/−) mice without corresponding changes in aglycone excretion. Likewise, inhibition of BCRP functions in Caco-2 cells altered polarized excretion of genistein conjugates by increasing their basolateral excretion. We further found that genistein glucuronides could be hydrolyzed back to genistein, whereas sulfates were stable in blood. Because genistein glucuronidation rates were 110% (liver) and 50% (colon) higher and genistein sulfation rates were 40% (liver) and 42% (colon) lower in Bcrp1(−/−) mice, the changes in genistein exposures are not mainly due to changes in enzyme activities. In conclusion, improved bioavailability of genistein and increased plasma area under the curve of its conjugates in Bcrp1(−/−) mice is due to altered distribution of genistein conjugates to the systemic circulation.
Introduction
Phase II metabolizing enzymes, especially UDP-glucuronosyltransferases (UGTs) and sulfotransferases (SULTs), play an important role in the metabolism, elimination, and detoxification of phenolics (Wu et al., 2011). UGT and SULT reactions greatly increase hydrophilicity of their substrate to facilitate their ultimate elimination via bile or kidney. Because of their high polarity, phase II conjugates cannot traverse through the cellular membrane by passive diffusion in enterocytes and hepatocytes, the two main first-pass metabolic organs, and require the action of efflux transporters for their exit from the cells (Jia et al., 2004).
Genistein, a plant phenolic and one of the most commonly consumed phytoestrogens, has been extensively studied because of its pharmacological and beneficial effects (Banerjee et al., 2008; Yang et al., 2012), however, with the realization that the full biological potential of genistein (i.e., aglycone) is severely hampered by its extensive metabolism into glucuronide and sulfate conjugates in humans after oral administration (Shelnutt et al., 2000; Coldham et al., 2002; Gu et al., 2006; Hosoda et al., 2010). On the other hand, although generally less active than the aglycone, genistein glucuronides retain the ability to prevent lipid oxidation and possess modest activity to enhance human natural killer cells at nutritionally relevant concentrations (Zhang et al., 1999b; Kgomotso et al., 2008). In addition, genistein conjugates including sulfates can serve as the reservoir of parent compound in vivo by being substrates for futile (both enteric and enterohepatic) recycling and for hydrolysis in tissues (Sfakianos et al., 1997; Chen et al., 2003).
Breast cancer resistance protein (BCRP/Bcrp/ABCG2), a member of the ATP-binding cassette transporter family, is one of the most important efflux transporters for drug disposition and plays an important role in the transport of phase II conjugates (Adachi et al., 2005). BCRP is expressed not only in multidrug resistance tumor cells but also in normal human tissues, including intestine, liver, kidney, brain endothelium, and placenta. BCRP is also a highly polymorphic transporter with different expression among populations, which might result in interindividual variations in the pharmacokinetics and hypersensitivity of normal cells to anticancer drugs (Imai et al., 2004; Ni et al., 2010).
Several studies showed that Bcrp1 plays an important role in the disposition of genistein, and knockout of Bcrp1 gene substantially improved oral bioavailability of genistein and its metabolites in mice (Enokizono et al., 2007; Álvarez et al., 2011). The mechanisms responsible for the improved bioavailability of genistein aglycone in Bcrp1 knockout mice were attributed to increased intestinal absorption because it is a weak substrate of BCRP (Enokizono et al., 2007). Alvarez et al. (2011) further proposed that improved bioavailability of genistein glucuronides and sulfates could be explained by an increased exposure level of genistein aglycone to UGTs and SULTs in Bcrp1 knockout mice. However, an ∼10-fold increase in exposure levels of genistein conjugates did not appear to correlate with a slight increase in aglycone (<2-fold compared with that in WT mice) in Bcrp1 knockout mice. More importantly, numerous previous studies showed that genistein is a highly permeable compound and has good oral absorption (>50%) in vivo (Coldham and Sauer, 2000; Coldham et al., 2002; Chen et al., 2005a; Zhou et al., 2008). Impaired BCRP-mediated efflux of genistein aglycone cannot explain the ∼6-fold increase of oral bioavailability of total genistein in Bcrp1 knockout mice, because the efflux ratio of genistein in a pair of BCRP/Bcrp-overexpressed cell lines were less than 2.5.
This is not the only case in which conjugates showed manyfold increases in efflux transport knockout mice, whereas the aglycones were comparable with wild-type mice (Zelcer et al., 2003; Zamek-Gliszczynski et al., 2011). It is clear that increased bioavailability of conjugates cannot be attributed only to changes in aglycone absorption and alternative mechanisms should be considered instead.
To reevaluate the role of Bcrp1 on disposition of orally administered genistein and reveal the mechanism for the substantially increased exposure level of genistein and its conjugates in Bcrp1(−/−) mice, we performed pharmacokinetic and biliary excretion studies in intestinal perfusion mode in Bcrp1(−/−) and WT mice. Human Caco-2 cells were used to delineate the polarized distribution mechanism of genistein glucuronides and sulfates in intestinal epithelial cells. Hydrolysis reactions in blood were performed to test the hypothesis that increased genistein aglycone might be derived from hydrolysis of conjugates in vivo. Finally, the activities of UTGs and SULTs were measured in Bcrp1(−/−) mice. Because intestine and liver are the major metabolism organs, the study of polarized distribution of genistein and its conjugates was mainly focused on these two organs, in addition to measurements of exposures to genistein and its metabolites in the systemic circulation.
Materials and Methods
Chemicals and Reagents.
Genistein and daidzein were purchased from LC Laboratories (Woburn, MA). Four genistein conjugates (Supplemental Fig. S1) were biosynthesized using mouse intestine and isolated from the mouse intestinal perfusate, as described in our previous publication (Yang et al., 2010b). Hanks' balanced salt solution (powder form) and methotrexate were purchased from Sigma-Aldrich (St. Louis, MO). Cloned Caco-2 cells (TC7) cells were a kind gift from Dr. Monique Rousset of Institut National de la Santé et de la Recherche Médicale U178 (Villejuit, France). 3-(6-Isobutyl-9-methoxy-1,4-dioxo-1,2,3,4,6,7,12,12α-octahydropyrazino[1′,2′:1,6]pyrido[3,4-b]indol-3-yl)-propionic acid tert-butyl ester (Ko143) were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA). The BCA protein assay kit was purchased from Thermo Fisher Scientific (Waltham, MA). Oral suspension vehicle was obtained from Professional Compounding Centers of America (Houston, TX). Nonsoy rat chow (AIN 76A) was purchased from Harlan Teklad (Madison, WI). All other materials (typically analytical grade or better) were used as received.
Animals.
Male wild-type FVB mice (6–10 weeks) were from Harlan (Indianapolis, IN), and male BCRP knockout mice (6–10 weeks) were purchased from Taconic Farm, Inc. (Hudson, NY). Wistar rats were bred in-house. Mice were kept in an environmentally controlled room (temperature 25 ± 2°C, humidity 50 ± 5%, and 12-h dark/light cycle) for at least 1 week before the experiments.
Cell Culture.
Caco-2 cells have been routinely cultured in our laboratory for more than 2 decades (Hu and Borchardt, 1990). The culture conditions for growing Caco-2 cells were described previously (Yang et al., 2010a). Porous polycarbonate cell culture inserts (3 μm) from Corning Life Sciences (Lowell, MA) were used to seed the cells at a seeding density of 60,000 cells/cm2. Caco-2 TC7 cells were fed every other day, and cell monolayers were ready for experiments from 19 to 22 days after seeding. Transendothelial electrical resistance values of the cell monolayers were measured before the experiment, and the monolayer was discarded if the net transendothelial electrical resistance values were less than 460 Ω/cm2 compared with those for the blank inserts.
Pharmacokinetic Studies of Genistein.
The animal protocols used in this study were approved by the University of Houston's Institutional Animal Care and Uses Committee. Mice were fed with nonsoy chow (AIN 76A) for at least 1 week before the pharmacokinetic study. Mice were fasted for 12 h with free access to water before the pharmacokinetic study and were fed again with nonsoy chow at 4 h after the commencement of a pharmacokinetic study. Bcrp1(−/−) and WT FVB mice were used in the pharmacokinetic study after oral gavages of genistein at 2 and 20 mg/kg. Intraperitoneal injection of genistein at 20 mg/kg was also performed using both Bcrp1(−/−) and WT mice. Genistein solution (2 mg/ml) was prepared in a solution of 100 mM sodium bicarbonate-polyethylene glycol 300 (7:3) at pH 10 for intraperitoneal injection. Genistein (4 mg/ml) was dispersed in oral suspending vehicle (PCCA, Houston, TX) for oral gavage. Each pharmacokinetic study was performed on five mice, and blood samples (20–25 μl) were collected by snipping the tail at 10 different time points after mice were anesthetized with isoflurane gas so each mouse had a complete pharmacokinetic profile. The blood samples collected in heparinized tubes were stored at −20°C until analysis.
Pharmacokinetic Analysis.
Pharmacokinetic parameters of genistein and its four metabolites including maximum plasma concentration (Cmax), elimination rate constant (ke), half-life, and area under the plasma concentration-time curve from time 0 to t (AUC0–t) were analyzed using the noncompartmental model in WinNonlin 3.3 (Pharsight, Mountain View, CA). To obtain accurate and reasonable pharmacokinetic parameters for comparison, the following criteria were adopted for noncompartmental fitting: 1) values of ke and half-life were not reported when the fitting goodness (R2) in the terminal phase was less than 0.8; 2) values of ke and half-life were not reported when extrapolated AUC was more than 30% of AUCinf; 3) half-life cannot exceed 1/2 of the time of collection of the last measurable blood sample, which was 12 h in this study; and 4) elimination rate constant should be fitted on the basis of the terminal phase not on the distribution phase. Because good fitting of ke and half-life were not always achievable because of the above restrictions, AUCinf could not always be determined. Therefore, AUC0–t was presented for comparison between WT and Bcrp1(−/−) mice.
In Situ Intestinal Perfusion.
Intestinal absorption of genistein was determined in WT and Bcrp1(−/−) mice (n = 4). The animal protocols used in this study were approved by the University of Houston's Institutional Animal Care and Uses Committee. The intestinal surgical procedures were similar to those described in our previous publications (Yang et al., 2011). In brief, small intestine and colon (8–12 cm each) were simultaneously perfused with 10 μM genistein in HBSS (pH 7.4) at a flow rate of 0.191 ml/min. To keep the temperature of the perfusate constant, the inlet cannula was insulated and kept warm by a 37°C circulating water bath. After a 30-min washout period with perfusate, perfusate samples were collected every 30 min (60, 90, 120, and 150 min). At the end of the perfusion experiment, the length of each segment of intestine was measured, and each tube containing sample was weighted after perfusion. Loss of water during perfusion was monitored by measuring the net weight of the perfusate sample, and the absorption data were discarded if the water flux is more than 10% per 10 cm of intestine. The 500-μl perfusate samples were added with 125 μl of internal standard solution (3 μM daidzein in acetonitrile), and the mixture was vortexed and sonicated for 30 s. After centrifugation at 15,000g for 15 min, 200 μl of supernatant was injected and analyzed by UPLC-MS/MS. The percentages of absorption were calculated using eq. 1:
In eq. 1, Cout and Cin are outlet and inlet perfusion concentrations of genistein after correction for water fluxes, respectively. To accurately compare intestinal absorption, percentage of absorption was normalized to 10 cm of intestine for all animals.
After 2.5 h of intestinal perfusion, the gallbladder was dissected intact carefully. Bile samples (5 μl) were taken and diluted 20 times with HBSS to make a mixture of 100 μl. Then 200 μl of internal standard solution (3 μM daidzein in acetonitrile) was added, and the final mixture (300 μl) was vortexed and sonicated for 2 min. After centrifugation at 15,000g for 15 min, 200 μl of supernatant was collected and air-dried. It was then reconstituted in 100 μl of 15% acetonitrile, and the concentrations of genistein and its metabolites in bile were determined by UPLC-MS/MS.
Transcellular Transport Study.
Transcellular transport of genistein was investigated in Caco-2 cells. The advantage of using the Caco-2 cell line instead of other BCRP-overexpressed cell lines is that this line contains both UGTs/SULTs and many other efflux transporters. The protocol for performing transport experiments in triplicate was the same as that described previously (Yang et al., 2010a). In brief, 2.5 ml of genistein in HBSS was loaded onto the apical side of the cell monolayer and 2.5 ml of blank HBSS was loaded onto the basolateral side. Five sequential samples (0.5 ml) were taken at different times (0, 1, 2, 3, and 4 h) from both sides. The same volume of donor genistein solution and receiver medium (fresh HBSS) was added immediately to replace the volume lost because of sampling. The pH values of HBSS in both the apical and basolateral side were 7.4. Two concentrations of genistein (2 and 10 μM) were studied, and 5 μM Ko143 was added to the apical side of the cell monolayers for BCRP inhibition studies. Methotrexate (2 μM), a well established BCRP substrate, was used as the positive control in Caco-2 cells.
Genistein and its four metabolites were determined in both the apical and basolateral side by UPLC-MS/MS. Rates of transport (Bt) were obtained using rates of change in concentration of transported genistein/its metabolites as a function of time (dC/dt) and volume of the chamber (V) (eq. 2). Both apical and basolateral excretion rates were calculated according to the following equation:
Intracellular concentrations of genistein and its metabolites were determined at the end of the 4-h transport study. The protocol for determining intracellular amounts of genistein and its metabolite in cells was described in our previous publication (Yang et al., 2010b).
The percentage of basolateral excretion, which represents basolateral distribution of genistein conjugates in Caco-2 cells, was calculated by eq. 3:
 |
The fraction of dose metabolized (Fmet) was calculated as in eq. 4 to describe the extent of genistein metabolism in a Caco-2 monolayer (Pang et al., 2009), where Mettotal is the total metabolites and Gentotal is the total genistein in receiver, donor, and cell compartments combined at 4 h, respectively:
Mass balance was obtained by comparing total amounts of genistein and its four metabolites at 4 h with amounts of genistein added in donor side at 0 h in the Caco-2 cell culture model.
Deconjugation of Genistein Glucuronides and Sulfates in Blood.
Genistein glucuronides and sulfates were biosynthesized through intestinal perfusion in rat and mice, which was described in our previous publication (Yang et al., 2010b). The mixture of 3 μM genistein glucuronide (combination of G-7-G and G-4′-G) and 3.8 μM genistein was incubated in fresh blood taken from Wistar rats in a 37°C shaking water bath for up to 4 h. Blood samples (50 μl) were taken at 0, 60, 120, and 240 min, and the reaction was stopped by addition of 50 μl of acetonitrile containing 3 μM daidzein (internal standard). Likewise, the hydrolysis reactions of genistein sulfate in fresh blood [0.8 μM genistein sulfates (combination of G-7-S and G-4′-S) and 2.2 μM aglycone] were conducted using the same conditions. The concentration used for hydrolysis reaction depends on the final concentration purified from biosynthesis. We also performed the same experiments by using 50 mM potassium phosphate buffer (pH 7.4) instead of blood for incubation at the same condition as the negative control.
Glucuronidation and Sulfation of Genistein in Hepatic and Intestinal S9 Fraction.
Hepatic and intestinal (small intestine and colon) S9 fractions from WT and Bcrp1(−/−) mice were prepared following the protocol described in our previous publication (Zhu et al., 2010). Protein concentrations of S9 fractions were determined using a protein assay kit, with bovine serum albumin as the standard.
Glucuronidation and sulfation of genistein by S9 fractions was measured using procedures described previously (Zhu et al., 2010). In brief, for the glucuronidation reaction, 10 μM genistein in 50 mM potassium phosphate buffer (pH 7.4) was added into the reaction mixture with S9 fractions (1 mg/ml), magnesium chloride (0.88 mM), saccharolactone (4.4 mM), and alamethicin (0.022 mg/ml). A solution of 3.5 mM UDPGA was added last to make a total volume of 200 μl, and the mixture was incubated in a 37°C water bath for 60 min. For the sulfation reaction, incubation conditions were the same except that 0.1 mM 3′-phosphoadenosine-5′-phosphosufate solution was added instead of the UDPGA coenzyme system.
Both glucuronidation and sulfation reactions were stopped by addition of 50 μl of stopping solution (94% acetonitrile-6% glacial acetic acid containing 100 μM testosterone as the internal standard). After centrifugation, the supernatants of samples were analyzed by UPLC. The assay was validated in our previous publication (Zhu et al., 2010). The same mixture with water in place of 3′-phosphoadenosine-5′-phosphosufate or UDPGA was used as the negative control.
Quantitation of Genistein and Its Four Metabolites.
A UPLC-MS/MS assay for analysis of genistein and its four conjugated metabolites in mouse blood was developed and validated previously (Yang et al., 2010b). In brief, an API 3200 QTRAP triple quadrupole mass spectrometer Applied Biosystems/MDS Sciex (Foster City, CA) coupled with an ultraperformance liquid chromatograph (Waters, Milford, MA) was used to analyze the blood samples. Because of the limited sample volume, whole blood was used directly. The concentrations of genistein, G-7-G, G-4′-G, G-4′-S, G-7-S and daidzein (internal standard) in blood samples were determined by multiple reaction monitoring method. The chromatogram of genistein and its four metabolites is shown in Supplemental Fig. S2. Methotrexate, the positive control compound used in the Caco-2 cell transport study, was measured by UPLC-MS/MS with the multiple reaction monitoring ion pair 455.0/308.1 (m/z).
Statistical Analysis.
The data in this article are presented as means ± S.D., if not specified otherwise. Significance differences were assessed by using Student's t test. p < 0.05 was considered statistically significant.
Results
Impact of Bcrp1 on Pharmacokinetics of Genistein Aglycone and Conjugates after Oral Administration.
Impact of Bcrp1 on oral pharmacokinetics of aglycone.
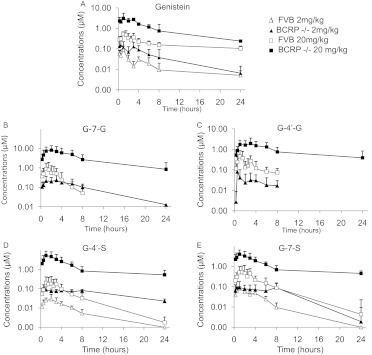
Genistein aglycone showed moderately higher blood concentrations (as expressed in AUC) in Bcrp1(−/−) mice than in WT mice at both 2 and 20 mg/kg (Fig. 1A). In Bcrp1(−/−) mice, the AUC0–t of aglycone was increased 2.0- and 2.2-fold (p < 0.05) at 2 and 20 mg/kg, respectively, and the Cmax of aglycone was increased 2.3-fold (p = 0.1) and 5.1-fold (p < 0.01) at 2 and 20 mg/kg, respectively (Table 1). These results were consistent with previous reports that AUC0–4 h of aglycone was ∼2-fold higher in Bcrp1(−/−) mice than in WT mice after oral administration of genistein (Enokizono et al., 2007; Alvarez et al., 2011). However, it is noted that these two previous publications had very different exposure levels of genistein aglycone in plasma of Bcrp1(−/−) mice, and our results (Fig. 1) were close to what Enokizono et al. (2007) observed (Cmax of ∼1.1 μM at 8 mg/kg oral genistein). The more recent study showed a Cmax of a little less than 0.2 μM at a dose of 50 mg/kg oral genistein (Alvarez et al., 2011). The ke and t1/2 were not available (N.A.) because of poor fitting in the terminal phase of the pharmacokinetic profile, which is probably the result of extensive enteric and enterohepatic recycling (Chen et al., 2005b; Zhou et al., 2008). It should be mentioned that half-life and elimination rate of genistein and its metabolites in wild-type FVB mice were somewhat different from those in our previous article (Yang et al., 2010b), because of the more stringent criteria adopted in the present noncompartmental analysis as detailed under Materials and Methods.
Fig. 1.
The blood concentrations of genistein (A), G-7-G (B), G-4′-G (C), G-4′-S (D), and G-7-S (E) after oral administration of genistein at both 2 and 20 mg/kg in wild-type and BCRP knockout mice. The blood concentrations of G-7-G and G-4′-G were below the detection limit in wild-type mice for the 2 mg/kg group and 24-h samples of BCRP(−/−) mice. Data are presented as means ± S.D; n = 5.
TABLE 1.
Pharmacokinetic parameters of genistein, G-7-G, G-4′-S, and G-7-S in WT and BCRP(−/−) mice after oral administration
Pharmacokinetic parameters in BCRP(−/−) mice were compared to WT mice at the same dose. Data are presented as means ± S.D.; n = 5.
| Strain | Dose | Cmax | ke | t1/2 | AUC0–t | |
|---|---|---|---|---|---|---|
| mg/kg | μM | h−1 | h | h · μM | ||
| Genistein | FVB | 2 | 0.10 ± 0.05 | N.A. | N.A. | 0.36 ± 0.17 |
| BCRP(−/−) | 2 | 0.23 ± 0.17 | 0.27 ± 0.20 | 3.8 ± 2.7 | 0.71 ± 0.24* | |
| FVB | 20 | 0.65 ± 0.22 | N.A. | N.A. | 6.05 ± 0.54 | |
| BCRP(−/−) | 20 | 3.35 ± 0.65** | N.A. | N.A. | 16.41 ± 4.23* | |
| G-7-G | WT | 2 | N.A. | N.A. | 0.15a | |
| BCRP(−/−) | 2 | 0.30 ± 0.16 | N.A. | N.A. | 1.87 ± 0.68* | |
| WT | 20 | 1.07 ± 1.03 | N.A. | N.A. | 3.49 ± 2.58 | |
| BCRP(−/−) | 20 | 9.63 ± 5.65* | 0.16 ± 0.10 | 5.5 ± 3.3 | 56.71 ± 43.87* | |
| G-4′-G | WT | 2 | N.A. | N.A. | 0.12a | |
| BCRP(−/−) | 2 | 0.11 ± 0.12 | 0.28 ± 0.23 | 3.7 ± 3.0 | 0.26 ± 0.17 | |
| WT | 20 | 0.52 ± 0.37 | 0.25 ± 0.25 | 6.5 ± 6.5 | 1.56 ± 0.79 | |
| BCRP(−/−) | 20 | 2.14 ± 1.44* | 0.15 ± 0.10 | 6.2 ± 3.3 | 15.19 ± 11.16* | |
| G-4′-S | WT | 2 | 0.04 ± 0.03 | 0.17 ± 0.01 | 4.0 ± 0.3 | 0.13 ± 0.04 |
| BCRP(−/−) | 2 | 0.14 ± 0.11 | N.A. | N.A. | 1.37 ± 1.17* | |
| WT | 20 | 0.25 ± 0.22 | 0.32 ± 0.19 | 2.7 ± 1.3 | 1.03 ± 0.64 | |
| BCRP(−/−) | 20 | 6.18 ± 4.14* | 0.30 ± 0.10 | 2.4 ± 0.7 | 26.78 ± 13.79* | |
| G-7-S | WT | 2 | 0.08 ± 0.02 | 0.27 ± 0.07 | 2.7 ± 0.6 | 0.32 ± 0.05 |
| BCRP(−/−) | 2 | 0.17 ± 0.05 | 0.21 ± 0.03 | 3.4 ± 0.5 | 1.13 ± 0.81* | |
| WT | 20 | 0.65 ± 0.56 | 0.21 ± 0.18 | 5.6 ± 4.5 | 2.49 ± 1.51 | |
| BCRP(−/−) | 20 | 4.39 ± 2.55* | 0.25 ± 0.09 | 2.9 ± 1.1 | 20.92 ± 8.86** |
N.A., not available because of poor fitting value (R2 < 0.8) in the terminal phase.
p < 0.05.
p < 0.01.
Estimated AUC0–t.
Impact of Bcrp1 on pharmacokinetics of conjugates after oral administration of genistein.
The blood concentrations of G-7-G, G-4′-G, G-4′-S, and G-7-S were significantly higher in Bcrp1(−/−) mice than in WT mice at both 2 and 20 mg/kg (Fig. 1, B–E). The Cmax values of G-7-G, G-4′-G, G-4′-S, and G-7-S were increased N.A., N.A., 3.6-, and 2.0-fold and 9.0-, 4.1-, 24.7-, and 6.8-fold in Bcrp1(−/−) mice compared with values in WT mice at 2 and 20 mg/kg, respectively. Consistent with the increase in Cmax values, AUC0–t of G-7-G, G-4′-G, G-4′-S, and G-7-S were increased N.A., N.A., 10.5-, and 3.5-fold and 16.2-, 9.7-, 26.0-, and 8.4-fold in Bcrp1(−/−) mice at 2 and 20 mg/kg, respectively (Table 1). When available, the ke and t1/2 values of G-7-G, G-4′-G, G-4′-S, and G-7-S were not statistically significantly altered in Bcrp1(−/−) mice compared with those in WT mice at 2 and 20 mg/kg, respectively.
Impact of Bcrp1 on total genistein AUC in blood.
We added the AUC0–t (t = 24 h) of aglycone and conjugates together (or AUCtotal) and used it as an indicator of overall exposure. The results indicated that at 2 mg/kg, AUCtotal was increased from ∼1.1 to ∼5.4 h · μM (or ∼5.0-fold). This change in AUCtotal was much greater than the change in AUC of genistein aglycone (less than 2-fold). At 20 mg/kg, the AUCtotal was increased from ∼14.6 to ∼136 h · μM (or ∼9.3-fold). Again, the change in AUCtotal was much greater than the change in AUC of genistein aglycone (less than 3-fold).
Previous reports showed that AUC0–t (t = 3 h) of total genistein (aglycone and conjugates) was increased from ∼0.9 to 5 h · μM (or 5.5-fold) at 50 mg/kg (Alvarez et al., 2011). Although the absolute exposure levels of total genistein were significantly lower than our current finding, the effect of Bcrp1 deficiency on percent changes in AUCtotal was consistent. It was noted that genistein showed nonlinear pharmacokinetics in terms of AUC between 2 and 20 mg/kg, which is consistent with results from a previous report (Zhou et al., 2008). The exact reason for the observed nonlinear pharmacokinetic phenomenon is not known, but a possible reason is the saturation of genistein glucuronidation at the higher dose.
Impact of Bcrp1 on Pharmacokinetics of Genistein Aglycone and Conjugates after Intraperitoneal Injection.
To investigate the impact of hepatic Bcrp1 on pharmacokinetics of genistein aglycone and conjugates, genistein was administered intraperitoneally in WT and Bcrp1(−/−) mice, which minimizes the role of intestinal Bcrp1 in genistein disposition.
Impact of Bcrp1 on intraperitoneal pharmacokinetics of aglycone.
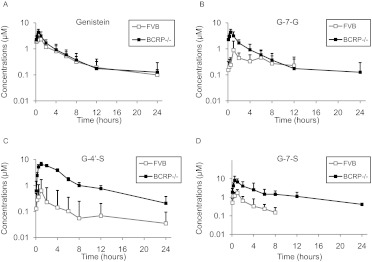
Genistein aglycone showed higher Cmax in Bcrp1(−/−) mice compared with that in WT mice after intraperitoneal administration, but the AUC0–t was not statistically increased in Bcrp1(−/−) mice compared with that in WT mice (Fig. 2A). The ke and half-life values of genistein were not significantly different between Bcrp1(−/−) and WT mice (Table 2).
Fig. 2.
The blood concentrations of genistein (A), G-7-G (B), G-4′-S (C), and G-7-S (D) after intraperitoneal administration of genistein at 20 mg/kg in wild-type FVB and Bcrp1(−/−) mice (n = 5). The blood concentrations of G-7-G and G-7-S were below the detection limit in FVB mice at 24 h. Data are presented as means ± S.D.
TABLE 2.
Pharmacokinetic parameters of genistein, G-7-G, G-4′-S, and G-7-S in WT and BCRP(−/−) mice after an intraperitoneal injection of 20 mg/kg
Pharmacokinetic parameters in Bcrp1(−/−) mice were compared to WT mice at the same dose. Data are presented as mean ± S.D.; n = 5.
| Strain | Cmax | ke | t1/2 | AUC0–t | |
|---|---|---|---|---|---|
| μM | h−1 | h | h · μM | ||
| Genistein | WT | 2.68 ± 0.86 | 0.23 ± 0.09 | 4.8 ± 4.2 | 10.0 ± 2.30 |
| BCRP(−/−) | 4.42 ± 1.04* | 0.52 ± 0.33 | 2.8 ± 2.8 | 11.54 ± 5.57 | |
| G-7-G | WT | 0.93 ± 0.38 | N.A. | N.A. | 3.26 ± 2.18 |
| BCRP(−/−) | 4.28 ± 2.08* | 0.55 ± 0.38 | 3.0 ± 3.0 | 10.84 ± 6.86* | |
| G-4′-S | WT | 0.64 ± 0.29 | 0.30 ± 0.30 | 4.2 ± 3.1 | 1.91 ± 1.16 |
| BCRP(−/−) | 8.18 ± 5.07* | 0.35 ± 0.22 | 3.1 ± 2.7 | 33.90 ± 31.78* | |
| G-7-S | WT | 1.73 ± 0.85 | 0.82 ± 0.53 | 1.1 ± 0.6 | 3.96 ± 3.41 |
| BCRP(−/−) | 11.31 ± 6.63* | 0.13 ± 0.03* | 5.4 ± 1.2* | 28.74 ± 24.13* |
N.A., not available because of poor fitting value (R2 < 0.8) in the terminal phase.
p < 0.05.
** p < 0.01.
Impact of Bcrp1 on intraperitoneal pharmacokinetics of conjugates.
Similar to results after oral administration, the exposure levels of genistein glucuronides and genistein sulfates were substantially higher in Bcrp1(−/−) mice than in WT mice after intraperitoneal administration at 20 mg/kg (Fig. 2, B–D). The Cmax values of G-7-G, G-4′-S, and G-7-S were increased 4.6-, 12.8-, and 6.5-fold and their AUC0–t values were increased 3.3-, 17.8-, and 7.3-fold in Bcrp1(−/−) mice, respectively (Table 2). The concentrations of G-4′-G were below the lower limit of quantification for most of the samples after intraperitoneal administration.
The ke of G-7-S was significantly decreased by 85% in BCRP knockout mice at 20 mg/kg (p < 0.05) (Table 2), but the ke of G-4′-S was not significantly changed in Bcrp1(−/−) mice compared with that in WT mice. Likewise, the t1/2 of G-7-S significantly increased 3.9-fold in Bcrp1 knockout mice, whereas that of G-4′-S did not increase significantly.
Impact of Bcrp1 on total genistein AUC in blood.
We added the AUC0–t values for aglycone and conjugates together (or AUCtotal) and used this as an indicator of overall exposure after intraperitoneal administration. At 20 mg/kg, the AUCtotal was increased from ∼19 to ∼85 h · μM (or ∼4.5-fold). This change in AUCtotal was much greater than the change in AUC of genistein aglycone (10% higher).
Role of Bcrp1 in Intestinal Absorption of Genistein.
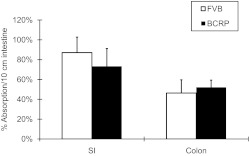
The intestinal absorption of genistein was investigated to determine whether Bcrp1 limits intestinal absorption of genistein aglycone. Absorption of genistein was rapid with minimal effects on water flux, and percentage of absorption of 10 μM genistein was almost complete in the small intestine in WT mice (87 ± 16%) and was not increased in Bcrp1(−/−) mice (74 ± 17%) compared with that in WT mice (Fig. 3). Percentage of absorption of genistein in colon segment was consistently very similar between WT mice (46 ± 14%) and Bcrp1(−/−) knockout mice (52 ± 8%). It was comparable with our previous report that intestinal absorption of genistein in rat small intestine is 77% or greater (Chen et al., 2005b). Genistein showed higher absorption in the small intestine than in the colon in mice, but this difference were not statistically significant. These data demonstrated that intestinal absorption of genistein is expected to be complete, and Bcrp1 did not significantly affect genistein intestinal absorption, which is very different from the substantial decreases in intestinal excretion of genistein conjugates that we reported earlier (Zhu et al., 2010).
Fig. 3.
The intestinal absorption of genistein aglycone in wild-type FVB and Bcrp1(−/−) mice (n = 4). Data are presented as means ± S.D. The perfusion containing 10 μM genistein was performed at a flow rate of 0.191 ml/min. SI, small intestine.
Role of Bcrp1 in Biliary Excretion of Genistein Aglycone and Conjugates.
Biliary excretion is one of the major elimination pathways for genistein (Prasain et al., 2006), and therefore the role of Bcrp1 in biliary excretion of genistein aglycone and conjugates was also investigated. Our previous results showed that biliary excretion of genistein and its metabolites was in steady state after 2 h of intestinal perfusion in rats (Chen et al., 2005b). Genistein concentrations in bile were not decreased in Bcrp1(−/−) mice (9.1 ± 3.2 μM) compared with those in WT mice (9.9 ± 4.4 μM), which indicated that Bcrp1 was not involved in biliary excretion of aglycone.
Of interest, knockout of Bcrp1 showed different effects on biliary excretion of genistein glucuronides and sulfates. The concentrations of genistein glucuronides (combination of G-7-G and G-4′-G) in bile did not display significant changes in WT mice (153.3 ± 69.3 μM) and Bcrp1(−/−) mice (186.1 ± 110.7 μM). In contrast, concentrations of genistein sulfates (combination of G-4′-S and G-7-S) in bile decreased substantially (93%) from 34.0 ± 14.3 μM in WT mice to 2.3 ± 0.6 μM in Bcrp1(−/−) mice (p < 0.05). The absolute amounts of genistein conjugates recovered in bile were much higher than that of the aglycone, indicating that genistein conjugates were efficiently eliminated via biliary excretion. Significantly decreased genistein sulfates showed that Bcrp1 plays a dominant role for its biliary excretion, whereas other transporters (e.g., MRP2) may be involved in the biliary excretion of genistein glucuronides in the absence of BCRP.
Role of BCRP in the Excretion of Genistein Glucuronides and Sulfates in Caco-2 Cells.
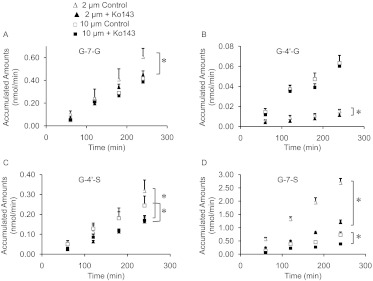
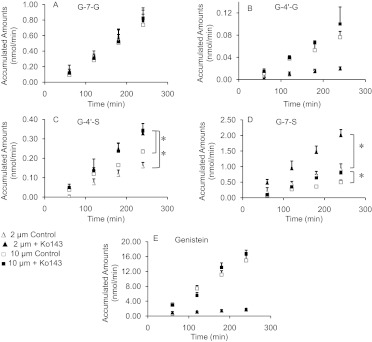
To further reveal the reasons for the higher exposure levels of genistein conjugates in Bcrp1(−/−) mice after oral administration, effects of BCRP on the polarized excretion of genistein conjugates in enterocytes were investigated. Before the transport study of genistein conjugates, methotrexate, the substrate of BCRP, showed significantly higher permeability and intracellular concentrations in the presence of 5 μM Ko143 in a positive control study (data not shown). Two concentrations of genistein (2 and 10 μM) were used in the Caco-2 cell transport study of how BCRP inhibition by Ko143 affected polarized excretion of conjugates.
In the presence of Ko143 treatment, the apical excretion of G-7-G and G-4′-G was significantly decreased (25 and 20%, p < 0.05) at 2 μM genistein (Fig. 4, A and B) but not at 10 μM (Supplement Table S3). Compared with results for glucuronides, we found a more potent inhibitory effect on sulfate excretion by 5 μM Ko143, which significantly inhibited (p < 0.05) the apical excretion of G-4′-S and G-7-S by 45 and 54% at 2 μM genistein and by 28 and 46% at 10 μM genistein (Fig. 4, C and D).
Fig. 4.
The apical excretion of G-7-G (A), G-4′-G (B), G-4′-S (C), and G-7-S (D) in Caco-2 cells in the presence or absence of KO143. Two concentrations of genistein (2 and 10 μM) and 5 μM KO143 were used as inhibitor. Data are presented as means ± S.D; n = 3. *, p < 0.05.
In contrast to the decreased apical excretion, basolateral excretion of genistein glucuronides and sulfates increased after Ko143 treatment. Whereas basolateral excretion of G-7-G and G-4′-G was only slightly increased (10–14%, p > 0.05) at both 2 and 10 μM genistein (Fig. 5, A and B), basolateral excretion of G-4′-S and G-7-S was significantly increased (p < 0.05) 90 and 110% at 2 μM genistein and 50 and 80% at 10 μM genistein (Fig. 5, C and D).
Fig. 5.
The basolateral excretion of G-7-G (A), G-4′-G (B), G-4′-S (C), and G-7-S (D) and the transport of genistein (E) from the apical to basolateral side in Caco-2 cells in the presence or absence of KO143. Two concentrations of genistein (2 and 10 μM) were used, and 5 μM KO143 were used as inhibitor. Data are presented as means ± S.D; n = 3. *, p < 0.05.
As a consequence of the inhibitory effects of Ko143, more genistein conjugates were basolaterally excreted after Ko143 treatment in Caco-2 cells as reflected by percentage of basolateral excretion. The percentage of basolateral excretion of G-7-G was significantly increased from 53% (control) to 63% (Ko143 treatment), and G-4′-G showed a similar change (p = 0.16) at a lower dose of genistein (2 μM). The excretion pathway of genistein sulfates was significantly changed from preferring apical excretion to basolateral excretion at 2 and 10 μM, but the extent of the preference was less at the higher concentration (Supplemental Fig. S1).
Unlike that of genistein conjugates, transport of genistein aglycone from the apical to the basolateral side did not significantly increase at either 2 or 10 μM after 5 μM Ko143 treatment (Fig. 5E), which is consistent with intestinal absorption in Bcrp1(−/−) mice. Likewise, intracellular amounts of genistein aglycone in Caco-2 cells in the presence of Ko143 did not change significantly compared with the control, indicating that inhibition of BCRP did not increase steady-state accumulation of genistein aglycone (Supplemental Fig. S4).
Fmet, a measurement of enzyme activities in intact cells, did not change at 2 or 10 μM after Ko143 treatment in terms of total and individual metabolism, indicating that inhibition of BCRP did not change the extent or the pathway of genistein metabolism in the Caco-2 cells (Supplemental Fig. S5). The recoveries of genistein and its conjugates in Caco-2 cells after 4 h were 92.3 to 108.8% in the control group and 87.8 to 104.2% in the Ko143 treatment group, indicating that these four conjugates are the major metabolites of genistein in Caco-2 cells.
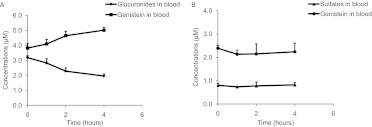
Deconjugation of Genistein Glucuronidation and Sulfation in Blood.
The concentration of genistein glucuronides decreased and that of aglycone increased with time in fresh blood after incubation, indicating that genistein glucuronides could be hydrolyzed in vivo into the aglycone (Fig. 6A). Unlike that of genistein glucuronides, the concentrations of both sulfates and aglycone were unaltered with time in fresh blood, indicating that genistein sulfates were not hydrolyzed in blood (Fig. 6B). Genistein glucuronides and sulfates were stable after incubation in buffer at the same conditions (Supplemental Fig. S6).
Fig. 6.
Deconjugation of genistein glucuronides and sulfates in fresh blood at 37°C in a shaking water bath for up to 4 h (n = 3). Genistein glucuronide (combination of G-7-G and G-4′-G; 3 μM) and genistein (3.8 μM) were incubated in fresh blood taken from Bcrp1(−/−) rats for genistein glucuronides hydrolysis. Genistein sulfates [combination of G-7-S and G-4′-S (0.8 μM) and aglycone (2.2 μM)] were incubated in the same condition as for genistein sulfate hydrolysis. SI, small intestine.
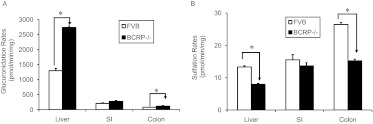
Glucuronidation and Sulfation Activities in Bcrp1 Knockout Mice.
Besides the role of BCRP, we also tested whether enzyme activities were altered in Bcrp1(−/−) mice. We found that rates of genistein glucuronidation in the hepatic S9 fraction (combination of G-7-G and G-4′-G) from Bcrp1(−/−) mice (2737 ± 46 pmol · min−1 · mg−1) were significantly increased (by 110%) compared with those from WT mice (1299 ± 71 pmol · min−1 · mg−1). Likewise, the glucuronidation rate was increased by 50% in colon (from 78 ± 5 pmol · min−1 · mg−1in WT mice to 117 ± 19 pmol · min−1 · mg−1 in Bcrp1(−/−) knockout mice). However, there were no significant changes in glucuronidation activities in the small intestinal S9 fraction between these two strains of mice (Fig. 7A).
Fig. 7.
The genistein glucuronidation rate (A) and sulfation rate (B) in liver, small intestine and colon S9 fractions prepared from FVB and BCRP(−/−) mice. Genistein (10 μM) was used. Data are presented as means ± S.D; n = 4. *, p < 0.05.
Unlike genistein glucuronidation, we found that rates of genistein sulfation (combination of G-4′-S and G-7-S) from Bcrp1(−/−) mice was significantly decreased by 40% (from 13.3 ± 0.4 to 7.9 ± 0.3 pmol · min−1 · mg−1) in liver S9 fractions and by 42% (from 26.5 ± 0.7 to 15.2 ± 0.5 pmol · min−1 · mg−1) in colonic S9 fractions compared with WT FVB mice. No significant change was observed in genistein sulfation activities in small intestinal S9 fraction (Fig. 7B). Although we saw similar exposure levels of genistein glucuronides and sulfates in blood circulation (Fig. 2), the in vitro metabolism rates of glucuronidation and sulfation had large differences, and the actual reasons for this apparent discrepancy remained unknown.
Discussion
Our results showed that knockout of Bcrp1 led to moderately increased bioavailability of aglycone (∼2-fold) and preferential distribution of genistein conjugates to plasma (as reflected by a substantial increase in plasma AUC of conjugates, usually >10-fold). Altered distribution of conjugates was the main mechanism behind the above-observed increases because the loss of Bcrp1 functionality (due to deletion of inhibition) had a substantial impact on the polarized distribution of genistein conjugates across the intestinal membrane and hepatocytes. Our results did not show increased absorption of genistein in the intestine of the knockout mouse model or across the Caco-2 cell monolayers in the presence of a potent BCRP inhibitor, Ko143.
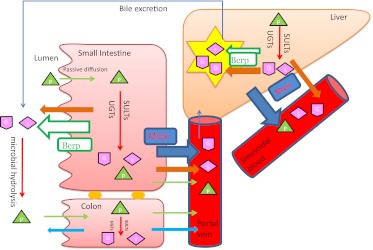
Previous publications have proposed that increased bioavailability of genistein aglycone was due to increased intestinal absorption in Bcrp1(−/−) mice (Enokizono et al., 2007), and elevated exposure level of genistein aglycone may be the major reason for increased AUC of genistein conjugates in Bcrp1(−/−) mice (Alvarez et al., 2011). Here, we clearly demonstrated that the intestinal absorption of genistein was not significantly altered in Bcrp1(−/−) mice nor was the absorption of genistein aglycone increased across the Caco-2 cell monolayers in the presence of a potent BCRP inhibitor, Ko143. We therefore concluded that the increased exposure of genistein glucuronides and sulfates in Bcrp1(−/−) mice was mainly caused by changes in polarized distribution of conjugates in intestine and liver. More specifically, genistein metabolites were being increasingly excreted to the systemic circulation (i.e., enhanced basolateral excretion), instead of being effluxed back to intestine and bile in Bcrp1(−/−) mice. Although increased plasma glucuronide levels were probably also aided by increased glucuronidation rates in the knockout mice, altered distribution is the major reason for the very large percentage increases in plasma AUC of genistein conjugates. We further demonstrated that increased bioavailability of genistein aglycone could be due to reconversion of glucuronides to aglycone in the blood. We propose a schematic diagram to explain the determinant impact of Bcrp1 on the distribution of genistein conjugates in enterocytes, hepatocytes, intestinal lumen, bile, and the systemic circulation (Fig. 8). Please notice the large role that Bcrp1 played in the excretion of conjugates to the apical side of the enterocytes and hepatocytes. Its deletion leads to major changes in distribution of the genistein conjugates (favoring the basolateral side), resulting in large percentage increases in plasma conjugate AUCs.
Fig. 8.
Schematic representation showing the absorption of genistein aglycone and distribution of its conjugates in intestine, bile and blood (or plasma). This is a schematic presentation and as such is an approximate representation of the major enzymes, and efflux transporters involved, and not exhaustive. Green triangle with P, genistein; magenta shape with G, genistein glucuronides; magenta diamond with s, genistein sulfates; arrows, efflux transporters for genistein conjugates transport; large green hollow arrows, proposed role of Bcrp; brown filled arrows, proposed role of other efflux transporters; small green solid arrows: aglycone transport via passive diffusion. Arrow size indicates the importance (or size) of its role in genistein conjugate transport. MRP, represented by large solid blue arrows, represents the MRP efflux transporters localized to the basolateral side. Currently, only one MRP transporter (i.e., MRP3) was shown to be involved in the basolateral efflux of conjugates (Kitamura et al., 2010), but other related MRP efflux transporters may also function. Note: Genistein has the same transport pathway in small intestine and colon. To keep the figure simple, some of the enzyme and transporter icons were omitted in the colon compartment. In the colon compartment, efflux transporters were represented by simple magenta solid arrows without identifying the actual efflux transporters, which are expected to be identical to those in the small intestine (although the expression levels may be different).
Four independent studies at in vivo, in situ, and in vitro levels were conducted to demonstrate the mechanism by which Bcrp1 deletion affected bioavailability of genistein aglycone and conjugates. In the first study, in which intestinal absorption was directly measured using an in situ intestinal perfusion model, we found that intestinal absorption of genistein aglycone did not increase in Bcrp1(−/−) mice compared with that in wild-type mice (Fig. 3). In the second study, in which the pharmacokinetics of genistein after intraperitoneal administration were determined, we found that AUC of genistein aglycone did not significantly increase in Bcrp1(−/−) mice but the exposure of genistein glucuronides and sulfates did (by 4–10-fold) (Fig. 2). The latter indicated that an increase in exposure of conjugates did not have to correlate with a corresponding increase in aglycone level in vivo, as postulated by an early investigation. In the third study of biliary excretion, biliary excretion of genistein aglycone did not change in Bcrp1(−/−) mice, whereas that of genistein sulfates significantly decreased (>90%), indicating that Bcrp1 knockout could alter the polarized excretion of conjugates in the hepatocytes. In the fourth study of genistein transport in the Caco-2 cell monolayers, inhibition of BCRP did not increase permeability or intracellular concentration of genistein aglycone but altered polarized excretion of genistein conjugates (to favoring basolateral efflux) (Figs. 4 and 5). The effects of BCRP inhibition on conjugate efflux were modest in Caco-2 cells because of the presence of other efflux transporters such as MRP2/3 and relatively low expression of BCRP in Caco-2 cells (Xia et al., 2005; Maubon et al., 2007). Taken together, these results suggest that in Caco-2 cells, inhibition of BCRP did not affect rate of transport or metabolism, but affected the polarized excretion of conjugates.
It is not surprising that intestinal absorption of genistein did not increase in Bcrp1(−/−) mice compared with that in WT mice (Fig. 3), because the compound is highly permeable and well absorbed. Genistein showed high permeability (>3 × 10−5 cm/s) in the human intestinal Caco-2 cell model (Liu and Hu, 2002; Chen et al., 2003), and the bidirectional transport study indicated that passive diffusion is the major transport mechanism of genistein in human Caco-2 cells (Liu and Hu, 2002). It was shown earlier that genistein was a weak substrate of BCRP (efflux ratio ∼2) in Bcrp-overexpressed Madin-Darby canine kidney II cells (Enokizono et al., 2007). However, the efflux transporter may not have any significant in vivo effect on a highly permeable compound with a low efflux ratio (∼2) because the substrate in the basolateral compartment or the mesenteric vein will be quickly removed and cannot be effluxed back efficiently in vivo because of unfavorable mass action (Huang et al., 2008; Zhang et al., 2009).
The reconversion of glucuronides to aglycone in blood as shown in Fig. 7 was probably responsible for the moderate increase in plasma genistein aglycone (i.e., oral bioavailability) seen in our studies. Reconversion of glucuronides to aglycone could better explain why genistein aglycone significantly increased after oral administration (for which glucuronides increased by ∼14-fold) but not after intraperitoneal administration (for which glucuronides increased by only ∼3-fold). Our results provide a new insight as to why increased exposure of aglycone may derive from hydrolysis of glucuronide conjugates, which is very different from the current understanding that increased levels of aglycone come directly from impaired aglycone efflux in Bcrp1(−/−) mice (Alvarez et al., 2011).
Because Bcrp1 controls polarized excretion of conjugates in the enterocytes and hepatocytes, deletion of Bcrp1 is probably the driving force behind the large increase in the distribution of genistein conjugates to mouse plasma (a big increase in their AUC0–t) in Bcrp1(−/−) mice. This is consistent with our previous observation that luminal excretion of genistein glucuronides and sulfates decreased 78 and >90%, respectively, in Bcrp1(−/−) mice compared with that in WT mice (Zhu et al., 2010). It is also consistent with results of Ko143 in Caco-2 cell monolayers, in which increased relative distribution of conjugates to the basolateral side was observed (Fig. 5). Because of unaltered intestinal absorption of genistein and slightly different enzyme activities in intestine, less luminal excretion of genistein conjugates in mouse intestine should lead to higher basolateral excretion of genistein conjugates (Fig. 5) to the systemic circulation as illustrated in Fig. 8. Likewise, Bcrp1 did not mediate the biliary excretion of genistein aglycone but played an important role in the biliary excretion of genistein sulfates. Hence, deletion of Bcrp1 would increase basolateral excretion of sulfates to the blood.
The transporter responsible for increased basolateral efflux in enterocytes and hepatocytes has not yet been defined, but the most likely candidate is MRP3, which may transport more genistein conjugates in the absence of Bcrp1 as a compensatory mechanism to prevent their accumulations inside the cell (Supplemental Fig. S4) (van de Wetering et al., 2009).
We observed a bigger impact of Bcrp1 deletion on plasma AUC of genistein and its conjugates after oral administration than after intraperitoneal administration, suggesting that Bcrp1 plays a more important role in intestinal conjugation than liver conjugation (of genistein). In liver conjugation of genistein, Bcrp1 plays a more important role in sulfonation than in glucuronidation, in that the biliary excretion of genistein glucuronides was not decreased in Bcrp1(−/−) mice but biliary excretion of sulfate was. As a consequence, there was a bigger change in sulfate AUC than in glucuronide AUC after intraperitoneal administration. Of course, the higher glucuronidation rate and lower sulfation rate in liver of Bcrp1(−/−) mice also probably contributed to the changes in plasma conjugate AUC.
Our result is the first report showing that knockout of Bcrp1 may also change the activities of relevant UGTs and SULTs (Fig. 7), which indicates that knockout of Bcrp1 could cause unexpected changes in SULT and UGT activities. The increased genistein glucuronidation rates and decreased sulfation rates in Bcrp1(−/−) mice further signified the predominant role of Bcrp1 in determining the formation and distribution of sulfates in mice. A previous publication has showed that the expression levels of MRP2 and MRP3 were not changed in Bcrp1 knockout mice (Nezasa et al., 2006).
In conclusion, Bcrp1 deficiency moderately increases the oral bioavailability of genistein aglycone. More importantly, Bcrp1 deficiency caused a major change in the distribution of conjugates, especially sulfates, resulting in large increases in plasma AUC of genistein glucuronides and sulfates. This impact on plasma conjugate AUC values occurred mainly in the intestine and liver and was greater after oral administration. Here, we want to advance the hypothesis that deficiency in efflux transporters could lead to altered plasma pharmacokinetics of these conjugates in Bcrp1(−/−) mice, which could increase plasma AUC of conjugates and bioavailability of genistein aglycone due to reconversion of glucuronides to aglycone in the blood.
Supplementary Material
Acknowledgments
We thank Drs. Vincent Tam and Diana Chow for their expertise on pharmacokinetic analysis.
The work was supported by the National Institutes of Health Institute of General Medical Sciences [Grants GM070737] (to M.H.).
Article, publication date, and citation information can be found at http://dmd.aspetjournals.org.
 The online version of this article (available at http://dmd.aspetjournals.org) contains supplemental material.
The online version of this article (available at http://dmd.aspetjournals.org) contains supplemental material.
- UGT
- UDP-glucuronosyltransferase
- SULT
- sulfotransferase
- BCRP/Bcrp
- breast cancer resistance protein
- WT
- wild type
- Ko143
- 3-(6-isobutyl-9-methoxy-1,4-dioxo-1,2,3,4,6,7,12,12α-octahydropyrazino[1′,2′:1,6]pyrido[3,4-b]indol-3-yl)-propionic acid tert-butyl ester
- AUC
- area under the plasma concentration curve
- HBSS
- Hanks' balanced salt solution
- UPLC
- ultraperformance liquid chromatography
- MS/MS
- tandem mass spectrometry
- G-7-G
- genistein-7-O-glucuronide
- G-4′-G
- genistein-4′-O-glucuronide
- UDPGA
- uridine-5′-diphosphate-β, d-glucuronic acid
- G-4′-S
- genistein-4′-O-sulfate
- G-7-S
- genistein-7-O-sulfate
- N.A.
- not available
- MRP
- multidrug resistance-associated protein.
Authorship Contributions
Participated in research design: Yang, Zhu, and Hu.
Conducted experiments: Yang, Gao, Zhu, and Yin.
Performed data analysis: Yang, Jiang, and Hu.
Wrote or contributed to the writing of the manuscript: Yang and Hu.
References
- Adachi Y, Suzuki H, Schinkel AH, Sugiyama Y. (2005) Role of breast cancer resistance protein (Bcrp1/Abcg2) in the extrusion of glucuronide and sulfate conjugates from enterocytes to intestinal lumen. Mol Pharmacol 67:923–928 [DOI] [PubMed] [Google Scholar]
- Álvarez AI, Vallejo F, Barrera B, Merino G, Prieto JG, Tomás-Barberán F, Espín JC. (2011) Bioavailability of the glucuronide and sulfate conjugates of genistein and daidzein in breast cancer resistance protein 1 knockout mice. Drug Metab Dispos 39:2008–2012 [DOI] [PubMed] [Google Scholar]
- Banerjee S, Li Y, Wang Z, Sarkar FH. (2008) Multi-targeted therapy of cancer by genistein. Cancer Lett 269:226–242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J, Lin H, Hu M. (2003) Metabolism of flavonoids via enteric recycling: role of intestinal disposition. J Pharmacol Exp Ther 304:1228–1235 [DOI] [PubMed] [Google Scholar]
- Chen J, Lin H, Hu M. (2005a) Absorption and metabolism of genistein and its five isoflavone analogs in the human intestinal Caco-2 model. Cancer Chemother Pharmacol 55:159–169 [DOI] [PubMed] [Google Scholar]
- Chen J, Wang S, Jia X, Bajimaya S, Lin H, Tam VH, Hu M. (2005b) Disposition of flavonoids via recycling: comparison of intestinal versus hepatic disposition. Drug Metab Dispos 33:1777–1784 [DOI] [PubMed] [Google Scholar]
- Coldham NG, Sauer MJ. (2000) Pharmacokinetics of [14C]genistein in the rat: gender-related differences, potential mechanisms of biological action, and implications for human health. Toxicol Appl Pharmacol 164:206–215 [DOI] [PubMed] [Google Scholar]
- Coldham NG, Zhang AQ, Key P, Sauer MJ. (2002) Absolute bioavailability of [14C] genistein in the rat; plasma pharmacokinetics of parent compound, genistein glucuronide and total radioactivity. Eur J Drug Metab Pharmacokinet 27:249–258 [DOI] [PubMed] [Google Scholar]
- Enokizono J, Kusuhara H, Sugiyama Y. (2007) Effect of breast cancer resistance protein (Bcrp/Abcg2) on the disposition of phytoestrogens. Mol Pharmacol 72:967–975 [DOI] [PubMed] [Google Scholar]
- Gu L, House SE, Prior RL, Fang N, Ronis MJ, Clarkson TB, Wilson ME, Badger TM. (2006) Metabolic phenotype of isoflavones differ among female rats, pigs, monkeys, and women. J Nutr 136:1215–1221 [DOI] [PubMed] [Google Scholar]
- Hosoda K, Furuta T, Yokokawa A, Ishii K. (2010) Identification and quantification of daidzein-7-glucuronide-4′-sulfate, genistein-7-glucuronide-4′-sulfate and genistein-4′,7-diglucuronide as major metabolites in human plasma after administration of kinako. Anal Bioanal Chem 397:1563–1572 [DOI] [PubMed] [Google Scholar]
- Hu M, Borchardt RT. (1990) Mechanism of l-alpha-methyldopa transport through a monolayer of polarized human intestinal epithelial cells (Caco-2). Pharm Res 7:1313–1319 [DOI] [PubMed] [Google Scholar]
- Huang SM, Strong JM, Zhang L, Reynolds KS, Nallani S, Temple R, Abraham S, Habet SA, Baweja RK, Burckart GJ, et al. (2008) New era in drug interaction evaluation: US Food and Drug Administration update on CYP enzymes, transporters, and the guidance process. J Clin Pharmacol 48:662–670 [DOI] [PubMed] [Google Scholar]
- Imai Y, Tsukahara S, Asada S, Sugimoto Y. (2004) Phytoestrogens/flavonoids reverse breast cancer resistance protein/ABCG2-mediated multidrug resistance. Cancer Res 64:4346–4352 [DOI] [PubMed] [Google Scholar]
- Jia X, Chen J, Lin H, Hu M. (2004) Disposition of flavonoids via enteric recycling: enzyme-transporter coupling affects metabolism of biochanin A and formononetin and excretion of their phase II conjugates. J Pharmacol Exp Ther 310:1103–1113 [DOI] [PubMed] [Google Scholar]
- Kgomotso T, Chiu F, Ng K. (2008) Genistein- and daidzein 7-O-β-d-glucuronic acid retain the ability to inhibit copper-mediated lipid oxidation of low density lipoprotein. Mol Nutr Food Res 52:1457–1466 [DOI] [PubMed] [Google Scholar]
- Kitamura Y, Kusuhara H, Sugiyama Y. (2010) Functional characterization of multidrug resistance-associated protein 3 (mrp3/abcc3) in the basolateral efflux of glucuronide conjugates in the mouse small intestine. J Pharmacol Exp Ther 332:659–666 [DOI] [PubMed] [Google Scholar]
- Liu Y, Hu M. (2002) Absorption and metabolism of flavonoids in the Caco-2 cell culture model and a perused rat intestinal model. Drug Metab Dispos 30:370–377 [DOI] [PubMed] [Google Scholar]
- Maubon N, Le Vee M, Fossati L, Audry M, Le Ferrec E, Bolze S, Fardel O. (2007) Analysis of drug transporter expression in human intestinal Caco-2 cells by real-time PCR. Fundam Clin Pharmacol 21:659–663 [DOI] [PubMed] [Google Scholar]
- Nezasa K, Tian X, Zamek-Gliszczynski MJ, Patel NJ, Raub TJ, Brouwer KL. (2006) Altered hepatobiliary disposition of 5 (and 6)-carboxy-2′,7′-dichlorofluorescein in Abcg2 (Bcrp1) and Abcc2 (Mrp2) knockout mice. Drug Metab Dispos 34:718–723 [DOI] [PubMed] [Google Scholar]
- Ni Z, Bikadi Z, Rosenberg MF, Mao Q. (2010) Structure and function of the human breast cancer resistance protein (BCRP/ABCG2). Curr Drug Metab 11:603–617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pang KS, Maeng HJ, Fan J. (2009) Interplay of transporters and enzymes in drug and metabolite processing. Mol Pharm 6:1734–1755 [DOI] [PubMed] [Google Scholar]
- Prasain JK, Xu J, Kirk M, Smith Johnson M, Sfakianos J, Barnes S. (2006) Differential biliary excretion of genistein metabolites following intraduodenal and intravenous infusion of genistin in female rats. J Nutr 136:2975–2979 [DOI] [PubMed] [Google Scholar]
- Shelnutt SR, Cimino CO, Wiggins PA, Badger TM. (2000) Urinary pharmacokinetics of the glucuronide and sulfate conjugates of genistein and daidzein. Cancer Epidemiol Biomarkers Prev 9:413–419 [PubMed] [Google Scholar]
- Sfakianos J, Coward L, Kirk M, Barnes S. (1997) Intestinal uptake and biliary excretion of the isoflavone genistein in rats. J Nutr 127:1260–1268 [DOI] [PubMed] [Google Scholar]
- van de Wetering K, Feddema W, Helms JB, Brouwers JF, Borst P. (2009) Targeted metabolomics identifies glucuronides of dietary phytoestrogens as a major class of MRP3 substrates in vivo. Gastroenterology 137:1725–1735 [DOI] [PubMed] [Google Scholar]
- Wu B, Kulkarni K, Basu S, Zhang S, Hu M. (2011) First-pass metabolism via UDP-glucuronosyltransferase: a barrier to oral bioavailability of phenolics. J Pharm Sci 100:3655–3681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia CQ, Liu N, Yang D, Miwa G, Gan LS. (2005) Expression, localization, and functional characteristics of breast cancer resistance protein in Caco-2 cells. Drug Metab Dispos 33:637–643 [DOI] [PubMed] [Google Scholar]
- Yang Z, Gao S, Wang J, Yin T, Teng Y, Wu B, You M, Jiang Z, Hu M. (2011) Enhancement of oral bioavailability of 20(S)-ginsenoside Rh2 through improved understanding of its absorption and efflux mechanisms. Drug Metab Dispos 39:1866–1872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Gao S, Yin T, Kulkarni KH, Teng Y, You M, Hu M. (2010a) Biopharmaceutical and pharmacokinetic characterization of matrine as determined by a sensitive and robust UPLC-MS/MS method. J Pharm Biomed Anal 51:1120–1127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Kulkarni K, Zhu W, Hu M. (2012) Bioavailability and pharmacokinetics of genistein: mechanistic studies on its ADME. Anticancer Agents Med Chem (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z, Zhu W, Gao S, Xu H, Wu B, Kulkarni K, Singh R, Tang L, Hu M. (2010b) Simultaneous determination of genistein and its four phase II metabolites in blood by a sensitive and robust UPLC-MS/MS method: application to an oral bioavailability study of genistein in mice. J Pharm Biomed Anal 53:81–89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamek-Gliszczynski MJ, Day JS, Hillgren KM, Phillips DL. (2011) Efflux transport is an important determinant of ethinylestradiol glucuronide and ethinylestradiol sulfate pharmacokinetics. Drug Metab Dispos 39:1794–1800 [DOI] [PubMed] [Google Scholar]
- Zelcer N, Reid G, Wielinga P, Kuil A, van der Heijden I, Schuetz JD, Borst P. (2003) Steroid and bile acid conjugates are substrates of human multidrug-resistance protein (MRP) 4 (ATP-binding cassette C4). Biochem J 371:361–367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Zhang YD, Zhao P, Huang SM. (2009) Predicting drug-drug interactions: an FDA perspective. AAPS J 11:300–306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Song TT, Cunnick JE, Murphy PA, Hendrich S. (1999b) Daidzein and genistein glucuronides in vitro are weakly estrogenic and activate human natural killer cells at nutritionally relevant concentrations. J Nutr 129:399–405 [DOI] [PubMed] [Google Scholar]
- Zhou S, Hu Y, Zhang B, Teng Z, Gan H, Yang Z, Wang Q, Huan M, Mei Q. (2008) Dose-dependent absorption, metabolism, and excretion of genistein in rats. J Agric Food Chem 56:8354–8359 [DOI] [PubMed] [Google Scholar]
- Zhu W, Xu H, Wang SW, Hu M. (2010) Breast cancer resistance protein (BCRP) and sulfotransferases contribute significantly to the disposition of genistein in mouse intestine. AAPS J 12:525–536 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.