Abstract
Eosinophils are multifunctional immune cells that contribute to innate and adaptive immune/repair responses. Lactoferrin (LF) is an iron-binding protein indicated to alter cell adhesion and immune function by receptor-mediated interactions or by participating in redox mechanisms. The eosinophil adhesion molecules, αMβ2 and α4β1, are differentially expressed following exposure to the cytokine granulocyte-macrophage colony-stimulating factor (GM-CSF) and various redox agents. We hypothesized that LF can alter the function and production of proteins involved in adhesion/migration. Utilizing eosinophil peroxidase activity or fluorescent labeling adhesion assays, LF reduced GM-CSF-induced eosinophil adhesion in the presence of fibronectin or vascular adhesion molecule-1 compared with GM-CSF treatment alone. Flow cytometric analysis of eosinophil αM (CD11b) and α4 (CD49d) integrins revealed that cotreatments (24 h) with LF plus GM-CSF induced a significant increase in CD11b compared with control and GM-CSF treatments but a significant decrease in CD49d compared with control and GM-CSF treatments. These changes in CD11b and CD49d levels were significantly correlated with the increased production of chemokines (macrophage inflammatory Protein-1α, monocyte chemotactic protein-1) and an identified increase in S100A9 production. Thus, LF release at sites of inflammation may alter eosinophil recruitment/activation and possibly the progression of diseases such as cancer and asthma where significant eosinophil influx has been described.
Introduction
Eosinophils are multifunctional immune cells that accumulate in various tissues in response to normal development (gastrointestinal, mammary, uterine, and thymic) and chronic inflammation (Kita 2011; Woodruff and others 2011). Eosinophils may participate in tissue homeostasis, wound/repair processes, and the innate and adaptive immune responses via the release of various granule proteins, growth factors, lipid mediators, cytokines, and chemokines (Kita 2011). Eosinophil migration from blood vessels into tissue has been proposed to involve mediator-induced activation of eosinophil cell adhesion molecules, eosinophil adhesion to endothelia, and subsequent eosinophil transmigration (Tachimoto and others 2002; Lampinen and others 2004). A functional role of certain cell adhesion molecules, that is, the integrins, has been identified in the progression of chronic inflammatory disease (Singh and others 2004; Garmy-Susini and others 2010).
The integrins α4β1 (CD49d/CD29, very late antigen-4) and αMβ2 (CD11b/CD18, Mac-1, complement receptor type 3) are 2 particular integrins that have been extensively studied in eosinophils (Barthel and others 2008). The α4β1 integrin has been reported to mediate cell–cell and cell–extracellular matrix contacts to vascular adhesion molecule (VCAM)-1 and fibronectin, respectively (Johansson and others 1997). The αMβ2 integrin is thought to initiate phagocytosis via interaction with complement component iC3b and, analogous to α4β1, αMβ2 is indicated to mediate cell–cell and cell–extracellular matrix contacts by binding interactions with intercellular adhesion molecule-1, VCAM-1, fibrinogen, and vitronectin (Ross 2002; Barthel and others 2008). In human primary blood eosinophils, increased cell surface expression of αMβ2, but not α4β1, has been found to occur in response to the cytokines interleukin (IL)-3, IL-5, and granulocyte-macrophage colony-stimulating factor (GM-CSF; Wong and others 2003). Research has also indicated that expression of αMβ2 is increased, whereas, the α4β1 is decreased in eosinophilia patients compared with normal controls (Kayaba and others 2001). Differential eosinophil expression of αMβ2 and α4β1 has also been found to occur in response to reactive oxygen species (Nagata and others 2000; Chuang and others 2003), suggesting that redox reactions are one of the underlying mechanisms involved in modulating eosinophil activation and recruitment.
Low affinity lactoferrin (LF) receptors have been identified on eosinophils (Spik and others 1994). LF is an iron-binding glycoprotein released from neutrophils at the site of inflammation and secreted by epithelial cells that contribute to the composition of bodily fluids (milk, tears, saliva, bile, and pancreatic fluid; Legrand and Mazurier 2010). Studies have indicated that either bovine or human LF is able to modulate the proliferation of various cell types (eg, tumor cells, osteoblasts, and fibroblasts) and alter the host immune responses to allergens and cancer (Brock 2002; Kruzel and others 2006; Chodaczek and others 2007; Nadolska and others 2010; Li and others 2011; Naot and others 2011). The functions of LF have been attributed to signaling events arising from the binding interactions of the protein with host (CD14, heparan sulfate proteoglycans, intelectin, and LDL receptor-related protein) or pathogen (lipopolysacharide and bacterial unmethylated CpG DNA) cell surface molecules (Brock 2002; Suzuki and others 2005; Legrand and Mazurier 2010) and from the iron-binding properties of the molecule (Klebanoff and Waltersdorph 1990; Sandomirsky and others 2003; Chodaczek and others 2007). These various processes have been reported to inhibit tumorigenesis (Brock 2002) and to reduce allergic inflammation (Kruzel and others 2006; Chodaczek and others 2007). In part, these biological effects may be a response to the reduced eosinophil migration found to occur after LF treatment (Bournazou and others 2010).
Based on this prior information regarding the biological functions of LF, we tested the hypothesis that LF can alter the function and production of proteins involved in migration by examining eosinophil adhesion, integrin expression, and the production of chemokines and calgranulins (S100A9). The data herein support the idea of LF as an important regulator of eosinophil responses and provide insight into the inflammatory reactions observed in cases of allergy and asthma in addition to certain cancers.
Materials and Methods
Isolation of peripheral blood eosinophils
Peripheral blood was obtained from human allergic subjects under informed consent. The study was approved by the University of Wisconsin-Madison Center for Health Sciences Human Subjects Committee. Eosinophils were purified from heparinized blood that was diluted with Hank's buffered salt solution without Ca2+, Mg2+, and phenol red (HBSS; Mediatech) and centrifuged for 20 min at 700×g over 1.090 g/mL Percoll. A granulocyte fraction was obtained after removal of the plasma, mononuclear cell band, and Percoll. Granulocytes were then subjected to red blood cell lysis via hypotonic shock, washed with 4°C HBSS supplemented with 2% new born calf serum (Life Technologies), and incubated 40 min with magnetic beads coated with anti-CD16, anti-CD14, and anti-CD3 (Miltenyi Biotechnology) prior to negative selection with an AutoMACS separator (Miltenyi Biotechnology). The recovered mixture (>97% purity, >98% viability) was evaluated via Giemsa's-based Diff-Quik stain (Baxter Scientific Products) and trypan blue exclusion respectively.
Assay of eosinophil peroxidase activity
Cellular eosinophil peroxidase (EPO) activity was determined using adherent cells in a manner similar to that detailed in previous studies (Strath and others 1985; Nagata and others 2000; Barthel and others 2006; Bates and others 2010). Purified human blood eosinophils were added to protein-coated surfaces that were prepared by applying 10 μg/mL of either soluble recombinant human VCAM-1, purified human fibronectin (both kindly provided by Dr. Deane Mosher, University of Wisconsin-Madison), or bovine serum albumin (BSA, A-6003; Sigma Chemical Co.) in TBS (pH 8) to triplicate wells of a 96-well plate (Sarstedt) for 4 h at 37°C. Coating buffers were aspirated and eosinophils (1×105/mL) suspended in 0.1% human serum albumin (HSA; Irvine Scientific) HBSS with Ca2+, Mg2+, but without phenol red (Mediatech) and treated with buffer control, 100 pM GM-CSF (R&D Systems) or 0.3–10 μg/mL bovine LF (Morinaga Milk Industry Co., Ltd.) for 1 h at 37°C. Additional eosinophils (1×105/mL) were saved on ice for use in a standard curve. After 1 h at 37°C, the plate was washed thrice with 200 μL of 37°C TBS and 100 μL of 37°C HBSS with Ca2+, Mg2+, but without phenol red, was added to each well. Additional eosinophils were plated in triplicate (100 μL/well) and serially diluted for use as a standard curve. To identify the presence of eosinophils in the assay, 100 μL of reaction buffer containing 0.1% Triton® X-100 (Sigma Chemical Co.), 50 mM Tris (pH 8), 1 mM H2O2, and 1 mM O-phenylenediamine dihydrochloride (OPD; Sigma Chemical Co.) was added to each well for 30 min to allow for oxidation of OPD and the consequent production of OPD diimine colored derivatives via the released EPO. After 30 min, the reaction was stopped with 4 M H2SO4 (50 μL) and optical density (OD) was measured at 490 nm. The absorbance levels of EPO activity were used as an indirect measurement of eosinophil adherence by examining the OD means of the samples relative to the standard curve.
Carboxyfluorescein diacetate succinimidyl ester labeling as a measure of adherence
Eosinophils (2×106) were stained with Cell Trace™ carboxyfluorescein diacetate succinimidyl ester (CFSE) (2 μM, C34554; Invitrogen-Molecular Probes) per manufacturer's instructions. CFSE-labeled eosinophils were suspended in 10 mL of 0.1% fetal bovine serum (FBS; Hyclone) HBSS with Ca2+, Mg2+, but without phenol red. In a 96-well plate (Sarstedt) coated with 10 μg/mL of fibronectin or VCAM-1, cells were plated in triplicate (100 μL) and treated with vehicle control, 100 pM GM-CSF, 10 μg/mL bovine LF (Morinaga) or 10 μg/mL human LF (Cell Sciences) for 1 h at 37°C. Additional eosinophils (2×105/mL) were saved on ice for use in a standard curve. After 1 h at 37°C, the plate was washed thrice with 200 μL of 37°C TBS and 100 μL of 37°C HBSS with Ca2+, Mg2+, but without phenol red, was added to each well. Additional eosinophils were plated in triplicate (100 μL/well) and serially diluted for use as a standard curve. The plate was immediately read on a Synergy™ 2 Multi-Detection Microplate Reader (Bioteck® Instruments, Inc.) at an excitation of 485/20 and emission of 528/20. The CFSE fluorescence levels were used as a measure of eosinophil adherence by examining the average fluorescence of the samples relative to the standard curve.
Flow cytometric analysis of cell surface molecules
Eosinophils (2×106/mL) were suspended in 0.5 mL of 0.1% HSA (Irvine Scientific) RPMI (Mediatech) and placed in 24-well plates (Sarstedt) coated with 10 μg/mL fibronectin. Cells were treated with buffer control, 100 pM GM-CSF (R&D Systems), 10 mM N-acetyl-l-cysteine (NAC; Sigma Chemical Co.), or bovine LF (Morinaga) for 24 h. Eosinophils were then aspirated from the wells and treated with 5 μL of phycoerythrin-conjugated anti-CD11b (Clone ICRF44, 555388; BD Biosciences Pharmingen), 5 μL of allophycocyanin-conjugated anti-CD49d (Clone 9F10, 304307; Biolegend), or antibody isotype control for 30 min at 4°C. Cells were washed with 1 mL of 1% FBS RPMI (Mediatech) and suspended in 300 μL PBS. To exclude dead cells, propidium iodide (3 μg/mL) was added to each sample. A total of 10,000 events were assessed on a FACScan flow cytometer (Becton–Dickinson) at the University of Wisconsin Carbone Cancer Center Flow Cytometry Core Facility. Data were analyzed with FlowJo data analysis software (TreeStar, Ashland, OR).
Enzyme-linked immunosorbent assay
Monoclonal anti-macrophage inflammatory protein (MIP)-1α antibodies (clone 14215, MAB670; R&D Systems, 1:250) or monoclonal anti-monocyte chemotactic protein (MCP)-1 antibodies (555055; BD Biosciences Pharmingen, 1:500) in 0.1 M sodium carbonate buffer (pH 9.6) were coated onto 96-well EIA/RIA plates (Costar). Blocking buffer containing 1% BSA (Sigma Chemical Co.) and 0.5% Tween®20 (Fischer Scientific) in PBS (pH 7.4) was added to wells for 2 h at room temperature. Serial dilutions of MIP-1α standard (270-LD; R&D Systems), MCP-1 standard (554620; BD Biosciences Pharmingen), or cell-free supernatants were aliquoted and incubated at 4°C overnight. Eosinophil-secreted proteins were assayed using biotinylated MIP-1α antibodies (BAF270; R&D Systems, 1:3,000) or biotinylated MCP-1 antibodies (554664; BD Biosciences Pharmingen, 1:1,000), and subsequent detection with strepavidin-horseradish peroxidase-40 (HRP-40; Fitzgerald Industries International). A colorimetric HRP substrate tetramethylbenzidine (Biofx Laboratories) was used to evaluate captured HRP activity and the enzymatic reaction was stopped by the addition of 0.18 M sulfuric acid. The OD was determined on an ELX800 Universal Microplate Reader (BioTek Instruments, Inc.). Absorbance was quantified at 450 nM, using 600 nM as a reference wavelength. Concentrations were calculated by interpolation from a standard curve with an assay sensitivity of 12.5 pg/mL. All determinations were performed in triplicate.
Immunoblotting
Cells were solubilized with lysis buffer [1% sodium dodecyl sulfate (SDS)], 1 mM dithiothreitol, 0.5 mM Na3VO4, 1 mM EDTA, 10% glycerol, 10 mM Tris (pH 8.0), sonicated, and boiled (5 min). Concentrated cell lysis buffer (10×) was added to conditioned media (1:10 dilution) and boiled (5 min). Proteins were loaded onto a 15% SDS-polyacrylamide gel electrophoresis gel and transferred onto 0.45 μm Immobilion-P polyvinylidene difluoride membranes (Millipore). Blots were cut for multiple detection and incubated with antibodies raised against human actin (BD Biosciences; 612656, 1:5,000) or S100A9 (Santa Cruz Biotechnology; 1:200). Blots were washed and subsequently incubated with HRP-conjugated secondary antibodies. Bound secondary antibody was visualized following incubation of the membrane with Super Signal West chemiluminescent HRP substrate (Thermo Scientific Pierce) and using an Epichemi II darkroom UVP equipped with a 12-bit cooled CCD camera. Luminescence was quantified and evaluated via the application of ImageJ software (National Institutes of Health).
Statistical analysis
Changes in CD49d, CD11b, MIP-1α, and MCP-1 from cell incubations involving treatments with GM-CSF alone to GM-CSF plus LF (GM-CSF+LF), GM-CSF plus the antioxidant NAC (GM-CSF+NAC), or GM-CSF plus LF and NAC (GM-CSF+LF+NAC) were expressed as ratios. The associations between changes in CD49d or CD11b with MIP-1α, and MCP-1 were evaluated using the Spearman rank correlation coefficient. All additional analyses were performed using a paired Student's t-test. A 2-sided P value of <0.05 was regarded as statistically significant.
Results
LF modulated human blood eosinophil adherence to fibronectin as measured by EPO activity
LF is found in airway lavage fluid (1–100 μg/mL) and has been indicated to inhibit cell adhesion by interacting with RGD motifs required for fibronectin binding to certain integrins (Travis and others 1999; Sakamoto and others 2006). Conversely, the cytokine GM-CSF is known to induce eosinophil adherence to fibronectin (Anwar and others 1993; Bates and others 2010). To test the idea that LF can block GM-CSF-induced eosinophil adhesion to fibronectin, we performed EPO assays as an indirect measurement of eosinophil adherence using bovine LF. Plates were coated with a positive control of 10 μg/mL BSA (a previously identified beta2 ligand; Zhu and others 2000) and 10 μg/mL fibronectin as described in Materials and Methods. As shown in Fig. 1, GM-CSF treatment alone significantly (P<0.02) enhanced eosinophil adherence/EPO activity to fibronectin compared with the control. Addition of increasing concentrations of LF to GM-CSF-treated wells significantly inhibited eosinophil adherence/EPO activity in the presence of fibronectin (P<0.02) but not in the presence of BSA compared with GM-CSF treatment alone in experiments using eosinophils from 5 different donors (see Fig. 1).
FIG. 1.
LF-modulated adherence to fibronectin compared to BSA. Purified human blood eosinophils (1×105cells/mL) were added to plates coated with 10 μg/mL BSA or 10 μg/mL FN. Cells were treated with 100 pM GM-CSF or increasing concentrations of bovine LF for 1 h. EPO activity was assessed at 490 nm as described under Materials and Methods. The average number of eosinophils adhered relative to a standard curve is displayed±SEM, n=5, *P<0.02 versus buffer control, †P<0.04 versus GM-CSF alone. GM-CSF, granulocyte-macrophage colony-stimulating factor; EPO, eosinophil peroxidase; BSA, bovine serum albumin; LF, lactoferrin; FN, fibronectin.
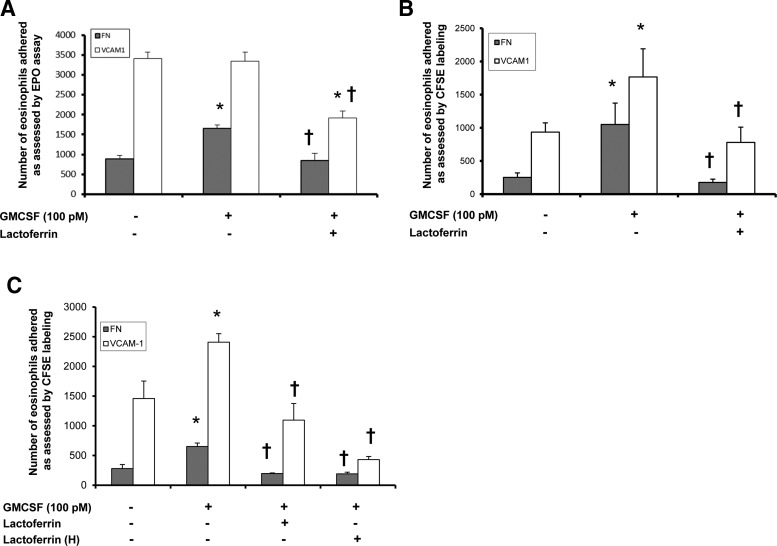
LF modulation of eosinophil adherence to VCAM-1 as measured by EPO activity
To test whether LF also modulates eosinophil adherence/EPO activity when plated on VCAM-1, EPO assays were performed on cells treated with 100 pM GM-CSF±10 μg/mL bovine LF and plated atop fibronectin or VCAM-1-coated wells as described in Materials and Methods. As shown in Fig. 2A, GM-CSF significantly enhanced eosinophil adherence/EPO activity to fibronectin (P<0.02) but not VCAM-1 compared with the control. In concurrence with previous data in Fig. 1, LF significantly reduced eosinophil adherence/EPO activity following GM-CSF treatment in the presence of fibronectin (P=0.03) and VCAM-1 (P=0.01) when compared with GM-CSF treatment alone. These assessments were performed using eosinophils from 5 different donors.
FIG. 2.
LF-modulated adherence to FN and VCAM-1. Purified human blood eosinophils were cultured in plates coated with 10 μg/mL FN or 10 μg/mL VCAM-1. Cells were treated with 100 pM GM-CSF and 10 μg/mL bovine or human (H) LF for 1 h and examined for eosinophil adherence via EPO activity or CFSE labeling. (A) The average number of eosinophils adhered relative to an EPO activity standard curve is displayed±SEM, n=5, *P<0.02 versus buffer control, †P<0.03 versus GM-CSF. (B) The average number of eosinophils adhered relative to a CFSE fluorescence standard curve is displayed±SEM, n=8, *P<0.03 versus buffer control, †P<0.04 versus GM-CSF. (C) Bovine versus human (H) LF: The average number of eosinophils adhered relative to a CFSE fluorescence standard curve is displayed±SEM, n=4, *P<0.04 versus buffer control, †P<0.05 versus GM-CSF; VCAM-1, vascular adhesion molecule-1; CFSE, carboxyfluorescein diacetate succinimidyl ester.
LF modulation of eosinophil adherence as measured by CFSE labeling
To further assess the potential functions of LF on eosinophil adhesion, and to validate the aforementioned cell adhesion measurements via EPO activity, we also labeled eosinophils with a fluorescent dye and assessed cell adhesion using fluorspectroscopic analyses. Specifically, fluorescently (CFSE) labeled eosinophils were plated in wells coated with 10 μg/mL fibronectin or VCAM-1 and treated with 100 pM GM-CSF±10 μg/mL bovine or human LF for 1 h. Residual fluorescence was examined after repeated plate washings as detailed in Materials and Methods. As shown in Fig. 2B and C, we observed that GM-CSF treatment significantly enhanced the relative fluorescence in the presence of fibronectin (P<0.03) and VCAM-1 (P<0.04) compared with the control. Similar to Fig. 2A, LF (bovine or human) treatment significantly reduced the relative fluorescence of GM-CSF cultures in the presence of fibronectin and VCAM-1 compared with GM-CSF treatment alone.
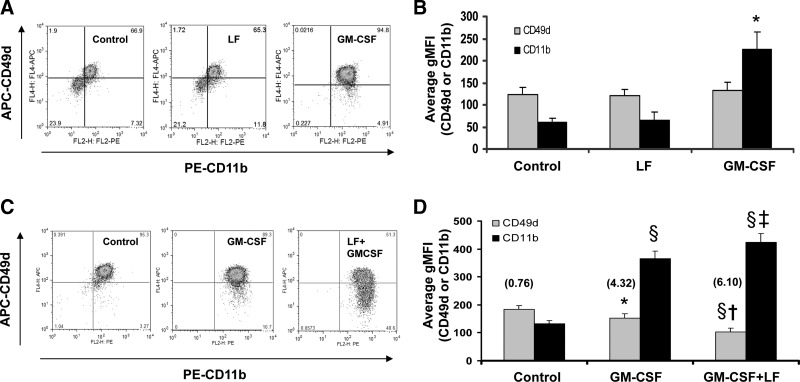
LF-modulation of eosinophil integrin expression
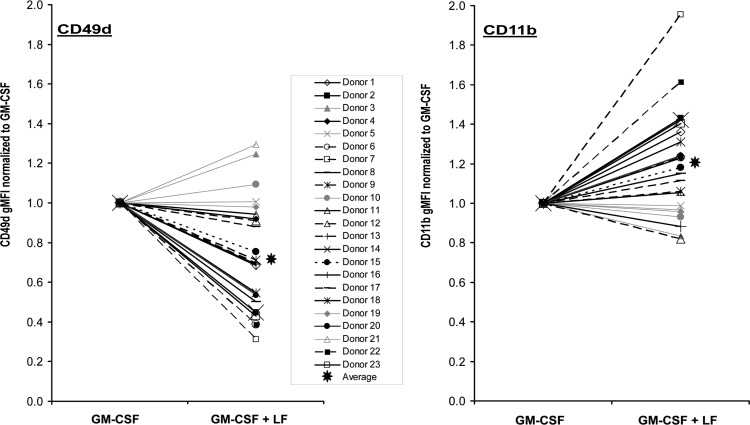
Because eosinophil adhesion can be mediated by the expression of integrins (Barthel and others 2006), we examined α4 (CD49d) and αM (CD11b) integrin subunit expression by flow cytometry after 24 h of cell culture on fibronectin-coated wells and following treatments with vehicle control, 100 pM GM-CSF, or 10 μg/mL bovine LF. As summarized in Fig. 3B, treatment of eosinophils from 4 different donors with LF alone did not yield a detectable effect on integrin expression when compared with the control. Also, treatment with GM-CSF enhanced CD11b expression but had no discernible effect on CD49d as previously reported (Wong and others 2003). However, as shown in Fig. 3C, representative dot plots of live cells reveal enhanced CD11b expression but reduced CD49d expression upon cotreatment with GM-CSF and LF when compared with cells treated with either buffer control or GM-CSF treatment alone. In Fig. 3D, the geometric mean fluorescent intensity (gMFI) from the flow cytometric analyses of CD49d and CD11b surface expression from 23 different donors is charted. We also listed the ratio of CD11b gMFI/CD49d gMFI. These data support the general capacity of LF to upregulate CD11b expression (P=0.0008) but downregulate CD49d expression (P=0.0002) in the context of cotreatment with GM-CSF. In Fig. 4, each of the 23 donors is plotted with respect to their response to GM-CSF alone and GM-CSF plus LF. Each data point is normalized to the GM-CSF treatment and the representative trends for these 23 donors and their expression of CD49d and CD11b are displayed. Using the Spearman rank correlation coefficient and comparing changes from GM-CSF to GM-CSF plus LF in these 23 donors, a significant inverse correlation was also identified (R=−0.82, P<0.0001) between CD49d and CD11b. This response was not related to gender (13 men, 10 women), medications, or forced expiratory volume (1.0 s) percent predicted values (data not shown). However, a few donors, who did not respond to LF treatment with reduced CD49d and increased CD11b, were found to have a higher than the average fixed exhalation nitric oxide rate or a higher than average percentage of eosinophils in their white blood cell count (data not shown).
FIG. 3.
LF-modulated CD49d and CD11b expression. Purified human blood eosinophils (2×106cells/mL) were cultured in plates coated with 10 μg/mL fibronectin (FN). Cells were treated with buffer control, 100 pM GM-CSF, and/or 10 μg/mL bovine LF for 24 h. Live cells were gated (A, C) and charted (B, D) for integrin expression. CD11b and CD49d are identified via flow cytometry and reported as the gMFI. Charts represent treatment with LF alone (B) (±SEM, n=4, *P=0.01 versus CD11b buffer control) or cotreatment with LF and GM-CSF (D) (±SEM, n=23 *P<0.02 versus CD49d buffer control, §P<0.0001 versus respective buffer control, †P=0.0002 versus CD49d GM-CSF, ‡P=0.0008 versus CD11b GM-CSF) compared to controls. The values in parenthesis indicate the ratio of CD11b gMFI/CD49d gMFI. gMFI, geometric mean fluorescent intensity.
FIG. 4.
Donor to donor variability in LF-modulated CD49d and CD11b expression. Purified human blood eosinophils from each of the 23 donors charted in Figure 3D are plotted. Identified markers represent the CD49d or CD11b gMFI of eosinophils treated with 100 pM GM-CSF±10 μg/mL bovine LF normalized to the GM-CSF treatment.
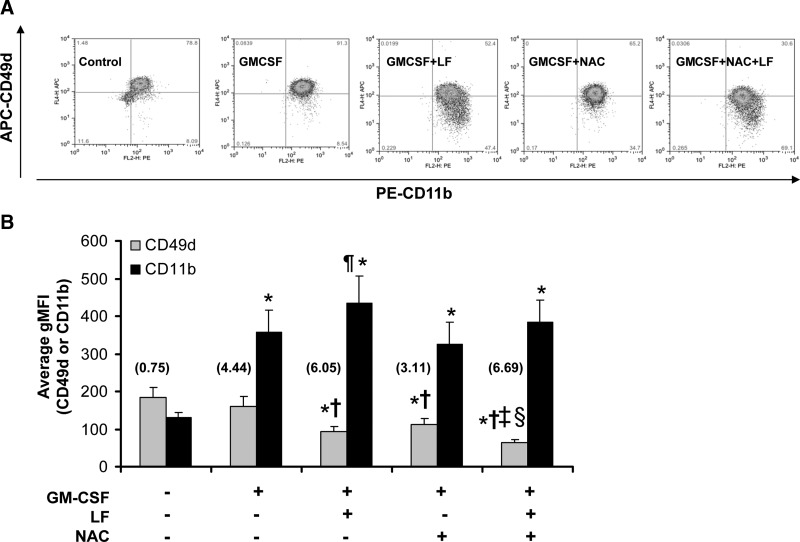
LF-modulated eosinophil integrin expression compared to NAC
The activation of eosinophil α4β1 has been reported to occur in response to superoxide exposure (Chuang and others 2003). To test the hypothesis that GM-CSF-treated eosinophil integrin expression is altered by antioxidants, eosinophils from 9 different donors were cultured in fibronectin-coated wells and treated with vehicle control, 100 pM GM-CSF, 10 μg/mL bovine LF, and/or 10 mM NAC for 24 h. As shown in Fig. 5, cotreatment of eosinophils with GM-CSF and LF led to enhanced CD11b expression but significantly decreased CD49d expression compared with GM-CSF treatment alone, which is similar to results presented in Figs. 3 and 4. Addition of NAC to eosinophils cultured with GM-CSF also promoted the downregulation of CD49d (P=0.03) but had no effect on CD11b surface expression when compared with GM-CSF treatment alone. Incubation with LF, NAC, and GM-CSF together resulted in a synergistic downregulation of CD49 that was significantly lower than that observed with eosinophils cultured with GM-CSF plus either LF (P=0.03) or NAC (P=0.002) alone. The expression of CD11b in these samples was not significantly different than the controls but trended lower than eosinophils treated with GM-CSF plus LF alone.
FIG. 5.
LF-modulated CD49d and CD11b expression compared to NAC. Purified human blood eosinophils (2×106cells/mL) were cultured in plates coated with 10 μg/mL FN. Cells were treated with buffer control, 100 pM GM-CSF, 10 mM NAC, and/or 10 μg/mL bovine LF for 24 h. Live cells were gated (A) and charted (B) for integrin expression. CD11b and CD49d are identified using flow cytometry and reported as the gMFI,±SEM, n=9, *P<0.02 versus respective buffer control, †P<0.03 versus GM-CSF, ‡P=0.03 versus GM-CSF+LF, §P=0.002 versus GM-CSF+NAC, ¶P<0.008 versus GM-CSF+NAC. The values in parenthesis indicate the ratio of CD11b gMFI/CD49d gMFI. NAC, N-acetyl-l-cysteine; FN, fibronectin.
LF-modulation of eosinophil chemokine production
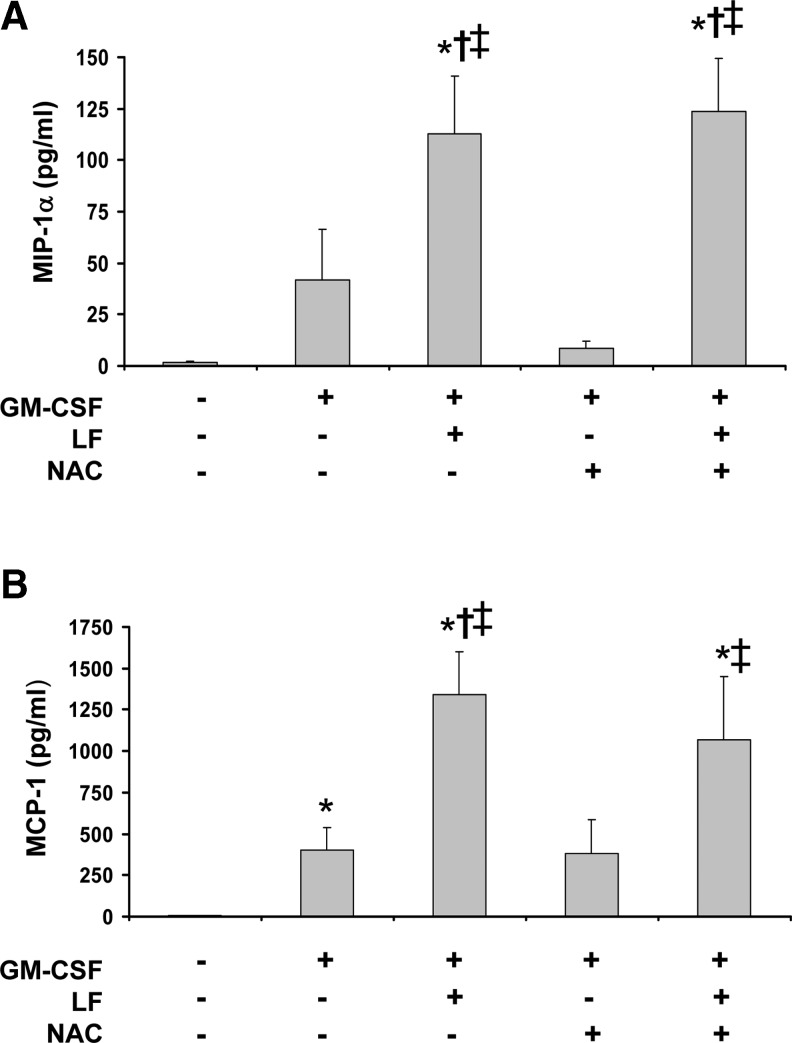
Leukocyte adhesion to fibronectin via β1 integrins can be rapidly induced and subsequently reduced by exposure to the chemokines, MIP-1α and MCP-1 (Carr and others 1996; Weber and others 1996). To test whether LF affects the production of these chemokines, cell-free conditioned media were isolated from eosinophils cultured on fibronectin-coated surfaces and treated with vehicle control, 100 pM GM-CSF, 10 μg/mL bovine LF, and/or 10 mM NAC for 24 h. Initial evaluation of MIP-1α and MCP-1 via ELISA indicated that LF alone did not induce chemokine production (n=7, data not shown). The additional study of 16 different donors (Fig. 6) revealed that in the presence of GM-CSF, LF, but not NAC, chemokine production was significantly induced (P<0.05) compared with either vehicle or GM-CSF treatment alone. Further, in examining changes in chemokine production versus changes in integrin expression from each of the 9 donors in Fig. 5 using the Spearman rank correlation coefficient, significant correlations were revealed (Table 1). Subjects with larger decreases in CD49d from GM-CSF to GM-CSF plus LF had larger increases in both MIP-1α (R=−0.75, P=0.03) and MCP-1 (R=−0.80, P=0.01) from GM-CSF to GM-CSF plus LF. Subjects with larger increases in CD11b from GM-CSF to GM-CSF plus LF also had larger increases in both MIP-1α (R=+0.72, P=0.04) and MCP-1 (R=−0.68, P=0.05) from GM-CSF to GM-CSF plus LF. Similar to data shown in Fig. 4, cells obtained from subjects that exhibited larger decreases in CD49d from GM-CSF alone to GM-CSF plus LF also had larger increases in CD11b (R=−0.85, P=0.006). These correlations were not significant in comparing changes between GM-CSF and GM-CSF plus NAC. Additionally, subjects with larger increases in MIP-1α had larger increases in MCP-1 (R=+0.92, P=0.001). These correlations suggest that the combined cell signals of LF and GM-CSF enhance chemokine production that may be indirectly involved in the upregulation of CD11b and downregulation of CD49d.
FIG. 6.
LF-modulated MIP-1α and MCP-1 production. Purified human blood eosinophils (2×106cells/mL) were cultured in plates coated with 10 μg/mL FN. Cells were treated with buffer control, 100 pM GM-CSF, 10 mM NAC, and/or 10 μg/mL bovine LF for 24 h. Cell-free conditioned media was measured in triplicate per experiment for the specified protein. Repeat experiments were averaged and are displayed as the mean concentration (pg/mL). Charted concentrations represent the chemokines MIP-1α (A) (mean±SEM, n=16, *P<0.003 versus control, †P<0.05 versus GM-CSF, ‡P<0.004 versus GM-CSF+NAC) and MCP-1 (B) (mean±SEM, n=16, *P<0.01 versus control, †p=0.001 versus GM-CSF, ‡P<0.004 versus GM-CSF+NAC). MIP-1α, macrophage inflammatory protein-1alpha; MCP-1, mononocyte chemotactic protein-1; FN, fibronectin.
Table 1.
Spearman Rank Correlation Coefficients Comparing Changes in Integrin Expression to Changes in Chemokine Production Based on the Treatments of GM-CSF and GM-CSF Plus LF and/or NAC
| CD49d versus MIP-1α | CD49d versus MCP-1 | CD11b versus MIP-1α | CD11b versus MCP-1 | MIP-1α versus MCP-1 | |
|---|---|---|---|---|---|
| GM-CSF-(GM-CSF+LF) | R=−0.75, P=0.03a | R=−0.80, P=0.01a | R=+0.72, P=0.04b | R=+0.68, P=0.05b | R=+0.92, P=0.001b |
| GM-CSF-(GM-CSF+NAC) | R=+0.03, P=0.95 | R=−0.32, P=0.41 | R=+0.60, P=0.10 | R=+0.40, P=0.29 | R=+0.58, P=0.11 |
| GM-CSF-(GM-CSF+LF+NAC) | R=−0.33, P=0.39 | R=−0.47, P=0.21 | R=+0.97, P=0.0002b | R=+0.88, P=0.003b | R=+0.93, P=0.0007b |
Purified human blood eosinophils treated with GM-CSF, GM-CSF plus LF (GM-CSF+LF), GM-CSF plus NAC (GM-CSF+NAC), or GM-CSF plus LF and NAC (GM-CSF+LF+NAC) from each of the 9 donors in Fig. 5 were examined for statistical dependence with respect to the variables of integrin gMFI (CD49d, CD11b) and chemokine production (MIP-1α, MCP-1). The changes in CD49d, CD11b, MIP-1(, and MCP-1 from GM-CSF alone to (GM-CSF+LF), (GM-CSF+NAC), or (GM-CSF+LF+NAC) were expressed as ratios. The associations between changes in CD49d or CD11b with MIP-1(and MCP-1 were evaluated using the Spearman rank correlation coefficient.
Inverse correlation.
Positive correlation (n=9).
GM-CSF, granulocyte-macrophage colony-stimulating factor; NAC, N-acetyl-l-cysteine; LF, lactoferrin; gMFI, geometric mean fluorescent intensity; MIP-1α, macrophage inflammatory protein-1α; MCP-1, monocyte chemotactic protein-1.
Eosinophil calgranulin production
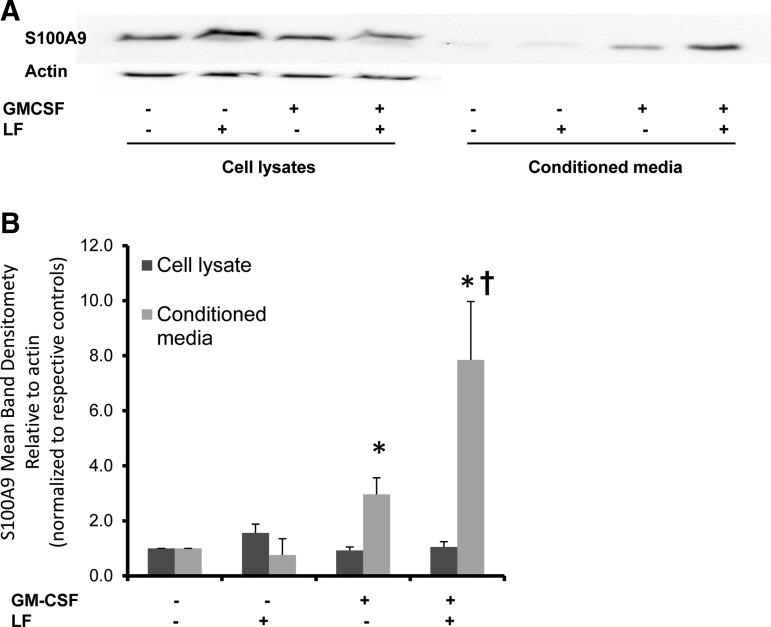
Increased expression of CD11b in neutrophils has been found to be regulated by calgranulin B (Newton and Hogg 1998; Manitz and others 2003). We have previously identified GM-CSF-induced calgranulin B (S100A9) production in eosinophils (Curran and Bertics 2011; Curran and others 2011). In Figs. 3–5, we show GM-CSF-induced CD11b expression that is enhanced by cotreatment with LF. To test whether LF enhances GM-CSF-induced S100A9 production, eosinophils from 4 different donors were cultured in fibronectin-coated wells and treated with vehicle control, 100 pM GM-CSF, or 10 μg/mL bovine LF for 24 h. Cell lysates and conditioned media were examined for calgranulin B (S100A9) by immunoblotting as detailed in Materials and Methods. As illustrated in Fig. 7, S100A9 is significantly produced in the conditioned media from eosinophils treated with GM-CSF compared with the conditioned media control (P=0.03). Cotreatment of LF with GM-CSF enhanced S100A9 production in the conditioned media compared with the controls of GM-CSF alone (P<0.05) and control buffer (P<0.04).
FIG. 7.
Immunoblot detection of intracellular and extracellular eosinophil S100A9 in response to LF. Purified human blood eosinophils (5×106/mL) were cultured in plates coated with 10 μg/mL FN. Cells were suspended in 0.1% human serum albumin RPMI and treated with buffer control, 100 pM GM-CSF or 10 μg/mL bovine LF for 24 h. Total eosinophil cell lysates from 5×105 cells and the total corresponding conditioned media were loaded onto a 15% sodium dodecyl sulfate–polyacrylamide gel electrophoresis gel. A representative immunoblot is displayed in (A). Data were also quantified with ImageJ software, expressed as S100A9 mean band densitometry relative to actin controls and normalized to the respective media control, n=4, *P<0.04 versus conditioned media buffer control, †P<0.05 versus conditioned media GM-CSF control.
Discussion
Eosinophils are innate immune cells that are characteristic of both healthy and diseased tissues (Kita 2011). The mechanisms that propagate eosinophils in homeostatic and inflammatory conditions are not clearly known (Kita 2011; Woodruff and others 2011). Like the eosinophil, the protein LF is found in homeostatic (eg, mammary gland) and inflamed [eg, asthmatic lung and inflammatory bowel disease (IBD)] tissue (Kayazawa and others 2002; Monteseirin 2009; Legrand and Mazurier 2010; Kita 2011). LF is secreted by activated neutrophils, a cell type also recruited in tandem with eosinophils in certain cases of asthma and IBD (Sampson 2000; Jeziorska and others 2001; Legrand and Mazurier 2010). Of interest, treatment of eosinophils with LF reportedly reduces eotaxin-stimulated eosinophil migration (Bournazou and others 2010), suggesting that LF may differentially affect cell signals associated with the expression of eosinophil adhesion molecules. To understand the nature of LF-induced eosinophil responses, we tested the hypothesis that LF can significantly alter the function, expression, and production of proteins involved in migration by examining eosinophil adhesion, integrin detection, and the production of chemokines and calgranulins.
Eosinophil adhesion to fibronectin and VCAM-1 can occur via binding interactions involving the integrins α4, αM, β1, β2, and β7 (Anwar and others 1994; Walsh and others 1996; Matsumoto and others 1997; Barthel and others 2006). The cytokine GMCSF is known to induce eosinophil adherence to fibronectin (Anwar and others 1993; Bates and others 2010), whereas, LF has been indicated to inhibit the adhesion of various cell lines to fibronectin (Sakamoto and others 2006). In response to 1 h culture, we show that increasing concentrations of LF inhibit GM-CSF-induced adhesion in response to fibronectin but not a characterized β2 ligand, BSA (Zhu and others 2000). We also identified that LF inhibits GM-CSF-induced adhesion to VCAM-1 via EPO assay (Fig. 2A) and a CFSE labeling adhesion assay (Fig. 2B). Interestingly, GM-CSF treatment enhanced adhesion via CFSE labeling (Fig. 2B) but not via EPO assay (Fig. 2C) in response to the VCAM-1 substrate, suggesting that donor to donor responses vary or that possibly during cell culture, EPO may be nonspecifically released from cells and thereby adhere to wells as has been previously described (Adamko and others 2004). In Fig. 2C, we assessed a comparison of bovine LF, derived from milk and used throughout this article, with human LF obtained from neutrophils. As shown, both variants of LF inhibited adhesion to fibronectin and VCAM-1 in the presence of GM-CSF. Differences in efficacy with respect to VCAM-1 could be due to differences in charge between bovine (+18) and human (+11) LF (Harmsen and others 1995), particularly if LF binds eosinophils via charge interactions. Alternatively, the structure of LF and downstream cell signals of the molecule have been reported to differ depending upon the iron bound to LF (Jiang and others 2011; Baker and Baker 2012; Jiang and Lonnerdal 2012). Bovine LF used in this study was 10% to 20% iron-saturated, whereas, the amount of iron bound to the human LF was not available.
After 24 h culture, we found that GM-CSF induced CD11b but not CD49d expression, whereas, LF treatment alone had no effect on the immunodetection of these integrins (Fig. 3B) or cell viability (data not shown). We also noted that cotreatment with GM-CSF and LF enhanced CD11b but downregulated CD49d expression (Figs. 3D and 4) and did not affect cell viability (data not shown). One potential mechanism for these data comes from the observation that LF may function, at least in part, as a superoxide dismutase and thus converts superoxide (O2−) to hydrogen peroxide (H2O2; Comhair and Erzurum 2010). Because CD49d and CD11b are respectively induced by O2−and H2O2 (Nagata and others 2000; Chuang and others 2003), LF-mediated functions as an antioxidant may reduce CD49d but enhance CD11b expression.
Because, as mentioned above, the regulation of CD49d and CD11b has been shown to be redox sensitive (Nagata and others 2000; Puig-Kroger and others 2000; Chuang and others 2003), we compared LF-induced responses in the presence of GM-CSF to similar treatments with NAC. Results demonstrated that NAC did induce CD49d downregulation but not CD11b upregulation (Fig. 5B). Interestingly, the NAC-modulated cell scatter was distinctly different from LF in that the NAC population was uniquely more uniform than that of LF (Fig. 5A), suggesting that the effects of LF may be directed to a distinct population subset. Including both LF and NAC in GM-CSF cultures enhanced CD49d downregulation compared with LF or NAC treatments alone (Fig. 5B). Interestingly, inclusion of LF in GM-CSF plus NAC treatments enhanced CD11b expression compared with NAC plus GM-CSF alone (Fig. 5B). These data support the idea that LF can operate synergistically with NAC via CD49d but not via CD11b expression. Thus, LF may have additional functions other than serving as an antioxidant.
To further investigate the LF-induced CD49d and CD11b modulation, we examined the production/release of chemokines (MIP-1α and MCP-1) and calgranulin B (S100A9), which are respectively known to regulate CD49d and CD11b expression (Carr and others 1996; Weber and others 1996; Newton and Hogg 1998; Manitz and others 2003). In Fig. 6, GM-CSF treatments with LF alone or in combination with NAC upregulated chemokine production compared with controls, whereas, NAC did not exhibit a response. The increased production of chemokines in GM-CSF plus LF-treated samples was also revealed to be correlative with reduced CD49d and increased CD11b immunodetection (Table 1). These associations were not revealed in GM-CSF plus NAC treatments, suggesting that LF and NAC do not operate along similar cell signaling pathways. We also assessed eosinophil intracellular and extracellular production of S100A9. The levels of extracellular S100A9 increased in response to GM-CSF treatment, as we have previously described (Curran and Bertics 2011), and the addition of LF enhanced the effect. Combined, these data suggest that in the presence of GM-CSF, LF induces the production of chemokines (MIP-1α, MCP-1, and S100A9) that may then alter eosinophil integrin expression and function. Because eosinophil priming with GM-CSF enhances cell signaling (Bates and others 2010) and the sensitivity of integrins to bind ligands (Lampinen and others 2004), the altered responses (chemokine production and integrin expression) to LF cotreatment may be an effect of the increased binding affinity of the LF receptor for its ligand and/or increased cell signals associated with GM-CSF priming.
Previous research has indicated that cationic-charged surfaces coupled with GM-CSF treatment induce increased eosinophil mediator release associated with the increased density of CD18 expression (Adamko and others 2008). The N1-terminal domain of bovine and human LF has a net positive charge and contains a heparin-binding peptide fragment termed lactoferricin that is known to survive digestion (van Berkel and others 1997; Kuwata and others 1998; Shimazaki and others 1998; Vogel and others 2002). Studies involving nasal polyp fibroblasts and the Caco-2 colon cancer cell line indicate that LF-binding interactions with these cells is inhibited by heparin (Ashida and others 2004; Nadolska and others 2010). Heparin and anti-thrombin III have been shown to modulate eosinophil chemokine production and migration via HSPGs (Culley and others 2003; Feistritzer and others 2004), indicating that the LF-induced eosinophil chemokine production (Fig. 6), S100A9 release (Fig. 7), CD49d downregulation, and CD11b upregulation (Figs. 3–5) may partially arise by virtue of the heparin-binding activity that has been ascribed to LF (Wu and others 1995).
In the pathobiology of atopic disease, eosinophils are known to accumulate in the skin, lung, and gastrointestinal tract (Simon and others 2010). Eosinophils are also identified in various tumors and in some clinical cases of multiple sclerosis where patients were treated with an anti-CD49d antibody called natalizumab (Samoszuk 1997; Abbas and others 2011). LF is an identified immunoregulator in diseases such as asthma (Kruzel and others 2006), cancer (Ward and others 2005), and multiple sclerosis (Zimecki and others 2007) where the integrin α4β1 has been found to be pivotal to the progression of these diseases (Singh and others 2004; Garmy-Susini and others 2010; Skarica and others 2011). The conditions by which LF may regulate the function of integrins on eosinophils or additional cell types may be integral in understanding disease development and immunotherapeutic treatment.
Acknowledgments
This work was supported by NIH grants HL088594, HL56396, AI070503, and HL069116 (to P.J.B.) as well as by the institutional grant 1UL1RR025011 from the Clinical and Translational Science Award (CTSA) program of the National Center for Research Resources, National Institutes of Health. The authors also wish to thank Greg J. Wiepz, Ph.D. for technical advice and article review, Michael D. Evans, M.S. for statistical analysis of correlation data, Beth Schwantes, B.S. for overseeing the eosinophil purification process, Paul Fichtinger for performing the eosinophil purification, and Mats W. Johansson, Ph.D. and Deane F. Mosher, M.D. for article review.
Author Disclosure Statement
The authors disclose that they do not have any commercial associations that might cause a conflict of interest in connection with the submitted article.
References
- Abbas M. Lalive PH. Chofflon M. Simon HU. Chizzolini C. Ribi C. Hypereosinophilia in patients with multiple sclerosis treated with natalizumab. Neurology. 2011;77(16):1561–1564. doi: 10.1212/WNL.0b013e318233b391. [DOI] [PubMed] [Google Scholar]
- Adamko DJ. Wu Y. Ajamian F. Ilarraza R. Moqbel R. Gleich GJ. The effect of cationic charge on release of eosinophil mediators. J Allergy Clin Immunol. 2008;122(2):383–390. doi: 10.1016/j.jaci.2008.03.020. 390 e1–e4. [DOI] [PubMed] [Google Scholar]
- Adamko DJ. Wu Y. Gleich GJ. Lacy P. Moqbel R. The induction of eosinophil peroxidase release: improved methods of measurement and stimulation. J Immunol Methods. 2004;291(1–2):101–108. doi: 10.1016/j.jim.2004.05.003. [DOI] [PubMed] [Google Scholar]
- Anwar AR. Moqbel R. Walsh GM. Kay AB. Wardlaw AJ. Adhesion to fibronectin prolongs eosinophil survival. J Exp Med. 1993;177(3):839–843. doi: 10.1084/jem.177.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anwar AR. Walsh GM. Cromwell O. Kay AB. Wardlaw AJ. Adhesion to fibronectin primes eosinophils via alpha 4 beta 1 (VLA-4) Immunology. 1994;82(2):222–228. [PMC free article] [PubMed] [Google Scholar]
- Ashida K. Sasaki H. Suzuki YA. Lonnerdal B. Cellular internalization of lactoferrin in intestinal epithelial cells. Biometals. 2004;17(3):311–315. doi: 10.1023/b:biom.0000027710.13543.3f. [DOI] [PubMed] [Google Scholar]
- Baker HM. Baker EN. A structural perspective on lactoferrin function. Biochem Cell Biol. 2012;90(3):320–328. doi: 10.1139/o11-071. [DOI] [PubMed] [Google Scholar]
- Barthel SR. Annis DS. Mosher DF. Johansson MW. Differential engagement of modules 1 and 4 of vascular cell adhesion molecule-1 (CD106) by integrins alpha4beta1 (CD49d/29) and alphaMbeta2 (CD11b/18) of eosinophils. J Biol Chem. 2006;281(43):32175–32187. doi: 10.1074/jbc.M600943200. [DOI] [PubMed] [Google Scholar]
- Barthel SR. Johansson MW. McNamee DM. Mosher DF. Roles of integrin activation in eosinophil function and the eosinophilic inflammation of asthma. J Leukoc Biol. 2008;83(1):1–12. doi: 10.1189/jlb.0607344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bates ME. Sedgwick JB. Zhu Y. Liu LY. Heuser RG. Jarjour NN. Kita H. Bertics PJ. Human airway eosinophils respond to chemoattractants with greater eosinophil-derived neurotoxin release, adherence to fibronectin, and activation of the Ras-ERK pathway when compared with blood eosinophils. J Immunol. 2010;184(12):7125–7133. doi: 10.4049/jimmunol.0900634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bournazou I. Mackenzie KJ. Duffin R. Rossi AG. Gregory CD. Inhibition of eosinophil migration by lactoferrin. Immunol Cell Biol. 2010;88(2):220–223. doi: 10.1038/icb.2009.86. [DOI] [PubMed] [Google Scholar]
- Brock JH. The physiology of lactoferrin. Biochem Cell Biol. 2002;80(1):1–6. doi: 10.1139/o01-212. [DOI] [PubMed] [Google Scholar]
- Carr MW. Alon R. Springer TA. The C-C chemokine MCP-1 differentially modulates the avidity of beta 1 and beta 2 integrins on T lymphocytes. Immunity. 1996;4(2):179–187. doi: 10.1016/s1074-7613(00)80682-2. [DOI] [PubMed] [Google Scholar]
- Chodaczek G. Saavedra-Molina A. Bacsi A. Kruzel ML. Sur S. Boldogh I. Iron-mediated dismutation of superoxide anion augments antigen-induced allergic inflammation: effect of lactoferrin. Postepy Hig Med Dosw (Online) 2007;61:268–276. [PubMed] [Google Scholar]
- Chuang KP. Tsai WS. Wang YJ. Shieh CC. Superoxide activates very late antigen-4 on an eosinophil cell line and increases cellular binding to vascular cell adhesion molecule-1. Eur J Immunol. 2003;33(3):645–655. doi: 10.1002/eji.200323446. [DOI] [PubMed] [Google Scholar]
- Comhair SA. Erzurum SC. Redox control of asthma: molecular mechanisms and therapeutic opportunities. Antioxid Redox Signal. 2010;12(1):93–124. doi: 10.1089/ars.2008.2425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Culley FJ. Fadlon EJ. Kirchem A. Williams TJ. Jose PJ. Pease JE. Proteoglycans are potent modulators of the biological responses of eosinophils to chemokines. Eur J Immunol. 2003;33(5):1302–1310. doi: 10.1002/eji.200323509. [DOI] [PubMed] [Google Scholar]
- Curran CS. Bertics PJ. Human eosinophils express RAGE, produce RAGE ligands, exhibit PKC-delta phosphorylation and enhanced viability in response to the RAGE ligand, S100B. Int Immunol. 2011;23(12):713–728. doi: 10.1093/intimm/dxr083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran CS. Evans MD. Bertics PJ. GM-CSF production by glioblastoma cells has a functional role in eosinophil survival, activation, and growth factor production for enhanced tumor cell proliferation. J Immunol. 2011;187(3):1254–1263. doi: 10.4049/jimmunol.1001965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feistritzer C. Kaneider NC. Sturn DH. Wiedermann CJ. Syndecan-4-dependent migration of human eosinophils. Clin Exp Allergy. 2004;34(5):696–703. doi: 10.1111/j.1365-2222.2004.1853.x. [DOI] [PubMed] [Google Scholar]
- Garmy-Susini B. Avraamides CJ. Schmid MC. Foubert P. Ellies LG. Barnes L. Feral C. Papayannopoulou T. Lowy A. Blair SL, et al. Integrin alpha4beta1 signaling is required for lymphangiogenesis and tumor metastasis. Cancer Res. 2010;70(8):3042–3051. doi: 10.1158/0008-5472.CAN-09-3761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harmsen MC. Swart PJ. de Bethune MP. Pauwels R. De Clercq E. The TH. Meijer DK. Antiviral effects of plasma and milk proteins: lactoferrin shows potent activity against both human immunodeficiency virus and human cytomegalovirus replication in vitro. J Infect Dis. 1995;172(2):380–388. doi: 10.1093/infdis/172.2.380. [DOI] [PubMed] [Google Scholar]
- Jeziorska M. Haboubi N. Schofield P. Woolley DE. Distribution and activation of eosinophils in inflammatory bowel disease using an improved immunohistochemical technique. J Pathol. 2001;194(4):484–492. doi: 10.1002/path.904. [DOI] [PubMed] [Google Scholar]
- Jiang R. Lonnerdal B. Apo- and holo-lactoferrin stimulate proliferation of mouse crypt cells but through different cellular signaling pathways. Int J Biochem Cell Biol. 2012;44(1):91–100. doi: 10.1016/j.biocel.2011.10.002. [DOI] [PubMed] [Google Scholar]
- Jiang R. Lopez V. Kelleher SL. Lonnerdal B. Apo- and holo-lactoferrin are both internalized by lactoferrin receptor via clathrin-mediated endocytosis but differentially affect ERK-signaling and cell proliferation in Caco-2 cells. J Cell Physiol. 2011;226(11):3022–3031. doi: 10.1002/jcp.22650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson S. Svineng G. Wennerberg K. Armulik A. Lohikangas L. Fibronectin-integrin interactions. Front Biosci. 1997;2:d126–d146. doi: 10.2741/a178. [DOI] [PubMed] [Google Scholar]
- Kayaba H. Yamada Y. Cui CH. Saito N. Honda K. Kobayashi Y. Urayama O. Chihara J. Expression of VLA-4 on eosinophils decreases in patients with eosinophilia. Int Arch Allergy Immunol. 2001;125(Suppl 1):33–37. doi: 10.1159/000053850. [DOI] [PubMed] [Google Scholar]
- Kayazawa M. Saitoh O. Kojima K. Nakagawa K. Tanaka S. Tabata K. Matsuse R. Uchida K. Hoshimoto M. Hirata I, et al. Lactoferrin in whole gut lavage fluid as a marker for disease activity in inflammatory bowel disease: comparison with other neutrophil-derived proteins. Am J Gastroenterol. 2002;97(2):360–369. doi: 10.1111/j.1572-0241.2002.05470.x. [DOI] [PubMed] [Google Scholar]
- Kita H. Eosinophils: multifaceted biological properties and roles in health and disease. Immunol Rev. 2011;242(1):161–177. doi: 10.1111/j.1600-065X.2011.01026.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klebanoff SJ. Waltersdorph AM. Prooxidant activity of transferrin and lactoferrin. J Exp Med. 1990;172(5):1293–1303. doi: 10.1084/jem.172.5.1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruzel ML. Bacsi A. Choudhury B. Sur S. Boldogh I. Lactoferrin decreases pollen antigen-induced allergic airway inflammation in a murine model of asthma. Immunology. 2006;119(2):159–166. doi: 10.1111/j.1365-2567.2006.02417.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuwata H. Yip TT. Yip CL. Tomita M. Hutchens TW. Direct detection and quantitative determination of bovine lactoferricin and lactoferrin fragments in human gastric contents by affinity mass spectrometry. Adv Exp Med Biol. 1998;443:23–32. doi: 10.1007/978-1-4757-9068-9_3. [DOI] [PubMed] [Google Scholar]
- Lampinen M. Carlson M. Hakansson LD. Venge P. Cytokine-regulated accumulation of eosinophils in inflammatory disease. Allergy. 2004;59(8):793–805. doi: 10.1111/j.1398-9995.2004.00469.x. [DOI] [PubMed] [Google Scholar]
- Legrand D. Mazurier J. A critical review of the roles of host lactoferrin in immunity. Biometals. 2010;23(3):365–376. doi: 10.1007/s10534-010-9297-1. [DOI] [PubMed] [Google Scholar]
- Li D. Sakashita S. Morishita Y. Kano J. Shiba A. Sato T. Noguchi M. Binding of lactoferrin to IGBP1 triggers apoptosis in a lung adenocarcinoma cell line. Anticancer Res. 2011;31(2):529–534. [PubMed] [Google Scholar]
- Manitz MP. Horst B. Seeliger S. Strey A. Skryabin BV. Gunzer M. Frings W. Schonlau F. Roth J. Sorg C, et al. Loss of S100A9 (MRP14) results in reduced interleukin-8-induced CD11b surface expression, a polarized microfilament system, and diminished responsiveness to chemoattractants in vitro. Mol Cell Biol. 2003;23(3):1034–1043. doi: 10.1128/MCB.23.3.1034-1043.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto K. Sterbinsky SA. Bickel CA. Zhou DF. Kovach NL. Bochner BS. Regulation of alpha 4 integrin-mediated adhesion of human eosinophils to fibronectin and vascular cell adhesion molecule-1. J Allergy Clin Immunol. 1997;99(5):648–656. doi: 10.1016/s0091-6749(97)70027-7. [DOI] [PubMed] [Google Scholar]
- Monteseirin J. Neutrophils and asthma. J Investig Allergol Clin Immunol. 2009;19(5):340–354. [PubMed] [Google Scholar]
- Nadolska B. Fraczek M. Krecicki T. Kocieba M. Zimecki M. Lactoferrin inhibits the growth of nasal polyp fibroblasts. Pharmacol Rep. 2010;62(6):1139–1147. doi: 10.1016/s1734-1140(10)70376-5. [DOI] [PubMed] [Google Scholar]
- Nagata M. Yamamoto H. Shibasaki M. Sakamoto Y. Matsuo H. Hydrogen peroxide augments eosinophil adhesion via beta2 integrin. Immunology. 2000;101(3):412–418. doi: 10.1046/j.1365-2567.2000.00123.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naot D. Chhana A. Matthews BG. Callon KE. Tong PC. Lin JM. Costa JL. Watson M. Grey AB. Cornish J. Molecular mechanisms involved in the mitogenic effect of lactoferrin in osteoblasts. Bone. 2011;49(2):217–224. doi: 10.1016/j.bone.2011.04.002. [DOI] [PubMed] [Google Scholar]
- Newton RA. Hogg N. The human S100 protein MRP-14 is a novel activator of the beta 2 integrin Mac-1 on neutrophils. J Immunol. 1998;160(3):1427–1435. [PubMed] [Google Scholar]
- Puig-Kroger A. Sanz-Rodriguez F. Longo N. Sanchez-Mateos P. Botella L. Teixido J. Bernabeu C. Corbi AL. Maturation-dependent expression and function of the CD49d integrin on monocyte-derived human dendritic cells. J Immunol. 2000;165(8):4338–4345. doi: 10.4049/jimmunol.165.8.4338. [DOI] [PubMed] [Google Scholar]
- Ross GD. Role of the lectin domain of Mac-1/CR3 (CD11b/CD18) in regulating intercellular adhesion. Immunol Res. 2002;25(3):219–227. doi: 10.1385/IR:25:3:219. [DOI] [PubMed] [Google Scholar]
- Sakamoto K. Ito Y. Mori T. Sugimura K. Interaction of human lactoferrin with cell adhesion molecules through RGD motif elucidated by lactoferrin-binding epitopes. J Biol Chem. 2006;281(34):24472–24478. doi: 10.1074/jbc.M604974200. [DOI] [PubMed] [Google Scholar]
- Samoszuk M. Eosinophils and human cancer. Histol Histopathol. 1997;12(3):807–812. [PubMed] [Google Scholar]
- Sampson AP. The role of eosinophils and neutrophils in inflammation. Clin Exp Allergy 30 Suppl. 2000;1:22–27. doi: 10.1046/j.1365-2222.2000.00092.x. [DOI] [PubMed] [Google Scholar]
- Sandomirsky BP. Galchenko SE. Galchenko KS. Antioxidative properties of lactoferrin from bovine colostrum before and after its lyophilization. Cryo Lett. 2003;24(5):275–280. [PubMed] [Google Scholar]
- Shimazaki K. Tazume T. Uji K. Tanaka M. Kumura H. Mikawa K. Shimo-Oka T. Properties of a heparin-binding peptide derived from bovine lactoferrin. J Dairy Sci. 1998;81(11):2841–2849. doi: 10.3168/jds.S0022-0302(98)75843-6. [DOI] [PubMed] [Google Scholar]
- Simon D. Wardlaw A. Rothenberg ME. Organ-specific eosinophilic disorders of the skin, lung, and gastrointestinal tract. J Allergy Clin Immunol. 2010;126(1):3–13. doi: 10.1016/j.jaci.2010.01.055. quiz 14–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh J. Adams S. Carter MB. Cuervo H. Lee WC. Lobb RR. Pepinsky RB. Petter R. Scott D. Rational design of potent and selective VLA-4 inhibitors and their utility in the treatment of asthma. Curr Top Med Chem. 2004;4(14):1497–1507. doi: 10.2174/1568026043387520. [DOI] [PubMed] [Google Scholar]
- Skarica M. Eckstein C. Whartenby KA. Calabresi PA. Novel mechanisms of immune modulation of natalizumab in multiple sclerosis patients. J Neuroimmunol. 2011;235(1–2):70–76. doi: 10.1016/j.jneuroim.2011.02.010. [DOI] [PubMed] [Google Scholar]
- Spik G. Legrand D. Leveugle B. Mazurier J. Mikogami T. Montreuil J. Pierce A. Rochard E. Characterization of two kinds of lactotransferrin (lactoferrin) receptors on different target cells. Adv Exp Med Biol. 1994;357:13–19. doi: 10.1007/978-1-4615-2548-6_2. [DOI] [PubMed] [Google Scholar]
- Strath M. Warren DJ. Sanderson CJ. Detection of eosinophils using an eosinophil peroxidase assay. Its use as an assay for eosinophil differentiation factors. J Immunol Methods. 1985;83(2):209–215. doi: 10.1016/0022-1759(85)90242-x. [DOI] [PubMed] [Google Scholar]
- Suzuki YA. Lopez V. Lonnerdal B. Mammalian lactoferrin receptors: structure and function. Cellular and molecular life sciences: CMLS. 2005;62(22):2560–2575. doi: 10.1007/s00018-005-5371-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tachimoto H. Ebisawa M. Bochner BS. Cross-talk between integrins and chemokines that influences eosinophil adhesion and migration. Int Arch Allergy Immunol. 2002;128(Suppl 1):18–20. doi: 10.1159/000059414. [DOI] [PubMed] [Google Scholar]
- Travis SM. Conway BA. Zabner J. Smith JJ. Anderson NN. Singh PK. Greenberg EP. Welsh MJ. Activity of abundant antimicrobials of the human airway. Am J Respir Cell Mol Biol. 1999;20(5):872–879. doi: 10.1165/ajrcmb.20.5.3572. [DOI] [PubMed] [Google Scholar]
- van Berkel PH. Geerts ME. van Veen HA. Mericskay M. de Boer HA. Nuijens JH. N-terminal stretch Arg2, Arg3, Arg4 and Arg5 of human lactoferrin is essential for binding to heparin, bacterial lipopolysaccharide, human lysozyme and DNA. Biochem J. 1997;328(Pt 1):145–151. doi: 10.1042/bj3280145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogel HJ. Schibli DJ. Jing W. Lohmeier-Vogel EM. Epand RF. Epand RM. Towards a structure-function analysis of bovine lactoferricin and related tryptophan- and arginine-containing peptides. Biochem Cell Biol. 2002;80(1):49–63. doi: 10.1139/o01-213. [DOI] [PubMed] [Google Scholar]
- Walsh GM. Symon FA. Lazarovils AL. Wardlaw AJ. Integrin alpha 4 beta 7 mediates human eosinophil interaction with MAdCAM-1, VCAM-1 and fibronectin. Immunology. 1996;89(1):112–119. doi: 10.1046/j.1365-2567.1996.d01-713.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward PP. Paz E. Conneely OM. Multifunctional roles of lactoferrin: a critical overview. Cell Mol Life Sci. 2005;62(22):2540–2548. doi: 10.1007/s00018-005-5369-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber C. Alon R. Moser B. Springer TA. Sequential regulation of alpha 4 beta 1 and alpha 5 beta 1 integrin avidity by CC chemokines in monocytes: implications for transendothelial chemotaxis. J Cell Biol. 1996;134(4):1063–1073. doi: 10.1083/jcb.134.4.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong CK. Ip WK. Lam CW. Interleukin-3, -5, and granulocyte macrophage colony-stimulating factor-induced adhesion molecule expression on eosinophils by p38 mitogen-activated protein kinase and nuclear factor-[kappa] B. Am J Respir Cell Mol Biol. 2003;29(1):133–147. doi: 10.1165/rcmb.2002-0289OC. [DOI] [PubMed] [Google Scholar]
- Woodruff SA. Masterson JC. Fillon S. Robinson ZD. Furuta GT. Role of eosinophils in inflammatory bowel and gastrointestinal diseases. J Pediatr Gastroenterol Nutr. 2011;52(6):650–661. doi: 10.1097/MPG.0b013e3182128512. [DOI] [PubMed] [Google Scholar]
- Wu HF. Monroe DM. Church FC. Characterization of the glycosaminoglycan-binding region of lactoferrin. Arch Biochem Biophys. 1995;317(1):85–92. doi: 10.1006/abbi.1995.1139. [DOI] [PubMed] [Google Scholar]
- Zhu X. Subbaraman R. Sano H. Jacobs B. Sano A. Boetticher E. Munoz NM. Leff AR. A surrogate method for assessment of beta(2)-integrin-dependent adhesion of human eosinophils to ICAM-1. J Immunol Methods. 2000;240(1–2):157–164. doi: 10.1016/s0022-1759(00)00192-7. [DOI] [PubMed] [Google Scholar]
- Zimecki M. Kocieba M. Chodaczek G. Houszka M. Kruzel ML. Lactoferrin ameliorates symptoms of experimental encephalomyelitis in Lewis rats. J Neuroimmunol. 2007;182(1–2):160–166. doi: 10.1016/j.jneuroim.2006.10.008. [DOI] [PubMed] [Google Scholar]