Abstract
Ferromagnetism was observed at room temperature in monodisperse CeO2 nanospheres synthesized by hydrothermal treatment of Ce(NO3)3·6H2O using polyvinylpyrrolidone as a surfactant. The structure and morphology of the products were characterized by X-ray diffraction (XRD), Raman spectroscopy, transmission electron microscopy, high-resolution transmission electron microscopy, and field-emission scanning electron microscopy (FE-SEM). The optical properties of the nanospheres were determined using UV and visible spectroscopy and photoluminescence (PL). The valence states of Ce ions were also determined using X-ray absorption near edge spectroscopy. The XRD results indicated that the synthesized samples had a cubic structure with a crystallite size in the range of approximately 9 to 19 nm. FE-SEM micrographs showed that the samples had a spherical morphology with a particle size in the range of approximately 100 to 250 nm. The samples also showed a strong UV absorption and room temperature PL. The emission might be due to charge transfer transitions from the 4f band to the valence band of the oxide. The magnetic properties of the samples were studied using a vibrating sample magnetometer. The samples exhibited room temperature ferromagnetism with a small magnetization of approximately 0.0026 to 0.016 emu/g at 10 kOe. Our results indicate that oxygen vacancies could be involved in the ferromagnetic exchange, and the possible mechanism of formation was discussed based on the experimental results.
Keywords: CeO2, Nanospheres, Dilute magnetic oxide, Ferromagnetism, Oxygen vacancies, Valence states
Background
Oxide-dilute magnetic semiconductors (O-DMSs) such as ZnO, TiO2, SnO2, and In2O3 doped with transition metal (TM) ions have recently attracted much attention due to their potential use in magneto-optoelectronic applications [1-3]. These O-DMSs are optically transparent and exhibit ferromagnetism (FM) at room temperature (RT) and even well above RT. Recently, TM-doped CeO2 have been also reported to exhibit ferromagnetism at and above room temperature [4-10]. Unlike other O-DMSs, CeO2 has a cubic structure with a lattice parameter a = 0.54113 nm [11] that will facilitate the integration of spintronic devices with advanced silicon microelectronic devices.
Early work on CeO2-based O-DMSs was focused on thin films [4-6] and only a few works have been carried out on powders, bulk, or nanocrystalline form [9-12]. Tiwari et al. [4] firstly discovered room temperature ferromagnetism (RT-FM) in Ce1−xCoxO2−δ (x ≤ 0.05) films deposited on a LaAlO3 (001) substrate by pulsed laser deposition (PLD) technique. These films are transparent in a visible regime and exhibit a very high Curie temperature (TC) at approximately 740 to 875 K with large magnetic moments of 6.1 ± 0.2 to 8.2 ± 0.2 μB/Co. Following the work by Tiwari et al., Song et al. [5] reported successful fabrication ofCe1−xCoxO2−δ (x = 0.03) thin films with (111) preferential orientation deposited on a Si (111) substrate by a PLD technique. Their deposited films show RT-FM with large magnetic moment of 5.8 μB/Co and coercivity of 560 Oe. The authors also showed that the films could be deposited on glass but with smaller magnetic moment and coercivity. These results suggested that the FM in Co-doped CeO2 depend not only on the doping concentration of transition element, but also on the microstructure of film, including its crystallization, defects, vacancies, etc. Vodungbo et al. [6] also reported FM in Co-doped CeO2 thin films grown by PLD on SrTiO3 and Si substrate. The films were ferromagnetic with a TC above 400 K. These authors found that the amount of structural defects had a little effect on FM, but the presence of oxygen during the growth or annealing reduced drastically the FM, suggesting that oxygen vacancies played an important role in the magnetic coupling between Co ions, while Wen et al. [9] reported the ferromagnetism observed in pure and Co-doped CeO2 powders. The RT-FM in pure CeO2 originated from oxygen vacancies while a slight Co doping in CeO2 caused a nearly two-order enhancement of saturation magnetization (Ms) to 0.47 emu/g as compared with the pure sample. The authors suggested that the large RT-FM observed in Co-doped CeO2 powder originated from a combination effect of oxygen vacancies and Co doping. Similarly, Ou et al. [10] reported RT-FM for Ce1−xCoxO2 (0 < x < 0.10) nanorods prepared by electrodeposition route. The nanorods were ferromagnetic with a high TC of about 870 K and the largest Ms of 0.015 emu/g. They suggested that the RT-FM observed in Co-doped CeO2 nanorods was adjusted by the structural defects including oxygen vacancies. The same behavior was found in nanoparticles of Fe-doped CeO2[11] with an Ms value of 0.0062 emu/g in 3 at % Fe prepared by a sol–gel method and Fe-doped CeO2[12] with an Ms value of 0.10 emu/g in 1 at % Fe prepared by the proteic sol–gel process. The authors suggested that the RT-FM originated from an exchange of F-center, which involved a combination of oxygen vacancies and TM doping.
Surprisingly, the researchers report RT-FM of undoping in different oxides, such as thin films of HfO2[13], TiO2 and In2O3[14], and nanoparticles of CeO2, Al2O3, ZnO, In2O3, and SnO2[15], while the corresponding bulk samples are diamagnetic. Most recently, there are some studies reporting ferromagnetism observed in pure CeO2 on powders, nanocrystalline, or cubes [16-18]. Liu et al. [16] studied the size-dependent ferromagnetism in CeO2 powders synthesized by precipitation route. They found that ferromagnetism was observed only in sub-20-nm powders with an Ms value of 0.08 emu/g. Similarly, Chen et al. [17] reported RT-FM in CeO2 nanoparticles prepared by thermal decomposition method with an Ms value of 0.12 emu/g. The authors showed that its crystallite size in nanometers would be ferromagnetic because of the large value of the surface-to-volume ratio, leading to the exchange interactions between electron spin moments that resulted from oxygen vacancies at the surface [4]. Recently, Ge et al. [18] observed ferromagnetism in CeO2 nanocubes with an Ms value of 0.0057 emu/g (an average size of 5.3 nm) prepared by a chemical method. They suggest that oxygen vacancy is essential for the formation of FM in CeO2 nanocubes.
However, magnetic properties of monodisperse nanospheres of pure CeO2 have not yet been reported. In this work, we report the ferromagnetism observed in monodisperse CeO2 nanospheres with a particle size of approximately 200 nm synthesized by hydrothermal treatment of Ce(NO3)3·6H2O using polyvinylpyrrolidone (PVP) as a surfactant. The technique of preparation and the effect of the type of cerium source on the crystallinity and morphology were investigated. The prepared samples were characterized by X-ray diffraction (XRD), Raman spectroscopy, field-emission scanning electron microscopy (FE-SEM), transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM), UV and visible spectroscopy (UV–vis), and photoluminescence (PL). The valence states of Ce ions were also investigated by using X-ray absorption near edge spectroscopy (XANES), and the magnetic properties of the samples were determined using a vibrating sample magnetometer (VSM). The origin of RT-FM in this pure CeO2 is also discussed.
Methods
In this study, cerium (III) nitrate hexahydrate, Ce(NO3)3·6H2O (99.99% purity; Kanto Corporation, Portland, OR, USA); cerium (III) acetate hydrate, Ce(CH3CO2)3·xH2O (99.9% purity; Sigma-Aldrich Corporation, St. Louis, MO, USA); cerium (III) chloride heptahydrate, CeCl3·7H2O (99.9% purity; Sigma-Aldrich Corporation); cerium (III) sulfate octahydrate, Ce2(SO4)3.8H2O (99.999% purity; Sigma-Aldrich Corporation); and PVP (Sigma-Aldrich Corporation) were used as starting materials. In a typical procedure, one gram of PVP was mixed with 40 mL of deionized water under vigorous magnetic stirring at room temperature (27°C) until a homogeneous solution was obtained. Subsequently, 3 mmol of cerium source was slowly added to the PVP solution under vigorous stirring at room temperature for 2 h, in order to obtain a well-dissolved solution. Throughout the whole process described, no pH adjustment was made. The homogeneous solution was transferred into a Teflon-lined stainless steel autoclave of 50-mL capacity and prepared at 160°C and 200°C for 12 h and 160°C and 200°C for 24 h. After the autoclave was cooled naturally to room temperature, the precipitate was collected and washed several times with distilled water. The final product was then dried in a vacuum at 80°C overnight. In addition, the as-prepared samples were also annealed in argon atmosphere at 400°C for 2 h to study the effect of oxygen vacancies on magnetic properties of the annealed samples.
The prepared samples were characterized using XRD, Raman spectroscopy, FE-SEM, TEM, HRTEM, UV–vis, PL, XANES, and VSM. A Philips X-ray diffractometer (Philips Tecnai, Amsterdam, The Netherlands) with CuKα radiation (λ = 0.15406 nm) was used to study the phases of the pure CeO2 samples. The Raman spectra were recorded at room temperature using a triple spectrometer (Jobin Yvon/Atago-Bussan T-64000, HORIBA Jobin Yvon S.A.S., Chilly-Mazarin, France). The morphology of the sample was obtained from TEM (JEM 2010 200 kV, JEOL Ltd., Akishima, Tokyo, Japan). FE-SEM was performed using a JEOL JSM-6335 F (JEOL Ltd.). The optical absorption spectrum was measured in the range of 200 to 800 nm using a UV-3101PC UV–vis-NIR scanning spectrometer (Shimadzu Corporation, Nakagyo-ku, Kyoto, Japan). PL was carried out on a luminescence spectrometer (PerkinElmer LS-55B, PerkinElmer Instrument, Waltham, MA, USA), using a Xenon lamp as the excitation source at room temperature. The Ce L3 XANES spectrum was studied using XANES in transmission mode at the BL4 Station at Siam Photon Laboratory (Synchrotron Light Research Institute (Public Organization), SLRI) in Nakhon Ratchasima, Thailand. The magnetic measurements were performed at room temperature using a vibrating sample magnetometer (VSM 7403, Lakeshore, Westerville, OH. USA).
Results and discussion
XRD analysis
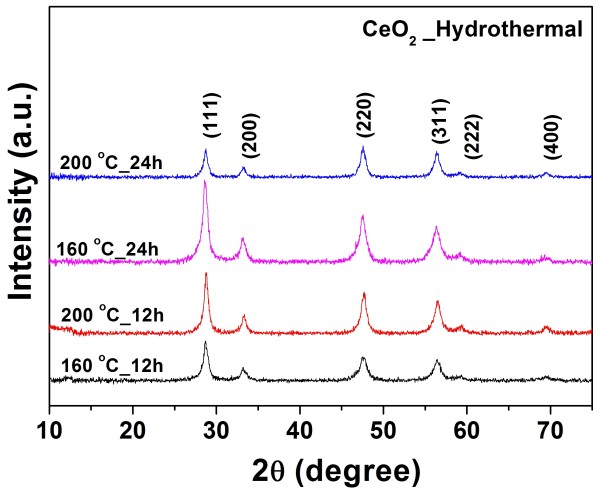
The XRD patterns of the samples prepared by hydrothermal reaction at 160°C for 12 h are shown in Figure 1a. The sample obtained with CeCl3·7H2O as the starting agent shows no XRD peaks, indicating that it is amorphous, whereas the sample from Ce(NO3)3·6H2O exhibits XRD peaks that correspond to the (111), (200), (220), (311), (222), and (400) planes, which are consistent with the face-centered cubic fluorite structure of CeO2 in the standard data from the Joint Committee on Powder Diffraction Standards (JCPDS) 34–0394, indicating that pure CeO2 was successfully synthesized via these procedures.
Figure 1.
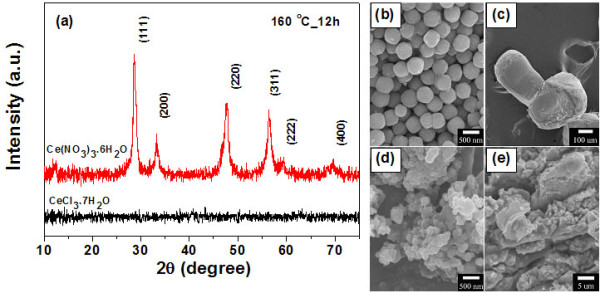
XRD pattern and FE-SEM images of the cerium source. (a) XRD patterns of cerium source as the starting materials prepared at 160°C for 12 h Ce(NO3)3·6H2O and CeCl3·7H2O. FE-SEM images of cerium source as the starting materials prepared at 160°C for 12 h (b) Ce(NO3)3·6H2O, (c) CeCl3·7H2O, (d) Ce(CH3CO2)3·xH2O, and (e) Ce2(SO4)3·8H2O.
In this study, we found that the type of cerium source has a great effect on the morphology of the final product. The cerium source from Ce(NO3)3·6H2O shows that the sample consisted of sphere-like particles with diameters of 100 to 250 nm (Figure 1b), whereas other cerium sources such as CeCl3·7H2O, Ce(CH3CO2)3·xH2O, and Ce2(SO4)3·8H2O resulted in irregular shapes and agglomerated particles as shown in Figure 1c,d,e, respectively. Therefore, it is clearly seen that cerium source from nitrate is most favorable for the formation of uniformly sized CeO2 nanospheres. It is possible that the absorption of PVP molecules on various crystallographic planes of cerium source played a major role in determining the product morphology, due to the fact that the supersaturation degree has a significant influence on the crystal nucleation rate and crystal growth rate [19]. However, the real reason for the morphology variation of the cerium source and surfactants has yet to be fully understood.
Figure 2 shows the XRD patterns of the pure CeO2 from cerium nitrate at various hydrothermal treatment durations and temperatures. All the samples exhibited six typical peaks corresponding to the (111), (200), (220), (311), (222), and (400) planes, which are consistent with the face-centered cubic fluorite structure of CeO2 in the standard data from JCPDS 34–0394, and this is in agreement with the selected area electron diffraction (SAED) patterns shown in Figure 3d. The values of the lattice constant calculated from the XRD spectra are shown in Table 1. The average crystallite size of all the samples was calculated from X-ray line broadening of the peaks at the (111), (200), (220), and (311) planes using Scherrer’s equation, (as listed in Table 1). We observed that the lattice parameter decreases with increasing crystallite size. This decrease is possibly due to the introduction of Ce3+ ions into the crystal lattice. Ce3+ ions have a higher ionic radius (1.034 Å) compared with the Ce4+ ions (0.92 Å) and introduce oxygen vacancies. Therefore, the concentration of Ce3+ ions increases, and there is also an increase in the number of oxygen vacancies. It is observed that the pure CeO2 nanoparticles experience considerable lattice distortion, which is in good agreement with earlier reports on CeO2 nanoparticles [9,20], which indicated that this causes a change in the Ce-O bond length (lattice distortion) and the overall lattice parameter.
Figure 2.
XRD patterns of obtained CeO2nanospheres. Ce(NO3)3·6H2O was used as starting materials prepared at 160°C and 200°C for 12 and 24 h.
Figure 3.
FE-SEM and TEM bright field images of CeO2nanospheres. (a) FE-SEM images of CeO2 nanospheres prepared at 200°C for 24 h; (b) TEM bright field images of CeO2 nanospheres prepared at 160°C for 12 h; (c) and (d) are TEM bright field images with corresponding SAED patterns (insets) of CeO2 nanospheres prepared at 200X°C for 12 h.
Table 1.
Summary of crystallite sizes, lattice constant, bandgap, and magnetization of pure CeO2nanospheres
| Sample | Crystallite size from XRD (nm) | Lattice constanta(nm) | Eg(eV) | Crystallite size from Raman spectroscopy (nm) |
Msat 10 kOe (emu/g) |
|
|---|---|---|---|---|---|---|
| Before Ar annealing | After Ar annealing | |||||
| CeO2 at 160 °C for 12 h |
9.43 ± 0.41 |
0.5430 ± 0.0021 |
3.00 |
7.39 |
- |
- |
| CeO2 at 200 °C for 12 h |
19.6 ± 0.53 |
0.5420 ± 0.0003 |
3.04 |
8.21 |
0.0026 |
0.011 |
| CeO2 at 160 °C for 24 h |
12.2 ± 0.13 |
0.5430 ± 0.0003 |
3.06 |
9.23 |
0.0053 |
0.0026 |
| CeO2 at 200 °C for 24 h | 15.6 ± 0.20 | 0.5428 ± 0.0006 | 3.10 | 12.28 | 0.016 | 0.015 |
Eg, bandgap energy; Ms, saturation magnetization. Data of magnetization of pure CeO2 nanospheres is before and after Ar annealing.
Raman analysis
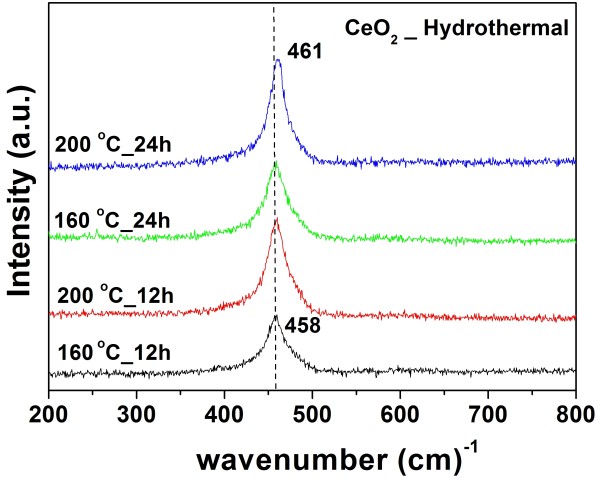
The formation of a cubic structure in the CeO2 nanospheres was further supported by the Raman spectra. Figure 4 shows typical spectra of CeO2. The Raman active modes are shifted from 458 to 461 cm−1 for the CeO2 samples heated at 160°C to 200°C. These Raman active modes are attributed to a symmetrical stretching mode of the Ce-80 vibrational unit, and therefore, they are very sensitive to any disorder in the oxygen sublattice that resulted from thermal, doping, or grain size [21-24]. The effect of the microstructure of CeO2 on the shape of the Raman spectra was observed by the broadening of the line and by increases in its asymmetry, which are attributed to the reduction of the phonon lifetime in the nanocrystalline regime [25, 26]. The particle size of the CeO2 sample can be also estimated from the Raman line broadening using the following Equation 1 [23, 24, 27]:
Figure 4.
Raman spectra of obtained CeO2nanospheres. Ce(NO3)3·6H2O was used as starting materials prepared at 160°C and 200°C for 12 and 24 h.
| (1) |
where is the full width at half maximum of the Raman active mode peak and DR is the particle size of a CeO2 sample. This relation can be used to determine the crystal size of the CeO2 samples as shown in Table 1. This calculated crystal size is near that obtained from X-ray line broadening.
FE-SEM, TEM, and HRTEM analyses
The morphology and structure of CeO2 nanospheres were investigated by FE-SEM and TEM as shown in Figure 3. TEM bright field images show that the samples contain monodisperse nanospheres with a narrow size distribution. The high-magnification TEM image of a single particle (Figure 3b) indicates that the sphere has a diameter of about 218 nm. This result is similar to the work reported by Zhou et al. [28], in which spherical CeO2 crystallites assembled by nanoparticles were synthesized by hydrothermal treatment because small nanoparticles of CeO2 aggregated and gradually evolved into a spherical assembly, achieving a low surface energy. The corresponding SAED patterns (inset in Figure 3d of the products show spotty ring patterns indicative of a face-centered cubic structure of CeO2 (JCPDS 34–0394), which is in agreement with the XRD results. The HRTEM images of the CeO2 sample prepared at 200°C for 12 h and the CeO2 sample prepared at 200°C for 12 h followed by annealing in Ar at 400°C for 2 h, are shown in Figure 5a,b, respectively. The d spacings of the lattice fringes of approximately 0.30 and 0.31 nm for the CeO2 sample prepared at 200°C for 12 h (Figure 5a) calculated from the HRTEM images correspond to the (111) plane of CeO2, whereas the d spacings of approximately 0.31 and 0.32 nm (Figure 5b) for the CeO2 sample prepared at 200°C for 12 h followed by annealing in Ar at 400°C for 2 h match with (111) plane of CeO2. This is in good agreement with the standard data (JCPDS 34–0394).
Figure 5.
HRTEM images of CeO2nanospheres prepared at 200°C for 24 h. (a) As-prepared CeO2 nanospheres and (b) CeO2 nanospheres after annealing in Ar at 400°C for 2 h.
Optical properties
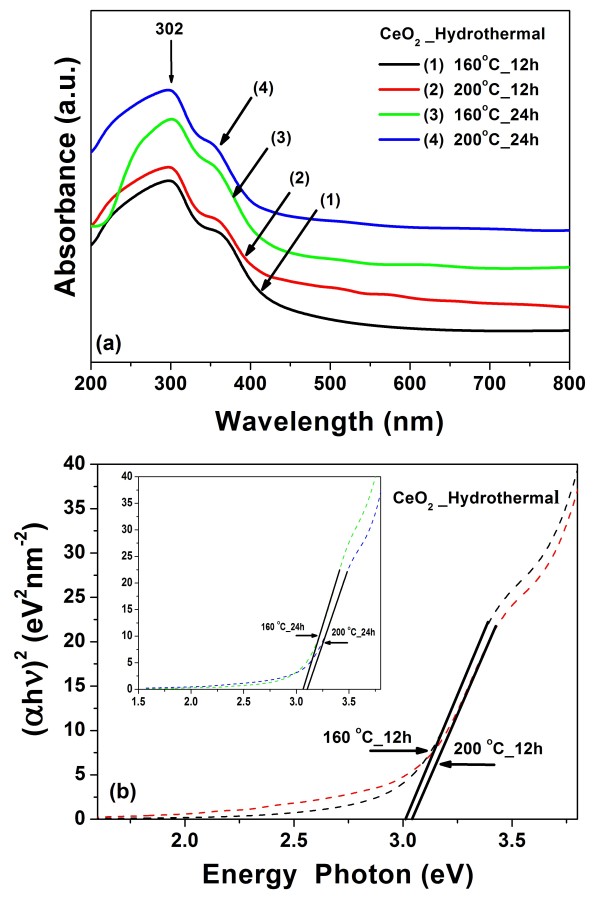
The UV–vis absorption spectra of the pure CeO2 nanospheres are shown in Figure 6a. All the samples show a strong absorption below 400 nm (3.10 eV) with a well-defined absorbance peak at approximately 302 nm (4.10 eV). The direct bandgap energy (Eg) is determined by fitting the absorption data to the direct transition as shown in Equation 2:
Figure 6.
Room temperature optical absorbance spectra of CeO2nanospheres and plot of (αhv)2. (a) Room temperature optical absorbance spectra of CeO2 nanospheres using Ce(NO3)3·6H2O as starting materials. (b) Plot of (αhv)2 as a function of photon energy for the CeO2 nanospheres.
| (2) |
where α is the optical absorption coefficient, hν is the photon energy, Eg is the direct bandgap, and A is a constant [29]. The extrapolation of the linear portions of the curves towards absorption equal to zero (y = 0) gives Eg for direct transitions (Figure 6b). The estimated direct bandgaps of all the samples are shown in Table 1. The bandgap of CeO2 reported in this work is lower than that reported in the literature. Chen and Chang [30] reported direct bandgap values ranging from 3.56 to 3.71 eV for CeO2 nanoparticles synthesized by precipitation method. Maensiri et al. [21] reported direct bandgap values ranging from 3.57 to 3.61 eV for CeO2 nanoparticles synthesized by the sol–gel method using egg white. Similarly, Masui et al. reported direct bandgap values of 4.1 and 2.6 nm for CeO2 nanoparticles prepared using reverse micelles to be 3.38 and 3.44 eV [31], respectively, due to quantum confinement effect [32]. This phenomenon has been well explained for particle sizes down to less than a few nanometers, but for our results, the bandgaps increased with increasing crystal size, which exhibit blueshifts in the UV absorption spectra inferred from the bandgap calculated for pure CeO2 nanospheres. This blueshift has been reported to be an electrostatic potential effect due to a cerium valence change when the particle size is larger than a few nanometers (e.g., ≥8 nm). The Ce4+ ions coexist with Ce3+ ions, and these ions can be attributed to oxygen vacancies at the surface [33]. Therefore, in our work, the bandgaps increased with increasing crystal size. The crystallite size is in the range of 9 to 19 nm as indicated by the existence of the blueshift for our CeO2 nanospheres.
PL analysis
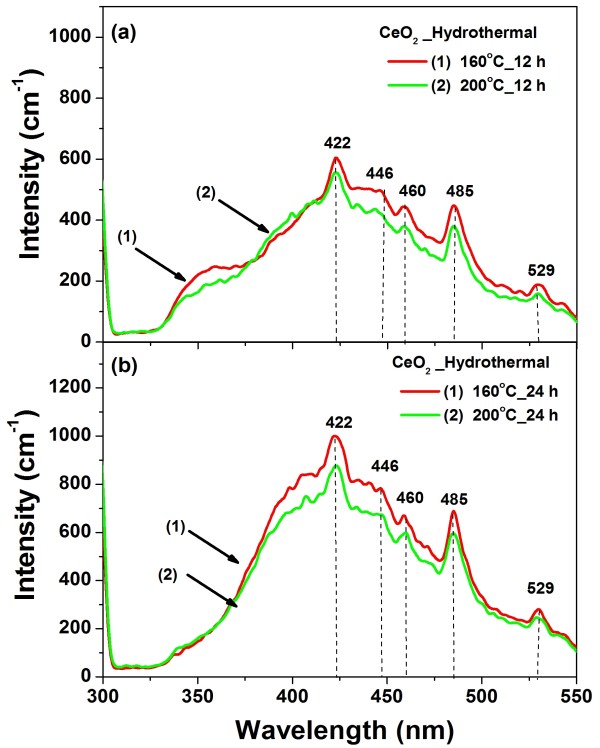
Figure 7 shows the room temperature PL spectra obtained using a Xenon laser of 290 nm as the excitation source of the CeO2 nanospheres. The spectra of all the samples are almost identical and mainly consist of five emission bands: a strong blue emission band at 422 nm (2.93 eV), a weak blue band at 446 nm (2.78 eV), a blue band at 460 nm (2.69 eV), a strong blue-green band at 485 nm (2.55 eV), and a green band at 529 nm (2.35 eV). Our results are consistent with that reported for CeO2 in the literature. The strong emission peak at 410 nm [34] and 422 nm [35] observed for CeO2 nanoparticles as-prepared at λex = 290 nm. Maensiri et al. [21] reported a blue band at approximately 443 nm, along with a green band at 529 nm for 400°C to 500°C calcined sample, and a strong UV emission band at 392 nm for 600°C calcined sample. The dependence of the PL blueshift peak on CeO2 particle concentration has also been observed by Sathyamurthy et al. [34] for CeO2 nanoparticles synthesized by a reverse micelle route. This phenomenon has been explained by charge transitions from the 4f band to the valence band of the CeO2 in both nanoparticles and thin films [36]. In addition, it is well known that the emission energy from the 4f band to the valence band energy gap of CeO2 is about 3.0 to 3.38 eV, as determined from the calculation of the electronic structure of CeO2[35,37]. Therefore, the emission in our CeO2 samples could be assumed to be the transition from the Ce 4f band to the O 2p band (valence band) in CeO2. The broad PL band ranging from 300 to 550 nm of all the samples could be the result of defects, including oxygen vacancies in the crystal with electronic energy levels below the 4f band [38]. This is confirmed by the enhanced absorption tail below 3 eV in nonstoichiometric CeO2 that was previously observed and attributed to the presence of oxygen vacancies [39].
Figure 7.
Room temperature photoluminescence spectrum of CeO2nanospheres using Ce(NO3)3·6H2O as starting materials. The sample was dispersed in methanol and the excitation wavelength used in PL measurement was 290 nm.
XANES analysis
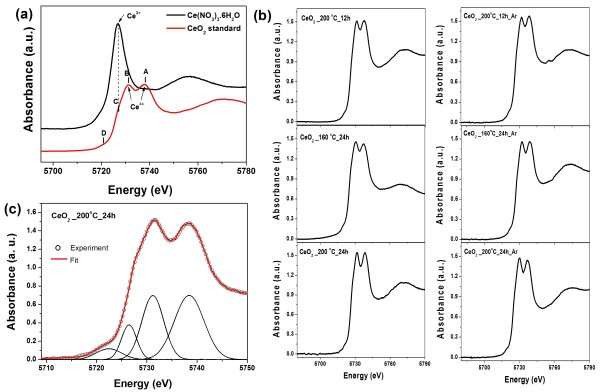
The valence state of Ce in pure CeO2 nanospheres was determined by XANES spectra measured at Ce L3 edge. Figure 8a shows the edge energies of the Ce(NO3)3·6H2O standard and CeO2 standard. The standard Ce(NO3)3·6H2O has a single peak illustrated by one intense white line at approximately 5,726.8 eV, which can be associated with the final state of 2p4f15degL, where L denotes an oxygen ligand 2p hole, corresponding to the Ce3+ valence state [40]. In the standard CeO2, there are four peaks comprising high energy peak A, main peak B, low energy peak C, and pre-edge peak D, which have been reported and assigned previously [41-43]. Peaks A and B are shifted to higher energies at approximately 5,737.9 and 5,731.3 eV, respectively, and were assigned as being due to a mixture of the multi-electron with the final state of 2p4f05d and 2p4f15dtgL, respectively, which characterizes the Ce in the Ce4+ valence state [40]. Peak C, at approximately 5,726.8 eV, is observed at the same energy as the white line of a typical Ce(NO3)3·6H2O standard, corresponding to the Ce3+ valence state. Peak D is assigned to the final states of 2p5d with a delocalized d character at the bottom of the conduction band due to the cubic crystal-field splitting of Ce 5d states [44].
Figure 8.
XANES spectra for Ce(NO3)3·6H2O and CeO2standard and pure CeO2and Gaussian fitting of XANES spectra. (a) XANES spectra at the Ce L3 absorption edge for Ce(NO3)3·6H2O and CeO2 standard showing features of A, B, C, and D. (b) XANES spectra of pure CeO2 at various hydrothermal treatment durations and temperature before and after annealing in Ar. (c) A Gaussian fitting of XANES spectra of pure CeO2 prepared at 20°C for 24 h.
Figure 8b shows the XANES spectra of pure CeO2 for various hydrothermal treatment durations and temperatures before and after annealing in Ar. The quantitative analysis of the valence state of Ce in each of the three states of the CeO2 nanospheres was performed using multi-peak Gaussian fitting obtained from the XANES spectra, as shown in Figure 8c. The analysis shows that Ce in the samples is in a mixed valence state of Ce3+ and Ce4+. From these results, we can obtain the valence state of Ce according to the fitting parameters of the peak positions and areas listed in Table 2. It is observed that the percentage of Ce3+ ranges from 7% to 13.7% in the pure CeO2 samples. These results provide confirmation of the formation of oxygen vacancies on the surface of the CeO2 samples. It is possible that the concentration and distribution of oxygen vacancies play an important role in the magnetism of our CeO2 nanospheres. The highest percentage of Ce3+ is 13.7% for CeO2 prepared at 200°C for 24 h, with the increase in the number of oxygen vacancies, leading to the highest Ms values. The concentration of Ce3+ in our pure CeO2 sample is higher than the values reported in the literature for CeO2. Zhang et al. [41] reported the average Ce3+ concentration of 10- and 6-nm CeO2 nanoparticles prepared by mixing cerium nitrate and hexamethylenetetramine in aqueous solution at room temperature to be 1% and 6.5%, respectively. Chen et al. [17] reported that the concentration of Ce3+ was higher than 21% for CeO2 nanoparticles synthesized by the thermal decomposition method, which is higher than the value for pure CeO2 nanospheres reported in this study.
Table 2.
Gaussian fitting for percentage of Ce3+of pure CeO2nanospheres before and after Ar annealing
| Sample |
Peak position (eV) |
Peak area (eV) |
Percentage of Ce3+(%) |
|||
|---|---|---|---|---|---|---|
| Before Ar annealing | After Ar annealing | Before Ar annealing | After Ar annealing | Before Ar annealing | After Ar annealing | |
| CeO2 at 200 °C for 12 h |
5737.881 |
5738.471 |
5.128 |
5.672 |
7.8 |
12.4 |
| 5730.733 |
5731.222 |
3.791 |
3.603 |
|
|
|
| 5724.617 |
5725.931 |
0.761 |
1.308 |
|
|
|
| CeO2 at 160 °C for 24 h |
5737.245 |
5738.888 |
6.734 |
4.703 |
9.8 |
10.4 |
| 5729.911 |
5731.695 |
3.549 |
3.427 |
|
|
|
| 5726.128 |
5726.769 |
1.013 |
0.949 |
|
|
|
| CeO2 at 200 °C for 24 h |
5738.440 |
5736.924 |
5.154 |
5.713 |
13.7 |
13.3 |
| 5731.205 |
5729.691 |
3.799 |
3.741 |
|
|
|
| 5726.465 | 5724.987 | 1.427 | 1.445 | |||
Magnetic properties
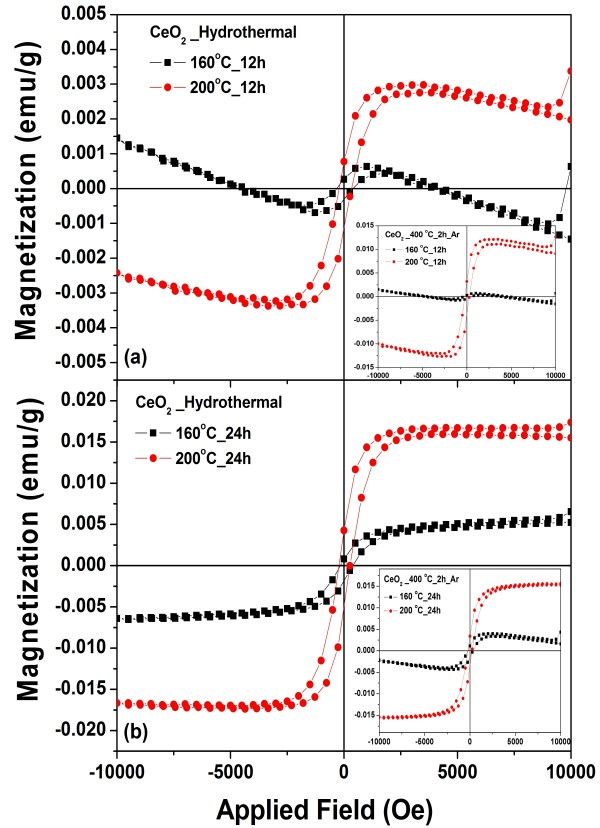
Figure 9a,b shows the field dependence of the specific magnetization (M-H curve) of pure CeO2 samples prepared at 160°C and 200°C for 12 h and prepared at 160°C and 200°C for 24 h, respectively, obtained from room temperature VSM measurements. The sample of CeO2 prepared at 160°C for 12 h exhibits mixed behaviors of ferromagnetism and diamagnetism having hysteresis loops at low field. The samples that were prepared at 200°C for 12 h showed weak RT-FM with magnetization (M) of approximately 0.0026 emu/g, and the samples that were prepared at 160°C and 200°C for 24 h showed weak RT-FM with saturation magnetization (Ms) of approximately 0.0053 and 0.016 emu/g, respectively (as listed in Table 1). These values are higher than the Ms values reported in the literature for pure CeO2 nanospheres [15,17]. Sundaresan et al. [15] reported an RT-FM with an Ms value of approximately 0.0019 emu/g for CeO2 nanoparticles with an average size of approximately 15 nm. Ge et al. [18] reported weak ferromagnetic behavior at an ambient temperature with an Ms value of approximately 0.0007 emu/g for pure CeO2 nanoparticles with an average size of approximately 100 nm obtained commercially from Sigma-Aldrich Corporation (purity of 99.9%). However, magnetic behavior (Ms of approximately 0.0057 emu/g) was also observed in monodisperse CeO2 nanocubes with an average size of approximately 5.3 nm prepared by a chemical method. The results obtained here (experimental and theoretical) provide evidence that pure CeO2 samples can indeed have a magnetic moment due to oxygen vacancies. This direct ferromagnetic coupling is called F-center exchange (FCE) [45], as cerium can have both variable valence states (Ce4+/Ce3+) and oxygen vacancies on the surface of the CeO2 nanoparticles. It is possible that oxygen vacancies can create magnetic moments on neighboring Ce ions [46]. In this work, the effect of Ar annealing at 400°C for 2 h on the magnetic properties was also performed to confirm the effect of oxygen vacancies on magnetic properties of the annealed samples. However, this effect was clearly observed only on the CeO2 sample prepared at 200°C for 12 h and followed by annealing in argon atmosphere at 400°C for 2 h, as its magnetization increased from 0.0025 emu/g to 0.010 emu/g as (see inset of Figure 9b). It is noted that for the samples prepared at 160°C and 200°C for 24 h, annealing did not affect much their magnetic behavior due to the short time of annealing.
Figure 9.
Magnetic properties of monodisperse CeO2nanospheres. Prepared at (a) 160°C and 200°C for 12 h and (b) 160°C and 200°C for 24 h. Inset shows CeO2 samples after annealing in argon atmosphere at 400°C for 2 h.
To explain the origin of the ferromagnetic contribution in the CeO2 nanostructures, the following arguments are proposed. The annealing of samples in an Ar atmosphere at 400°C for 2 h could possibly increase the number of oxygen vacancies and Ce3+ ions in the samples. The high concentration of Ce3+ (approximately 13.3% Ce3+ for the sample prepared at 200 °C for 24 h) suggests that defects could be present in the majority of the samples, which activate more coupling between the Ce ions, leading to an increase in Ms. Wen et al. [9] reported the variation of RT-FM in oxygen and H2 (10%)/Ar (90%) annealed samples of 1% Co-doped CeO2 powder. They found that the sample showed little hysteresis loop after O2 annealing and that the FM signal decreased significantly, while the H2 (10%)/Ar (90%) annealed sample showed an enhanced FM with Ms of about 0.4 emu/g. However, further work is needed to achieve a thorough understanding, and this will be of great interest to researchers in the field of dilute magnetic oxides.
Conclusions
In summary, spheres of pure CeO2 with Ce(NO3)3·6H2O using PVP as a surfactant have been successfully synthesized by hydrothermal method, and their structures, valence state, and magnetic properties were investigated. The XRD and Raman spectroscopy results suggested the formation of CeO2 cubic fluorite structures in the CeO2 samples, which was in agreement with the SAED patterns. It is observed that there is a decrease in the lattice parameters with increasing crystallite size, possibly due to the formation of structure defects/oxygen vacancies in the CeO2 lattice. The bandgaps of our CeO2 nanospheres increased with increasing crystal size indicated by the existence of a blueshift due to a cerium valence change, and this can be attributed to oxygen vacancies at the surface. The surface defects in the CeO2 nanospheres play an important role in the PL properties of our sample. The XANES results reveal that a fraction of the Ce ions are in the 3+ state, and these cause the samples to show weak RT-FM with an Ms value of 0.0026 to 0.016 emu/g. A ferromagnetic exchange mechanism in the pure CeO2 samples is discussed by FCE, and the Ms of samples was shown to change, as well as the proportion of oxygen vacancies.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
SP designed and carried out all the experiments and data analysis, and participated in preparing the draft of the manuscript. SP co-supervised the research and offered technical support for TEM. PC and YP offered technical support for XANES measurement and analysis. SM, the project coordinator, supervised the research, designed the experiments, participated in preparing the draft of the manuscript, and revised the manuscript. All authors read and approved the final manuscript.
Contributor Information
Sumalin Phokha, Email: kaekip@hotmail.com.
Supree Pinitsoontorn, Email: psupree@kku.ac.th.
Prae Chirawatkul, Email: prae@slri.or.th.
Yingyot Poo-arporn, Email: yingyot@slri.or.th.
Santi Maensiri, Email: santimaensiri@g.sut.ac.th.
Acknowledgments
The authors would like to thank the Department of Chemistry of Khon Kaen University for providing VSM and UV–vis facilities, Ubon Ratchathani University for providing XRD and PL facilities, and the National Metal and Materials Technology Center (MTEC) for providing TEM facilities. We thank the Synchrotron Light Research Institute (Public Organization), Nakhon Ratchasima, Thailand for the XANES facilities. S. Phokha would like to acknowledge the financial support for her Ph.D. studies from the Thailand Research Fund through the Royal Golden Jubilee Ph.D. program (grant no. PHD/0275/2550) and the Graduate School of Khon Kaen University (grant no. 53142103). This work is supported by the ‘Industry/University Cooperative Research Center (I/UCRC) in HDD Component, the Faculty of Engineering, Khon Kaen University, and National Electronics and Computer Technology Center, National Science and Technology Development Agency.’
References
- Prellier W, Fouchet A, Mercey B. Oxide-diluted magnetic semiconductors: a review of the experimental status. J Phys Condens Matter. 2003;15:R1583. doi: 10.1088/0953-8984/15/37/R01. [DOI] [Google Scholar]
- Pearton SJ, Heo WH, Ivill M, Norton DP, Steiner T. Dilute magnetic semiconducting oxides. Semicond Sci Technol. 2004;19:R59. doi: 10.1088/0268-1242/19/10/R01. [DOI] [Google Scholar]
- Coey JMD. Dilute magnetic oxides. Curr Opin Solid State Mater Sci. 2006;10:83. doi: 10.1016/j.cossms.2006.12.002. [DOI] [Google Scholar]
- Tiwari A, Bhosle VM, Ramachandran S, Sudhakar N, Narayan J, Budak S, Gupta A. Ferromagnetism in Co doped CeO2: observation of a giant magnetic moment with a high Curie temperature. Appl Phys Lett. 2006;88:142511. doi: 10.1063/1.2193431. [DOI] [Google Scholar]
- Song YQ, Zhang HW, Wen QY, Li YX, Xiao JQ. Room-temperature ferromagnetism of Co-doped CeO2 thin films on Si(111) substrates. Chin Phys Lett. 2007;24:218. doi: 10.1088/0256-307X/24/1/059. [DOI] [Google Scholar]
- Vodungbo B, Zheng Y, Vidal F, Demaille D, Etgens VH, Mosca DH. Room temperature ferromagnetism of Co doped CeO2-δ diluted magnetic oxide: Effect of oxygen and anisotropy. Appl Phys Lett. 2007;90:062510. doi: 10.1063/1.2472520. [DOI] [Google Scholar]
- Fernandes V, Klein JJ, Mattoso N, Mosca DH, Silveira E, Ribeiro E, Schreiner WH, Varalda J, de Oliveira AJA. Room temperature ferromagnetism in Co-doped CeO2 films on Si(001) Phys Rev B. 2007;75:121304R. [Google Scholar]
- Thurber A, Reddy KM, Shutthanandan V, Engelhard MH, Wang C, Hays J, Punnoose A. Ferromagnetism in chemically synthesized CeO2 nanoparticles by Ni doping. Phys Rev B. 2007;76:165206. [Google Scholar]
- Wen QY, Zhang HW, Song YQ, Yang QH, Zhu H, Xiao JQ. Room-temperature ferromagnetism in pure and Co doped CeO2 powders. J Phys Condens Matter. 2007;19:246205. doi: 10.1088/0953-8984/19/24/246205. [DOI] [PubMed] [Google Scholar]
- Ou YN, Li GR, Liang JH, Feng ZP, Tong YX. Ce1-xCoxO2-δ nanorods grown by electrochemical deposition and their magnetic properties. J Phys Chem C. 2010;114:13509–13514. doi: 10.1021/jp1038128. [DOI] [Google Scholar]
- Paula CAB, Daniel AAS, Jose’ GSD, Marcelo AM. Structural and magnetic study of Fe-doped CeO2. Physica B. 2010;405:1821–1825. doi: 10.1016/j.physb.2010.01.054. [DOI] [Google Scholar]
- Maensiri S, Phokha S, Laokul P, Seraphin S. Room temperature ferromagnetism in Fe-doped CeO2 nanoparticles. J Nanosci Nanotechnol. 2009;9:6415–6420. doi: 10.1166/jnn.2009.1372. [DOI] [PubMed] [Google Scholar]
- Venkatesan M, Fitzgerald CB, Coey JMD. Thin films: unexpected magnetism in a dielectric oxide. Nature. 2004;430:630. doi: 10.1038/430630a. [DOI] [PubMed] [Google Scholar]
- Coey JMD, Venkatesan M, Stamenov P, Fitzgerald CB, Dorneles LS. Magnetism in hafnium dioxide. Phys Rev B. 2005;72:024450. [Google Scholar]
- Sundaresan A, Bhargavi R, Rangarajan N, Siddesh U, Rao CNR. Ferromagnetism as a universal feature of nanoparticles of the otherwise nonmagnetic oxides. Phys Rev B. 2006;74:161306R. [Google Scholar]
- Liu Y, Lockman Z, Aziz A, Macmanus DJ. Size dependent ferromagnetism in cerium oxide (CeO2) nanostructures independent of oxygen vacancies. J Phys Condens Matter. 2008;20:165201. doi: 10.1088/0953-8984/20/16/165201. [DOI] [Google Scholar]
- Chen SY, Lu YH, Huang TW, Yan DC, Dong CL. Oxygen vacancy dependent magnetism of CeO2 nanoparticles prepared by thermal decomposition method. J Phys Chem C. 2010;114:19576–19581. doi: 10.1021/jp1045172. [DOI] [Google Scholar]
- Ge MY, Wang H, Liu EZ, Liu JF, Jiang JZ, Li YK, Xu ZA, Li HY. On the origin of ferromagnetism in CeO2 nanocubes. Appl Phys Lett. 2008;93:062505. doi: 10.1063/1.2972118. [DOI] [Google Scholar]
- Lu X, Li X, Chen F, Ni C, Chen Z. Hydrothermal synthesis of prism-like mesocrystal CeO2. J Alloys Comp. 2009;476:958–962. doi: 10.1016/j.jallcom.2008.09.198. [DOI] [Google Scholar]
- Deshpande S, Patil S, Kuchibhatla SVNT, Seal S. Size dependency variation in lattice parameter and valency states in nanocrystalline cerium oxide. Appl Phys Lett. 2005;87:133113. doi: 10.1063/1.2061873. [DOI] [Google Scholar]
- Maensiri S, Marsingboon C, Loakul P, Jareonboon W, Promarak V, Anderson PL, Seraphin S. Egg white synthesis and photoluminescence of platelike clusters of CeO2 nanoparticles. Cryst Growth Des. 2007;7:950–955. doi: 10.1021/cg0608864. [DOI] [Google Scholar]
- Phoka S, Laokul P, Swatsitang E, Promarak V, Seraphinc S, Maensiri S. Synthesis, structural and optical properties of CeO2 nanoparticles synthesized by a simple polyvinyl pyrrolidone (PVP) solution route. Mater Chem Phys. 2009;115:423–428. doi: 10.1016/j.matchemphys.2008.12.031. [DOI] [Google Scholar]
- Kosacki I, Suzuki T, Petrovsky V, Anderson HU, Colomban P. Raman scattering and lattice defects in nanocrystalline CeO2 thin films. Solid State Ionics. 2002;149:99–105. [Google Scholar]
- Kosacki I, Petrovsky V, Anderson HU, Colomban P. Raman spectroscopy of nanocrystalline ceria and zirconia thin films. J Am Ceram Soc. 2002;85:2646–2650. [Google Scholar]
- Lee SH, Lu ZY, Babu SV, Matijevic E. Chemical mechanical polishing of thermal oxide films using silica particles coated with ceria. J Materials Res. 2002;7:2744. [Google Scholar]
- Laberty-Robert C, Long JW, Lucas EM, Pettigrew KA, Stroud RM, Doescher MS, Rolison DR. Sol–gel derived ceria nanoarchitectures: synthesis, characterization and electrical. Chem Mater. 2006;18:50–58. doi: 10.1021/cm051385t. [DOI] [Google Scholar]
- Weber WH, Hass KC, McBride JR. Raman study of CeO2: second-order scattering, lattice dynamics and particle-size effects. Phys Rev B. 1993;48:178–185. doi: 10.1103/PhysRevB.48.178. [DOI] [PubMed] [Google Scholar]
- Zhou F, Ni X, Zhang Y, Zheng H. Size-controlled synthesis and electrochemical characterization of spherical CeO2 crystallites. J Coll Interf Sci. 2007;307:135–138. doi: 10.1016/j.jcis.2006.11.005. [DOI] [PubMed] [Google Scholar]
- Ziegler E, Heinrich A, Oppermann H, Stover G. Electrical properties and non-stoichiometry in ZnO single crystals. Phys Status Solidi A. 1981;66:635. doi: 10.1002/pssa.2210660228. [DOI] [Google Scholar]
- Chen HI, Chang HY. Synthesis of nanocrystalline cerium oxide particles by the precipitation method. Ceram Int. 2005;31:795–802. doi: 10.1016/j.ceramint.2004.09.006. [DOI] [Google Scholar]
- Masui T, Fujiwara K, Machida K, Adachi G, Sakata T, Mori H. Characterization of cerium(IV) oxide ultrafine particles prepared using reversed micelles. Chem Mater. 1997;9:2197–2204. doi: 10.1021/cm970359v. [DOI] [Google Scholar]
- Tsunekawa S, Wang JT, Kawazoe Y, Kasuya A. Blueshifts in the ultraviolet absorption spectra of cerium oxide nanocrystallites. J Appl Phys. 2003;94:3654. doi: 10.1063/1.1600520. [DOI] [Google Scholar]
- Barreca D, Bruno G, Gasparotto A, Losurdob M, Tondello E. Nanostructure and optical properties of CeO2 thin films obtained by plasma-enhanced chemical vapor deposition. Mater Sci Eng C. 2003;23:1013–1016. doi: 10.1016/j.msec.2003.09.103. [DOI] [Google Scholar]
- Sathyamurthy S, Leonard KJ, Dabestani RT, Paranthaman MP. Reverse micellar synthesis of cerium oxide nanoparticles. Nanotechnology. 2005;16:1960–1964. doi: 10.1088/0957-4484/16/9/089. [DOI] [Google Scholar]
- Yu SH, Colfen H, Fischer A. High quality CeO2 nanocrystals stabilized by a double hydrophilic block copolymer. Colloids Surfaces A Physicochem Eng Aspects. 2004;243:49–52. doi: 10.1016/j.colsurfa.2004.05.006. [DOI] [Google Scholar]
- Gao F, Li GH, Zhang JH, Qin FG, Yao ZY, Liu ZK, Wang ZG, Lin LY. Growth and photoluminescence of epitaxial CeO2 films on Si (111) substrate. Chin Phys Lett. 2001;18:443. doi: 10.1088/0256-307X/18/3/345. [DOI] [Google Scholar]
- Morshed AH, Moussa ME, Bedair SM, Leonard R, Liu SX, Masry NE. Violet/blue emission from epitaxial cerium oxide films on silicon substrates. Appl Phys Lett. 1997;70:1647. doi: 10.1063/1.118658. [DOI] [Google Scholar]
- Marabelli F, Wachter P. Covalent insulator CeO2: optical reflectivity measurements. Phys Rev B. 1987;36:1238–1243. doi: 10.1103/PhysRevB.36.1238. [DOI] [PubMed] [Google Scholar]
- Koelling DD, Boring AM, Wood JH. The electronic structure of CeO2 and PrO2. Solid State Commun. 1983;47:227–232. doi: 10.1016/0038-1098(83)90550-1. [DOI] [Google Scholar]
- Hormes J, Pantelouris M, Balazs GB, Rambaby B. X-ray absorption near edge structure (XANES) measurements of ceria-based solid electrolytes. Solid State Ionics. 2000;136-137:945–954. [Google Scholar]
- Zhang F, Wang P, Koberstein J, Khalid S, Chan SW. Cerium oxidation state in ceria nanoparticles studied with X-ray photoelectron spectroscopy and absorption near edge spectroscopy. Surf Sci. 2004;563:74–82. doi: 10.1016/j.susc.2004.05.138. [DOI] [Google Scholar]
- Beck DD, Capehart TW, Hoffman RW. Determination of Ce valence in Rh-Ce/Al2O3 catalysts by X-ray absorption. Chem Phys Lett. 1989;159:205–213. doi: 10.1016/0009-2614(89)87411-1. [DOI] [Google Scholar]
- Shahin AM, Grandjean F, Long GJ, Schuman TP. Cerium L3-edge XAS investigation of the structure of crystalline and amorphous cerium oxides. Chem Mater. 2005;17:315–321. doi: 10.1021/cm0492437. [DOI] [Google Scholar]
- Zhang J, Wu Z, Liu T, Hu T, Wu Z, Ju X. XANES study on the valence transitions in cerium oxide nanoparticles. J Synchrotron Rad. 2001;8:531–532. doi: 10.1107/S0909049500016022. [DOI] [PubMed] [Google Scholar]
- Coey JMD, Douvalis AP, Fitzgerald CB, Venkatesan M. Ferromagnetism in Fe-doped SnO2 thin films. Appl Phys Lett. 2004;84:1332. doi: 10.1063/1.1650041. [DOI] [Google Scholar]
- Esch F, Fabris S, Zhou L, Montini T, Africh C, Fornasiero P, Comelli G, Renzo R. Electron localization determines defect formation on ceria substrates. Science. 2005;309:752–755. doi: 10.1126/science.1111568. [DOI] [PubMed] [Google Scholar]