Abstract
Anorexia nervosa (AN) is characterized by subnormal estrogen and dehydroepiandrosterone (DHEA) levels. We sought to determine whether the combination of DHEA + estrogen/progestin is superior to placebo in preserving skeletal health over 18 months in AN. Females with AN, aged 13 to 27 years, were recruited for participation in this double-blind, placebo-controlled, randomized trial. Ninety-four subjects were randomized, of whom 80 completed baseline assessments and received either study drug (oral micronized DHEA 50 mg + 20 µg ethinyl estradiol/0.1 mg levonorgestrel combined oral contraceptive pill [COC] daily; n = 43) or placebo (n = 37). Serial measurements of areal bone mineral density (aBMD), bone turnover markers, and serum hormone concentrations were obtained. Sixty subjects completed the 18-month trial. Spinal and whole-body aBMD z scores were preserved in the DHEA + COC group, but decreased in the placebo group (comparing trends, P = .008 and P = .001, respectively). Bone turnover markers initially declined in subjects receiving DHEA + COC and then returned to baseline. No differences in body composition, adverse effects of therapy, or alterations in biochemical safety parameters were observed. Combined therapy with DHEA + COC appears to be safe and effective for preventing bone loss in young women with AN, whereas placebo led to decreases in aBMD. Dehydroepiandrosterone + COC may be safely used to preserve bone mass as efforts to reverse the nutritional, psychological, and other hormonal components of AN are implemented.
1. Introduction
The many patients who have anorexia nervosa (AN) are at increased risk for fracture and early osteoporosis [1–4]. The mechanisms of bone loss observed in adolescent girls with AN are complex. Identified factors that may affect attainment of peak bone mass include low levels of estrogens, androgens, and insulin-like growth factor I (IGF-I); increased cortisol levels; poor nutrition; family history of osteoporosis; and the low body mass that is characteristic of AN. A combination of these and other factors likely leads to low bone mineral density (BMD) in young women with this disease. Effective therapies for preventing bone loss in patients with AN have remained elusive. Hormone therapy has yielded disappointing results except in the most severely malnourished patients [4–10]. As adolescence is a critical period for bone acquisition [11], the identification of strategies to preserve areal BMD (aBMD) in adolescents with AN is paramount.
Secretion of the adrenal steroid dehydroepiandrosterone (DHEA) increases dramatically during adolescence and decreases with age [12,13]. Dehydroepiandrosterone concentrations are positively correlated with BMD, suggesting an important role for DHEA in the prevention of bone loss associated with low-DHEA states [14–16]. Dehydroepiandrosterone has antiresorptive properties, related to suppression of marrow production of the proresorptive cytokine interleukin-6, among others [17]. Dehydroepiandrosterone may also directly affect osteoblasts independent of androgen or estrogen receptors [18,19]. Dehydroepiandrosterone administration has been investigated in the elderly [13]. Postmenopausal women receiving oral or percutaneous DHEA maintained spinal or hip aBMD compared with controls [20–23], but DHEA’s beneficial effects were not observed in elderly men [23–25] or in short-term trials [26].
Some reports demonstrate subnormal DHEA levels in adolescents with AN, similar to concentrations observed in the elderly [27,28]. We reported that micronized DHEA reduced markers of bone resorption and increased indices of bone formation in adolescents and young women with AN [29]. In a randomized clinical trial, we previously demonstrated that DHEA monotherapy increased hip aBMD in young women with AN, but the treatment effect was similar to a group receiving estrogen monotherapy, and effects were attenuated by accompanied weight gain [4].
Combination therapy with androgen and estrogen/progestin combined oral contraceptive pill (COC) may be the ideal regimen to normalize altered bone turnover in AN. Estrogen inhibits bone resorption, while decreasing bone formation [6]. Androgens, in turn, stimulate bone formation [29]. Dehydroepiandrosterone has been described as an “estrogen precursor” and is converted into estrogen and androgen [21,23]. Thus, both bone formation and resorption may be affected to attenuate bone loss. Combination androgen and estrogen therapy administered to postmenopausal women led to anabolic trends in bone formation markers [30] and skeletal gains [20]. Our objective was to determine the effects of an 18-month regimen of oral DHEA + COC vs placebo on BMD in young women with AN.
2. Methods
2.1. Participant selection
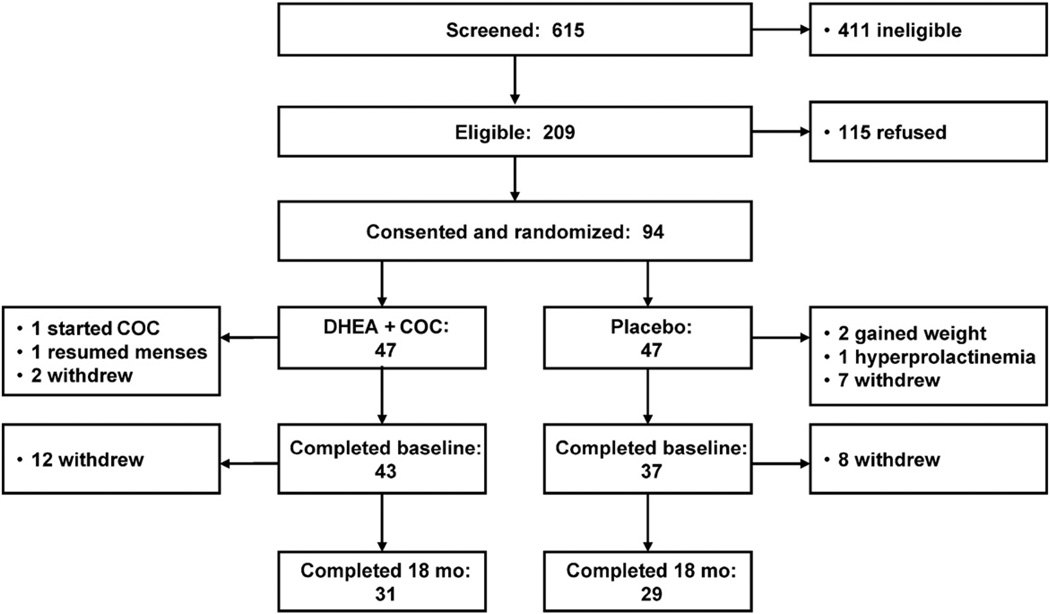
From 2003 to 2008, 615 young women were screened, of whom 209 met study eligibility criteria (Fig. 1). Eligible females were aged 13 to 27 years, were skeletally mature, and had been diagnosed with AN (amenorrhea, fear of weight gain, and malnutrition with body weight ≤85% median body weight for age and sex) by their outpatient medical provider within the Division of Adolescent Medicine at Children’s Hospital Boston. Patients were excluded for other medical diagnoses (ie, celiac disease, diabetes) or medications affecting BMD (ie, glucocorticoids). Informed consent was obtained, with parental consent/subject assent for subjects younger than 18 years.
Fig. 1.
Patient recruitment, enrollment, and disposition.
2.2. Study design
The study was a single-site, randomized, placebo-controlled trial conducted at Children’s Hospital Boston. Ninety-four subjects were enrolled and assigned sequential randomization codes, interpretable as drug or placebo only by the pharmacy staff according to a key prepared by the study statistician. The sequence of assignments was randomly permuted in blocks of 2, 4, and 6 to ensure unpredictability as well as temporal balance. The pharmacy dispensed drug or placebo in identical gelatin capsules, indistinguishable by patients or staff. Assignments were not revealed to any investigators, subjects, or staff aside from the pharmacist and statistician until the entire trial was complete. The treatment arm received 18 months of oral micronized DHEA (50 mg daily; Belmar Pharmacy, Lakewood, CO; Investigational New Drug [IND] 52 192). We contracted with a single vendor (Belmar Pharmacy) for the duration of the trial to ensure purity and consistency of the compound. Dehydroepiandrosterone was initially given in combination with conjugated equine estrogens (0.3 mg daily; Premarin, Wyeth Pharmaceuticals, Collegeville, PA, USA) for the first 3 months. As we have found that adolescents with AN are exquisitely sensitive to estrogen-related adverse effects, we first began low-dose conjugated estrogens to minimize these symptoms, a regimen that has worked well in our clinical practice and a previous trial [4]. After 3 months, subjects in the treatment arm received 15 months of DHEA + COC (20 µg ethinyl estradiol/0.1 mg levonorgestrel; Alesse, Wyeth). The other group received placebo for the entire 18 months. All participants received routine care including medical, nutritional, and psychological monitoring. Participants returned for assessments at 3, 6, 12, and 18 months. Medication compliance was measured at each visit with pill counts. All subjects were advised to consume the recommended daily intake of calcium (1300 mg) and vitamin D (600 IU).
2.3. Study assessments
The primary outcomes were aBMD of the total body, total hip, and lumbar spine by dual-energy x-ray absorptiometry (DXA; QDR 4500, Hologic, Bedford, MA) obtained at 0, 6, 12, and 18 months. Measurements were compared with age- and sex-matched controls [31–33], and aBMD z scores (number of standard deviations away from the average value of the reference group) were calculated. The average in vivo precision for aBMD (percentage coefficient of variation) was 0.62% at the spine and 0.72% at the hip.
Fasting blood samples and second-morning urine specimens were collected for bone formation (osteocalcin [polyclonal radioimmunoassay]) [34] and bone resorption markers (urinary N-telopeptides [NTx; enzyme-linked immunosorbent assay, Wampole Laboratories, Princeton, NJ, USA]). Serum hormones were measured, including DHEA sulfate (DHEA-S; radioimmunoassay, Esoterix, Calabasas Hills, CA, USA), estradiol and total testosterone (high-performance liquid chromatography tandem mass spectrometry, Esoterix), free testosterone (equilibrium dialysis, Esoterix), IGF-I (double-antibody radioimmunoassay, ALPCO Diagnostics, Salem, NH, USA), and sex hormone–binding globulin (SHBG; immunoradiometric assay, Esoterix). Safety studies (aspartate aminotransferase [AST], alanine aminotransferase [ALT], lipid profiles, fasting glucose, and insulin; electrochemiluminescence immunoassay, Roche Molecular Biochemicals, Indianapolis, IN, USA) were also measured. At each visit, acne and hirsutism were assessed on physical examination using a 4-point scale ranging from none (0) to severe (4), as has been used previously. Other end points included anthropometrics and body composition by DXA.
2.4. Statistical analyses
The trial was designed with a target of 50 completers, providing 80% power to detect a difference between treatment arms of 0.017 g cm−2y−1 in the rate of change of hip aBMD as observed in our prior trial of DHEA monotherapy.
Baseline comparison of continuous measures between arms was made by Student t test, corroborated by Wilcoxon 2-sample test for variables with a skewed distribution. Dichotomies were compared by Fisher exact test.
The course of each measurement from baseline to 18 months was compared between arms by linear smoothing-spline analysis, a semiparametric regression technique that furnishes a precise estimate of the general time trend in longitudinal data while permitting arbitrary changes of linear direction, both above and below the general trend, at designated junction points [35]. In a 2-arm trial, spline analysis produces a test for the difference in trend between arms (PΔtrend) as well as a test for nonzero trend in each arm (Ptrend). Analyses of aBMD and bone markers were adjusted for time-varying weight and age. Analyses of aBMD z were adjusted for weight only, as age is accounted for in the z score calculation. All other longitudinal analyses were adjusted for age. Variables with skewed distribution were log-transformed for analysis and retransformed for reporting, with linear trend estimates expressed as percentage change per month.
Spline-fitting computations were implemented as a mathematically equivalent linear mixed model, with random effects (Gaussian deviates) serving to express subject-to-subject variation in overall slope and junction-to-junction variation in local slope. The mixed-model formulation accounts for visit-to-visit correlation within subjects and carries the additional advantage of being unbiased in the presence of missing data, so long as the missingness is either random or predictable by available variables [36].
We identified variables associated with missingness by comparing baseline characteristics of dropouts to those who completed the trial. In cases where dropouts differed significantly from completers, we tested whether the difference was uniform across trial arms using 2-factor analysis of variance (for continuous characteristics) or exact 2-factor logistic regression (for dichotomies). In one instance of significant nonuniformity, we used multiple imputation techniques [36] to simulate complete data and check for impact on the primary findings even though, theoretically, the mixed-model analysis would not be affected by missing data related to known variables. Compliance with medication at the 4 follow-up visits was compared between arms by 2-factor logistic regression.
All analyses followed the intention-to-treat principle, ascribing the randomly assigned treatment to each subject regardless of compliance. SAS software (version 9.2; Cary, NC) was used for all computations. Statistical test results from spline analysis are reported with specific reference to the tested hypothesis as described above (Ptrend, PΔtrend); other test results are reported simply as P. Findings with P < .05 were considered statistically significant.
3. Results
3.1. Patients
Among the 94 randomized subjects, 14 (4 DHEA + COC, 10 placebo) became ineligible or withdrew before completing baseline measurements (Fig. 1), resulting in a final sample of 80 (43 DHEA + COC, 37 placebo), aged 18.1 ± 2.7 years (mean ± SD). The groups did not differ in baseline demographic characteristics (Table 1). Between the time of study recruitment and the actual baseline study visit, subjects had restored varying degrees of body weight. Thus, not all subjects were still below 85% median body weight at baseline; however, all subjects remained amenorrheic. Subjects had low body mass index (BMI) (18.0 ± 1.5 kg/m2; range, 14.8–22.9 kg/m2) and amenorrhea of median duration 11 months (range, 1–144 months). Subjects designated as amenorrheic for 1 month were 6 participants receiving COCs at recruitment who discontinued COCs at least 1 month before participation. At baseline, moderate skeletal deficits were noted at the spine, with 13 subjects (16%) showing low aBMD (aBMD z ≤ −2 SD; Table 1). Vitamin D concentrations were measured at baseline. If deficient (25-hydroxy-vitamin D [25OHD] <20 ng/mL), subjects were supplemented with oral ergocalciferol (vitamin D2) or cholecalciferol (vitamin D3) 50 000 IU once weekly for 2 months, then 400 to 600 IU daily, to achieve normal levels. If a participant’s baseline measure of 25OHD = 21 to 30 ng/mL (vitamin D insufficiency), the subject was instructed to supplement with vitamin D 1000 IU PO daily. Concentrations of 25OHD were measured at each study visit until documented to be greater than 30 ng/mL.
Table 1.
Baseline participant characteristics by study arm.
| All (80) | DHEA + COC arm (43) | Placebo arm (37) | P a | ||
|---|---|---|---|---|---|
| Mean ± SD | Minimum, maximum | Mean ± SD | |||
| Age, y | 18.1 ± 2.7 | 13.3, 27.1 | 18.0 ± 2.5 | 18.3 ± 2.8 | .84 |
| Height, cm | 164.0 ± 7.0 | 148.3, 180.1 | 164.4 ± 6.7 | 163.6 ± 7.5 | .47 |
| Weight, kg | 48.6 ± 5.8 | 37.4, 67.9 | 49.1 ± 5.9 | 48.0 ± 5.6 | .55 |
| BMI, kg/m2 | 18.0 ± 1.5 | 14.8, 22.9 | 18.1 ± 1.5 | 17.8 ± 1.5 | .24 |
| BMI, % of median for age | 86 ± 7 | 68, 109 | 86 ± 7 | 85 ± 8 | .15 |
| Fat mass, kg | 8.6 ± 2.7 | 3.7, 17.4 | 8.8 ± 2.8 | 8.4 ± 2.6 | .55 |
| Lean mass, kg | 37.4 ± 4.5 | 27.2, 47.9 | 37.5 ± 4.2 | 37.2 ± 4.8 | .68 |
| 25(OH) D, ng/mL | 38.3 ± 14.4 | 9, 83 | 39.8 ± 13.4 | 36.7 ± 15.6 | .14 |
| Parathyroid hormone, pg/mL | 29.7 ± 11.2 | 10.2, 70.2 | 30.1 ± 11.4 | 29.4 ± 11.1 | .71 |
| Lumbar spine, b g/cm2 | 0.89 ± 0.10 | 0.67, 1.15 | 0.89 ± 0.10 | 0.88 ± 0.10 | .51 |
| z, SD | −0.90 ± 1.00 | −3.3, 1.2 | −0.84 ± 1.02 | −0.98 ± 1.00 | .57 |
| Total hip, g/cm2 | 0.89 ± 0.11 | 0.66, 1.19 | 0.89 ± 0.10 | 0.89 ± 0.12 | .84 |
| z, SD | −0.36 ± 0.92 | −2.2, 2.0 | −0.37 ± 0.87 | −0.35 ± 1.00 | .90 |
| Whole body, g/cm2 | 1.06 ± 0.07 | 0.92, 1.28 | 1.07 ± 0.07 | 1.05 ± 0.08 | .28 |
| z, SD | 0.07 ± 0.94 | −2.0, 2.2 | 0.19 ± 0.90 | −0.06 ± 0.97 | .23 |
| Median (Q1–Q3) | Minimum, maximum | Median (Q1–Q3) | |||
| Duration of AN, mo | 12 (4–28) | 1, 132 | 12 (6–40) | 9 (4–18) | .07 |
| Months amenorrhea c | 11 (5–20) | 1, 144 | 9 (6–18) | 12 (5–20) | .63 |
| n (%) | n (%) | ||||
| White | 71 (89) | 39 (91) | 32 (86) | .73 | |
| Hispanic | 2 (3) | 1 (2) | 1 (3) | 1.0 | |
| Lumbar spine: z ≤ −1 SD | 41 (51) | 20 (47) | 21 (57) | .38 | |
| z ≤ −2 SD | 13 (16) | 8 (19) | 5 (14) | .76 | |
| Total hip: z ≤ −1 SD | 19 (24) | 9 (21) | 10 (29) | .60 | |
| z ≤ −2 SD | 3 (4) | 1 (2) | 2 (6) | .58 | |
| Whole body: z ≤ −1 SD | 11 (14) | 4 (9) | 7 (19) | .33 | |
| z ≤ −2 SD | 1 (1) | 0 (0) | 1 (3) | .46 | |
Kruskal-Wallis or Fisher exact test comparing distribution in DHEA + COC and placebo arms.
Areal bone mineral density (BMD).
Excludes 5 participants with primary amenorrhea; includes 6 participants who were on COCs at the time of recruitment and discontinued use of the medication for at least 1 month before the baseline visit.
Twelve subjects receiving DHEA + COC and 8 receiving placebo discontinued participation after baseline for reasons unrelated to the study (Fig. 1). Dropouts were generally younger than completers (17.0 vs 18.5 years, P = .02) and nonwhite (6/20 = 30% vs 3/60 = 5%, P = .006); but the age gap did not differ between groups (P = .84 for dropout × treatment interaction), nor did race imbalance (P = .31). Dropouts had higher baseline spinal aBMD z than completers in the treated, but not placebo, group (interaction P = .008). This finding prompted multiple imputation analysis, described below. Dropouts and completers did not differ with respect to other baseline characteristics.
Thirty-nine percent of subjects reported missing at least 1 dose of medication before the 3-month visit, 53% at 6 months, 60% at 12 months, and 48% at 18 months. Compliance did not vary between trial arms (P = .67). During study participation, 5 subjects were medically hospitalized. Activity level was deter mined by the subjects’ clinicians; no participants were confined to long-term bed rest. Sixty women completed the trial.
3.2. Bone mineral density
The time course of aBMD differed significantly between treated and placebo subjects whether measured at the spine, hip, or whole body (Fig. 2). At each location, treated subjects showed a modest upward trend in aBMD over 18 months and a virtually flat course of aBMD z, as assessed by the overall trend estimate in spline analysis. In contrast, placebo subjects showed a convex or downward trend in both aBMD and aBMD z at each location. The active − placebo difference was statistically significant in every case (PΔtrend < .05).
Fig. 2.
Areal BMD and aBMD z scores in adolescent and young adult females with AN randomized to 18 months of treatment with DHEA + COC or placebo. Lines represent the fitted spline ± standard error. P(trend) tests for change over time in each treatment group. P(Δtrend) compares the time course between treatment groups.
When we restricted the analysis to the 36 of 80 women whose BMI was less than 85% of the median for age, the treatment difference for hip aBMD retained its magnitude and statistical significance (PΔtrend = .02). For hip aBMD z, the difference in trend was attenuated by 25%, but remained statistically significant (PΔtrend = .03). At the other sites in this smaller group, the difference in trend for aBMD and aBMD z shrank by 25% to 50%, whereas the standard error of the difference swelled by 25% to 40%, resulting in loss of statistical significance with PΔtrend between .08 and .28.
Body weight strongly influenced hip measures, with a coefficient for aBMD of 0.0018 g cm−2 kg−1 (P = .001) and a coefficient for aBMD z of 0.016 SD per kilogram (P = .0004). Weight had a lesser influence on whole-body aBMD (P = .07) and an inverse effect on whole-body aBMD z (−0.014 SD per kilogram, P = .001). Weight did not influence the spine (P > .90). Age did not affect aBMD (P > .30) or modify the treatment effect at any site (time × treatment × age, P > .20).
3.3. Bone turnover markers
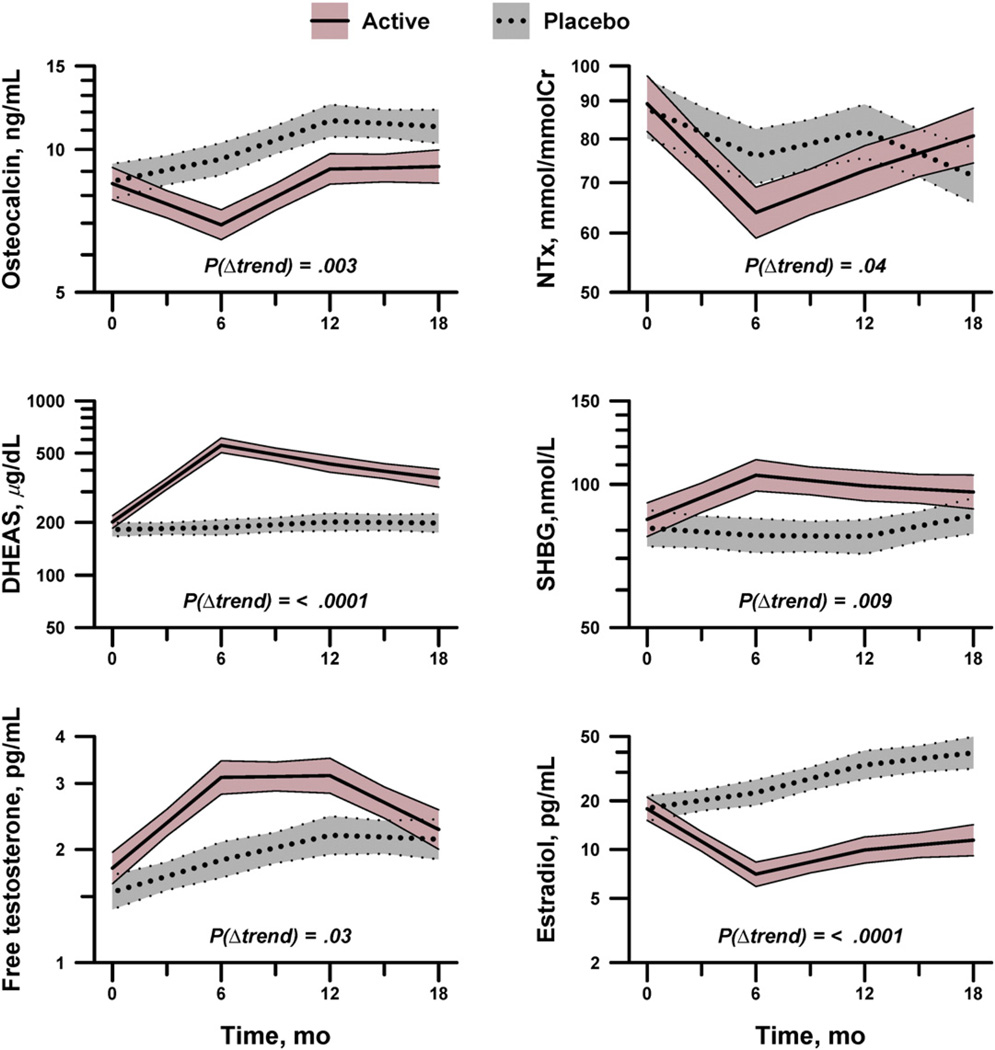
The pattern of change in bone biomarkers differed distinctly between treatment groups (Fig. 3). Osteocalcin rose initially in placebo-treated subjects and plateaued after 12 months, with a nonsignificant overall upward trend of 1.0%/mo (Ptrend = .19), whereas in DHEA + COC subjects, it showed a concave course, initially declining and then returning to near baseline by 18 months (Ptrend = .01; PΔtrend = .003). The NTx declined overall at 2.5%/mo in placebo subjects (Ptrend < .05), but showed a sharp concave course in DHEA + COC subjects (Ptrend < .0001; PΔtrend = .04).
Fig. 3.
Bone turnover markers and steroid hormones in adolescent and young adult females with AN randomized to 18 months of treatment with DHEA + COC or placebo. Lines represent the fitted spline ± standard error. P(Δtrend) compares the time course between treatment groups.
3.4. Body mass and composition
Among trial completers, 5 lost weight (1.4–5.3 kg), 1 subject showed no change, and the remainder gained up to 21.7 kg. One quarter of completers (15/60) attained a BMI higher than the median for age by study’s end, only one of whom had been at that level at baseline. Steady increases were observed for all body mass and composition measures (Table 2), including weight (5–6 kg/y), lean mass (1–2 kg/y), fat mass (3 kg/y), and BMI, whether expressed absolutely (2 kg m−2 y−1), standardized (0.9–1.0 SD per year), or relative to median for age (10%–11%/y). The rate of change in body mass and composition measures did not differ between groups (Table 2).
Table 2.
Baseline mean and 18-month trends in anthropometry, body composition, IGF-1, serum lipids, liver function measures, and glucose tolerance in young women with AN treated with DHEA + COC (active) or placebo.
| Group | Baseline a | Trend b | |||
|---|---|---|---|---|---|
| Change/y ± SE | Ptrend | PΔtrend | |||
| Weight, kg | Active | 49.1 ± 0.9 | 5.9 ± 1.0 | <.0001 | .52 |
| Placebo | 48.0 ± 0.9 | 5.2 ± 1.1 | <.0001 | ||
| BMI, kg/m2 | Active | 18.1 ± 0.2 | 2.3 ± 0.4 | <.0001 | .39 |
| Placebo | 17.8 ± 0.2 | 1.9 ± 0.4 | <.0001 | ||
| BMI z, SD | Active | −1.24 ± 0.12 | 0.97 ± 0.17 | <.0001 | .82 |
| Placebo | −1.47 ± 0.15 | 0.94 ± 0.18 | <.0001 | ||
| BMI, % of median for age | Active | 86 ± 1 | 10.7 ± 1.7 | <.0001 | .38 |
| Placebo | 85 ± 1 | 9.0 ± 1.8 | <.0001 | ||
| Lean mass, kg | Active | 37.5 ± 0.6 | 1.8 ± 0.4 | <.0001 | .17 |
| Placebo | 37.2 ± 0.8 | 1.4 ± 0.4 | .002 | ||
| Fat mass, kg | Active | 8.8 ± 0.4 | 3.2 ± 0.7 | <.0001 | .92 |
| Placebo | 8.4 ± 0.4 | 3.1 ± 0.8 | .0002 | ||
| IGF-1, ng/mL | Active | 268 ± 15 | 25 ± 17 | .14 | .88 |
| Placebo | 277 ± 17 | 27 ± 17 | .11 | ||
| Total cholesterol, mg/dL | Active | 169 ± 6 | 7.2 ± 8.2 | .38 | .26 |
| Placebo | 169 ± 5 | −4.6 ± 11.4 | .69 | ||
| HDL cholesterol, mg/dL | Active | 68 ± 2 | −2.7 ± 2.7 | .33 | .63 |
| Placebo | 70 ± 2 | −1.7 ± 2.8 | .54 | ||
| LDL cholesterol, mg/dL | Active | 95 ± 6 | 0.9 ± 3.5 | .80 | .57 |
| Placebo | 90 ± 4 | −1.8 ± 3.5 | .61 | ||
| Triglycerides, mg/dLc | Active | 78 ± 7 | 0.1 ± 5.1 | .99 | .89 |
| Placebo | 71 ± 7 | −1.0 ± 5.3 | .85 | ||
| AST, IU/L c | Active | 33 ± 3 | −6.9 ± 1.9 | .0006 | .09 |
| Placebo | 29 ± 2 | −2.6 ± 2.0 | .19 | ||
| ALT, IU/L c | Active | 33 ± 5 | −9.5 ± 3.7 | .01 | .38 |
| Placebo | 29 ± 5 | −4.8 ± 3.9 | .22 | ||
| Fasting glucose, mg/dL | Active | 75 ± 2 | 1.5 ± 3.3 | .66 | .72 |
| Placebo | 78 ± 2 | 0.8 ± 3.3 | .82 | ||
| Fasting insulin, µIU/mL | Active | 8.9 ± 1.8 | −1.0 ± 1.9 | .60 | .32 |
| Placebo | 7.2 ± 2.6 | 1.8 ± 2.0 | .38 | ||
HDL indicates high-density lipoprotein; LDL, low-density lipoprotein.
Unadjusted baseline mean ± standard error.
Mean linear trend over 18 months, estimate ± standard error from spline analysis. Ptrend tests for nonzero slope within treatment group. PΔtrend tests for equal slope in active and placebo groups.
Outliers excluded from analysis: triglyceride >400 mg/dL (1), AST >100 IU/L (3), and ALT >200 IU/L (3).
3.5. Hormone concentrations
Hormone response differed between the 2 treatment groups as shown in Fig. 3. Dehydroepiandrosterone sulfate concentrations increased rapidly among treated subjects (Ptrend < .0001) and remained above twice baseline through-out, with negligible changes in placebo subjects (Ptrend = .80; PΔtrend < .0001). Among completers, DHEA-S exceeded the upper limit of normal (248 µg/dL) at least once in 28 (90%) of 31 subjects receiving DHEA + COC, contrasted with 14 (48%) of 29 placebo-treated subjects (P = .0006). Sex hormone–binding globulin increased transiently in the DHEA + COC group (Ptrend = .002) while remaining flat in placebo subjects (Ptrend =.63; PΔtrend = .009). Free testoster one showed a similar pattern, rising transiently in DHEA + COC subjects (Ptrend < .0001) but not in placebo subjects (Ptrend =.12; PΔtrend = .03). Free testosterone exceeded the upper limit of normal (6.3 pg/mL) at least once in 7 (23%) of 31 completers in the DHEA + COC arm compared with 2 (7%) of 29 in the placebo arm (P = .15). Insulin-like growth factor I did not vary by treatment group (Table 2). Serum estradiol differed markedly between arms, rising slowly in placebo subjects (1.0%/mo, P = .28) but falling to less than half of baseline in DHEA + COC subjects and never recovering (Ptrend < .0001; PΔtrend < .0001). Among completers, all 31 subjects receiving DHEA + COC had episodic withdrawal bleeding. In the placebo group, 22 (76%) of the 29 subjects resumed menses during the 18-month trial.
3.6. Safety measures
Despite elevated serum androgen concentrations, no group differences in acne or hirsutism were seen. Alanine aminotransferase and AST were similar between groups (Table 2). Only 2 subjects developed ALT or AST greater than 100 IU/L, a relatively common finding in malnourished adolescents with AN [37]; one received placebo and one received treatment, and the elevated level spontaneously resolved. No group differences in lipids, glucose, or insulin occurred (Table 2); nor did clinically significant abnormalities in these measures.
3.7. Missing-data analysis
Because we detected differing baseline spinal aBMD z according to dropout status and treatment arm, we examined the possible influence of missing data by conducting multiple imputation analysis. Each missing spinal aBMD z value was estimated by a linear regression equation fitted to the available spinal aBMD z data at the prior visit, including terms for age, race, and aBMD z × treatment interaction, to account for all identified influences on dropout. The imputation was repeat ed 15 times with random perturbations of the regression coefficients. Each resulting complete data set was analyzed by repeated-measures analysis of variance with autoregressive covariance, a simplified version of the spline analysis. Time × treatment interaction in the 15 replicates ranged in significance from P = .003 to .080, with median P = .019. The combined significance, taking account of between-imputation and within-imputation variance, was P = .035, corroborating the finding for spinal aBMD z in the original data.
4. Discussion
We found combination therapy with oral DHEA + COC to be safe and effective for preserving aBMD in young women with AN. Benefits of treatment were seen at each skeletal site; the DHEA + COC arm maintained aBMD for the trial’s duration, whereas the placebo-treated group lost aBMD. Given that BMD should be stable or increasing during young adulthood, any degree of bone loss is concerning. The advantageous effects of DHEA + COC persisted even after controlling for weight gain.
The current regimen of combination androgen and estrogen hormone replacement differs from those previously studied. Estrogen monotherapy has not yielded a beneficial effect on skeletal health in women with AN [4–6,8,38]. Leptin concentrations are low in women with AN [39–42]; nutritional rehabilitation and weight gain result in increased leptin concentrations [39,43]. The skeletal effects of leptin differ depending on whether an individual is deficient, tolerant, or resistant to leptin [44–46]. Short-term leptin replacement in women with hypothalamic amenorrhea led to resumption of menses and increases in bone formation markers, but no change in BMD [47]. Longer-term (12-month) treatment with metreleptin led to BMD increases of 4% to 6% at the lumbar spine in heavily exercising women with hypothalamic amenorrhea; but skeletal sites (hip, radius), each composed primarily of cortical bone, did not exhibit a similar response [46]. Preliminary research has investigated the use of bisphosphonates for treatment of bone loss and maintenance of BMD in adolescents with AN [48,49]. Insulin-like growth factor I stimulates osteoblast function and collagen synthesis, and is abnormally low in patients with AN [28,50]. In a randomized trial, women with AN who were treated with rhIGF-I demonstrated beneficial changes in BMD [38]. Combined oral contraceptive pills appeared to augment the effects of rhIGF-I when used in combination [38]. Proof of concept regarding the beneficial skeletal effects of combined antiresorptive and anabolic therapies in AN was suggested by these data and explored further in the current study. Although these therapies hold promise, further research is needed regarding patient acceptance of a subcutaneous medication, administration of these agents to a younger age group, and effects of prolonged treatment.
The decrease in bone resorption markers exhibited by the treatment arm provides a potential mechanism of effect for our findings. In patients with AN, bone remodeling differs from that observed in other estrogen-deficient states. The patho genesis of bone loss in adolescents with AN is characterized by both impaired bone formation and accelerated bone resorption [8,28,51]. These abnormalities improve with refeeding; weight gain is associated with normalization of both elevated bone resorption and depressed bone formation [52]. Previously, DHEA treatment led to anabolic effects on bone formation, whereas adolescents receiving COC exhibited decreases in bone formation [4]. Given the potential synergistic mecha nisms of action, we explored the effect of combination DHEA + COC on bone acquisition. Similar to previous work, the shape of the time course of osteocalcin differed significantly between groups, with no significant change for placebo subjects compared with an initial decline followed by a slow rise in DHEA + COC subjects. Osteocalcin reflects overall bone turnover in addition to bone formation. Thus, group differences in osteocalcin may have been driven in part by our NTx findings. Bone resorption initially declined in the active group (perhaps due to the estrogen/estradiol supplementation) and then started to rise. Similar reductions in bone resorption occurred in postmenopausal women receiving DHEA, in whom DHEA also preserved aBMD [20,25]. In addition, our therapy may have acted through other mechanisms that are not currently understood. Recent data obtained in young women with AN have demonstrated a paradoxical increase in marrow fat in the peripheral skeletal despite a paucity of subcutaneous fat in these malnourished patients [53]. More work is needed to understand how the current therapy exerts its skeletal effects and whether it is due to altering the fate of mesenchymal stem cells within bone marrow.
Both groups gained weight, which alone leads to improve ments in skeletal health in patients with AN [4]. Given this fact, we call attention to our weight-adjusted results. The combination of DHEA + COC was effective in preserving skeletal health even after controlling for weight gain, whereas the placebo group exhibited bone losses. Serum estradiol concentrations rose in the placebo group to twice the baseline levels by 18 months, likely because of recovery from AN and increased weight/body fat. In the DHEA + COC arm (who received ethinyl estradiol), serum estradiol fell below baseline for the trial duration, reflecting probable suppression of ovarian estradiol production after receipt of exogenous gonadal hormones [54,55]. Our serum estradiol measure ments likely detected only endogenous 17β-estradiol, not ethinyl estradiol provided by the COC tested in this study.
One concern regarding androgen therapy is the potential development of adverse cosmetic effects, including hirsutism and acne. Despite increased DHEA-S and free testosterone in the DHEA + COC arm, no group differences in androgenic signs were seen. The observed rise in SHBG may have mitigated potential androgenic effects. Safety laboratory studies also remained stable.
Study limitations should be acknowledged. In this challenging population of patients, we found a high refusal rate, which may have skewed our sample toward young women with less refractory AN. These subjects also had a less severe degree of malnutrition. Although all subjects remained amenorrheic at the start of the study, delays between study recruitment and study initiation may have been associated with interim weight gain. However, our study cohort is representative of adolescents and young women seen routinely in our clinical practice and adolescent specialty practices throughout the country. Thus, our results are generalizable to a more “typical,” less severely ill patient. Our sample was limited to skeletally mature young women [56]; results may not be applicable to growing adolescents. Measurements of aBMD are 2-dimensional and do not yield information regarding skeletal strength or microarchitecture. However, DXA measurements are the current clinical standard of care for assessing skeletal health. Hip aBMD was chosen as the primary study outcome before the publication of recommendations regarding limitations of measuring hip aBMD [57]. The effects of DHEA + COC on the peripheral skeleton, a common fracture site among youth, are unknown and deserve further study. Despite these limitations, our randomized, controlled trial is the first to test the current regimen and has many strengths. We explored a novel therapy that is widely available and able to be administered orally, unlike other therapies that require subcutaneous or parenteral administration. Importantly, our safety data show that the treatment was well tolerated and acceptable to subjects, in addition to being effective.
In summary, in young women with AN who are at increased risk of low BMD and fracture, a combination regimen of oral DHEA + COC appears to be safe and effective for preserving aBMD, whereas placebo therapy led to de creases in aBMD. Measures of clinical and biochemical parameters showed that the DHEA + COC intervention was safe. Given the striking prevalence of AN, these data suggest that the combination of DHEA + COC holds promise as a safe and useful short-term therapy to prevent aBMD loss in these young women while instituting aggressive treatment of the underlying eating disorder and its skeletal and many non-skeletal complications. Further information will eventually come forward from future trials as to the mechanism of action of this regimen’s bone-protective effects.
Acknowledgment
We wish to acknowledge the excellent technical assistance of Julie Ringelheim, Julia Brown, Caitlin Stone, Kristen van der Veen, Nicolle Quinn, Jamie Nydegger, Yailka Cardenas, and Diane DiFabio; the Clinical and Translational Study Unit nurses; and our patients, who made this research possible.
Funding
Funding received from National Institute of Child Health and Human Development R01 HD043869, National Institute of Child Health and Human Development K23 HD060066, Children’s Hospital Boston Clinical and Translational Study Unit, and Department of Defense, US Army Bone Health and Military Readiness Program.
Footnotes
Author contributions: CMG, HF, CR, and ML designed the research. AD, CG, and CMG conducted the research. AD, CG, HF, and CMG performed data management and data analyses. CR and ML gave additional scientific input.
Manuscript preparation: The manuscript has been read and approved by all authors. The first draft of the manuscript was prepared by Dr DiVasta; all authors contributed to manuscript revision and the writing of the final paper. No author was paid to produce the manuscript.
Conflict of Interest
Dr Gordon is Co-director of the Harvard Clinical Investigation Training Program, sponsored in part by Pfizer and Merck. No funding for our study was provided by these entities.
REFERENCES
- 1.Treasure J, Claudino AM, Zucker N. Eating disorders. Lancet. 2010;375:583–593. doi: 10.1016/S0140-6736(09)61748-7. [DOI] [PubMed] [Google Scholar]
- 2.Bachrach LK, Guido D, Katzman D, et al. Decreased bone density in adolescent girls with anorexia nervosa. Pediatrics. 1990;86:440–447. [PubMed] [Google Scholar]
- 3.Rigotti NA, Neer RM, Skates SJ, et al. The clinical course of osteoporosis in anorexia nervosa. A longitudinal study of cortical bone mass. Jama. 1991;265:1133–1138. [PubMed] [Google Scholar]
- 4.Gordon CM, Grace E, Emans SJ, et al. Effects of oral dehydroepiandrosterone on bone density in young women with anorexia nervosa: a randomized trial. J Clin Endocrinol Metab. 2002;87:4935–4941. doi: 10.1210/jc.2002-020545. [DOI] [PubMed] [Google Scholar]
- 5.Golden NH, Lanzkowsky L, Schebendach J, et al. The effect of estrogen-progestin treatment on bone mineral density in anorexia nervosa. J Pediatr Adolesc Gynecol. 2002;15:135–143. doi: 10.1016/s1083-3188(02)00145-6. [DOI] [PubMed] [Google Scholar]
- 6.Liu SL, Lebrun CM. Effect of oral contraceptives and hormone replacement therapy on bone mineral density in premenopausal and perimenopausal women: a systematic review. Br J Sports Med. 2006;40:11–24. doi: 10.1136/bjsm.2005.020065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Munoz MT, Morande G, Garcia-Centenera JA, et al. The effects of estrogen administration on bone mineral density in adolescents with anorexia nervosa. Eur J Endocrinol. 2002;146:45–50. doi: 10.1530/eje.0.1460045. [DOI] [PubMed] [Google Scholar]
- 8.Klibanski A, Biller BM, Schoenfeld DA, et al. The effects of estrogen administration on trabecular bone loss in young women with anorexia nervosa. J Clin Endocrinol Metab. 1995;80:898–904. doi: 10.1210/jcem.80.3.7883849. [DOI] [PubMed] [Google Scholar]
- 9.Seeman E, Szmukler GI, Formica C, et al. Osteoporosis in anorexia nervosa: the influence of peak bone density, bone loss, oral contraceptive use, and exercise. J Bone Miner Res. 1992;7:1467–1474. doi: 10.1002/jbmr.5650071215. [DOI] [PubMed] [Google Scholar]
- 10.Strokosch GR, Friedman AJ, Wu SC, et al. Effects of an oral contraceptive (norgestimate/ethinyl estradiol) on bone mineral density in adolescent females with anorexia nervosa: a double-blind, placebo-controlled study. J Adolesc Health. 2006;39:819–827. doi: 10.1016/j.jadohealth.2006.09.010. [DOI] [PubMed] [Google Scholar]
- 11.Bonjour JP, Theintz G, Buchs B, et al. Critical years and stages of puberty for spinal and femoral bone mass accumulation during adolescence. J Clin Endocrinol Metab. 1991;73:555–563. doi: 10.1210/jcem-73-3-555. [DOI] [PubMed] [Google Scholar]
- 12.Orentreich N, Brind JL, Rizer RL, et al. Age changes and sex differences in serum dehydroepiandrosterone sulfate concentrations throughout adulthood. J Clin Endocrinol Metab. 1984;59:551–555. doi: 10.1210/jcem-59-3-551. [DOI] [PubMed] [Google Scholar]
- 13.Labrie F, Belanger A, Cusan L, et al. Marked decline in serum concentrations of adrenal C19 sex steroid precursors and conjugated androgen metabolites during aging. J Clin Endocrinol Metab. 1997;82:2396–2402. doi: 10.1210/jcem.82.8.4160. [DOI] [PubMed] [Google Scholar]
- 14.Taelman P, Kaufman JM, Janssens X, et al. Persistence of increased bone resorption and possible role of dehydroepiandrosterone as a bone metabolism determinant in osteoporotic women in late post-menopause. Maturitas. 1989;11:65–73. doi: 10.1016/0378-5122(89)90121-7. [DOI] [PubMed] [Google Scholar]
- 15.Haden ST, Glowacki J, Hurwitz S, et al. Effects of age on serum dehydroepiandrosterone sulfate, IGF-I, and IL-6 levels in women. Calcif Tissue Int. 2000;66:414–418. doi: 10.1007/s002230010084. [DOI] [PubMed] [Google Scholar]
- 16.Steinberg KK, Freni-Titulaer LW, DePuey EG, et al. Sex steroids and bone density in premenopausal and perimenopausal women. J Clin Endocrinol Metab. 1989;69:533–539. doi: 10.1210/jcem-69-3-533. [DOI] [PubMed] [Google Scholar]
- 17.Gordon CM, LeBoff MS, Glowacki J. Adrenal and gonadal steroids inhibit IL-6 secretion by human marrow cells. Cytokine. 2001;16:178–186. doi: 10.1006/cyto.2001.0962. [DOI] [PubMed] [Google Scholar]
- 18.Wang L, Wang YD, Wang WJ, et al. Differential regulation of dehydroepiandrosterone and estrogen on bone and uterus in ovariectomized mice. Osteoporos Int. 2009;20:79–92. doi: 10.1007/s00198-008-0631-1. [DOI] [PubMed] [Google Scholar]
- 19.Wang L, Wang YD, Wang WJ, et al. Dehydroepiandrosterone improves murine osteoblast growth and bone tissue morphometry via mitogen-activated protein kinase signaling pathway independent of either androgen receptor or estrogen receptor. J Mol Endocrinol. 2007;38:467–479. doi: 10.1677/jme.1.02173. [DOI] [PubMed] [Google Scholar]
- 20.Labrie F, Diamond P, Cusan L, et al. Effect of 12-month dehydroepiandrosterone replacement therapy on bone, vagina, and endometrium in postmenopausal women. J Clin Endocrinol Metab. 1997;82:3498–3505. doi: 10.1210/jcem.82.10.4306. [DOI] [PubMed] [Google Scholar]
- 21.Jankowski CM, Gozansky WS, Kittelson JM, et al. Increases in bone mineral density in response to oral dehydroepiandrosterone replacement in older adults appear to be mediated by serum estrogens. J Clin Endocrinol Metab. 2008;93:4767–4773. doi: 10.1210/jc.2007-2614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nair KS, Rizza RA, O’Brien P, et al. DHEA in elderly women and DHEA or testosterone in elderly men. N Engl J Med. 2006;355:1647–1659. doi: 10.1056/NEJMoa054629. [DOI] [PubMed] [Google Scholar]
- 23.Weiss EP, Shah K, Fontana L, et al. Dehydroepiandrosterone replacement therapy in older adults: 1-and 2-y effects on bone. Am J Clin Nutr. 2009;89:1459–1467. doi: 10.3945/ajcn.2008.27265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.von Muhlen D, Laughlin GA, Kritz-Silverstein D, et al. Effect of dehydroepiandrosterone supplementation on bone mineral density, bone markers, and body composition in older adults: the DAWN trial. Osteoporos Int. 2008;19:699–707. doi: 10.1007/s00198-007-0520-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Baulieu EE, Thomas G, Legrain S, et al. Dehydroepiandrosterone (DHEA), DHEA sulfate, and aging: contribution of the DHEAge Study to a sociobiomedical issue. Proc Natl Acad Sci U S A. 2000;97:4279–4284. doi: 10.1073/pnas.97.8.4279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Morales AJ, Haubrich RH, Hwang JY, et al. The effect of six months treatment with a 100 mg daily dose of dehydroepiandrosterone (DHEA) on circulating sex steroids, body composition and muscle strength in age-advanced men and women. Clin Endocrinol (Oxf) 1998;49:421–432. doi: 10.1046/j.1365-2265.1998.00507.x. [DOI] [PubMed] [Google Scholar]
- 27.Zumoff B, Walsh BT, Katz JL, et al. Subnormal plasma dehydroisoandrosterone to cortisol ratio in anorexia nervosa: a second hormonal parameter of ontogenic regression. J Clin Endocrinol Metab. 1983;56:668–672. doi: 10.1210/jcem-56-4-668. [DOI] [PubMed] [Google Scholar]
- 28.Gordon CM, Goodman E, Emans SJ, et al. Physiologic regulators of bone turnover in young women with anorexia nervosa. J Pediatr. 2002;141:64–70. doi: 10.1067/mpd.2002.125003. [DOI] [PubMed] [Google Scholar]
- 29.Gordon CM, Grace E, Emans SJ, et al. Changes in bone turnover markers and menstrual function after short-term oral DHEA in young women with anorexia nervosa. J Bone Miner Res. 1999;14:136–145. doi: 10.1359/jbmr.1999.14.1.136. [DOI] [PubMed] [Google Scholar]
- 30.Raisz LG, Wiita B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37–43. doi: 10.1210/jcem.81.1.8550780. [DOI] [PubMed] [Google Scholar]
- 31.Kelly TL. Bone mineral density reference databases for American men and women. J Bone Miner Res. 1990;5:S249. [Google Scholar]
- 32.Looker AC, Wahner HW, Dunn WL, et al. Proximal femur bone mineral levels of US adults. Osteoporos Int. 1995;5:389–409. doi: 10.1007/BF01622262. [DOI] [PubMed] [Google Scholar]
- 33.Kelly TL, Specker B, Binkely N. Pediatric BMD reference database for US white children. Bone. 2005;36:S30. [Google Scholar]
- 34.Gundberg CM, Hauschka PV, Lian JB, et al. Osteocalcin: isolation, characterization, and detection. Methods Enzymol. 1984;107:516–544. doi: 10.1016/0076-6879(84)07036-1. [DOI] [PubMed] [Google Scholar]
- 35.Ruppert D, Wand WP, Carroll RJ. Semiparametric regression. Cambridge, UK: Cambridge University Press; 2003. [Google Scholar]
- 36.Gadbury GL, Coffey CS, Allison DB. Modern statistical methods for handling missing repeated measurements in obesity trial data: beyond LOCF. Obes Rev. 2003;4:175–184. doi: 10.1046/j.1467-789x.2003.00109.x. [DOI] [PubMed] [Google Scholar]
- 37.Fong H, DiVasta AD, DiFabio D, et al. Prevalence and predictors of abnormal liver enzymes in young women with anorexia nervosa. J Pediatrics. 2008 doi: 10.1016/j.jpeds.2008.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Grinspoon S, Thomas L, Miller K, et al. Effects of recombinant human IGF-I and oral contraceptive administration on bone density in anorexia nervosa. J Clin Endocrinol Metab. 2002;87:2883–2891. doi: 10.1210/jcem.87.6.8574. [DOI] [PubMed] [Google Scholar]
- 39.Bosy-Westphal A, Brabant G, Haas V, et al. Determinants of plasma adiponectin levels in patients with anorexia nervosa examined before and after weight gain. Eur J Nutr. 2005;44:355–359. doi: 10.1007/s00394-005-0533-3. [DOI] [PubMed] [Google Scholar]
- 40.Hebebrand J, Blum WF, Barth N, et al. Leptin levels in patients with anorexia nervosa are reduced in the acute stage and elevated upon short-term weight restoration. Mol Psychiatry. 1997;2:330–334. doi: 10.1038/sj.mp.4000282. [DOI] [PubMed] [Google Scholar]
- 41.Housova J, Anderlova K, Krizova J, et al. Serum adiponectin and resistin concentrations in patients with restrictive and binge/purge form of anorexia nervosa and bulimia nervosa. J Clin Endocrinol Metab. 2005;90:1366–1370. doi: 10.1210/jc.2004-1364. [DOI] [PubMed] [Google Scholar]
- 42.Baranowska B, Wasilewska-Dziubinska E, Radzikowska M, et al. Neuropeptide Y, galanin, and leptin release in obese women and in women with anorexia nervosa. Metabolism. 1997;46:1384–1389. doi: 10.1016/s0026-0495(97)90136-0. [DOI] [PubMed] [Google Scholar]
- 43.Heer M, Mika C, Grzella I, et al. Changes in bone turnover in patients with anorexia nervosa during eleven weeks of inpatient dietary treatment. Clin Chem. 2002;48:754–760. [PubMed] [Google Scholar]
- 44.Gordon CM. Leptin and the skeleton—where is the fat? Metabolism. 2011;60:1203–1206. doi: 10.1016/j.metabol.2011.05.020. [DOI] [PubMed] [Google Scholar]
- 45.Conroy R, Girotra M, Shane E, et al. Leptin administration does not prevent the bone mineral metabolism changes induced by weight loss. Metabolism. 2011;60:1222–1226. doi: 10.1016/j.metabol.2011.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sienkiewicz E, Magkos F, Aronis KN, et al. Long-term metreleptin treatment increases bone mineral density and content at the lumbar spine of lean hypoleptinemic women. Metabolism. 2011;60:1211–1221. doi: 10.1016/j.metabol.2011.05.016. [DOI] [PubMed] [Google Scholar]
- 47.Chou SH, Chamberland JP, Liu X, et al. Leptin is an effective treatment for hypothalamic amenorrhea. Proc Natl Acad Sci U S A. 2011;108:6585–6590. doi: 10.1073/pnas.1015674108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Golden NH, Iglesias EA, Jacobson MS, et al. Alendronate for the treatment of osteopenia in anorexia nervosa: a randomized, double-blind, placebo-controlled trial. J Clin Endocrinol Metab. 2005;90:3179–3185. doi: 10.1210/jc.2004-1659. [DOI] [PubMed] [Google Scholar]
- 49.Miller KK, Grieco KA, Mulder J, et al. Effects of risedronate on bone density in anorexia nervosa. J Clin Endocrinol Metab. 2004;89:3903–3906. doi: 10.1210/jc.2003-031885. [DOI] [PubMed] [Google Scholar]
- 50.Soyka LA, Grinspoon S, Levitsky LL, et al. The effects of anorexia nervosa on bone metabolism in female adolescents. J Clin Endocrinol Metab. 1999;84:4489–4496. doi: 10.1210/jcem.84.12.6207. [DOI] [PubMed] [Google Scholar]
- 51.Soyka LA, Misra M, Frenchman A, et al. Abnormal bone mineral accrual in adolescent girls with anorexia nervosa. J Clin Endocrinol Metab. 2002;87:4177–4185. doi: 10.1210/jc.2001-011889. [DOI] [PubMed] [Google Scholar]
- 52.Caillot-Augusseau A, Lafage-Proust MH, Margaillan P, et al. Weight gain reverses bone turnover and restores circadian variation of bone resorption in anorexic patients. Clin Endocrinol (Oxf) 2000;52:113–121. doi: 10.1046/j.1365-2265.2000.00879.x. [DOI] [PubMed] [Google Scholar]
- 53.Ecklund K, Vajapeyam S, Feldman HA, et al. Bone marrow changes in adolescent girls with anorexia nervosa. J Bone Miner Res. 2010;25:298–304. doi: 10.1359/jbmr.090805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sullivan H, Furniss H, Spona J, et al. Effect of 21-day and 24-day oral contraceptive regimens containing gestodene (60 microg) and ethinyl estradiol (15 microg) on ovarian activity. Fertil Steril. 1999;72:115–120. doi: 10.1016/s0015-0282(99)00205-8. [DOI] [PubMed] [Google Scholar]
- 55.Fitzgerald C, Feichtinger W, Spona J, et al. A comparison of the effects of two monophasic low dose oral contraceptives on the inhibition of ovulation. Adv Contracept. 1994;10:5–18. doi: 10.1007/BF01986524. [DOI] [PubMed] [Google Scholar]
- 56.Chevalley T, Bonjour JP, Ferrari S, et al. The influence of pubertal timing on bone mass acquisition: a predetermined trajectory detectable five years before menarche. J Clin Endocrinol Metab. 2009;94:3424–3431. doi: 10.1210/jc.2009-0241. [DOI] [PubMed] [Google Scholar]
- 57.Gordon CM, Bachrach LK, Carpenter TO, et al. Dual energy x-ray absorptiometry interpretation and reporting in children and adolescents: the 2007 ISCD pediatric official positions. J Clin Densitom. 2008;11:43–58. doi: 10.1016/j.jocd.2007.12.005. [DOI] [PubMed] [Google Scholar]