Abstract
Human luteinizing hormone (hLH) and chorionic gonadotropin (hCG) act on the same receptor (LHCGR) but it is not known whether they elicit the same cellular and molecular response. This study compares for the first time the activation of cell-signalling pathways and gene expression in response to hLH and hCG. Using recombinant hLH and recombinant hCG we evaluated the kinetics of cAMP production in COS-7 and hGL5 cells permanently expressing LHCGR (COS-7/LHCGR, hGL5/LHCGR), as well as cAMP, ERK1/2, AKT activation and progesterone production in primary human granulosa cells (hGLC). The expression of selected target genes was measured in the presence or absence of ERK- or AKT-pathways inhibitors. In COS-7/LHCGR cells, hCG is 5-fold more potent than hLH (cAMP ED50: 107.1±14.3 pM and 530.0±51.2 pM, respectively). hLH maximal effect was significantly faster (10 minutes by hLH; 1 hour by hCG). In hGLC continuous exposure to equipotent doses of gonadotropins up to 36 hours revealed that intracellular cAMP production is oscillating and significantly higher by hCG versus hLH. Conversely, phospho-ERK1/2 and -AKT activation was more potent and sustained by hLH versus hCG. ERK1/2 and AKT inhibition removed the inhibitory effect on NRG1 (neuregulin) expression by hLH but not by hCG; ERK1/2 inhibition significantly increased hLH- but not hCG-stimulated CYP19A1 (aromatase) expression. We conclude that: i) hCG is more potent on cAMP production, while hLH is more potent on ERK and AKT activation; ii) hGLC respond to equipotent, constant hLH or hCG stimulation with a fluctuating cAMP production and progressive progesterone secretion; and iii) the expression of hLH and hCG target genes partly involves the activation of different pathways depending on the ligand. Therefore, the LHCGR is able to differentiate the activity of hLH and hCG.
Introduction
Luteinizing hormone (LH) and chorionic gonadotropin (CG) are heterodimeric glycoprotein hormones acting on the same receptor, the luteinizing hormone-chorionic gonadotropin receptor (LHCGR) [1], which is found as dimer/oligomer at the cell membrane [2]. LH is the physiological hormone in non-pregnant women, produced by the pituitary in a pulsatile fashion. LH binds to LHCGR on the granulosa cells surface, resulting in progesterone production, ovulation, luteinization and corpus luteum formation [3]. Moreover, LH stimulates androstenedione and testosterone production in theca cells. In the human ovary, androstenedione is aromatized to estrone by granulosa cells and finally converted to estradiol by 17-β-hydroxysteroid dehydrogenase type I, representing the system known as the two-cell-two-gonadotropin regulation of estrogen synthesis [3]. After the ovulation, LH supports the transient life span of the corpus luteum acting on the luteinized granulosa cells [3]. Conversely, CG is a hormone produced mainly by placental trophoblast cells during pregnancy in an increasing, non-pulsatile fashion [4]. Apart from equine CG (eCG), which mediates a predominant FSH-like activity [5], CG with exclusive, unique LH-like activity exists only in primates and its β-subunit gene is present in increasing copy number, according to the increasing structural complexity of placental implantation in the primate species [6].
Human LH (hLH) and CG (hCG) differ in their half-life (60–120 minutes for hLH, several hours for hCG [7]–[13] and in some structural features, such as the presence of a carboxyl terminal peptide (CTP) and the type and amount of glycosylation. Due to this heterogeneity and derivation from extractive preparations, gonadotropins have been difficult to quantify accurately in the past, and most in vitro experiments have been conducted using urinary hCG calibrated by in vivo bioassay against standard preparations expressed in activity units [14]. With the advent of recombinant gonadotropins, highly homogeneous and consistent r-hLH and r-hCG can be accurately quantified in molar terms [15] and used to compare their effects in vitro at exactly equimolar concentrations.
Being structurally different, it should be expected that hLH and hCG display different hormone-receptor interaction features and, consequently, might be not equivalent at molecular and cellular level. There are some hints that hLH and hCG may not have the same activity. Some residues of the LHCGR extracellular domain are indeed able to differentiate binding of hLH and hCG [16] and the human LHCGR can react differently to hLH and hCG when exon 10 is lacking (LHCGR-10) [17]. LHCGR-10 can bind either gonadotropin with similar affinity but cAMP production is drastically impaired upon hLH but not hCG stimulation [17], [18], suggesting that exon 10 of the LHCGR can distinguish between hLH and hCG. Whether this translates physiologically into preferential activation of different signal transduction pathways and, eventually, different cell responses, is not known.
Some LHCGR-dependent effects are mediated by the activation of the cyclic AMP-protein kinase A (cAMP/PKA) pathway, which stimulates progesterone production and has been associated to morphological changes [19] and apoptosis [20], [21] in granulosa cells. However, additional signalling pathways (e.g. AKT- and ERK1/2-pathways) are involved [1] in LHCGR-dependent events such as proliferation, differentiation and survival [22], for example via expression of EGF-like growth factors [23]. Lastly, aromatase expression and steroidogenic function via LHCGR activation are likely to involve cAMP/PKA, ERK1/2 and AKT pathways, all playing a crucial role in the final stages of maturation of human oocytes and follicles [24], [25].
While equivalence of hLH and hCG in activating the downstream signaling of LHCGR is presumed, this issue has never been investigated thoroughly so far. In this work, after confirming that extractive and recombinant gonadotropins are equivalent in essence as far as their effects in vitro are concerned, we evaluated the effects of hLH and hCG on the cell response by systematically assessing the activation of cAMP/PKA-, ERK1/2- and AKT-pathways in three validated in vitro models consisting of a COS-7 cell line permanently transfected with human LHCGR (COS-7/LHCGR) [18], the immortalized human granulosa cell line hGL5 [26] permanently transfected with the human LHCGR (hGL5/LHCGR) and human primary granulosa cells (hGLC) [27]. The COS-7/LHCGR cell system is a standardized model, constitutively expressing a fix number of LHCGR under the transcriptional regulation of the cytomegalovirus promoter [18]. Conversely, primary hGLC obtained from women undergoing assisted reproduction procedures naturally express the LHCGR, which is presumably subjected to “physiological” control of LHCGR gene transcription and different modulation of the desensitization mechanisms [28]. Finally, the hGL5 cell line derives from human granulosa cells immortalized by transformation with the E6 and E7 regions of human papillomavirus. Despite their steroidogenic activity, hGL5 cells do not naturally express gonadotropin receptors [26] and were permanently transfected with the human LHCGR for this study. These cell model were used to assess the kinetics of cAMP and progesterone production and to investigate some intracellular events following acute and chronic exposure to equipotent hLH and hCG doses. Remarkable differences in LH and hCG action were demonstrated.
Methods
A detailed description of the methods is available as Methods S1.
Recombinant and extractive gonadotropins
Human, highly purified recombinant LH (r-hLH; Luveris) and CG (r-hCG; Ovitrelle) were kindly provided by Merck-Serono S.p.A. (Rome, Italy). LH extracted from human pituitary (ex-hLH) and CG extracted from human pregnancy urine (ex-hCG) were purchased by Sigma-Aldrich (Sigma-Aldrich, St. Louis, MO).
Cell lines
A COS-7 cell line permanently transfected with LHCGR wild type (COS-7/LHCGR) was kindly provided by Prof. Gromoll (Centre for Reproductive Medicine and Andrology, University of Münster, Germany). The immortalized human granulosa cell line hGL5 [26] was permanently transfected by electroporation with LHCGR wild type. For stable transfection we used the pTracer vector (Invitrogen, Leek, The Netherlands), containing the cytomegalovirus promoter in front of the multiple cloning site and the green fluorescent protein reporter gene [18]. COS-7/LHCGR cells were cultured in DMEM supplemented with 10% FBS, 2 mM L-glutamine, 100 U/ml penicillin and 100 µg/ml streptomycin. This cell line, overexpressing the human LHCGR, was extensively validated previously [18]. hGL5/LHCGR cells were cultured in DMEM/F12 supplemented with 10% FBS, 2% Ultroser G, 2 mM L-glutamine, 100 U/ml penicillin and 100 µg/ml streptomycin. All cell lines were maintained in an incubator at 37°C and with 5% CO2.
Granulosa-lutein cell isolation and culture
Human primary granulosa-lutein cells (hGLC) were isolated from ovarian follicles of women undergoing oocyte retrieval for assisted reproduction technologies (ART). The cells from 3–4 different patients were pooled and collected using a 50% Percoll gradient (GE Healthcare, Little Chalfont, UK) following a procedure previously described [27], then cultured in a CO2 cell incubator. Before each experiment, granulosa cells were maintained in culture until day 6, to allow the recovery of the response to gonadotropins (Fig. S1).
Patients selection
Women undergoing ovarian stimulation for infertility due to tubal or male factor were included. Study approval was obtained from the local ethics committee and informed, written consent was obtained from each patient. (Ethics committee approval - dossier number: Pratica 161/11, date: 21 september 2011, Prot. n. 3186/C.E. Comitato Etico Provinciale di Modena). The patients had to match the following criteria: absence of endocrine abnormalities; absence of severe viral or bacterial infections; age between 25–45 years.
cAMP stimulation protocols
For cAMP stimulation a validated protocol was followed [27]. The COS-7/LHCGR, hGL5/LHCGR and granulosa cells were seeded in triplicate in 24-well plates for cAMP dose-response experiments and serum starved 12 hours before the experiments. Cells were stimulated using increasing doses of r-hLH, r-hCG, ex-hLH or ex-hCG as appropriate (ranging between 0.1 pM-1 mM) in the presence of 500 µM IBMX (Sigma-Aldrich). Total cAMP was measured after 3 hours incubation and the cAMP ED50 values for hLH and hCG were calculated. A total of 4 independent experiments were performed.
To evaluate the kinetics of response to continuous exposure to gonadotropins, time-course experiments were performed. The COS-7/LHCGR and hGLC were stimulated using the cAMP equipotent ED50 doses of r-hLH or r-hCG, for different times ranging between 5 minutes and 36 hours in the presence of IBMX. The intracellular cAMP was measured after each incubation. Cell viability was also evaluated [29]. A total of 3 independent experiments were performed.
The quantitative detection of cAMP was performed in triplicate by a competitive ELISA kit and the data were entered into a curve fitting software which extrapolates the cAMP concentration against a standard curve. The data were represented using a log regression analysis.
Immunofluorescence analysis of human granulosa cells
Immunofluorescence analysis was performed to evaluate the kinetics of receptor internalization resulting from continuous in vitro stimulation of hGLC by gonadotropins. Serum-starved hGLC were stimulated for different times with the ED50 dose of hLH or hCG. After stimulation, the cells were fixed and sequentially incubated with anti-LHCGR [30] and anti-ERK1/2 antibody to be observed in confocal microscopy. Negative and positive controls for the antibodies and non-permeabilized cells were also included.
Phospho-ERK1/2 and phospho-AKT stimulation and Western blot analysis
To evaluate the doses of r-hLH, r-hCG, ex-hLH or ex-hCG resulting in the maximum level of stimulation of ERK1/2- and AKT-pathway (EDMAX), dose-response experiments were performed. 12 hours serum-starved hGLC were stimulated for 15 minutes [31], [32] with increasing doses of r-hLH or r-hCG (ranging between 0.1 pM-1 mM), including negative controls. To compare the response to recombinant versus extractive gonadotropins also hGL5/LHCGR cells were used. In time-course experiments the cells were then stimulated over 1 hour with 100 pM r-hLH or r-hCG (EDMAX), previously obtained in dose-response experiments. Negative controls were included in each step of the time-course experiment. The cells were immediately lysed for protein extraction in 4°C-cold RIPA buffer added with protease and phosphatase inhibitors. A total of 4 independent experiments were performed by using each time a different pool of granulosa cells obtained from 3–4 different patients.
Phospho-ERK1/2 and phospho-AKT activation were evaluated by Western blot analysis after 12% SDS-PAGE. Equal protein loading was confirmed against total ERK1/2 after stripping the membranes. The signals were revealed by chemiluminescence, then acquired and semi-quantitatively evaluated by an image analysis system.
Stimulation for gene expression analysis
Serum-starved hGLC were stimulated for 12 hours with r-hLH or r-hCG EDMAX. One-hour pre-incubated samples with ERK1/2- or AKT-pathway inhibitors were also included. After stimulation, total RNA was extracted and an equal amount in cDNA synthesis reactions was used.
Real-time PCR analysis
Real-time PCR was performed in triplicates with the primers shown in Table 1. The ribosomal protein S7 (RPS7) gene was used as normalizer. The thermal cycling settings for all genes were the following: 45 cycles of 30 s of melting at 95°C followed by 10 s of annealing and extension at 60°C. Normalized gene expression was evaluated using the 2−ΔCt method [33] and expressed as fold increase over its unstimulated sample (basal level). A total of four experiments were performed.
Table 1. Primer sequences used in real-time PCR experiments.
| Gene | Oligonucleotide sequences | Product length (bp) | NCBI Ref. Sequence | Protein name |
| AREG | F:GACACCTACTCTGGGAAGCG | 121 | NM_001657.2 | Amphiregulin |
| R:AAGGCATTTCACTCACAGGG | ||||
| EREG | F:TACTGCAGGTGTGAAGTGGG | 139 | NM_001432.2 | Epiregulin |
| R:TGGAACCGACGACTGTGATA | ||||
| NRG1* | F:CCCCGATTGAAAGAGATGAA | 116 | NM_001159999.1 | Neuregulin 1 |
| R:TTCCCATTCTTGAACCACTTG | ||||
| CYP19A1 | F:CCCTTCTGCGTCGTGTCAT | 86 | NM_000103.3 | Aromatase |
| R:GATTTTAACCACGATAGCACTTTCG | ||||
| RPS7** | F: AATCTTTGTTCCCGTTCCTCA | 135 | NM_001011.3 | Ribosomal protein S7 |
| R: CGAGTTGGCTTAGGCAGAA |
Primer chosen to include all NRG1 transcript variants, verified by NCBI BLAST.
Endogenous control gene.
Statistical analysis
Each value obtained from hLH and hCG-stimulated cells was normalized for the corresponding control value. In time-course experiments for ERK1/2 and AKT, the semi-quantitative evaluations were graphically expressed in relative units. Data are expressed as means ± SEM. Mann Whitney's U-test, unpaired T-test or two-way analysis of variance were performed where appropriate. In time-course experiments, each data-set was verified with D'Agostino and Pearson normality test (alpha = 0.05). Values were considered statistically significant for P<0.05.
Results
Short-term kinetics of cAMP production
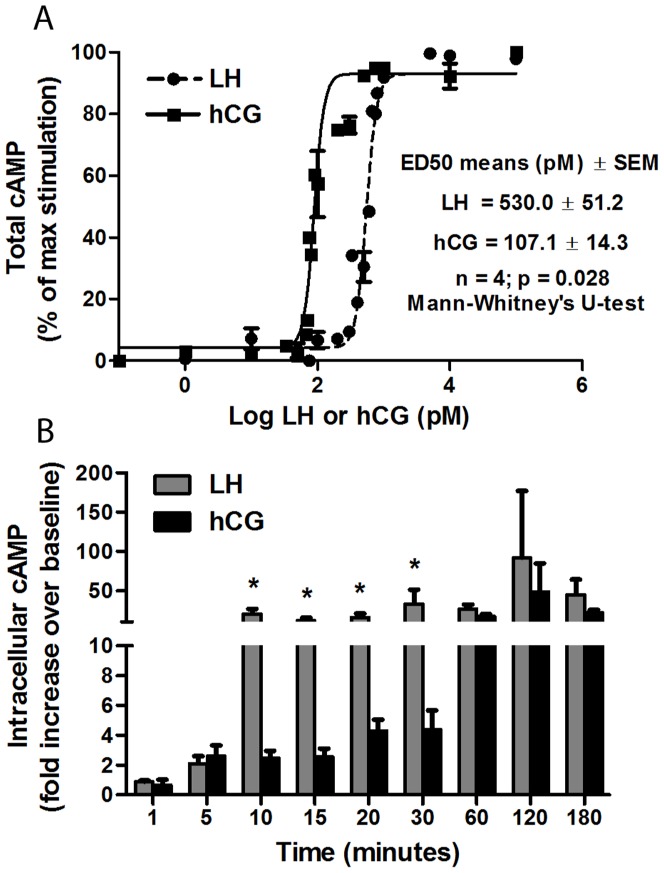
To evaluate whether r-hLH and r-hCG stimulation of the human LHCGR results in the same kinetics of cAMP production we performed dose-response and time course-experiments using the COS-7/LHCGR cell line. This model allows testing the early effects of the two gonadotropins in “steady” receptor conditions, since no regulation of LHCGR gene transcription occurs in these stably transfected cells constitutively expressing the LHCGR under the control of the cytomegalovirus promoter. Dose-response studies were performed by measuring total cAMP after 3 hours of stimulation with increasing doses of hCG or hLH in the presence of IBMX (500 µM). A significant difference in the kinetics of total cAMP production was found, with dose-response curves shifted significantly to the left in the case of hCG. As shown in Fig. 1, the ED50 of r-hCG (107.1±14. 3 pM) resulted approximately 5 times lower than the ED50 of r-hLH (530.0±51.2 pM) (n = 4, p<0.05, Fig. 1A). Since recombinant gonadotropins could differ from pituitary hLH and urinary hCG in the extent and type of glycosylation this could affect cAMP production, making the results obtained with “artificial” gonadotropins potentially different from those with “natural” gonadotropins. To assess this, a comparison between both type of gonadotropins was performed in different cell models (Fig. S2). The results obtained were similar, indicating that recombinant and extractive gonadotropins result in comparable effects on activation of signaling pathways, as otherwise well known from clinical practice.
Figure 1. Dose-response and time-course experiments.
a. Dose-response experiment to r-hLH and r-hCG in COS7/LHCGR in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours of incubation and the results of four independent experiments were plotted (Mean±SEM). b. Time-course experiment performed by continuous incubation of COS7/LHCGR for different time-points in the presence of 500 µM IBMX and gonadotropins at ED50 doses (500 pM r-hLH; 100 pM r-hCG). Intracellular cAMP was measured (n = 3; Mean±SEM). Asterisk indicates the significant differences of hLH vs hCG (t-test; p<0.05).
The ED50 doses of r-hLH and r-hCG were then used in time-course experiments, based on the measurement of intracellular cAMP in COS-7/LHCGR cells stimulated for up to 180 minutes by r-hLH (500 pM) or r-hCG (100 pM), in the presence of 500 µM IBMX (500 µM). Using equipotent doses, accumulation of intracellular cAMP by r-hLH resulted significantly faster, with maximal activation achieved in 10 minutes, while, by r-hCG, the same levels of maximal stimulation were attained only after 60 minutes of stimulation (Fig. 1B). Maximal intracellular cAMP accumulation was approximately 20–30 times higher than basal levels in untreated cells and was equally reached in both r-hLH- and r-hCG-stimulated cells, confirming that the ED50 doses were indeed equipotent in acute activation. After the first 60 min of continuous exposure to r-hLH or r-hCG intracellular cAMP concentrations remained elevated (Fig. 1B). Therefore, equimolar concentrations r-hLH and r-hCG possess significantly different in vitro potency (in terms of cAMP) and equipotent concentrations of r-hLH and r-hCG stimulate intracellular cAMP accumulation with significantly different kinetics.
When repeated in hGLC cells the cAMP dose-response and time course experiments confirmed the same difference between r-hLH and r-hCG (Fig. S3).
Long-term kinetics of cAMP and progesterone production in hGLC continuously exposed to hLH or hCG
hGLC obtained at oocyte pickup in ART programs are a well-known, widely used model for the study of cellular response to gonadotropins [27] and constitute a more physiological model than COS-7/LHCGR cells, since they naturally express the LHCGR and display physiological control of the receptor gene transcription and downregulation in vitro [31]. In preliminary experiments we determined that hGLC do not respond significantly, in terms of cAMP production, to r-hLH or r-hCG for up to four days in culture but respond optimally on day 6 (Fig. S1), as previously described for FSH as well [27]. To compare the kinetics of intracellular cAMP beyond the first 60 min hGLC were stimulated on culture day 6 with ED50 doses of r-hLH or r-hCG for up to six hours. Intracellular cAMP levels decreased after the first three hours but were not extinguished by 360 min (Fig. S4).
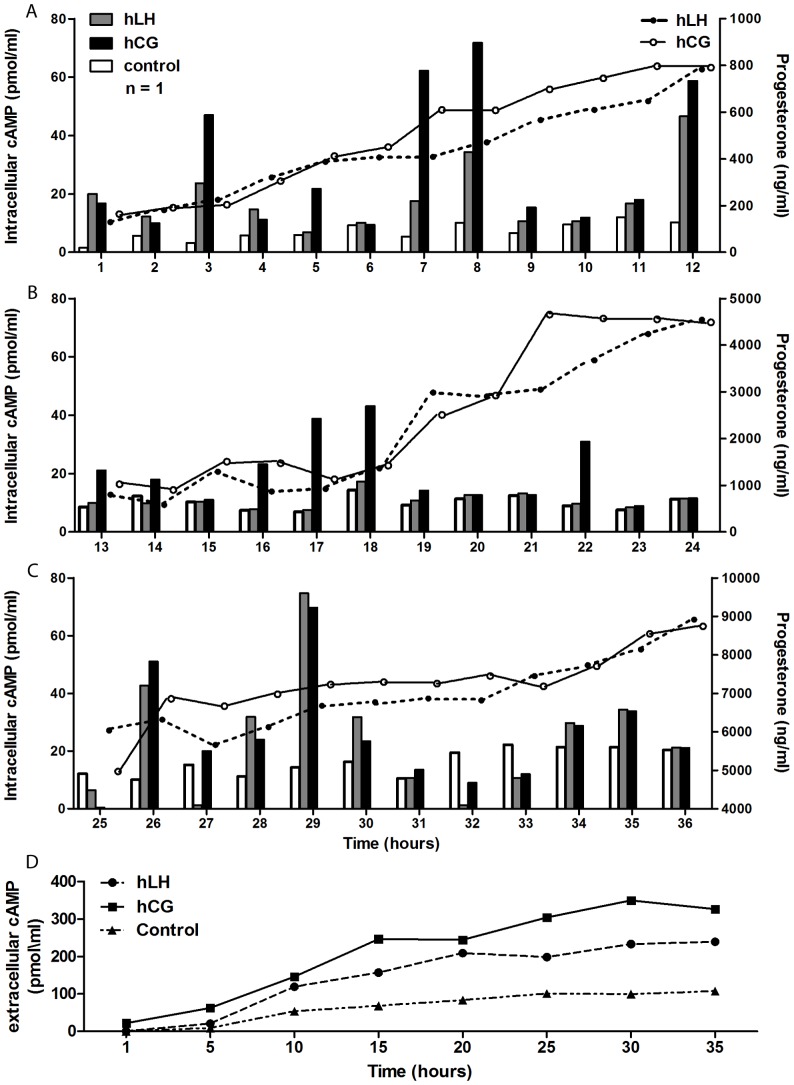
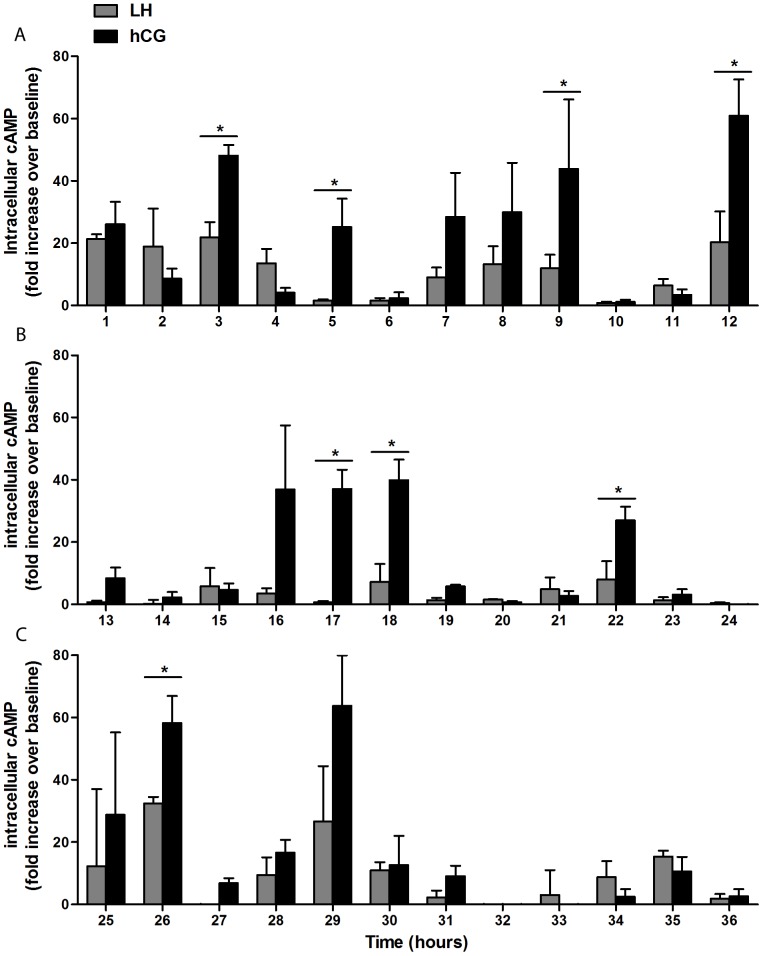
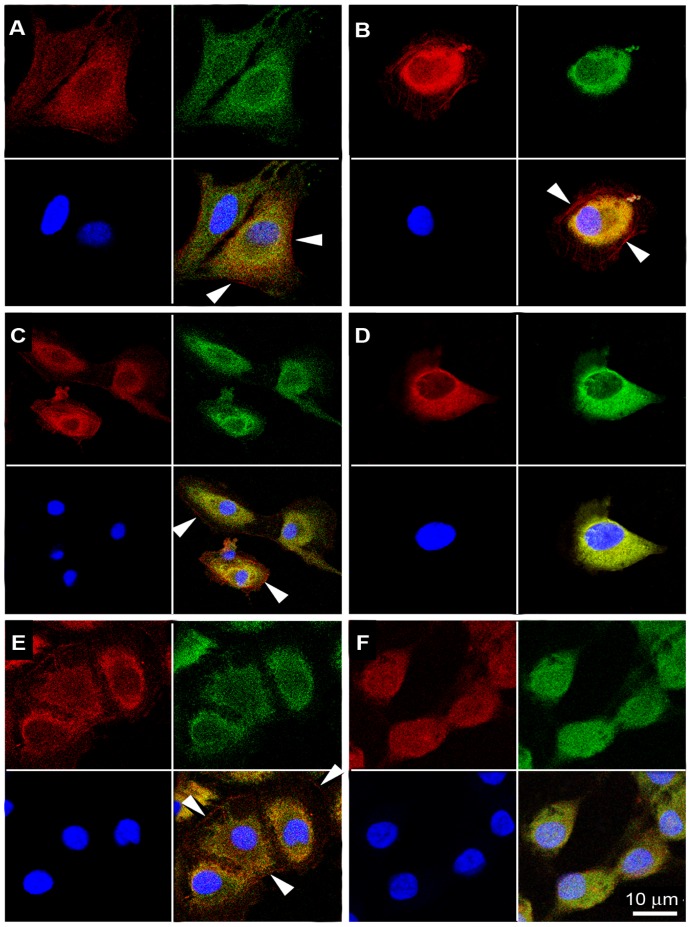
We therefore performed long-term stimulation experiments in which day 6 hGLC were exposed to constant ED50 doses of r-hLH (500 pM) or r-hCG (100 pM) for up to 36 hours. As shown in Fig. 2, continuous exposure to r-hLH or r-hCG resulted in repetitive, cyclic rises and falls of intracellular cAMP with peaks approximately every 4–5 hours, while cAMP remained substantially constant in the unstimulated control cells. Moreover, cAMP concentrations during the peaks were often significantly higher upon r-hCG stimulation, in spite of the five-fold lower molar concentrations used, for the first 36 hours (Fig. 3). Intracellular cAMP response tended to extinguish after 30 hours for both r-hLH and r-hCG stimulations. The fluctuations in cellular cAMP response to constant r-hLH or r-hCG doses resulted in progressive, linear accumulation of progesterone in the supernatant, which increased 45-times, from about 200 ng/ml to about 9000 ng/ml, by 36 hours (Fig. 2) for both r-hLH and r-hCG stimulations in spite of overall lower cAMP level stimulated by r-hLH. r-hLH and r-hCG concentrations in the supernatant (measured by an immunometric assay) and cell viability (measured by MTT assay) remained constant during the entire stimulation period (data not shown). The fluctuations in intracellular cAMP were accompanied a constant increase of extracellular cAMP measured in the culture medium (Fig. 2D), suggesting the existence of a mechanism regulating the efflux of the second messenger outside the cell membrane of viable hGLC cells, according to previous observation in a wide variety of cell types [34]. The mechanism of the cyclic hGLC response to constant r-hLH/r-hCG exposure requires a set of ad hoc experiments and was not further explored in this setting. However, immunofluorescence staining revealed membrane localization of the LHCGR in unstimulated hGLC at each time-point, while the receptor was visible at the membrane after one hour of stimulation by r-hLH or r-hCG but prevalently intracellular after 15 and 24 hours, suggesting possible internalization (Fig. 4). In addition, stimulated cells tended to assume a typical rounded shape with time, a phenomenon previously described in granulosa cells exposed to FSH [35].
Figure 2. Long-term kinetics of intracellular cAMP production and extracellular progesterone and cAMP release in a primary hGLC sample continuously exposed to r-hLH or r-hCG.
Absolute values of intracellular cAMP (bars) and extracellular progesterone (lines) production by hGLC cultured in the presence IBMX (500 µM) under continuous stimulation by hLH (500 pM) or hCG (100 pM) for up to 36 hours: (a) 0–12 hours, (b) 13–24 hours, (c) 25–36 hours. cAMP basal level of the unstimulated control cells is also shown for each time-point. d. Measurement of extracellular cAMP, released in culture medium. One representative of three independent measurement is shown for each graph. Please notice the different progesterone scale on the Y axis on right side.
Figure 3. Long-term kinetics of intracellular cAMP production in hGLC continuously exposed to r-hLH or r-hCG.
hGLC were cultured in the presence IBMX (500 µM) under continuous stimulation by r-hLH (500 pM) or r-hCG (100 pM) for up to 36 hours: (a) 0–12 hours, (b) 13–24 hours, (c) 25–36 hours. Results are means±SEM (n = 3). Asterisks indicate significant differences between hCG and hLH at the given time point (n = 3; t-test; p<0.05).
Figure 4. Cellular localization of LHCGR and morphological changes of hGLC under continuous exposure to hLH 500 (pM) by confocal microscopy.
a. Unstimulated cells (control) after 1 hour. b. hLH-treated cells after 1 hour. c. Unstimulated cells after 15 hours. d. LHCGR sequestration from cell surface in hLH-treated hGLC, after 15 hours. e. Unstimulated cells after 24 hours. f. Cell-rounding in hLH-treated cells after 24 hours. LHCGR is labeled in red (Tritc), the cytoplasmic marker ERK1/2 in green (Fitch) and cell nuclei marker (DAPI) in blue. The merging of the three images is in the lower right plate of each panel. Images are from one experiment and are representative of three independent experiments with similar results. White arrows indicated LHCGR; Scale bar: 10 µm. Western blot control for the anti-LHCGR antibody and non-permeabilized cells control for immunofluorescence staining were also included (Fig. S5).
Acute effects of hLH and hCG on the ERK1/2 and AKT pathways in hGLC
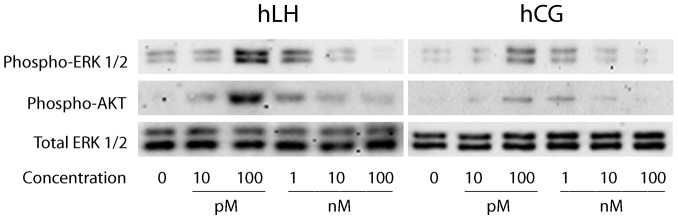
We next examined the activation of the ERK1/2- and AKT-pathways in hGLC. First, we performed dose-response experiments in which phospho-ERK1/2 and phospho-AKT were detected by Western blotting of protein extracts obtained from day 6 hGLC stimulated for 15 minutes with different doses of r-hCG or r-hLH in the pM-nM range. As shown in Fig. 5, the highest activation of both ERK1/2 and AKT was reached with 100 pM of either gonadotropin. Interestingly, r-hLH appeared more effective than r-hCG at all doses tested. Differently from what was observed for cAMP, an equipotent (ED50) dose of the two gonadotropins on ERK1/2 and AKT activation could not be determined. Therefore, the maximally stimulating 100 pM dose was used for both r-hLH and r-hCG in further experiments.
Figure 5. Dose-response experiment evaluating the maximal phospho-ERK1/2 and phospho-AKT activation in hGLC by Western blotting.
The cells were stimulated for 15 minutes by different r-hLH or r-hCG doses and the phospho-ERK1/2 and phospho-AKT signals were normalized for total ERK. The image is representative of 3 independent experiments.
As shown for cAMP dose-response experiments (Fig. S2), a comparison between recombinant and extractive gonadotropins was performed in hGL5/LHCGR cells by Western blot, to assess whether the carbohydrate structure related to recombinant gonadotropins could affect the phosphorylation rate of ERK1/2 and AKT (Fig. S6). The results obtained were similar, indicating that recombinant and extractive gonadotropins lead to essentially the same effects.
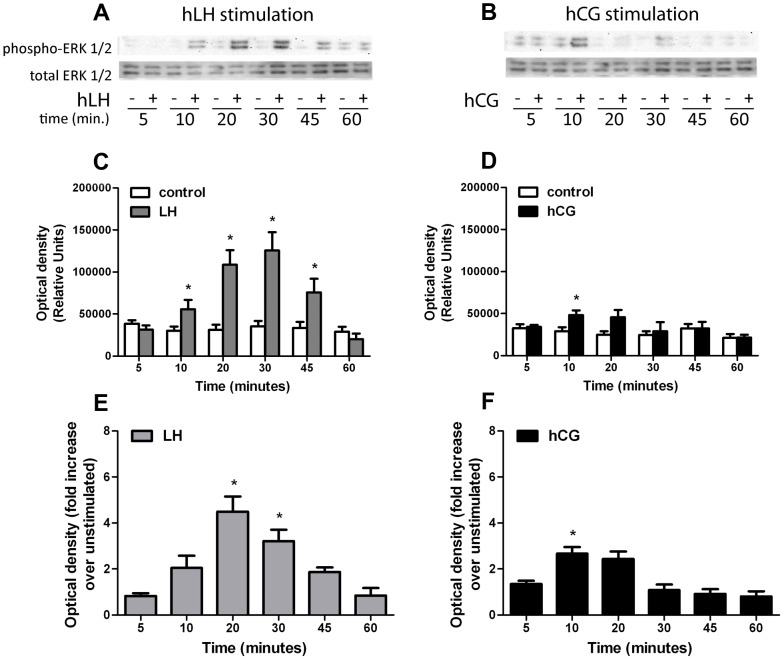
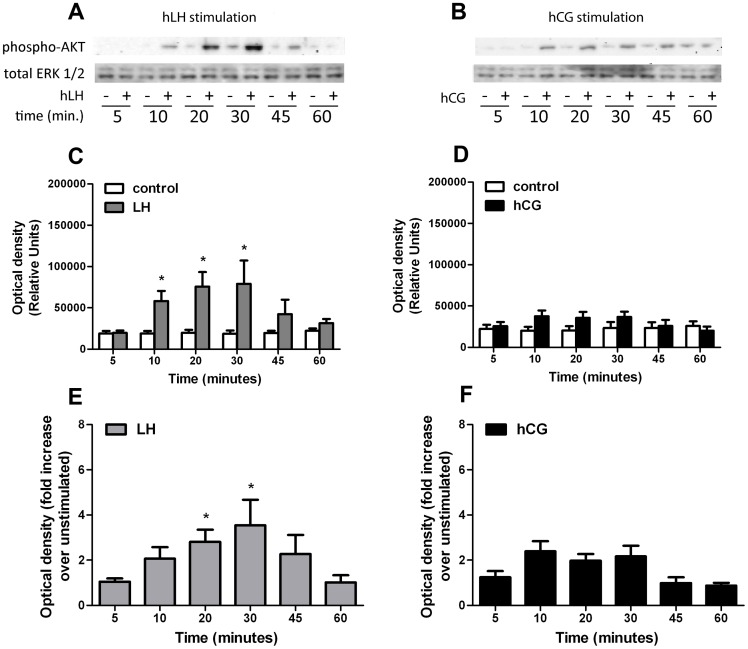
The kinetics of ERK1/2 and AKT activation were studied in time-course experiments using day 6 hGLC in a time range of 5–60 min. Phospho-ERK1/2 and phospho-AKT signals were detected by Western blot and semiquantified by image analysis. Stimulation with 100 pM of r-hLH resulted in a strong, significant activation of ERK1/2 between 10 and 45 minutes, while 100 pM of r-hCG induced a much weaker and short-lived stimulation, reaching significance only at 10 min (Fig. 6). Similar effects were observed for phospho-AKT. Here again r-hLH (100 pM) provoked a significant increase in phospho-AKT between 10 to 30 minutes, while the r-hCG (100 pM) stimulation appeared to be minimal and did not reach statistical significance at any time point (Fig. 7). The ERK1/2 and AKT activation tended to extinguish within 60 minutes for both r-hLH and r-hCG stimulations.
Figure 6. Time-course analysis of the phospho-ERK1/2 activation in hGLC under 100 pM hLH or 100 pM hCG stimulation.
a. Comparison of phospho-ERK1/2 activation in hLH-stimulated vs unstimulated (control) at different time-points, by Western blotting (image is representative of 4 independent experiments; total ERK as normalizer). b. Comparison of phospho-ERK1/2 activation in hCG-stimulated vs unstimulated (control) at different time-points, by Western blotting (image is representative of 4 independent experiments; total ERK as normalizer). c, d. Relative semi-quantification of the optical density representing phospho-ERK1/2 activation (shown in figs. 6A,B) stimulated by (c) hLH or (d) hCG, compared to unstimulated (Mean±SEM; n = 4; * = significant vs unstimulated; t-test; p<0.05). e. Normalization of the phospho-ERK1/2 signals measured in hLH-stimulated samples (represented as relative units in Fig. 6C) over each unstimulated (Mean±SEM; n = 4; * = significant vs unstimulated; two-way analysis of variance; p<0.05). f. Normalization of the phospho-ERK1/2 signals measured in hCG-stimulated samples (represented as relative units in Fig. 6D) over each unstimulated (Mean±SEM; n = 4; * = significant vs unstimulated; two-way analysis of variance; p<0.05).
Figure 7. Time-course analysis of the phospho-AKT activation in hGLC under 100 pM hLH or 100 pM hCG stimulation.
a. Comparison of phospho-AKT activation in hLH-stimulated vs unstimulated (control) at different time-points, by Western blotting (image is representative of 4 independent experiments; total ERK as normalizer). b. Comparison of phospho-AKT activation in hCG-stimulated vs unstimulated (control) at different time-points, by Western blotting (image is representative of 4 independent experiments; total ERK as normalizer). c, d. Relative semi-quantification of the optical density representing phospho-AKT activation (shown in figs. 7A,B) stimulated by (C) hLH or (D) hCG, compared to unstimulated (Mean±SEM; n = 4; * = significant vs unstimulated; t-test; p<0.05). e. Normalization of the phospho-AKT signals measured in hLH-stimulated samples (represented as relative units in Fig. 7C) over each unstimulated (Mean±SEM; n = 4; * = significant vs unstimulated; two-way analysis of variance; p<0.05). f. Normalization of the phospho-AKT signals measured in hCG-stimulated samples (represented as relative units in Fig. 7D) over each unstimulated (Mean±SEM; n = 4; * = significant vs unstimulated; two-way analysis of variance; p<0.05).
These data demonstrate that, in hGLC, r-hLH acutely activates ERK1/2 and AKT, while r-hCG action is weaker, less sustained and significant only on the ERK1/2 pathway.
Effects of early blockade of the ERK1/2 or AKT pathway on hLH- and hCG-mediated gene expression
Given the different effect of r-hLH and r-hCG on acute ERK and AKT activation, we assessed whether the selective blockade of these pathways could affect the expression of genes known to be under hLH and/or hCG control. Day 6 hGLC where exposed to the specific inhibitors U0126 or LY294002 for one hour and then stimulated with either r-hLH or r-hCG (100 pM). After 12 hours gene expression of the EGF-like factors amphiregulin (AREG), epiregulin (EREG) and neuregulin 1 (NRG1) and of CYP19A1 (aromatase) was quantified by real-time RT-PCR. r-hLH or r-hCG stimulation resulted in significant stimulation of the expression of AREG and EREG and inhibition of NRG1 (Fig. 8A and 8B) and significant stimulation of aromatase (Fig. 8C). In addition, r-hLH was significantly more potent than r-hCG on AREG.
Figure 8. Evaluation of the gene expression induced in hGLC by hLH or hCG stimulation after 12 hours, performed by real-time PCR.
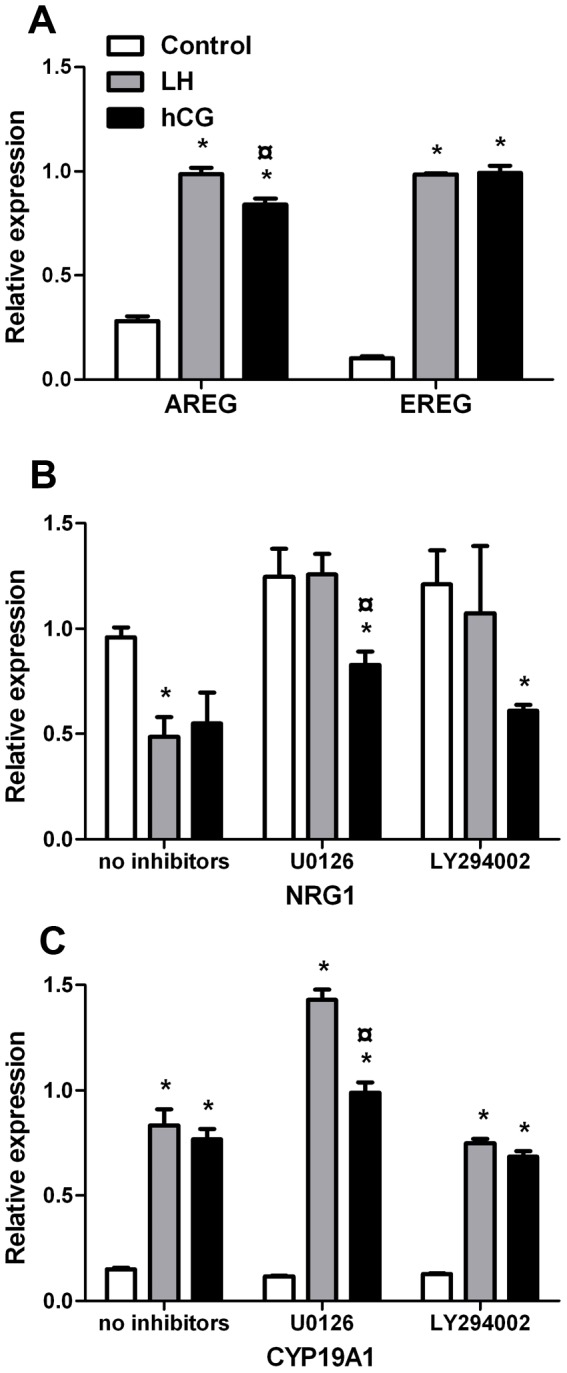
Specific ERK1/2- or AKT-pathways inhibitor (U0126 and LY294002 respectively) were also used where indicated. a. Increase in the gene expression of the EGF-like factors amphiregulin (AREG) and epiregulin (EREG) induced by LH or hCG. b. Effects of the hLH or hCG stimulation and of the ERK1/2- or AKT pathway early inhibition on neuregulin 1 (NRG1) gene expression. c. Effects of the hLH or hCG stimulation and of the ERK1/2- or AKT pathway early inhibition on NRG1 gene expression. In each treatments, RPS7 gene expression was used as normalizer (mean±SEM; n = 4; * = significant vs control; ° = significant vs hLH-stimulated; t-test; p<0.05).
Pre-incubation of hGLC with U0126 or LY294002 resulted in sustained inhibition of ERK1/2 and AKT phosphorylation, respectively (Fig. S7). The r-hLH- and r-hCG-stimulated expression of AREG and EREG was not changed by U0126 or LY294002 (data not shown), while inhibition of the ERK or AKT pathway exerted significant effects on NRG1 expression r-hLH- but not r-hCG-mediated (Fig. 8B). In fact, both U0126 and LY294002 prevented the decrease of NRG1 expression induced by r-hLH, indicating that both ERK- and AKT-pathways are involved in the inhibition of NRG1 expression controlled by r-hLH. Conversely, neither U0126 nor LY294002 had any effect on r-hCG-inhibited NRG1 expression, suggesting that the early activation of these pathways is not involved in r-hCG-mediated action on neuregulin 1, a result consistent with the very weak (or lacking) activation of these pathways by r-hCG as demonstrated in Fig. 6 and Fig. 7.
As shown in Fig. 8C, the r-hLH- and r-hCG-stimulated gene expression of CYP19A1 was not affected by LY294002, while U0126 significantly and specifically increased only r-hLH-stimulated expression. These results demonstrate that the early activation of the ERK1/2 pathway is involved in hLH- but not in hCG action on CYP19A1 expression while AKT activation plays no role.
Discussion
We systematically analyzed whether hLH and hCG are equivalent in vitro in terms of biopotency, kinetics of response and molecular effects. It is traditionally believed that hLH and hCG are biologically equivalent since they act via the same receptor, for which both molecules are assumed to have the same binding affinity. hCG has long been the preferred hormone both in clinical practice and in in vitro experiments because, before the advent of recombinant gonadotropins, it could be obtained easily by urinary extraction and owing to its much longer half-life. However, hLH and not hCG is the physiological hormone in non-pregnant women and the evolutionary reason for the presence of hCG in primates has not been clearly established. The vast majority of our current knowledge about in vitro hLH action on Leydig and granulosa/theca cells was obtained using hCG. Our experiments challenge the concept that hLH and hCG have the same biopotency and bioactivity.
Acute cAMP response
Our results demonstrate that, in vitro, in the presence of a standardized receptor milieau and at maximal stimulation, hCG is about 5-times more biopotent than equimolar concentrations of hLH in terms of cAMP production. Previous in vitro [36] and in vivo [37]–[39] studies did not pick up differences between the effects of hCG and hLH, probably because they were based on gonadotropins not quantified in exact molar terms but in “units” calibrated against a third preparation, e.g. the standard International Standard WHO 80/522 [18], or using in vivo bioassay (rat seminal vesicle weight gain assay). In the latter system, the biopotency ratio between the two molecules has been evaluated as 1∶6 [40] meaning that, when amounts of hLH and hCG equipotent in vivo are used in vitro, the molar ratio favours hLH, thereby masking the higher activity of hCG at the receptor level, which becomes evident when the two gonadotropins are used in vitro at equimolar concentrations. Essentially the same results were observed when hGLC were used to assess hLH and hCG ED50 (Fig. S3).
The equipotent doses of hLH and hCG were determined by measuring total cAMP over 3 hours (Fig. 1A) and were confirmed by progesterone production in hGLC, which reached equal levels upon hLH or hCG stimulation (Fig. 2). When half-maximal, ED50 concentrations were used in time course-experiments, intracellular cAMP accumulation resulted about 6-times faster in response to hLH than hCG (Fig. 1B). Whether the rate of cAMP increase depends on the levels of receptor occupation, on receptor dimerization, or on G protein coupling is still unknown. Binding studies performed formerly with the same cell line [18] showed that hLH was significantly less potent than hCG in displacing [125I]hCG, suggesting different binding kinetics. However, this is not sufficient to establish a difference in receptor affinity for the two hormones, since the same studies have not been performed in parallel using [125I]hLH. Binding studies with [125I]hCG and [125I]hLH would be necessary to assess whether the two hormones have different affinity/dissociation features. Future experiments should investigate which residues on the LHCGR, possibly in exon 10, are involved in distinguishing between the two hormones, whether both hLH and hCG induce receptor dimerization, and whether the same G proteins are activated by the two hormones.
Since hCG is a primate-specific hormone naturally produced mainly during pregnancy, it has been speculated that its occurrence in the evolution of primates might be related to the complexity of hemochorial placentation [41], [42]. At pregnancy onset the rescue of the corpus luteum is ensured by very low amounts of hCG produced by the early embryo and pituitary hLH is not sufficient for this task. Our cAMP in vitro data suggest that, in an in vivo, “physiological” setting, sustained progesterone production could take advantage from the higher activity of hCG, compared to hLH, at the receptor level, in addition to its longer in vivo half-life.
Comparison between recombinant and extractive gonadotropins
The use of recombinant hLH/hCG provides the unique possibility of comparing effects of both hormones at equimolar concentrations. Nevertheless, recombinant and extractive gonadotropins could differ in extent and type of glycosylation. To assess whether the possibly different tertiary structure of recombinant and extractive gonadotropins affect the cell signaling, a comparison between both type of gonadotropins was performed in different cell models, showing similar results and indicating that recombinant and extractive gonadotropins result in essentially comparable effects (Fig. S2 and Fig. S6).
hLH is secreted as multiple forms by the pituitary, as suggested by the structural and functional heterogeneity of at least 20 different hLH isoforms with various sialic acid content [43], [44], depending from the purification method used and the source, pituitary or urinary [45].
hCG is also a heterogenic molecule. It is produced from several cell types in at least five different forms, (e.g. from cytotrophoblast, syncytiotrophoblast, pituitary, tumor tissue, etc.) each having the same amino acid sequence but showing different function depending on the type and heaviness of glycosylation due to O-linked and N-linked sugars [46], [47]. Recombinant hLH and hCG are well described [48]–[50] and they are produced industrially with the goal of achieving highly consistent molecules by using a consistent and standardized manufacturing process. However, there is no extensive physicochemical comparative characterization of recombinant and natural, extractive hLH and hCG molecules, due to the lack of a highly purified reference preparation and to the different analytical methods used [51].
Using chromatographic procedures the retention time of the natural and recombinant hLH and hCG molecules are similar, demonstrating similar hydrophobicity of the alpha and beta subunits [45]. The only available comparative data between the commercial r-hLH and r-hCG versus the natural forms are referred to pharmacokinetics and pharmacodynamic studies. Both recombinant hLH and extractive hLH were eliminated with a terminal half-life of few hours. The pharmacokinetics of recombinant human LH are dose-dependent and similar to those of extractive human LH [52]. The same holds true for hCG, since r-hCG and ex-hCG produced bioequivalent pharmacodynamic responses consistent with the natural physiology of hCG [53].
Chronic cAMP and progesterone response
The experiments with hGLC revealed for the first time that intracellular cAMP response to constant, chronic stimulation by hLH or hCG is cyclic, with a period of about 4–5 hours. The hGLC cell model is naturally expressing the LHCGR and it can be presumed that the receptor undergoes a kind of “physiological” regulation upon stimulation. Differently, LHCGR gene transcription is regulated by the CMV promoter in COS-7/LHCGR and some discrepancies between hGLC could exist, in terms of intracellular cAMP regulation. This is the main reason why the hGLC cell model was used in long-term time-course experiments. Interestingly, although equipotent doses of hLH and hCG were used, after the first two-three hours of stimulation cAMP production was significantly higher in response to hCG than to hLH at a few time points. The consequence of the intermittent, intracellular cAMP production was a progressive, massive secretion of progesterone (a terminal product of steroidogenesis in granulosa-lutein cells) in the supernatant. Therefore, constant exposure to hLH or hCG at concentrations close to those circulating in vivo (as assessed by measuring hLH and hCG by ELISA in the supernatant) results in the amplification of the steroidogenic response over time. This is consistent with the clinical experience that gonadotropin administration does not need to be pulsatile and with experimental studies in mice [54], in which continuous and pulsatile infusions of hLH have identical steroidogenic effects in rats passively immunized against gonadotropin-releasing hormone. Therefore, hLH/hCG-responsive cells, such as hGLC, have developed a mechanism of cyclic refractoriness of the LHCGR to the chronic, non-pulsatile and increasing stimulation by hCG occurring in pregnancy, resulting in a progressive increase of progesterone production. Conversely, pituitary hLH secretion in non-pregnant women is physiologically pulsatile and at much lower concentrations compared to hCG during pregnancy. We speculate that the evolutionary reason why the two hormones result in these different cAMP production features could be found in the main role of cAMP plays in sustaining steroidogenesis as required in pregnancy [55], while LH has higher activity during folliculogenesis, when the cell cycle regulators AKT and ERK play a crucial role [56], [57]. The molecular mechanism regulating the fluctuations in intracellular cAMP response remains to be extensively studied. The presence of cAMP in the culture medium (Fig. 2D) strongly suggests the existence of a mechanism regulating the efflux of the second messenger outside the cell membrane of viable hGLC cells, according to previous observations in a wide variety of cell types [34]. In this regard, in primary rat skeletal muscle cultures the loss of intracellular cAMP from peak levels, induced by activation of adenylyl cyclase, was followed by an increase of extracellular cAMP. This effect was not dependent on phosphodiesterases activation, since it was obtained in the presence of IBMX [58]. Moreover, hGLC are able to downregulate this receptor after in vivo or in vitro stimulation with gonadotropins [28]. Internalization of the ligand-receptor complex, downregulation of the mRNA transcript [31] and phosphorylation of serine or threonine residues on the receptor molecules [1], [28] could be involved. In addition, receptor dimerization, a feature common to all the G-protein coupled receptors (GPCRs) [2], might play a role in the activation of different signalling pathways [59], resulting in the cyclic waving of intracellular cAMP. Finally, post-endocytotic trafficking of the hormone-receptor complex, which has already been extensively studied for the FSH receptor (FSHR) [61], the thyroid-stimulating hormone receptor (TSHR) [61], [62] and others GPCRs [63] could be involved.
Interestingly, hLH and hCG were equipotent in terms of progesterone production in spite of overall lower cAMP levels when stimulated by hLH. Progesterone production in preovulatory granulosa cells depends on the cAMP/PKA-pathway [64] but other signalling pathways could be involved [57], e.g. through molecules of the EGF family such as neuregulin 1 and amphiregulin [22], [65], modulated by ERK1/2 and AKT [66].
Acute activation of the ERK1/2 and AKT pathways and gene expression
Our data show for the first time in hGLC, that the acute effect of maximally stimulating doses of hCG on ERK1/2 phosphorylation is low when compared to the powerful effect of hLH. These data were corroborated by the experiments investigating the consequences of selective inhibition of early ERK1/2 and AKT activation on the expression of selected hLH- and hCG-dependent genes. Therefore, while hCG is more potent than LH on the PKA pathway, hLH is more potent than hCG on the ERK1/2 and AKT pathways. Interestingly, similar findings were very recently reported in goat ovarian granulosa cells after prolonged treatment with supra physiological doses of hLH and hCG [67]. In these experiments the role of hLH and hCG was investigated in relation to tumorigenesis and it was shown that hLH promoted growth and proliferation in caprine ovarian granulosa cells, while hCG was more active on stimulating cAMP levels and decreased the rate of proliferation.
The functional role of the cAMP, ERK and AKT signalling pathways in fertility is actively investigated both in humans [22] and animal models [57], [68], [69], revealing that hLH/hCG stimulation of the same receptor results in activation of different, complex signal transduction pathways and molecules [70]–[72]. Although recent findings attribute to cAMP the ability to mediate multiple and opposite effects [73], the in vitro activation of cAMP-pathway by gonadotropins is traditionally associated to structural changes, consisting in cell-rounding [19], [74], apoptotic events [74]–[76] and to prevention of meiosis resumption in the oocyte [77]. In contrast, gonadotropin-dependent activation of anti-apoptotic pathways [22], [78] and proliferative effects [79] seems to be mediated by ERK1/2 and AKT, and reduction of ERK1/2 signalling activates apoptotic signals [80]. Granulosa cell death in vitro can be induced by the specific AKT inhibitor LY294006 and is completely blocked by hLH co-treatment [78]. Taken together, these results indicate that hCG and hLH action on the regulation of cell cycle and apoptosis in granulosa cell might be divergent and/or dependent on which signal transduction pathway is activated. This is particularly relevant in determining the cell fate during folliculogenesis, when the activation of different signal transduction pathways mediates a delicate balance between pro- and anti-apoptotic signals [80].
The continuous stimulation of granulosa cells by hLH/hCG over 12 hours resulted in the increase of AREG, EREG and CYP19A1 gene expression and in the reduction of NRG1 gene expression. The EGF-like factors AREG, EREG and NRG1 are well known target genes of hLH/hCG action [66], [81], [82]. These molecules may play a role in the ovulatory process and oocyte maturation [66], [83] exerted via both ERK- and AKT-pathways activation [22],[71]. By using the U0126 or LY294002 inhibitors, we confirmed the different involvement of the ERK1/2 and AKT pathways on gene expression in dependence of hLH or hCG. In accordance with the lower AKT activation by hCG versus hLH, the inhibition of this pathway by LY294002 did not result in any relief of the hCG-dependent inhibition of NRG1 expression, while the inhibitory effect mediated by hLH was prevented by blocking AKT. Conversely, only the decrease of NRG1 gene expression induced by hLH was counteracted by inhibition of ERK1/2, indicating again the relevance of this pathway for hLH- but not for hCG action. Finally we investigated the expression of CYP19A1 gene, an early-response target of hLH and hCG both in human [25], [84] and in mice [85]. Our data demonstrated a further increase of CYP19A1 gene expression in U0126 treated cells under hLH- but not hCG stimulation. In contrast, AKT-pathway blockade did not affect CYP19A1 gene expression. This suggests that the ERK-pathway exerts a negative control on aromatase expression mediated by hLH but not hCG, pinpointing another relevant difference in the mechanism of action of the two gonadotropins in hGLC.
Conclusions
The present work provides novel insights in the downstream signaling of the LHCGR in response to hLH and hCG stimulation. For the first time we provide evidence that equimolar concentrations of hLH and hCG result in a higher in vitro activity of hCG versus hLH in terms of cAMP. Moreover, in hGLC the LHCGR is able to differentiate the in vitro hLH and hCG action at the molecular level, affecting the kinetics of cAMP production and ERK1/2- and AKT-pathway activation. Different signal transduction pathways were also differentially used to reach an equal transcriptional level of target genes by both gonadotropins, but how LHCGR modulates the activation of different signalling pathways depending on the ligand remains to be clarified. Our study was so far limited at assessing the acute effects of hLH and hCG and future experiments should be dedicated to the events following the early activation of the various pathways and their interaction, resulting in the final, full biological effects. Finally, while the in vivo effects of the differential activation of the various pathways remain to be studied in detail, the nonequivalence of hLH and hCG deserves consideration in the development of future therapeutic strategies [86].
Supporting Information
Recovery of hGLC response to hLH and hCG, over 0–6 days from in vivo pick-up. Total cAMP measured on the (a) third and (b) sixth day of culture are shown.
(TIF)
Comparison between recombinant and extractive gonadotropins effects on total cAMP production. a. Dose-response experiment with r-hLH and r-hCG versus ex-hLH and ex-hCG in COS7/LHCGR, in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours. One representative experiment is shown. b. The experiment shown in panel “a” has been repeated using hGL5/LHCGR, in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours. One representative experiment is shown.
(TIF)
Dose-response and time-course experiments in hGLC. a. Dose-response experiment with r-hLH and r-hCG in hGLC in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours. One of three independent experiments is shown. b. Time-course experiment performed by continuous incubation of hGLC for different time-points in the presence of 500 µM IBMX and gonadotropins at ED50 doses (500 pM r-hLH; 100 pM r-hCG). Intracellular cAMP was measured. One of three independent experiments is shown in absolute levels.
(TIF)
Intracellular cAMP production over 6 hours in hGLC stimulated by hLH (500 pM) or hCG (100 pM). Each value was normalized vs unstimulated. One representative experiment of two is shown.
(TIF)
Control samples of immunofluorescence analysis. Non-permeabilized cells control of LHCGR sequestration from cell surface in hLH-treated hGLC, after 15 hours. a. Unstimulated cells. b. hLH-treated hGLC. LHCGR is labeled in red (Tritc), the cytoplasmic marker ERK1/2 in green (Fitch) and cell nuclei marker (DAPI) in blue. The merging of the three images is in the lower right plate of each panel. Images are from one experiment and are representative of three independent experiments with similar results. c. Western blot control for anti-LHCGR antibody performed on 1) COS7/LHCGR cell lysates; 2) hGL5/LHCGR cell lysates; 3) Untrasfected hGL5 cell lysates (negative control); 4) and 5) hGLC cell lysates from two different donors.
(TIF)
Comparison between recombinant and extractive gonadotropin effect on ERK1/2 and AKT phosphorylation. Dose-response experiment evaluating the maximal phospho-ERK1/2 and phospho-AKT activation in hGL5/LHCGR by Western blotting. The cells were stimulated for 15 minutes by different recombinant or extractive hLH or hCG doses and the phospho-ERK1/2 and phospho-AKT signals were normalized for total ERK. One representative experiment is shown.
(TIF)
Western blot analysis of phospho ERK1/2 (a) and phospho AKT (b) and efficacy of the relative inhibitors U0126 and LY294002 in hGLC stimulated for 15 with hLH or hCG.
(TIF)
Supplementary Methods
(DOCX)
Acknowledgments
We thank Dr. Tiziana Marsella, Mother-Infant Department, and Dr. Chiara Grisolia, Laboratory of Clinical Chemistry, University Hospital of Modena, for their help.
Funding Statement
This work was supported by: Fondazione Cassa di Risparmio di Modena (http://www.fondazione-crmo.it) and IDEAS RBID08777T from Italian Ministry of University and Research (http://www.istruzione.it/web/hub). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Ascoli M, Fanelli F, Segaloff DL (2002) The lutropin/choriogonadotropin receptor, a 2002 perspective. Endocr Rev 23 2:141–74. [DOI] [PubMed] [Google Scholar]
- 2. Guan R, Feng X, Wu X, Zhang M, Zhang X, et al. (2009) Bioluminescence resonance energy transfer studies reveal constitutive dimerization of the human lutropin receptor and a lack of correlation between receptor activation and the propensity for dimerization. J Biol Chem 284 12:7483–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Khan-Sabir N, Beshay VE, Carr BR (2008) The Normal Menstrual Cycle and the Control of Ovulation. In: Endotext, Chapter 3. 2008. Avaiable: http://www.endotext.org/female/female3/femaleframe3.htm
- 4. Birken S, Maydelman Y, Gawinowicz MA, Pound A, Liu Y, et al. (1996) Isolation and characterization of human pituitary chorionic gonadotropin. Endocrinology 137 4:1402–11. [DOI] [PubMed] [Google Scholar]
- 5. Stewart F, Allen WR (1981) Biological functions and receptor binding activities of equine chorionic gonadotrophins. J Reprod Fertil 62 2:527–36. [DOI] [PubMed] [Google Scholar]
- 6. Cole LA (2009) New discoveries on the biology and detection of human chorionic gonadotropin. Reprod Biol Endocrinol 7: 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Santen RJ, Bardin CW (1973) Episodic luteinizing hormone secretion in man. Pulse analysis, clinical interpretation, physiologic mechanisms. J Clin Invest 52 10:2617–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Penny R, Olambiwonnu NO, Frasier SD (1977) Episodic fluctuations of serum gonadotropins in pre- and post-pubertal girls and boys. J Clin Endocrinol Metab 45 2:307–11. [DOI] [PubMed] [Google Scholar]
- 9. Diebel ND, Bogdanove EM (1978) Analysis of luteinizing hormone and follicle-stimulating hormone release kinetics during a dynamic secretory event, the postpartum preovulatory surge in the rat, based on quantitative changes in stored and circulating luteinizing hormone and follicle-stimulating hormone and metabolic clearance data for these hormones. Endocrinology 103 3:665–73. [DOI] [PubMed] [Google Scholar]
- 10. Lähteenmäki P (1978) The disappearance of HCG and return of pituitary function after abortion. Clin Endocrinol (Oxf) 9 2:101–12. [DOI] [PubMed] [Google Scholar]
- 11. Mann K, Lamerz R, Hellmann T, Kümper HJ, Staehler G, et al. (1980) Use of human chorionic gonadotropin and alpha-fetoprotein radioimmunoassays: specificity and apparent half-life determination after delivery and in patients with germ cell tumors. Oncodev Biol Med 1 4–5:301–12. [PubMed] [Google Scholar]
- 12. Norman RJ, Buchholz MM, Somogyi AA, Amato F (2000) hCGbeta core fragment is a metabolite of hCG: evidence from infusion of recombinant hCG. J Endocrinol 164 3:299–305. [DOI] [PubMed] [Google Scholar]
- 13. Ludwig M, Westergaard LG, Diedrich K, Andersen CY (2003) Developments in drugs for ovarian stimulation. Best Pract Res Clin Obstet Gynaecol 17 2:231–47. [DOI] [PubMed] [Google Scholar]
- 14. Rose MP, Gaines Das RE, Balen AH (2000) Definition and measurement of follicle stimulating hormone. Endocr Rev 21 1:5–22. [DOI] [PubMed] [Google Scholar]
- 15. Lunenfeld B (2002) Development of gonadotrophins for clinical use. Reprod Biomed Online 4 Suppl 1: 11–7. [DOI] [PubMed] [Google Scholar]
- 16. Galet C, Ascoli M (2005) The differential binding affinities of the luteinizing hormone (LH)/choriogonadotropin receptor for LH and choriogonadotropin are dictated by different extracellular domain residues. Mol Endocrinol 19 5:1263–76. [DOI] [PubMed] [Google Scholar]
- 17. Gromoll J, Eiholzer U, Nieschlag E, Simoni M (2000) Male hypogonadism caused by homozygous deletion of exon 10 of the luteinizing hormone (LH) receptor: differential action of human chorionic gonadotropin and LH. J Clin Endocrinol Metab 85 6:2281–6. [DOI] [PubMed] [Google Scholar]
- 18. Müller T, Gromoll J, Simoni M (2003) Absence of exon 10 of the human luteinizing hormone (LH) receptor impairs LH, but not human chorionic gonadotropin action. J Clin Endocrinol Metab 88 5:2242–9. [DOI] [PubMed] [Google Scholar]
- 19. Schiffer Z, Keren-Tal I, Deutsch M, Dantes A, Aharoni D, et al. (1996) Fourier analysis of differential light scattering for the quantitation of FSH response associated with structural changes in immortalized granulosa cells. Mol Cell Endocrinol 118 1–2:145–53. [DOI] [PubMed] [Google Scholar]
- 20. Amsterdam A, Sasson R, Keren-Tal I, Aharoni D, Dantes A, et al. (2003) Alternative pathways of ovarian apoptosis: death for life. Biochem Pharmacol 66 8:1355–62. [DOI] [PubMed] [Google Scholar]
- 21. Amsterdam A, Keren-Tal I, Aharoni D, Dantes A, Land-Bracha A, et al. (2003) Steroidogenesis and apoptosis in the mammalian ovary. Steroids 68 10–13:861–7. [DOI] [PubMed] [Google Scholar]
- 22. Ben-Ami I, Armon L, Freimann S, Strassburger D, Ron-El R, et al. (2009) EGF-like growth factors as LH mediators in the human corpus luteum. Hum Reprod 24 1:176–84. [DOI] [PubMed] [Google Scholar]
- 23. Park J-Y, Su Y-Q, Ariga M, Law E, Jin S-LC, et al. (2004) EGF-like growth factors as mediators of LH action in the ovulatory follicle. Science 303 5658:682–4. [DOI] [PubMed] [Google Scholar]
- 24. Donadeu FX, Ascoli M (2005) The differential effects of the gonadotropin receptors on aromatase expression in primary cultures of immature rat granulosa cells are highly dependent on the density of receptors expressed and the activation of the inositol phosphate cascade. Endocrinology 146 9:3907–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Lindeberg M, Carlström K, Ritvos O, Hovatta O (2007) Gonadotrophin stimulation of non-luteinized granulosa cells increases steroid production and the expression of enzymes involved in estrogen and progesterone synthesis. Hum Reprod 22 2:401–6. [DOI] [PubMed] [Google Scholar]
- 26. Rainey WH, Sawetawan C, Shay JW, Michael MD, Mathis JM, et al. (1994) Transformation of human granulosa cells with the E6 and E7 regions of human papillomavirus. J Clin Endocrinol Metab 78 3:705–10. [DOI] [PubMed] [Google Scholar]
- 27. Nordhoff V, Sonntag B, von Tils D, Götte M, Schüring AN, et al. (2011) Effects of the FSH receptor gene polymorphism p.N680S on cAMP and steroid production in cultured primary human granulosa cells. Reprod Biomed Online 23 2:196–203. [DOI] [PubMed] [Google Scholar]
- 28. Amsterdam A, Hanoch T, Dantes A, Tajima K, Strauss JF, et al. (2002) Mechanisms of gonadotropin desensitization. Mol Cell Endocrinol 187 1–2:69–74. [DOI] [PubMed] [Google Scholar]
- 29. Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65 1–2:55–63. [DOI] [PubMed] [Google Scholar]
- 30. Lin PC, Li X, Lei ZM, Rao ChV (2003) Human cervix contains functional luteinizing hormone/human chorionic gonadotropin receptors. J Clin Endocrinol Metab 88 7:3409–14. [DOI] [PubMed] [Google Scholar]
- 31. Menon B, Franzo-Romain M, Damanpour S, Menon KMJ (2011) Luteinizing hormone receptor mRNA down-regulation is mediated through ERK-dependent induction of RNA binding protein. Mol Endocrinol 25 2:282–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Palaniappan M, Menon KM (2010) Human chorionic gonadotropin stimulates theca-interstitial cell proliferation and cell cycle regulatory proteins by a cAMP-dependent activation of AKT/mTORC1 signaling pathway. Mol Endocrinol 24 9:1782–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)). Method Methods 25 4:402–8. [DOI] [PubMed] [Google Scholar]
- 34. Hofer AM, Lefkimmiatis K (2007) Extracellular calcium and cAMP: Second messengers as “third messenger”? Physiology (Bethesda) 22: 320–7. [DOI] [PubMed] [Google Scholar]
- 35. Lawrence TS, Ginzberg RD, Gilula NB, Beers WH (1979) Hormonally Induced Cell Shape Changes in Cultured Rat Ovarian Granulosa Cells. The Journal of Cell Biology 80 1:21–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Molskness TA, Zelinski-Wooten MB, Hild-Petito SA, Stouffer RL (1991) Comparison of the steroidogenic response of luteinized granulosa cells from rhesus monkeys to luteinizing hormone and chorionic gonadotropin. Biol Reprod 45 2:273–81. [DOI] [PubMed] [Google Scholar]
- 37. Duijkers IJ, Beerens MC, Coelingh Bennink HJ, Huisman JA, Rombout F, et al. (1995) Pharmacokinetics of two human menopausal gonadotrophin preparations after single intravenous administration during pituitary suppression. Hum Reprod 10 6:1367–72. [DOI] [PubMed] [Google Scholar]
- 38. Zeleznik AJ (1998) In vivo responses of the primate corpus luteum to luteinizing hormone and chorionic gonadotropin. Proc Natl Acad Sci USA 95 18:11002–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Cailleux-Bounacer A, Reznik Y, Cauliez B, Menard JF, Duparc C, et al. (2008) Evaluation of endocrine testing of Leydig cell function using extractive and recombinant human chorionic gonadotropin and different doses of recombinant human LH in normal men. Eur J Endocrinol 159 2:171–8. [DOI] [PubMed] [Google Scholar]
- 40. Van Hell, Matthijsen R, Overbeek GA (1964) Effects of human menopausal gonadotrophin preparations in different bioassay methods. Acta Endocrinol 47: 409–18. [DOI] [PubMed] [Google Scholar]
- 41. Cole LA (2009) hCG and hyperglycosylated hCG in the establishment and evolution of hemochorial placentation. J Reprod Immunol 82 2:112–8. [DOI] [PubMed] [Google Scholar]
- 42. Nagirnaja L, Rull K, Uusküla L, Hallast P, Grigorova M, et al. (2010) Genomics and genetics of gonadotropin beta-subunit genes: Unique FSHB and duplicated LHB/CGB loci. Mol Cell Endocrinol 329 1–2:4–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Stanton PG, Pozvek G, Burgon PG, Robertson DM, Hearn MT (1993) Isolation and characterization of human LH isoforms. J Endocrinol 138 3:529–43. [DOI] [PubMed] [Google Scholar]
- 44. Stanton PG, Burgon PG, Hearn MT, Robertson DM (1996) Structural and functional characterisation of hFSH and hLH isoforms. Mol Cell Endocrinol 125 1–2:133–41. [DOI] [PubMed] [Google Scholar]
- 45. Almeida BE, Oliveira JE, Carvalho CM, Dalmora SL, Bartolini P, et al. (2010) Analysis of human luteinizing hormone and human chorionic gonadotropin preparations of different origins by reversed-phase high-performance liquid chromatography. J Pharm Biomed Anal 53 1:90–7. [DOI] [PubMed] [Google Scholar]
- 46. Lottersberger C, Hoermann R, Mann K, Schwarz S, Berger P (2003) Tumor- and pregnancy-derived isoforms of human chorionic gonadotropin: biological and diagnostic relevance. Horm Res 59 3:125–34. [DOI] [PubMed] [Google Scholar]
- 47. Cole LA (2012) hCG, the wonder of today's science. Reprod Biol Endocrinol 10: 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Amoresano A, Siciliano R, Orrù S, Napoleoni R, Altarocca V, et al. (1996) Structural characterisation of human recombinant glycohormones follitropin, lutropin and choriogonadotropin expressed in Chinese hamster ovary cells. Eur J Biochem 242 3:608–18. [DOI] [PubMed] [Google Scholar]
- 49. Lambert A, Talbot JA, Anobile CJ, Robertson WR (1998) Gonadotrophin heterogeneity and biopotency: implications for assisted reproduction. Mol Hum Reprod 4 7:619–29. [DOI] [PubMed] [Google Scholar]
- 50. Gervais A, Hammel YA, Pelloux S, Lepage P, Baer G, et al. (2003) Glycosylation of human recombinant gonadotrophins: characterization and batch-to-batch consistency. Glycobiology 13 3:179–89. [DOI] [PubMed] [Google Scholar]
- 51. Talbot JA, Mitchell R, Hoy AM, Lambert A, Gauher A, et al. (1996) Recombinant human luteinizing hormone: a partial physicochemical, biological and immunological characterization. Mol Hum Reprod 2 10:799–806. [DOI] [PubMed] [Google Scholar]
- 52. le Cotonnec JY, Porchet HC, Beltrami V, Munafo A (1998) Clinical pharmacology of recombinant human luteinizing hormone: Part I. Pharmacokinetics after intravenous administration to healthy female volunteers and comparison with urinary human luteinizing hormone. Fertil Steril 1998 69 2:189–94. [DOI] [PubMed] [Google Scholar]
- 53. Trinchard-Lugan I, Khan A, Porchet HC, Munafo A (2002) Pharmacokinetics and pharmacodynamics of recombinant human chorionic gonadotrophin in healthy male and female volunteers. Reprod Biomed Online 4 2:106–15. [DOI] [PubMed] [Google Scholar]
- 54. Gibson-Berry KL, Chase DJ (1990) Continuous and pulsatile infusions of luteinizing hormone have identical effects on steroidogenic capacity and sensitivity of Leydig cells in rats passively immunized against gonadotropin-releasing hormone. Endocrinology 126 6:3107–15. [DOI] [PubMed] [Google Scholar]
- 55. Albrecht ED, Pepe GJ (1990) Placental steroid hormone biosynthesis in primate pregnancy. Endocr Rev 11 1:124–50. [DOI] [PubMed] [Google Scholar]
- 56. Evans AC, Martin F (2000) Kinase pathways in dominant and subordinate ovarian follicles during the first wave of follicular development in sheep. Anim Reprod Sci 64 3–4:221–31. [DOI] [PubMed] [Google Scholar]
- 57. Ryan KE, Glister C, Lonergan P, Martin F, Knight PG, et al. (2008) Functional significance of the signal transduction pathways Akt and Erk in ovarian follicles: in vitro and in vivo studies in cattle and sheep. J Ovarian Res 1 1:2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Godinho RO, Costa VL Jr (2003) Regulation of intracellular cyclic AMP in skeletal muscle cells involves the efflux of cyclic nucleotide to the extracellular compartment. Br J Pharmacol 138 5:995–1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Segaloff DL (2010) Constitutive activity of the lutropin receptor and its allosteric modulation by receptor heterodimerization. Meth Enzymol 484: 231–52. [DOI] [PubMed] [Google Scholar]
- 60. Rozenfeld R, Devi LA (2011) Exploring a role for heteromerization in GPCR signalling specificity. Biochem J 433 1:11–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Krishnamurthy H, Kishi H, Shi M, Galet C, Bhaskaran RS, et al. (2003) Postendocytotic trafficking of the follicle-stimulating hormone (FSH)-FSH receptor complex. Mol Endocrinol 17 11:2162–76. [DOI] [PubMed] [Google Scholar]
- 62. Quellari M, Desroches A, Beau I, Beaudeux E, Misrahi M (2003) Role of cleavage and shedding in human thyrotropin receptor function and trafficking. Eur J Biochem 270 17:3486–97. [DOI] [PubMed] [Google Scholar]
- 63. Magalhaes AC, Dunn H, Ferguson SS (2012) Regulation of GPCR activity, trafficking and localization by GPCR-interacting proteins. Br J Pharmacol 165 6:1717–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Karlsson AB, Maizels ET, Flynn MP, Jones JC, Shelden EA, et al. (2010) Luteinizing hormone receptor-stimulated progesterone production by preovulatory granulosa cells requires protein kinase A-dependent activation/dephosphorylation of the actin dynamizing protein cofilin. Mol Endocrinol 24 9:1765–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Noma N, Kawashima I, Fan H-Y, Fujita Y, Kawai T, et al. (2011) LH-induced neuregulin 1 (NRG1) type III transcripts control granulosa cell differentiation and oocyte maturation. Mol Endocrinol 25 1:104–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Chin EC, Abayasekara DRE (2004) Progesterone secretion by luteinizing human granulosa cells: a possible cAMP-dependent but PKA-independent mechanism involved in its regulation. J Endocrinol 183 1:51–60. [DOI] [PubMed] [Google Scholar]
- 67. Gupta C, Chapekar T, Chhabra Y, Singh P, Sinha S, et al. (2012) Differential response to sustained stimulation by hCG and LH on goat ovarian granulosa cells. Indian J Med Res 331–40. [PMC free article] [PubMed] [Google Scholar]
- 68. Brown C, LaRocca J, Pietruska J, Ota M, Anderson L, et al. (2010) Subfertility caused by altered follicular development and oocyte growth in female mice lacking PKB alpha/Akt1. Biol Reprod 82 2:246–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Yamashita S, Tai P, Charron J, Ko C, Ascoli M (2011) The Leydig cell MEK/ERK pathway is critical for maintaining a functional population of adult Leydig cells and for fertility. Mol Endocrinol 25 7:1211–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Choi J-H, Chen C-L, Poon SL, Wang H-S, Leung PCK (2009) Gonadotropin-stimulated epidermal growth factor receptor expression in human ovarian surface epithelial cells: involvement of cyclic AMP-dependent exchange protein activated by cAMP pathway. Endocr Relat Cancer 16 1:179–88. [DOI] [PubMed] [Google Scholar]
- 71. Yamashita Y, Hishinuma M, Shimada M (2009) Activation of PKA, p38 MAPK and ERK1/2 by gonadotropins in cumulus cells is critical for induction of EGF-like factor and TACE/ADAM17 gene expression during in vitro maturation of porcine COCs. J Ovarian Res 2: 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Ulloa-Aguirre A, Crépieux P, Poupon A, Maurel M-C, Reiter E (2011) Novel pathways in gonadotropin receptor signaling and biased agonism. Rev Endocr Metab Disord 12 4:259–74. [DOI] [PubMed] [Google Scholar]
- 73. Insel PA, Zhang L, Murray F, Yokouchi H, Zambon AC (2012) Cyclic AMP is both a pro-apoptotic and anti-apoptotic second messenger. Acta Physiol (Oxf) 204 2:277–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Keren-Tal I, Suh BS, Dantes A, Lindner S, Oren M, et al. (1995) Involvement of p53 expression in cAMP-mediated apoptosis in immortalized granulosa cells. Exp Cell Res 218 1:283–95. [DOI] [PubMed] [Google Scholar]
- 75. Amsterdam A, Dantes A, Liscovitch M (1994) Role of phospholipase-D and phosphatidic acid in mediating gonadotropin-releasing hormone-induced inhibition of preantral granulosa cell differentiation. Endocrinology 135 3:1205–11. [DOI] [PubMed] [Google Scholar]
- 76. Aharoni D, Dantes A, Oren M, Amsterdam A (1995) cAMP-mediated signals as determinants for apoptosis in primary granulosa cells. Exp Cell Res 218 1:271–82. [DOI] [PubMed] [Google Scholar]
- 77. Sun Q-Y, Miao Y-L, Schatten H (2009) Towards a new understanding on the regulation of mammalian oocyte meiosis resumption. Cell Cycle 8 17:2741–7. [DOI] [PubMed] [Google Scholar]
- 78. Johnson AL, Bridgham JT, Swenson JA (2001) Activation of the Akt/protein kinase B signaling pathway is associated with granulosa cell survival. Biol Reprod 64 5:1566–74. [DOI] [PubMed] [Google Scholar]
- 79. Peter AT, Dhanasekaran N (2003) Apoptosis of granulosa cells: a review on the role of MAPK-signalling modules. Reprod Domest Anim 38 3:209–13. [DOI] [PubMed] [Google Scholar]
- 80. Craig J, Orisaka M, Wang H, Orisaka S, Thompson W, et al. (2007) Gonadotropin and intra-ovarian signals regulating follicle development and atresia: the delicate balance between life and death. Front Biosci 12: 3628–39. [DOI] [PubMed] [Google Scholar]
- 81. Motola S, Popliker M, Tsafriri A (2008) Response of follicle cells to ovulatory stimuli within the follicle and in primary culture. Mol Cell Endocrinol 282 1–2:26–31. [DOI] [PubMed] [Google Scholar]
- 82. Zamah AM, Hsieh M, Chen J, Vigne JL, Rosen MP, et al. (2010) Human oocyte maturation is dependent on LH-stimulated accumulation of the epidermal growth factor-like growth factor, amphiregulin. Hum Reprod 25 10:2569–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Sekiguchi T, Mizutani T, Yamada K, Kajitani T, Yazawa T, et al. (2004) Expression of epiregulin and amphiregulin in the rat ovary. J Mol Endocrinol 33 1:281–91. [DOI] [PubMed] [Google Scholar]
- 84. McAllister JM, Mason JI, Byrd W, Trant JM, Waterman MR, et al. (1990) Proliferating human granulosa-lutein cells in long term monolayer culture: expression of aromatase, cholesterol side-chain cleavage, and 3 beta-hydroxysteroid dehydrogenase. J Clin Endocrinol Metab 71 1:26–33. [DOI] [PubMed] [Google Scholar]
- 85. Fan H-Y, Liu Z, Johnson PF, Richards JS (2011) CCAAT/enhancer-binding proteins (C/EBP)-α and -β are essential for ovulation, luteinization, and the expression of key target genes. Mol Endocrinol 25 2:253–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Baer G, Loumaye E (2003) Comparison of recombinant human luteinising hormone (r-hLH) and human menopausal gonadotropin (hMG) in assisted reproductive technology. Curr Med Res Opin 19 2:83–8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Recovery of hGLC response to hLH and hCG, over 0–6 days from in vivo pick-up. Total cAMP measured on the (a) third and (b) sixth day of culture are shown.
(TIF)
Comparison between recombinant and extractive gonadotropins effects on total cAMP production. a. Dose-response experiment with r-hLH and r-hCG versus ex-hLH and ex-hCG in COS7/LHCGR, in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours. One representative experiment is shown. b. The experiment shown in panel “a” has been repeated using hGL5/LHCGR, in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours. One representative experiment is shown.
(TIF)
Dose-response and time-course experiments in hGLC. a. Dose-response experiment with r-hLH and r-hCG in hGLC in the presence of 500 µM IBMX. Total cAMP was measured after 3 hours. One of three independent experiments is shown. b. Time-course experiment performed by continuous incubation of hGLC for different time-points in the presence of 500 µM IBMX and gonadotropins at ED50 doses (500 pM r-hLH; 100 pM r-hCG). Intracellular cAMP was measured. One of three independent experiments is shown in absolute levels.
(TIF)
Intracellular cAMP production over 6 hours in hGLC stimulated by hLH (500 pM) or hCG (100 pM). Each value was normalized vs unstimulated. One representative experiment of two is shown.
(TIF)
Control samples of immunofluorescence analysis. Non-permeabilized cells control of LHCGR sequestration from cell surface in hLH-treated hGLC, after 15 hours. a. Unstimulated cells. b. hLH-treated hGLC. LHCGR is labeled in red (Tritc), the cytoplasmic marker ERK1/2 in green (Fitch) and cell nuclei marker (DAPI) in blue. The merging of the three images is in the lower right plate of each panel. Images are from one experiment and are representative of three independent experiments with similar results. c. Western blot control for anti-LHCGR antibody performed on 1) COS7/LHCGR cell lysates; 2) hGL5/LHCGR cell lysates; 3) Untrasfected hGL5 cell lysates (negative control); 4) and 5) hGLC cell lysates from two different donors.
(TIF)
Comparison between recombinant and extractive gonadotropin effect on ERK1/2 and AKT phosphorylation. Dose-response experiment evaluating the maximal phospho-ERK1/2 and phospho-AKT activation in hGL5/LHCGR by Western blotting. The cells were stimulated for 15 minutes by different recombinant or extractive hLH or hCG doses and the phospho-ERK1/2 and phospho-AKT signals were normalized for total ERK. One representative experiment is shown.
(TIF)
Western blot analysis of phospho ERK1/2 (a) and phospho AKT (b) and efficacy of the relative inhibitors U0126 and LY294002 in hGLC stimulated for 15 with hLH or hCG.
(TIF)
Supplementary Methods
(DOCX)