Abstract
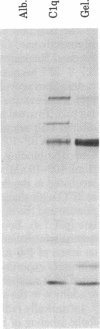
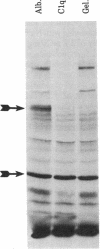
Fibronectin immobilized to plastic tubes binds soluble C1q with a Kd of 82 +/- 2.6 nM. The binding of fibronectin to C1q is relatively insensitive to pH but is sensitive to ionic conditions. C1q covalently bound to Sepharose selectively binds cellular fibronectin produced by a hamster fibroblast cell line. The globular head regions of C1q have no effect on the binding of C1q to fibronectin but the collagenous tails of C1q interfere competitively with a Ki of 59 nM. We conclude that fibronectin binds C1q via its collagen-like tail region and thus the process resembles the binding of fibronectin to gelatin. This is further emphasized by our observation that gelatin binds to fibronectin immobilized on plastic tubes with a Kd of 131 nM. Because fibronectin stimulates endocytosis in several systems and promotes the clearance of particulate material from the circulation, these results suggest the possibility that fibronectin could function in the clearance of C1q-coated material such as immune complexes or cellular debris.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bing D. H., Andrews J. M., Morris K. M., Cole E., Irish V. Purification of subcomponents Clq, Cl(-)r and Cl(-)s of the first component of complement from Cohn Fraction I by affinity chromatography. Prep Biochem. 1980;10(3):269–296. doi: 10.1080/10826068009412829. [DOI] [PubMed] [Google Scholar]
- Blumenstock F. A., Saba T. M., Weber P., Laffin R. Biochemical and immunological characterization of human opsonic alpha2SB glycoprotein: its identity with cold-insoluble globulin. J Biol Chem. 1978 Jun 25;253(12):4287–4291. [PubMed] [Google Scholar]
- Burton D. R., Boyd J., Brampton A. D., Easterbrook-Smith S. B., Emanuel E. J., Novotny J., Rademacher T. W., van Schravendijk M. R., Sternberg M. J., Dwek R. A. The Clq receptor site on immunoglobulin G. Nature. 1980 Nov 27;288(5789):338–344. doi: 10.1038/288338a0. [DOI] [PubMed] [Google Scholar]
- Chiu A. T., Ryan J. W., Stewart J. M., Dorer F. E. Formation of angiotensin III by angiotensin-converting enzyme. Biochem J. 1976 Apr 1;155(1):189–192. doi: 10.1042/bj1550189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuatrecasas P. Affinity chromatography. Annu Rev Biochem. 1971;40:259–278. doi: 10.1146/annurev.bi.40.070171.001355. [DOI] [PubMed] [Google Scholar]
- Doran J. E., Mansberger A. R., Reese A. C. Cold insoluble globulin-enhanced phagocytosis of gelatinized targets by macrophage monolayers: a model system. J Reticuloendothel Soc. 1980 May;27(5):471–483. [PubMed] [Google Scholar]
- Emmerling M. R., Johnson C. D., Mosher D. F., Lipton B. H., Lilien J. E. Cross-linking and binding of fibronectin with asymmetric acetylcholinesterase. Biochemistry. 1981 May 26;20(11):3242–3247. doi: 10.1021/bi00514a040. [DOI] [PubMed] [Google Scholar]
- Engvall E., Ruoslahti E. Binding of soluble form of fibroblast surface protein, fibronectin, to collagen. Int J Cancer. 1977 Jul 15;20(1):1–5. doi: 10.1002/ijc.2910200102. [DOI] [PubMed] [Google Scholar]
- Giclas P. C., Pinckard R. N., Olson M. S. In vitro activation of complement by isolated human heart subcellular membranes. J Immunol. 1979 Jan;122(1):146–151. [PubMed] [Google Scholar]
- Hughes-Jones N. C., Gardner B. Reaction between the isolated globular sub-units of the complement component C1q and IgG-complexes. Mol Immunol. 1979 Sep;16(9):697–701. doi: 10.1016/0161-5890(79)90010-5. [DOI] [PubMed] [Google Scholar]
- Isliker H., Bing D. H., Lahan J., Hynes R. O. Fibronectin interacts with Clq, a subcomponent of the first component of complement. Immunol Lett. 1982 Jan;4(1):39–43. doi: 10.1016/0165-2478(82)90075-x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K., Favre M. Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol. 1973 Nov 15;80(4):575–599. doi: 10.1016/0022-2836(73)90198-8. [DOI] [PubMed] [Google Scholar]
- McConahey P. J., Dixon F. J. A method of trace iodination of proteins for immunologic studies. Int Arch Allergy Appl Immunol. 1966;29(2):185–189. doi: 10.1159/000229699. [DOI] [PubMed] [Google Scholar]
- Molnar J., Gelder F. B., Lai M. Z., Siefring G. E., Jr, Credo R. B., Lorand L. Purification of opsonically active human and rat cold-insoluble globulin (plasma fibronectin). Biochemistry. 1979 Sep 4;18(18):3909–3916. doi: 10.1021/bi00585a010. [DOI] [PubMed] [Google Scholar]
- Mosher D. F. Fibronectin. Prog Hemost Thromb. 1980;5:111–151. [PubMed] [Google Scholar]
- Porter R. R., Reid K. B. Activation of the complement system by antibody-antigen complexes: the classical pathway. Adv Protein Chem. 1979;33:1–71. doi: 10.1016/s0065-3233(08)60458-1. [DOI] [PubMed] [Google Scholar]
- Porter R. R., Reid K. B. The biochemistry of complement. Nature. 1978 Oct 26;275(5682):699–704. doi: 10.1038/275699a0. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Engvall E., Hayman E. G. Fibronectin: current concepts of its structure and functions. Coll Relat Res. 1981;1(1):95–128. doi: 10.1016/s0174-173x(80)80011-2. [DOI] [PubMed] [Google Scholar]
- Saba T. M., Jaffe E. Plasma fibronectin (opsonic glycoprotein): its synthesis by vascular endothelial cells and role in cardiopulmonary integrity after trauma as related to reticuloendothelial function. Am J Med. 1980 Apr;68(4):577–594. doi: 10.1016/0002-9343(80)90310-1. [DOI] [PubMed] [Google Scholar]
- Senger D. R., Hynes R. O. C3 component of complement secreted by established cell lines. Cell. 1978 Oct;15(2):375–384. doi: 10.1016/0092-8674(78)90006-5. [DOI] [PubMed] [Google Scholar]
- Sim R. B., Arlaud G. J., Colomb M. G. C1 inhibitor-dependent dissociation of human complement component C1 bound to immune complexes. Biochem J. 1979 Jun 1;179(3):449–457. doi: 10.1042/bj1790449a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sledge C. R., Bing D. H. Binding properties of the human complement protein Clq. J Biol Chem. 1973 Apr 25;248(8):2818–2823. [PubMed] [Google Scholar]
- Strang C. J., Siegel R. C., Phillips M. L., Poon P. H., Schumaker V. N. Ultrastructure of the first component of human complement: electron microscopy of the crosslinked complex. Proc Natl Acad Sci U S A. 1982 Jan;79(2):586–590. doi: 10.1073/pnas.79.2.586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaheri A., Mosher D. F. High molecular weight, cell surface-associated glycoprotein (fibronectin) lost in malignant transformation. Biochim Biophys Acta. 1978 Sep 18;516(1):1–25. doi: 10.1016/0304-419x(78)90002-1. [DOI] [PubMed] [Google Scholar]
- Wagner D. D., Hynes R. O. Domain structure of fibronectin and its relation to function. Disulfides and sulfhydryl groups. J Biol Chem. 1979 Jul 25;254(14):6746–6754. [PubMed] [Google Scholar]
- Yamada K. M., Olden K. Fibronectins--adhesive glycoproteins of cell surface and blood. Nature. 1978 Sep 21;275(5677):179–184. doi: 10.1038/275179a0. [DOI] [PubMed] [Google Scholar]
- Ziccardi R. J. Activation of the early components of the classical complement pathway under physiologic conditions. J Immunol. 1981 May;126(5):1769–1773. [PubMed] [Google Scholar]
- van de Water L., 3rd, Schroeder S., Crenshaw E. B., 3rd, Hynes R. O. Phagocytosis of gelatin-latex particles by a murine macrophage line is dependent on fibronectin and heparin. J Cell Biol. 1981 Jul;90(1):32–39. doi: 10.1083/jcb.90.1.32. [DOI] [PMC free article] [PubMed] [Google Scholar]