Abstract
Purpose
We examined the efficacy of retinol palmitate (VApal) for dry eyes using dry eye model rabbits whose lacrimal glands were resected.
Materials and methods
After alkaline injury on keratoconjunctival epithelium, VApal eye drops were administered 6 times a day for 7 days. The efficacy of VApal was also compared with that of 0.1% hyaluronic acid eye drops.
Results
The fluorescein staining and rose bengal scores showed a significant decrease compared with the score in the vehicle group at 7 days (P < 0.05) in the 1000 IU/mL VApal group and at both 3 days (P < 0.05) and 7 days (P < 0.01) in the 1500 IU/mL VApal group. Histological examination revealed recovery of the corneal epithelium, and PAS staining disclosed the recovery of mucin-producing lower palpebral conjunctival goblet cells after 7 days in the 1500 IU/mL VApal group compared with the vehicle group. Results from impression cytology showed a significant increase in density of conjunctival goblet cells compared with that in the vehicle group after 7 days in the 1000 IU/mL VApal group and after 3 and 7 days in the 1500 IU/mL VApal group. There were no significant changes in tear flow in either group. Topical application of VApal at 1500 IU/mL showed greater improvement than 0.1% hyaluronic acid in both fluorescein and rose bengal score and in the density of conjunctival goblet cells.
Conclusion
It is suggested that VApal is effective for the improvement of keratoconjunctival epithelial damage associated with tear abnormalities, such as dry eyes.
Keywords: cornea, dry eye, hyaluronic acid, vitamin A, wound healing, retinol palmitate
Introduction
Vitamin A (VA) regulates the proliferation and differentiation of corneal epithelial cells and preserved conjunctival goblet cells.1 It has been used in the treatment of disease of the ocular surface.2–5 Vitamin A regulates the differentiation of the keratoconjunctival epithelium on the ocular surface, is released from the lacrimal gland, and exists in retinol in the tears.6 It is known that VA deficiency leads to keratoconjunctival epithelial damage, resulting in conditions such as superficial punctate keratitis and dry keratoconjunctivitis, as well as abnormalities such as loss of conjunctival goblet cells.7,8 It has been reported that keratoconjunctival epithelial damage and loss of conjunctival goblet cells occur in rabbits with VA deficiency9 and that expression of a mucin gene (rMuc4) is decreased in the conjunctival epithelium of rats with VA deficiency.10 These findings suggest the possibility of VA deficiency leading to decreased mucin secretion, resulting in the aggravation of dry eyes, because inadequate tear retention on the ocular surface accelerates tear film break-up, causing epithelial damage.
With regard to the effect of VA on mucin production, we previously reported that retinol palmitate (VApal) promotes mucin production in cultured rabbit conjunctival tissue (in vitro)11 and that topical application of VA markedly improves keratoconjunctival epithelial damage and loss of conjunctival goblet cells.9 It was also reported that topical application of VApal promotes the healing of keratoconjunctival damage and recovery of conjunctival goblet cells and that its effect on keratoconjunctival epithelial damage is particularly strong in the mucin layer.12 Additionally, it was reported that retinoic acid ointment promotes the healing in rabbits after corneal epithelial injury.13
The most traditional drug delivery system to the anterior segment of eye is eye drops. However, ocular drug delivery is hampered by the barriers protecting the eye. Conventional ophthalmic drug-delivery systems, including eye drops and ophthalmic ointments, are no longer sufficient for treating eye diseases and may lead to side effects. Particularly, VA is readily inactivated by oxidation and shows poor stability. There are several reports concerned with side effects of retinoic acid ophthalmic ointment.2,3 In contrast, it has been reported that ophthalmic solutions containing retinol palmitate (VApal, an ester of VA) are effective and cause few side effects.4
Therefore, to clarify the efficacy of VApal for dry eyes, we resected the lacrimal glands of rabbits as a model of dry eyes that was considered to be similar to the actual clinical situation and evaluated the effect of VApal eye drops in this model. The model decreased tear secretion and density of functional goblet cells and induced keratoconjunctival epithelial damage, which has never been reported in animal models. The efficacy of VApal was also compared with that of 0.1% hyaluronic acid eye drops for the treatment of keratoconjunctival epithelial damage. Hyaluronic acid has an average molecular weight of 500,000 to 1,200,000 and is widely used for dry eyes in clinical practice.
Materials and methods
Animals
This study employed 28 healthy male New Zealand White rabbits (Japan SLC Inc, Hamamatsu, Japan) weighing 2.5–3 kg. The animals were used in compliance with the Association for Research in Vision and Ophthalmology and the National Institutes of Health statement for the use of Animals in Research.
Experimental procedure
Resection of the lacrimal gland
Under general anesthesia by intramuscular administration of a mixture of ketamine hydrochloride (100 mg/kg) and xylazine hydrochloride (10 mg/kg), a rabbit model of dry eyes was prepared according to the method of Toshida et al.14 Briefly, an incision was made in the buccal region bilaterally, and the main lacrimal gland, Harderian gland, and nictitating membrane were removed. After applying an appropriate amount of Terramycin eye ointment (0.5% oxytetracycline hydrochloride/polymyxin B sulfate, Terramycin; Pfizer Japan, Tokyo, Japan), each incision was sutured. For 3 days after surgery, Terramycin eye ointment and tobramycin eye drops (0.3% tobracin; Nitto Medic, Toyama, Japan) were applied, while 0.2 mL of 4% gentamicin sulfate for injection (Gentacin; MSD, Whitehouse Station, NJ, USA) was administered intramuscularly. The animals were followed up for 4 weeks after lacrimal gland resection, and those with evidence of dry eyes (inadequate tear volume, decreased goblet cell density in the lower eyelid, and keratoconjunctival epithelial damage) were used in this study.
Alkaline injury to the lower palpebral conjunctiva
At 4 weeks after lacrimal gland resection, animals were placed under general anesthesia by intramuscular administration of a mixture of ketamine hydrochloride (100 mg/kg) and xylazine hydrochloride (10 mg/kg) and topical anesthesia with 0.4% oxybuprocaine hydrochloride (Benoxil; Santen Pharmaceutical Co, Ltd, Osaka, Japan). Alkaline injury was induced by placing a piece of filter paper 5 mm in diameter (Whatman Qualitative 1; GE Healthcare, Little Chalfont, Buckinghamshire, UK) soaked in 0.5% sodium hydroxide for 30s on the central part of the lower palpebral conjunctiva bilaterally. Exfoliated cells and excess sodium hydroxide solution were removed by rinsing the lower palpebral conjunctiva with 0.9 g/dL (0.9%) sodium chloride solution.
Topical application and observation
Twenty-eight rabbits were randomly divided into 4 groups of 7 animals each. In 3 groups (21 animals), retinol palmitate (VApal) (DMS Nutrition Japan, Inc, Tokyo, Japan) was applied to one eye at concentrations of 500, 1000, or 1500 IU/mL (eye drops in which VApal was dissolved with a surfactant (0.5% polyoxyethylene hydrogenated castor oil 60) in 0.9% sodium chloride solution) and vehicle was applied to the other eye. Topical application was performed six times daily (100 μL) for 7 days beginning immediately after alkaline injury. The frequency of topical application was set at six times daily to ensure sufficient delivery of VApal.5
In the other group (seven animals), VApal (1500 IU/mL) was applied to one eye and 0.1% hyaluronic acid eye drops (Hyalein Mini 0.1; Santen Pharmaceutical Co, Ltd, Osaka, Japan) were applied to the other eye six times daily (100 μL) for 7 days beginning immediately after alkaline injury. Fluorescein staining and rose bengal staining were performed before alkaline injury, as well as at 3 and 7 days after treatment, and the severity of keratoconjunctival damage was scored according to the criteria of Lemp15 and van Bijsterveld16 under blinded conditions.
Histological evaluation
Histological evaluation was performed in 3/7 animals that were selected at random from the group in which VApal (1500 IU/mL) was applied to one eye and vehicle was applied to the other eye. Seven days later, these animals were sacrificed using an overdose of anesthesia (ketamine hydrochloride and xylazine hydrochloride) and the cornea and the lower palpebral conjunctiva were removed and fixed in formalin. Paraffin sections (5-μm thick) were prepared, stained with hematoxylin and eosin (HE), or periodic acid–Schiff (PAS) stain, and observed under a light microscope in at least three randomly selected visual fields.
Evaluation of goblet cell density
Conjunctival goblet cell density in lower palpebral area was evaluated by impression cytology before alkaline injury and at 3 and 7 days after treatment as described previously.14 Briefly, after the animals were placed under general anesthesia with a mixture of ketamine hydrochloride (100 mg/kg) and xylazine hydrochloride (10 mg/kg) plus topical anesthesia with 0.4% oxybuprocaine hydrochloride (Benoxil), a nitrocellulose membrane (Bio- Rad Laboratories, Hercules, CA) was applied to the lower palpebral conjunctiva, fixed with formalin, and stained with PAS reagent to reveal goblet cells. After computer capture of light microscope images, the number of PAS-positive spots was counted using Scion image software (Beta 4.02; Scion Corp, Frederick, MD). The density of PAS-positive spots was determined in three visual fields that were selected randomly. Then the mean was calculated and converted to cell density per unit area (1 mm2).
Measurement of tear volume
Tear volume was measured by Schirmer’s tear test in all animals before alkaline injury of the lower palpebral conjunctiva. Briefly, at 5 minutes after application of topical anesthesia with 0.4% oxybuprocaine hydrochloride (Benoxil), a paper strip for Schirmer’s test was inserted into the temporal conjunctival sac of the lower eyelid and the tear volume was measured for 5 minutes. The strip was then removed from the lower palpebral conjunctiva, and the value reached on the strip was recorded. Tear volume was also measured in this manner after 3 and 7 days in the group of seven animals receiving VApal (1500 IU/mL) in one eye and the vehicle in the other eye.
Statistical analysis
Fluorescein and rose bengal staining scores were compared using Wilcoxon’s test between the VApal 500, 1000, and 1500 IU/mL groups and the vehicle group, and between the VApal 1500 IU/mL group and the 0.1% hyaluronic acid group. For the density of conjunctival goblet cells, analysis of variance was performed using Bartlett’s test (to confirm the homogeneity of variance) to compare the VApal 500, 1000, and 1500 IU/mL groups with the vehicle group, followed by the Tukey–Kramer multiple comparison test for comparison of all groups. Homogeneity of variance was assessed using the F-test for data on the density of conjunctival goblet cells in the group of seven animals receiving topical VApal (1500 IU/mL) and 0.1% hyaluronic acid, as well as for the tear volume in the group of seven animals receiving topical VApal (1500 IU/mL) and vehicle. If a significant difference was not found (if the variance was equal), Student’s t-test was performed for comparisons between the two groups. If the variance was not equal, the Aspin–Welch t-test was employed for comparisons between the two groups. In all analyses, P < 0.05 (two-tailed) was considered to be statistically significant. Data are shown as the mean ± standard error.
Results
Effect of retinol palmitate (VApal) eye drops on keratoconjunctival epithelial damage
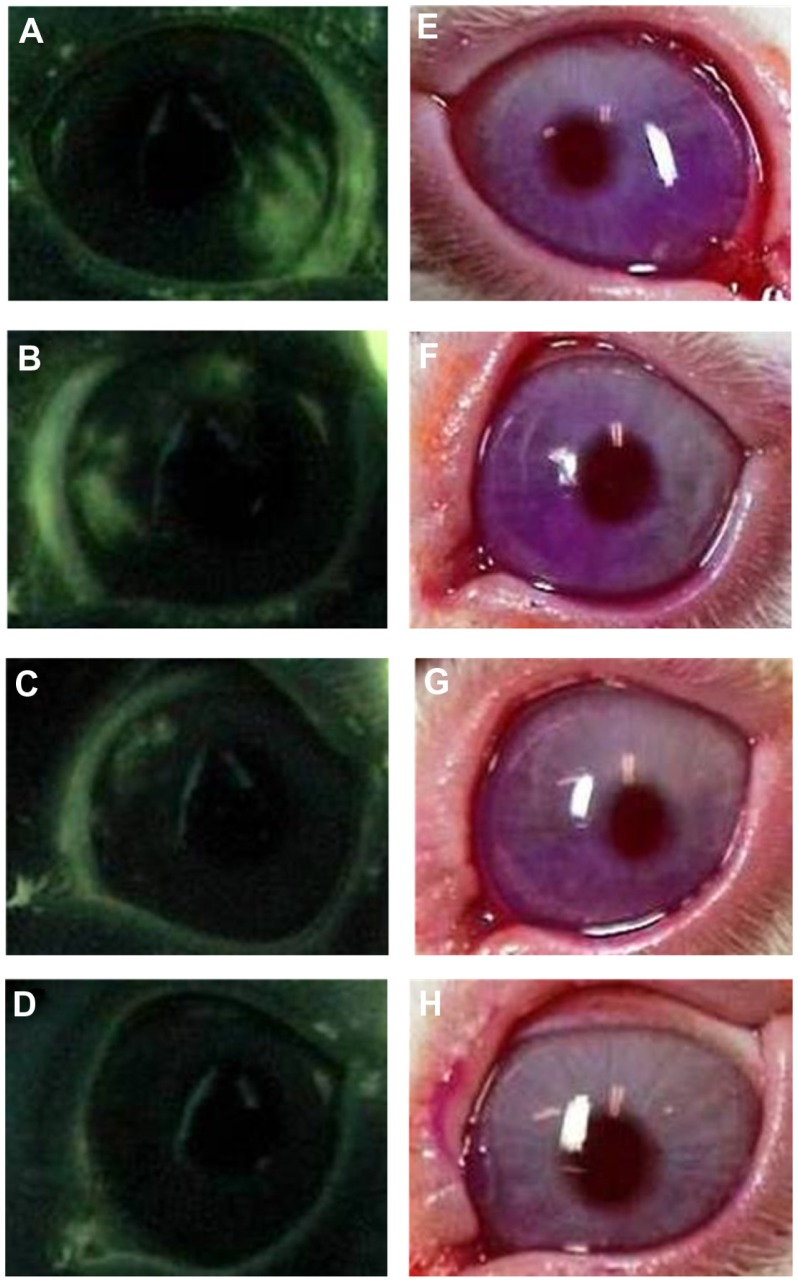
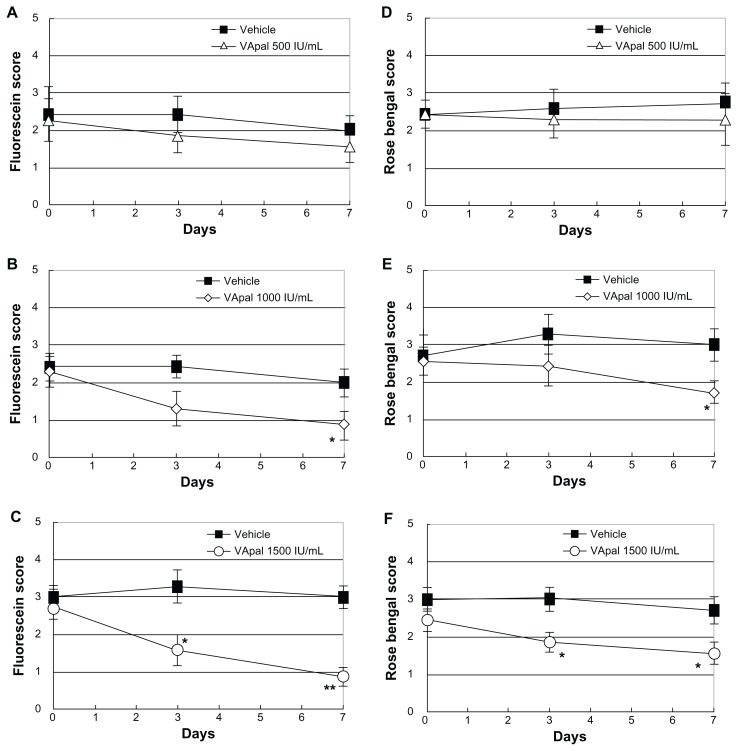
Figure 1 shows slit-lamp microscopic photos of the anterior segment of the eye with fluorescein staining and rose bengal staining at 7 days of treatment with the VApal (500, 1000, or 1500 IU/mL) or vehicle. Figure 2 shows the changes of the fluorescein and rose bengal staining scores in the three VApal groups and the vehicle group. In the 500 IU/mL VApal group, the fluorescein staining score showed no decrease compared with the score in the vehicle group throughout the study period (Figure 2A). However, it showed a significant decrease compared with the score in the vehicle group at 7 days (P < 0.05) in the 1000 IU/mL VApal group and at both 3 days (P < 0.05) and 7 days (P < 0.01) in the 1500 IU/mL VApal group (Figure 2B and C). Similarly to the fluorescein score, the rose bengal staining score showed no decrease in the 500 IU/mL VApal group compared with the vehicle group (Figure 2D), but showed a significant decrease compared with the vehicle group at 7 days (P < 0.05) in the 1000 IU/mL VApal group and at both 3 days (P < 0.05) and 7 days (P < 0.05) in the 1500 IU/mL VApal group (Figure 2E and F).
Figure 1.
Slit-lamp microscopic images of fluorescein (A–D) and rose bengal (E–H) staining in rabbit eyes at 7 days of treatment with the VApal or the vehicle. Corneal findings stained with fluorescein in 500 IU/mL (B), 1000 IU/mL (C) and 1500 IU/mL (D) of VApal-treated eye and the vehicle-treated eye (A), stained with rose bengal in 500 IU/mL (F), 1000 IU/mL (G), and 1500 IU/mL (H) of VApal-treated eye and vehicle-treated eye (E).
Abbreviation: VApal, vitamin A retinol palmitate.
Figure 2.
Changes of the fluorescein score (A–C) and rose bengal score (D–F) treated with the VApal or vehicle. The fluorescein score and rose bengal score in VApal-treated eyes: Concentrations of VApal are 500 IU/mL (Δ) (A and D), 1000 IU/mL (⋄) (B and E) and 1500 IU/mL (○) (C and F) respectively.
Notes: All data were compared to vehicle-treated eyes (■). Significant decreases of both fluorescein score and rose bengal score were shown in both 1000 IU/mL and 1500 IU/mL VApal-treated eyes compared to the vehicle-treated eyes after 7 days. *P < 0.05; **P < 0.01. All data are mean ± SE (n = 7).
Abbreviations: VApal, vitamin A retinol palmitate; SE, standard error.
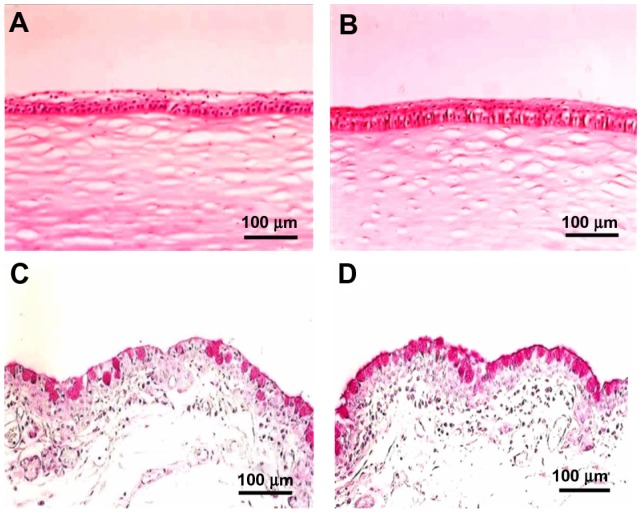
Histological evaluation after topical application of VApal
Figure 3 shows the cornea (HE stain) and the lower palpebral conjunctiva (PAS stain) at 7 days after starting treatment in the VApal (1500 IU/mL) and vehicle groups. Histological examination revealed recovery of the corneal epithelium after 7 days in the 1500 IU/mL VApal group, compared with the persistence of damage (exfoliation of superficial corneal cells) in the vehicle group (Figure 3A and B). PAS staining revealed the recovery of mucin-producing lower palpebral conjunctival goblet cells in the 1500 IU/mL VApal group (Figure 3C and D).
Figure 3.
Light microscopic images showing the structure of the cornea stained with HE (A and B), and the lower tarsal conjunctiva stained with PAS (C and D). Corneal epithelium and conjunctival epithelium were recovered, goblet cells were increased at 7 days after treatment with 1500 IU/mL of VApal (B and D) compared with vehicle-treated eye (A and C).
Note: Bars are 100 μm.
Abbreviations: PAS, periodic acid–Schiff; VApal, vitamin A retinol palmitate.
Density of lower palpebral conjunctival goblet cells after topical application of VApal; impression cytology
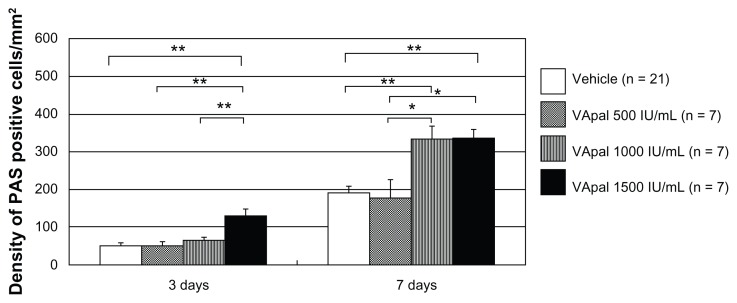
Figure 4 shows the density of goblet cells (PAS-positive cells) per unit area (1 mm2) of the lower palpebral conjunctiva after 3 and 7 days in the VApal and vehicle groups. The density of conjunctival goblet cells showed no significant difference after 3 and 7 days between the 500 IU/mL VApal group (49.3 ± 11.4 and 179.8 ± 48.0 cells/mm2 at 3 and 7 days, respectively) and the vehicle group (51.3 ± 7.1 and 190.8 ± 19.0 cells/mm2 at 3 and 7 days, respectively). However, the density showed a significant increase (P < 0.01) compared with that in the vehicle group after 7 days (332.6 ± 34.9 cells/mm2) in the 1000 IU/mL VApal group and after 3 days (131.6 ± 16.6 cells/mm2) and 7 days (335.8 ± 24.1 cells/mm2) in the 1500 IU/mL VApal group. It also showed a significant difference after 3 days (P < 0.01) and 7 days (P < 0.05) between the 500 and 1500 IU/mL VApal groups, after 3 days (P < 0.01) between the 1000 and 1500 IU/mL VApal groups, and after 7 days (P < 0.05) between 500 and 1000 IU/mL VApal groups.
Figure 4.
Density of PAS positive cells in lower eyelid conjunctiva.
Notes: Data were averaged from randomly selected three visual fields after treatments in vehicle-treated eyes (n = 21) and in VApal-treated eyes (n = 7 each). The VApal-treated eyes showed significantly greater numbers of density compared with the vehicle-treated eyes at 3 and 7 days. *P < 0.05; **P < 0.01. All data are mean ± SE.
Abbreviations: PAS, periodic acid–Schiff; SE, standard error; VApal, vitamin A retinol palmitate.
The density of lower palpebral conjunctival goblet cells was 651.2 ± 21.9 cells/mm2 (n = 14) in normal rabbit eyes and 525.9 ± 19.9 cells/mm2 (n = 42: all VApal-treated animals) in the eyes of rabbits with lacrimal gland resection (before alkaline injury of the lower palpebral conjunctiva) (data not shown). Therefore, in animals with lacrimal gland resection, the density of lower palpebral conjunctival goblet cells was decreased to approximately 80% of that in normal eyes. In this model, the density of lower palpebral conjunctival goblet cells was always lower than that in normal eyes or eyes with lacrimal gland resection (before alkaline injury).
Evaluation of tear volume after topical application of VApal
Tear volume (Schirmer’s tear test) was compared between before treatment and 3 or 7 days after the start of treatment with VApal at 1500 IU/mL. No change was observed from before alkaline injury and VApal treatment of the conjunctiva (8.6 ± 0.7 mm) at 3 days (9.4 ± 0.4 mm) and 7 days (8.2 ± 1.2 mm) after the start of VApal administration. There was little difference between the VApal group and the vehicle group (8.6 ± 1.3 before alkaline injury and vehicle treatment and 9.1 ± 1.1 and 8.3 ± 1.1 mm at 3 and 7 days, respectively). Thus, topical application of VApal had no influence on tear volume.
The tear volume was 15.5 ± 1.2 mm (n = 14) in normal rabbit eyes and 7.5 ± 0.5 mm (all 42 animals in the topical application study) in the eyes with lacrimal gland resection before alkaline injury of the conjunctiva. Therefore, tear volume in the rabbits with lacrimal gland resection was decreased to approximately 50% of the volume in normal rabbits.
Comparison with hyaluronic acid eye drops
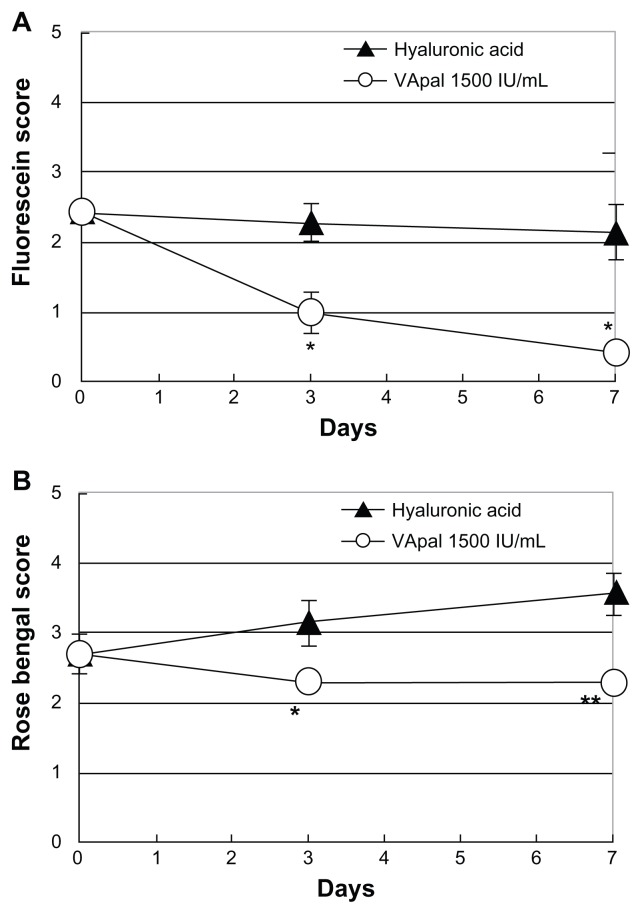
Figure 5 shows changes in fluorescein and rose bengal staining scores during treatment with VApal at 1500 IU/mL or 0.1% hyaluronic acid 6 times daily for 7 days. In the 1500 IU/mL VApal group, both the fluorescein and rose bengal staining scores showed a significant decrease compared with the scores in the hyaluronic acid group after 3 days (P < 0.05) and 7 days (fluorescein staining score: P < 0.05, rose bengal staining score: P < 0.01) of treatment (Figure 5A and B). When the density of lower palpebral conjunctival goblet cells (PAS-positive cells) was compared between the 1500 IU/mL VApal group and the hyaluronic acid group at 7 days after starting treatment, the density was significantly higher (P < 0.05) in the 1500 IU/mL VApal group (524.3 ± 34.2 cells/mm2) than in the hyaluronic acid group (375.3 ± 54.8 cells/mm2).
Figure 5.
Comparison of the effects with VApal and hyaluronic acid eye drops. The fluorescein score (A) and rose bengal score (B) in the 1500 IU/mL of VApal-treated eyes (○) showed significant decrease compared with the scores in the hyaluronic acid-treated eyes (▲) at 3 and 7 days.
Notes: *P < 0.05; **P < 0.01. All data are mean ± SE (n = 7).
Abbreviations: SE, standard error; VApal, vitamin A retinol palmitate.
Discussion
Dry eyes are defined as “a chronic disease of tears and of the keratoconjunctival epithelium which is induced by different causes and is associated with eye discomfort and visual dysfunction”.15,17 The number of patients with dry eyes is estimated to be more than 8 million in Japan and more than 10 million in the United States, with the number of patients with this disease increasing each year.18
For the treatment of dry eyes, artificial tears and hyaluronic acid eye drops are used to supply moisture and improve keratoconjunctival epithelial damage. In the development of new treatments for dry eyes, improving keratoconjunctival epithelial damage and quantitative and qualitative tear abnormalities has been emphasized,15,19 with a focus on the promotion of tear mucin production. Tear mucin is produced by conjunctival goblet cells and keratoconjunctival epithelial cells and forms a mucin layer under the tear film, playing an important role in protecting the ocular surface, stabilizing tears, and healing keratoconjunctival epithelial damage.20,21 Attempts have been made to apply the agents gefarnate14,22–24 and rebamipide,25–27 which promote not only gastric but also corneal mucin production, diquafosol, which is a P2Y2 receptor agonist inducing PAS-positive glycoproteins,28–30 and 15(S)-HETE (eicosanoid) that stimulates the production of keratoconjunctival epithelial mucin (MUC1).31 Attempts have also been made to apply DA-6034 (a flavonoid derivative that protects the gastric mucosa),32 ecabet sodium, and the anti-inflammatory agent cyclosporine.33–36
Vitamin A is also known not only to regulate the proliferation and differentiation of corneal epithelial cells and preserved conjunctival goblet cells but also to promote tear mucin production.1,11,37 Clinical studies have already indicated that eye ointments and eye drops containing VA (such as retinoic acid and VApal) cure keratoconjunctival disease by improving keratoconjunctival epithelial keratosis and increasing the density of conjunctival goblet cells.1–5
In the present study, we investigated the efficacy of VApal eye drops in a lacrimal gland-removed dry eye rabbit model. This model is considered to closely resemble the pathologic state of dry eyes associated with both tear abnormalities and keratoconjunctival epithelial damage, since the tear volume was decreased to approximately 50% of the normal value and the density of conjunctival goblet cells was decreased to approximately 80% of the normal value, resulting in the occurrence of keratoconjunctival epithelial damage.
In order to determine the usefulness of VApal for dry eyes, investigation after its topical application (at 500, 1000, or 1500 IU/mL) and comparison with 0.1% hyaluronic acid eye drops, an existing treatment for dry eyes, were performed using this animal model. The results suggested that topical application of VApal is effective for the improving of keratoconjunctival epithelial damage associated with tear abnormalities because it promoted healing of corneal epithelial damage and recovery of conjunctival goblet cells in our rabbit model of dry eyes induced by lacrimal gland resection (Figures 1, 2, and 4), as previously reported in a rabbit model of keratoconjunctival epithelial damage.12
No improvement was noted after topical application of VApal at 500 IU/mL (Figure 2A and D). However, both the fluorescein and rose bengal staining scores showed significant dose-dependent improvement after topical application of VApal at 1000 IU/mL (7 days) and at 1500 IU/mL (3 and 7 days), indicating that use of VApal at higher concentrations (1000 or 1500 IU/mL) is effective for treating keratoconjunctival epithelial damage associated with tear abnormalities in the present model of dry eyes (Figure 2B, C, E, and F). With topical application of VApal at 1500 IU/mL, improvement occurred sooner (at 3 days) after starting treatment and the recovery of keratoconjunctival epithelial damage was similar to the change in the number of PAS-positive goblet cells in the lower palpebral conjunctiva (Figure 4). Because it has been reported that topical application of mucin promotes the healing of corneal damage in rabbits,38 the results of this study suggest that VApal may promote the differentiation of conjunctival goblet cells and improve keratoconjunctival epithelial damage via recovery of the goblet cell population, promotion of mucin production from goblet cells, and stabilization of the tear film due to the promotion of tear mucin layer production.
We previously reported that keratoconjunctival epithelial damage was improved after recovery of conjunctival goblet cells and improvement of the mucin layer, resulting from topical application of VApal which promoted the healing of keratoconjunctival epithelial damage and recovery of conjunctival goblet cells (with the rose bengal staining score improving earlier than the fluorescein staining score) in a rabbit model of keratoconjunctival epithelial damage induced by treatment with n-heptanol.12 In the animal model of lacrimal gland resection used in this study, however, there was no difference between the fluorescein and rose bengal staining scores with regard to the pattern of improvement after topical application of VApal. This was presumably related to a difference in severity of damage to conjunctival goblet cells. In the animal model of keratoconjunctival epithelial damage, not only the cornea but also the ocular and palpebral conjunctivae was damaged (conjunctival rose bengal staining score: approximately 6 points), while the animal model of lacrimal gland resection only featured conjunctival damage to part of the lower palpebral conjunctiva of the eye with lacrimal gland resection, and only slight damage affected the entire conjunctiva (conjunctival rose bengal staining score: approximately 2–3 points). As a result, improvement of the mucin layer by VApal as shown by the rose bengal staining score was not detected before improvement of the fluorescein staining score.
In this study, a comparison of VApal and 0.1% hyaluronic acid eye drops showed that topical application of VApal at 1500 IU/mL achieved significantly better improvement after 3 and 7 days compared with that following treatment with hyaluronic acid (Figure 5). It has been reported that hyaluronic acid improves tears because it retains a significant amount of water, has a long residence time, and can promote growth of the corneal epithelial cell layer39 and healing of corneal epithelial damage.40,41 On the other hand, VApal increases not only mucin production by the conjunctival epithelium but also hyaluronic acid production by the corneal epithelium.42 Because the comparison of VApal and hyaluronic acid showed that VApal was more effective, the mechanism involved in the healing of keratoconjunctival epithelial damage is considered to primarily involve promotion of mucin production by conjunctival goblet cells secondary to promotion of keratoconjunctival epithelial cell differentiation by VApal rather than an increase of corneal epithelial hyaluronic acid production by this agent.
In the present lacrimal gland resection rabbit model of dry eyes, topical application of VApal improved keratoconjunctival epithelial damage and a dose of 1500 IU/mL improved it more effectively than 0.1% hyaluronic acid eye drops. Therefore, it is suggested that VApal is effective for improving keratoconjunctival epithelial damage associated with tear abnormalities, such as dry eyes. Additionally, the mucin-producing effect of VApal has an important role in improving keratoconjunctival epithelial damage after topical application of VApal. However, it is unclear whether VApal has the same beneficial effects in human eyes without any side effects. It is necessary to conduct further studies at the protein or gene expression levels to elucidate the mechanisms underlying the increased production of mucin and healing of keratoconjunctival epithelial damage by VApal.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Kobayashi T, Tsubota K, Takamura E, Sawa M, Ohashi Y, Usui M. Effect of retinol palmitate as a treatment for dry eye: A cytological evaluation. Ophthalmologica. 1997;211(6):358–361. doi: 10.1159/000310829. [DOI] [PubMed] [Google Scholar]
- 2.Wright P. Topical retinoic acid therapy for disorders of the outer eye. Trans Ophthalmol Soc UK. 1985;104(8):869–874. [PubMed] [Google Scholar]
- 3.Soong HK, Martin NF, Wagoner MD, et al. Topical retinoid therapy for squamous metaplasia of various ocular surface disorders. A multicenter, placebo-controlled double-masked study. Ophthalmology. 1988;95(10):1442–1446. doi: 10.1016/s0161-6420(88)33009-5. [DOI] [PubMed] [Google Scholar]
- 4.Ohashi Y, Watanabe H, Kinoshita S, Hosotani H, Umemoto M, Manabe R. Vitamin A eye drops for superior limbic keratoconjunctivitis. Am J Ophthalmol. 1988;105(5):523–527. doi: 10.1016/0002-9394(88)90245-0. [DOI] [PubMed] [Google Scholar]
- 5.Ohashi Y, Mano T, Umemoto M, et al. Effects of vitamin A instillation on corneal and conjunctival disorders. Rinsho Ganka (Jpn J Clip Ophthalmol) 1987;41(4):368–369. Japanese. [Google Scholar]
- 6.Ubels JL, MacRae SM. Vitamin A is present as retinol in the tears of humans and rabbits. Curr Eye Res. 1984;3(6):815–822. doi: 10.3109/02713688409000793. [DOI] [PubMed] [Google Scholar]
- 7.Sullivan WR, McCulley JP, Dohlman CH. Return of goblet cells after vitamin A therapy in xerosis of the conjunctiva. Am J Ophthalmol. 1973;75(4):720–725. doi: 10.1016/0002-9394(73)90828-3. [DOI] [PubMed] [Google Scholar]
- 8.Pfister RR, Renner ME. The corneal and conjunctival surface in vitamin A deficiency: a scanning electron microscopy study. Invest Ophthalmol Vis Sci. 1978;17(9):874–883. [PubMed] [Google Scholar]
- 9.Kubo Y, Arimura A, Watanabe Y, Nakayasu K, Kanai A. Effect of vitamin A palmitate on vitamin A-deficient rabbits. Nippon Ganka Gakkai Zasshi (J Jpn Ophthalmol Soc) 1999;103(10):729–733. Japanese. [PubMed] [Google Scholar]
- 10.Tei M, Spurr-Michaud SJ, Tisdale AS, Gipson IK. Vitamin A deficiency alters the expression of mucin genes by the rat ocular surface epithelium. Invest Ophthalmol Vis Sci. 2000;41(1):82–88. [PubMed] [Google Scholar]
- 11.Kubo Y, Arimura A, Nakayasu K, Kanai A. Effect of vitamin A palmitate on the synthesis of mucins in cultured conjunctiva. Nippon Ganka Gakkai Zasshi (J Jpn Ophthalmol Soc) 1999;103(8):580–583. Japanese. [PubMed] [Google Scholar]
- 12.Toshida H, Odaka A, Koike D, Murakami A. Effect of retinol palmitate eye drops on experimental keratoconjunctival epithelial damage induced by n-heptanol in rabbit. Curr Eye Res. 2008;33(1):13–18. doi: 10.1080/02713680701827696. [DOI] [PubMed] [Google Scholar]
- 13.Ubels JL, Edelhauser HF, Foley KM, Liao JC, Gressel P. The efficacy of retinoic acid ointment for treatment of xerophthalmia and corneal epithelial wounds. Curr Eye Res. 1985;4(10):1049–1057. doi: 10.3109/02713688509003350. [DOI] [PubMed] [Google Scholar]
- 14.Toshida H, Nakata K, Hamano T, Nakamura M, Nguyen D, Beuerman R. Gefarnate stimulates goblet cell repopulation following an experimental wound to the tarsal conjunctiva in the dry eye rabbit. Adv Exp Med Biol. 2002;506(Pt A):353–357. doi: 10.1007/978-1-4615-0717-8_51. [DOI] [PubMed] [Google Scholar]
- 15.Lemp MA. Report of the National Eye Institute/Industry workshop on Clinical Trials in Dry Eyes. CLAO J. 1995;21(4):221–232. [PubMed] [Google Scholar]
- 16.van Bijsterveld OP. Diagnostic tests in the sicca syndrome. Arch Ophthalmol. 1969;82(1):10–14. doi: 10.1001/archopht.1969.00990020012003. [DOI] [PubMed] [Google Scholar]
- 17.The International Dry Eye WorkShop. The Definition and Classification of Dry Eye Disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop. Ocul Surf. 2007;5(2):75–92. doi: 10.1016/s1542-0124(12)70081-2. [DOI] [PubMed] [Google Scholar]
- 18.The International Dry Eye WorkShop. Research in dry eye: report of the Research Subcommittee of the International Dry Eye WorkShop. Ocul Surf. 2007;5(2):179–193. doi: 10.1016/s1542-0124(12)70086-1. [DOI] [PubMed] [Google Scholar]
- 19.Nelson JD. Diagnosis and treatment of the dry eye: a clinical perspective. Adv Exp Med Biol. 2002;506(Pt B):1067–1078. doi: 10.1007/978-1-4615-0717-8_151. [DOI] [PubMed] [Google Scholar]
- 20.Kessing SV. Mucous gland system of the conjunctiva. A quantitative normal anatomical study. Acta Ophthalmol. 1968;95(Suppl):1. [PubMed] [Google Scholar]
- 21.Watanabe H, Fabricant M, Tisdale AS, Spurr-Michaud SJ, Lindberg K, Gipson IK. Human corneal and conjunctival epithelia produce a mucin-like glycoprotein for the apical surface. Invest Ophthalmol Vis Sci. 1995;36(2):337–344. [PubMed] [Google Scholar]
- 22.Toshida H, Nakata K, Hamano T, Nakamura M, Nguyen D, Beuerman RW. Effect of gefarnate on the ocular surface in squirrel monkeys. Cornea. 2002;21(3):292–299. doi: 10.1097/00003226-200204000-00011. [DOI] [PubMed] [Google Scholar]
- 23.Hamano T. Dry eye treatment with eye drops that stimulate mucin production. Adv Exp Med Biol. 1998;438:965–968. doi: 10.1007/978-1-4615-5359-5_137. [DOI] [PubMed] [Google Scholar]
- 24.Nakamura M, Endo K, Nakata K, Hamano T. Gefarnate stimulates secretion of mucin-like glycoproteins by corneal epithelium in vitro and protects corneal epithelium from desiccation in vivo. Exp Eye Res. 1997;65(4):569–574. doi: 10.1006/exer.1997.0366. [DOI] [PubMed] [Google Scholar]
- 25.Urashima H, Okamoto T, Takeji Y, Shinohara H, Fujisawa S. Rebamipide increases the amount of mucin-like substances on the conjunctiva and cornea in the N-acetylcysteine -treated in vivo model. Cornea. 2004;23(6):613–619. doi: 10.1097/01.ico.0000126436.25751.fb. [DOI] [PubMed] [Google Scholar]
- 26.Takeji Y, Urashima H, Aoki A, Shinohara H. Rebamipide increases the mucin-like glycoprotein production in corneal epithelial cells. J Ocul Pharmacol Ther. 2012;28(3):259–263. doi: 10.1089/jop.2011.0142. [DOI] [PubMed] [Google Scholar]
- 27.Urashima H, Takeji Y, Okamoto T, Fujisawa S, Shinohara H. Rebamipide increases mucin-like substance contents and periodic acid Schiff reagent-positive cells density in normal rabbits. J Ocul Pharmacol Ther. 2012;28(3):264–270. doi: 10.1089/jop.2011.0147. [DOI] [PubMed] [Google Scholar]
- 28.Murakami T, Fujihara T, Nakamura M, Nakata K. P2Y2 receptor elicits PAS-positive glycoprotein secretion from rabbit conjunctival goblet cells in vivo. J Ocul Pharmacol Ther. 2003;19(4):345–352. doi: 10.1089/108076803322279390. [DOI] [PubMed] [Google Scholar]
- 29.Fujihara T, Murakami T, Nagano T, Nakamura M, Nakata K. INS365 suppresses loss of corneal epithelial integrity by secretion of mucin-like glycoprotein in a rabbit short-term dry eye model. J Ocular Pharmacol Ther. 2002;18(4):363–370. doi: 10.1089/10807680260218524. [DOI] [PubMed] [Google Scholar]
- 30.Fujihara T, Murakami T, Fujita H, Nakamura M, Nakata K. Improvement of corneal barrier function by the P2Y2 agonist INS365 in a rat dry eye model. Invest Ophthalmol Vis Sci. 2001;42(1):96–100. [PubMed] [Google Scholar]
- 31.Gamache DA, Wei ZY, Weimer LK, Miller ST, Spellman JM, Yanni JM. Corneal protection by the ocular mucin secretagogue 15(S)-HETE in a rabbit model of desiccation-induced corneal defect. J Ocular Pharmacol Ther. 2002;18(4):349–361. doi: 10.1089/10807680260218515. [DOI] [PubMed] [Google Scholar]
- 32.Choi SM, Lee YG, Seo MJ, Kang KK, Ahn BO, Yoo M. Effects of DA-6034 on aqueous tear fluid secretion and conjunctival goblet cell proliferation. J Ocular Pharmacol Ther. 2009;25(3):209–213. doi: 10.1089/jop.2008.0126. [DOI] [PubMed] [Google Scholar]
- 33.Kunert KS, Tisdale AS, Gipson IK. Goblet cell numbers and epithelial proliferation in the conjunctiva of patients with dry eye syndrome treated with cyclosporine. Arch Ophthalmol. 2002;120(3):330–337. doi: 10.1001/archopht.120.3.330. [DOI] [PubMed] [Google Scholar]
- 34.Toshida H, Nguyen DH, Beuerman RW, Murakami A. Neurologic evaluation of acute lacrimomimetic effect of cyclosporine in an experimental rabbit dry eye model. Invest Ophthalmol Vis Sci. 2009;50(6):2736–2741. doi: 10.1167/iovs.08-1880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Toshida H, Nakayasu K, Kanai A. Effect of cyclosporin A eye drops on tear secretion in rabbit. Jpn J Ophthalmol. 1998;42(3):168–173. doi: 10.1016/s0021-5155(98)00003-3. [DOI] [PubMed] [Google Scholar]
- 36.Kim EC, Choi JS, Joo CK. A comparison of vitamin a and cyclosporine a 0.05% eye drops for treatment of dry eye syndrome. Am J Ophthalmol. 2009;147(2):206–213. doi: 10.1016/j.ajo.2008.08.015. [DOI] [PubMed] [Google Scholar]
- 37.Hori Y, Spurr-Michaud SJ, Russo CL, Argueso P, Gipson IK. Effect of retinoic acid on gene expression in human conjunctival epithelium: Secretory phospholipase A2 mediates retinoic acid induction of MUC16. Invest Ophthalmol Vis Sci. 2005;46(11):4050–4061. doi: 10.1167/iovs.05-0627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shigematsu T, Shimizu Y, Majima Y. Effects of mucin ophthalmic solution on epithelial wound healing in rabbit cornea. Ophthalmic Res. 1997;29(2):61–66. doi: 10.1159/000267998. [DOI] [PubMed] [Google Scholar]
- 39.Nishida T, Nakamura M, Mishima H, Otori T. Hyaluronan stimulates corneal epithelial migration. Exp Eye Res. 1991;53(6):753–758. doi: 10.1016/0014-4835(91)90110-z. [DOI] [PubMed] [Google Scholar]
- 40.Nakamura M, Hikida M, Nakano T. Concentration and molecular weight dependency of rabbit corneal epithelial wound healing on hyaluronan. Curr Eye Res. 1992;11(10):981–986. doi: 10.3109/02713689209033496. [DOI] [PubMed] [Google Scholar]
- 41.Nakamura M, Sato N, Chikama TI, Hasegawa Y, Nishida T. Hyaluronan facilitates corneal epithelial wound healing in diabetic rats. Exp Eye Res. 1997;64(6):1043–1050. doi: 10.1006/exer.1997.0302. [DOI] [PubMed] [Google Scholar]
- 42.Toshida H, Tabuchi N, Koike D, et al. The effects of vitamin A compounds on hyaluronic acid released from cultured rabbit corneal epithelial cells and keratinocytes. J Nutr Sci Vitaminol. 2012;58(4):223–229. doi: 10.3177/jnsv.58.223. [DOI] [PubMed] [Google Scholar]