SUMMARY
Haemangiomas represent the most common benign tumours in infancy, affecting 1-2% of newborns. The present meta-analysis aimed to critically review the current evidence on the efficacy of propranolol in the management of airway haemangiomas, and explore potential adverse events and treatment failures. A literature review was performed in Medline and other available database sources, along with critical analysis of pooled data. Seventeen studies were included in the analysis. No study represented Level I evidence. The total number of treated patients was 61; 14 patients received propranolol as single-treatment. The comparative effectiveness of propranolol vs. systemic steroids was documented in 35 children, and showed superior outcome in the vast majority (94%, p < 0.001). The mean obstruction before propranolol administration was 72%, and after intervention was 20% (p < 0.001). The mean referral-age for children with airway haemangiomas was 2.4 months, the mean starting-age of propranolol treatment was 5.1 months and the mean follow-up period was 8.4 months. Four children failed to respond (6.5%), and in seven the haemangioma relapsed after discontinuation of treatment (11.5%). The results of the present study suggest that propranolol can be recommended for the treatment of airway haemangiomas, as it was found to be effective and outperformed the previously-considered gold standard treatment methods, with fewer side-effects. Immediate treatment with propranolol should be initiated once a diagnosis of symptomatic airway haemangioma is confirmed, and cardiovascular assessment has been performed. Children should remain on propranolol until the haemangioma enters the phase of involution. Active parental monitoring is essential to ensure treatment safety.
KEY WORDS: Haemangioma, Airway, Propranolol, Steroids
RIASSUNTO
Gli emangiomi rappresentano i tumori benigni più comuni durante l'infanzia, interessando l'1-2% dei neonati. L'obbiettivo della nostra meta-analisi è stato una revisione critica delle attuali evidenze sull'efficacia del propranololo nel trattamento degli emangiomi delle prime vie aeree, sottolineando in particolare i potenziali eventi avversi e i fallimenti terapeutici. Un'analisi critica è stata fatta su dati raccolti dopo un'accurata revisione della letteratura effettuata mediante Medline e gli altri database disponibili. Diciassette studi sono stati inclusi nell'analisi. Nessuno studio ha riportato prove di I livello. In totale sono stati trattati 61 pazienti di cui 14 avevavo ricevuto propanololo come singolo trattamento. In 35 bambini è stato possibile confrontare l'efficacia del propanololo con quella degli steroidi per via sistemica dimostrando la superiorità del primo nella maggioranza dei casi (94% - p < 0,001). L'ostruzione media prima della somministrazione di propranololo era del 72%, dopo la terapia invece del 20% (p < 0,001). L'età media alla diagnosi dei bambini affetti da emangioma delle prime vie aeree era di 2,4 anni, l'età media di inizio del trattamento con propranololo era invece di 5,1, e il periodo medio di follow-up di 8,4 mesi. Quattro bambini non hanno risposto (6,5%), e in sette casi l'emangioma ha recidivato dopo interruzione del trattamento (11,5%). In conclusione, i risultati della presenta metanalisi dimostrano che il propranololo può essere raccomandato per il trattamento degli emangiomi delle prime vie aeree, in quanto di efficacia superiore e con minori effetti collaterali rispetto ai metodi precedentemente considerati come il gold standard terapeutico per questo tipo di patologia. Il trattamento con propranololo dovrebbe essere iniziato non appena confermata la diagnosi di emangioma sintomatico delle prime vie aeree ed effettuata una valutazione cardiovascolare. I bambini devono essere trattati con propranololo fino a quando l'emangioma entra nella fase di involuzione. Il monitoraggio attivo dei genitori è essenziale per garantire la sicurezza del trattamento.
Introduction
Haemangiomas represent the most common benign tumours in infancy, affecting 1-2% of newborns 1. Although they are often confused with other vascular malformations, they demonstrate a unique course characterized by proliferation during the first year of life, and involution thereafter.
Fortunately, many haemangiomas are superficial involving only skin and subcutaneous tissue, and can be allowed to follow their self-limiting natural course, and resolve with minimal sequelae. However, approximately 10% of haemangiomas can affect the child's quality of life due to aesthetic reasons or cause functional impairment, which can even be life-threatening 2.
Airway haemangiomas in particular may lead to feeding difficulties, barking cough, stridor, respiratory distress and even acute airway obstruction. Hence, such haemangiomas usually require early and aggressive treatment; however, the treatment modalities employed so far have been associated with their own co-morbidities and potential long-term adverse effects for the developing child. In other words, the traditional management (medical or surgical) of severe or life threatening airway haemangiomas may have a serious impact on the child's quality of life, or be associated with severe complications.
Recently, Leaute-Lambreze et al. reported the accidental regression of an infantile haemangioma following the administration of propranolol for steroid-induced hypertrophic cardiomyopathy 3. This had led other otolaryngology centres to try the same method of management in order to achieve a good clinical result with minimal complications. The first related studies, usually case reports or case series, were very encouraging claiming that a new revolutionary method of management had been discovered 4-7.
The aim of the present meta-analysis is to critically review the current evidence on the efficacy of propranolol in the management of airway haemangiomas. Adverse events and treatment failures are also explored.
Materials and Methods
An extensive search of the literature was performed in Medline and other available database sources, establishing two main categories of outcomes:
assessment of the clinical effectiveness of systematically administered propranolol in halting the progress of growing airway haemangiomas, and
comparison of the clinical outcome of propranolol versus the systemic, or intralesional steroid administration as a first-line approach for the treatment of proliferating airway haemangiomas.
Using this framework of results, the retrieved studies were critically appraised according to evidence-based guidelines for the categorization of medical studies (Tab. I).
Table I.
Levels of evidence regarding in studies that investigate the results of treatment.
| Category of evidence | Study design |
|---|---|
| Level I |
|
| Level II |
|
| Level III |
|
| Level IV |
|
| Level V |
|
In addition, three secondary end-points were also analysed:
clinical effectiveness of propranolol in eradication of proliferating airway haemangiomas,
clinical effectiveness of propranolol compared to previously utilized second-line treatments for infantile haemangiomas, and
protocol for the safe administration of propranolol, and appropriate patient follow-up until complete resolution of the disease.
During the search, the keywords "haemangioma", "infantile", "airway", "proliferation", "propranolol", "steroids", and "treatment" were utilized. The keywords "haemangioma", "airway", and "propranolol" were considered primary, and were either combined to each of the other keywords individually, or used in groups of three. In addition, reference lists from the retrieved articles were manually searched. Language restrictions limited the search to English-language articles only.
The clinical effectiveness of propranolol was assessed using chi-square analysis. Chi-square was also used to determine the comparative effectiveness of propranolol vs. steroid administration. The degree of airway obstruction before and after treatment with propranolol was compared using a paired t-test. The correlation coefficient between treatment with propranolol and the change in the percentage of airway obstruction post-treatment was calculated using linear regression. Statistical importance was accepted at the level of P < 0.05.
Results
Eighteen studies met the defined criteria and were initially included in study selection. Among these studies, one included patients with overlapping haemangiomas, frequently involving more than one anatomic site in the same patient, without, however, clear identification of the number of patients with airway haemangiomas. This study was therefore excluded from the analysis of pooled data.
Overall, one multi-centre retrospective cohort study, two prospective cohort and two retrospective studies, six caseseries, and six case reports were systematically analyzed. Two studies represented Level II, three studies Level III and 12 studies Level IV evidence (Tab. I).
The total number of treated patients was 61; among these 35 had exclusive airway lesions. Co-existing segmental distribution of haemangiomas was present in 11 children, cutaneous lesions in nine, deep neck/mediastinal components in four and finally PHACE S syndrome (posterior fossa malformations, haemangiomas, arterial anomalies, coarctation of the aorta/cardiac defects, eye abnormalities and sternal defects) in two of the treated children. The vast majority of patients (n = 43) received 2 mg of oral propranonolol per day divided in 2 or 3 doses; a dose of 3 mg per day was given in 10 children, whereas the administered dose was not reported in 4 cases.
Fourteen patients received propranolol as single treatment. Four patients were treated with another selective β-blocker (acebutalol), but showed similar responses, and were hence included in further analysis of treatment outcomes. The comparative effectiveness of propranolol vs. systemic steroids was documented in 35 children (Tab. II). Propranolol treatment outperformed steroids in the vast majority of patients (33 cases, 94%), whereas steroids showed superior outcome only in two cases (6%). The result was statistically significant (p < 0.001). Propranolol was also used as salvage therapy in 10 children previously treated with laser, and in three children who had previously received vincristine; better results demonstrating a clear advantage for propranolol were reported in all of these patients.
Table II.
Comparative effectiveness of propranololol vs. systemic steroids in the management of airway haemangiomas (raw data).
| Case | Systemic prednisolone | Steroid effectiveness | Propranolol | β-blocker efficiency | Additional treatment |
|---|---|---|---|---|---|
| 1 | High doses (details missing) | No | 3 mg/kg/day | Success | No |
| 2 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 3 | 2 mg/kg/day | No | 2 mg/kg/day≠ | Success | No |
| 4 | 3 mg/kg/day | No | 3 mg/kg/day | Success | No |
| 5 | 2 mg/kg/day | No | 3 mg/kg/day | Success | No |
| 6 | 2 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 7 | 2 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 8 | 2 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 9 | 2 mg/kg/day | No | 3 mg/kg/day | Success | No |
| 10 | 1 mg/kg/day | No | 8 mg/kg/day† | Success | No |
| 11 | 1 mg/kg/day | No | 8 mg/kg/day† | Success | No |
| 12 | 1.5 mg/kg/day | No | 3 mg/kg/day | Success | No |
| 13 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 14 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 15 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 16 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 17 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 18 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 19 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 20 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 21 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 22 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 23 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 24 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 25 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 26 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 27 | 0.1 mg/kg/day‡ | No | 2 mg/kg/day | Success | No |
| 28 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 29 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 30 | 3 mg/kg/day | No | 2 mg/kg/day | Success | No |
| 31 | 3 mg/kg/day | Yes | 2 mg/kg/day | Failure | No |
| 32 | High doses (details missing) | No | 2 mg/kg/day | Success | No |
| 33 | 100 μg/kg/day§ | No | 2 mg/kg/day | Success | No |
| 34 | High doses (details missing) | No | Details missing | Success | No |
| 35 | High doses (details missing) | Yes | Details missing | Failure | No |
the patient was initially treated with propranolol, but later received acebutalol, due to asthmatic attack
the patient received acebutalol
the patient received betamethasone
the patient received dexamethasone
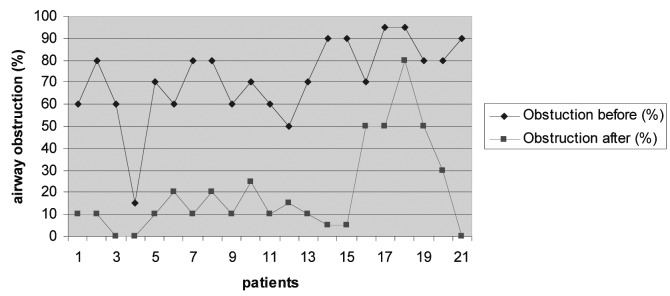
Drawing on the degree of improvement of airway obstruction, this was clearly documented as a respective percentage in 21 patients. The mean obstruction before the administration of propranolol was 72%, and after intervention was 20%. The result proved statistically significant (p < 0.001).
The mean referral age for children with airway haemangiomas was 2.4 months (range 0.7-8 months), and the mean starting age of propranolol treatment 5.1 months (range 0.75-22 months). Treated children were followed up for a mean of 8.4 months. Four children from our study population failed to respond to propranolol treatment (6.5%), and in seven the haemangioma relapsed after discontinuation of treatment (11.5%).
Discussion
Epidemiology-Pathophysiology
Infantile haemangiomas seem to affect 4-10% of Caucasian infants, with a female to male ratio of 2.5-4:1 8-10. They have also been associated with prematurity, low birth weight and multiple pregnancies 11-13. Sixty per cent of haemangiomas represent single lesions, and 80% are found in the head and neck area 8 14. Airway haemangiomas can involve any site from the nares to the tracheobronchial tree. However, they are most commonly seen in the subglottis, and account for approximately 1.5% of congenital laryngeal anomalies 15. The natural history of haemangiomas in the subglottis may be accompanied by an alarming mortality rate of around 50% if left untreated 16 17. Indeed, approximately two-thirds of these haemangiomas present with 70% or worse airway narrowing 18 19. Histologically, infantile haemangiomas are composed of a complex mixture of clonal epithelial cells associated with pericytes, fibroblasts, dendritic and mast cells 6 8-10 20 21. Although theories regarding their origin involve potential somatic endothelial mutations, intrinsic defects, extrinsic factors (i.e. tocolytics) and placental derivation 8 22 23, no current hypothesis can explain all their characteristics. It has been proposed recently that the tumour may be a developmental disorder of primitive mesodermal proliferation and differentiation governed by the renin-angiotensin system 24 25.
Haemangioma growth is controlled by pro-angiogenic factors during the proliferative phase, predominantly basic fibroblast growth factor (bFGF) and vascular endothelial growth factor (VE GF). Conversely, the involutional phase is associated with cell apoptosis resulting in tumour regression 6 and replacement with fibro-fatty tissue 26. Immunochemistry shows persistent reactivity to the erythrocytetype glucose transporter protein isoform 1 (GL UT-1) 8 27.
Treatment modalities-Mechanism of propranolol action
A variety of treatment modalities have been used in the management of airway haemangiomas. Although the treatment plan depends on many factors (i.e. size and extend of the lesion, social situations, clinical experience etc.), a widely accepted aim is to maintain airway patency, promote lesion regression, avoid tracheostomy and minimize therapeutic side effects until the lesions enter the natural phase of involution 4 15. Unfortunately, a watchful-waiting policy can only be applied in the minority of patients who do not demonstrate respiratory or feeding difficulties 19.
The gold standard for the treatment of symptomatic airway haemangiomas until recently has been the administration of steroids. Systemic steroids can be effective in controlling haemangioma growth in over 50% of patients 10 28-30, even though subglottic lesions tend to show poorer response 29. However, their side-effects are also well known; cushingoid face, insomnia, irritability, growth retardation, gastro-intestinal lesions, delayed wound healing, hirsutism, and even adrenal suppression, hypertension, or hypertrophic cardiomyopathy.
Interferon-α, on the other hand, has been widely used for the treatment of refractory haemangiomas, but is associated with significant neurotoxic effects, such as spastic diplegia and developmental delay 9, especially in young infants 18, while it may also be responsible for a variety of other sideeffects (flu-type malaise, neutropenia, elevation of liver enzymes, hypothyroidism). Vincristine therapy has also been employed in life-threatening conditions, but may cause peripheral neuropathy resulting in constipation, abdominal pain and paraesthesia, in addition to inappropriate secretion of antidiuretic hormone. In these latter cases, cyclophosphamide has also been reported to have promising results 31. However, the serious side-effects related to cyclophosphamide (i.e. avascular necrosis, cardiomyopathy, pulmonary fibrosis, gonadal damage, and later malignancies 32) have to be carefully considered prior to the initiation of treatment. In addition, it should not be forgotten that vincristine and cyclophosphamide are mainly anti-cancer drugs with potentially very serious side effects in the long-term, especially when given to very young children.
A number of surgical interventions have also been attempted, especially in cases that do not respond to medical therapy. Tracheostomy 33 can no longer be considered as first-line treatment, not only because of the relatively low risk of mortality (approximately 1%), but also for the associated morbidity (including the serious impact on the child's and family's quality of life), and the need for close postoperative tracheostomy care 19 34. However, in emergency cases tracheostomy may be the only immediate solution to save the child's life.
Intra-lesional steroids may reduce the related systematic morbidity, although their results have been variable 35, and they often involve prolonged intubation and stay in a paediatric ICU; the associated risks are also not considered acceptable by many authors 15.
Laser ablation is considered a reasonable option for localized haemangiomas not causing extreme airway narrowing. As with other modalities of treatment, the respective efficacy varies, whereas the associated risk includes high recurrence rates that may lead to multiple treatments 36 37. Extensive circumferential narrowing is not suitable for laser debulking, due to the risk of postoperative scarring, and the ensuing subglottic stenosis. Hence, open resection and laryngo-tracheal augmentation may be ultimately required. However, these are relatively major surgical procedures with potentially serious complications. Moreover, they demand inpatient management, intensive care units and vast related experience on behalf of the operating surgeon.
The incidental discovery that propranolol induced early regression of infantile haemangiomas during their proliferative phase by Leaute-Labreze et al, along with its low side-effect profile, attracted significant attention, and propranolol- based regimens have emerged as a new method of treatment in a growing volume of literature. However, the use of propranolol for proliferative infantile haemangiomas in the first year of life is still officially considered "off label".
The exact mechanism of propranolol action in infantile haemangiomas is equally unclear to their origin. Propranonol is a non-selective β-blocker inhibitor, with pure antagonistic effects. Proposed mechanisms of action include control of hypoxic stress, vasoconstriction, inhibition of angiogenesis and induction of endothelial cell apoptosis. Vasoconstriction can be visible as a change in colour, associated with palpable tissue softening, which can be observed as early as 1-3 days from treatment onset 8 15 38. The anti-angiogenic effect of propranolol can be exercised through its anti-VE GF activity 3 11, as well as through reduced activity of the renin-angiotensin system and reduced expression of matrix metalloproteinases (MMPs) 8. The resulting growth arrest is followed by regression of the lesions through an increase in the rate of apoptosis. The latter, however, is not complete in young infants, as partial re-growth of haemangiomas has been observed after the discontinuation of propranolol administration. In addition, variability in response can be suggestive of variability in tumour composition 39.
Meta-analysis
The results of the present meta-analysis suggest that propranolol is indeed effective in the treatment of airway haemangiomas (p < 0.001). The vast majority of children that received propranolol for airway hemangiomas showed improvement or resolution of symptoms, and normal thriving. Non-responsiveness to propranolol was only reported in four children (6.5%), whereas seven additional children relapsed following the discontinuation of treatment (11.5%). An interesting observation is that the administration of propranolol as monotherapy for airway haemangiomas in 14 infants resulted in 2 treatment failures (14.3%), but only one relapse after cessation of therapy (7.1%). Considering the respective percentages of the entire study population, this observation suggests that enhanced alert is required, when it is decided to treat infants exclusively with propranolol for airway haemangiomas, as the risk of non-responsiveness is increased. Indeed, in an area where compromise can be lethal, these patients should be monitored closely as they may require adjuvant therapy. In addition, despite the great efficiency of propranolol, recurrences of airway haemangiomas are possible after early or even late discontinuation of treatment. The cause of these relapses is equally unknown to the mechanism of propranolol action; the present metaanalysis identified a specific cause of haemangioma recurrence in only one of the 7 relapsing infants (administration of a sympathomimetic drug during cardiac surgery). It should be noted, however, that the mean follow up period of treated children identified in the present metaanalysis (8.4 months) is somewhat restrictive in assessing the above-mentioned important issues.
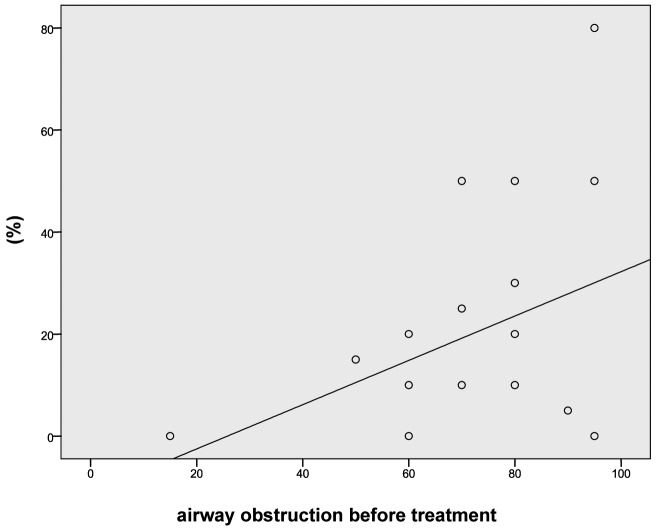
Unfortunately, sizing of the obstructed airway in order to quantify the exact value of improvement following the administration of propranolol, especially in cases of subglottic haemangiomas, was not reported in all studies. However, the present meta-analysis identified a subpopulation of treated children (n = 21), where such measurements had been performed during microlaryngoscopy and bronchoscopy under general anaesthesia (MLB), before and after intervention. As seen in Figure 1, the percentage of obstruction had dramatically decreased following propranolol administration in most cases. The related difference was statistically significant (p < 0.001). In addition, the correlation co-efficient between treatment with propranolol and the change in the percentage of obstruction post-treatment was found to be moderately positive (r = 0.39), and again statistically significant (p < 0.05) (Fig. 2).
Fig. 1.
Percentage of airway obstruction before and after propranolol treatment.
Fig. 2.
Scatter plot illustrating the relationship of propranolol to the result of treatment. The line of best fit shows a moderately positive correlation (r = 0.39).
The administration of propranolol as single-therapy is also a case-in-point, especially if we consider the relatively increased risk of non-responsiveness mentioned above. In particular, among the aforementioned 21 cases, five children had received propranolol as monotherapy. The mean obstruction before the administration of propranolol was 66%, and after intervention was 24%. Despite the small number of cases, the result was statistically significant (p = 0.01).
It should be noted that not all children show complete eradication of the haemangioma after being treated with propranolol. Indeed, only four children were reported as having no obstruction, and two additional children as having 5% of the subglottic airway obstructed. However, steroids and other medical methods of treatment do not eradicate the disease either. Therefore, symptom improvement/resolution and the ability of the infant to thrive, although subjective, can be considered the most important criteria of success. Nevertheless, if we put an arbitrary cut-off criterion of success – 10% of the airway to be obstructed after the intervention – then over 50% of responders can be expected to reach this significant target. The latter observation can be used for parent counselling before obtaining consent for the initiation of treatment.
In addition, propranolol was found more effective than steroids in treating airway haemangiomas (p < 0.001). Indeed, the respective odds ratio for successful outcome for propranolol vs. steroid treatment was positive and statistically significant (p < 0.05). In comparison to the previously considered second-line treatments for proliferating infantile haemangiomas, propranolol tended to outperform both laser treatment and vincristine, although the small number of patients treated with these modalities did not allow significant comparisons to be made. Nevertheless, when propranolol was used as salvage therapy in 10 children previously treated with laser, and in three children who had previously received vincristine, it achieved better symptom control in all.
Admittedly, the present meta-analysis did not identify any Level I studies regarding the administration of propranolol for the treatment of proliferating airway haemangiomas. The vast majority of studies were small case series, and the number of treated children was also relatively small. However, the reported results seem encouraging. In addition, the uniform nature of these results, along with the quality of the studies performed, allow us to adopt a grade C strength of recommendation regarding the effectiveness of propranolol in airway haemangiomas (Tab. III). However, the rather small number of patients with airway haemangiomas, who were treated solely with propranolol (Tab. IV), precludes us from drawing definite conclusions regarding the strength of recommendation for the effectiveness of propranolol as single-therapy for airway haemangiomas. We hope that the results of the present study will encourage further high quality studies with long term follow-up in order to clarify this important issue.
Table III.
Strength of recommendation by category of evidence for guideline development 40.
| Strength of recommendation | Category of evidence |
|---|---|
| A | Directly based on category I evidence |
| B | Directly based on category II evidence or extrapolated recommendation from category I evidence |
| C | Directly based on category III evidence or extrapolated recommendation from category I or II evidence |
| D | Directly based on category IV evidence or extrapolated recommendation from category I, II or III evidence |
Table IV.
Propranolol administration for airway haemangiomas (patient series).
| Authors | Study type | Evidence level | Number of subjects with airway haemangioma | Treatment failure/relapse | Adverse effects |
|---|---|---|---|---|---|
| Holmes et al., 2011 | Prospective study | II | 1 | 0 | None |
| Guye et al., 2011 41 | Case report | IV | 1 | 0 | None |
| Leboulanger et al., 2010 42 | Multicentre retrospective cohort | III | 14 | 2 | |
| Blanchet et al., 2010 43 | Case series | IV | 4 | 1 | None |
| Truong et al., 2010 44 | Case series | IV | 6 | 0 | None |
| Santos et al., 2010 | Case series | IV | 6 | 1 | Asthma (n = 1) |
| Rosbe et al., 2010 45 | Case series | IV | 3 | 1 | Soft palate ulcer (N = 1) |
| Canadas et al., 2010 46 | Case report | IV | 1 | 1 | None |
| * Manunza et al, 2010 47 | Case-series | IV | 9 | Pallor (n = 1) Asthma (n = 1) | |
| Jepshon et al, 2009 | Case report | 1 | 0 | None | |
| Maturo and Hartnick, 2010 48 | Case series | IV | 2 | 0 | None |
| Larrier, 2010 49 | Retrospective study | III | 3 | 2 | None |
| Anderson de Moreno et al, 2009 50 |
Retrospective study | III | 4 | 0 | None |
| Sans et al, 2009 | Prospective cohort | II | 4 | 0 | Not specified for patients With airway haemangiomas |
| Buckmiller et al, 2009 | Case report | IV | 1 | 0 | None |
| Theletsane et al, 2009 | Case report | IV | 1 | 0 | None |
| Mistry and Tzifa, 2009 | Case report | IV | 1 | 0 | None |
authors belong to the same investigating team
Treatment regimen-Follow up algorithm
Propranolol, a non-selective β-blocker, was considered one of the most important contributions to clinical medicine and pharmacology of the 20th century 8. It is almost completely absorbed from the gastro-intestinal tract, highly protein-bound (80-95%), and there is a significant first pass metabolism and hepatic tissue binding, with up to 90% of an oral dose being eliminated. Like many drugs adapted for use in infants, there are no pharmacokinetic data in this specific population, and no prospective, controlled studies have described optimal dosing or monitored the respective side-effects in this age group 51.
The potential risks associated with the use of propranolol include cardio-pulmonary complications, such as bradycardia, and hypotension, especially in syndromic children (i.e. PHACES, PELVIS etc.) and neonates in the first week of life 48, bronchospasm in patients with atopic predisposition or underlying spastic respiratory illness 52, hypoglycaemia, especially in low birth weight newborns 9 and children with low caloric intake 53 or diabetes, electrolytic abnormalities (i.e. hyperkalaemia 52), gastro-oesophageal reflux, somnolence/fatigue, agitated sleep, rash and resistance to the drug's re-introduction after early discontinuation of therapy 42. These effects, however, appear minor, compared to the serious side effects of more conventional agents, such as the ones mentioned earlier. In addition, after over 40 years of extensive clinical experience with propranolol, no serious cardiovascular event has been recorded in a child on chronic β-blocker therapy 54. Nevertheless, the adoption of this treatment modality by non-familiar specialties, such ENT, certainly warrants careful monitoring, close liaise with the affiliated paediatric Cardiology Department, and a treatment algorithm to minimize the potential adverse effects 15 28 39 51 55. The present meta-analysis identified only four adverse effects in children receiving propranolol for the treatment of airway haemangiomas; two asthmatic attacks, which had necessitated cessation of treatment, one episode of pallor without loss of consciousness, which was attributed to a possible vagal reaction, but did not require discontinuation of treatment or dose adjustment and a soft palate ulcer, which responded to steroid treatment.
The pharmacologically optimal dosing interval for propranolol is 6 hours, although better compliance is achieved when the medication is given 2 or 3 times daily. Before starting the treatment, baseline vital signs are obtained 51, along with blood glucose, especially if the child has not established a routine feeding pattern 48. Electrocardiogram and echocardiogram are also necessary, and if either is abnormal a cardiology review is required 51. Infants with large haemangiomas and especially segmental distribution are at increased risk of high output cardiac failure, and propranolol can conceal the related tachypnoea and hyperidrosis 51. Syndromic children are also evaluated with trans-cranial Doppler and/or MRI /MRA of the head and neck 48. Otherwise, a CT scan of the neck with intravenous contrast gives optimal resolution of the haemangioma 56, without the need to intubate the infant.
Hospitalized infants receive a starting dose of 0.17 mg/kg three times daily. Vital signs and blood glucose are checked 1 hour after each dose, as this corresponds to the peak absorption time. If the first 2 doses are tolerated, the dose is doubled to 0.33 mg/kg, and after 2 more doses to 0.67 mg/ kg. For infants less than 3 months of age, slower dose escalation to avoid hypoglycaemia may be required. Frequent feeds are strongly encouraged. Parents need to be informed about the clinical signs of hypotension, bradycardia and hypoglycaemia, as for the need of dose adjustment according to the child's weight 48. The optimal duration of treatment is not known, but it is currently believed that the child should remain on propranolol until the haemangioma enters the phase of involution, which usually happens after the first year of life. Earlier discontinuation of treatment may result in unresponsiveness to propranolol re-introduction, should the latter be required. Moreover, the lack of long-term follow up in the published studies does not allow any definite conclusions with regard to relapses, even after the beginning of the involutional phase.
If a child is unresponsive to propranolol, biopsy (taking all the necessary precautions to control a potentially persistent and life threatening bleeding) and immunohistochemical staining for GLUT-1 may be considered to confirm diagnosis 28 44. Finally, propranolol is gradually tapered over 2-3 weeks, and not stopped immediately, due to issues involving cardiac hypersensitivity.
Conclusions
Airway haemangiomas may require early and aggressive treatment in life-threatening cases. However, the treatment modalities employed so far have been associated with their own comorbidities and potential long-term adverse effects for the developing child. Propranolol has the potential to alter the management of proliferating haemangiomas in infancy radically. Although high-quality evidence is still not available, the results of the present study suggest that propranolol can be recommended for the treatment of airway haemangiomas, as it was found to be effective and outperformed the previously-considered gold standard methods of treatment (p < 0.001) with fewer side-effects. Immediate treatment with propranolol should commence once a diagnosis of symptomatic airway haemangioma is confirmed and a cardiovascular assessment has taken place. Active parental monitoring is also essential to ensure treatment safety.
Acknowledgements
The authors would like to thank Dr Marie Lyons MD, FRC S (ORL -HNS) for her key remarks in the revised version of the manuscript.
References
- 1.Brown RL, Azizkhan RG. Pediatric head and neck lesions. Pediatr Clin N Am. 1998;45:889–905. doi: 10.1016/s0031-3955(05)70052-3. [DOI] [PubMed] [Google Scholar]
- 2.Enjolras O. Classification and management of the various superficial vascular anomalies: hemangiomas and vascular malformations. J Dermatol. 1997;24:701–710. doi: 10.1111/j.1346-8138.1997.tb02522.x. [DOI] [PubMed] [Google Scholar]
- 3.Leaute-Labreze C, Roque ED, Hubiche T. Propranolol for severe hemangiomas of infancy. N Engl J Med. 2008;358:2649–2651. doi: 10.1056/NEJMc0708819. [DOI] [PubMed] [Google Scholar]
- 4.Buckmiller L, Dyamenahalli U, Richter GT. Propranolol for airway hemangiomas: case report of novel treatment. Laryngoscope. 2009;119:2051–2054. doi: 10.1002/lary.20633. [DOI] [PubMed] [Google Scholar]
- 5.Jephson CG, Manunza F, Syed S, et al. Successful treatment of isolated subglottic haemangioma with propranolol alone. Int J Pediatr Otorhinolaryngol. 2009;73:1821–1823. doi: 10.1016/j.ijporl.2009.08.020. [DOI] [PubMed] [Google Scholar]
- 6.Denoyelle F, Leboulanger N, Enjolras O, et al. Role of propranolol in the therapeutic strategy of infantile laryngotracheal hemangioma. Int J Pediatr Otorhinolaryngol. 2009;73:1168–1172. doi: 10.1016/j.ijporl.2009.04.025. [DOI] [PubMed] [Google Scholar]
- 7.Theletsane T, Redfern A, Raynham O, et al. Life-threatening infantile haemangioma: a dramatic response to propranolol. J Eur Acad Dermatol Venereol. 2009;23:1465–1466. doi: 10.1111/j.1468-3083.2009.03261.x. [DOI] [PubMed] [Google Scholar]
- 8.Zimmermann AP, Wiegand S, Werner JA, et al. Propranolol therapy for infantile haemangiomas: Review of the literature. Int J Pediatr Otorhinolaryngol. 2010;74:338–342. doi: 10.1016/j.ijporl.2010.01.001. [DOI] [PubMed] [Google Scholar]
- 9.Sans V, Roque ED, Berge J , et al. Propranolol for severe infantile hemangiomas: follow-up report. Pediatrics. 2009;124:e423–e431. doi: 10.1542/peds.2008-3458. [DOI] [PubMed] [Google Scholar]
- 10.Frieden IJ, Haggstrom AN, Drolet BA, et al. Infantile hemangiomas: current knowledge, future directions. Proceedings of a research workshop on infantile hemangiomas. Pediatr Dermatol. 2005;22:383–406. doi: 10.1111/j.1525-1470.2005.00102.x. [DOI] [PubMed] [Google Scholar]
- 11.Truong MT, Chang KW, Berk DR, et al. Propranolol for the treatment of a life-threatening subglottic and mediastinal infantile hemangioma. J Pediatr. 2010;156:335–338. doi: 10.1016/j.jpeds.2009.10.010. [DOI] [PubMed] [Google Scholar]
- 12.Drolet BA, Swanson EA, Frieden IJ. Infantile hemangiomas: an emerging health issue linked to an increased rate of low birth weight infants. J Pediatr. 2008;153:712–715. doi: 10.1016/j.jpeds.2008.05.043. [DOI] [PubMed] [Google Scholar]
- 13.Haggstrom AN, Drolet BA, Baselga E, et al. Prospective study of infantile hemangiomas: demographic, prenatal, and perinatal characteristics. J Pediatr. 2007;150:291–294. doi: 10.1016/j.jpeds.2006.12.003. [DOI] [PubMed] [Google Scholar]
- 14.Leaute-Labreze C, Taieb A. Efficacy of beta-blockers in infantile haemangiomas: the physiopathological significance and therapeutic consequences. Ann Dermatol Venerol. 2008;135:860–862. doi: 10.1016/j.annder.2008.10.006. [DOI] [PubMed] [Google Scholar]
- 15.Mistry N, Tzifa K. Use of propranolol to treat multicentric airway haemangioma. J Laryngol Otol. 2010;124:1329–1332. doi: 10.1017/S002221511000068X. [DOI] [PubMed] [Google Scholar]
- 16.Bailey CM, Froehlich P, Hoeve HL. Management of subglottic haemangioma. J Laryngol Otol. 1998;112:765–768. doi: 10.1017/s0022215100141623. [DOI] [PubMed] [Google Scholar]
- 17.Ferguson CF, Flake CG. Subglottic hemangioma as a cause of respiratory obstruction in infants. Ann Otol Rhinol Laryngol. 1961;70:1095–1112. doi: 10.1177/000348946107000414. [DOI] [PubMed] [Google Scholar]
- 18.Pransky SM, Canto C. Management of subglottic hemangioma. Curr Opin Otolaryngol Head Neck Surg. 2004;12:509–512. doi: 10.1097/01.moo.0000143980.41120.38. [DOI] [PubMed] [Google Scholar]
- 19.Rahbar R, Nicollas R, Roger G, et al. The biology and management of subglottic hemangioma:past, present, future. Laryngoscope. 2004;114:1880–1891. doi: 10.1097/01.mlg.0000147915.58862.27. [DOI] [PubMed] [Google Scholar]
- 20.Boye E, Yu Y, Paranya G, et al. Clonality and altered behaviour of endothelial cells from hemangiomas. J Clin Invest. 2001;107:745–756. doi: 10.1172/JCI11432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Khan ZA, Boscolo E, Picard A, et al. Multipotential stem cells recapitulate human infantile hemangioma in immunodeficient mice. J Clin Invest. 2008;118:2592–2599. doi: 10.1172/JCI33493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Eivazi B, Ardelean M, Baumler W, et al. Update on haemangiomas and vascular malformations of the head and neck. Eur Arch Otorhinolaryngol. 2009;266:187–197. doi: 10.1007/s00405-008-0875-6. [DOI] [PubMed] [Google Scholar]
- 23.Werner JA, Dunne AA, Lippert BM, et al. Optimal treatment of vascular birthmarks. Am J Clin Dermatol. 2003;4:745–756. doi: 10.2165/00128071-200304110-00003. [DOI] [PubMed] [Google Scholar]
- 24.Itinteang T, Brasch HD, Tan ST, et al. Expression of components of the renin-angiotensin system in proliferating infantile haemangioma may account for the propranolol-induced accelerated involution. J Plast Reconstr Aesthet Surg. 2011;64:759–765. doi: 10.1016/j.bjps.2010.08.039. [DOI] [PubMed] [Google Scholar]
- 25.Itinteang T, Withers AH, Leadbitter P, et al. Pharmacologic therapies for infantile hemangioma: is there a rational basis? Plast Reconstr Surg. 2011;128:499–507. doi: 10.1097/PRS.0b013e31821b63a0. [DOI] [PubMed] [Google Scholar]
- 26.Phung TL, Hochman M, Mihm MC, et al. Current knowledge of the pathogenesis of infantile hemangiomas. Arch Facial Plast Surg. 2005;7:319–321. doi: 10.1001/archfaci.7.5.319. [DOI] [PubMed] [Google Scholar]
- 27.North PE, Waner M, Mizeracki A, et al. GLUT1: a newly discovered immunohistochemical marker for juvenile hemangiomas. Hum Pathol. 2000;31:11–22. doi: 10.1016/s0046-8177(00)80192-6. [DOI] [PubMed] [Google Scholar]
- 28.Santos S, Torrelo A, Tamariz-Martel A, et al. Clinical observations on propranolol use for paediatric airway hemangiomas. Acta Otorrinolaringol Esp. 2010;61:365–370. doi: 10.1016/j.otorri.2010.01.014. [DOI] [PubMed] [Google Scholar]
- 29.Bennett ML, Fleischer AB, Chamlin SL, et al. Oral corticosteroid use is effective for cutaneous hemangiomas: an evidencebased evaluation. Arch Dermatol. 2001;137:1208–1213. doi: 10.1001/archderm.137.9.1208. [DOI] [PubMed] [Google Scholar]
- 30.Enjolras O, Riche MC, Merland JJ, et al. Management of alarming hemangiomas in infancy: a review of 25 cases. Pediatrics. 1990;85:e491–e498. [PubMed] [Google Scholar]
- 31.Vlahovic A, Simic R, Djokic D, et al. Diffuse neonatal hemangiomatosis treatment with cyclophosphamide: a case report. J Pediatr Hematol Oncol. 2009;31:858–860. doi: 10.1097/MPH.0b013e3181b536ad. [DOI] [PubMed] [Google Scholar]
- 32.Gottschling S, Schneider G, Meyer S, et al. Two infants with life-threatening diffuse neonatal hemangiomatosis treated with cyclophosphamide. Pediatr Blood Cancer. 2006;46:239–242. doi: 10.1002/pbc.20522. [DOI] [PubMed] [Google Scholar]
- 33.Suehs OW, Herbur PA. Hemangiomas of the larynx in infants. Arch Otolaryngol. 1940;32:783–789. [Google Scholar]
- 34.O-Lee TJ, Messner A. Subglottic hemangioma. Otolaryngol Clin North Am. 2008;41:903–911. doi: 10.1016/j.otc.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 35.Bitar MA, Mourkarbel RV, Zalzal GH, et al. Management of congenital subglottic hemangioma: trends and success over the past 17 years. Otolaryngol Head Neck Surg. 2005;132:226–231. doi: 10.1016/j.otohns.2004.09.136. [DOI] [PubMed] [Google Scholar]
- 36.Brodsky L, Yoshpe N, Ruben RJ. Clinical-pathological correlates of congenital subglottic hemangiomas. Ann Otol Rhinol Laryngol. 1983;105:4–18. doi: 10.1177/00034894830920s401. [DOI] [PubMed] [Google Scholar]
- 37.Chatrath P, Black M, Jani P, et al. A review of the current management of infantile subglottic haemangioma, including a comparison of CO2 laser therapy versus tracheostomy. Int J Pediatr Otorhinolaryngol. 2002;64:143–157. doi: 10.1016/s0165-5876(02)00072-1. [DOI] [PubMed] [Google Scholar]
- 38.Holmes WJ, Mishra A, Gorst C, et al. Propranolol as firstline treatment for rapidly proliferating Infantile Haemangiomas. J Plast Reconstr Aesthet Surg. 2011;64:445–451. doi: 10.1016/j.bjps.2010.07.009. [DOI] [PubMed] [Google Scholar]
- 39.Buckmiller LM, Munson PD, Dyamenahalli U, et al. Propranolol for infantile hemangiomas: early experience at a tertiary vascular anomalies center. Laryngoscope. 2010;120:676–681. doi: 10.1002/lary.20807. [DOI] [PubMed] [Google Scholar]
- 40.Shekelle PG, Woolf SH, Eccles M, et al. Clinical guidelines: developing guidelines. BMJ. 1999;318:593–596. doi: 10.1136/bmj.318.7183.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Guye E, Chollet-Rivier M, Schröder D, et al. Propranolol treatment for subglottic haemangioma. Arch Dis Child Fetal Neonatal Ed. 2011;96:F263–F264. doi: 10.1136/adc.2010.201905. [DOI] [PubMed] [Google Scholar]
- 42.Leboulanger N, Fayoux P, Teissier N, et al. Propranolol in the therapeutic strategy of infantile laryngotracheal hemangioma: A preliminary retrospective study of French experience. Int J Pediatr Otorhinolaryngol. 2010;74:1254–1257. doi: 10.1016/j.ijporl.2010.07.025. [DOI] [PubMed] [Google Scholar]
- 43.Blanchet C, Nicollas R, Bigorre M, et al. Management of infantile subglottic hemangioma: acebutolol or propranolol? Int J Pediatr Otorhinolaryngol. 2010;74:959–961. doi: 10.1016/j.ijporl.2010.05.013. [DOI] [PubMed] [Google Scholar]
- 44.Truong MT, Perkins JA, Messner AH, et al. Propranolol for the treatment of airway hemangiomas: a case series and treatment algorithm. Int J Pediatr Otorhinolaryngol. 2010;74:1043–1048. doi: 10.1016/j.ijporl.2010.06.001. [DOI] [PubMed] [Google Scholar]
- 45.Rosbe KW, Suh KY, Meyer AK, et al. Propranolol in the management of airway infantile hemangiomas. Arch Otolaryngol Head Neck Surg. 2010;136:658–665. doi: 10.1001/archoto.2010.92. [DOI] [PubMed] [Google Scholar]
- 46.Canadas KT, Baum ED, Lee S, et al. Case report: Treatment failure using propanolol for treatment of focal subglottic hemangioma. Int J Pediatr Otorhinolaryngol. 2010;74:956–958. doi: 10.1016/j.ijporl.2010.05.012. [DOI] [PubMed] [Google Scholar]
- 47.Manunza F, Syed S, Laguda B, et al. Propranolol for complicated infantile haemangiomas: a case series of 30 infants. Br J Dermatol. 2010;162:466–468. doi: 10.1111/j.1365-2133.2009.09597.x. [DOI] [PubMed] [Google Scholar]
- 48.Maturo S, Hartnick C. Initial experience using propranolol as the sole treatment for infantile airway hemangiomas. Int J Pediatr Otorhinolaryngol. 2010;74:323–325. doi: 10.1016/j.ijporl.2009.12.008. [DOI] [PubMed] [Google Scholar]
- 49.Larrier D. Propranolol for the treatment of subglottic hemangiomas. Otolaryngol Head Neck Surg. 2010;143(2S2):P264–P264. [Google Scholar]
- 50.Anderson de Moreno L, Matt B, Horn D, et al. Propranolol in the treatment of subglottic hemangiomas. Otolaryngol Head Neck Surg. 2010;143(2S2):P264–P265. [Google Scholar]
- 51.Lawley LP, Siegfried E, Todd JL. Propranolol treatment for hemangioma of infancy: risks and recommendations. Pediatr Dermatol. 2009;26:610–614. doi: 10.1111/j.1525-1470.2009.00975.x. [DOI] [PubMed] [Google Scholar]
- 52.Pavlakovic H, Kietz S, Lauerer P, et al. Hyperkalemia complicating propranolol treatment of an infantile hemangioma. Pediatrics. 2010;126:e1589–e1593. doi: 10.1542/peds.2010-0077. [DOI] [PubMed] [Google Scholar]
- 53.Bonifazi E, Acquafredda A, Milano A, et al. Severe hypoglycemia during successful treatment of diffuse hemangiomatosis with propranolol. Pediatr Dermatol. 2010;27:195–196. doi: 10.1111/j.1525-1470.2009.01081.x. [DOI] [PubMed] [Google Scholar]
- 54.Love JN, Sikka N. Are 1-2 tablets dangerous? Beta-blocker exposure in toddlers. J Emerg Med. 2004;26:309–314. doi: 10.1016/j.jemermed.2003.11.015. [DOI] [PubMed] [Google Scholar]
- 55.Holland KE, Frieden IJ, Frommelt PC, et al. Hypoglycemia in children taking propranolol for the treatment of infantile hemangioma. Arch Dermatol. 2010;146:775–778. doi: 10.1001/archdermatol.2010.158. [DOI] [PubMed] [Google Scholar]
- 56.Perkins JA, Duke W, Chen E, et al. Emerging concepts in airway infantile hemangioma assessment and management. Otolaryngol Head Neck Surg. 2009;141:207–212. doi: 10.1016/j.otohns.2009.04.013. [DOI] [PubMed] [Google Scholar]