Abstract
Obesity at diagnosis of breast cancer is associated with higher all-cause mortality and treatment-associated toxicities. We evaluated the association between parity and obesity in the Ella study, a population of Mexican and Mexican–American breast cancer patients with high parity. Obesity outcomes included body mass index (BMI) ≥30 kg/m2, waist circumference (WC) ≥35 in (88 cm), and waist-to-hip-ratio (WHR) ≥0.85. Prevalence of obesity ([BMI] ≥30 kg/m2) was 38.9 %. For WC, the multivariate odds ratio (OR) (95 % confidence interval [CI]) for having WC ≥ 35 inches in women with ≥4 pregnancies relative to those with 1–2 pregnancies was 1.59 (1.01–2.47). Higher parity (≥4 pregnancies) was non-significantly associated with high BMI (OR = 1.10; 95 % CI 0.73–1.67). No positive association was observed for WHR. Our results suggest WC is independently associated with high parity in Hispanic women and may be an optimal target for post-partum weight loss interventions.
Keywords: Parity, Obesity, Hispanic women, Body mass index, Waist circumference
Introduction
The rapid increase in obesity rates is a growing concern in the U.S. and in several low- and middle-resource countries worldwide [1, 2]. In the U.S., racial/ethnic minorities suffer disproportionately from the obesity epidemic, particularly women [3]. According to the 2007–2008 National Health and Nutrition Examination Survey (NHANES), 45.1 % of Mexican–American women were obese, compared with 33.0 % of non-Hispanic white women [4]. High rates of obesity contribute to chronic conditions, such as hypertension, cardiovascular disease, type 2 diabetes, and cancers of the breast (postmenopausal), colon, endometrium, kidney and esophagus [1]. Among pregravid women, obesity increases the risk for gestational diabetes and congenital malformations, namely fetal anencephaly [5]. For women with breast cancer, obesity is associated with higher all-cause mortality [6] and higher rates of intentionally reduced doses of chemotherapy, particularly in the morbidly obese, compared to normal weight individuals [7, 8].
Parity may contribute to obesity due to postpartum weight retention. However, this relationship is complex given that fertility rates have decreased in recent years, yet obesity prevalence continues to rise. Several published studies have reported positive associations between parity and weight gain or being obese [9–18]. Most of these reports have included only weight-related measures such as BMI or weight change as obesity outcomes; fewer have included measures of central adiposity [10–12, 17]. Understanding the relationship between parity and risk of obesity in later years in specific racial/ethnic groups and in individuals living in less-developed countries is a crucial step in identifying women most likely to benefit from weight loss interventions.
The Hispanic population in the U.S. has grown substantially in the last two decades. From 2000 to 2010, the number of Hispanics increased from 35.3 to 50.5 million; the largest subgroup is comprised of individuals of Mexican descent (63 %) [19]. Among Hispanic women, rates of both obesity and fertility are high [20]. However, little is known about the contribution of parity to body size (e.g., obesity, central adiposity) in this ethnic group. Furthermore, since it has been proposed that the parity-obesity association is dependent on the level of development of the country where the women reside [21], it is important to assess this relationship in both developed and less-developed countries. We used data from an existing binational study of breast cancer to evaluate the relationship between parity and obesity, including body mass index (BMI), waist circumference (WC), and waist-to-hip ratio (WHR) measures, in a highly parous population of Mexican and Mexican–American women with a recent diagnosis of breast cancer.
Conceptual Framework
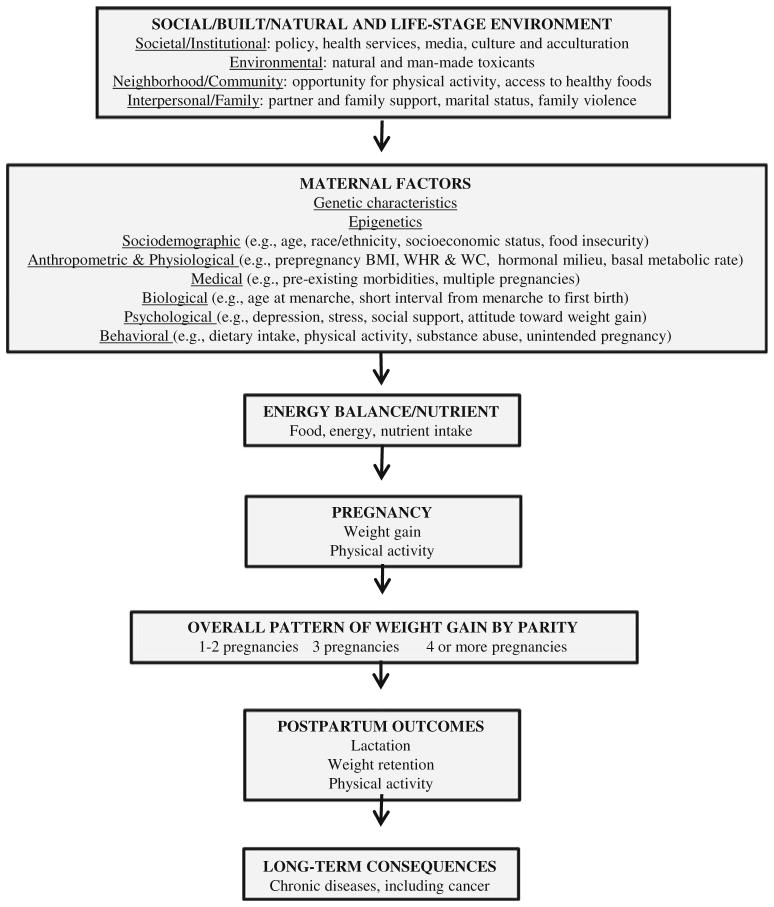
Figure 1 shows the conceptual framework for key determinants related to parity and obesity. The framework expands on previous work presented in the 2009 Institute of Medicine (IOM) and National Research Council Report (NRC) report on weight gain during pregnancy (Fig. 1) [22]. Additional determinants include biological maternal factors proposed by Gunderson [23]. Our contribution to the framework includes expanded anthropometric measures (WC and WHR) and detailed categories of multiparity.
Fig. 1.
Conceptual framework for key determinants related to parity and obesity. Arrows indicate possible causal influence
Methods
Participants
The Ella Binational Breast Cancer Study is a case-only study of women of self-reported Mexican descent who were 18 and older and diagnosed with invasive breast cancer within 12 months of enrollment. Participants were recruited from two study sites in the U.S. (University of Arizona and MD Anderson Cancer Center) and three study sites in Mexico (Universidad de Sonora, Instituto Tecnológico de Sonora and Universidad de Guadalajara). Complete details regarding the study population and recruitment strategy have been previously published [24]. Eligibility criteria for this sub-study required that participants have complete risk factor questionnaire or medical record data available to compute BMI and at least one pregnancy. This resulted in a study population of 974 participants (482 U.S. and 492 Mexico participants). The Institutional Review Board from each institution approved the study and all participants provided written informed consent.
Data Collection
A face-to-face interview was conducted where participants were administered a risk factor questionnaire and provided consent to abstract their medical records. In the U.S., 48.1 % of participants elected to have their interviews conducted in Spanish, with the remaining in English. The risk factor questionnaire included information on sociode-mographic data, reproductive factors, anthropometric measures, and other breast cancer risk factor data. Height and weight prior to breast cancer diagnosis were primarily obtained from the risk factor questionnaire. If self-reported weight was not available from the questionnaire (n = 48, 4.9 %), this was obtained from the medical record at a time point nearest to diagnosis; data on weight between the two sources were highly correlated (rho = 0.85). When self-reported height was not available from the questionnaire, this was considered missing due to low correlation between self-reported and medical record height (rho = 0.40); this resulted in the exclusion of 56 women. Waist and hip circumference were obtained by trained interviewers at the time the risk factor questionnaire was administered. Interviewers instructed participants to remove excess layers of clothing and stand with weight distributed evenly between both feet, with their abdomen relaxed, and arms placed at their side. Interviewers faced the participant and placed the tape measure at the level of the natural waist. The interviewer used a tape measure to measure the smallest horizontal circumference in the area between the participant’s ribs and the iliac crest after the participant completed a normal expiration of breath. Hip circumference was measured at the maximum extension of the buttocks.
BMI was calculated using weight in kilograms divided by the square of height in meters.
Using World Health Organization [25] standards derived from European populations, participants were categorized according to BMI as non-obese (BMI < 30 kg/m2) or obese (BMI ≥ 30 kg/m2). Guidelines from the National Heart, Lung and Blood Institute (NHLBI) [26] were used to define high-risk WC and WHR based on risk associated with developing obesity-related metabolic disorders, where a WC measuring ≥35 inches (88 cm) or a WHR ≥ 0.85 were considered to be high.
Parity or number of full-term births was self-reported via the interview-administered questionnaire. A full-term birth was defined as any pregnancy lasting longer than 5 months, regardless of outcome. Nulliparous women were not considered due to the low prevalence of nulliparity in the study population (9.8 %).
Menopausal status was derived primarily from the medical record (91 %) but was substituted with self-reported menopausal status from the risk factor questionnaire when necessary (agreement between medical record data and self-reported menopausal status was 90.1 %). Variables such as nativity (country of birth), interview language, and acculturation were evaluated only among U.S. participants. Acculturation was derived using the risk factor questionnaire, which included two orthogonal four-item measures of cultural orientation; one scale assessed the degree of English language use and exposure, and the other assessed the degree of Spanish language use and exposure [27]. This bi-dimensional derivation of cultural orientation has been previously validated by Marin and Gamba [28].
Statistical Analysis
Logistic regression models were constructed to test the relationship between parity and obesity using the three separate obesity outcome measures: BMI ≥ 30 kg/m2, WC ≥ 35 in, and WHR ≥ 0.85. Parity was categorized into three groups based on its distribution in this study population: 1–2 (referent), 3, and ≥ 4 pregnancies. After adjustment for age at interview (continuous) and recruitment site (University of Arizona, MD Anderson Cancer Center, Universidad de Sonora, Instituto Tecnológico de Sonora, and Universidad de Guadalajara), sociodemo-graphic factors were assessed individually for confounding. If inclusion of the variable conferred ≥10 % change in the association between parity and obesity, the variable was included in the final multivariate model. As previously reported in the literature (23–29), potential confounders included education (<high school, high school, post-high school), employment history (none, <10 years, 10–19 years, ≥20 years), smoking status (never, former, current), alcohol use (never, ever), physical activity (total METs/day, continuous), age at menarche (continuous), age at first full-term pregnancy (continuous), breastfeeding (never, <12 months, 12 to <24 months, ≥24 months), hormone contraception use (never, ever), menopausal status (pre, post), and hormone replacement therapy use (never, ever). Education and age at first full-term pregnancy were identified as confounders for models of all three obesity outcomes (BMI, WC, and WHR). Breast-feeding was also selected for models of WC and WHR but not BMI. Four additional factors (smoking, physical activity, hormone contraception use, and hormone replacement therapy use) were selected and included in the multivariate model for WHR. Although additional variables qualified as confounders for the WHR model (alcohol, employment, and menopausal status), these factors were not included as covariates in the final model due to substantial missing data for these variables; however, including these covariates did not substantially change the results (data not shown). In stratified analyses by country, we considered language use, nativity and acculturation as confounding variables in the U.S. models. Given the high correlation among these three variables, we only included acculturation in the multivariate models; however, its inclusion in the models did not materially change the point estimates. Tests for trend were calculated by treating parity as a continuous variable and assessing dose–response relationships between parity and each of the three obesity outcome measures. All statistical tests were conducted using Stata 12.1 (StataCorp, College Station, Texas).
Results
Characteristics of the Ella study population by level of parity are presented in Table 1. Compared to women with 1–2 pregnancies, those with ≥ 4 were older, had a lower level of education, a longer length of employment, an earlier age at first birth, and a longer duration of breast-feeding. For women in the U.S., those with higher parity were less likely to be U.S.-born, and more likely to be Spanish-dominant than women with fewer pregnancies.
Table 1.
Ella study population characteristics by number of pregnancies
| Characteristic | Total (n = 974) | Number of pregnancies
|
||
|---|---|---|---|---|
| 1–2 (n = 328) | 3 (n = 281) | ≥4 (n = 365) | ||
| Age at interview (y), mean ± SD | 52.6 ± 11.9 | 48.6 ± 10.8 | 50.9 ± 10.9 | 57.5 ± 11.9 |
| Education [n (%)] | ||||
| <High school | 487 (50.4) | 100 (30.8) | 134 (47.7) | 253 (70.3) |
| High school | 265 (27.4) | 108 (33.2) | 85 (30.3) | 72 (20.0) |
| ≥High school | 214 (22.2) | 117 (36.0) | 62 (22.1) | 35 (9.72) |
| Unknown | 8 | 3 | 0 | 5 |
| Employment [n (%)] | ||||
| None | 176 (18.4) | 30 (9.32) | 48 (17.5) | 98 (27.3) |
| <10 years | 315 (33.0) | 107 (33.2) | 98 (35.6) | 110 (30.6) |
| 10–19 years | 272 (28.5) | 107 (33.2) | 72 (26.2) | 93 (25.9) |
| ≥20 years | 193 (20.2) | 78 (24.2) | 57 (20.7) | 58 (16.2) |
| Unknown | 18 | 6 | 6 | 6 |
| Age at menarche (years), mean ± SD | 12.8 ± 1.6 | 12.7 ± 1.6 | 12.8 ± 1.7 | 12.9 ± 1.5 |
| Age at first pregnancy (years), mean ± SD | 22.8 ± 5.5 | 26.2 ± 6.0 | 22.3 ± 4.5 | 20.2 ± 3.8 |
| Breastfeeding [n (%)] | ||||
| Never | 228 (23.4) | 118 (36.0) | 67 (23.8) | 43 (11.8) |
| 0 to <12 months | 317 (32.6) | 137 (41.8) | 95 (33.8) | 85 (23.3) |
| 12 to <24 months | 164 (16.8) | 47 (14.3) | 60 (21.4) | 57 (15.6) |
| ≥24 months | 265 (27.2) | 26 (7.93) | 59 (21.0) | 180 (49.3) |
| Hormone contraceptive use [n (%)] | 595 (61.1) | 201 (61.3) | 188 (66.9) | 206 (56.4) |
| Hormone replacement therapy [n (%)] | ||||
| Never | 841 (86.8) | 280 (85.6) | 234 (84.5) | 327 (89.6) |
| Ever | 128 (13.2) | 47 (14.4) | 43 (15.5) | 38 (10.4) |
| Unknown | 5 | 1 | 4 | 0 |
| Smoking status [n (%)] | ||||
| Never | 681 (69.9) | 238 (72.6) | 199 (70.8) | 244 (66.9) |
| Former | 246 (25.3) | 71 (21.7) | 73 (26.0) | 102 (28.0) |
| Current | 47 (4.83) | 19 (5.79) | 9 (3.20) | 19 (5.21) |
| Alcohol use [n (%)] | ||||
| No | 573 (62.4) | 158 (51.8) | 162 (31.1) | 253 (72.7) |
| Yes | 345 (37.6) | 147 (48.2) | 103 (38.9) | 95 (27.3) |
| Unknown | 56 | 23 | 16 | 17 |
| Physical activity (METs/day), mean ± SD | 107.5 ± 124 | 104.3 ± 125 | 107.8 ± 120 | 110.2 ± 125 |
| Menopausal status, n (%) | ||||
| Premenopausal | 483 (50.3) | 204 (63.4) | 154 (55.0) | 125 (34.9) |
| Postmenopausal | 477 (49.7) | 118 (36.7) | 126 (45.0) | 233 (65.1) |
| Unknown | 14 | 6 | 1 | 7 |
| Nativity [n (%)]a | ||||
| U.S.-born | 218 (45.2) | 96 (50.5) | 71 (48.3) | 51 (35.2) |
| Foreign-born, living in U.S. <10 years | 208 (43.2) | 71 (37.4) | 61 (41.5) | 76 (52.4) |
| Foreign-born, living in U.S. ≥10 years | 56 (11.6) | 23 (12.1) | 15 (10.2) | 18 (12.4) |
| Interview language [n (%)]a | ||||
| English | 250 (51.9) | 112 (59.0) | 83 (56.5) | 55 (37.9) |
| Spanish | 232 (48.1) | 78 (41.1) | 64 (43.5) | 90 (62.1) |
| Acculturation [n (%)]a | ||||
| English-dominant | 86 (17.8) | 45 (23.7) | 30 (20.4) | 11 (7.59) |
| Bilingual | 234 (48.6) | 91 (47.9) | 75 (51.0) | 68 (46.9) |
| Spanish-dominant | 162 (33.6) | 54 (28.4) | 42 (28.6) | 66 (45.5) |
| Recruitment site [n (%)] | ||||
| Arizona | 181 (18.6) | 63 (19.2) | 52 (18.5) | 66 (18.1) |
| Houston | 301 (30.9) | 127 (38.7) | 95 (33.8) | 79 (21.6) |
| Universidad de Sonora | 137 (14.1) | 42 (12.8) | 34 (12.1) | 61 (16.7) |
| Instituto Tecnológico de Sonora | 183 (18.8) | 44 (13.4) | 56 (19.9) | 83 (22.7) |
| Universidad de Guadalajara | 172 (17.7) | 52 (15.9) | 44 (15.7) | 76 (20.8) |
U.S. participants only
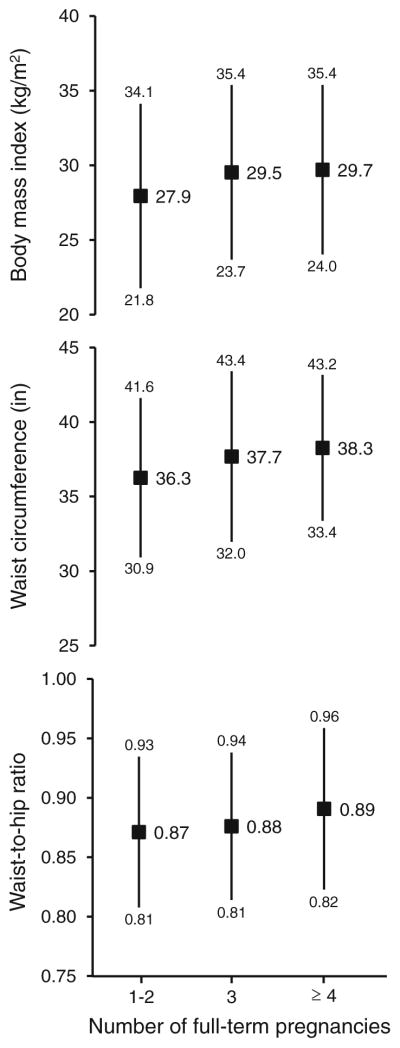
Obesity in the total population was high: 38.9 % (379/974) had a BMI ≥ 30 kg/m2; 70.5 % (568/806) had a WC ≥ 35 inches (88 cm); and 69.6 % (558/802) had a WHR ≥ 0.85. Mean values (±SD) for the total population were: 29.1 ± 5.9 for BMI, 37.4 ± 5.3 for WC, and 0.88 ± 0.07 for WHR. BMI significantly increased with parity category, where women reporting ≥4 or more pregnancies had a mean BMI of 29.7 ± 5.7 kg/m2 compared to 27.9 ± 6.2 kg/m2 among those with 1–2 pregnancies; the greatest increase was between the first two categories (Fig. 2). WC also significantly increased with increasing parity level (36.3, 37.7, and 38.3 in for 1–2, 3, and ≥4 pregnancies, respectively); again, the greatest increase occurred between the first two categories. Likewise, there was a significant monotonic increase in WHR with increasing parity category.
Fig. 2.
Mean values (squares) and standard deviations (lines) for BMI, waist circumference, and waist-to-hip ratio, according to number of pregnancies in the Ella study. All associations were statistically significant based on linear regression modeling (p < 0.001)
Table 2 describes the association between parity and obesity outcomes separately for BMI, WC, and WHR. While an increased odds of having a BMI ≥ 30 kg/m2 was shown for women reporting ≥4 pregnancies compared with those with 1–2 in the crude model, the OR was attenuated and no longer significant in the age- and site-adjusted and multivariate models. In the WC models, women with ≥4 pregnancies were significantly more likely to have a WC ≥ 35 inches than those with 1–2 (multivariate OR = 1.59; 95 % CI, 1.01–2.47; p-trend = 0.04). Although a significant positive association was shown for women with ≥4 pregnancies and having a WHR ≥ 0.85, this was greatly attenuated and no longer positive in the multivariate model.
Table 2.
Association between parity and measures of obesity
| Obesity measure | Number of pregnancies | N | Odds ratio (95 % confidence interval)
|
||
|---|---|---|---|---|---|
| Crude | Age- and site-adjusted | Multivariatea | |||
| Body mass index ≥30 kg/m2 | 1–2 | 328 | 1.00 | 1.00 | 1.00 |
| 3 | 281 | 1.56 (1.12–2.17) | 1.53 (1.09–2.15) | 1.32 (0.91–1.90) | |
| ≥4 | 365 | 1.57 (1.15–2.14) | 1.42 (1.02–1.99) | 1.10 (0.73–1.67) | |
| p trend | 0.006 | 0.042 | 0.674 | ||
| Waist circumference ≥35 in | 1–2 | 264 | 1.00 | 1.00 | 1.00 |
| 3 | 228 | 1.66 (1.13–2.42) | 1.59 (1.08–2.37) | 1.49 (0.98–2.24) | |
| ≥4 | 314 | 2.39 (1.66–3.45) | 1.84 (1.25–2.73) | 1.59 (1.01–2.47) | |
| p trend | <0.001 | 0.002 | 0.044 | ||
| Waist-to-hip ratio ≥0.85 | 1–2 | 262 | 1.00 | 1.00 | 1.00 |
| 3 | 227 | 1.17 (0.80–1.72) | 1.07 (0.72–1.59) | 0.95 (0.61–1.49) | |
| ≥4 | 313 | 1.43 (1.00–2.04) | 1.10 (0.74–1.62) | 0.77 (0.47–1.28) | |
| p trend | 0.051 | 0.645 | 0.316 | ||
BMI model is adjusted for age, site, age at first full-term pregnancy, and breastfeeding, and the sample size is reduced from 974 to 966 due to missing data for covariates. WC model is adjusted for age, site, education and age at first full-term pregnancy, and the sample size is reduced from 806 to 798 due to missing data for covariates. WHR multivariate model is adjusted for age, site, education, age at first full-term pregnancy, breastfeeding, smoking, physical activity, hormone contraception use, hormone replacement therapy use, and age at menarche, and the sample size is reduced from 802 to 789 due to missing data for covariates
When we conducted stratified analysis by country (Table 3), a non-significant positive association was shown between parity and BMI for the U.S. but not Mexico (OR = 1.29; 95 % CI, 0.74–2.27 and OR = 0.92; 95 % CI, 0.49–1.73 for ≥4 pregnancies vs. 1–2 for the U.S. and Mexico, respectively). For the association between parity and WHR, a positive non-significant association was shown for women in the U.S. (OR = 1.21; 95 % CI, 0.60–2.44) while a significant inverse association was shown for those in Mexico (OR = 0.42; 95 % CI, 0.19–0.90). In contrast, the magnitude of the association for the WC outcome was nearly identical in both countries (OR = 1.51; 95 % CI, 0.79–2.90 and OR = 1.48; 95 % CI, 0.76–2.87 for the U.S. and Mexico, respectively). Adjustment for acculturation in the multivariate models did not materially change the point estimates for any of the obesity outcomes. When we assessed the interaction by country for the association between parity and the obesity measures, this was not statistically significant for any of the obesity measures.
Table 3.
Association between parity and measures of obesity, stratified by country
| Obesity measure | Number of pregnancies | U.S.
|
Mexico
|
||
|---|---|---|---|---|---|
| N | Odds ratio (95 % confidence interval)a | N | Odds ratio (95 % confidence interval)a | ||
| Body mass index ≥30 kg/m2 | 1–2 | 190 | 1.00 | 135 | 1.00 |
| 3 | 147 | 1.60 (0.98–2.59) | 134 | 1.10 (0.61–1.98) | |
| ≥ 4 | 145 | 1.29 (0.74–2.27) | 215 | 0.92 (0.49–1.73) | |
| p trend | 0.324 | 0.714 | |||
| Waist circumference ≥35 in | 1–2 | 131 | 1.00 | 130 | 1.00 |
| 3 | 100 | 1.73 (0.96–3.10) | 128 | 1.22 (0.67–2.23) | |
| ≥ 4 | 103 | 1.51 (0.79–2.90) | 206 | 1.48 (0.76–2.87) | |
| p trend | 0.155 | 0.246 | |||
| Waist-to-hip ratio ≥0.85 | 1–2 | 131 | 1.00 | 127 | 1.00 |
| 3 | 99 | 1.10 (0.60–1.99) | 124 | 0.64 (0.32–1.31) | |
| ≥ 4 | 103 | 1.21 (0.60–2.44) | 205 | 0.42 (0.19–0.90) | |
| p trend | 0.587 | 0.025 | |||
BMI model is adjusted for age, site, age at first full-term pregnancy, and breastfeeding, and the sample size is reduced from 974 to 966 (US, 482; Mexico, 484) due to missing data for covariates. WC model is adjusted for age, site, education and age at first full-term pregnancy, and the sample size is reduced from 806 to 798 (US, 334; Mexico, 464) due to missing data for covariates. WHR multivariate model is adjusted for age, site, education, age at first full-term pregnancy, breastfeeding, smoking, physical activity, hormone contraception use, hormone replacement therapy use, and age at menarche, and the sample size is reduced from 802 to 789 (US, 333; Mexico, 456) due to missing data for covariates
There were no significant interactions between parity and country on any of the three obesity measures (likelihood ratio test, all p > 0.2)
Although we are likely underpowered to assess effect modification for several variables, we considered these analyses for family history of breast cancer and menopausal status. We found no significant effect modification by family history. However, for menopausal status, we observed a significant interaction with parity on BMI (p = 0.024), but not WC or WHR. However, no clear association by menopausal status was shown for parity and BMI. The OR for premenopausal women with 4 or more pregnancies versus those with 1–2 was 1.14 (95 % CI, 0.61–2.14); the corresponding OR (95 % CI) for post-menopausal women was 1.06 (0.58–1.93).
Discussion
Results of the present study show that among parous women, number of pregnancies is positively associated with increasing body size, particularly central adiposity. The positive association with WC was independent of age, recruitment site, education, and age at first full-term pregnancy and it was observed in both U.S. and Mexico. These findings are important given that WC, more than BMI, is linked to disturbances in insulin and diabetes risk [29] as well as metabolic syndrome [30]. Our results do not support an association between parity and WHR, a measure that is not recommended by the most recent NHLBI guidelines as it provides no advantage over WC alone [26].
Obesity, a chronic disease which disproportionately impacts Hispanic women is associated with a variety of metabolic disorders and several malignancies [26]. For women who develop breast cancer, the adverse consequences of obesity are complex. While dosing by surface area to achieve greatest benefit is recommended, patient level risk for cardiotoxicity in the context of underlying obesity associated co-morbid conditions (e.g., pre-existing heart disease, hypertension and diabetes) increase the risk for anthracycline associated cardiac risk [31]. Further, obese breast cancer patients suffer a 30 % higher risk of all-cause mortality than non-obese breast cancer patients [6]. This has raised discussions about how to manage the individual benefits versus risk of cancer treatment in women with co-morbid conditions that often cluster with obesity and advancing age [32]; treatment associated risks that may accelerate progression of heart disease, hypertension, and other competing causes of death.
Davis et al., [13] reported data from a prospective study of 2,923 women, which showed that parous women were 3.5 more likely to be obese (BMI ≥ 30 kg/m2) than nulliparous women. Their findings were similar for white, African-American, and Hispanic women. Additional published reports also support the direct association between parity and obesity [23]. However, results of some studies do not show a dose–response relationship between parity and having a BMI > 30 kg/m2 [14, 15, 33]. Findings from a study of African-American women showed that compared to nulliparous women, those who had one child gained more weight following childbirth; however, women who had additional children did not gain more weight than primiparous women [14]. Thus, there is no consensus in the literature regarding a linear association between parity and obesity [23]. In our study, which did not include nulliparous women, no dose–response relationship between number of pregnancies and having a BMI ≥ 30 kg/m2 was shown in the multivariate model.
Of the studies that have reported data for measures of central adiposity associated with parity, two reported positive associations [10, 11] and one did not [17]. Luoto et al., [10] showed that multiparous women had the highest WC and were 35 % more likely to have visceral obesity (≥88 cm) than nulliparous women. Results of a study among Iraqi women [11] support those in the present study, where parity significantly increased risk of having a higher WC (≥80 cm) after adjustment for various confounding variables. In regard to WHR, published data show no positive associations with parity after multivariate adjustment [11, 17], which is consistent with results from our study; however, a report published in the mid-1990s, showed that primiparas had greater increases in WHR compared to nulliparous women [12].
Limitations of the present study relate to the lack of a non-diseased/control group and its cross-sectional design, prohibiting assessment of parity as an etiologic factor for measures of obesity. Also, although we ascertained self-reported data on height and weight prior to breast cancer diagnosis, this was not possible for WC, which was measured within one year following diagnosis. Another limitation relates to the lack of data on measures of obesity prior to any pregnancy, as it has been shown that baseline BMI modifies the effect of the parity-obesity association [9, 14, 23]. This is especially important since 45 % of U.S. women are overweight or obese when they begin child-bearing [34]; furthermore, recommendations for weight gain during pregnancy vary by pre-pregnancy BMI [22]. In addition, due to the low prevalence of nulliparity in our study population, we were unable to compare obesity measures of parous versus nulliparous women. In spite of these limitations, our results for BMI and WC are supported by published studies that include a prospective design and a non-diseased population [10, 13]. Strengths of our study include the large number of women of Mexican descent and the binational design which addresses the association for women in both Mexico and Mexican–American women in the U.S. Availability of both BMI and measured values of central adiposity is an additional asset of this study.
New Contributions to the Literature
Our results show that WC is independently associated with high parity in Hispanic women. Given that the association was shown for women in Mexico and the U.S., this represents an optimal target for post-partum weight loss interventions. Participants in the Ella study represent a unique group composed entirely of women of Mexican origin living in the U.S. and Mexico. Given its binational design, our study also provides data on the parity-obesity association for women in Mexico. As the obesity epidemic continues to increase in high-, middle-, and low-resource countries, it is important to understand its etiology among populations that are most affected by this condition in order to develop targeted tailored interventions.
In summary, results of the present study show that in a highly parous population of women of Mexican descent, higher parity is associated with a greater likelihood of being obese. The effect of parity was particularly influential for visceral adiposity. Specific interventions targeting weight gain in the post-partum period for Hispanic women in the U.S. are already underway [35, 36] and may benefit from our findings especially the need to consider WC as an important target for intervention. Addressing the obesity epidemic will require identifying targeted interventions for women at different stages of life when they are at highest risk for weight gain: obesity status before conception, weight gain during pregnancy, and weight retention in the post-partum period.
Acknowledgments
We are indebted to Ana Lilia Amador, Leticia Cordova, Carole Kepler, Giovanna Cruz, Rachel Zenuk Garcia, and Fang Wang for their contributions. Grant support: Avon Foundation, Arizona Cancer Center Core Grant from the National Cancer Institute (CA-023074-2953), M.D. Anderson Cancer Center Specialized Program of Research Excellence in Breast Cancer (P50 CA116199-02S1), and Susan G. Komen for the Cure® (KG090934).
Contributor Information
María Elena Martínez, Email: e8martinez@ucsd.edu, Moores UCSD Cancer Center, University of California, San Diego, 3855 Health Sciences Drive, #0901, La Jolla, CA 92093-0901, USA.
Erika Pond, Mel and Enid Zuckerman College of Public Health, University of Arizona, Tucson, AZ, USA. Arizona Cancer Center, Tucson, AZ, USA.
Betsy C. Wertheim, Arizona Cancer Center, Tucson, AZ, USA
Jesse N. Nodora, Moores UCSD Cancer Center, University of California, San Diego, 3855 Health Sciences Drive, #0901, La Jolla, CA 92093-0901, USA
Elizabeth T. Jacobs, Mel and Enid Zuckerman College of Public Health, University of Arizona, Tucson, AZ, USA. Arizona Cancer Center, Tucson, AZ, USA
Melissa Bondy, Dan L. Duncan Cancer Center, Baylor College of Medicine, Houston, TX, USA.
Adrian Daneri-Navarro, Universidad de Guadalajara, Guadalajara, Jalisco, Mexico.
Maria Mercedes Meza-Montenegro, Instituto Tecnológico de Sonora, Ciudad Obregón, Sonora, Mexico.
Luis Enrique Gutierrez-Millan, Universidad de Sonora, Hermosillo, Sonora, Mexico.
Abenaa Brewster, Department of Clinical Cancer Prevention, University of Texas M.D. Anderson Cancer Center, Houston, TX, USA.
Ian K. Komenaka, Department of Surgery, Maricopa Medical Center, Phoenix, AZ, USA
Patricia Thompson, Arizona Cancer Center, Tucson, AZ, USA. Department of Cellular and Molecular Medicine, College of Medicine, University of Arizona, Tucson, AZ, USA.
References
- 1.Kumanyika S, Jeffery RW, Morabia A, Ritenbaugh C, Antipatis VJ. Obesity prevention: the case for action. Int J Obes Relat Metab Disord. 2002;26(3):425–36. doi: 10.1038/sj.ijo.0801938. (Epub 2002/03/16) [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization. Obesity: preventing and managing the global epidemic—Introduction. WHO Technical Report Series. 2000;894:1–253. [PubMed] [Google Scholar]
- 3.Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA. 2006;295(13):1549–55. doi: 10.1001/jama.295.13.1549. [DOI] [PubMed] [Google Scholar]
- 4.Flegal KM, Carroll MD, Ogden CL, Curtin LR. Prevalence and trends in obesity among US adults, 1999–2008. JAMA. 2010;303(3):235–41. doi: 10.1001/jama.2009.2014. (Epub 2010/01/15) [DOI] [PubMed] [Google Scholar]
- 5.Sarwer DB, Allison KC, Gibbons LM, Markowitz JT, Nelson DB. Pregnancy and obesity: a review and agenda for future research. J Womens Health. 2006;15(6):720–33. doi: 10.1089/jwh.2006.15.720. (Epub 2006/08/17) [DOI] [PubMed] [Google Scholar]
- 6.Patterson RW, Cadmus LA, Emond JA, Pierce JP. Physical activity, diet, adiposity and female breast cancer prognosis: a review of the epidemiologic literature. Maturitas. 2010;66:5–15. doi: 10.1016/j.maturitas.2010.01.004. [DOI] [PubMed] [Google Scholar]
- 7.Griggs JJ, Sorbero MES, Lyman GH. Undertreatment of obese women receiving breast cancer chemotherapy. Arch Intern Med. 2005;165(11):1267–73. doi: 10.1001/archinte.165.11.1267. [DOI] [PubMed] [Google Scholar]
- 8.Greenman CG, Jagielski CH, Griggs JJ. Breast cancer adjuvant chemotherapy dosing in obese patients. Cancer. 2008;112(10): 2159–65. doi: 10.1002/cncr.23416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Coitinho DC, Sichieri R, D’Aquino Benicio MH. Obesity and weight change related to parity and breast-feeding among parous women in Brazil. Public Health Nutr. 2001;4(4):865–70. doi: 10.1079/phn2001125. (Epub 2001/08/31) [DOI] [PubMed] [Google Scholar]
- 10.Luoto R, Mannisto S, Raitanen J. Ten-year change in the association between obesity and parity: results from the National FINRISK Population Study. Gend Med. 2011;8(6):399–406. doi: 10.1016/j.genm.2011.11.003. (Epub 2011/12/14) [DOI] [PubMed] [Google Scholar]
- 11.Mansour AA, Ajeel NA. Parity is associated with increased waist circumference and other anthropometric indices of obesity. Eat Weight Disord. 2009;14(2–3):e50–5. doi: 10.1007/BF03327800. (Epub 2009/11/26) [DOI] [PubMed] [Google Scholar]
- 12.Smith DE, Lewis CE, Caveny JL, Perkins LL, Burke GL, Bild DE. Longitudinal changes in adiposity associated with pregnancy. The CARDIA Study. Coronary Artery Risk Development in Young Adults Study. JAMA. 1994;271(22):1747–51. (Epub 1994/06/08) [PubMed] [Google Scholar]
- 13.Davis EM, Zyzanski SJ, Olson CM, Stange KC, Horwitz RI. Racial, ethnic, and socioeconomic differences in the incidence of obesity related to childbirth. Am J Public Health. 2009;99(2): 294–9. doi: 10.2105/AJPH.2007.132373. (Epub 2008/12/09) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rosenberg L, Palmer JR, Wise LA, Horton NJ, Kumanyika SK, Adams-Campbell LL. A prospective study of the effect of childbearing on weight gain in African-American women. Obes Res. 2003;11(12):1526–35. doi: 10.1038/oby.2003.204. [DOI] [PubMed] [Google Scholar]
- 15.Williamson DF, Madans J, Pamuk E, Flegal KM, Kendrick JS, Serdula MK. A prospective study of childbearing and 10-year weight gain in U.S. white women 25 to 45 years of age. Int J Obes Relat Metab Disord. 1994;18(8):561–9. [PubMed] [Google Scholar]
- 16.Wolfe WS, Sobal J, Olson CM, Frongillo EAJ, Williamson DF. Parity-associated weight gain and its modification by sociode-mographic and behavioral factors: a prospective analysis in US women. Int J Obes Relat Metab Disord. 1997;21(9):802–10. doi: 10.1038/sj.ijo.0800478. [DOI] [PubMed] [Google Scholar]
- 17.Koch E, Bogado M, Arahya F, Romero T, Diaz C, Manriquez L, Paredes M, Roman C, Taylor A, Kirschbaum A. Impact of parity on anthropometric measures of obesity controlling by multiple confounders: a cross sectional study in Chilean women. J Epi-demiol Community Health. 2007;62:461–70. doi: 10.1136/jech.2007.062240. [DOI] [PubMed] [Google Scholar]
- 18.Arroyo P, Avila-Rosas H, Fernandez V, Casanueva E, Galvan D. Pairty and the prevalence of overweight. Int Fed Byn Obstet. 1995;48:269–72. doi: 10.1016/0020-7292(94)02284-6. [DOI] [PubMed] [Google Scholar]
- 19.Ennis SR, Rios-Vargas M, Albert NG. 2010 Census briefs. 2011. The Hispanic Population: 2010. Contract no.: C210BR-04. [Google Scholar]
- 20.Dye JL. Fertility of American Women: 2006. U.S. Department of Commerce. Economics and Statistics Administration. U.S. Census Bureau; 2008. [Google Scholar]
- 21.Kim SA, Yount KM, Ramakrishnan U, Martorell R. The relationship between parity and overweight varies with household wealth and national development. Int J Epidemiol. 2007;36(1): 93–101. doi: 10.1093/Ije/Dyl252. [DOI] [PubMed] [Google Scholar]
- 22.Institute of Medicine (IOM) and National Research Council (NRC) Weight gain during pregnancy: reexamining the guidelines. Washingon, DC: The National Academies Press; 2009. [PubMed] [Google Scholar]
- 23.Gunderson EP. Childbearing and obesity in women: weight before, during, and after pregnancy. Obstet Gynecol Clin North Am. 2009;36(2):317–32. ix. doi: 10.1016/j.ogc.2009.04.001. (Epub 2009/06/09) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Martínez ME, Gutiérrez-Millan LE, Bondy M, Daneri-Navarro A, Meza-Montenegro MM, Anduro-Corona I, Aramburo-Rubio MI, Balderas-Peña LMA, Barragan-Ruiz JA, Brewster A, Caire-Juvera G, Castro-Cervantes JM, Zamudio MAC, Cruz G, Del Toro-Arreola A, Edgerton ME, Flores-Marquez MR, Franco-Topete RA, Garcia H, Gutierrez-Rubio SA, Hahn K, Jimenez-Perez LM, Komenaka IK, Bujanda ZAL, Lu D, Morgan-Villela G, Murray JL, Nodora JN, Oceguera-Villanueva A, Martínez MAO, Michel LP, Quintero-Ramos A, Sahin A, Shim JY, Stewart M, Vazquez-Camacho G, Wertheim B, Zenuk R, Thompson P. Comparative study of breast cancer in Mexican and Mexican-American women. Health. 2010;2(9):1040–8. [Google Scholar]
- 25.World Health Organization. Teport of a WHO consultiation. Geneva: 2000. Obesity: preventing and managing the global epidemic. [PubMed] [Google Scholar]
- 26.NHLBI. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. Bethesda, MD: National Heart, Lung, and Blood Institute; 1998. Obesity education initiative expert panel on the identi-fication, evaluation, and treatment of overweight and obesity in adults. [Google Scholar]
- 27.Garcia RZ, Carvajal SC, Wilkinson AV, Thompson PA, Nodora JN, Komenaka IK, Brewster A, Cruz GI, Wertheim BC, Bondy ML, Martínez ME. Factors that influence mammography use and breast cancer detection among Mexican-American and African-American women. Cancer Causes Control. 2012;23(1):165–73. doi: 10.1007/s10552-011-9865-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Marin G, Gamba RJ. A new measurement of acculturation for Hispanics: the bidimensional acculturation scale for Hispanics (BAS) Hispanic J Behav Sci. 1996;18(3):297–316. [Google Scholar]
- 29.Despres JP, Lemieux I, Prud’homme D. Treatment of obesity: need to focus on high risk abdominally obese patients. BMJ. 2001;322(7288):716–20. doi: 10.1136/bmj.322.7288.716. (Epub 2001/03/27) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Despres JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444(7121):881–7. doi: 10.1038/Nature05488. [DOI] [PubMed] [Google Scholar]
- 31.Barrett-Lee PJ, Dixon JM, Farrell C, Jones A, Leonard R, Murray N, Palmieri C, Plummer CJ, Stanley A, Verrill MW. Expert opinion on the use of anthracyclines in patients with advanced breast cancer at cardiac risk. Ann Oncol. 2009;20(5):816–27. doi: 10.1093/annonc/mdn728. [DOI] [PubMed] [Google Scholar]
- 32.Giordano SH, Hortobagyi GN. Time to remove the subspecialty blinders: breast cancer does not exist in isolation. J Natl Cancer Inst. 2008;100(4):230–1. doi: 10.1093/jnci/djn015. [DOI] [PubMed] [Google Scholar]
- 33.Brown JE, Kaye SA, Folsom AR. Parity-related weight change in women. Int J Obes Relat Metab Disord. 1992;16(9):627–31. (Epub 1992/09/01) [PubMed] [Google Scholar]
- 34.Pediatric & pregnancy nutrition surveillance system. 2009 Available at http://www.cdc.gov/pednss/pnss_tables/tables_health_indicators.htm [Internet] [cited February 10, 2012]
- 35.Chasan-Tabber L, Silveira M, Marcus BH, Braum B, Stanek E, Markenson G. Feasibility and efficacy of a physical activity intervention among pregnant women: the Behaviors Affecting Baby and You (B.A.B.Y.) Study. J Phys Act Health. 2011;8(Suppl 2):S228–38. [PMC free article] [PubMed] [Google Scholar]
- 36.Keller C, Records K, Ainsworth B, Belyea M, Permana P, Coonrod D, Vega-Lopez S, Nagle-Williams A. Madres para la Salud: design of a theory-based intervention for postpartum Latinas. Contemp Clin Trials. 2011;32:418–27. doi: 10.1016/j.cct.2011.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]