Abstract
NAD+ is a central metabolite in the cell. Changes in NAD+ abundance and the activity of NAD+-dependent enzymes, such as the sirtuins, are at the core of metabolic/mitochondrial diseases, such as obesity and diabetes, and of cancer and neurodegeneration. Here, we discuss how maintaining or raising NAD+ levels can improve metabolism and prevent age-related functional decline and associated disease, and how basic scientific discoveries in the NAD+ signaling pathway are being translated to the clinic.
Nicotinamide adenine dinucleotide (NAD) is a metabolite that was first identified over 100 years ago. At that time, Nobel laureate Sir Arthur Harden discovered a molecular fraction involved in yeast fermentation that he called “cozymase.” Subsequent work of, among others, three additional Nobel laureates, notably Otto Warburg, led to the identification of NAD+ as an essential cofactor of many biochemical reactions either in its oxidized (NAD+) or reduced (NADH) form (Berger et al., 2004; Houtkooper et al., 2010a). For instance, NAD+ is used as a cofactor in glucose and fat breakdown, whereas NADH is used as a substrate for the electron transport chain (Fig. 1). NAD+ precursors have been used extensively as dietary supplements: nicotinic acid (NA; also known as niacin) and nicotinamide (NAM) have been long used to treat pellagra, a niacin deficiency (Elvehjem et al., 1937). Niacin is also a commonly used anti-hyperlipidemic drug. Yet it is only in recent years, after the discovery of the sirtuin protein family, that NAD+ metabolism has emerged as a hot topic for drug development. The key discoveries that led to this increased interest in NAD+ metabolism were the finding that yeast Sir2p was required for the lifespan extension brought about by caloric restriction (Lin et al., 2000), and the identification of sirtuins as NAD+-dependent enzymes (Imai et al., 2000). These two key findings suggested that this class of enzymes could “sense” the metabolic state of the cell, as the NAD+/NADH ratio is largely influenced by the availability and breakdown of nutrients. Because some of the sirtuins in turn regulate the activity of key (transcriptional) metabolic regulators, such as FOXOs, PGC-1α, and p53, the NAD+/sirtuin axis was shown to play a predominant regulatory role in metabolism (Fig. 2 A). Clinical interest was primarily raised by the finding that NAD+/sirtuins promote mitochondrial function, which has made this pathway an attractive target for treating diseases with a mitochondrial contribution, ranging from genetic mitochondrial diseases to cancer and common age-related diseases, including those of the metabolic and nervous systems (Guarente, 2008; Houtkooper et al., 2012). Here, we discuss different means of NAD+ modulation with a particular focus on compounds such as NAD+ precursors, PARP inhibitors, and resveratrol, and describe how these induce mitochondrial function and translate to clinical applications.
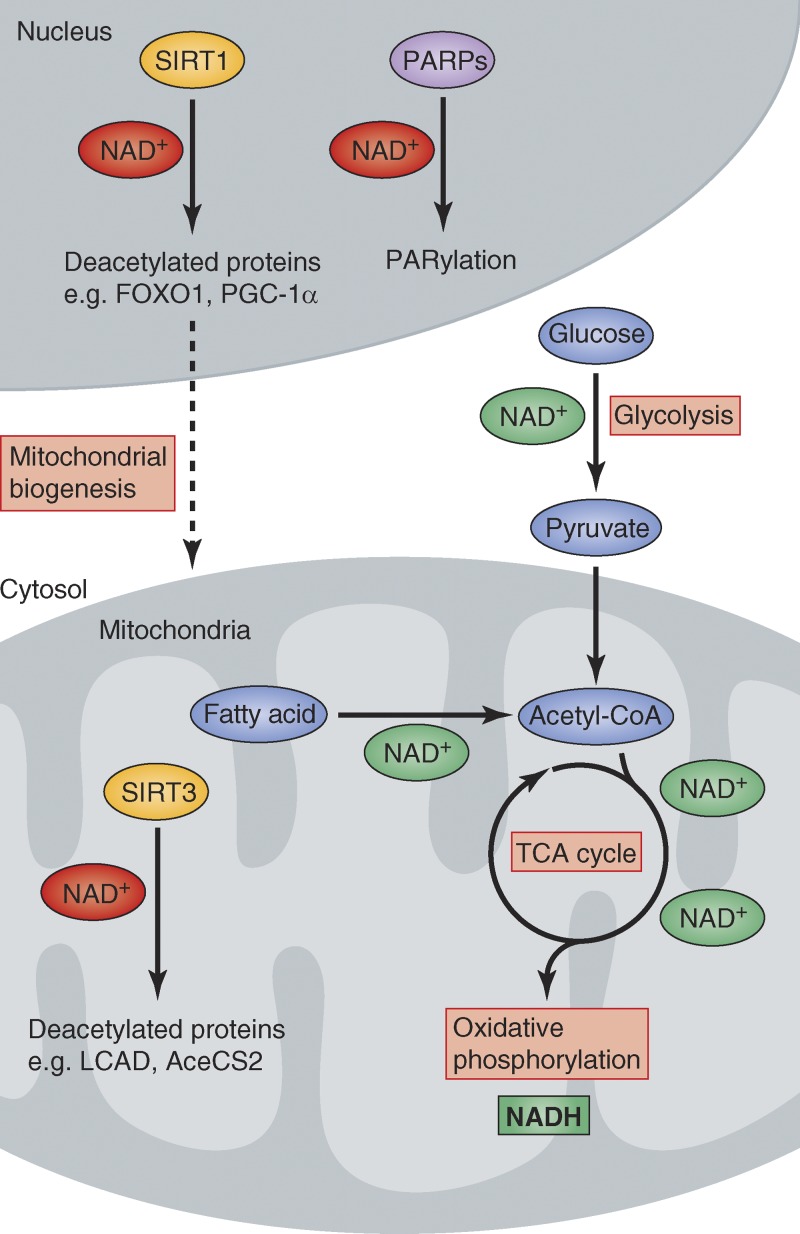
Figure 1.
Diverse metabolic actions of NAD+. Schematic overview of how NAD+ and NADH are used as metabolic cofactors, either in more traditional oxidoreductase reactions (green NAD+) or in more recently discovered regulatory roles as a cosubstrate for the reactions catalyzed by sirtuins and PARPs (red NAD+). Note: reactions shown here are only a small representation of those that involve NAD+.
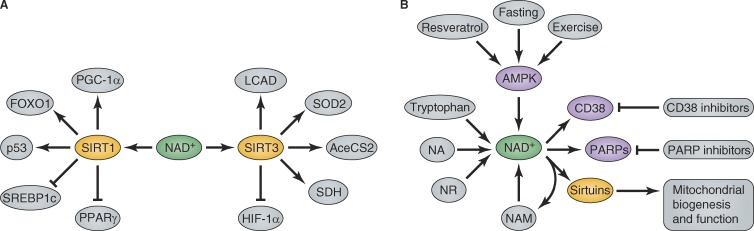
Figure 2.
Metabolic control by modulating NAD+ levels. (A) NAD+ levels dictate the activity of SIRT1 and SIRT3, which in turn regulate a variety of metabolic proteins. The protein targets shown here just represent a selection. (B) Mitochondrial function is regulated by sirtuin enzymes, notably SIRT1 and SIRT3, which are under the control of NAD+ levels. Modulating NAD+ levels results in improved mitochondrial function with clinically beneficial effects.
NAD+ metabolism—the bench
The biochemistry of NAD+ metabolism.
The central role of NAD+ in nutrient breakdown and metabolic sensing requires a delicate balance in its production and utilization. NAD+ levels are controlled by balancing biosynthesis and salvage on one side, and breakdown on the other (Fig. 2 B). NAD+ can be synthesized from the amino acid tryptophan, but its prime precursors include NA and NAM and the more recently identified nicotinamide riboside (NR; Fig. 2 B; Bieganowski and Brenner, 2004). Conversely, the main NAD+ breakdown pathways involves three enzyme classes, i.e., sirtuins, poly(ADP-ribose) polymerases (PARPs), and cyclic ADP-ribose synthases (Fig. 2 B). Although PARPs and cyclic ADP-ribose synthases play a crucial role in NAD+ homeostasis, their function by itself is less relevant for our story, which is why we will not discuss them in detail and refer the reader to a recent review (Houtkooper et al., 2010a).
Modulation of NAD+ levels.
The NAD+/sirtuin pathway adapts metabolism in response to environmental (nutrition, exercise) challenges. During energy crises such as caloric restriction, fasting, and exercise, NAD+ levels rise, concomitant with sirtuin activation (Chen et al., 2008; Cantó et al., 2010), while energy overload, such as seen with a high-fat diet, reduces the NAD+/NADH ratio (Kim et al., 2011).
Besides physiological processes, NAD+ levels can be modulated pharmacologically. As a case in point, the polyphenol resveratrol, often described as a red wine compound, activates the energy sensor AMP-activated protein kinase (AMPK; Baur et al., 2006; Cantó et al., 2009), which in turn stimulates NAD+ (re)synthesis (Fulco et al., 2008; Cantó et al., 2009). Alternatively, supplying precursors or inhibiting NAD+ consumers also increases NAD+ levels. Supplementation of NA, NR, or NAD+ biosynthesis intermediates, like nicotinamide mononucleotide (NMN), increases NAD+ levels in cultured cells or in tissues of mice receiving the compounds (Yoshino et al., 2011; Cantó et al., 2012). Similarly, treatment with either inhibitors of PARPs (Bai et al., 2011b) or CD38 (Barbosa et al., 2007), both enzymes that consume NAD+, will induce NAD+ levels and results in the activation of the sirtuins. Importantly from a therapeutic perspective, because NR and probably also the other precursors can be metabolized both in the nucleus and in mitochondria, NR supplementation leads to accumulation of nuclear and mitochondrial NAD+, and subsequent activation of both the nuclear SIRT1 and the mitochondrial SIRT3 (Cantó et al., 2012). On the other hand, the NAD+ consumer PARP1 is confined to the nucleus. This implies that increased NAD+ levels in tissues of Parp1 knockout mice are accompanied by nuclear SIRT1, but not mitochondrial SIRT3, activation (Bai et al., 2011b). Future work will have to address whether PARP inhibitors also activate the other nuclear sirtuins, SIRT6 and SIRT7, and how inhibitors of the ectoenzyme CD38 modulate subcellular NAD+ levels and sirtuin activity.
Sirtuin activation and mitochondrial metabolism.
The yeast sirtuin enzyme Sir2p was described to prevent genome instability in yeast, thereby acting as an anti-aging factor (Haigis and Sinclair, 2010; Houtkooper et al., 2012). In mammals, the sirtuin family consists of seven members, SIRT1–7, with distinct enzymatic activities and subcellular localization (Houtkooper et al., 2012). As discussed earlier, sirtuins function mainly as deacetylases and are optimally placed to translate changes in nutritional state to metabolic adaptations. SIRT1 is the best-characterized sirtuin and is implicated in many nonmetabolic processes as well. The prime metabolic target for SIRT1 is PGC-1α, a cofactor that regulates mitochondrial biogenesis and function (Fig. 2 A; Lin et al., 2005). From the other SIRT1 deacetylation targets, the forkhead transcription factor FOXO1 is worth mentioning in this context as it modulates mitochondrial fatty acid metabolism and protects against oxidative stress (van der Horst and Burgering, 2007). The key mitochondrial sirtuin, SIRT3, targets several proteins involved in fatty acid metabolism and ketogenesis, and antioxidant enzymes (Fig. 2 A; Houtkooper et al., 2012), although its real physiological role in metabolic control is still debated (Hirschey et al., 2010; Fernandez-Marcos et al., 2012). Through this multitude of sirtuin targets, NAD+ has a profound effect on mitochondrial function in general, although future work will need to clarify how these pleiotropic actions are controlled by specific sirtuins.
Clinical application of NAD+ metabolism—the bedside
With the central position of NAD+ in cellular metabolism in mind, the question is whether increasing its level could indeed be clinically beneficial, similar to the effects of caloric restriction. The key to answering this question is whether NAD+ is a limiting factor for the enzymes that require NAD+ as a cosubstrate. In support of this premise is the fact that high-dose vitamin and micronutrient therapy have been known to yield pleiotropic beneficial effects on multiple metabolic pathways (Ames, 2006). Boosting NAD+ levels may hence have similar favorable effects on pathways that require NAD+ as a cosubstrate.
Hyperlipidemia.
For a long time, patients with hyperlipidemia have been treated with niacin (Altschul et al., 1955), often in combination with statins to lower cholesterol biosynthesis. Although the G protein–coupled receptor GPR109A was linked to the effects of niacin (Tunaru et al., 2003), an increase in NAD+ could also contribute, especially considering the central role of sirtuin enzymes in lipid metabolism (Schug and Li, 2011; Houtkooper et al., 2012). Arguing in favor of the latter hypothesis is the fact that high doses of niacin (grams/day) are required for lipid lowering, while the EC50 for GPR109A (i.e., concentration to activate 50% of the receptor; ∼250 nmol/L) is rather low (Wise et al., 2003). Interestingly, supplementation with NR decreased the LDL/HDL ratio in mice without activating GPR109A, suggesting that this may be true (Cantó et al., 2012). This would circumvent the GPR109A-mediated adverse effects, such as flushing, observed upon high-level niacin intake (Cantó et al., 2012). Further mechanistic studies with other NAD-centered approaches leading to sirtuin activation are required to establish the potential of the NAD+/sirtuin axis for lipid lowering.
Inherited mitochondrial diseases.
As described in the “Sirtuin activation and mitochondrial metabolism” section, modulating NAD+ levels has profound effects on mitochondrial function, through the actions of SIRT1 and SIRT3 primarily (Houtkooper et al., 2012). Hence, it is tempting to speculate that compounds altering NAD+ levels could be used to treat or alleviate inherited diseases of mitochondrial function that often affect young children. Indications of the success of such an approach in mouse models are emerging. For instance, treatment with the AMPK agonist, AICAR, rescued the mitochondrial dysfunction and the limited exercise capacity of cytochrome c oxidase–deficient mice (Viscomi et al., 2011). This effect could well be linked to the fact that AMPK activation boosts NAD+ generation, subsequently stimulating SIRT1 and mitochondrial homeostasis (Cantó and Auwerx, 2012), although this has not been conclusively demonstrated. Inherited metabolic diseases, which are often rare, may therefore be good therapeutic indications to test the potential effects of strategies to induce NAD+ levels.
Acquired mitochondrial diseases.
Mitochondrial dysfunction is critically involved in the development of metabolic (e.g., obesity, type 2 diabetes) and neurodegenerative diseases (e.g., Alzheimer’s, Parkinson’s; Lin and Beal, 2006; Houtkooper et al., 2010b; Patti and Corvera, 2010). This suggests that improving mitochondrial homeostasis, for instance by activating the sirtuin pathway, would result in clinically relevant effects. In support of this hypothesis, high Sirt1 mRNA expression in adipose tissue of nondiabetic offspring of type 2 diabetic patients corresponds to increased energy expenditure and insulin sensitivity (Rutanen et al., 2010). The link is further corroborated as several NAD+ boosters, including resveratrol, NMN, and NR, improve mitochondrial function in mice and correct the metabolic disturbances instigated by a high-fat diet regimen (Baur et al., 2006; Lagouge et al., 2006; Yoshino et al., 2011; Cantó et al., 2012). PARP inhibitors could be an attractive alternative to increase NAD+ and improve metabolic flexibility (Bai et al., 2011b), as these compounds are in phase III trials as anti-cancer agents (Rouleau et al., 2010). The fact that PARP inhibitors increase oxidative metabolism—an anti-Warburg effect—could actually contribute to their efficacy against cancers (Bai et al., 2011b). Based on the phenotype of the Parp2-deficient mice, however, care should be taken to exclude that the pancreatic dysfunction observed in these mice interferes with the beneficial effects seen in PARP inhibitor–treated mice (Bai et al., 2011a). Finally, it is encouraging that the AMPK agonist resveratrol also activates mitochondrial function in humans (Timmers et al., 2011) at a dose that is more than 200-fold lower than the dose used in mice (Baur et al., 2006; Lagouge et al., 2006). Clinically, resveratrol improved metabolic parameters such as blood glucose and inflammatory markers (Timmers et al., 2012). Given the fact that several compounds are on the market (e.g., niacin), in clinical development (e.g., PARP inhibitors), or can be easily developed because they are natural products (e.g., NR), new detailed clinical studies targeting the NAD+/sirtuin pathway are to be expected soon. It is important to keep in mind, however, that side effects that may be acceptable for those afflicted with devastating and debilitating mitochondrial genetic diseases may be unacceptable in the wider, generally healthy population suffering from acquired mitochondrial diseases.
Aging and age-related metabolic diseases.
Aging is intimately linked with metabolism (Houtkooper et al., 2010b). Based on data in lower model organisms and beneficial effects on metabolism and many age-related diseases, SIRT1 was considered as a bona fide anti-aging protein (for more detailed discussion, see Houtkooper et al., 2012). This initial excitement is now toned down, as SIRT1 overexpression or resveratrol treatment by themselves may not always be sufficient to extend lifespan in a major fashion (Tissenbaum and Guarente, 2001; Howitz et al., 2003; Bass et al., 2007; Burnett et al., 2011). This circumstance may be analogous to the situation for many nuclear receptors, where the activation of a given endocrine pathway requires both the presence of the receptor and availability of the corresponding ligand. This premise was underscored by the fact that nuclear receptor knockout mice are almost never a mirror image of ligand activation. Therefore, it is interesting to see whether pharmacological activation, by boosting levels of the rate-limiting NAD+ cosubstrate, in the context of SIRT1 overexpression could synergistically prolong life.
Still, there is ample evidence that SIRT1 activation can improve health and prevent age-related disease. SIRT1 overexpressing transgenic mice are protected against age-related development of diabetes, and have lower incidence of carcinomas and sarcomas, even though they do not live longer (Herranz et al., 2010), suggesting that Sirt1 regulates healthy aging rather than lifespan itself. Mice that were metabolically challenged with a high-fat diet also lived longer when resveratrol was supplemented in the diet (Pearson et al., 2008). Unfortunately, even though there is strong evidence that resveratrol and other AMPK activators increase NAD+ levels and SIRT1 activity, they may have pleiotropic effects, and therefore pure NAD+ boosters should undergo further testing for their effects on aging.
Perspectives
The identification of the NAD+–sirtuin–mitochondria axis opened an exciting new area of research with great clinical potential. Even though the field suffered from overexcitement in early years, when the miracle genes and drugs were glorified in scientific and public media, a more realistic sense of belief has settled in now. Given the robust impact of the NAD+–sirtuin pathway on mitochondrial and metabolic homeostasis, it is almost certain that future studies will further bolster the clinical value of this ancient and evolutionary conserved signaling pathway. It is therefore expected that a variety of new pharmaceutical and nutriceutical “NAD+ compounds” may find their way to the clinic. Even though such “NAD+ therapeutics” may not become life extension drugs, we are convinced they will maintain mitochondrial fitness and support a healthy life.
Acknowledgments
R.H. Houtkooper is supported by an AMC Postdoc fellowship. J. Auwerx is the Nestlé Chair in Energy Metabolism and is supported by the École Polytechnique Fédérale de Lausanne, the EU Ideas program (ERC-2008-AdG-23118), the Velux Stiftung, and the Swiss National Science Foundation (31003A-124713 and CRSII3-136201). Illustrations were provided by Neil Smith, www.neilsmithillustration.co.uk.
Footnotes
Abbreviations used in this paper:
- AMPK
- AMP-activated protein kinase
- NAD
- nicotinamide adenine dinucleotide
- NR
- nicotinamide riboside
- PARP
- poly(ADP-ribose) polymerase
References
- Altschul R., Hoffer A., Stephen J.D. 1955. Influence of nicotinic acid on serum cholesterol in man. Arch. Biochem. Biophys. 54:558–559 10.1016/0003-9861(55)90070-9 [DOI] [PubMed] [Google Scholar]
- Ames B.N. 2006. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc. Natl. Acad. Sci. USA. 103:17589–17594 10.1073/pnas.0608757103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai P., Canto C., Brunyánszki A., Huber A., Szántó M., Cen Y., Yamamoto H., Houten S.M., Kiss B., Oudart H., et al. 2011a. PARP-2 regulates SIRT1 expression and whole-body energy expenditure. Cell Metab. 13:450–460 10.1016/j.cmet.2011.03.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai P., Cantó C., Oudart H., Brunyánszki A., Cen Y., Thomas C., Yamamoto H., Huber A., Kiss B., Houtkooper R.H., et al. 2011b. PARP-1 inhibition increases mitochondrial metabolism through SIRT1 activation. Cell Metab. 13:461–468 10.1016/j.cmet.2011.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbosa M.T., Soares S.M., Novak C.M., Sinclair D., Levine J.A., Aksoy P., Chini E.N. 2007. The enzyme CD38 (a NAD glycohydrolase, EC 3.2.2.5) is necessary for the development of diet-induced obesity. FASEB J. 21:3629–3639 10.1096/fj.07-8290com [DOI] [PubMed] [Google Scholar]
- Bass T.M., Weinkove D., Houthoofd K., Gems D., Partridge L. 2007. Effects of resveratrol on lifespan in Drosophila melanogaster and Caenorhabditis elegans. Mech. Ageing Dev. 128:546–552 10.1016/j.mad.2007.07.007 [DOI] [PubMed] [Google Scholar]
- Baur J.A., Pearson K.J., Price N.L., Jamieson H.A., Lerin C., Kalra A., Prabhu V.V., Allard J.S., Lopez-Lluch G., Lewis K., et al. 2006. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 444:337–342 10.1038/nature05354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger F., Ramírez-Hernández M.H., Ziegler M. 2004. The new life of a centenarian: signalling functions of NAD(P). Trends Biochem. Sci. 29:111–118 10.1016/j.tibs.2004.01.007 [DOI] [PubMed] [Google Scholar]
- Bieganowski P., Brenner C. 2004. Discoveries of nicotinamide riboside as a nutrient and conserved NRK genes establish a Preiss-Handler independent route to NAD+ in fungi and humans. Cell. 117:495–502 10.1016/S0092-8674(04)00416-7 [DOI] [PubMed] [Google Scholar]
- Burnett C., Valentini S., Cabreiro F., Goss M., Somogyvári M., Piper M.D., Hoddinott M., Sutphin G.L., Leko V., McElwee J.J., et al. 2011. Absence of effects of Sir2 overexpression on lifespan in C. elegans and Drosophila. Nature. 477:482–485 10.1038/nature10296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantó C., Auwerx J. 2012. Targeting sirtuin 1 to improve metabolism: all you need is NAD(+)? Pharmacol. Rev. 64:166–187 10.1124/pr.110.003905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantó C., Gerhart-Hines Z., Feige J.N., Lagouge M., Noriega L., Milne J.C., Elliott P.J., Puigserver P., Auwerx J. 2009. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 458:1056–1060 10.1038/nature07813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantó C., Jiang L.Q., Deshmukh A.S., Mataki C., Coste A., Lagouge M., Zierath J.R., Auwerx J. 2010. Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab. 11:213–219 10.1016/j.cmet.2010.02.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantó C., Houtkooper R.H., Pirinen E., Youn D.Y., Oosterveer M.H., Cen Y., Fernandez-Marcos P.J., Yamamoto H., Andreux P.A., Cettour-Rose P., et al. 2012. The NAD(+) precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell Metab. 15:838–847 10.1016/j.cmet.2012.04.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D., Bruno J., Easlon E., Lin S.J., Cheng H.L., Alt F.W., Guarente L. 2008. Tissue-specific regulation of SIRT1 by calorie restriction. Genes Dev. 22:1753–1757 10.1101/gad.1650608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elvehjem C., Madden R., Strong F., Woolley D. 1937. Relation of nicotinic acid and nicotinic acid amide to canine black tongue. J. Am. Chem. Soc. 59:1767–1768 10.1021/ja01288a509 [DOI] [Google Scholar]
- Fernandez-Marcos P.J., Jeninga E.H., Canto C., Harach T., de Boer V.C., Andreux P., Moullan N., Pirinen E., Yamamoto H., Houten S.M., et al. 2012. Muscle or liver-specific Sirt3 deficiency induces hyperacetylation of mitochondrial proteins without affecting global metabolic homeostasis. Sci Rep. 2:425 10.1038/srep00425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fulco M., Cen Y., Zhao P., Hoffman E.P., McBurney M.W., Sauve A.A., Sartorelli V. 2008. Glucose restriction inhibits skeletal myoblast differentiation by activating SIRT1 through AMPK-mediated regulation of Nampt. Dev. Cell. 14:661–673 10.1016/j.devcel.2008.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guarente L. 2008. Mitochondria—a nexus for aging, calorie restriction, and sirtuins? Cell. 132:171–176 10.1016/j.cell.2008.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haigis M.C., Sinclair D.A. 2010. Mammalian sirtuins: biological insights and disease relevance. Annu. Rev. Pathol. 5:253–295 10.1146/annurev.pathol.4.110807.092250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herranz D., Muñoz-Martin M., Cañamero M., Mulero F., Martinez-Pastor B., Fernandez-Capetillo O., Serrano M. 2010. Sirt1 improves healthy ageing and protects from metabolic syndrome-associated cancer. Nat Commun. 1:3 10.1038/ncomms1001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirschey M.D., Shimazu T., Goetzman E., Jing E., Schwer B., Lombard D.B., Grueter C.A., Harris C., Biddinger S., Ilkayeva O.R., et al. 2010. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature. 464:121–125 10.1038/nature08778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houtkooper R.H., Cantó C., Wanders R.J., Auwerx J. 2010a. The secret life of NAD+: an old metabolite controlling new metabolic signaling pathways. Endocr. Rev. 31:194–223 10.1210/er.2009-0026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houtkooper R.H., Williams R.W., Auwerx J. 2010b. Metabolic networks of longevity. Cell. 142:9–14 10.1016/j.cell.2010.06.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houtkooper R.H., Pirinen E., Auwerx J. 2012. Sirtuins as regulators of metabolism and healthspan. Nat. Rev. Mol. Cell Biol. 13:225–238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howitz K.T., Bitterman K.J., Cohen H.Y., Lamming D.W., Lavu S., Wood J.G., Zipkin R.E., Chung P., Kisielewski A., Zhang L.L., et al. 2003. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature. 425:191–196 10.1038/nature01960 [DOI] [PubMed] [Google Scholar]
- Imai S., Armstrong C.M., Kaeberlein M., Guarente L. 2000. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature. 403:795–800 10.1038/35001622 [DOI] [PubMed] [Google Scholar]
- Kim H.J., Kim J.H., Noh S., Hur H.J., Sung M.J., Hwang J.T., Park J.H., Yang H.J., Kim M.S., Kwon D.Y., Yoon S.H. 2011. Metabolomic analysis of livers and serum from high-fat diet induced obese mice. J. Proteome Res. 10:722–731 10.1021/pr100892r [DOI] [PubMed] [Google Scholar]
- Lagouge M., Argmann C., Gerhart-Hines Z., Meziane H., Lerin C., Daussin F., Messadeq N., Milne J., Lambert P., Elliott P., et al. 2006. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell. 127:1109–1122 10.1016/j.cell.2006.11.013 [DOI] [PubMed] [Google Scholar]
- Lin M.T., Beal M.F. 2006. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 443:787–795 10.1038/nature05292 [DOI] [PubMed] [Google Scholar]
- Lin J., Handschin C., Spiegelman B.M. 2005. Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab. 1:361–370 10.1016/j.cmet.2005.05.004 [DOI] [PubMed] [Google Scholar]
- Lin S.J., Defossez P.A., Guarente L. 2000. Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science. 289:2126–2128 10.1126/science.289.5487.2126 [DOI] [PubMed] [Google Scholar]
- Patti M.E., Corvera S. 2010. The role of mitochondria in the pathogenesis of type 2 diabetes. Endocr. Rev. 31:364–395 10.1210/er.2009-0027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson K.J., Baur J.A., Lewis K.N., Peshkin L., Price N.L., Labinskyy N., Swindell W.R., Kamara D., Minor R.K., Perez E., et al. 2008. Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab. 8:157–168 10.1016/j.cmet.2008.06.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rouleau M., Patel A., Hendzel M.J., Kaufmann S.H., Poirier G.G. 2010. PARP inhibition: PARP1 and beyond. Nat. Rev. Cancer. 10:293–301 10.1038/nrc2812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutanen J., Yaluri N., Modi S., Pihlajamäki J., Vänttinen M., Itkonen P., Kainulainen S., Yamamoto H., Lagouge M., Sinclair D.A., et al. 2010. SIRT1 mRNA expression may be associated with energy expenditure and insulin sensitivity. Diabetes. 59:829–835 10.2337/db09-1191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schug T.T., Li X. 2011. Sirtuin 1 in lipid metabolism and obesity. Ann. Med. 43:198–211 10.3109/07853890.2010.547211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timmers S., Konings E., Bilet L., Houtkooper R.H., van de Weijer T., Goossens G.H., Hoeks J., van der Krieken S., Ryu D., Kersten S., et al. 2011. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 14:612–622 10.1016/j.cmet.2011.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timmers S., Auwerx J., Schrauwen P. 2012. The journey of resveratrol from yeast to human. Aging (Albany NY). 4:146–158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tissenbaum H.A., Guarente L. 2001. Increased dosage of a sir-2 gene extends lifespan in Caenorhabditis elegans. Nature. 410:227–230 10.1038/35065638 [DOI] [PubMed] [Google Scholar]
- Tunaru S., Kero J., Schaub A., Wufka C., Blaukat A., Pfeffer K., Offermanns S. 2003. PUMA-G and HM74 are receptors for nicotinic acid and mediate its anti-lipolytic effect. Nat. Med. 9:352–355 10.1038/nm824 [DOI] [PubMed] [Google Scholar]
- van der Horst A., Burgering B.M. 2007. Stressing the role of FoxO proteins in lifespan and disease. Nat. Rev. Mol. Cell Biol. 8:440–450 10.1038/nrm2190 [DOI] [PubMed] [Google Scholar]
- Viscomi C., Bottani E., Civiletto G., Cerutti R., Moggio M., Fagiolari G., Schon E.A., Lamperti C., Zeviani M. 2011. In vivo correction of COX deficiency by activation of the AMPK/PGC-1α axis. Cell Metab. 14:80–90 10.1016/j.cmet.2011.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wise A., Foord S.M., Fraser N.J., Barnes A.A., Elshourbagy N., Eilert M., Ignar D.M., Murdock P.R., Steplewski K., Green A., et al. 2003. Molecular identification of high and low affinity receptors for nicotinic acid. J. Biol. Chem. 278:9869–9874 10.1074/jbc.M210695200 [DOI] [PubMed] [Google Scholar]
- Yoshino J., Mills K.F., Yoon M.J., Imai S. 2011. Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 14:528–536 10.1016/j.cmet.2011.08.014 [DOI] [PMC free article] [PubMed] [Google Scholar]