Abstract
Androgens initiate a complex network of signals within the UGS that trigger prostate lineage commitment and bud formation. Given its contributions to organogenesis in other systems, we investigated a role for canonical Wnt signaling in prostate development. We developed a new method to achieve complete deletion of beta-catenin, the transcriptional coactivator required for canonical Wnt signaling, in early prostate development. Beta-catenin deletion abrogated canonical Wnt signaling and yielded prostate rudiments that exhibited dramatically decreased budding and failed to adopt prostatic identity. This requirement for canonical Wnt signaling was limited to a brief critical period during the initial molecular phase of prostate identity specification. Deletion of beta-catenin in the adult prostate did not significantly affect organ homeostasis. Collectively, these data establish that beta-catenin and Wnt signaling play key roles in prostate lineage specification and bud outgrowth.
Keywords: Wnt signaling, Prostate development, Urogenital sinus, Mouse
Introduction
The adult rodent prostate is an exocrine gland divided into distinct lobes, with each lobe consisting of branching ducts and blind ending tubules. In the embryo, this complex structure originates from a simple cone-shaped structure called the urogenital sinus (UGS), which is present in both males and females (Sugimura et al., 1986). Between embryonic days 14.5 and 17.5 (E14.5 to E17.5) in males, rising levels of testicular androgens act directly on urogenital sinus mesenchyme to initiate prostate development (Cunha, 1973). In particular, androgens induce a variety of mesenchymal signals which instruct the adjacent urogenital sinus epithelium (UGE) to adopt prostatic identity, invade the surrounding mesenchyme, and form solid tubular buds. Although androgens play a crucial role in initiating prostate organogenesis from the mesenchymal compartment, the subsequent signaling events driving lineage commitment and branching morphogenesis have not been fully defined.
Prostate bud formation at embryonic day E17.5 represents the first morphologic evidence of prostate development, but molecular evidence of prostate differentiation is present shortly after circulating androgen levels begin to rise. The molecular phase of prostate development is defined by the earliest known marker of prostatic differentiation, the homeobox gene Nkx3.1. Nkx3.1 expression is first detectable in the UGS around E16, when androgen is present but prostate buds have yet to form (Bhatia-Gaur et al., 1999; Bieberich et al., 1996). Although Nkx3.1 expression is a reliable marker of prostatic differentiation, it is not required for prostate formation (Bhatia-Gaur et al., 1999; Kim et al., 2002). This suggests other signaling pathways are responsible for prostate lineage commitment. Our previous work, complemented by several other studies, have identified Wnt pathway related genes as significantly induced by androgens at the initiation of prostatic differentiation (Blum et al., 2010; Mehta et al., 2011; Schaeffer et al., 2008; Zhang et al., 2006). Moreover, we have shown a link between the signaling pathways active during early lineage commitment and pathways aberrantly activated in prostate pathology. Clearly defining the role for Wnt signaling in early prostate development will provide a foundation for future studies in prostate development, and may provide insights into prostatic disease.
Wnt signaling is critical for cell fate determination in a variety of organs outside of the prostate. For example, canonical Wnt signaling is required for specification of distal epithelial cells in the mouse lung (Mucenski et al., 2003), and is required for the initiation of mammary development (Chu et al., 2004). During pancreas development, canonical Wnt signaling is required for exocrine lineage specification (Murtaugh et al., 2005; Wells et al., 2007). In the prostate, investigations into roles for canonical Wnt signaling in embryonic prostate development are limited. Constitutively active beta-catenin induces hyperplasia and prostatic intraepithelial neoplasia (mPIN) if expressed in the embryo (Yu et al., 2009). The non-canonical, beta-catenin independent, Wnt pathway has been shown to regulate bud positioning and outgrowth through examination of Wnt5a knockout mice (Allgeier et al., 2008; Huang et al., 2009). Postnatal manipulation of canonical Wnt signaling in organ culture of rat UGSs inhibits branching, increases proliferation, and alters differentiation of prostate epithelium (Wang et al., 2008). Investigations of Wnt function in adult cells have shown, in addition to oncogenic effects, that activation of canonical Wnt signaling leads to a change in cellular differentiation, either preventing maturation or inducing transdifferentiation into squamous epithelium (Bierie et al., 2003; Bruxvoort et al., 2007; Yu et al., 2009). Overexpression of Wif1, an antagonist of Wnt signaling, in recombinant tissue grafts prevents transdifferentiation of adult bladder transitional epithelial cells to prostate epithelial cells (Li et al., 2009). Thus, multiple lines of evidence indicate a potential role for canonical Wnt signaling in prostate lineage commitment, but the precise role of this pathway in early prostate organogenesis is undefined.
Canonical Wnt signaling is activated when a Wnt ligand binds a low-density lipoprotein receptor-related protein (LRP) and one of the frizzled (Fzd) transmembrane receptor family members (Gordon and Nusse, 2006). These complex receptor-ligand interactions all funnel into a single transcriptional co-activator, beta-catenin, encoded by a single gene, Ctnnb1. Thus, deletion of Ctnnb1 can effectively silence canonical Wnt signaling. Beta-catenin levels are strictly regulated through an elegant series of phosphorylation and degradation events. In the absence of Wnt ligand, cytoplasmic beta-catenin is phosphorylated and is subsequently ubiquitinated and targeted for proteasomal degradation (Aberle et al., 1997; Behrens et al., 1998). Wnt signaling inhibits phosphorylation of beta-catenin, allowing it to accumulate in the cytoplasm and translocate to the nucleus where it associates with TCF/LEF family transcription factors and regulates target gene expression. One such target is Axin2, a negative feedback regulator of Wnt signaling and a useful marker of canonical pathway activity, both generally (Jho et al., 2002) and in the prostate (Ontiveros et al., 2008). Unlike other Wnt targets that can also be induced by other pathways, Axin2 transcription appears to be a specific indicator of canonical Wnt signaling (Yan et al., 2001).
Because Wnt signaling is broadly active in development, genetic modes to explore a role for canonical Wnt signaling in prostate lineage determination are challenging. Beta-catenin germline knockout mice show severe defects at gastrulation and die at E7, and thus are not useful for assessing a prostate phenotype at E16 and later (Haegel et al., 1995). Furthermore, existing systems for conditional deletion of genes in the prostate, such as Nkx3.1-Cre and Probasin-Cre, are expressed after prostate development has begun, so are unsuitable for studying lineage commitment (Jin et al., 2003; Lin et al., 2007). We therefore adapted a method used previously to study kidney development (Jokela and Vainio, 2007) and accomplished controlled, conditional deletion of beta-catenin in early prostate development (Ghosh et al., 2011). Tamoxifen-inducible Cre allows rapid and efficient deletion of target genes flanked by LoxP sites in vivo and in vitro. With this method, we demonstrate an absolute requirement for canonical Wnt signaling in lineage commitment and bud outgrowth.
Materials and Methods
Animals
All experimental procedures were approved by the Johns Hopkins Institutional Animal Care and Use Committee (IACUC). All mouse lines were maintained on a C57BL/6J background. Wildtype C57BL/6J, Ctnnb1fl/fl, NOD.SCID, and Cre-ERT1 mice (Tamoxifen-inducible Cre inserted into Rosa26 locus) were obtained from Jackson Laboratories (Bar Harbor, ME, Stocks 664, 4152, 1303, and 4847). Probasin-Cre (Pb-Cre4) mice were obtained from NCI Mouse Repository (Frederick, MD). Nkx3.1-Cre mice were a gift from Michael Shen (Columbia University, New York, NY). For tamoxifen mediated deletion of beta-catenin, Cre-ERT1 mice were crossed with Ctnnb1fl/fl mice to generate breeder pairs homozygous for both mutant alleles. For in vivo deletion of beta-catenin, Pb-Cre4 hemizygous, Ctnnb1fl/fl males were crossed with Ctnnb1fl/fl females. Nkx3.1-Cre heterozygous, Ctnnb1fl/+ males were crossed with Ctnnb1fl/fl females to generate Nkx3.1-Cre heterozygous, Ctnnb1fl/fl embryos. Genotyping was performed using primer sets and protocols recommended by the source of each strain. DNA for PCR was isolated from tails (adult mice) or liver (embryos).
UGS organ culture and subcapsular renal grafts
Timed pregnant female mice were obtained from overnight mating, with the following day considered E0.5. The UGS was harvested from embryos at the specified ages (E15.5–E17.5) and sex was determined by gonadal inspection. UGSs were harvested into ice-cold serum free UGS media [DMEM-F12 (1:1) media (Invitrogen), supplemented with nonessential amino acids (Cellgro), ITS liquid media supplement (Sigma), Penicillin/Streptomycin (Invitrogen), 1 g/L D-glucose (Sigma), L-glutamine (Invitrogen), and 1×10−8M dihydrotestosterone (Sigma-Aldrich)]. After harvest, UGSs were submerged in cold UGS media for 1 hour containing (Z)-4-hydroxytamoxifen (referred to in text as tamoxifen, Sigma-Aldrich), PKF118-310, or vehicle (ethanol and DMSO, respectively). Following treatment, the UGSs were cultured on 0.4 micron membranes (Millipore), oriented with the ventral surface up, overlying UGS media containing the indicated drugs or vehicle for 2–5 days. Media was changed every 48 hours. In all experiments, a minimum of 5 UGSs were treated per condition. For subcapsular renal grafts, UGSs and prostates were similarly harvested and cultured for 48 hours in the presence of tamoxifen or vehicle, then grafted under the kidney capsule of adult male NOD.SCID host mice and harvested after 4 weeks.
Histology, Immunohistochemistry (IHC), and Immunofluorescence
Tissues were fixed in formalin, routinely processed, embedded, and sectioned. For immunohistochemistry, the sections were deparaffinized and rehydrated before steaming in Target Retrieval Solution (Dako) for 40 minutes. For Nkx3.1 staining, slides were steamed for 40 minutes in EDTA pH 9. Endogenous peroxidases were quenched by peroxide treatment, and the slides were blocked for one hour with Serum Free Protein Block (Dako). Slides were incubated with antibodies directed against beta-catenin (BD Biosciences, #14), E-cadherin (Cell Signaling, #4065), AR (Santa Cruz, N-20), FoxA2 (Cell Signaling, D56D6), Nkx3.1 (Chen and Bieberich, 2005), or Hoxb13 (McMullin et al., 2009), and visualized with ImmPRESS Polymer detection kit (Vector Labs). For two color immunohistochemistry, the procedure was repeated and visualized with Vector VIP Peroxidase Substrate. For immunofluorescence, Alexafluor conjugated secondary antibodies (Invitrogen) were applied after incubation with the primary antibody.
Real-time reverse-transcription PCR
To separate UGE and UGM, the UGSs were incubated in 1% Trypsin for 75 minutes and tissue compartments were separated by fine dissection as previously described (Staack et al., 2003). Total RNA was isolated using RNeasy Mini-kit (Qiagen) and first strand cDNA was synthesized using Ready-To-Go You-Prime First-Strand Beads (GE Healthcare) according to manufacturer’s directions. Quantitative real-time PCR was performed using Fast SYBR Green Master Mix (Applied Biosystems) with oligonucleotides specific for Hprt, Ctnnb1, Axin2, Nkx3.1, Ar, Cdh1, Fgf10, Tcf7, Foxa1, and Foxa2 (sequences listed in Supplementary Table S1). Individual expression values were normalized by comparison to Hprt, and relative levels of mRNA expression were calculated using the delta-delta Ct method (Livak and Schmittgen, 2001).
Immunoblotting
UGSs were lysed in NuPAGE LDS sample buffer and were fractionated on NuPAGE gels (Invitrogen). After transfer to PVDF membranes, the samples were blocked and incubated with antibodies directed against beta-catenin (BD Biosciences, #14) and GAPDH (Santa Cruz, Clone 6C5). Blots were developed using Odyssey IRDye (LI-COR Biosciences).
Results
Temporal and spatial pattern of Wnt signaling in the UGS
Multiple components of the Wnt pathway are induced by androgens in a temporal and spatial pattern that suggests a role for Wnt signaling in early prostate development (Blum et al., 2010; Mehta et al., 2011; Schaeffer et al., 2008; Zhang et al., 2006). To understand the relationship between Wnt signaling and prostate lineage specification, we measured the relative expression of Axin2 and Nkx3.1 mRNA. UGSs were harvested at E16.5 from male and female embryos and mRNA was isolated from enzymatically separated UGM and UGE. At this time point of prostate lineage specification, when molecular prostatic differentiation subsequent to androgen exposure is beginning but morphologic changes have not begun, Axin2 transcript expression was significantly higher in males than in females. Furthermore, differential expression was limited to the UGE (Fig. 1A), suggesting a role for Wnt in specifying the prostate epithelial lineage. To refine the timing of these events, we cultured androgen-naive E15.5 UGSs with dihydrotestosterone (DHT), a potent androgen capable of inducing prostatic differentiation in vitro (Lasnitzki and Mizuno, 1977), and harvested UGE after 24 and 48 hours of androgen exposure. Compared to controls harvested before culture, UGE showed significantly increased Axin2 transcript expression at both time points after androgen exposure, coincident with increased Nkx3.1 expression (Fig. 1B). Together, these observations confirm that canonical Wnt/beta-catenin signaling is active in UGE during the earliest phases of prostate lineage specification, including time points before the emergence of prostate buds.
Figure 1. Androgen induces Wnt signaling in UGE during prostate specification.
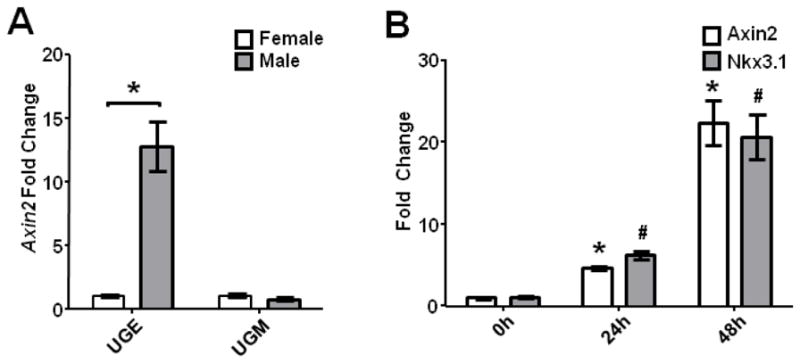
(A) qRT-PCR of male and female E16.5 separated UGE and UGM for Axin2 expression normalized to respective female samples. Differential expression of Axin2 was limited to the UGE (p<0.01). (B) E15.5 female UGSs cultured in vitro with DHT demonstrated a significant increase in Axin2 expression at 24 and 48 hours (p<0.01) by qRT-PCR of separated UGE, coinciding with significant increases in Nkx3.1 expression (p<0.01). Graphs represent mean ± standard error of the mean (SEM), p-values calculated using unpaired two-tailed Student’s t-test with unequal variance.
Conditional deletion of beta-catenin in the UGS
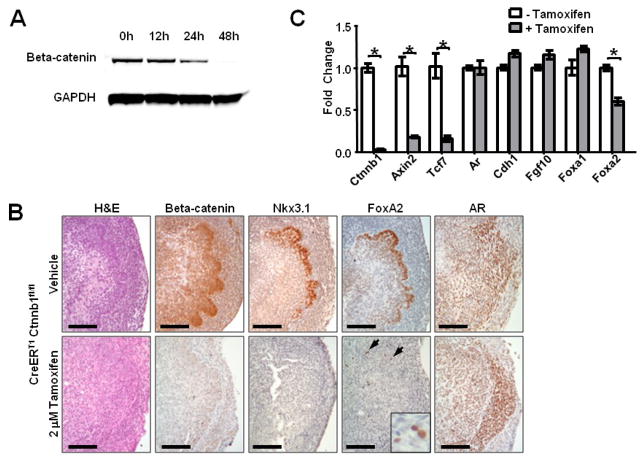
Because germline deletion of beta-catenin is embryonic lethal, we used a conditional Cre-LoxP system to delete Ctnnb1, the gene encoding beta-catenin. E15.5 UGSs from embryos homozygous for tamoxifen-inducible Cre (CreERT1) and floxed beta-catenin (Ctnnb1fl/fl) were cultured with 2 M tamoxifen or vehicle for 48 hours, and immunoblots were used to determine the time course and efficiency of gene deletion (Badea et al., 2003; Brault et al., 2001). Beta-catenin protein levels began to decrease within 24 hours of tamoxifen exposure and were nearly undetectable by the 48 hour time point (Fig. 2A). As expected, canonical Wnt signaling was dramatically suppressed after beta-catenin deletion, indicated by marked reduction in Axin2 and Tcf7 mRNA extracted from whole UGS cultures 48 hours after tamoxifen exposure (Fig. 2C). Several other genes known to influence prostate development were unaffected by beta-catenin loss, including the beta-catenin binding partner E-cadherin (Cdh1) and known mediators of prostate development, such as androgen receptor (Ar), Fgf10, and Foxa1 (Fig. 2C). In contrast, expression of the transcription factor Foxa2, which has been shown to be induced by activated beta-catenin in the prostate (Yu et al., 2011; Yu et al., 2009), was suppressed in the gene-deleted UGS compared to controls. Immunohistochemistry (IHC) confirmed uniform deletion of beta-catenin throughout the UGS and a marked decrease in FoxA2 expression (Fig. 2B). In the control group, FoxA2 expression closely matched Nkx3.1 expression at the UGE/UGM border and in the nascent buds. After beta-catenin deletion, Nkx3.1 was undetectable, and FoxA2 expression was limited to scattered individual cells. Androgen receptor, which is expressed throughout the UGS in both sexes before prostate development, continued to be expressed in both the UGE and UGM of beta-catenin deleted UGSs (Fig. 2B).
Figure 2. Inducible knockout of beta-catenin is rapid and results in decreased Foxa2 expression.
(A) Immunoblot analysis of E15.5 CreERT1 Ctnnbfl/fl UGSs grown in vitro with 2 M tamoxifen showed decreased beta-catenin protein level after 24 hours and loss within 48 hours. (B) H&E stain and IHC confirmation of beta-catenin deletion after 48 hours tamoxifen treatment (scale bar 100 micrometers). Nkx3.1 expression is undetectable, and FoxA2 expression is limited to scattered individual cells (arrows and inset). AR expression was similar between the two groups. Scale bars are 100 micrometers. (C) qRT-PCR analysis of whole UGSs after 48 hour tamoxifen treatment demonstrates decreased beta-catenin (Ctnnb1), Axin2, Tcf7, and Foxa2 expression relative to vehicle treated controls (p<0.01). Graph represents mean ± standard error of the mean (SEM), p-values calculated using unpaired two-tailed Student’s t-test with unequal variance.
Loss of Wnt signaling prevents bud formation and prostate determination
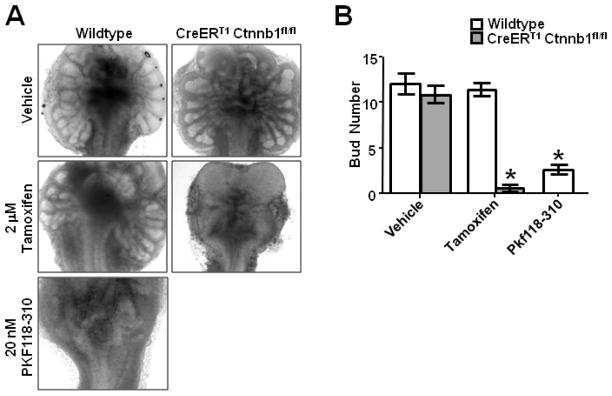
In a well established in vitro organ culture system (Doles et al., 2005), wild type controls showed robust bud development, whereas tamoxifen-treated beta-catenin null UGSs showed little or no bud formation (Fig. 3A, B). To confirm that decreased budding was due to loss of canonical Wnt signaling and not a peripheral role of beta-catenin, we treated UGSs with the small molecule Wnt inhibitor PKF118-310. This drug targets the beta-catenin/TCF complex to disrupt Wnt signaling (Lepourcelet et al., 2004). We observed a similar phenotype with both genetic and pharmacologic methods of Wnt inhibition, with significant decreases in bud number in both beta-catenin deleted and PKF118-310 treated UGSs compared to controls (Fig. 3A, B). Treatment of wildtype UGSs in organ culture with neither 2 M tamoxifen, the dose used for conditional deletion of beta-catenin, nor a log fold higher dose had any discernable effect on bud formation or growth (Fig. 3A, S1), indicating the observed effect is not from tamoxifen exposure, but loss of beta-catenin.
Figure 3. Deletion of beta-catenin prevents prostate differentiation and bud formation in vitro.
(A) Wildtype (left column) and CreERT1 Ctnnb1fl/fl (right column) E15.5 UGSs cultured in vitro for five days with 2 M tamoxifen, vehicle, or 20nM PKF118-310. (B) Quantitative analysis of bud formation in vitro demonstrates significantly reduced bud formation in beta-catenin deleted and inhibitor treated tissues compared to controls (n=5, p<0.01), but no difference in tamoxifen-treated versus vehicle treated wildtype UGSs (n=5, p=0.663). Graph represents mean ± standard error of the mean (SEM), p-values calculated using unpaired two-tailed Student’s t-test with unequal variance.
Several steps are required for prostate bud formation, most notably lineage commitment, followed by bud initiation and bud elongation (Bhatia-Gaur et al., 1999). We investigated the acquisition of prostate identity using immunohistochemistry for the prostate epithelial lineage marker Nkx3.1. Expression of Nkx3.1 by immunohistochemistry was similar in control UGSs grown in vitro or in vivo, but beta-catenin deleted UGSs failed to express Nkx3.1, suggesting a failure of prostate lineage commitment (Fig. 2B).
Beta-catenin deletion prevents prostate development
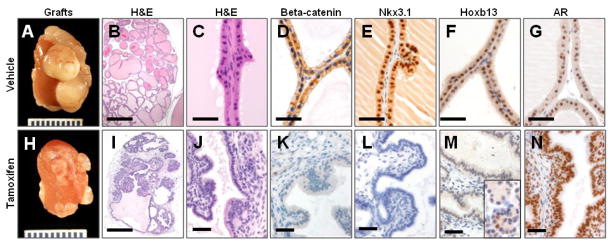
Growth of UGSs as subcapsular renal grafts mirrors normal development in vivo, allowing assessment of long term prostate growth and maturation (Doles et al., 2005). After four weeks of growth in a host mouse, tamoxifen-treated grafts were significantly smaller than vehicle-treated controls (Fig 4A, H, S2). Histology and immunohistochemical analysis showed the control grafts to be composed primarily of mature prostate ducts (Fig 4B–G). The prostatic epithelium in these grafts expressed normal levels of beta-catenin, Nkx3.1, Hoxb13, and AR (Fig 4D–G). The grafts from beta-catenin deleted UGSs were examined by two experienced pathologists (BWS, DMB) for histologic evidence of prostatic differentiation. The grafts were found to be disorganized, and did not contain histologically recognizable seminal vesicle or prostatic structures (Fig 4I, J, S3). Immunohistochemistry showed a complete absence of beta-catenin and Nkx3.1 expression in the tamoxifen-treated and grafted UGSs (Fig. 4K, L), confirming a lack of prostate differentiation. Although Nkx3.1 is a robust marker of prostate lineage, its expression is regulated by androgens (Sciavolino et al., 1997). An androgen-independent marker of prostate identity can be found in Hoxb13. Although it is more widely expressed in the developing embryo, expression in adult tissues is limited to colon and prostate (Sreenath et al., 1999). All grafted tissues remained AR positive (Fig. 4N), but Hoxb13 expression was limited to scattered cells within areas of periurethral gland differentiation (Fig. 4M). The lack of significant prostate differentiation and failure of bud formation confirmed that canonical Wnt/beta-catenin signaling is required for prostate lineage specification.
Figure 4. Beta-catenin knockout grafts do not show prostatic differentiation.
Renal grafts from E15.5 CreERT1 Ctnnb1fl/fl UGSs grown after 48 hour in vitro culture with vehicle (A–G) or tamoxifen (H–N) before 4 weeks growth as subcapsular renal grafts. Vehicle treated grafts show normal prostate gland formation (B, C), and express beta-catenin (D), Nkx3.1 (E), Hoxb13 (F), and AR (G). Beta-catenin deletion resulted in smaller grafts compared to controls (A, H), and H&E staining demonstrates disorganized grafts without recognizable prostatic differentiation (I, J). IHC analysis showed loss of beta-catenin (K), and Nkx3.1 (L). Hoxb13 expression was absent in the majority of tissue (M), with expression limited to small foci of cells with periurethral gland differentiation (inset). Grafted tissue retains AR expression (N). Scale bars 500 micrometers in B, I. All others 50 micrometers.
After lineage commitment, prostate development can occur without beta-catenin
To determine if canonical Wnt signaling is continuously required for prostate lineage commitment or if it is only required during prostatic differentiation, we utilized methods to delete beta-catenin at time points after lineage commitment and bud initiation. The Nkx3.1-Cre mouse has been indispensible for studies of prostatic development and differentiation, but reporter-based studies of Cre activity show expression after E17.0 (Lin et al., 2007), and expression of floxed target genes may not be eliminated until after E18.5 (Thomsen et al., 2008). We took advantage of this Cre expression timeline and compared deletion with our inducible system, giving loss of beta-catenin within 24–48 hours, to Nkx3.1-Cre Ctnnb1fl/fl tissues, predicted to show complete deletion at a later time point. The Nkx3.1-Cre cross was embryonic lethal, possibly due to expression in the developing sclerotome (Stanfel et al., 2006; Thomsen et al., 2008), but we were able to harvest E15.5 UGSs and culture them in vitro. In contrast to the inducible system, these UGSs were able to show evidence of prostatic differentiation by forming buds which lacked beta-catenin, but expressed Nkx3.1 (Fig. 5).
Figure 5. Beta-catenin deletion after differentiation does not prevent bud formation.
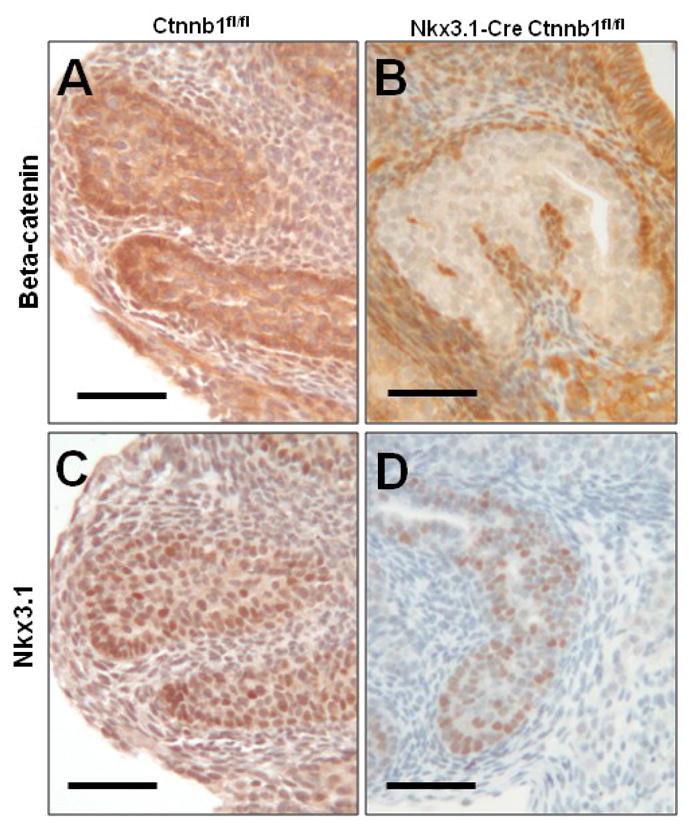
Organ cultures of E15.5 Nkx3.1-Cre Ctnnb1fl/+ and Ctnnb1fl/fl UGSs show normal beta-catenin (A) and Nkx3.1 (C) expression in control UGS prostate buds after 5 days in controls. Nkx3.1-Cre Ctnnb1fl/fl mice form (B) beta-catenin deficient prostate buds that (D) maintain Nkx3.1 expression. Scale bars are 50 micrometers.
To further assess the timeline of beta-catenin dependent prostate differentiation, we allowed prostate development to proceed in vivo until birth, and harvested prostates from pups at postnatal day zero (P0), approximately five days after prostate development begins. These prostates were treated with vehicle or tamoxifen in vitro for two days to delete beta-catenin, and grown for four weeks as subcapsular renal grafts. When harvested, control grafts contained mature prostate tissue (Fig. 6 A–D). Tamoxifen-treated grafts showed scattered areas of prostatic differentiation and Nkx3.1 expression despite complete beta-catenin deletion (Fig. 6E–H). In addition to Nkx3.1, these grafts expressed basal and luminal markers (CK14, p63, CK8) in the expected pattern (Fig. S5). However, the SMA positive stroma was frequently discontinuous around the beta-catenin deleted glands.
Figure 6. After androgen action, prostate glands can form without beta-catenin.
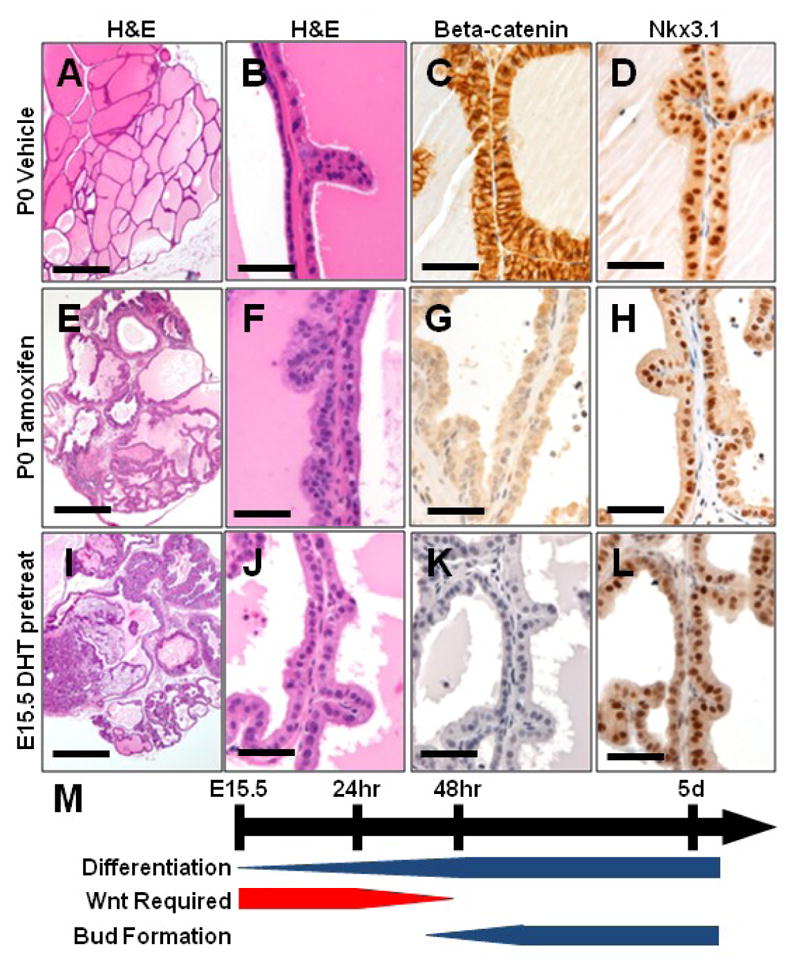
Grafts from P0 CreERT1 Ctnnb1fl/fl prostates treated in vitro with vehicle (A–D) or tamoxifen (E–H) for 48 hours show normal development in controls (A–D). Tamoxifen treated grafts are smaller than controls, but show prostate gland formation (E,F) despite loss of beta-catenin (G), and retain Nkx3.1 expression (H). Grafts from E15.5 female UGSs pretreated with DHT for 24 hours before exposure to tamoxifen (I–L) show a small number of prostate glands (I, J) forming without beta-catenin (K) that retain Nkx3.1 expression (L). (M) Timeline depicting normal prostatic differentiation, bud formation, and the transient requirement for Wnt signaling on which these processes rely. Scale bars 500 micrometers in A, E, I. All others 50 micrometers.
In order to further shorten the window of active beta-catenin signaling, we harvested E15.5 female UGSs for culture with DHT for 24 hours to initiate prostatic differentiation before deleting beta-catenin with tamoxifen. Grown in vitro, UGSs harvested from male or female embryos at E15.5 show an absence of budding after beta-catenin deletion (Fig. S4). However, pretreatment of these grafts with DHT for 24 hours before tamoxifen treatment, or physiologic in vivo exposure to DHT in male embryos harvested at E16.5, resulted in rudimentary bud formation despite beta-catenin deletion (Fig. S4). Grafts from these E15.5 UGSs pretreated with DHT before tamoxifen treatment contained occasional mature prostate glands which maintained Nkx3.1 expression despite the absence of beta-catenin (Fig 6I–L). Thus, these results indicate a window between E14.5–E16.5 where prostatic differentiation requires canonical Wnt/beta-catenin signaling (Fig. 6M).
Loss of beta-catenin in luminal adult epithelial cells does not affect glandular homeostasis
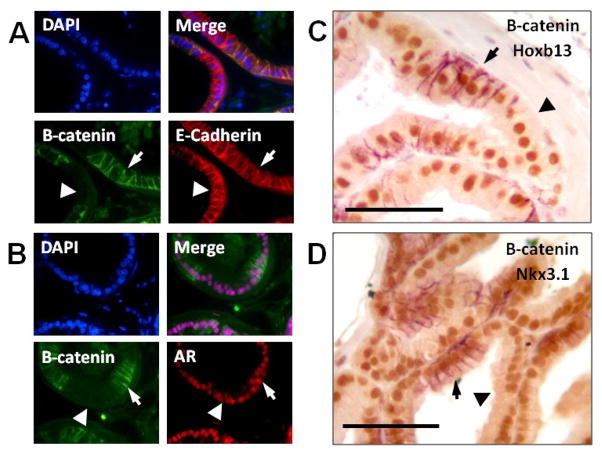
In addition to roles in canonical Wnt signaling, beta-catenin participates in adherens junctions by binding E-cadherin and may play a role in androgen signaling through interactions with AR (Aberle et al., 1996; Truica et al., 2000). We sought to exclude the possibility that loss of beta-catenin would interfere with E-cadherin localization or androgen signaling. To accomplish this, we examined the prostates from 12 week old Probasin-Cre (Pb-Cre4) Ctnnb1fl/fl mice. Probasin-driven Cre expression is initiated postnatally, is active in the luminal epithelial cells, and should have no effect on prostate determination and initial branching. Because Probasin-Cre shows mosaic expression in the prostate, we were able to compare beta-catenin deleted and replete cells in the same gland. Both cell types showed normal histology and expression of E-cadherin and AR. Expression of the androgen-independent differentiation marker Hoxb13 was unchanged, as was the androgen-dependent Nkx3.1 (Fig. 7A–D). These data indicate that loss of beta-catenin in adult prostate cells does not significantly affect homeostasis and suggest that androgen signaling and cellular adhesion sufficient to maintain mature prostate glands can occur in the absence of beta-catenin.
Figure 7. Mature secretory luminal cells maintain prostatic identity after beta-catenin deletion.
Pb-Cre4 Ctnnb1fl/fl mice show mosaic deletion of beta-catenin in luminal prostate epithelial cells. In areas with beta-catenin expression (arrow) and in areas of beta-catenin deletion (arrowhead) epithelial cells retain normal expression and localization of E-cadherin (A) and Androgen receptor (B) by immunofluorescence. (C,D) Two color immunohistochemistry shows mosaic beta-catenin expression (purple) and normal expression of Hoxb13 (C), and Nkx3.1 (D) in brown. Scale bars are 50 micrometers.
Discussion
A variety of Wnt pathway members have been shown to be expressed in the developing prostate in an androgen dependent fashion. However, the role of canonical Wnt signaling in early prostate development was not previously addressed. Here, we show that prostate lineage specification depends on canonical Wnt signaling through beta-catenin, and that this signal is required at the earliest stages of prostate lineage commitment. Deletion of beta-catenin or pharmacologic inhibition of canonical Wnt signaling blocks prostatic differentiation, as illustrated by a failure to initiate bud formation or form recognizable glandular tissue, and by undetectable levels of Nkx3.1 and greatly reduced Hoxb13 expression. Previous reports indicate a role for beta-catenin in epithelial differentiation and cell fate determination in other organs (Chu et al., 2004; Mucenski et al., 2003; Murtaugh et al., 2005; Wells et al., 2007). Our findings are consistent with these reports, and suggest that Wnt mediated cell-fate determination may be a widely conserved feature of branching morphogenesis.
Investigations into the genetic programs which control prostate morphogenesis are limited by available strategies for conditional mutagenesis. Germline knockouts and other mutant mice have proven useful for studying some pathways, but certain mutations result in early embryonic death or prevent normal formation of the UGS. Currently available models for conditional mutagenesis, such as Cre-LoxP systems, are useful for studies of late embryonic prostate morphogenesis and adult homeostasis, but are activated too late in development to study the initial stages of prostate differentiation. We show that conditional deletion of genes in the developing prostate can be accomplished rapidly and efficiently with complete temporal control.
In addition to its role in canonical Wnt signaling, beta-catenin functions in cell-cell adhesion though participation in the adherens junction. In the prostate, loss of beta-catenin did not effect on E-cadherin expression or localization, and previous reports show no interruption of adherens junctions and cell-cell adhesion after loss of beta-catenin (Bridgewater et al., 2008). A pharmacologic inhibitor of Wnt signaling produced similar results to genetic deletion. Since the inhibitor affects transcriptional activity of beta-catenin through disruption of its association with Tcf/LEF family members, it should not affect adherens functions of beta-catenin. This result further supports the requirement for canonical Wnt signaling in prostate specification.
Previously demonstrated roles of Wnt signaling in the prostate include postnatal regulation of branching and stem cell maintenance (Ontiveros et al., 2008; Wang et al., 2008). Our findings do not contradict these reports, as we focused on the role of canonical Wnt signaling in earlier prenatal events. Prostate tissue forming from beta-catenin deleted tissue was smaller than controls, but because we used in vitro organ culture and subcapsular renal grafts, we were unable to enumerate branch points. We cannot make determinations about the role of Wnt signaling in prostatic stem cell maintenance because many of these phenotypes are evident only after castration and regeneration cycling of adult mice. Just as we have shown a short window of absolute requirement for canonical Wnt signaling in early prostate development, it is reasonable to hypothesize that the function of the Wnt signaling pathway can change during different phases of prostate maturation.
Prostate development is controlled by androgens, but estrogen and estrogenic endocrine disruptors have shown a variety of effects on prostate growth and branching prostate (Prins et al., 2001). By using an inducible system for gene deletion that requires the presence of tamoxifen, a partial agonist of the estrogen receptor in mice, we were compelled to carefully control for potential effects of tamoxifen on prostate lineage specification and branching. Fortunately, no observable effects of tamoxifen on UGS organ culture were noted, even at one log higher concentration than those necessary to induce Cre recombination.
Androgen induced signals from the UGM to the UGE initiate prostate development (Cunha, 1973). So called “andromedins” are postulated to relay this signal and induce prostatic differentiation. A number of Wnt family members are expressed in the UGS in a manner consistent with a role in epithelial-mesenchymal interactions during prostate induction, and could function as andromedins (Mehta et al., 2011; Schaeffer et al., 2008; Zhang et al., 2006). Here we show that canonical Wnt signaling in the epithelium is induced after androgen exposure. Although we have determined that canonical Wnt signaling is required for prostate specification, the source and identity of the initiating Wnt ligand has yet to be determined. Because this inducible system deletes beta-catenin in both the epithelium and mesenchyme, we could not determine which compartment requires Wnt signaling. Although Nkx3.1-Cre expression is specific to the epithelium, the ability of prostatic differentiation to occur after Nkx3.1-Cre mediated deletion of beta-catenin suggests that either the Wnt requirement is mesenchymal, or, as we hypothesize, that the requirement is epithelial, but transient, and abates prior to Nkx3.1-Cre expression. While it remains unclear whether UGE or UGM initiates Wnt signaling, we detect androgen induced Wnt signaling exclusively in the UGE.
An additional aspect of this pathway which remains undetermined is the precise role of Wnt/beta-catenin signaling in prostate lineage commitment, and the downstream mediators that carry out this role. In our study, the absence of identifiable prostatic structures in beta-catenin null tissue indicates an absolute requirement for this protein in prostatic induction. However, it is unclear if Wnt signaling is directly inducing prostatic lineage committment, or if it plays another, more permissive role, such as promoting survival or proliferation of committed prostate progenitors. A key to determining the function of Wnt signaling will be determining the downstream targets which are subsequently induced. Previous studies have indicated that Foxa2 is positively regulated by activated Wnt signaling (Yu et al., 2011; Yu et al., 2009). Here we show the complement, that Foxa2 expression is diminished after beta-catenin deletion. The timing and localization of Foxa2 during prostate development, expression only during early morphogenesis, suggest it may play a role in the earliest stages of prostate lineage commitment. Our data confirm that Foxa2 is regulated by Wnt signaling, suggesting a potential pathway which could mediate the transient requirement for Wnt signaling in prostate development.
In summary, we show a critical requirement for canonical Wnt/beta-catenin signaling in early prostate lineage commitment. To accomplish this, we adapted a strategy for gene deletion to use in studies of prostatic differentiation and development that allows careful interrogation of gene function at the earliest stages of prostate development. Future studies should focus on the identity and source of the signaling molecules that initiate Wnt signaling in the UGE, and what downstream targets mediate this effect.
Supplementary Material
Highlights.
Canonical Wnt signaling is induced by androgen in urogenital sinus epithelium.
Abrogation of Wnt signaling prevents prostatic differentiation and bud formation.
Beta-catenin deletion in mature prostatic epithelium does not affect glandular homeostasis.
Acknowledgments
The authors thank Dr. Michael Shen, University of Columbia, for providing Nkx3.1-Cre mice. BWS was supported by NIH T32 RR007002 and DOD PC080778. LM was supported by P30CA006973. EMS was supported by NIH DK081019. EMS is an AUA Astellas Rising Star and HHMI Early Careers Physician Scientist.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Aberle H, Bauer A, Stappert J, Kispert A, Kemler R. beta-catenin is a target for the ubiquitin-proteasome pathway. EMBO J. 1997;16:3797–804. doi: 10.1093/emboj/16.13.3797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aberle H, Schwartz H, Kemler R. Cadherin-catenin complex: protein interactions and their implications for cadherin function. J Cell Biochem. 1996;61:514–23. doi: 10.1002/(SICI)1097-4644(19960616)61:4%3C514::AID-JCB4%3E3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- Allgeier SH, Lin T-M, Vezina CM, Moore RW, Fritz WA, Chiu S-Y, Zhang C, Peterson RE. WNT5A selectively inhibits mouse ventral prostate development. Developmental biology. 2008;324:10–17. doi: 10.1016/j.ydbio.2008.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badea TC, Wang Y, Nathans J. A noninvasive genetic/pharmacologic strategy for visualizing cell morphology and clonal relationships in the mouse. J Neurosci. 2003;23:2314–22. doi: 10.1523/JNEUROSCI.23-06-02314.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrens J, Jerchow BA, Wurtele M, Grimm J, Asbrand C, Wirtz R, Kuhl M, Wedlich D, Birchmeier W. Functional interaction of an axin homolog, conductin, with beta-catenin, APC, and GSK3beta. Science. 1998;280:596–9. doi: 10.1126/science.280.5363.596. [DOI] [PubMed] [Google Scholar]
- Bhatia-Gaur R, Donjacour AA, Sciavolino PJ, Kim M, Desai N, Young P, Norton CR, Gridley T, Cardiff RD, Cunha GR, Abate-Shen C, Shen MM. Roles for Nkx3.1 in prostate development and cancer. Genes Dev. 1999;13:966–77. doi: 10.1101/gad.13.8.966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bieberich CJ, Fujita K, He WW, Jay G. Prostate-specific and androgen-dependent expression of a novel homeobox gene. J Biol Chem. 1996;271:31779–82. doi: 10.1074/jbc.271.50.31779. [DOI] [PubMed] [Google Scholar]
- Bierie B, Nozawa M, Renou JP, Shillingford JM, Morgan F, Oka T, Taketo MM, Cardiff RD, Miyoshi K, Wagner KU, Robinson GW, Hennighausen L. Activation of beta-catenin in prostate epithelium induces hyperplasias and squamous transdifferentiation. Oncogene. 2003;22:3875–87. doi: 10.1038/sj.onc.1206426. [DOI] [PubMed] [Google Scholar]
- Blum R, Gupta R, Burger PE, Ontiveros CS, Salm SN, Xiong X, Kamb A, Wesche H, Marshall L, Cutler G, Wang X, Zavadil J, Moscatelli D, Wilson EL. Molecular signatures of the primitive prostate stem cell niche reveal novel mesenchymal-epithelial signaling pathways. PLoS ONE. 2010:5. doi: 10.1371/journal.pone.0013024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brault V, Moore R, Kutsch S, Ishibashi M, Rowitch DH, McMahon AP, Sommer L, Boussadia O, Kemler R. Inactivation of the beta-catenin gene by Wnt1-Cre-mediated deletion results in dramatic brain malformation and failure of craniofacial development. Development. 2001;128:1253–64. doi: 10.1242/dev.128.8.1253. [DOI] [PubMed] [Google Scholar]
- Bridgewater D, Cox B, Cain J, Lau A, Athaide V, Gill PS, Kuure S, Sainio K, Rosenblum ND. Canonical WNT/beta-catenin signaling is required for ureteric branching. Dev Biol. 2008;317:83–94. doi: 10.1016/j.ydbio.2008.02.010. [DOI] [PubMed] [Google Scholar]
- Bruxvoort KJ, Charbonneau HM, Giambernardi TA, Goolsby JC, Qian CN, Zylstra CR, Robinson DR, Roy-Burman P, Shaw AK, Buckner-Berghuis BD, Sigler RE, Resau JH, Sullivan R, Bushman W, Williams BO. Inactivation of Apc in the mouse prostate causes prostate carcinoma. Cancer Res. 2007;67:2490–6. doi: 10.1158/0008-5472.CAN-06-3028. [DOI] [PubMed] [Google Scholar]
- Chen H, Bieberich CJ. Structural and functional analysis of domains mediating interaction between NKX-3.1 and PDEF. J Cell Biochem. 2005;94:168–77. doi: 10.1002/jcb.20297. [DOI] [PubMed] [Google Scholar]
- Chu EY, Hens J, Andl T, Kairo A, Yamaguchi TP, Brisken C, Glick A, Wysolmerski JJ, Millar SE. Canonical WNT signaling promotes mammary placode development and is essential for initiation of mammary gland morphogenesis. Development. 2004;131:4819–29. doi: 10.1242/dev.01347. [DOI] [PubMed] [Google Scholar]
- Cunha GR. The role of androgens in the epithelio-mesenchymal interactions involved in prostatic morphogenesis in embryonic mice. Anat Rec. 1973;175:87–96. doi: 10.1002/ar.1091750108. [DOI] [PubMed] [Google Scholar]
- Doles JD, Vezina CM, Lipinski RJ, Peterson RE, Bushman W. Growth, morphogenesis, and differentiation during mouse prostate development in situ, in renal grafts, and in vitro. The Prostate. 2005;65:390. doi: 10.1002/pros.20321. [DOI] [PubMed] [Google Scholar]
- Ghosh S, Lau H, Simons BW, Powell JD, Meyers DJ, De Marzo AM, Berman DM, Lotan TL. PI3K/mTOR signaling regulates prostatic branching morphogenesis. Dev Biol. 2011;360:329–42. doi: 10.1016/j.ydbio.2011.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon MD, Nusse R. Wnt signaling: multiple pathways, multiple receptors, and multiple transcription factors. J Biol Chem. 2006;281:22429–33. doi: 10.1074/jbc.R600015200. [DOI] [PubMed] [Google Scholar]
- Haegel H, Larue L, Ohsugi M, Fedorov L, Herrenknecht K, Kemler R. Lack of beta-catenin affects mouse development at gastrulation. Development. 1995;121:3529–37. doi: 10.1242/dev.121.11.3529. [DOI] [PubMed] [Google Scholar]
- Huang L, Pu Y, Hu WY, Birch L, Luccio-Camelo D, Yamaguchi T, Prins GS. The role of Wnt5a in prostate gland development. Developmental biology. 2009;328:188–199. doi: 10.1016/j.ydbio.2009.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jho EH, Zhang T, Domon C, Joo CK, Freund JN, Costantini F. Wnt/beta-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol Cell Biol. 2002;22:1172–83. doi: 10.1128/MCB.22.4.1172-1183.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin C, McKeehan K, Wang F. Transgenic mouse with high Cre recombinase activity in all prostate lobes, seminal vesicle, and ductus deferens. Prostate. 2003;57:160–4. doi: 10.1002/pros.10283. [DOI] [PubMed] [Google Scholar]
- Jokela T, Vainio S. Conditional tamoxifen Cre induced mutagenesis in the embryonic kidney in organ culture. Genesis. 2007;45:757–61. doi: 10.1002/dvg.20352. [DOI] [PubMed] [Google Scholar]
- Kim MJ, Bhatia-Gaur R, Banach-Petrosky WA, Desai N, Wang Y, Hayward SW, Cunha GR, Cardiff RD, Shen MM, Abate-Shen C. Nkx3.1 mutant mice recapitulate early stages of prostate carcinogenesis. Cancer Res. 2002;62:2999–3004. [PubMed] [Google Scholar]
- Lasnitzki I, Mizuno T. Induction of the rat prostate gland by androgens in organ culture. J Endocrinol. 1977;74:47–55. doi: 10.1677/joe.0.0740047. [DOI] [PubMed] [Google Scholar]
- Lepourcelet M, Chen YN, France DS, Wang H, Crews P, Petersen F, Bruseo C, Wood AW, Shivdasani RA. Small-molecule antagonists of the oncogenic Tcf/beta-catenin protein complex. Cancer Cell. 2004;5:91–102. doi: 10.1016/s1535-6108(03)00334-9. [DOI] [PubMed] [Google Scholar]
- Li X, Wang Y, Sharif-Afshar A-R, Uwamariya C, Yi A, Ishii K, Hayward SW, Matusik RJ, Bhowmick NA. Urothelial transdifferentiation to prostate epithelia is mediated by paracrine TGF-[beta] signaling. Differentiation. 2009;77:95–102. doi: 10.1016/j.diff.2008.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin Y, Liu G, Zhang Y, Hu YP, Yu K, Lin C, McKeehan K, Xuan JW, Ornitz DM, Shen MM, Greenberg N, McKeehan WL, Wang F. Fibroblast growth factor receptor 2 tyrosine kinase is required for prostatic morphogenesis and the acquisition of strict androgen dependency for adult tissue homeostasis. Development. 2007;134:723–34. doi: 10.1242/dev.02765. [DOI] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods. 2001;25:402–8. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- McMullin RP, Mutton LN, Bieberich CJ. Hoxb13 regulatory elements mediate transgene expression during prostate organogenesis and carcinogenesis. Dev Dyn. 2009;238:664–72. doi: 10.1002/dvdy.21870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta V, Abler LL, Keil KP, Schmitz CT, Joshi PS, Vezina CM. Atlas of Wnt and R-spondin gene expression in the developing male mouse lower urogenital tract. Dev Dyn. 2011 doi: 10.1002/dvdy.22741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mucenski ML, Wert SE, Nation JM, Loudy DE, Huelsken J, Birchmeier W, Morrisey EE, Whitsett JA. beta-Catenin is required for specification of proximal/distal cell fate during lung morphogenesis. J Biol Chem. 2003;278:40231–8. doi: 10.1074/jbc.M305892200. [DOI] [PubMed] [Google Scholar]
- Murtaugh LC, Law AC, Dor Y, Melton DA. Beta-catenin is essential for pancreatic acinar but not islet development. Development. 2005;132:4663–74. doi: 10.1242/dev.02063. [DOI] [PubMed] [Google Scholar]
- Ontiveros CS, Salm SN, Wilson EL. Axin2 expression identifies progenitor cells in the murine prostate. The Prostate. 2008;68:1263–1272. doi: 10.1002/pros.20770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prins GS, Birch L, Habermann H, Chang WY, Tebeau C, Putz O, Bieberich C. Influence of neonatal estrogens on rat prostate development. Reprod Fertil Dev. 2001;13:241–52. doi: 10.1071/rd00107. [DOI] [PubMed] [Google Scholar]
- Schaeffer EM, Marchionni L, Huang Z, Simons B, Blackman A, Yu W, Parmigiani G, Berman DM. Androgen-induced programs for prostate epithelial growth and invasion arise in embryogenesis and are reactivated in cancer. Oncogene. 2008;27:7180–91. doi: 10.1038/onc.2008.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciavolino PJ, Abrams EW, Yang L, Austenberg LP, Shen MM, Abate-Shen C. Tissue-specific expression of murine Nkx3.1 in the male urogenital system. Dev Dyn. 1997;209:127–38. doi: 10.1002/(SICI)1097-0177(199705)209:1<127::AID-AJA12>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Sreenath T, Orosz A, Fujita K, Bieberich CJ. Androgen-independent expression of hoxb-13 in the mouse prostate. Prostate. 1999;41:203–7. doi: 10.1002/(sici)1097-0045(19991101)41:3<203::aid-pros8>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- Staack A, Donjacour AA, Brody J, Cunha GR, Carroll P. Mouse urogenital development: a practical approach. Differentiation; research in biological diversity. 2003;71:402. doi: 10.1046/j.1432-0436.2003.7107004.x. [DOI] [PubMed] [Google Scholar]
- Stanfel MN, Moses KA, Carson JA, Zimmer DB, DeMayo F, Schwartz RJ, Zimmer WE. Expression of an Nkx3.1-CRE gene using ROSA26 reporter mice. Genesis. 2006;44:550–5. doi: 10.1002/dvg.20250. [DOI] [PubMed] [Google Scholar]
- Sugimura Y, Cunha GR, Donjacour AA. Morphogenesis of ductal networks in the mouse prostate. Biol Reprod. 1986;34:961–71. doi: 10.1095/biolreprod34.5.961. [DOI] [PubMed] [Google Scholar]
- Thomsen MK, Butler CM, Shen MM, Swain A. Sox9 is required for prostate development. Dev Biol. 2008;316:302–11. doi: 10.1016/j.ydbio.2008.01.030. [DOI] [PubMed] [Google Scholar]
- Truica CI, Byers S, Gelmann EP. Beta-catenin affects androgen receptor transcriptional activity and ligand specificity. Cancer research. 2000;60:4709. [PubMed] [Google Scholar]
- Wang B-E, Wang X-D, Ernst JA, Polakis P, Gao W-Q. Regulation of Epithelial Branching Morphogenesis and Cancer Cell Growth of the Prostate by Wnt Signaling. PLoS ONE. 2008;3:e2186. doi: 10.1371/journal.pone.0002186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wells JM, Esni F, Boivin GP, Aronow BJ, Stuart W, Combs C, Sklenka A, Leach SD, Lowy AM. Wnt/beta-catenin signaling is required for development of the exocrine pancreas. BMC Dev Biol. 2007;7:4. doi: 10.1186/1471-213X-7-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan D, Wiesmann M, Rohan M, Chan V, Jefferson AB, Guo L, Sakamoto D, Caothien RH, Fuller JH, Reinhard C, Garcia PD, Randazzo FM, Escobedo J, Fantl WJ, Williams LT. Elevated expression of axin2 and hnkd mRNA provides evidence that Wnt/beta -catenin signaling is activated in human colon tumors. Proc Natl Acad Sci U S A. 2001;98:14973–8. doi: 10.1073/pnas.261574498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Wang Y, DeGraff DJ, Wills ML, Matusik RJ. Wnt/beta-catenin activation promotes prostate tumor progression in a mouse model. Oncogene. 2011;30:1868–79. doi: 10.1038/onc.2010.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Wang Y, Jiang M, Bierie B, Roy-Burman P, Shen MM, Taketo MM, Wills M, Matusik RJ. Activation of β-Catenin in mouse prostate causes HGPIN and continuous prostate growth after castration. The Prostate. 2009;69:249–262. doi: 10.1002/pros.20877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang TJ, Hoffman BG, Ruiz de Algara T, Helgason CD. SAGE reveals expression of Wnt signalling pathway members during mouse prostate development. Gene expression patterns: GEP. 2006;6:310. doi: 10.1016/j.modgep.2005.07.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.