Abstract
Telomere attrition, DNA damage and constitutive mitogenic signaling can all trigger cellular senescence in normal cells and serve as a defense against tumor progression. Cancer cells may circumvent this cellular defense by acquiring genetic mutations in checkpoint proteins responsible for regulating permanent cell cycle arrest. A small family of tumor suppressor genes encoding the retinoblastoma susceptibility protein family (Rb, p107, p130) exerts a partially redundant control of entry into S phase of DNA replication and cellular proliferation. Here we report that activation of the p53-dependent DNA damage response has been found to accelerate senescence in human prostate cancer cells lacking a functional Rb protein. This novel form of irradiation-induced premature cellular senescence reinforces the notion that other Rb family members may compensate for loss of Rb protein in the DNA damage response pathway. Consistent with this hypothesis, depletion of p107 potently inhibits the irradiation-induced senescence observed in DU145 cells. In contrast, p130 depletion triggers a robust and unexpected form of premature senescence in unirradiated cells. The dominant effect of depleting both p107 and p130, in the absence of Rb, was a complete blockade of irradiation-induced cellular senescence. Onset of the p107-dependent senescence was temporally associated with p53-mediated stabilization of the cyclin-dependent kinase inhibitor p27 and decreases in c-myc and cks1 expression. These results indicate that p107 is required for initiation of accelerated cellular senescence in the absence of Rb and introduces the concept that p130 may be required to prevent the onset of terminal growth arrest in unstimulated prostate cancer cells lacking a functional Rb allele.
Keywords: apoptosis, cell cycle arrest, cks1, DU145, c-myc, p16, p27, skp2
Introduction
Untransformed cells have a finite reproductive lifespan determined by telomere length, termed replicative senescence. This program eliminates aging cells that may potentially turn carcinogenic by permanently halting the cell cycle.1 In addition, DNA damage and oncogene activation can induce this terminal arrest, known as “accelerated” or “premature” senescence before telomere attrition reaches a critical length and induces replicative senescence. Both replicative and premature senescence pathways are activated in a similar fashion. However, multiple redundancies exist and can vary according to cell type and nature of stress. p53 is critical for initiating senescence in response to DNA double-strand breaks (DSBs) caused by ionizing radiation.2–4 The ataxia-telangiectasia mutated (ATM/ATR) kinases play a critical role upstream of p53 and serve to detect DSBs and initiate the DNA damage response signaling cascade that converges on p53 directly or indirectly through the Chk1/2 kinases. Permanent growth arrest in response to genotoxic stress relies on functional p53 and p16-retinoblastoma (Rb) pathways. Both the p53 and p16-Rb pathways depend on the cyclin-dependent kinase inhibitors (CDKIs) p16 and the downstream target of p53, p21WAF1/Cip1 (p21) to maintain Rb in an active, hypophosphorylated state. Active Rb can sequester E2F and repress target genes such as cyclin A and cyclin B, necessary for cell cycle progression. While p53 stabilization is crucial for senescence induction, the Rb protein serves to maintain permanent cellular arrest by establishing senescence-associated heterochromatin foci (SAHF), permanently silencing E2F target genes by establishing discrete regions of repressive chromatin.5
Products of the retinoblastoma (Rb) gene family comprises tumor suppressor pRb/p105 gene and related Rb-like 1/p107 and Rb-like 2/p130. These three factors share homologous structures and cooperatively interact to restrict G1 to S phase cell cycle progression. The Rb proteins also have distinct functions, as demonstrated by the binding of distinct E2F transcription factors.6,7 E2F promotes S phase entry by binding the promoter region of many genes required for S phase entry, such as DNA polymerase subunits and cyclins A and E. Rb family members are able to bind E2F and prevent it from interacting with the promoter of these critical E2F target genes.
Cancer cells often bypass the premature senescence that is induced by traditional chemotherapeutics and IR by accumulating mutations within the p53 and p16-Rb pathways. The p53 gene has a very important role in the biological properties of tumor cells, and is the most frequently mutated tumor suppressor gene in human cancers. Mutations of Rb are also common and the product of this gene clearly plays a major role in DNA damage induced premature senescence, however compensation by other Rb family members has not thoroughly been investigated.
The purpose of the present study was to investigate mechanisms of irradiation-induced premature senescence in a prostate cancer cell line lacking a functional Rb protein. We previously demonstrated that DU145 cells (p53−/−, Rb−/−) fail to undergo irradiation-induced senescence (Lehmann et al., 2007). However, re-expression of a WT p53 transgene was sufficient to re-establish senescence, even in the absence of a functional Rb protein. Thus, other members of the pocket protein family (p107/p130) may populate the downstream p53-dependent senescence pathway in at least some human prostate cancer (CaP) cells.
Results
DU145 cells lacking a functional Rb and stably expressing WT p53 are capable of irradiation-induced senescence
DU145 cells are an example of a radio-resistant prostate cancer cell line that fails to undergo irradiation-induced senescence and displays typical mutations that abrogate the function of two critical proteins that mediate senescence. These cells harbor two different missense mutations in each of the two p53 alleles (Phe223Leu and Val274Phe), both located in the core domain necessary to bind cognate promoter sites of target genes.8 Such mutations result in a stabilized p53 protein that is incapable of forming functional tetramers necessary for p53-dependent transcription accompanying various cellular stresses. As a result, DU145 cells fail to elicit a premature senescence response to the double strand breaks created by ionizing radiation. Therefore, to re-establish a functional p53-dependent pathway for inducing cellular senescence, DU145 cells were stably infected with a retroviral vector containing WT p53 (DU/WT p53).
DU145 cells also lack one Rb allele and the other contains a single base substitution within exon 21 that converts a lysine codon (AAG; codon 715) to an in-frame stop codon (TAG), resulting in a loss of 105 base pairs (35 amino acids).9 The Rb pocket is composed of two domains separated by a spacer region, A (379–572) and B (646–792) that functionally interact to form a repressor motif capable of binding E2F transcription factors.10 The DU145 Rb deletion lies in the heart of the B domain, producing a truncated protein incapable of binding E2F.
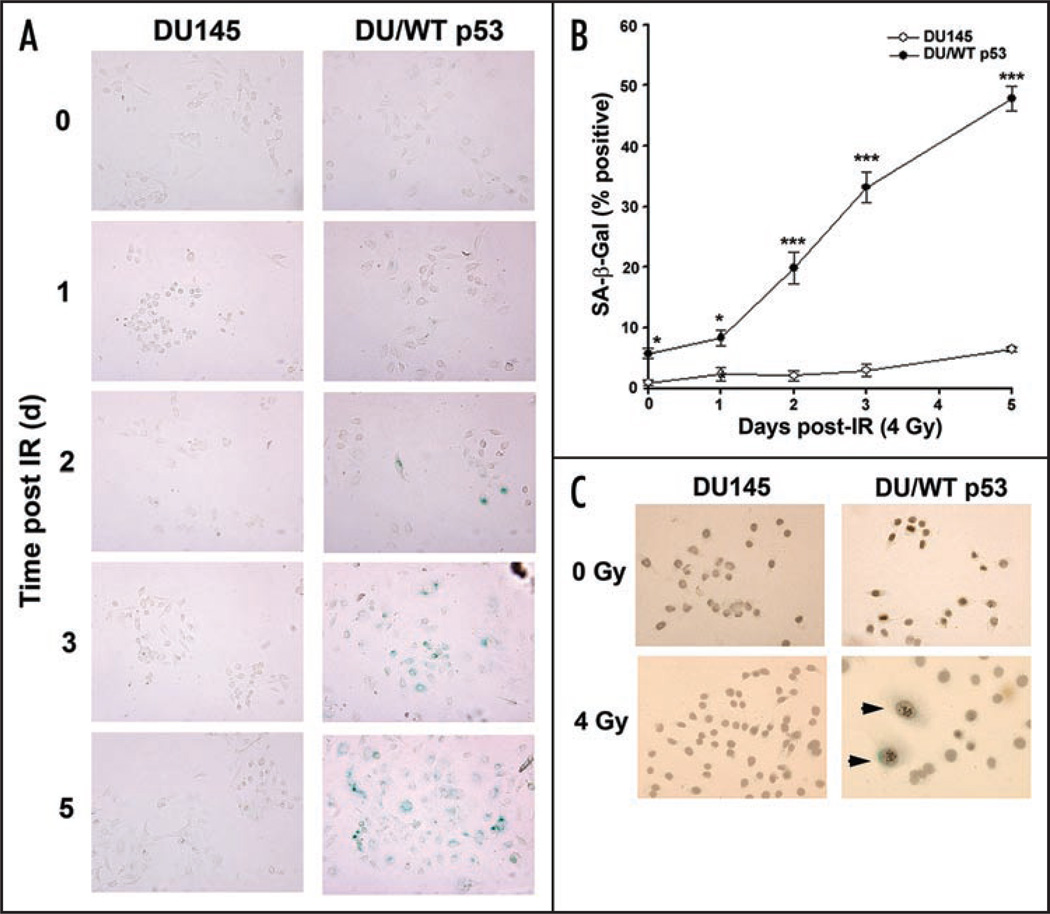
DNA-damaging agents induces phenotypic changes similar to replicative senescence that include an enlarged, flattened morphology that stain positive for acidic (pH6) β-galactosidase activity, termed senescence-associated β-galactosidase (SA-β-gal) staining.11,12 However, DU145 cells escape premature senescence as a 4 Gy dose of irradiation failed to stain greater than 5% of DU145 cells positive for SA-β-gal (Fig. 1A). Unexpectedly, re-expression of WT p53 was sufficient for re-establishing irradiation-induced senescence in the absence of a functional Rb protein, as greater than 60% of the cells stained positive for SA-β-gal at 5 days (Fig. 1A). SA-β-gal positive cells appeared 2 days after irradiation and the percentage of cells staining SA-β-gal positive increased further after 3 and 5 days (Fig. 1B). To ensure the p53 transgene was indeed functional, a p53-responsive promoter luciferase reporter assay demonstrated that the transgene was capable of binding transcriptional targets as irradiation induced a six-fold increase in transcriptional activity.3
Figure 1.
Irradiation-induced senescence is dependent on functional p53 protein. (A) DU145 and DU/WT p53 cells were either mock (0 Gy) or irradiated (4 Gy) and stained for β-galactosidase activity five days later. (B) The percentage of β-galactosidase positive cells were scored using bright-field microscopy. (C) DU145 and DU/WT p53 cells were stained for β-galactosidase activity and DAPI to reveal SAHF. DAPI fluorescence was converted to grayscale and inverted to demonstrate areas of condensed DNA.
Alterations of chromatin structure are believed to contribute to the irreversible nature of the senescent state forming characteristic heterochromatin structure called senescence-associated heterochromatic foci (SAHFs).5,13 Senescent cells maintain permanent arrest by chromatin remodeling, which can be visualized by brightly stained foci using 4'6-diamidino-2-phenylindole (DAPI). To confirm DU/WT p53 cells are permanently arrested, DU145 and DU/WT p53 cells were stained for SAHF using β-galactosidase and the nuclear counterstain DAPI. Images taken with DAPI were converted to black and white and inverted, so that they can be superimposed on bright-field images taken of SA-β-gal staining. Combined images revealed replicating cells (unirradiated DU145 and DU/WT p53 cells, irradiated DU145 cells) lacked SA-β-gal staining with even nuclear DAPI staining (Fig. 1C). In contrast, irradiated DU/WT p53 cells displayed a large flattened morphology that stained positive for SA-β-gal with SAHF consistent with terminal arrest (Fig. 1C, arrows).
p53-dependent stabilization of p27 coincides with the onset of irradiation-induced senescence
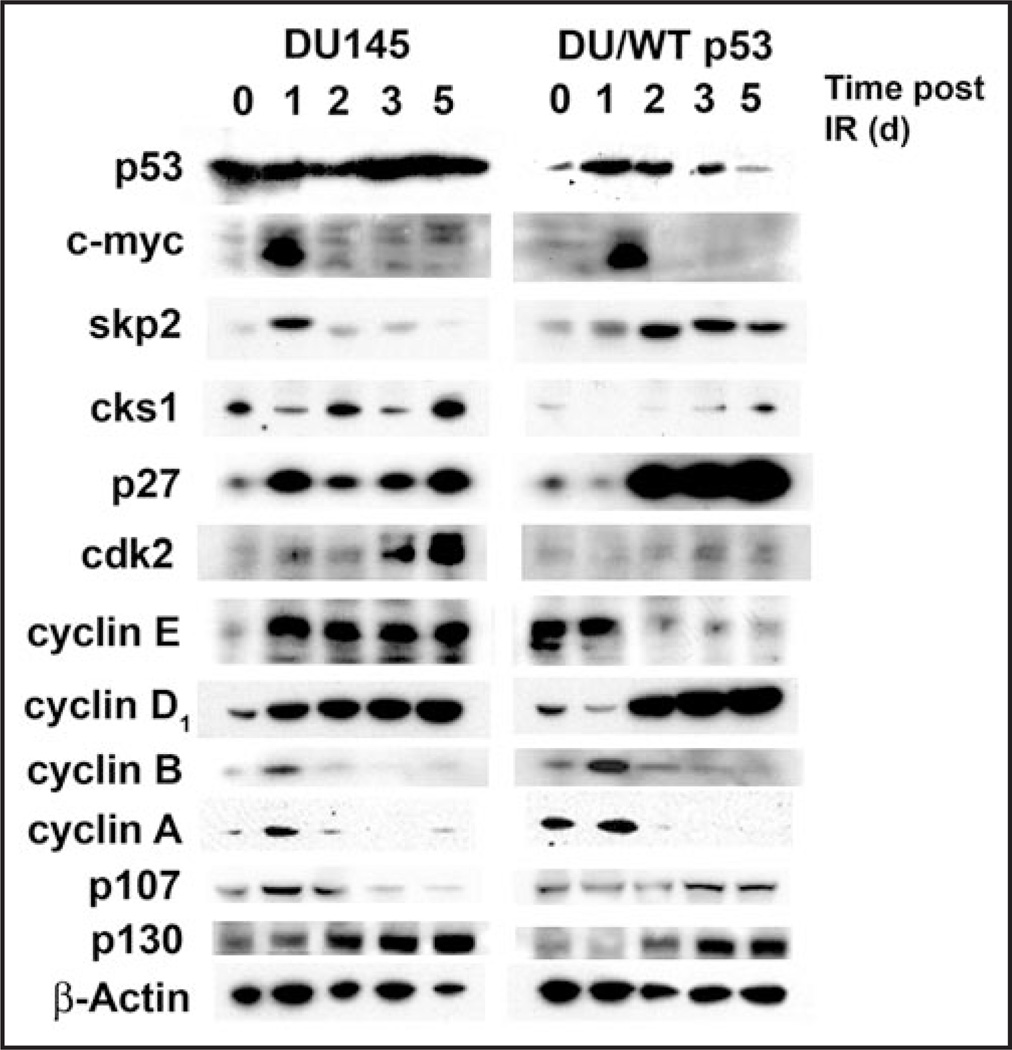
Senescent cells display changes in gene expression not observed in proliferating cells or quiescent cells that include increased expression of classical CDKIs, p16 and p21.14,15 To determine the molecular changes associated with senescence downstream of p53, cell lysates were prepared from irradiated DU145 and DU/WT p53 cells at 0, 1, 2, 3 and 5 days post-irradiation. Typically, intracellular levels of p53 protein remain low in unstressed cells due to its ongoing ubiquitin-dependent proteasomal degradation that is regulated by interactions with E3-ubiquitin ligases, such as MDM2.16 Consistent with most p53 mutations, the two dominant negative mutations in both p53 alleles of DU145 cells resulted in high basal expression of p53. However, p53 levels were not altered by irradiation exposure. In contrast, DU/WT p53 cells stabilized p53 protein levels at 1 and 2 days, consistent with irradiation-induced activation of p53 (Fig. 2). Thus re-expression of WT p53 was sufficient to overcome the dominant negative effects of the endogenous mutants. p53 initiates permanent cell cycle arrest by inducing the transcription of the CDKI, p21. However, p21 expression was neither detected in replicating DU145 cells nor DU/WT p53 cells entering senescence (data not shown). The absence of p21 expression in DU145 cells may be attributed to promoter methylation as it has been previously shown that mRNA levels were undetectable unless cells are treated with demethylating agent 5-Aza-2 deoxycytidine.17 Thus, DU/WT p53 cells undergoing premature senescence must utilize alternative mechanisms other than p21 transcription.
Figure 2.
Irradiation-induced senescence is independent of Rb. DU145 and DU/WT p53 cells were monitored for changes in protein expression associated with the onset of senescence. Cells lysates were immunoblotted for p53, c-myc, skp2, cks1, p27, cdk2, cyclin E, cyclin D1, cyclin B, cyclin A, p107 and p130 at 0, 1, 2, 3 and 5 days post-irradiation (4 Gy). β-Actin served as a protein loading control.
To investigate other possible cell cycle inhibitors that may mediate permanent cell cycle exit, lysates were probed for the CDKI’s p16 and p27. Both DU145 and DU/WT p53 failed to express p16 protein (data not shown), however p27 levels increased markedly in DU/WT p53 cells at 2 days and remained elevated throughout 5 days, in contrast to DU145 cells that maintained relatively constant p27 expression (Fig. 2). The absence of p16 expression may be attributed to a missense mutation in codon 76 (GAC-TAC) in exon 2 of CDK2N, that is absolutely conserved through 7 distant species and likely to be functionally critical.18,19
Since p27 is not a direct transcriptional target of p53, control of p27 stabilization was investigated by probing lysates for the F-box protein, S-phase kinase associated protein (Skp2), part of the SCFSkp2 ubiquitin ligase complex necessary for p27 degradation.20 Skp2 levels transiently increased in DU145 cells at 1 day, while Skp2 levels not only increased and remained high in DU/WT p53 cells, but band migration was slightly retarded, suggesting a posttranslational modification (Fig. 2). Degradation of p27 requires Skp2 binding, however its binding is drastically reduced in the absence of another SCF complex member, cyclin-dependent kinase subunit 1 (Cks1).21 Targeted deletion of either Skp2 or Cks1 leads to substantial increases in p27 and reduces cell proliferation.21,22 Since changes in Skp2 protein apparently did not account for p27 stabilization, similar lysates were probed for Cks1.23 Cks1 protein expression begins in late G1 and peaks in S/G2 phases of the cell cycle. Cks1 protein levels in DU145 cells cycled from high to low through 5 days, typical of replicating cells (Fig. 2). However, DU/WT p53 cells expressed lower basal and induced (4 Gy) levels of Cks1 protein. Thus, p27 stabilization during onset of premature senescence may in part rely on p53-dependent suppression of cks1 protein. One possible mechanism may involve direct repression of the Cks1 promoter by p53.24 However, p53 may also initiate senescence indirectly by repressing c-myc. Recently, inactivation of c-myc has been shown to induce cellular senescence and requires expression of p53.25 Activation of p53 has been known to downregulate c-myc and shown to repress c-myc transcription through a mechanism involving histone deacetylation.26,27 Expression of c-myc transiently increased in both DU145 and DU/WT p53 cells 1 day post-irradiation, and returned to basal level in 24 hours (Fig. 2). Ultimately, p27 inhibits cyclin E-Cdk2 complexes that are necessary for progression through S-phase. Immunoblots revealed diminished basal levels of both cdk2 and cyclin E, while cyclin E levels decreased and remained low 2 days post irradiation in DU/WT p53 cells (Fig. 2). Thus, the p53-dependent stabilization of p27 was associated with decreases in both cyclin E and cdk2, effectively reducing the phosphorylation-dependent inactivation of Rb family proteins. Currently, the ability of p27 to inhibit cyclin D- cdk4/6 remains controversial, however studies show that differential phosphorylation of p27 can modulate activity between non-inhibitory and inhibitory roles.29 Immunoblots from irradiated DU/WT p53 cells showed that cyclin D levels rose 2 days post-irradiation in concert with increased p27 expression (Fig. 2). Interestingly, the decrease in cyclin E occurred simultaneously as cyclin D levels rose. In contrast to the G1 cyclins, the G2 cyclins (cyclin A and cyclin B) followed similar expression patterns between replicating DU145 cells and senescent DU/WT p53 cells (Fig. 2). Therefore, senescent DU/WT p53 cells may have permanently halted the cell cycle by inhibiting both cyclin E-cdk2 and cyclin D- cdk4/6, necessary for G1-S progression. Inhibition of cyclin-cdk complexes typically relieves the phosphorylation-dependent inhibition of Rb. Since Rb belongs to a family of proteins that perform partially overlapping functions, lysates were immunoblotted for changes in expression of p107 and p130 following irradiation. While p130 protein levels followed similar patterns in DU145 and DU/WT p53 cells, p107 levels decreased in replicating DU145 and increased in DU/WT p53 cells entering senescence (Fig. 2).
p107 is required for irradiation-induced senescence in the absence of Rb
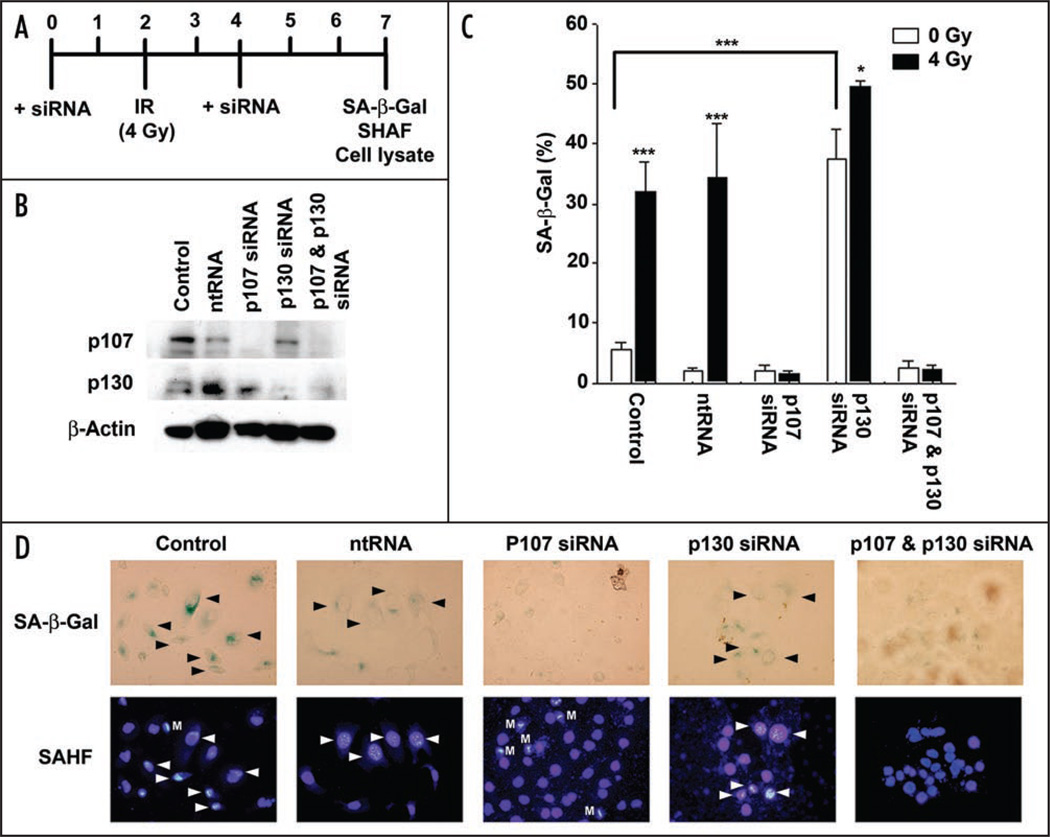
To determine which of the two remaining pocket proteins were dispensable for irradiation-induced senescence, siRNA silencing of p107 and p130 were performed. DU/WT p53 cells were either treated with 5 nM of nontargeting siRNA (ntRNA) or siRNA targeted to p107 and/or p130 2 days prior to irradiation (4 Gy). Cells were re-treated with siRNA or ntRNA 2 days post-irradiation to ensure knockdown throughout the experiment. Cell lysates and SA-β-gal staining were performed 3 days later, on day 5 after irradiation (Fig. 3A). Cell lysates confirmed protein knockdown of both p107 and p130 when treated with the corresponding siRNA constructs (Fig. 3B). SA-β-gal staining was performed on mock or irradiated DU/WT p53 cells for each of the treatments and counterstained with DAPI. ntRNA had no effect on cellular senescence, with DU/WT p53 cells displaying basal (<5%) and induced (>30%) SA-β-gal staining similar to control levels (Fig. 3C). Knockdown of p107 levels prevented DU/WT p53 cells from entering irradiation-induced senescence with SA-β-gal staining similar to basal levels (Fig. 3C). Interestingly, knockdown of just p130 forced DU/WT p53 cells into premature senescence regardless of DNA damage induced by irradiation, with basal levels of SA-β-gal staining approximately 35%. This novel finding that premature senescence requires p107 is further illustrated by the inability of untreated and irradiated DU/WT p53 cells to senesce when treated with siRNA for both p107 and p130. Staining for SAHF confirmed the results obtained with SA-β-gal staining, demonstrating many foci in the nuclei of irradiated control, ntRNA and p130 siRNA treated cells (Fig. 3D, arrows). Also of note are the increased numbers of mitotic cells appearing in the p107 siRNA treated cells that failed to enter senescence. Therefore, p107 plays a dominant role in irradiation-induced senescence in the absence of Rb and also knockdown of p130 triggers an unexpected form of premature senescence even in the absence of cellular stress.
Figure 3.
Rb-independent senescence in DU/WT p53 cells requires functional p107 protein. (A) Experimental timeline for DU/WT p53 cells treated with either missense siRNA, p107 siRNA, p130 siRNA or p107 and p130 siRNA. Cells were treated with siRNA for 2 d and re-treated at 4 d. Senescence-associated β-galactosidase staining was done 5 d after IR (4Gy). (B) Cell lysates were prepared prior to IR at 2 d post siRNA transfection and at 5 d after IR to evaluate knockdown of p107 and p130. (C) Number of SA-β-gal positive cells were scored as a percentage for the various treatments. (D) Images show representative SA-β-galactosidase staining and SAHF using DAPI of DU/WT p53 cells. Arrows indicate individual cells staining positive for SA-β-gal (black) and SAHF (white). Cell containing condensed chromosomes undergoing mitosis (M) are labeled accordingly.
Discussion
Normal cells possess an innate mechanism for preventing proliferation in the event of severe DNA damage and both p53 and Rb are known as critical mediators of premature cell senescence. Malignant cells often lose this ability by acquiring mutations within proteins controlling G1 progression. While Rb activation is a clear component in the cellular brakes to oncogenic stress and DNA damage, less is known about remaining members of the Rb family (p107 and p130). Rb and p53 are commonly mutated in the majority of cancer cells and the prostate cell line, DU145, lacking functional Rb and p53, provided a unique model to investigate whether p107 or p130 can induce irradiation-induced senescence.
Our results confirmed that irradiation-induced senescence requires functional p53 as demonstrated by increased SA-β-gal staining and SAHF in irradiated DU145 cells re-expressing WT p53. However, this form of premature senescence did not require Rb and revealed that p107 can compensate for a loss in Rb function. siRNA knockdown of p107 prevented irradiation-induced senescence previously re-instated by expression of WT p53 in DU145 cells lacking Rb. Further evidence for redundancy in the Rb family has been previously demonstrated by the prevention of oncogene-induced senescence by ras transformation in epidermal cells simultaneously ablated for both p107 and Rb.30 Previous studies have implicated p130 as a major player in senescence induction, however these experiments were carried out in cells containing all three Rb family members.31,32 Recently, p130 has been implicated in mediating quiescence rather than senescence by forming an evolutionarily conserved multisubunit protein complex containing p130 and E2F4 that mediates the repression of cell cycle-dependent genes.33 Unexpectedly, knockdown of p130 in cells lacking Rb triggered premature senescence in the absence of DNA damage, demonstrating a novel form of growth arrest that may mimic oncogene-induced senescence. p107 has previously been shown to inhibit proliferation in ras-mediated transformed fibroblasts and perform a tumor-suppressive role when Rb is inactivated.34 Therefore the absence of both Rb and p130 may provide a cellular environment similar to oncogene overexpression that can be suppressed as long as p107 and p53 remain functional.
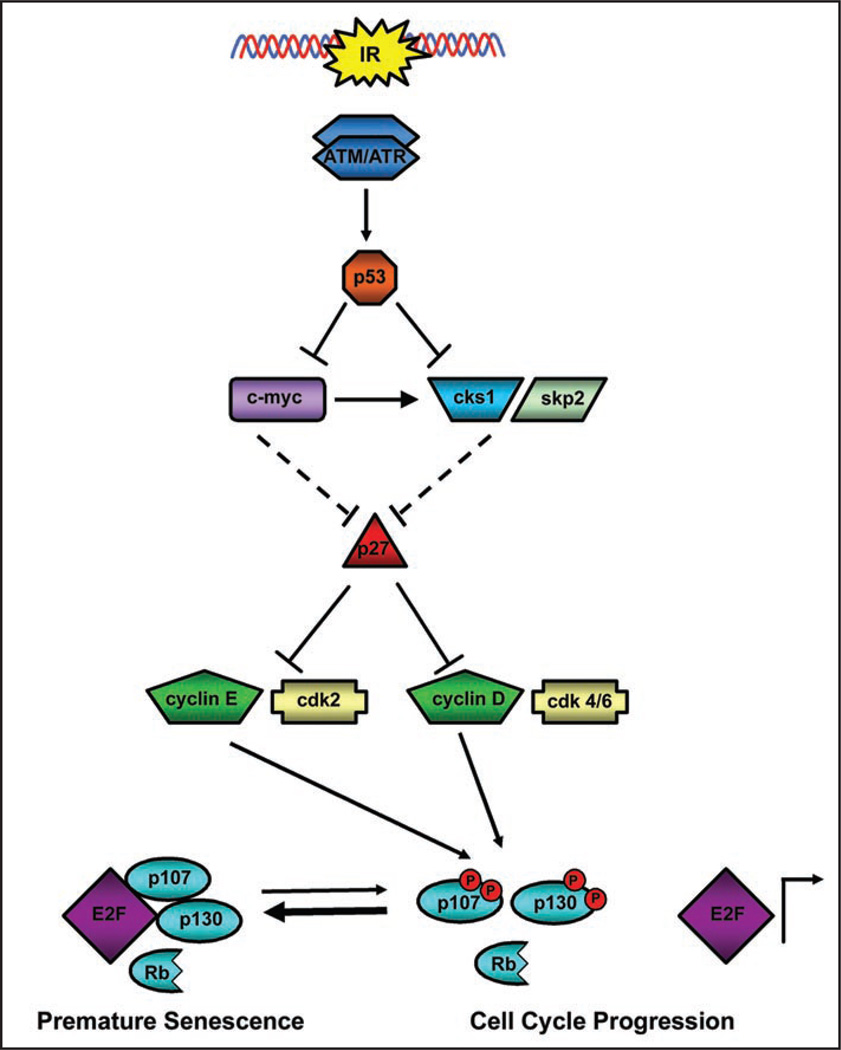
The mechanism of senescence by p107 is unclear, but most likely involves chromatin remodeling, as cells treated with p107 siRNA failed to form SAHF. While Rb preferentially binds E2F1/2/3 in late G1, p103 and p107 form transcriptional repression complexes with E2F4/5, HDAC and SUV39H1 in G0 and early G1 respectively.35 While p107 was shown to be required for irradiation-induced senescence, functional p53 was absolutely required to initiate downstream signaling. Typically p53 induces the transcription of p21, arresting the cell cycle in Rb+/+ cells, however we demonstrated a novel mechanism by which p53 can also indirectly stabilize another CDKI, p27. Stabilized p27 then can inhibit cyclin E-cdk2 and possibly cyclin D-cdk4/6, allowing p107 to play the role of Rb and permanently halt the cell cycle. Results shown in Figure 4 describe a mechanism by which p53 may stabilize p27. Stabilization of p27 was associated with decreased expression of both c-myc and Cks1 (Fig. 4). The decrease in c-myc protein may serve to stabilize p27 by preventing the transcriptional repression of p27 by c-myc and inhibiting c-myc dependent transcription of Cks1, part of the SCF ubiquitin ligase complex.28 However, the absence of c-myc expression in DU/WT p53 did not account for the initial stabilization of p27 as p27 levels increased at day 3. More likely p27 may have been stabilized by direct repression of the Cks1 promoter by p53.24
Figure 4.
Rb-independent senescence is mediated by p53-dependent stabilization of p27. Substantial DNA damage activates ATM/ATR, which in turn stabilizes p53. p53 can stabilize p27 directly by repressing the transcription of cks1 or indirectly by repressing c-myc transcription, relieving p27 repression and preventing c-myc dependent transcription of cks1. The stabilized p27 then inhibits both cyclin E and cyclin D, essential for G1-S progression. Unphosphorylated p107 is then able to permanently halt the cell cycle and maintain premature senescence. (------) lines indicate tentative and hypothetical interactions.
Cellular senescence has recently been show to be a major player in in vivo tumor regression as restoration of p53 function in sarcoma and liver carcinoma increases p53 dependent cellular senescence, rather than apoptosis.36,37 Since most prostate cancers frequently contain mutations in Rb and not p107 or p130, treatment of these cancers with traditional chemotherapy and irradiation may still be effective as long as p53 function remains intact.
Experimental Procedures
Cell line models and culture conditions
DU145 cells were acquired from the American Type Culture Collection (Manassas, VA USA; ATCC HTB-81). DU145 cells were cultured in MEM eagle with 2 mM L-glutamine and Earle’s BSS adjusted to contain 1.5 g/L sodium biocarbonate, 0.1 mM nonessential amino acids, 0.1 mM sodium pyruvate, supplemented with 10% FBS and 100 U/ml penicillin and 100 mg/ml streptomycin.
The effects of p53 activation on cellular senescence were examined by infecting DU145 cells with retroviral vectors encoding WT p53 as described.23 The WT p53 encoding retroviral vectors24 were generously provided by Dr. Moshe Oren (The Weizmann Institute of Science, Rehovot, Israel). DU145 cells infected by WT p53 retroviral vector are designated as DU/WT p53.
Western blot analysis
Immunoblots were performed as described previously.27 Antibodies purchased from Santa Cruz Biotechnology (Santa Cruz, CA) were raised against cdk2 (M2), c-myc (C-19), cyclin A (H-432), cyclin B (D-11), cyclin E (M-20), E2F-1 (C-20), p16 (N-20), p21 (M-19), p27 (C-19), p53 (DO-1), p107 (C-18), p130 (C-20), Rb (IF-8). Anti-cyclin D1 (DCS-6) and DP-1 were purchased from BD biosciences (San Jose, CA) and anti-cks1 was purchased from Zymed (Carlsbad, CA). Anti-β-actin antibody (Ab-1) was purchased from Calbiochem (La Jolla, CA).
siRNA knockdown of endogenous p107 and p130
DU/WT p53 cells were plated on coverslips and media was changed to 0.5% FBS absent of antibiotic supplement the day before transfection. Cells were transfected with 5 nM of nontargeting siRNA (sc-37007), p107 (sc-29423), p130 (sc-29425) or with both p107 and p130 siRNA from Santa Cruz Biotechnology (Santa Cruz, CA) diluted in Opti-MEM Reduced Serum Media using Lipofectamine 2000 (Invitrogen, Carlsbad, CA). Cells were either irradiated (4 Gy) or lysed 48 h post-transfection. Irradiated cells were then incubated for 2 days and transfected again with 5 nM of siRNA. Three days after second transfection cells were either lysed or stained for β-galactosidase activity.
Senescence-associated β-galactosidase (SA-β-gal)
DU145 cells were seeded on German glass coverslips, allowed to attach overnight and either mock treated or exposed to a dose of 4 Gy IR. SA-β-gal staining was performed either three or five days after irradiation, at pH 6.0, using a β-gal staining kit (Cell Signaling, Danvers, MA). Briefly, after three PBS washes and fixation in 2% formaldehyde and 0.2% glutaraldehyde, cells were stained with 1 mg/ml of 5-bromo-4-chloro-3-indolyl-β-galactoside. The percentage of SA-β-gal positive cells was determined by scoring 100 cells, in triplicate, as either positive or negative for staining using bright-field microscopy (Zeiss, West Germany).
Staining of heterochromatin-associated histone modification
SAHF were identified by the preferential binding of the DNA dye, 4'6-diamidino-2-phenylindole (DAPI). Irradiated (4 Gy) DU145 and DU/WT p53 cells were stained for β-galactosidase activity at 5 d. Coverslips were then washed 3X with PBS, permeabilized with 0.2% Trition X-100/PBS and stained with 1 µg/ml DAPI. Bright field microscopy was used to capture images of β-galactosidase staining. Fluorescence images of DAPI-stained nuclei were captured, converted to grayscale and inverted. The resulting image was then merged with the β-galactosidase stained image.
Data analysis
Values shown are representative of three or more experiments, unless otherwise specified, and treatment effects were evaluated using one-way ANOVA (InStat 3.1). Errors are standard errors of the mean (SE). p values < 0.05 were taken as a significant difference between means (*p < 0.05, **p < 0.01 and ***p < 0.001).
Acknowledgements
Supported in part by grants from the NIH (R01098195) and the Brody Brothers Foundation Endowment (#997729).
Abbreviations
- ATM
ataxia-telangiectasia mutated
- CaP
prostate carcinoma
- CDKIs
cyclin-dependent kinase inhibitors
- DN
dominant negative
- DSB
double-strand break
- IR
irradiation
- MDM2
murine double mutant 2
- Rb
retinoblastoma
- SA-β-gal
senescence-associated β-galactosidase
- SAHF
senescence-associated heterochromatin formation
- Skp2
S-phase kinase associated protein
- WT
wild-type
References
- 1.Campisi J, d’Adda di Fagagna F. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol. 2007 doi: 10.1038/nrm2233. [DOI] [PubMed] [Google Scholar]
- 2.Seluanov A, Gorbunova V, Falcovitz A, Sigal A, Milyavsky M, Zurer I, Shohat G, Goldfinger N, Rotter V. Change of the death pathway in senescent human fibroblasts in response to DNA damage is caused by an inability to stabilize p53. Mol Cell Biol. 2001;21:1552–1564. doi: 10.1128/MCB.21.5.1552-1564.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lehmann BD, McCubrey JA, Jefferson HS, Paine MS, Chappell WH, Terrian DM. A dominant role for p53-dependent cellular senescence in radiosensitization of human prostate cancer cells. Cell Cycle. 2007;6:595–605. doi: 10.4161/cc.6.5.3901. [DOI] [PubMed] [Google Scholar]
- 4.Di Leonardo A, Linke SP, Clarkin K, Wahl GM. DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev. 1994;8:2540–2551. doi: 10.1101/gad.8.21.2540. [DOI] [PubMed] [Google Scholar]
- 5.Narita M, Nunez S, Heard E, Lin AW, Hearn SA, Spector DL, Hannon GJ, Lowe SW. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell. 2003;113:703–716. doi: 10.1016/s0092-8674(03)00401-x. [DOI] [PubMed] [Google Scholar]
- 6.DeGregori J, Johnson DG. Distinct and Overlapping Roles for E2F Family Members in Transcription, Proliferation and Apoptosis. Curr Mol Med. 2006;6:739–748. doi: 10.2174/1566524010606070739. [DOI] [PubMed] [Google Scholar]
- 7.Macaluso M, Montanari M, Giordano A. Rb family proteins as modulators of gene expression and new aspects regarding the interaction with chromatin remodeling enzymes. Oncogene. 2006;25:5263–5267. doi: 10.1038/sj.onc.1209680. [DOI] [PubMed] [Google Scholar]
- 8.Isaacs WB, Carter BS, Ewing CM. Wild-type p53 suppresses growth of human prostate cancer cells containing mutant p53 alleles. Cancer Res. 1991;51:4716–4720. [PubMed] [Google Scholar]
- 9.Bookstein R, Shew JY, Chen PL, Scully P, Lee WH. Suppression of tumorigenicity of human prostate carcinoma cells by replacing a mutated RB gene. Science. 1990;247:712–715. doi: 10.1126/science.2300823. [DOI] [PubMed] [Google Scholar]
- 10.Chow KN, Dean DC. Domains A and B in the Rb pocket interact to form a transcriptional repressor motif. Mol Cell Biol. 1996;16:4862–4868. doi: 10.1128/mcb.16.9.4862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chang BD, Broude EV, Dokmanovic M, Zhu H, Ruth A, Xuan Y, Kandel ES, Lausch E, Christov K, Roninson IB. A senescence-like phenotype distinguishes tumor cells that undergo terminal proliferation arrest after exposure to anticancer agents. Cancer Res. 1999;59:3761–3767. [PubMed] [Google Scholar]
- 12.Stein GH, Namba M, Corsaro CM. Relationship of finite proliferative lifespan, senescence, and quiescence in human cells. J Cell Physiol. 1985;122:343–349. doi: 10.1002/jcp.1041220303. [DOI] [PubMed] [Google Scholar]
- 13.Ye X, Zerlanko B, Zhang R, Somaiah N, Lipinski M, Salomoni P, Adams PD. Definition of pRB- and p53-dependent and -independent steps in HIRA/ASF1a-mediated formation of senescence-associated heterochromatin foci. Mol Cell Biol. 2007;27:2452–2465. doi: 10.1128/MCB.01592-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Robles SJ, Adami GR. Agents that cause DNA double strand breaks lead to p16INK4a enrichment and the premature senescence of normal fibroblasts. Oncogene. 1998;16:1113–1123. doi: 10.1038/sj.onc.1201862. [DOI] [PubMed] [Google Scholar]
- 15.Chang BD, Xuan Y, Broude EV, Zhu H, Schott B, Fang J, Roninson IB. Role of p53 and p21waf1/cip1 in senescence-like terminal proliferation arrest induced in human tumor cells by chemotherapeutic drugs. Oncogene. 1999;18:4808–4818. doi: 10.1038/sj.onc.1203078. [DOI] [PubMed] [Google Scholar]
- 16.Lavin MF, Gueven N. The complexity of p53 stabilization and activation. Cell Death Differ. 2006;13:941–950. doi: 10.1038/sj.cdd.4401925. [DOI] [PubMed] [Google Scholar]
- 17.Bott SR, Arya M, Kirby RS, Williamson M. p21WAF1/CIP1 gene is inactivated in metastatic prostatic cancer cell lines by promoter methylation. Prostate Cancer Prostatic Dis. 2005;8:321–326. doi: 10.1038/sj.pcan.4500822. [DOI] [PubMed] [Google Scholar]
- 18.Jarrard DF, Bova GS, Ewing CM, Pin SS, Nguyen SH, Baylin SB, Cairns P, Sidransky D, Herman JG, Isaacs WB. Deletional, mutational, and methylation analyses of CDKN2 (p16/MTS1) in primary and metastatic prostate cancer. Genes Chromosomes Cancer. 1997;19:90–96. [PubMed] [Google Scholar]
- 19.Okamoto A, Demetrick DJ, Spillare EA, Hagiwara K, Hussain SP, Bennett WP, Forrester K, Gerwin B, Serrano M, Beach DH, et al. Mutations and altered expression of p16INK4 in human cancer. Proc Natl Acad Sci USA. 1994;91:11045–11049. doi: 10.1073/pnas.91.23.11045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ganoth D, Bornstein G, Ko TK, Larsen B, Tyers M, Pagano M, Hershko A. The cell cycle regulatory protein Cks1 is required for SCF(Skp2)-mediated ubiquitinylation of p27. Nat Cell Biol. 2001;3:321–324. doi: 10.1038/35060126. [DOI] [PubMed] [Google Scholar]
- 21.Spruck C, Strohmaier H, Watson M, Smith AP, Ryan A, Krek TW, Reed SI. A CDKindependent function of mammalian Cks1: targeting of SCF(Skp2) to the CDK inhibitor p27Kip1. Mol Cell. 2001;7:639–650. doi: 10.1016/s1097-2765(01)00210-6. [DOI] [PubMed] [Google Scholar]
- 22.Nakayama K, Nagahama H, Minamishima YA, Matsumoto M, Nakamichi I, Kitagawa K, Shirane M, Tsunematsu R, Tsukiyama T, Ishida N, Kitagawa M, Hatakeyama S. Targeted disruption of Skp2 results in accumulation of cyclin E and p27(Kip1), polyploidy and centrosome overduplication. Embo J. 2000;19:2069–2081. doi: 10.1093/emboj/19.9.2069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bartek J, Lukas J. p27 destruction: Cks1 pulls the trigger. Nat Cell Biol. 2001;3:E95–E98. doi: 10.1038/35070160. [DOI] [PubMed] [Google Scholar]
- 24.Rother K, Li YY, Tschop K, Kirschner R, Muller GA, Mossner J, Engeland K. Expression of cyclin-dependent kinase subunit 1 (Cks1) is regulated during the cell cycle by a CDE/CHR tandem element and is downregulated by p53 but not by p63 or p73. Cell Cycle. 2007;6:853–862. doi: 10.4161/cc.6.7.4017. [DOI] [PubMed] [Google Scholar]
- 25.Wu CH, van Riggelen J, Yetil A, Fan AC, Bachireddy P, Felsher DW. Cellular senescence is an important mechanism of tumor regression upon c-Myc inactivation. Proc Natl Acad Sci USA. 2007;104:13028–13033. doi: 10.1073/pnas.0701953104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ragimov N, Krauskopf A, Navot N, Rotter V, Oren M, Aloni Y. Wild-type but not mutant p53 can repress transcription initiation in vitro by interfering with the binding of basal transcription factors to the TATA motif. Oncogene. 1993;8:1183–1193. [PubMed] [Google Scholar]
- 27.Ho JS, Ma W, Mao DY, Benchimol S. p53-Dependent transcriptional repression of c-myc is required for G1 cell cycle arrest. Mol Cell Biol. 2005;25:7423–7431. doi: 10.1128/MCB.25.17.7423-7431.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Keller UB, Old JB, Dorsey FC, Nilsson JA, Nilsson L, MacLean KH, Chung L, Yang C, Spruck C, Boyd K, Reed SI, Cleveland JL. Myc targets Cks1 to provoke the suppression of p27Kip1, proliferation and lymphomagenesis. Embo J. 2007;26:2562–2574. doi: 10.1038/sj.emboj.7601691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.James M, Ray A, Leznova D, Blain SW. Differential modification of p27Kip1 controls its cyclin D-cdk4 inhibitory activity. Mol Cell Biol. 2007 doi: 10.1128/MCB.02171-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lara MF, Santos M, Ruiz S, Segrelles C, Moral M, Martinez-Cruz AB, Hernandez P, Martinez-Palacio J, Lorz C, Garcia-Escudero R, Paramio JM. p107 acts as a tumor suppressor in pRb-deficient epidermis. Mol Carcinog. 2007 doi: 10.1002/mc.20367. [DOI] [PubMed] [Google Scholar]
- 31.Kapic A, Helmbold H, Reimer R, Klotzsche O, Deppert W, Bohn W. Cooperation between p53 and p130(Rb2) in induction of cellular senescence. Cell Death Differ. 2006;13:324–334. doi: 10.1038/sj.cdd.4401756. [DOI] [PubMed] [Google Scholar]
- 32.Jackson JG, Pereira Smith OM. Primary and compensatory roles for RB family members at cell cycle gene promoters that are deacetylated and downregulated in doxorubicin-induced senescence of breast cancer cells. Mol Cell Biol. 2006;26:2501–2510. doi: 10.1128/MCB.26.7.2501-2510.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Litovchick L, Sadasivam S, Florens L, Zhu X, Swanson SK, Velmurugan S, Chen R, Washburn MP, Liu XS, DeCaprio JA. Evolutionarily conserved multisubunit RBL2/p130 and E2F4 protein complex represses human cell cycle-dependent genes in quiescence. Mol Cell. 2007;26:539–551. doi: 10.1016/j.molcel.2007.04.015. [DOI] [PubMed] [Google Scholar]
- 34.Williams JP, Stewart T, Li B, Mulloy R, Dimova D, Classon M. The retinoblastoma protein is required for Ras-induced oncogenic transformation. Mol Cell Biol. 2006;26:1170–1182. doi: 10.1128/MCB.26.4.1170-1182.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sun A, Bagella L, Tutton S, Romano G, Giordano A. From G0 to S phase: a view of the roles played by the retinoblastoma (Rb) family members in the Rb-E2F pathway. J Cell Biochem. 2007;102:1400–1404. doi: 10.1002/jcb.21609. [DOI] [PubMed] [Google Scholar]
- 36.Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L, Newman J, Reczek EE, Weissleder R, Jacks T. Restoration of p53 function leads to tumour regression in vivo. Nature. 2007;445:661–665. doi: 10.1038/nature05541. [DOI] [PubMed] [Google Scholar]
- 37.Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V, Cordon-Cardo C, Lowe SW. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature. 2007;445:656–660. doi: 10.1038/nature05529. [DOI] [PMC free article] [PubMed] [Google Scholar]