Abstract
Background
Dopamine (DA) and the DA D2 receptor (D2R) are involved in the rewarding and the conditioned responses to food and drug rewards. Osborne-Mendel (OM) rats are genetically prone and S5B/P rats are genetically resistant to obesity when fed a high-fat diet. We hypothesized that the differential sensitivity of these two rat strains to natural rewards may also be reflected in sensitivity to drugs of abuse. Therefore, we tested whether OM and S5B/P rats showed a differential preference to cocaine using conditioned place preference (CPP). To also evaluate whether there is specific involvement of the D2R in this differential conditioning sensitivity, we then tested whether the D2R agonist bromocriptine (BC) would differentially affect the effects of cocaine in the two strains.
Methods
OM and S5B/P rats were conditioned with cocaine (5 or 10mg/kg) in one chamber and saline in another for 8 days. Rats were then tested for cocaine preference. The effects of BC (0.5, 1, 5, 10, 20mg/kg) on cocaine preference were then assessed in subsequent test sessions.
Results
OM rats did not show a significant preference for the cocaine-paired chamber on test day. Only the S5B/P rats showed cocaine CPP. Later treatment with only the highest dose of BC resulted in reduced cocaine CPP in S5B/P rats when treated with 5mg/kg cocaine and in OM rats treated with 10mg/kg cocaine.
Conclusion
Our results indicated obesity-resistant S5B rats showed greater cocaine CPP than the obesity-prone OM rats. These findings do not support a theory of common vulnerability for reinforcer preferences (food and cocaine). However, they show that BC reduced cocaine conditioning effects supporting at least a partial regulatory role of D2R in conditioned responses to drugs.
Keywords: addiction, psychostimulant, bromocriptine, conditioned place preference
1. Introduction
Obesity is one of the fastest growing public health problems worldwide. Nearly 30% of the adult US population is obese; an alarming statistic considering the increased morbidity and mortality linked with obesity, including an estimate of as many as 300,000 deaths per year in the US [1].
Similarly, drug addiction continues to be a pervasive problem worldwide. In the US alone it is estimated that 21.6 million people aged 12 or older (9.1% of the US population) need treatment for illicit drug or alcohol abuse [2]. Dopamine (DA) and more specifically DA D2 receptors (D2R) have been previously implicated in obesity as well as drug addiction and are specifically involved in the rewarding and conditioned responses to natural (food) and drug rewards [3–7].
In addition, the DA transporter (DAT) has also been implicated in both cocaine abuse and obesity. Cocaine has been known to block the action of DAT, therefore increasing levels of extra synaptic DA [8]. Clinical studies have shown that an intravenous dose of (0.3–0.6mg/kg) cocaine produces a “high” and leads to a 60–77% blockade of DAT [8]. In obese individuals, age and body mass index were negatively correlated with DAT levels [9]. Similarly, lower levels of DAT were found in C57 mice that were fed a high fat (40%) diet [10].
Therefore, both obesity and addiction have been linked with impaired brain DA function. Specifically for both conditions, clinical and preclinical studies have shown lower than normal levels of D2R in the striatum [11–14], [15], [16], [17],[2], [16, 18]. Similarly, when fed a high fat diet, rats exhibited decreased DA turnover in the mesolimbic pathways and show reduced preference for amphetamines in the CPP paradigm [19]. It follows from these observations that this shared mechanism may result in enhanced responses to natural rewards as well as drugs of abuse. We assessed this hypothesis by examining cocaine preference in two inbred rat strains with differing susceptibilities to diet-induced obesity. For this, we first examined cocaine conditioned place preference (CPP) in Osborne Mendel (OM) rats, which favor high fat diets over carbohydrates or proteins [20–23] and S5B/P (S5B) rats, which favor low fat diets [20, 21]. Because of these preferences, OM rats are considered susceptible to dietary obesity while S5B rats are considered obesity-resistant [20, 21, 23]. Furthermore, to be able to assess the role of D2R in cocaine CPP, we also assessed the effects of bromocriptine (BC), a D2R agonist drug that reduces food intake in leptin receptor deficient obese zucker (fa/fa) and in diet induced obese rats [24], on cocaine’s reinforcing effects. We hypothesized that OM and S5B rats will show differences in CPP to cocaine and in their response to BC.
2. Methods and Materials
2.1 Animals
This study used 13 OM and 13 S5B male 3–4 month old rats which were obtained from a colony at Pennington Biomedical Research Center. Rats were individually housed in clear plexi-glass cages with wire covers under standard laboratory conditions (22±2 °C, 50±10% relative humidity) and a reverse12h/12h light/dark cycle with lights on at 2000h and off at 0800h. All experimental sessions occurred during the rat’s dark cycle. Rat chow (Lab Diet, St. Louis, MO; laboratory rodent diet 5001: 13.496% fat, 28.507% protein, 57.996% carbohydrates) and tap water were available ad libitum. Body weight and food intake was measured on a daily basis at 1000h. All experiments were conducted in conformity with the National Academy of Sciences Guide for the Care and Use of Laboratory Animals [25] and Brookhaven National Laboratory Institutional Animal Care and Use Committee Protocols.
2.2 Drugs
Cocaine hydrochloride and BC were both purchased from Sigma-Aldrich (St. Louis, MO). Cocaine hydrochloride was calculated as a salt base and prepared by dissolving the cocaine in saline for doses of 5mg/kg and 10mg/kg. The rats were randomly assigned to the order of receiving these different cocaine doses. BC was prepared by dissolving it in an ethanol (10%), distilled water (10%) and peanut oil (80%) solution as previously described [26] to produce concentrations of 0.5mg/kg, 1mg/kg, 5mg/kg, 10mg/kg and 20mg/kg. Both drugs were administered intraperitoneally (IP).
2.4 Apparatus
The CPP apparatus (Coulbourn Instruments, Whitehall, PA) contained three compartments. The two end compartments (30.5 × 26.5 × 37cm) were connected by a central corridor (12.75 × 23 × 15.25cm). The compartment on the left had black walls with a perforated stainless steel floor with round holes on staggered centers, the central corridor was transparent with a smooth plexi-glass floor, and the right compartment had white walls with a stainless steel mesh floor. Infrared activity monitors measured locomotor activity in each compartment for each session. The Graphic State 3.02 program (Coulbourn Instruments, Whitehall, PA) was used to collect the experimental variables.
2.5 Procedures: Cocaine Conditioned Place Preference (CPP)
Habituation
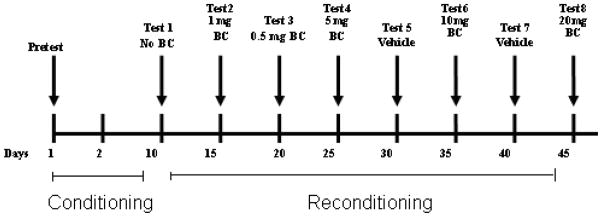
The CPP procedure consisted of the following phases: habituation, pretest, conditioning, test, reconditioning and retest (see Figure 1 for timeline). To habituate the rats to the transportation from the animal facility to the room where the experiments were conducted, rats were brought from the animal facility to the CPP room. They were left inside the room with the lights off for 30 minutes prior to any experimental procedures.
Figure 1.
Timeline of study.
Pretest
On day 1 of the experiment, a pretest was conducted to determine initial chamber preference. Rats were placed in the middle chamber with the doors open and were given 10 minute access to all chambers without cocaine or saline. Data was recorded by photo beam breaks within each chamber to determine baseline chamber preferences.
Conditioning
Rats were split into randomized groups based on strain and cocaine dose resulting in 4 different groups: OM rats that received 5mg/kg (OM 5mg/kg; n=6) or 10mg/kg cocaine (OM 10mg/kg (n=7); and S5B rats that received 5mg/kg (S5B 5mg/kg; n=7) or 10mg/kg cocaine (S5B 10mg/kg; n=6). Using baseline preference measurements from pretest, rats were given cocaine in the opposite chamber of preference. Cocaine and saline were administered on alternate days just prior to the rats being placed in the CPP apparatus. Rats were only allowed access to one of the two chambers each day during the conditioning session which lasted for a total of 30 minutes each. This procedure continued for 8 alternating days. Locomotor activity was also measured and analyzed inside the CPP chambers during the conditioning sessions by an infrared locomotor activity sensor located in the middle of the tap panel of the chamber.
Test
After the conditioning phase of the experiment, rats were tested for preference to cocaine. The procedure was similar to the pretest. Rats were placed in the middle chamber with doors open for 10 minutes and given access to both chambers. Preference to the cocaine-paired or saline paired chambers was measured by recording time spent in each box.
Reconditioning
After the first test session, rats were reconditioned using the same procedures as the conditioning phase of the experiment. However, instead of 8 alternating days, rats were only reconditioned for 4 alternating days.
Retest
After every 4 reconditioning days, rats were tested for cocaine CPP in response to different doses of BC (0.5, 1, 5, 10 and 20mg/kg) which were administered 15 minutes prior to the test session. Retest procedures were identical to Test and Pretest procedures.
2.6) Data and Statistical Analysis
Time spent in each chamber (during testing) and locomotor activity (during conditioning sessions) was analyzed. Each rat was said to have established cocaine CPP if significantly more time was spent in the cocaine paired chamber on test day compared to the pretest. In response to different doses of BC, time spent in the cocaine paired chamber after BC administration was calculated and comparisons were made to the initial test day to examine if BC had any effects on the time each rat spent in the cocaine paired chamber.
First, a two-way repeated measures analysis of variance (two-way RM ANOVA) was used to determine baseline cocaine preference (test) and subsequent cocaine preference in response to BC (retest). All pair-wise comparisons were assessed using the Holm-Sidak method. The statistical analyses were conducted using the SigmaPlot (v11.0) program.
3. Results
3.1) Cocaine Conditioned Place Preference
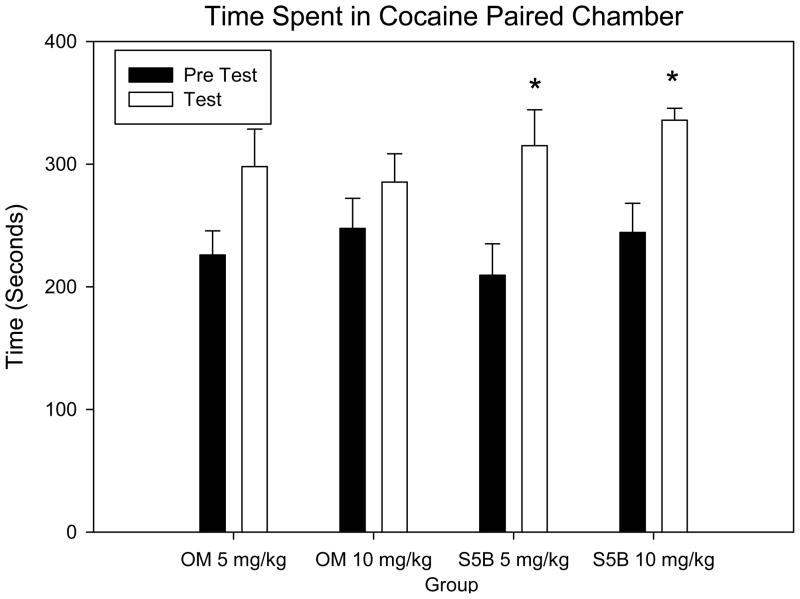
A two-way RM ANOVA with test session (pretest vs. test) and group set as factors showed no significant difference between groups [F (3,51) = 0.594, p >.05]; but a significant difference between test sessions [F (1, 51) = 20.855, p <.001; figure 2]. A pair-wise comparison of test sessions within each group showed significant increases in time spent in the cocaine paired chamber on test day compared to the pretest in the S5B rats both for the 5mg/kg (t = 3.273, p < .05; figure 2) and the 10mg/kg cocaine doses (t = 2.624, p <.05; figure 2). In the OM rats, cocaine only induced a trend for significance for CPP for the 5mg/kg cocaine dose (p = .051; figure 2).
Figure 2.
Cocaine CPP. Mean time (+SEM) spent in the cocaine paired chamber on pretest and test day. A pair-wise comparison of test sessions within each group show significant increases in time spent in the cocaine paired chamber on test day compared to the pretest only in the S5B 5mg/kg cocaine group (t = 3.273, p < .05) and the S5B 10mg/kg cocaine group (t = 2.624, p <.05). *Denotes significant difference (p <.05) in time spent in the cocaine paired chamber when compared to pretest.
3.2) Cocaine CPP after BC Treatment
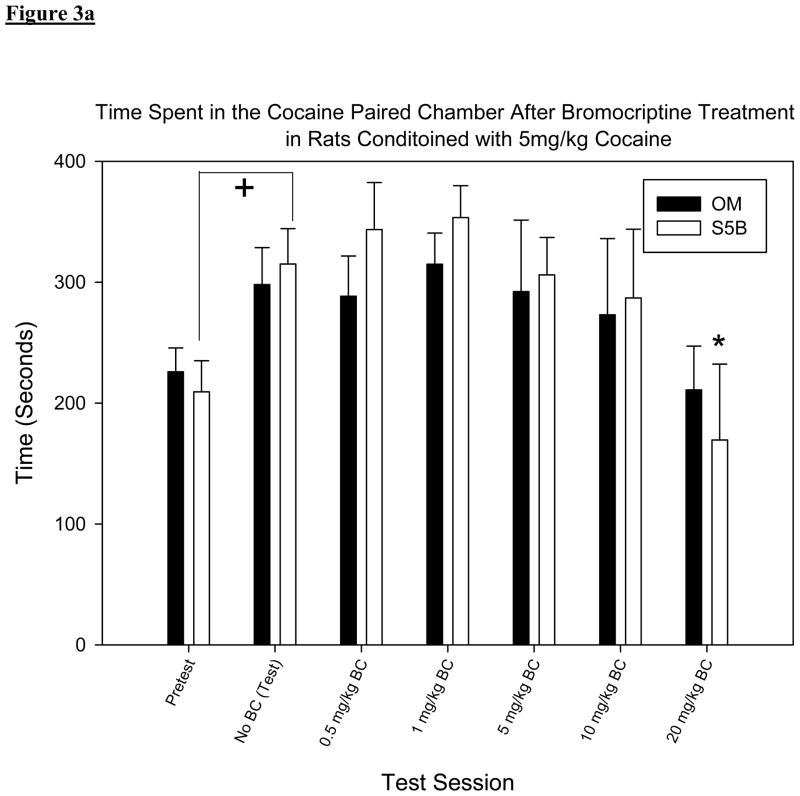
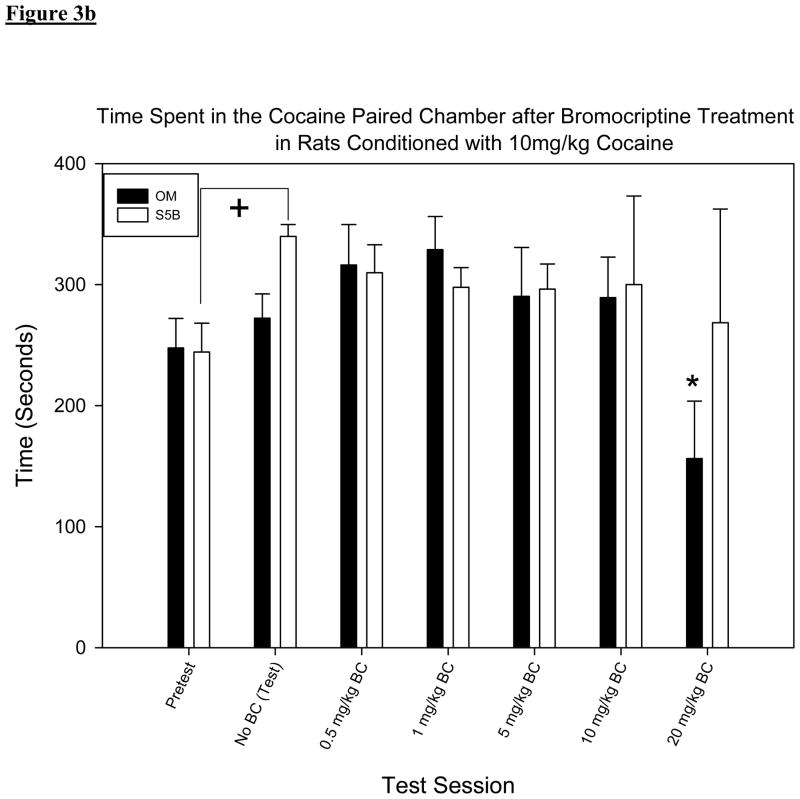
A two-way RM ANOVA with test session and group set as factors showed no significant differences between groups [F (3, 155) = 0.364, p > .05] but showed a significant difference between test sessions [F (5, 155) = 5.527, p < .001 figures 3a & b]. Pair-wise comparisons made between test day, when BC was not administered, and subsequent test sessions when BC was administered showed significant decreases in time spent in the cocaine paired chamber only following the highest dose of BC (20mg/kg) in the S5B rats when given 5mg/kg cocaine (t = 2.980, p <.05; figure 3a) and in the OM rats when given 10mg/kg cocaine (t = 3.092, p < .05; figure 3b).
Figure 3.
(a): Mean time spent in the cocaine paired chamber after BC treatment in rats conditioned with 5mg/kg cocaine. Reduced CPP is only found in the S5B 5mg/kg cocaine group (t = 2.980, p <.05) at the 20mgkg dose of BC.
(b)Mean time spent in the cocaine paired chamber after BC treatment in rats conditioned with 10mg/kg cocaine. Reduced CPP is only found in the OM 10mg/kg cocaine group (t = 3.092, p < .05 at the 20mgkg dose of BC.*Denotes significant difference (p <.05) in time spent in the cocaine paired chamber when compared to test day (no BC). + Indicates significant increase in time spent in the cocaine paired chamber on test day when compared to pretest.
3.3) Locomotor Activity
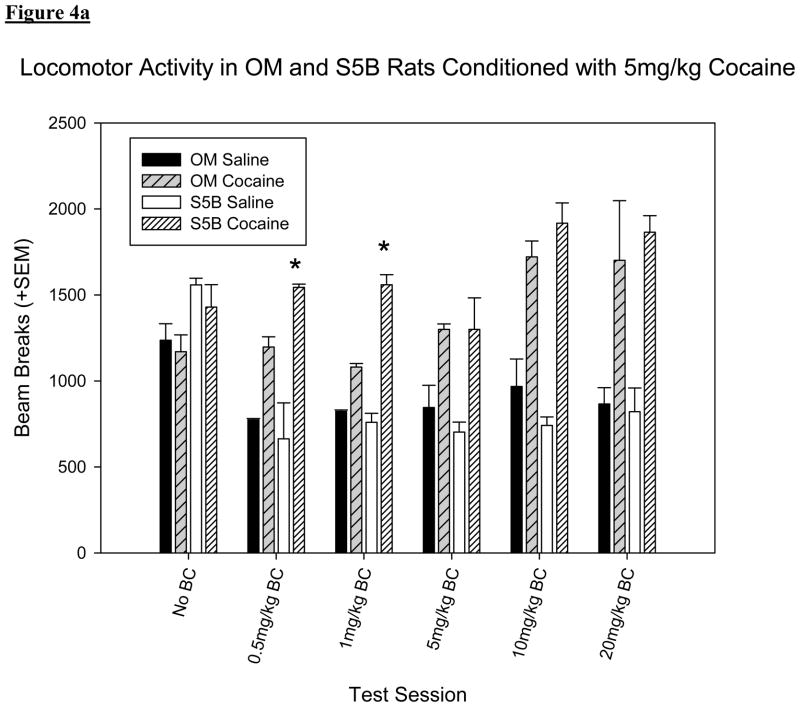
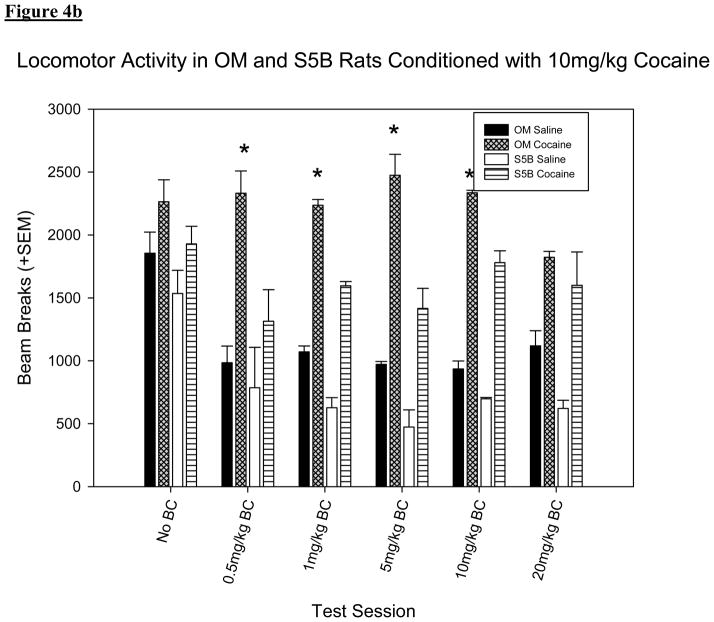
A three-way ANOVA with strain, drug (saline or cocaine) and cocaine dose (5 vs. 10mg/kg) set as factors show a significant difference in locomotor activity between strain [F (1, 935) = 13.489, p < .001; figure 4] and treatment [F (1, 935) = 72.782, p < .001; figure 4]. In contrast to the data on preference (section 3.1), OM rats showed more activity than S5B rats when conditioned with 10mg/kg but S5B rats showed more activity when conditioned with 5mg/kg cocaine. This difference in locomotor activity was not seen when higher doses of BC were administered. Both strains of rats showed more activity on cocaine conditioning days as opposed to saline conditioning days. The interaction between strain and drug was not significant [F (1, 935) = 1.965, p > .05].
Figure 4.
(a): Locomotor activity on conditioning days in OM and S5B rats conditioned with 5mg/kg cocaine. *Denotes significant difference (p <.05) compared to OM rats conditioned with the same drug. (b) Locomotor activity on conditioning days in OM and S5B rats conditioned with 10mg/kg cocaine. OM rats showed more activity than S5B rats (F (1, 935) = 15.255, p < .001) and both strains of rats showed more activity on cocaine conditioning days as opposed to saline conditioning days (F (1, 935) = 70.684, p < .001). *Denotes significant difference (p <.05) compared to S5B rats conditioned with the same drug.
3.4) Body Weight and Food Intake
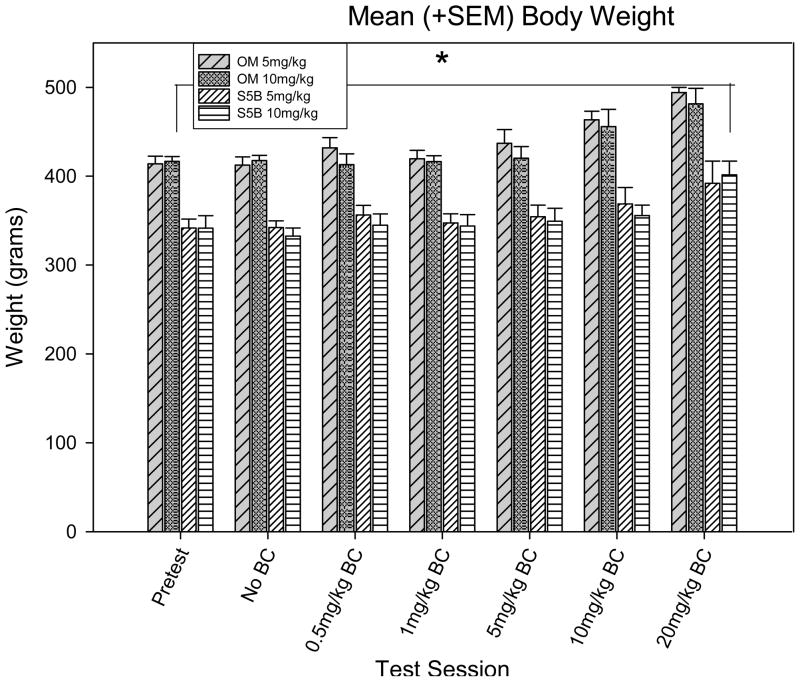
Body weight and food intake data confirmed that the two rat strains maintained their characteristic phenotypes throughout the study despite the conditioning procedures. A three-way ANOVA with test session, strain and cocaine dose set as factors show a significant difference between test session [F (8,35) = 70.567, p <.001) and strain [F (1,35) = 1737.450, p <.001; figure 5]. OM rats weighed more than S5B rats (t = 41.683, p < .001) and this difference was maintained throughout all test sessions (data not shown).
Figure 5.
Mean body weight (+SEM) between groups between test sessions.
*Denotes significant (p <.05) difference in body weight between OM and S5B rats.
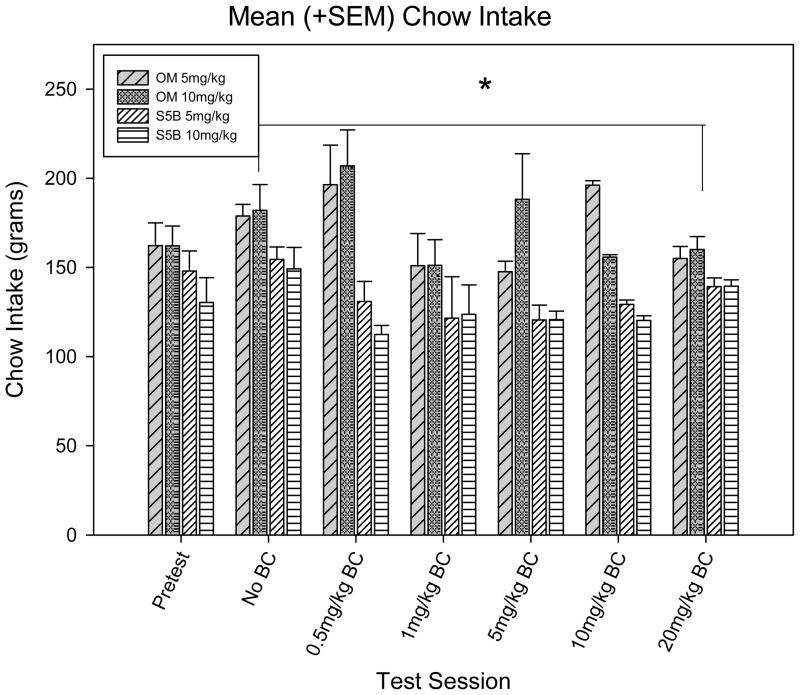
A three-way ANOVA with test session, strain and cocaine dose set as factors showed a significant difference between strains [F (1,35) = 44.328, p <.001], as would be expected, OM rats exhibited greater food intake than S5B rats (t = 6.658, p < .001).
4. Discussion
The present study examined whether differential sensitivity to diet-induced obesity in non-obese rats would be mirrored by a similar differential sensitivity to the reinforcing motor simulating effects of cocaine. The specific rat strains used express either genetic susceptibility or resistance to obesity and thus may serve as an appropriate model to make generalizations on drug abuse susceptibility in genetically susceptible and resistant obese populations.
Here we show that cocaine only induced a non-significant trend to CPP in the OM rats, but produced a robust CPP in the S5B rats. Despite the appeal of the notion of shared vulnerability for obesity and drug abuse, we found decreased CPP to cocaine in the obesity prone OM rats in contrast to the CPP observed in the obesity resistant S5B rats. Our findings are consistent with findings reported in the Fischer and Lewis rats, which also showed an opposite pattern in their sensitivity to cocaine than to food. Fischer rats, showed a greater motivation for food reward than Lewis rats whereas Lewis rats showed a greater motivation for cocaine than the Fischer rats [27]. This study together with the present study showed no cocaine preference in obesity prone rats (OM) compared to obesity resistant (S5B) rats. Thus, showing preference for food was associated with decreased preference for cocaine and vice-versa.
Although we used non-obese rats in this study, the two strains differed in body weight and such differences may have contributed to the CPP effects we observed. However, the strain differences in body weight between these two strains are evident across time, between sexes and even with different diets. OM rats show greater body weight than sex and age matched S5B rats at 10 weeks and 20 weeks of age, even when fed a grain diet (23.4 % protein, 3% fat, 53.5 carbohydrate) [23]. However, when fed a high fat diet (24.5% protein, 60% fat, 7.5% carbohydrate) this difference in body weight was superior and the OM rats weighed nearly twice as much as S5B rats [23]. Likewise, differences in body fat content between the two strains were only noticeable when given a high fat diet [23].
Chronic food restriction has also been shown to lead to increased sensitivity to drug abuse [28]. Food restriction led to an increase in the reward magnitude of drugs and an increase in the locomotor inducing effects of drugs as a result of neuroadaptations and not changes in drug disposition [28]. Although OM and S5B rats in this study were not subject to food restriction, if these two strains were to be fed a different diet, one either high in fat or high in starch, as opposed to regular rat chow, the results in the CPP might have differed. Since the OM rats prefer a high fat diet, if they were given this diet, we would predict that they would show a decrease in cocaine CPP; while an increase may be predicted in S5B rats. These predictions are based on previous studies in Sprague Dawley rats fed a high fat diet that showed delayed acquisition of cocaine self-administration in contrast to those fed regular chow which showed cocaine self- administration acquisition in a shorter amount of time [29]. Similarly, when given a choice between water sweetened with saccharin and intravenous cocaine 94% of rats showed a preference for the water sweetened with saccharin [30]. Therefore, if OM and S5B rats were fed either a diet high in fat or starch, given their strain preferences for these diets, it is very possible that they may respond differently to cocaine.
Although OM rats did not show cocaine CPP, they were not insensitive to cocaine, showing enhanced activity during conditioning sessions to the 10 mg/kg cocaine dose. At the 5mg/kg dose, this trend was reversed and S5B rats showed greater locomotor activity than OM rats. However, at both doses, the strain differences in locomotor activity were not seen with the highest dose of BC. This result was in contrast to previous studies where daily injections of 10mg/kg BC for 4 weeks resulted in increased locomotor activity in diet-induced obese Sprague Dawley rats [24].
BC is a D2R agonist and previous studies have shown that it can decrease motivation for cocaine. Other studies have shown that BC may be beneficial in treating obesity and diabetes. For example, dose dependent decreases in cocaine self-administration were found if the rat was pretreated with BC [31]. Likewise, BC was found to attenuate the responding for cocaine associated stimuli [32]. In addition, BC has also been shown to decrease food intake, decrease body fat composition and increase locomotor activity in both obese Zucker rats as well as diet-induced obese rats [24, 33]. In addition, BC reduced body fat composition in several different species including Djungarian hamsters, Swiss Webster mice and obese Zucker rats [26]. In the present study, it was only at the highest dose (20mg/kg) of BC, that effects were observed, but a consistent differential effect across strain or cocaine dose was not evident. One possible explanation for this differential effect of BC is that OM and S5B rats differ in their response curve to cocaine and therefore, might be differentially affected by BC, based on the dose of cocaine. OM rats showed a greater preference for the lower dose, 5mg/kg of cocaine. Perhaps for these rats the higher dose of 10mg/kg cocaine was aversive or simply not as preferred. In contrast to the OM rats, S5B rats showed a preference for both doses of cocaine. These differential responses to cocaine may account for the differential effects of BC on both strain and cocaine dose. Conceivably BC efficacy is affected by specific genetic differences inherent between the two strains which remain to be defined.
This disruption of CPP is supportive of prior studies that have shown that selective DA agonists can disrupt cocaine self-administration [34–36] and CPP [37–39]. Thus, BC as shown in the present study to reduce cocaine preference should warrant further investigation with respect to clinical application, perhaps limited to only a relatively high dose. However, one is given pause in doing this as, clinical studies conducted to date have not shown a beneficial effect of BC in the treatment of cocaine addiction [40, 41]. Future studies will examine the D2R availability in response to BC treatment as well as D2R levels between these two strains of rats. Furthermore, with advances in pharmacogenomics, future research may help us better understand and predict BC efficacy based on the genomic analysis of the patient.
In conclusion, our data highlight most the need to include in theoretical models of excess consumption several components of the brain’s reinforcement pathways that may elicit both food and drug seeking appetitive behavior.
Figure 6.
Mean food intake (+SEM) between groups between test sessions.
*Denotes significant (p <.05) difference in chow intake between OM and S5B rats.
Highlights.
Obesity-resistant S5B rats showed greater cocaine CPP than obesity-prone OM rats.
Vulnerability for reinforcer preferences (food and cocaine) are not the same.
Bromocriptine (BC) reduced cocaine CPP in both OM and S5B rats
BC efficacy on cocaine CPP was influenced by strain (genetic differences).
Data suggest partial regulatory role of D2R in conditioned responses to drugs.
Acknowledgments
Support Contributed by the NIAAA (Intramural Research Program, LNI).
Abbreviations
- DA, D2R, OM, S5B, CPP, BC
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Flegal KM, et al. Estimating deaths attributable to obesity in the United States. Am J Public Health. 2004;94(9):1486–9. doi: 10.2105/ajph.94.9.1486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Volkow ND, Li T-K. Drugs and alcohol: Treating and preventing abuse, addiction and their medical consequences. Pharmacology & Therapeutics. 2005;108(1):3–17. doi: 10.1016/j.pharmthera.2005.06.021. [DOI] [PubMed] [Google Scholar]
- 3.Volkow ND, et al. Overlapping neuronal circuits in addiction and obesity: evidence of systems pathology. Philos Trans R Soc Lond B Biol Sci. 2008;363(1507):3191–200. doi: 10.1098/rstb.2008.0107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Volkow ND, Wise RA. How can drug addiction help us understand obesity? Nat Neurosci. 2005;8(5):555–60. doi: 10.1038/nn1452. [DOI] [PubMed] [Google Scholar]
- 5.Wang GJ, et al. Positron emission tomographic evidence of similarity between obesity and drug addiction. Psychiatric Annals. 2003;33(2):104–111. [Google Scholar]
- 6.Wang G, et al. Similarity between obesity and drug addiction as assessed by neurofunctional imaging: a concept review. J Addict Dis. 2004;23(3):39–53. doi: 10.1300/J069v23n03_04. [DOI] [PubMed] [Google Scholar]
- 7.Wang GJ, et al. Similarity between obesity and drug addiction as assessed by neurofunctional imaging: a concept review. J Addict Dis. 2004;23(3):39–53. doi: 10.1300/J069v23n03_04. [DOI] [PubMed] [Google Scholar]
- 8.Volkow N, et al. Relationship between subjective effects of cocaine and dopamine transporter occupancy. Nature. 1997;6627:827–830. doi: 10.1038/386827a0. [DOI] [PubMed] [Google Scholar]
- 9.Chen PS, et al. Correlation between body mass index and striatal dopamine transporter availability in healthy volunteers--A SPECT study. Neuroimage. 2008;40(1):275–279. doi: 10.1016/j.neuroimage.2007.11.007. [DOI] [PubMed] [Google Scholar]
- 10.Huang XF, et al. Dopamine transporter and D2 receptor binding densities in mice prone or resistant to chronic high fat diet-induced obesity. Behav Brain Res. 2006;175(2):415–419. doi: 10.1016/j.bbr.2006.08.034. [DOI] [PubMed] [Google Scholar]
- 11.Wang G, et al. Brain dopamine and obesity. Lancet. 2001;357(9253):354–7. doi: 10.1016/s0140-6736(00)03643-6. [DOI] [PubMed] [Google Scholar]
- 12.Hamdi A, Porter J, Prasad C. Decreased striatal D2 dopamine receptors in obese Zucker rats: changes during aging. Brain Res. 1992;589(2):338–40. doi: 10.1016/0006-8993(92)91296-q. [DOI] [PubMed] [Google Scholar]
- 13.Thanos P, et al. Food restriction markedly increases dopamine D2 receptor (D2R) in a rat model of obesity as assessed with in-vivo muPET imaging ([(11)C] raclopride) and in-vitro ([(3)H] spiperone) autoradiography. Synapse. 2008;62(1):50–61. doi: 10.1002/syn.20468. [DOI] [PubMed] [Google Scholar]
- 14.Stice E, et al. Relation between obesity and blunted striatal response to food is moderated by TaqIA A1 allele. Science. 2008;322(5900):449–52. doi: 10.1126/science.1161550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Volkow ND, et al. Decreased dopamine D2 receptor availability is associated with reduced frontal metabolism in cocaine abusers. Synapse. 1993;14(2):169–77. doi: 10.1002/syn.890140210. [DOI] [PubMed] [Google Scholar]
- 16.McBride WJ, et al. Regional CNS densities of serotonin and dopamine receptors in high alcohol-drinking (HAD) and low alcohol-drinking (LAD) rats. Alcohol. 1997;14(6):603–9. doi: 10.1016/s0741-8329(97)00072-4. [DOI] [PubMed] [Google Scholar]
- 17.Thanos PK, et al. D2R DNA Transfer Into the Nucleus Accumbens Attentuates Cocaine Self Administration in Rats Synapse. 2008;62(5):481–486. doi: 10.1002/syn.20523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Thanos PK, et al. DRD2 gene transfer into the nucleus accumbens core of the alcohol preferring and nonpreferring rats attenuates alcohol drinking. Alcohol Clin Exp Res. 2004;28(5):720–8. doi: 10.1097/01.alc.0000125270.30501.08. [DOI] [PubMed] [Google Scholar]
- 19.Davis JF, et al. Exposure to elevated levels of dietary fat attenuates psychostimulant reward and mesolimbic dopamine turnover in the rat. Behavioral Neuroscience. 2008;122(6):1257–1263. doi: 10.1037/a0013111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schaffhauser AO, et al. Effects of a high-fat diet and strain on hypothalamic gene expression in rats. Obes Res. 2002;10(11):1188–96. doi: 10.1038/oby.2002.161. [DOI] [PubMed] [Google Scholar]
- 21.Smith KR. A critical literature review submitted in partial fulfillment of the requirements for the senior research thesis. 2006. Understanding Obesity through a Comparison of Osborne-Mendel and S5B/P1 Strains of Rat. [Google Scholar]
- 22.Nagase H, Bray GA, York DA. Pyruvate and hepatic pyruvate dehydrogenase levels in rat strains sensitive and resistant to dietary obesity. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 1996;270:489–495. doi: 10.1152/ajpregu.1996.270.3.R489. [DOI] [PubMed] [Google Scholar]
- 23.Schemmel R, Mickelsen O, Gill JL. Dietary Obesity in Rats: Body Weight and Body Fat Accretion in Seven Strains of Rats the Journal of Nutrition. 1969;100:1041–1048. doi: 10.1093/jn/100.9.1041. [DOI] [PubMed] [Google Scholar]
- 24.Davis LM, et al. Bromocriptine Administration Reduces Hyperphagia and Adiposity and Differentially Affects Dopamine D2 Receptor and Transporter Binding in Leptin-Receptor-Deficient Zucker Rats and Rats with Diet-Induced Obesity. Neuroendocrinology. 2008 Nov 4; doi: 10.1159/000170586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.NAS and NRC, Guide for the Care and Use of Laboratory Animals. Washington D.C: National Academy Press; 1996. [Google Scholar]
- 26.Cincotta AH, Meier AH. Reductions of body fat stores and total plasma cholesterol and triglyceride concentrations in several species by bromocriptine treatment. Life Sci. 1989;45(23):2247–54. doi: 10.1016/0024-3205(89)90066-0. [DOI] [PubMed] [Google Scholar]
- 27.Christensen CJ, et al. Demand for food and cocaine in Fischer and Lewis rats. Behav Neurosci. 2009;123:165–171. doi: 10.1037/a0013736. [DOI] [PubMed] [Google Scholar]
- 28.Carr KD. Chronic food restriction: enhancing effects on drug reward and striatal cell signaling. Physiol Behav. 2007;91(5):459–72. doi: 10.1016/j.physbeh.2006.09.021. [DOI] [PubMed] [Google Scholar]
- 29.Wellman PJ, Nation JR, Davis KW. Impairment of acquisition of cocaine self-administration in rats maintained on a high-fat diet. Pharmacol Biochem Behav. 2007;88(1):89–93. doi: 10.1016/j.pbb.2007.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lenoir M, et al. Intense Sweetness Surpasses Cocaine Reward. PLoS ONE. 2007;2(8):e698. doi: 10.1371/journal.pone.0000698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hubner C, Koob GF. Bromocriptine produces decreases in cocaine self-administration in the rat. Neuropsychopharmacology. 1990;3(2):101–108. [PubMed] [Google Scholar]
- 32.Koob G, et al. Effects of dopamine agonists and antagonists on cocaine-induced operant responding for a cocaine-associated stimulus. Psychopharmacology (Berl) 1996;126(4):311–322. doi: 10.1007/BF02247382. [DOI] [PubMed] [Google Scholar]
- 33.Davis LM, et al. Bromocriptine reduces steatosis in obese rodent models. J Hepatol. 2006;45(3):439–44. doi: 10.1016/j.jhep.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 34.Corrigall WA, Coen KM. Cocaine self-administration is increased by both D1 and D2 dopamine antagonists. Pharmacol Biochem Behav. 1991;39(3):799–802. doi: 10.1016/0091-3057(91)90168-2. [DOI] [PubMed] [Google Scholar]
- 35.Hubner CB, Moreton EJ. Effects of Selective D1 and D2 Dopamine Antagonists on Cocaine Self-Administration in the Rat. Psychopharmacology. 1991;105:151–156. doi: 10.1007/BF02244301. [DOI] [PubMed] [Google Scholar]
- 36.Koob GF, Le HT, Creese I. The D1 Dopamine Receptor Antagonist SCH 23390 Increases Cocaine Self-Administration in the Rat. Neuroscience Letters. 1987;79:315–320. doi: 10.1016/0304-3940(87)90451-4. [DOI] [PubMed] [Google Scholar]
- 37.Vorel SR, et al. Dopamine D3 receptor antagonism inhibits cocaine-seeking and cocaine-enhanced brain reward in rats. J Neurosci. 2002;22(21):9595–603. doi: 10.1523/JNEUROSCI.22-21-09595.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sora I, et al. Cocaine reward models: conditioned place preference can be established in dopamine- and in serotonin-transporter knockout mice. Proc Natl Acad Sci U S A. 1998;95(13):7699–704. doi: 10.1073/pnas.95.13.7699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baker DA, et al. Effects of intraaccumbens administration of SCH-23390 on cocaine- induced locomotion and conditioned place preference. Synapse. 1998;30(2):181–93. doi: 10.1002/(SICI)1098-2396(199810)30:2<181::AID-SYN8>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- 40.Handelsman L, et al. Bromocriptine for cocaine dependence. A controlled clinical trial. Am J Addict. 1997;6(1):54–64. [PubMed] [Google Scholar]
- 41.Gorelick DA, Wilkins JN. Bromocriptine treatment for cocaine addiction: Association with plasma prolactin levels. Drug Alcohol Depend. 2006;81:189–195. doi: 10.1016/j.drugalcdep.2005.06.010. [DOI] [PubMed] [Google Scholar]