Abstract
Synthetic CpG oligonucleotides (ODN) have potent immunostimulatory properties exploited in clinical vaccine trials. How CpG ODN are captured and delivered to the intracellular receptor TLR9, however, has been elusive. Here we show that DEC-205, a multilectin receptor expressed by a variety of cells, is a receptor for CpG ODN. When CpG ODN are used as an adjuvant, mice deficient in DEC-205 have impaired dendritic cell (DC) and B-cell maturation, are unable to make some cytokines such as IL-12, and display suboptimal cytotoxic T-cell responses. We reveal that DEC-205 directly binds class B CpG ODN and enhances their uptake. The CpG-ODN binding function of DEC-205 is conserved between mouse and man, although human DEC-205 preferentially binds a specific class B CpG ODN that has been selected for human clinical trials. Our findings identify an important receptor for class B CpG ODN and reveal a unique function for DEC-205.
Dendritic cells (DC) constantly sample their environment and present captured antigens to T cells in the secondary lymphoid compartment (1). Presentation of innocuous antigens captured under steady-state conditions leads to tolerance, whereas antigen presentation in the context of infection results in the priming of naïve T cells (2). The conversion of DC from tolerogenic to immunogenic during infection is thought to depend on their capacity to sense pathogen components or damage associated with disease (2). DC recognize infectious agents and their products using an array of receptors, including Toll-like receptors (TLR), C-type lectin-like receptors, retinoic-acid-inducible gene I–like receptors, nucleotide-binding oligomerisation domain–like receptors, and cytoplasmic DNA sensors (3). TLR9 detects nonmethylated cytosine-guanosine (CpG) motifs, which are underrepresented in the mammalian genome and therefore serve to alert DC to the presence of bacterial or viral DNA (3,4). Synthetic CpG oligonucleotide (ODN) agonists for TLR9 have therapeutic applications and consequently have been extensively characterized (5,6). They come in several classes, differing in their nucleotide sequence, their single- or double-stranded nature, their CpG content, and their degree of stabilization by phosphorothioation (6). Each class of CpG ODN differs somewhat in its biological outcome, which in part is determined by the cell types they activate and their trafficking within the endosomal network (7). Phase I/II clinical trials have focused on the use of class B ODN (B-ODN) (5), which are single stranded, are fully phosphorothioated, and induce B-cell proliferation, plasmacytoid DC (pDC) maturation, and in the mouse, maturation of conventional DC. The phosphorothioate backbone not only confers resistance to nuclease degradation but also increases ODN uptake (8,9), increases affinity for TLR9, and alters the sequence specificity for TLR9-mediated responses (9–11). The optimized B-ODN sequences for the stimulation of mice and humans are different. However, this species sequence specificity is not seen with natural phosphodiester DNA (11), and these phosphorothioate-specific sequence effects could act either at the level of ODN uptake or TLR9 binding.
The immunogenicity of B-ODN has been extensively documented in mouse experimental models, and B-ODN are now in phase I/II clinical trials as vaccine adjuvants to enhance immune responses against infectious diseases and cancer (5). Mechanistically, it is well established that activation by CpG ODN requires TLR9 (6). However, TLR9 resides in intracellular compartments (12), so how inoculated CpG ODN gain access to TLR9 remains unclear. There is some evidence to implicate receptor(s) in their uptake (13), including the natural killer cell Ig-like receptor KIR3DL2 (14) and CD14 (15) for some other cell types. Whether professional antigen presenting cells (APC) such as DC and B cells express unique receptors that facilitate uptake of CpG ODN has not been established. This is an important issue as presentation of vaccine antigens by these APC is critical for induction of T-cell immunity and antibody production.
DEC-205 (CD205) is a 205 kDa molecule that has a cysteine-rich domain, a fibronectin type II domain, and 10 C-type lectin-like domains, as well as an internalization sequence in its cytoplasmic tail (16,17). It is expressed at different levels on DC, B cells, T cells, and thymic epithelial cells (18). The function of DEC-205 is unknown, but it has endocytic capacity, recycles through late endosomal/lysosomal compartments (19), and facilitates antigen presentation when targeted with antigen-bearing monoclonal antibodies (20). Furthermore, its domain structure suggests the potential for recognition of multiple ligands. These features have lent support to the hypothesis that DEC-205 may function as a “promiscuous” antigen receptor (16,20). However, its natural ligands have yet to be identified (21,22). Here we show that DEC-205 is a key receptor involved in the uptake of B-ODN, a clinically relevant adjuvant, facilitating biological activities in DC and B cells. Our study also suggests that DEC-205 may dictate the species specificity of stimulatory ODN used in man.
Results
Mouse and Human DEC-205 Bind CpG ODN.
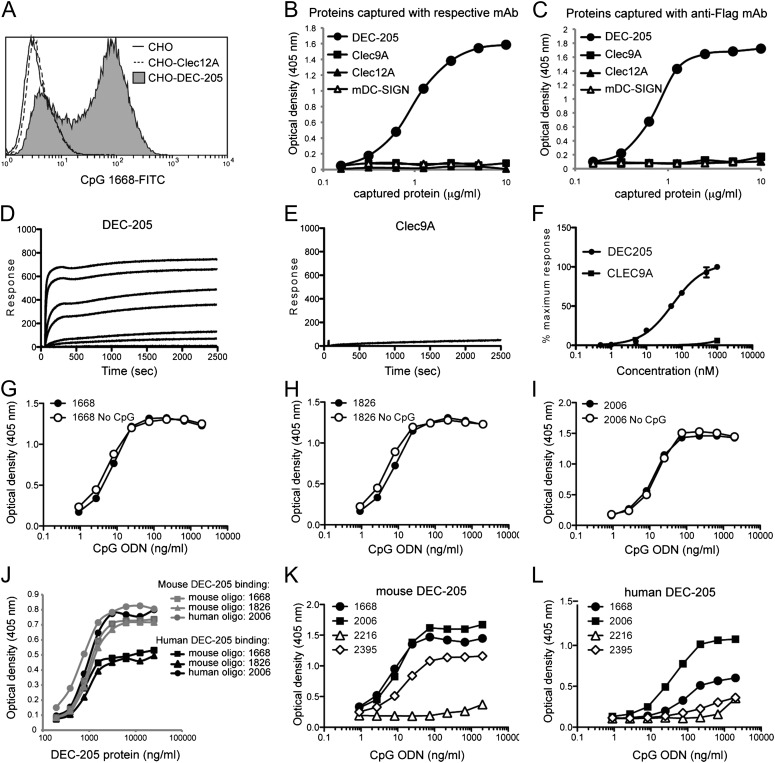
To investigate whether DEC-205 can bind CpG ODN, we transfected CHO-K1 cells with mouse DEC-205. In contrast to their untransfected counterparts or a control transfectant expressing another cell surface lectin (Clec12A), cells transfected with DEC-205 captured CpG ODN 1668 (Fig. 1A). To directly assess whether DEC-205 on its own was able to bind CpG ODN, we generated recombinant soluble DEC-205 and measured binding to CpG ODN by ELISA. Indeed, DEC-205 but not three other control lectins (mDC-SIGN, Clec12A, and Clec9A) bound CpG ODN 1668 (Fig. 1 B and C). We further confirmed the capacity of DEC-205 but not Clec9A to bind CpG ODN by using surface plasmon resonance (SPR) (Fig. 1 D–F). To determine whether the CpG motif was critical for binding DEC-205, we compared binding of three B-ODN (1668, 1826, and 2006) that contain the CpG motif to that of control counterparts in which the CpG motif was reversed to GpC (i.e., no CpG) so they cannot activate TLR9 (Fig. 1 G–I) (10). This revealed that the CpG motif was not necessary for binding DEC-205, despite its requirement for signaling via TLR9 (10).
Fig. 1.
DEC-205 directly binds CpG ODN. (A) CHO-K1 cells or CHO-K1 cells expressing DEC-205 or Clec12A were incubated on ice with 125 nM of FITC-conjugated 1668 CpG ODN (1 h). Binding was analyzed by flow cytometry. Data are representative of three experiments. ELISA plates were coated with 10 μg/mL of mAb against (B) DEC-205, Clec9A, Clec12A, or mouse DC-SIGN or (C) the Flag peptide. Graded doses of soluble flag-tagged DEC-205, Clec9A, Clec12A, or mouse DC-SIGN were then captured onto ELISA plates. Captured proteins were incubated with 1668 CpG ODN-biotin (10 μg/mL), and this binding was detected using SA-HRP and visualized with ABTS. (D and E) Sensorgrams showing binding of increasing concentrations of (D) DEC-205 protein or (E) control protein (Clec9A) injected over immobilized 1668 CpG ODN-biotin and (F) comparison of maximum response of DEC-205 and Clec9A binding as a percentage of maximal response as a function of concentration. Circles represent the mean values of two experiments ± SEM. The binding of CpG ODN by DEC-205 is not dependent on the CpG motif. (G–I) ELISA plates were coated with 10 μg/mL of 9B4 mAb against the Flag peptide and then incubated with mouse DEC-205 (5 μg/mL). Bound DEC-205 was interrogated with graded doses of biotinylated (G) 1668 CpG ODN, (H) 1826 CpG ODN, or (I) 2006 CpG ODN that either contained the CpG motif or not (No CpG). Representative data of three experiments are shown. (J) ELISA plates were coated with 10 μg/mL of mAb against the Flag peptide. Graded doses of either human or mouse flag-tagged DEC-205 were captured by the anti-Flag mAb and then exposed to various biotinylated CpG ODN (1 μg/mL). Alternatively, captured (K) mouse or (L) human DEC-205 (5 μg/mL) were exposed to graded doses of A- (2216), B- (1668 and 2006), or C- (2395) ODN. Bound CpG ODN were detected using SA-HRP and visualized using ABTS. This analysis was performed twice with similar findings, and one experiment is presented.
Because DEC-205 is conserved between mouse and man, we performed similar experiments using recombinant human DEC-205 with three different class B CpG ODN: 1668, 1826, and 2006. All three ODN stimulate mouse cells (23), but ODN 2006 is considered the most potent CpG ODN for human application (5). Strikingly, whereas mouse DEC-205 had the capacity to bind all three CpG ODN similarly, human DEC-205 displayed a greater capacity to bind ODN 2006 (Fig. 1 J–L). Although clinical trials predominantly use class B CpG ODN, class C CpG ODN are also used, and in the mouse, class A ODN (A-ODN) are also immunostimulatory. Thus, we asked whether DEC-205 could bind A-ODN and class C ODN (C-ODN). Neither mouse nor human DEC-205 bound A-ODN 2216 efficiently (Fig. 1 K and L). However, mouse DEC-205 could bind C-ODN 2395, whereas human DEC-205 did so very poorly (Fig. 1 K and L), reiterating that the ideal sequence of ODN recognized by human DEC-205 may differ from that recognized by mouse DEC-205.
DEC-205 Is Required for the Uptake and Response to CpG ODN.
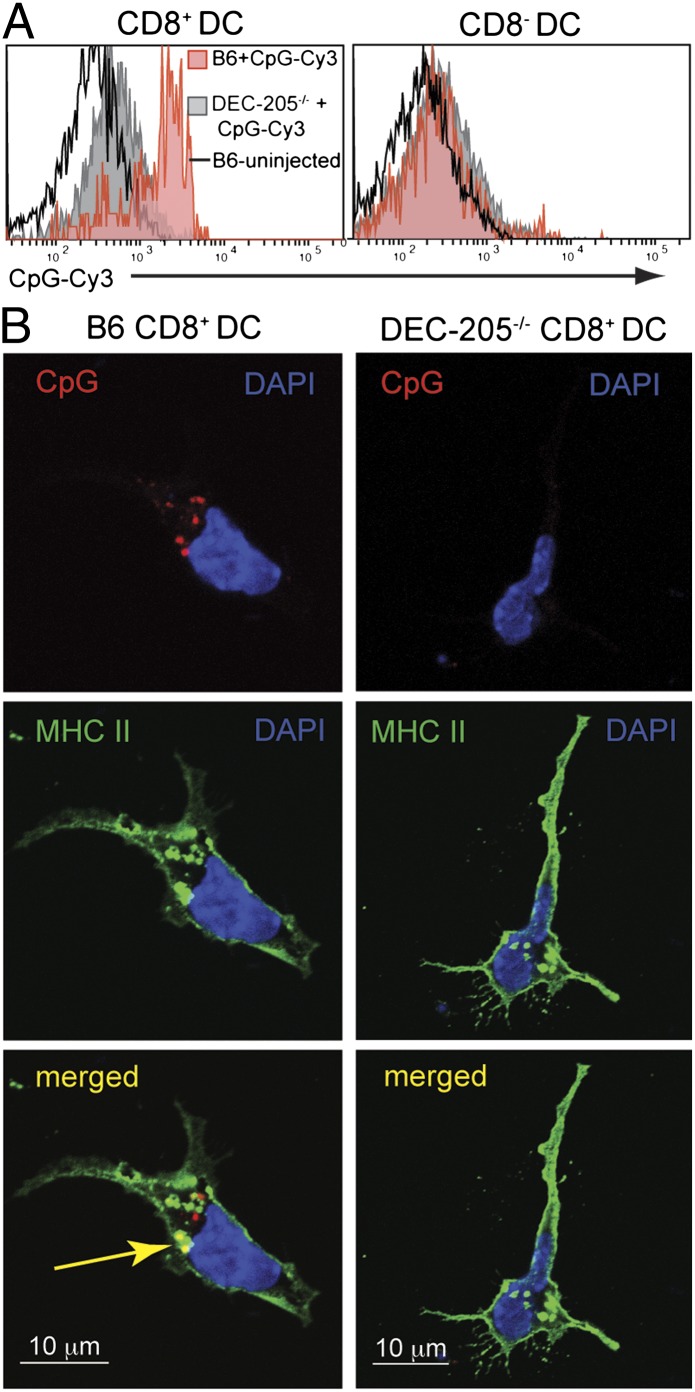
DEC-205 is expressed at high levels on CD8+ DC (Fig. S1) and has been successfully used to target antigen to this DC subset in vivo (24). Therefore, we sought to determine whether DEC-205 was also required for the acquisition of CpG ODN by CD8+ DC in vivo. Normal C57BL/6 (B6) or DEC-205–/– mice were injected with fluorescent CpG 1668, and 30 min later splenic DC were purified. Indeed, CD8+ DC acquired (Fig. 2A) and internalized (Fig. 2B) the fluorescent CpG ODN in a DEC-205-specific manner. Similarly, DC purified from naïve mice also required DEC-205 in vitro for optimal uptake of CpG ODN, but in this case, a significant amount of CpG ODN was also acquired independent of this receptor (Fig. S2A).
Fig. 2.
DEC-205 facilitates the uptake of CpG ODN. B6 or DEC-205–/– mice were injected i.v. with 20 μg 1668 CpG ODN-Cy3. Spleens were extracted 30 min later, DC purified, and examined by flow cytometry (A) for their uptake of CpG ODN. CD8+ DC were sorted by flow cytometry and then analyzed by confocal microscopy (B) for their internalization of CpG ODN. Nuclei were stained with DAPI (blue), MHC class II (green), and CpG ODN-Cy3 (red). Pictures depict one slice in the middle of the cell. Experiment was conducted four times, with this particular staining combination performed twice. Representative data are presented.
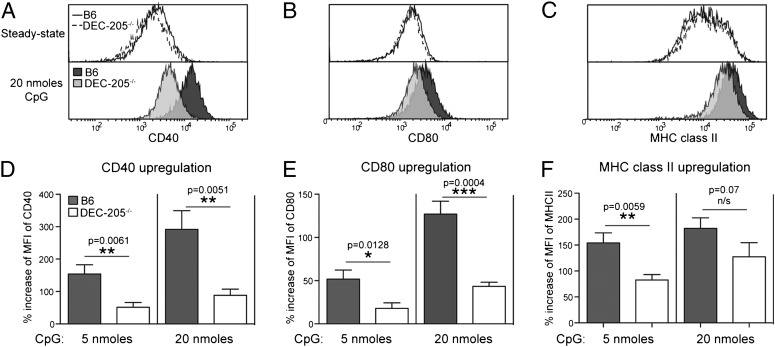
Next we asked whether DEC-205 was required by DC to respond to CpG ODN. Upon CpG ODN encounter, DC undergo maturational changes, up-regulating molecules that promote optimal antigen presentation. Thus, 5 h after injection of CpG ODN into B6 mice, splenic CD8+ DC had up-regulated CD40, CD80, and MHC class II. By contrast, CD8+ DC from CpG-injected DEC-205–/– mice showed significantly lower levels of these markers (Fig. 3 A–F). The inability of DEC-205–/– DC to fully mature in response to CpG ODN did not reflect generalized unresponsiveness to TLR ligands because DEC-205–/– DC showed equivalent activation to B6 DC when mice were injected with the TLR4 ligand, LPS (Fig. S3A). TLR9 is essential for DC activation caused by CpG ODN (Fig. S3B) (4, 6), and it is well established that this requires TLR9 engagement in late-endosomal/lysosomal compartments (12). Because DEC-205 is known to traffic to late endosomes and lysosomes (16,19), our results suggest that DEC-205 participates in CpG ODN binding on the DC surface and internalization to TLR9+ compartments. Nevertheless, DEC-205-deficient CD8+ DC showed partial, dose-dependent responses to CpG ODN (Fig. 3 D–F), suggesting they retained some capacity for CpG ODN internalization in the absence of DEC-205 expression. This is not surprising because DC are constitutively capable of engulfing large volumes of extracellular medium by macropinocytosis (25). We therefore sought to assess the role of DEC-205 on B cells, which are much less capable of fluid-phase (nonspecific) endocytosis.
Fig. 3.
DEC-205 facilitates DC maturation induced by CpG ODN. B6 or DEC-205–/– mice were injected i.v. with 5 or 20 nmols of 1668 CpG ODN, and then 5 h later spleens were extracted, DC isolated, and CD8+ DC examined by flow cytometry for expression of CD40, CD80, and MHC class II. Representative histograms of (A) CD40, (B) CD80, and (C) MHC class II expression of steady-state or 1668 CpG ODN-activated CD8+ DC. Percent increase of mean fluorescence intensity of (D) CD40, (E) CD80, and (F) MHC class II in CD8+ DC and CD8– DC in response to CpG. Cumulative data of five experiments ± SEM. Statistical significance was determined by a one-tailed unpaired Student t test.
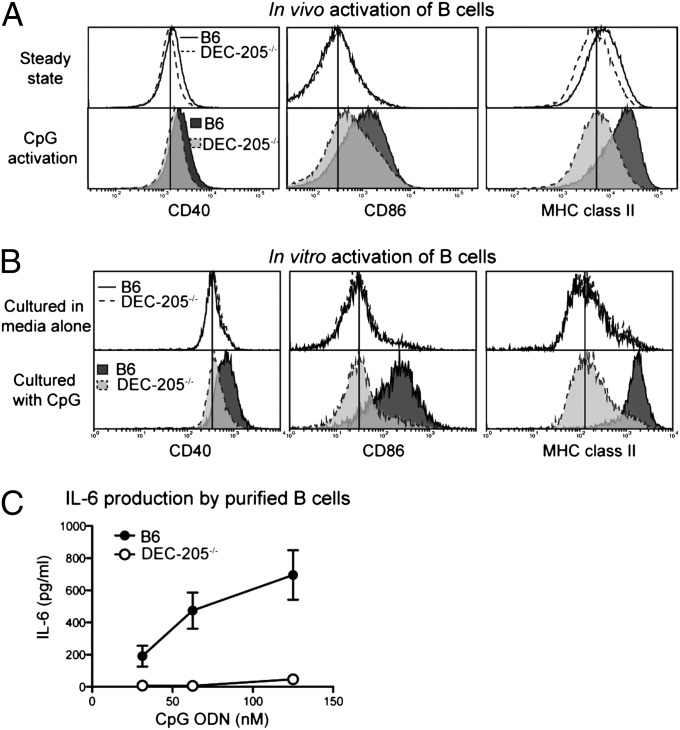
B cells express significant levels of DEC-205 (Fig. S1) particularly upon activation (24). To test the role of this receptor in CpG ODN activation of B cells, B6 or DEC-205–/– mice were injected with CpG ODN 1668. B-cell activation was assessed 1 d later by measuring surface expression of CD40, CD86, and MHC class II. B cells were more dependent on DEC-205 for CpG ODN recognition than DC (compare Figs. 4A and 3A). This was even more evident in vitro where DEC-205-deficient B cells showed a profound inability to up-regulate CD40, CD86, and MHC class II in response to CpG ODN (Fig. 4B). Indeed, purified DEC-205–/– B cells were also severely hampered in their ability to produce the cytokine IL-6 in response to CpG ODN (Fig. 4C) (6). To establish whether DEC-205 facilitated B-cell responses to CpG ODN by mediating uptake, we assessed the ability of B cells to capture CpG ODN in vivo and in vitro (Fig. S2 C and D). Indeed DEC-205 was required for optimal uptake of CpG ODN, although B cells could also acquire CpG ODN in a DEC-205-independent manner, particularly in vitro. Thus, although it is possible that the enhanced uptake of CpG ODN facilitated by DEC-205 resulted in the superior activation of B cells, an alternative possibility is that DEC-205 facilitates activation by promoting the delivery of captured CpG ODN to TLR9.
Fig. 4.
DEC-205 facilitates B-cell activation induced by CpG ODN. (A) B6 or DEC-205–/– mice were injected i.v. with 20 nmols of 1668 CpG ODN, and then 24 h later spleens were extracted and B cells analyzed for their expression of CD40, CD80, and MHC class II by flow cytometry. Representative histograms of three experiments are presented. (B) B cells (CD19+CD3–) from B6 and DEC-205–/– mice were purified by flow cytometry, incubated with 31.25 nM of 1668 CpG ODN for 16–18 h, and then assessed for their expression of CD40, CD86, and MHC class II. This experiment was performed three times, and representative histograms are shown. (C) The supernatants from the cultured B cells from (B) were collected, and IL-6 concentrations were measured by ELISA. Pooled data from two experiments are presented. Samples were run in duplicates or triplicates.
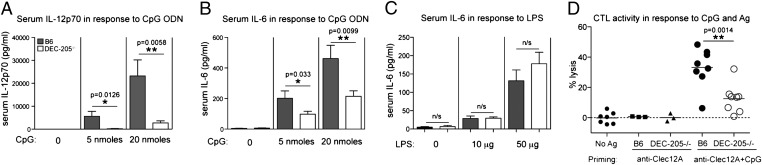
Because CpG ODN are well known for their capacity to induce cytokines and CD8+ DC are particularly efficient at producing IL-12p70 in response to CpG ODN (26), we asked whether DEC-205–/– mice were compromised in their ability to release a range of cytokines into the serum upon injection of CpG ODN. Bio-Plex cytokine array analysis showed that DEC-205–/– mice were significantly less effective at producing IL-12p70 and IL-6 compared with normal B6 mice (Fig. 5 A and B), consistent with an effect of DEC-205 deficiency on DC and B-cell activation in response to CpG ODN. Furthermore, the mutant mice were significantly less effective at producing IL-1α, TNF-α, MCP-1 MIP1α, and MIP1β (Fig. S4). However, DEC-205–/– mice were comparable to B6 mice in their ability to produce cytokines in response to LPS (Fig. 5C). We conclude that DEC-205 plays an important role in the specific recognition of CpG ODN for the production of a range of cytokines in vivo. Interestingly, when tested in vitro, purified DC captured significant amounts of CpG ODN independent of DEC-205 (Fig. S2A) and therefore produced IL-12 independent of this receptor (Fig. S2B). This suggests that in vitro isolation of DC enhances their DEC-205-independent binding of CpG ODN, perhaps by increasing pinocytosis or by revealing nonspecific receptors not available in vivo.
Fig. 5.
DEC-205 facilitates production of cytokines and the induction of CTL in response to CpG. B6 or DEC-205–/– mice were injected i.v. with (A and B) 5 or 20 nmols of 1668 CpG ODN or (C) graded doses of LPS. Plasma samples were taken 3 h later, and levels of (A) IL-12p70 and (B and C) IL-6 were measured by Bio-Plex Pro cytokine bead arrays. For CpG-treatment, the cumulative data of four experiments are presented (n = 8) as mean ± SEM. For LPS-treated mice, the cumulative data of two experiments are presented (n = 4) as mean ± SEM. Statistical significance was determined by a one-tailed unpaired Student t test. n/s, not significant. (D) B6 or DEC-205–/– mice were injected i.v. with 1 μg of anti-Clec12A-OVA mAb in the presence or absence of 5 nmols of CpG (1668). Six days later, mice were injected with OVA-peptide-coated target cells, and killing was measured 18 h later. The experiment was performed twice, and each symbol represents the lytic activity of an individual mouse.
Given the impaired function of CD8+ DC in DEC-205–/– mice in response to CpG ODN, we questioned whether this would translate to ineffective priming of T cells in vivo. To examine this question, mice were immunized with a CpG ODN together with a monoclonal antibody specific for Clec12A linked to ovalbumin (anti-Clec12A-OVA), an approach known to be dependent on adjuvant for CTL priming (27,28). This revealed that DEC-205–/– were significantly impaired in their ability to generate a CTL response compared with their B6 counterparts (Fig. 5D). These findings underscore the importance of DEC-205 in optimal responses to CpG ODN.
Discussion
Unmethylated cytosine-guanosine (CpG) motifs prevalent in prokaryotic DNA are recognized by TLR9 and serve to alert the immune system to the presence of microbial invaders. The immunostimulatory capacity of bacterial DNA is mimicked by synthetic ODN, which are either fully or partially modified with phosphorothioated backbones to confer protection against nuclease degradation and extend their in vivo half-life (29). Although A-, B-, and C-ODN have been used in mice to elicit immunity, it is predominantly the B-ODN that have been taken into human clinical trials. Here, we reveal a role for DEC-205 in the uptake of B-ODN and provide evidence that DEC-205 is required for the optimal adjuvancy effects of B-ODN.
For cells to respond to injected CpG ODN via intracellular TLR9, they need to internalize the DNA and shuttle it to the intracellular organelle that contains TLR9. Although there is evidence to suggest receptors may be involved in the uptake of CpG ODN (13), it remains unclear if different cell types use different receptors or if multiple receptors contribute to binding by a single cell type. Recently, NK cells were shown to be capable of using several KIR, including KIR3DL2, to capture and deliver CpG ODN to TLR9 in vitro (14). Similarly, CD14 was shown to bind nucleic acid and shuttle it to TLR9 in macrophages (15). The importance of CD14 in responding to CpG ODN in vivo was shown at the level of cytokine production, with CD14–/– mice showing a decrease in their ability to produce IL-6, IL-8, and IL-1β. However, direct uptake via CD14, which is expressed at low levels by DC, was not shown in vivo, questioning the precise mechanism by which this molecule influences CpG ODN uptake. Whether the observations for murine CD14 will also extend to human CD14 is untested. Thus, although other receptors have been implicated in CpG ODN binding, their influence in vivo, their function in humans, and their precise actions in the critical cell types for adaptive immunity, namely DC and B cells, have yet to be clarified.
Here we identified DEC-205 as a critical receptor involved in the capture of CpG ODN in vivo. DEC-205 directly bound CpG ODN and facilitated uptake by CD8+ DC, the key producers of IL-12. In the absence of DEC-205, DC showed impaired maturation in response to CpG ODN, diminished production of IL-12, and poor induction of CTL. Importantly, DEC-205 is well conserved between species, and human DEC-205 retained the capacity to bind CpG ODN. Interestingly, human DEC-205 had a higher binding capacity for the 2006 ODN, correlating with the ability of this CpG ODN to optimally stimulate human cells (5). As the empirical selection of effective CpG ODN in the human coincides with an optimal DEC-205 binding motif, it is feasible that more efficient CpG ODN sequences might be scientifically selected based on their capacity to optimally bind DEC-205. Because, in humans, B cells and pDC express TLR9 (30,31) and both of these cell types express DEC-205 (32,33), characterization of the ideal sequence required to bind DEC-205 coupled to the ideal sequence required to activate TLR9 sets the scene for the rational design of maximally stimulatory ODN.
Materials and Methods
Mice.
C57BL/6J wehi (B6), DEC-205–/–, (back-crossed 12 generations onto B6), and TLR9–/– [kindly made available by S. Akira (Osaka University, Osaka, Japan)] were bred under specific pathogen-free conditions at the Walter and Eliza Hall Institute. Sex- and age-matched mice (6–12 wk) were used and handled according to the guidelines of the National Health and Medical Research Council of Australia. Experimental procedures were approved by the Animal Ethics Committee of the Melbourne Health Research Directorate.
Reagents.
The following fully phosphorothioated 3′ biotinylated ODN were purchased from Geneworks: 1668 CpG ODN: 5′TCC ATG ACG TTC CTG ATG CT; 1668 No CpG ODN: 5′TCC ATG AGC TTC CTG ATG CT; 1826 CpG ODN: 5′ TCC ATG ACG TTC CTG ACG TT; 1826 No CpG ODN: 5′ TCC ATG AGC TTC CTG ACG TT; 2006 CpG ODN: 5′ TCG TCG TTT TGT CGT TTT GTC GTT; 2006 No CpG ODN: 5′ TGC TGC TTT TGT GCT TTT GTG CTT; and 2395 CpG ODN: 5′ TCG TCG TTT TCG GCG CGC GCC G. The A-ODN 2216 was only phosophorothioated at the sites indicated by an asterisk: 5′G*G*GGGACGATCGTC*G*G*G*G*G*G. For in vitro and in vivo experiments, 1668 CpG was also synthesized without the 3′ biotin tag or with a 5′ Cy3-tag. 1668 CpG ODN-FITC was purchased from Sigma.
Cloning of DEC-205.
The ectodomains of mouse DEC-205 (CD205; NP_038853.2; 1-MRT ... PLS-1665) and human DEC-205 (CD205; NP_002340; 1-MRT ... PLG-1664) were synthesized by GeneArt, in frame with a C-terminal FLAG tag and a biotinylation consensus sequence (34), and subcloned into pcDNA3.1+ expression vector (Invitrogen). Control FLAG-tagged ectodomains of three other mouse C-type lectins were also generated: CIRE (mouse-DC-SIGN) (35), Clec12A (28), and Clec9A (36). Recombinant soluble ectodomains were expressed in mammalian cells (FreeStyle 293F cells; Invitrogen). Secreted proteins were purified from media by affinity chromatography using an anti-FLAG M2 agarose resin and were further purified by size-exclusion chromatography using a prepacked Superose 6 column. Full-length mouse DEC-205 cDNA was expressed in CHO-K1 cells, and DEC-205 expressing CHO-K1 cells were sorted by flow cytometry.
Fluorescent Staining of DC and Other Cell Types.
Detailed information on mAb used and staining procedures have been described elsewhere (27,28). DC were isolated as described previously (37). Briefly, spleens were chopped, digested with collagenase and DNase at room temperature, and treated with EDTA. Low-density cells were obtained by density centrifugation (1.077 g/cm3 Nycodenz), and non-DC-lineage cells were then depleted. Depletion of non-DC was achieved by labeling these cells with mAb (KT3-1.1, anti-CD3; T24/31.7, anti-Thy1; TER119, anti-erythrocytes; RA36B2, anti-CD45R or 1D3 anti-CD19; and RB6-8C5, anti-Ly6C/G or 1A8, Ly6G) and then removing them using anti-rat Ig magnetic beads (Biomag beads; QIAGEN). CD8+ DC (CD11c+CD8+120G8–), CD8– DC (CD11c+CD8–120G8–), and pDC (CD11cint120G8+) were analyzed for their expression of CD40, CD80, CD86, and MHC class II. B cells (CD19+CD3–) were purified from splenocyte suspensions by flow cytometry and incubated with graded doses of 1668 CpG ODN in RPMI-1640 medium containing 10% (vol/vol) FCS, 100 U/mL penicillin, and 100 μg/mL streptomycin, 10–4 M 2-mercaptoethanol. To analyze the binding of CpG ODN to CHO-K1 cells expressing DEC-205, cell suspensions were prepared by treating adherent cells with 0.01 M EDTA. Cells were washed twice (3% FCS in RPMI) before being incubated on ice with 125 nM of 1668-CpG-ODN-FITC in 3% FCS/PBS. Bound CpG ODN was visualized by flow cytometry. Cell sorting was performed on a FACSDIVA (Becton Dickinson), FACSAria (Becton Dickinson), or MoFlo (Dako Cytomation) instrument. Analysis was performed on FACSCalibur or LSR II (Becton Dickinson) or FACScan (Becton Dickinson).
Cytokine Assays and in Vivo CTL Assay.
Mouse blood was collected in heparin. Plasma cytokines were measured using Bio-Plex Pro Mouse Group I 23-Plex Panel Assay (Bio-Rad) according to the manufacturer’s guidelines. The mouse cytokines and chemokines detected included: IL-1α,β; IL-2 to IL-6, IL-9, IL-10, IL-12p40, IL-12p70, IL-13, and IL-17A; Eotaxin; G-CSF; GM-CSF; IFN-γ; KC; MCP-1; MIP-1α, β, and RANTES; and TNF-α. IL-6 was also measured by ELISA using the eBioscience kit according to the manufacturer’s instructions. For the induction of in vivo CTL, mice were immunized with 1 μg of anti-Clec12A-OVA mAb in the presence or absence of 5 nmols of CpG (1668). Six days later, mice were injected with target cells, and in vivo killing was determined as described elsewhere (27,38).
Confocal Microscopy.
Mice were injected with 20 μg 1668 CpG ODN-Cy3. Spleens were extracted 30 min later and DC isolated. CD8+ DC were further purified by flow cytometry. DC were placed on anti-MHC class II–coated coverslips (1.5 h at 37 °C), fixed for 30 min with 4% paraformaldehyde, permeabilized with 0.3% Triton X-100, stained with biotinylated mAb against MHC class II and Streptavidin-Alexa488, counterstained with 5 μg/mL DAPI, and mounted using Prolong Gold Antifade Reagent (Invitrogen). Images were acquired on a LSM700 confocal microscope (Zeiss) and analyzed using the Imaris software (Bitplane).
ELISA for the Detection of DEC-205/CpG ODN Interaction.
ELISA plates (Costar) were coated overnight at 4 °C with 10 μg/mL of specified mAb. Unbound mAb was washed away (PBS, 0.05% Tween-20) before proteins (diluted in PBS/3% BSA) were added (4 °C, overnight). Captured protein was exposed to various biotinylated CpG ODN at specified concentrations (2 h, room temperature), detected with SA-HRP (2 h room temperature) (GE Healthcare) and colorimetric reaction visualized using ABTS (2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulphonic acid). Each titration point was performed in duplicate.
SPR.
Binding of DEC-205 1668 CpG ODN was assessed using a Biacore 3000. Approximately 50 RU (response units) of 3′-biotinylated CpG was immobilized on to streptavidin-coated SA sensor chips (GE Healthcare) according to the manufacturer’s instructions. Free streptavidin sites were blocked using biotin. To detect interactions, different concentrations (0.5, 1, 5, 10, 50, 100, 500, and 1,000 nM) of DEC-205 were injected for 2,500 s using a flow rate of 2 μL/min. Specific binding to CpG was calculated by subtracting the response from that of an empty flow cell (biotin-blocked streptavidin). Following DEC-205 binding, the surface was regenerated by injecting 20 μL of 0.1% SDS, 3 mM EDTA at a flow rate of 80 μL/min. Sensorgram data were plotted using BIAevaluation software and exported to Prism for further analysis.
Statistical Analysis.
Unpaired t test was performed on the data. The significance of differences is listed as follows: not significant (n/s); P > 0.05; *P < 0.05; **P < 0.01; ***P < 0.001. Analysis was performed in Prism (GraphPad).
Supplementary Material
Acknowledgments
We thank Belinda Phipson for her advice on statistical analysis. This work was supported by the National Health and Medical Research Council of Australia (NHMRC) Program Grants 454465 (to K.S. and W.R.H.) and 461269 (to N.A.N. and J.-G.Z.); NHMRC Project Grants 575546 (to K.S., M.H.L., and I.C.), 1003355 (to W.R.H. and I.C.), 461254 (J.A.V.), 461255 (to J.A.V.), and 637362 (to J.A.V.); and Australian Research Council Federation Fellowship (to W.R.H.). S.M. was supported by the Swiss National Science Foundation, and J.D.M. and L.C.S. are the recipients of NHMRC Career Development Awards. This work was made possible through Victorian State Government Operational Infrastructure Support and Australian Government NHMRC Independent Research Institute Infrastructure Support Scheme.
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1208796109/-/DCSupplemental.
*This Direct Submission article had a prearranged editor.
References
- 1.Carbone FR, Belz GT, Heath WR. Transfer of antigen between migrating and lymph node-resident DCs in peripheral T-cell tolerance and immunity. Trends Immunol. 2004;25:655–658. doi: 10.1016/j.it.2004.09.013. [DOI] [PubMed] [Google Scholar]
- 2.Joffre O, Nolte MA, Spörri R, Reis e Sousa C. Inflammatory signals in dendritic cell activation and the induction of adaptive immunity. Immunol Rev. 2009;227:234–247. doi: 10.1111/j.1600-065X.2008.00718.x. [DOI] [PubMed] [Google Scholar]
- 3.Kumar H, Kawai T, Akira S. Pathogen recognition by the innate immune system. Int Rev Immunol. 2011;30:16–34. doi: 10.3109/08830185.2010.529976. [DOI] [PubMed] [Google Scholar]
- 4.Wagner H. The sweetness of the DNA backbone drives Toll-like receptor 9. Curr Opin Immunol. 2008;20:396–400. doi: 10.1016/j.coi.2008.06.013. [DOI] [PubMed] [Google Scholar]
- 5.Bode C, Zhao G, Steinhagen F, Kinjo T, Klinman DM. CpG DNA as a vaccine adjuvant. Expert Rev Vaccines. 2011;10:499–511. doi: 10.1586/erv.10.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Krieg AM. Therapeutic potential of Toll-like receptor 9 activation. Nat Rev Drug Discov. 2006;5:471–484. doi: 10.1038/nrd2059. [DOI] [PubMed] [Google Scholar]
- 7.Guiducci C, et al. Properties regulating the nature of the plasmacytoid dendritic cell response to Toll-like receptor 9 activation. J Exp Med. 2006;203:1999–2008. doi: 10.1084/jem.20060401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sester DP, Naik S, Beasley SJ, Hume DA, Stacey KJ. Phosphorothioate backbone modification modulates macrophage activation by CpG DNA. J Immunol. 2000;165:4165–4173. doi: 10.4049/jimmunol.165.8.4165. [DOI] [PubMed] [Google Scholar]
- 9.Roberts TL, Dunn JA, Sweet MJ, Hume DA, Stacey KJ. The immunostimulatory activity of phosphorothioate CpG oligonucleotides is affected by distal sequence changes. Mol Immunol. 2011;48:1027–1034. doi: 10.1016/j.molimm.2011.01.011. [DOI] [PubMed] [Google Scholar]
- 10.Haas T, et al. The DNA sugar backbone 2ʹ deoxyribose determines toll-like receptor 9 activation. Immunity. 2008;28:315–323. doi: 10.1016/j.immuni.2008.01.013. [DOI] [PubMed] [Google Scholar]
- 11.Roberts TL, Sweet MJ, Hume DA, Stacey KJ. Cutting edge: species-specific TLR9-mediated recognition of CpG and non-CpG phosphorothioate-modified oligonucleotides. J Immunol. 2005;174:605–608. doi: 10.4049/jimmunol.174.2.605. [DOI] [PubMed] [Google Scholar]
- 12.Barber GN. Innate immune DNA sensing pathways: STING, AIMII and the regulation of interferon production and inflammatory responses. Curr Opin Immunol. 2011;23:10–20. doi: 10.1016/j.coi.2010.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Häcker H, et al. CpG-DNA-specific activation of antigen-presenting cells requires stress kinase activity and is preceded by non-specific endocytosis and endosomal maturation. EMBO J. 1998;17:6230–6240. doi: 10.1093/emboj/17.21.6230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sivori S, et al. Natural killer cells expressing the KIR2DS1-activating receptor efficiently kill T-cell blasts and dendritic cells: implications in haploidentical HSCT. Blood. 2011;117:4284–4292. doi: 10.1182/blood-2010-10-316125. [DOI] [PubMed] [Google Scholar]
- 15.Baumann CL, et al. CD14 is a coreceptor of Toll-like receptors 7 and 9. J Exp Med. 2010;207:2689–2701. doi: 10.1084/jem.20101111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jiang W, et al. The receptor DEC-205 expressed by dendritic cells and thymic epithelial cells is involved in antigen processing. Nature. 1995;375:151–155. doi: 10.1038/375151a0. [DOI] [PubMed] [Google Scholar]
- 17.East L, Isacke CM. The mannose receptor family. Biochim Biophys Acta. 2002;1572:364–386. doi: 10.1016/s0304-4165(02)00319-7. [DOI] [PubMed] [Google Scholar]
- 18.Inaba K, et al. Tissue distribution of the DEC-205 protein that is detected by the monoclonal antibody NLDC-145. I. Expression on dendritic cells and other subsets of mouse leukocytes. Cell Immunol. 1995;163:148–156. doi: 10.1006/cimm.1995.1109. [DOI] [PubMed] [Google Scholar]
- 19.Mahnke K, et al. The dendritic cell receptor for endocytosis, DEC-205, can recycle and enhance antigen presentation via major histocompatibility complex class II–positive lysosomal compartments. J Cell Biol. 2000;151:673–684. doi: 10.1083/jcb.151.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bonifaz L, et al. Efficient targeting of protein antigen to the dendritic cell receptor DEC-205 in the steady state leads to antigen presentation on major histocompatibility complex class I products and peripheral CD8+ T cell tolerance. J Exp Med. 2002;196:1627–1638. doi: 10.1084/jem.20021598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shrimpton RE, et al. CD205 (DEC-205): a recognition receptor for apoptotic and necrotic self. Mol Immunol. 2009;46:1229–1239. doi: 10.1016/j.molimm.2008.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang SS, et al. Plasminogen activator Pla of Yersinia pestis utilizes murine DEC-205 (CD205) as a receptor to promote dissemination. J Biol Chem. 2008;283:31511–31521. doi: 10.1074/jbc.M804646200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blazar BR, Krieg AM, Taylor PA. Synthetic unmethylated cytosine-phosphate-guanosine oligodeoxynucleotides are potent stimulators of antileukemia responses in naive and bone marrow transplant recipients. Blood. 2001;98:1217–1225. doi: 10.1182/blood.v98.4.1217. [DOI] [PubMed] [Google Scholar]
- 24.Kamphorst AO, Guermonprez P, Dudziak D, Nussenzweig MC. Route of antigen uptake differentially impacts presentation by dendritic cells and activated monocytes. J Immunol. 2010;185:3426–3435. doi: 10.4049/jimmunol.1001205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Watts C. Capture and processing of exogenous antigens for presentation on MHC molecules. Annu Rev Immunol. 1997;15:821–850. doi: 10.1146/annurev.immunol.15.1.821. [DOI] [PubMed] [Google Scholar]
- 26.Hochrein H, et al. Differential production of IL-12, IFN-alpha, and IFN-gamma by mouse dendritic cell subsets. J Immunol. 2001;166:5448–5455. doi: 10.4049/jimmunol.166.9.5448. [DOI] [PubMed] [Google Scholar]
- 27.Lahoud MH, et al. Targeting antigen to mouse dendritic cells via Clec9A induces potent CD4 T cell responses biased toward a follicular helper phenotype. J Immunol. 2011;187:842–850. doi: 10.4049/jimmunol.1101176. [DOI] [PubMed] [Google Scholar]
- 28.Lahoud MH, et al. The C-type lectin Clec12A present on mouse and human dendritic cells can serve as a target for antigen delivery and enhancement of antibody responses. J Immunol. 2009;182:7587–7594. doi: 10.4049/jimmunol.0900464. [DOI] [PubMed] [Google Scholar]
- 29.Sands H, et al. Biodistribution and metabolism of internally 3H-labeled oligonucleotides. I. Comparison of a phosphodiester and a phosphorothioate. Mol Pharmacol. 1994;45:932–943. [PubMed] [Google Scholar]
- 30.Bernasconi NL, Onai N, Lanzavecchia A. A role for Toll-like receptors in acquired immunity: Up-regulation of TLR9 by BCR triggering in naive B cells and constitutive expression in memory B cells. Blood. 2003;101:4500–4504. doi: 10.1182/blood-2002-11-3569. [DOI] [PubMed] [Google Scholar]
- 31.Hornung V, et al. Quantitative expression of toll-like receptor 1-10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides. J Immunol. 2002;168:4531–4537. doi: 10.4049/jimmunol.168.9.4531. [DOI] [PubMed] [Google Scholar]
- 32.Kato M, et al. Expression of human DEC-205 (CD205) multilectin receptor on leukocytes. Int Immunol. 2006;18:857–869. doi: 10.1093/intimm/dxl022. [DOI] [PubMed] [Google Scholar]
- 33.Tel J, et al. DEC-205 mediates antigen uptake and presentation by both resting and activated human plasmacytoid dendritic cells. Eur J Immunol. 2011;41:1014–1023. doi: 10.1002/eji.201040790. [DOI] [PubMed] [Google Scholar]
- 34.Brown MH, et al. 2B4, the natural killer and T cell immunoglobulin superfamily surface protein, is a ligand for CD48. J Exp Med. 1998;188:2083–2090. doi: 10.1084/jem.188.11.2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Caminschi I, et al. Molecular cloning of a C-type lectin superfamily protein differentially expressed by CD8alpha(-) splenic dendritic cells. Mol Immunol. 2001;38:365–373. doi: 10.1016/s0161-5890(01)00067-0. [DOI] [PubMed] [Google Scholar]
- 36.Caminschi I, et al. The dendritic cell subtype-restricted C-type lectin Clec9A is a target for vaccine enhancement. Blood. 2008;112:3264–3273. doi: 10.1182/blood-2008-05-155176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vremec D, et al. Production of interferons by dendritic cells, plasmacytoid cells, natural killer cells, and interferon-producing killer dendritic cells. Blood. 2007;109:1165–1173. doi: 10.1182/blood-2006-05-015354. [DOI] [PubMed] [Google Scholar]
- 38.White PJ, et al. Generic construction of single component particles that elicit humoural and cellular immune responses without the need for adjuvants. Vaccine. 2008;26:6824–6831. doi: 10.1016/j.vaccine.2008.09.087. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.