Abstract
Young, developing fruits of nasturtium (Tropaeolum majus L.) accumulate large deposits of nonfucosylated xyloglucan (XG) in periplasmic spaces of cotyledon cells. This “storage” XG can be fucosylated by a nasturtium transferase in vitro, but this does not happen in vivo, even as a transitory signal for secretion. The only XG that is clearly fucosylated in these fruits is the structural fraction (approximately 1% total) that is bound to cellulose in growing primary walls. The two fucosylated subunits that are formed in vitro are identical to those found in structural XG in vivo. The yield of XG-fucosyltransferase activity from membrane fractions is highest per unit fresh weight in the youngest fruits, especially in dissected cotyledons, but declines when storage XG is forming. A block appears to develop in the secretory machinery of young cotyledon cells between sites that galactosylate and those that fucosylate nascent XG. After extensive galactosylation, XG traffic is diverted to the periplasm without fucosylation. The primary walls buried beneath accretions of storage XG eventually swell and lose cohesion, probably because they continue to extend without incorporating components such as fucosylated XG that are needed to maintain wall integrity.
XG that accumulates in cotyledons of developing nasturtium (Tropaeolum majus L.) seeds as a temporary “storage” polysaccharide (NXG) differs from primary wall “structural” XG of most dicots in three major respects: (a) NXG is deposited in massive amounts (up to 20% of seed dry weight) in periplasmic spaces between plasma membranes and primary walls, i.e. in apposition to the wall (Hoth et al., 1986; Hoth and Franz, 1986; Ruel et al., 1990). It is not mobilized until about 8 d after germination (Edwards et al., 1985) in an auxin-dependent event (Hensel et al., 1991). (b) NXG is readily extracted with hot water (Hsu and Reeves, 1967; Hoth et al., 1986) or dilute alkali (Edwards et al., 1985; Hensel et al., 1991), whereas wall XG is so well integrated between and even into the cellulose framework (Hayashi, 1989; Carpita and Gibeaut, 1993; Edelmann and Fry, 1992) that the microfibril:XG complex must be swollen and hydrogen bonds broken (e.g. by 24% KOH) before this bound XG will dissolve. (c) NXG contains Glc, Xyl, and Gal in a molar ratio of 4:3:1.7, but no trace of Fuc, as determined by sensitive analyses using high-performance anion-exchange chromatography and PAD (Fanutti et al., 1996; Faik et al., 1997a).
The typical structural XG in primary walls contains Fuc and less Gal than storage XG. Nevertheless, Gal is an essential part of wall XG because terminal Fuc residues are attached to it by an α-1,2 linkage in a three-sugar side chain. Such side chains facilitate XG binding to cellulose (Levy et al., 1991, 1997). Small amounts of Fuc have been detected in hydrolysates of nasturtium seed extracts, but it was not shown to derive from structural components of the wall (Ruel et al., 1990). If expanding nasturtium fruit cells also contain fucosylated XG in primary walls, cotyledons must be capable of synthesizing two forms of XG with quite different compositions and extracellular locations.
Recently, we detected (Faik et al., 1997b) XG-dependent fucosyltransferase activity in extracts of particulate membranes from developing nasturtium fruits. This raises the question of how or whether the great bulk of NXG avoids being fucosylated in vivo. There are several possible explanations. Assuming that XG:fucosyltransferase is localized and active in the Golgi toward the end of the secretory process, either in trans cisternae or secretory vesicles (Brummell et al., 1990) or in the trans Golgi network (Zhang and Staehelin, 1992; Driouich et al., 1993), it could be that two forms of XG are synthesized at the same time but in different Golgi compartments, with XG:fucosyltransferase confined to the site that leads to wall XG. It is also possible that structural and storage XG are formed at different times during cell expansion, or in separate cells or tissues, and that fucosyltransferase is active only when or if the wall is incorporating XG.
An alternative and more speculative explanation is that newly synthesized NXG is fucosylated, but as a transitory decoration with Fuc cleaved from the polymer before or during the time it is deposited in periplasmic spaces. This would require the action of an α-fucosidase with the capacity to defucosylate XG. However, those plant α-fucosidases that have been studied to date, those in extracts of germinated nasturtium seeds and pea epicotyls (Farkas et al., 1991; Augur et al., 1993), are only able to hydrolyze Fuc from XG oligosaccharide when it is free in solution, not when it is combined as a subunit in intact XG. Moreover, there is no evidence that terminal fucosylation of XG is required as a signal for XG secretion; in fact, mutants of Arabidopsis that are unable to synthesize Fuc continue to incorporate normal levels of XG into cell walls (Reiter et al., 1993). Therefore, one aim of this study was to clarify how developing nasturtium fruits can harbor an active XG:fucosyltransferase and also generate large amounts of nonfucosylated storage XG.
With respect to the timing of the deposition of storage XG in relation to cotyledon growth, Hoth and Franz (1986) reported the first visible periplasmic deposits in electron micrographs of cells from developing nasturtium cotyledons at 23 d after anthesis. The cotyledons continue to grow rapidly while generating protein bodies, depositing NXG and greatly increasing dry weight (Hoth et al., 1986). The periplasmic deposits stain with the Thiery silver proteinate reagent for polysaccharide and with a polyclonal antibody to XG (Ruel et al., 1990). They also stain strongly with Coomassie brilliant blue (Hoth et al., 1986), indicating the presence of protein in these accretions. The light micrographs in the latter study show enough cotyledon cells to calculate statistically significant values for average cell size (cross-sectional area). Sizes increased between cells of sections observed before NXG deposition, those measured during deposition with some naked primary wall still visible, and those measured after heavy deposition with the entire periplasm filled with accretions, leaving no intercellular connections. Thus, the ratio of the relative sizes of tissue cells cut at 18, 26, and 35 d after anthesis was 1:1.6:2.5, respectively. Clearly, the deposition of periplasmic XG does not restrict substantial cell expansion, although it may erect a barrier to the incorporation of new wall materials.
In the present study HPLC and PAD were used to identify the XG subunits that are present in cellulase digests of NXG and nasturtium wall XG, and to compare the XG subunits that are fucosylated in vivo and in vitro. The point during fruit development at which NXG begins to be deposited was estimated from dry:fresh weight ratios and direct examination of electron micrographs. Levels of fucosyltransferase activity with or without added TXG were measured in detergent extracts of membranes from homogenates of whole fruits prepared before and after endogenous NXG began to be generated. The activity was also compared in extracts of excised cotyledons versus pericarp tissue. The results demonstrate that structural XG in primary walls of nasturtium fruits is fucosylated, and that the level of membrane-bound, XG-dependent fucosyltransferase activity declines when cotyledons form storage XG.
MATERIALS AND METHODS
Plant Materials and Chemicals
Dry nasturtium (Tropaeolum majus L. var Climbing Giants) seeds were purchased from W.H. Perron Ltd. (Montreal, Canada), washed for 10 min in 10% commercial bleach, soaked in water overnight, and germinated in moist vermiculite at room temperature. After about 1 month, seedlings were transferred to pots containing sand, Pro-Mix (Pharmacia), and black earth in a ratio of 2:1:1, plus lime (1 g L−1). Plants were grown in a growth chamber under 16 h of light (21°C) and 8 h of dark (18°C) with 70% RH throughout the day. Flowers were self-pollinated and the first fruits appeared about 10 d after anthesis. Three fruits often develop from one flower. The term “fruit” as used here refers to the seed with cotyledons enveloped by integument plus the outer pericarp, composed mainly of green, spongy parenchyma.
Individual fruits were harvested from their first appearance (<1 mm diameter, 50 mg fresh weight) to the time about 30 d after anthesis when they were fully grown (about 1.4 cm in diameter, 1 g fresh weight). They were weighed at daily intervals, surface sterilized for 5 min in 10% (v/v) commercial bleach, washed, and frozen for later use. Dry weight was measured after freeze drying. To obtain growth curves for developing fruits, the diameters of individual fruits were estimated daily with calipers at the widest point, and fresh and dry weights were calculated from standard curves obtained by relating diameters to weights. These weights were plotted versus age in days after anthesis, using 15 d as the reference point, which is when the fruits, including the seeds with integuments, began their most rapid period of expansion (Hoth et al., 1986).
The substrate for fucosyltransferase, GDP-l-[U-14C]Fuc, was purchased from New England Nuclear/DuPont (8.3 GBq mmol−1). The NXG provided as the potential Fuc acceptor was prepared from mature seeds by the method described by Edwards et al. (1985), except that extraction was in 2 n NaOH, 0.01% NaBH4 at room temperature without heat to avoid possible peeling reactions. TXG and partially purified cellulase from Trichoderma sp. were purchased from Megazyme International (Bray, Ireland). BSA, Chaps, DTT, leupeptin, Pipes buffer, PMSF, Pronase, tosyl Lys chloromethyl ketone, and tosyl Phe chloromethyl ketone were purchased from Sigma. CarboPac PA-100 columns for HPLC and the apparatus for PAD were from Dionex (Sunnyvale, CA). Sepharose CL-6B was from Pharmacia and Bio-Gel P2 was from Bio-Rad.
Solubilization of XG-Dependent Fucosyltransferase Activity
The method used to extract and assay fucosyltransferase activity from nasturtium fruit particulate membrane was a modification of the procedures described previously for enzyme from pea epicotyls (Hanna et al., 1991; Faik et al., 1997a). Frozen whole fruits or excised cotyledons and pericarp tissues were homogenized in an Osterizer blender (Sunbeam, Canada) (4 × 30 s at 1-min intervals) in 2 volumes of cold extraction buffer composed of 0.1 m Pipes-KOH, pH 6.8, 0.4 m Suc, 5 mm MgCl2, 5 mm MnCl2, 1 mm DTT, plus 10 μm leupeptin, 1 mm PMSF, 0.1 mm tosyl Lys chloromethyl ketone, and 0.1 mm tosyl Phe chloromethyl ketone. The resulting mixture was filtered through nylon cloth to remove cell wall debris, and the filtrate was centrifuged (model L8–80 centrifuge, Beckman) at high speed (100,000g) with an angle rotor (50 titanium) for 60 min at 4°C to obtain pellets containing total particulate membranes. Enzyme was solubilized from particulate pellets derived from 10 g fresh weight of whole fruits, or a minimum of 3 g fresh weight of cotyledons, by stirring at 4°C for 30 min in one-third volume of extraction buffer ± 0.3 to 0.4% (w/v) Chaps detergent, and removing insoluble material by recentrifugation (100,000g). In some experiments this was followed by a second extraction with Chaps. The detergent extracts were analyzed for fucosyltransferase activity, as described below. Protein concentrations were measured using an assay kit (Bio-Rad) with BSA as a standard.
Enzymic Assays
The basic assay medium for fucosyltransferase was composed of 0.1 m Hepes-KOH, pH 6.8, 25 mm MnCl2, 5 mm DTT, and 0.5 m Suc. Standard reaction mixtures (50 μL total volume) contained 20 μL of assay medium, 10 μL of GDP-[14C]Fuc (92.5 pmol, 85,000 dpm), 10 μL of 1% XG (100 μg of NXG or TXG), and 10 μL of enzyme (up to 10 μg of protein). The final concentration of substrate GDP-[14C]Fuc was 1.85 μm. Water replaced XG in controls. Reactions were initiated by the addition of enzyme and terminated after 30 min of incubation at room temperature. To avoid precipitation of insoluble salts of the labeled substrate, and to precipitate protein but not XG, reaction mixtures were terminated first by the addition of cold 10% TCA (for at least 1 h at 4°C) and centrifugation, followed by the addition of cold ethanol to the supernatant (final concentration 67%). The mixtures were chilled to −20°C and centrifuged to precipitate XG and other insoluble materials. The pellets were washed three times with 67% ethanol, redissolved/suspended in 200 μL of water, and radioactivity was determined by liquid-scintillation spectroscopy.
Oligosaccharide subunits of XG were prepared by digesting the reaction-mixture components that were insoluble in 67% ethanol with Trichoderma sp. cellulase (0.5 mg mL−1) in 50 mm sodium acetate buffer, pH 5.0, at 35°C for 16 h. The reaction was terminated by boiling. The products were fractionated on columns (1.1 × 126 cm) of Bio-Gel P2 with 0.01% NaN3 as an eluent. In this system, [14C]Fuc-labeled XG oligosaccharides eluted at the high-Mr end of the carbohydrate peak (Maclachlan et al., 1992).
α-Fucosidase activity was assayed versus TXG that had been fucosylated by incubation with GDP-[14C]Fuc in solubilized pea fucosyltransferase (Hanna et al., 1991), or versus a mixture of oligosaccharides generated from this labeled XG by partial hydrolysis with Trichoderma sp. cellulase. The assay procedure (Farkas et al., 1991) used paper chromatography to measure the initial rate of release of free [14C]Fuc from these substrates by various enzyme preparations.
Primary Wall XG
The buffer-insoluble debris that were retained by nylon filters from enzyme homogenates of developing nasturtium fruits were collected frozen from several experiments using a wide size range of fruits. The combined debris from 100 g fresh weight of fruits were stirred in approximately 5 volumes of 2 n NaOH, 0.01% NaBH4 at room temperature overnight. This was repeated four times to ensure that all traces of starch, NXG, and other unbound wall matrix materials had been dissolved (see also Edwards et al., 1985; Hanna et al., 1991).
The insoluble residue remaining from the debris after this scouring treatment was stirred overnight in 24% KOH and 0.1% NaBH4 to dissociate any residual cellulose:XG macromolecular complex. The mixture was centrifuged to remove cellulose and the primary cell wall XG was precipitated from the supernatant with 2 volumes of cold ethanol (Hayashi and Maclachlan, 1984). The precipitate was redissolved in hot water and reprecipitated as an insoluble XG:copper complex by adding Fehling's solutions (Rao, 1959). The blue pellet was resuspended in 0.5 m EDTA, pH 6.5, and the XG was precipitated with 2 volumes of ethanol. Washes were repeated until copper was completely eluted from the flocculent wall XG. Total carbohydrate was assayed with phenol sulfuric acid (Dubois et al., 1956) and XG specifically by the I-KI method of Kooiman (1960), using commercial TXG as a standard.
Analysis of Monosaccharides and Oligosaccharides
Oligosaccharides obtained from NXG or nasturtium cell wall XG, as described above, were concentrated by heating, and 5 to 10 μg was injected into a CarboPac PA-100 column attached to an apparatus for HPLC (Beckman). Carbohydrate was eluted with 30 mm sodium acetate in 0.1 m NaOH (degassed with helium). Elution profiles were recorded automatically using PAD. To determine the distribution of 14C in oligosaccharides purified from 10 enzymic reaction mixtures, fractions were collected manually as they eluted from the PAD apparatus at times that related to identifiable peaks. 14C was determined in neutralized fractions pooled from several injections until peaks containing several hundred disintegrations per minute were recovered for accurate scintillation spectrometry. Monosaccharides were prepared from XG or oligosaccharides by complete hydrolysis with 2 n trifluoroacetic acid in sealed tubes at 120°C for 1 h. The acid was removed by evaporation and aliquots (5 μg of carbohydrate) were analyzed by elution from a CarboPac PA-100 column and PAD with degassed 12 mm NaOH.
Electron Microscopy
Nasturtium seeds were collected at different stages of development, and sections cut from cotyledons with a razor were fixed for 2 h in 3.5% glutaraldehyde, 0.1 m phosphate buffer, pH 7.2. The tissue was postfixed in 1% OsO4 in the same buffer at 4°C for 1 h, dehydrated in cold 25% ethanol, followed by 50%, 75%, and 95% ethanol at room temperature, and embedded in Spurr's epoxy resin. Thin sections (700 Å) were cut with a microtome (Ultra Cut E, Reichert, Vienna, Austria), transferred to copper grids, and stained with 2% uranyl acetate (10 min) followed by lead citrate (20 min). Sections were viewed and photographed with an electron microscope (model EM 410, Philips, Eindhoven, The Netherlands).
RESULTS
Composition of Primary Cell Wall XG from Developing Nasturtium Fruits
Primary cell wall XG, as extracted from the filtered debris obtained from enzymic homogenates (Methods), yielded 150 mg of XG per 100 g of nasturtium fruit, which was similar to the value of 0.2% fresh weight recorded for primary wall XG extracted from pea epicotyls by a similar process (Hayashi and Maclachlan, 1984). The purified wall XG was hydrolyzed to subunits with Trichoderma sp. cellulase, fractionated through a Bio-Gel P2 column, and the oligosaccharide peak was analyzed by HPLC.
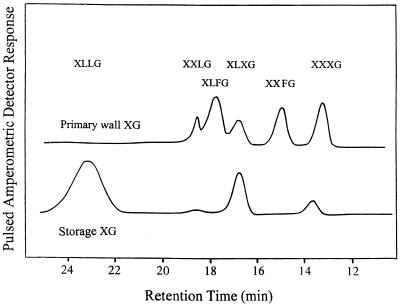
Figure 1 presents the PAD profiles obtained for oligosaccharide subunits, with elution positions from the CarboPac column of known XG oligosaccharides indicated by the abbreviated nomenclature proposed by Fry et al. (1993). The wall XG contained the following subunits in order of their elution, along with the ratio of recoveries (mol %) in parentheses: XXXG (24), XXFG (18), XLXG (17), XLFG (30), and XXLG (11). Fucosylated subunits made up about one-half of the XG components on a molar basis, which is typical of primary wall XG oligosaccharide PAD profiles reported for other dicots, e.g. Arabidopsis stem and apple fruit (Zablackis et al., 1995; Vincken et al., 1996).
Figure 1.
Profiles of oligosaccharide subunits produced by cellulase hydrolysis of NXG and primary cell wall XG from developing nasturtium fruits. NXG and wall XG were extracted sequentially using 2 n NaOH and 24% KOH, respectively, as described in Methods. XG was digested with Trichoderma sp. cellulase and the resulting oligosaccharides were recovered from peak fractions after gel filtration on Bio-Gel P2 columns. The two sets of oligosaccharides were then fractionated one after the other by HPLC through a CarboPac PA-100 column and assayed using PAD, with calibration by purified authentic XG subunits (Vincken et al., 1995; Faik et al., 1997a). Subunits are designated according to the nomenclature devised by Fry et al. (1993).
When nasturtium wall XG was totally hydrolyzed to monosaccharides and analyzed by HPLC to obtain PAD profiles, the only sugars detected were Glc, Xyl, Gal, and Fuc, with a ratio of values for relative mol % close to 4:3:1:0.5. This corresponds exactly to the monosaccharide mol %, which can be calculated from the relative concentrations and Mr values of oligosaccharide peaks in Figure 1.
XG-Dependent Fucosyltransferase Activity
Enzyme extracted with or without Chaps detergent from particulate membranes of young, developing nasturtium fruits was able to catalyze incorporation of label from GDP-[14C]Fuc into product(s) soluble in 10% TCA but insoluble in 67% ethanol if reaction mixtures contained added storage seed XG. As also observed with pea microsomal extracts (Hanna et al., 1991; Faik et al., 1997a), fucosyl transfer to insoluble products was distinctly greater when detergent was used in the extraction medium. This implies that at least some of the enzyme responsible was intrinsically bound to membrane and that availability of acceptor XG in these extracts was a limiting factor. We could not detect any α-fucosidase activity in supernatants or membrane extracts of fruit homogenates that could act on intact fucosylated XG. The assay method that was used readily measured XG nonasaccharide-dependent fucosidase activity in extracts of germinated nasturtium seeds (Farkas et al., 1991).
Incorporation of [14C]Fuc into an ethanol-insoluble product was greater in the presence of XG from tamarind than from nasturtium seed, presumably because of the differences in affinity of nasturtium fucosyltransferase for these two acceptors, i.e. differences in their subunit composition. This difference is most marked in the relative concentrations of octasaccharides, which could act as fucosyl acceptors: TXG contains mainly XXLG (galactosylated nearest the unsubstituted Glc), whereas XLXG predominates in NXG (Fanutti et al., 1996; Faik et al., 1997a; see also Fig. 1). XXFG is the major fucosylated nonasaccharide in pea cell wall XG (Hayashi and Maclachlan, 1984; Guillén et al., 1995), which must have derived from XXLG. Thus, nasturtium fucosyltransferase may prefer TXG over NXG as an acceptor because the former contains more of the octasaccharide needed to form XXFG.
Fucosylated Products
In preliminary experiments, reaction mixtures incubated in the absence of added storage XG and terminated without adding TCA generated relatively substantial amounts of 14C-labeled product that was insoluble in 67% ethanol. This was not labeled XG or protein but eluted near the total volume of effluent from a column of Sepharose CL-6B. It cofractionated without further treatment on columns of Bio-Gel P2 and CarboPac PA-100 with GDP-Fuc and sugar phosphate. It dissolved readily in 10% TCA, like XG, but in the presence of acid it was not precipitated by 67% ethanol. This product was apparently a charged derivative of [14C]Fuc that was dissociated by acid. It was probably a divalent salt of the substrate or a phosphorylated degradation product because these are not soluble in neutral 67% ethanol. In subsequent tests reaction mixtures were first acidified and precipitated with 10% TCA and then precipitated with 67% ethanol. This treatment reduced values for controls (without XG) to near zero by leaving salts of the substrate and derivatives in the acidic ethanol supernatant.
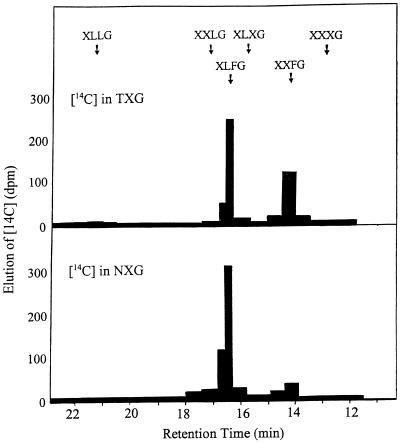
The 67% ethanol-insoluble 14C product formed by nasturtium fruit extract in the presence of TXG was dissolved in boiling water and fractionated with 0.1 n NaOH as a solvent on a column of Sepharose CL-6B. The 14C profile paralleled exactly the profile of TXG (phenol sulfuric acid assay), with a peak eluting at a size equivalent to dextran (>106 D). There was no shoulder corresponding to the peak of protein in these preparations, and the profile was not altered by incubation with protease (Pronase). However, the Sepharose peak was completely degraded by treatment with Trichoderma sp. cellulase to products that fractionated on columns of Bio-Gel P2 at the upper end of the peak of XG oligosaccharide subunits. Aliquots of these Bio-Gel P2 peaks were passed through a CarboPac PA-100 column for oligosaccharide and 14C analysis, as described in Methods. Figure 2 represents the disintegrations per minute recovered from TXG and NXG digests as a function of retention time in this chromatographic system. There were two peaks of 14C that corresponded to the known elution positions of fucosylated nonasaccharide (XXFG) and decasaccharide (XLFG). It is unlikely that these two products were fucosylated on a different Gal unit (e.g. XFXG and XFLG), because if such subunits existed, they would be expected to elute from Dionex columns at different times than authentic XXFG and XLFG, just as the two oligosaccharides XXLG and XLXG elute separately (Buckeridge et al., 1992; Vincken et al., 1995, 1996; Faik et al., 1997a).
Figure 2.
Incorporation of label from GDP-[14C]Fuc into XG subunits by fucosyltransferase from developing nasturtium fruits. 14C products insoluble in 67% ethanol that were formed by Chaps extract of particulate membrane in the presence of storage NXG or TXG were hydrolyzed by Trichoderma sp. cellulase to generate labeled oligosaccharides subsequently isolated by fractionation through a Bio-Gel P2 column. Individual oligosaccharides were resolved by HPLC through a CarboPac PA-100 column. Arrows indicate the relative retention times of subunit oligosaccharides in storage NXG and TXG compared with those in fucosylated structural wall XG. 14C was determined in fractions as they eluted from the PAD apparatus.
When NXG was the acceptor in these tests, the yield of labeled XXFG was much lower than the yield of XLFG (1:8, respectively; Fig. 2), as predicted above. This reflects the relative concentrations of precursor subunits in reserve NXG (XXLG ≪ XLLG; see Fig. 1). There was no sign of a third fucosylated product that might have formed from XLXG, the main octasaccharide in NXG. In TXG, however, the precursor octasaccharide for XXFG was predominant, and with this as an acceptor, the XXFG:XLFG ratio (2:3; Fig. 2) reflected a much higher yield of nonasaccharide. It is concluded that nasturtium XG fucosyltransferase preferentially transferred Fuc to the Gal residue closest to the unsubstituted Glc of the oligosaccharides XXLG or XLLG, and that the yields of nonasaccharide:decasaccharide depended on the relative availability of the appropriate precursor subunits in the XG acceptor.
XG Deposition during Nasturtium Fruit Development
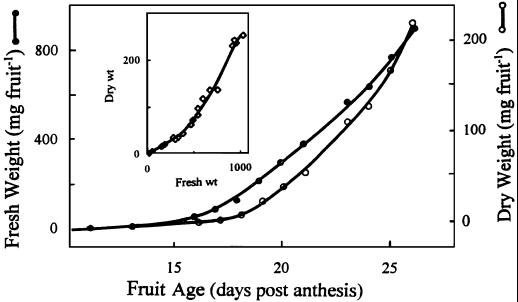
Figure 3 shows the fresh and dry weights of whole fruits measured at daily intervals from when they first emerged to when they abruptly stopped expanding and began senescing. A plot of dry versus fresh weight (inset) indicates a point about midway into the period of most rapid expansion when the slope increased, i.e. at approximately 500 mg fresh weight per fruit. This corresponds to 22 to 23 d after anthesis, and is attributable to relatively rapid increments of dry weight (storage reserves) after this time.
Figure 3.
Increases in fresh and dry weights of nasturtium fruits during development. In a plot of dry/fresh weight (inset), the slope increases at a time when fruits are about one-half of their full size, which is exactly when periplasmic XG and protein bodies begin to be deposited (see Fig. 4).
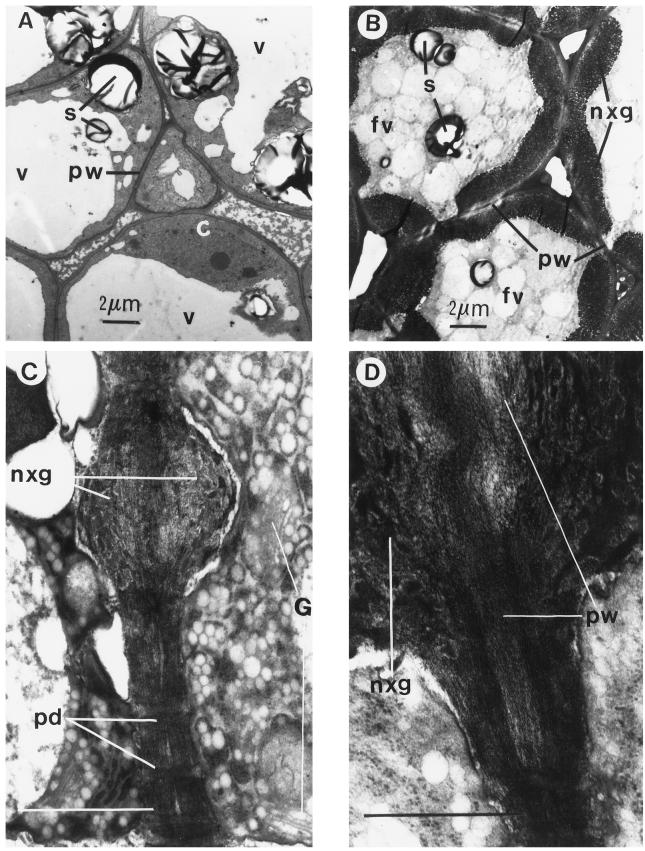
Electron micrographs of fruit sections that were approaching half-maximum size (400 μg fresh weight per fruit, 21 d after anthesis; Fig. 4A) show cells from the fleshy part of cotyledons that are fully vacuolated with typical primary walls but no periplasmic XG. Two days later (600 μg fresh weight per fruit; Fig. 4B), cells contained many fragmented vacuoles in the process of developing into protein bodies (see also Hoth et al., 1986), plus prodigious thickenings of electron-dense material between the plasma membrane and wall. At a higher magnification (Fig. 4, C and D), the Golgi and ER configurations were often seen near these periplasmic thickenings. Adjacent cells generally deposited periplasmic accretions on opposite sides of the primary wall (Fig. 4, B–D). Regions in which visible plasmodesmata traverse adjacent walls seemed to be the last to be invaded by the deposits. Periplasmic XG and protein-body formation took place exactly when the major increase in dry weight was observed (Fig. 3). A few days later, when cells were fully grown, the extracellular deposits were even more extensive, completely covering most intercellular connections, and preventing contact between any part of the plasma membrane and wall. The primary wall buried underneath the periplasmic thickenings tended to swell and lose definition (Fig. 4, B and D) compared with the typical compact and cohesive walls observed when they and the plasma membrane were in contact.
Figure 4.
Transmission electron micrographs of nasturtium cotyledon parenchyma examined when fruits were about half-maximum size (400–600 mg per fruit). Seeds were sectioned, fixed, postfixed in OsO4, embedded in Spurr's epoxy resin, and thin sections (700 Å) of cotyledon tissues were stained with uranium and lead as described in Methods. At 22 d after anthesis (A), no periplasmic deposits were detected, but by 24 d (B), deposits filled the spaces between the plasma membrane and the primary wall (periplasm) to a thickness many times that of the walls. Most deposits were first visible as accretions localized in adjacent cells on opposite sides of the wall (C). The walls under these deposits were swollen and contained microfibrils that were visibly dispersed (D). c, Cytoplasm; fv, fragmented vacuole; G, Golgi; nxg, periplasmic XG; pd, plasmodesmata; pw, primary wall; s, starch; and v, vacuole. Bars in C and D = 1 μm.
Fragmented vacuoles often contain electron-transparent crystals (see Fig. 4B) that are also visible in light micrographs as irregular clear deposits within Coomassie blue-stained protein bodies (Hoth et al., 1986). These inclusions are probably insoluble salts (e.g. Ca2+ phytate), which are commonly found in seeds as a storage component confined to protein bodies (Loewus, 1982; Bewley and Black, 1994). This is a potential source of divalent ions that precipitated the substrate GDP-[14C]Fuc, which was observed during incubations with particulate nasturtium extracts.
XG:Fucosyltransferase Activity during Nasturtium Fruit Development
XG-dependent fucosyltransferase activity recovered in two sequential detergent washes of particulate fractions from whole fruits was measured from the early growing period (16–22 d after anthesis), before periplasmic XG appeared in cotyledons, to the period of continued growth and the beginning of maturation, when the rate of periplasmic XG deposition was maximal (Figs. 3 and 4). The results (Table I) show that dry weight increased most markedly after the fruits reached an average fresh weight of about 400 g (22–24 d after anthesis). The recovery in both extracts of fucosyltransferase activity per unit fresh weight of fruit, assayed in the presence of added TXG, peaked before this time and then declined during the surge of NXG deposition.
Table I.
Yields of XG-dependent fucosyltransferase activity in detergent extracts of particulate membranes of developing nasturtium fruits
| Days after Anthesis | Weight
|
XG-Dependent
Fucosyltransferase Activity
|
||
|---|---|---|---|---|
| Fresh | Dry | 0.3% Chaps | 0.4% Chaps | |
| mg fruit−1 | dpm h−1 g−1 fresh wt | |||
| 16 | 48 | 5 | 12,100 | 8500 |
| 18 | 153 | 16 | 18,800 | 9400 |
| 22 | 416 | 43 | 13,700 | 8100 |
| 24 | 665 | 135 | 7,100 | 5300 |
| 27 | 927 | 185 | 6,800 | 3500 |
Whole fruits were harvested over a period corresponding to 16 to 27 d after anthesis, which encompassed the time when growth was most rapid and the age (21–23 d after anthesis, 400–600 g fresh weight per fruit; Figs. 3 and 4) when periplasmic XG began to be deposited in cotyledons. Particulate pellets prepared by centrifugation (100,000g) of filtered homogenates were extracted twice in sequence with 0.3% and 0.4% Chaps detergent as indicated, and the two solubilized extracts were assayed for fucosyltransferase activity in reaction medium with or without added TXG, as described in Methods. 14C was measured in products soluble in 10% TCA but insoluble in 67% ethanol. The effect of TCA was to dissociate insoluble salts of the labeled substrate and render them soluble in ethanol, thereby reducing artifactual 14C in controls to less than about 600 dpm h−1 g−1 fresh weight in all reactions.
Direct tests were carried out to determine whether any part of the relatively low levels of XG-dependent fucosyltransferase activity that were recovered from fruits during the last half of their development (Table I) was derived from cotyledons. Fruits were selected at sizes at which they had either not quite begun to deposit or were actively in the process of depositing NXG. Enveloping pericarps were separated manually from seeds, which were then dissected into integuments (discarded) and the remaining embryonic tissue (mainly cotyledons). Pericarps and cotyledons were weighed, homogenized, and particulate membrane fractions were extracted with detergent to produce separate enzyme preparations for comparison of XG-dependent fucosyltransferase activity. The results (Table II) show that extracts of cotyledons from 400-mg fruit were about 10 times richer in XG:fucosyltransferase activity on a fresh-weight basis than extracts of the much larger pericarp.
Table II.
Recovery of XG-dependent fucosyltransferase activity in detergent extracts of particulate membranes from excised nasturtium cotyledons and pericarp
| Fruits | Fresh Wt
|
XG-Dependent
Fucosyltransferase Activity
|
||
|---|---|---|---|---|
| Cotyledons | Pericarp | Cotyledons | Pericarp | |
| mg fruit−1a | dpm h−1 g−1 fresh wt | |||
| 400 | 13 | 364 | 33,100 | 3400 |
| 600 | 92 | 432 | 12,400 | 5100 |
| 800 | 153 | 518 | 4,300 | 1700 |
Whole fruits, weighing approximately 400, 600, or 800 mg each, were dissected on an iced tray to separate seeds minus integuments (mainly cotyledons) from the fleshy protective pericarp. These were weighed, homogenized, filtered, centrifuged, and extracted once with 0.4% Chaps, as described in Methods. Fucosyltransferase activity was assayed in the detergent extracts with or without added TXG and expressed per unit fresh weight.
The sum of values for cotyledons and pericarp do not add up to the initial weights of whole fruits because integuments (seed coats) were discarded.
Chromatography (Bio-Gel P2) confirmed that the product formed by cotyledons was entirely digested by cellulase to labeled XG oligosaccharide. When whole fruits increased from 400 to 600 and 800 mg fresh weight, the cotyledons expanded 7- and 12-fold, respectively. This must have depended in part on cell division in the meristem followed by cell expansion, because the increase observed in average cell size of the fleshy part of the cotyledons (see Hoth et al., 1986; Fig. 4, A and B) was not sufficient by itself to explain such a great increase in fresh weight. During these developments the cotyledons lost most of their XG:fucosyltransferase activity per unit fresh weight. As observed with whole fruits (Table I), there was no sign of any burst of this activity when NXG began or continued to be deposited, whether calculated per unit fresh weight or per fruit (Table II). By comparison, the pericarp tissue was nearing the end of its growing period during these tests, and fresh weight only increased by about 40%. XG:fucosyltransferase activity from the pericarp, calculated per unit fresh weight or per fruit, increased slightly and then declined.
DISCUSSION
Our results (Figs. 1–4) demonstrate that nasturtium fruits deposit two kinds of XG during their development to maturity: storage XG specifically into periplasmic spaces of cotyledon cells, and structural XG into growing primary walls. Structural XG is fucosylated in the subunits XLFG and XXFG, which make up 30 and 18 mol %, respectively, of all subunits; the others are heptasaccharide (XXXG) and two octasaccharides (XXLG and XLXG). The absence of digalactosylated nonasaccharide (XLLG) in this wall preparation is notable, for it indicates that there was no contamination of the extracted wall XG by storage XG, in which this is the most abundant subunit (Fig. 1). When the fruits were about half-maximum size, cotyledons suddenly began to deposit storage NXG, which contains relatively more Gal than wall XG but no Fuc. Detergent extracts of particulate membranes from whole nasturtium fruits had the enzymic capacity to fucosylate storage XG to form the same subunits that are found in structural XG. To our knowledge, this is the first published report that identifies XXFG and XLFG as the products formed in vitro by fucosyltransferase activity from any plant source. The level of this XG:fucosyltransferase activity per unit fresh weight is highest in the youngest nasturtium fruits (Table I), especially cotyledons (Table II) before they develop the capacity to form NXG. It then diminishes but does not disappear. Thus, the question is what function(s) the XG:fucosyltransferase activity may have in nasturtium cotyledons, which are especially known for their capacity to generate relatively large amounts of nonfucosylated storage XG.
Several observations in this study mitigate against any requirement for the transitory fucosylation of storage XG as a signal to facilitate its secretion. First, no Fuc was detected in subunits of NXG (Fig. 1). Second, there was no increase in XG:fucosyltransferase activity that coincided with the burst of NXG biosynthesis (Tables I and II; compare with Fig. 4). Third, NXG was a relatively ineffectual fucosyl acceptor for the nasturtium transferase, probably because this enzyme preferentially fucosylated the octasaccharide subunit XXLG (Fig. 2), which is only a minor constituent of NXG (Fig. 1). Finally, no α-fucosidase activity was detectable in nasturtium fruits, which could have hydrolyzed Fuc from an XG precursor to form NXG. It is concluded that nasturtium fruit XG:fucosyltransferase does not act to fucosylate, even temporarily, any XG that is destined for storage in periplasmic spaces.
If we assume, therefore, that XG:fucosyltransferase activity only functions to catalyze the last step of biosynthesis of XG that is destined for integration into the primary wall, there are two possible uses for the reduced levels of enzyme activity observed (Table II) after NXG starts to fill periplasmic spaces. Cotyledons increase in size at least 10-fold while this is happening (Table II), cell division must continue for a time, and primary walls must extend to keep pace. XG:fucosyltransferase would be expected to be active in new growing cells that coexist with maturing cells that deposit NXG. In maturing cells it is unlikely that any structural XG that might be formed could diffuse through the periplasmic accretions to reach the wall, although it could be directed to and secreted near the shrinking regions of the plasma membrane, where intercellular connections still remain. It is not surprising that these are the only parts of the primary wall that continue to show a compact form and borders that are as well defined as they were in earlier stages of growth. Walls buried under NXG swell, and it is difficult to discern their edges. Microfibrils visibly separate from one another (Fig. 4D) as if they were being pulled apart and are no longer tethered by sufficient structural XG or other matrix-binding agents that are required to maintain the integrity of the wall framework.
The basic problem for future research that is raised by this study is to understand the mechanism of how maturing cotyledon cells suddenly increase the rate of XG biosynthesis and channel almost all of it into secretion of NXG, effectively avoiding the final fucosylation step reserved for biosynthesis of structural XG. It appears that nascent NXG is processed in the secretory machinery only to the point at which it is well galactosylated, and then it exits in traffic directed toward quiescent regions of the plasma membrane, where periplasmic XG is secreted. NXG deposits particularly on all sides of intercellular spaces and on opposite sides of the primary walls of adjacent cells, where there are no plasmodesmata (Fig. 4). The factors that trigger and regulate this diversion are unknown. Perhaps maturing cotyledon cells develop a block between the steps that catalyze galactosylation of XG and terminal fucosylation, as is observed when the fungal antibiotic brefeldin A is added to actively secreting plant cells. Driouich et al. (1993) reported that suspension-cultured cells of sycamore react to brefeldin A by blocking transfer of XG via vesicles from the trans Golgi cisternae to the trans Golgi network, with the result that fucosylation is inhibited but XG biosynthesis is not, and truncated XG accumulates in secretory vesicles.
In any event, because nasturtium cotyledons do not extinguish the capacity to form wall XG when the biosynthesis of periplasmic XG becomes the predominant pathway, XG fucosylation may continue in a transition period in a few cells that are slow to differentiate or in a few strategically located Golgi configurations. It remains to be established whether storage and structural XG are ever assembled in the same Golgi at the same time.
ACKNOWLEDGMENT
We thank Kathy Hewitt (Electron Microscopy Centre, Biology Department, McGill University) for preparing the electron micrographs.
Abbreviations:
- Chaps
3-[(3-cholamidopropyl)-dimethylammonio]-1-propanesulfonate
- NXG and TXG
storage xyloglucans containing no Fuc derived from nasturtium and tamarind seed, respectively
- PAD
pulsed amperometric detection
- XG
xyloglucan. Oligosaccharide subunits of XG are abbreviated according to the nomenclature proposed by Fry et al. (1993)
Footnotes
This study was funded by the Natural Sciences and Engineering Research Council of Canada via a scholarship (to D.D.) and a research grant (to G.M.).
LITERATURE CITED
- Augur C, Benhamou N, Darvill A, Albersheim P. Purification, characterization and cell wall localization of an α-fucosidase that inactivates a xyloglucan oligosaccharin. Plant J. 1993;3:415–426. doi: 10.1046/j.1365-313x.1993.t01-24-00999.x. [DOI] [PubMed] [Google Scholar]
- Bewley DJ, Black M (1994) Seeds: Physiology of Development and Germination. Plenum Press, New York
- Brummell DA, Camirand A, Maclachlan G. Differential distribution of glycosyltransferase in pea Golgi dictyosomes and secretory vesicles. J Cell Sci. 1990;96:705–710. [Google Scholar]
- Buckeridge MS, Rochon DC, Reid JSG, Dietrich SMC. Xyloglucan structure and post-germination metabolism in seeds of Copaifera langsdorfii from savanna and forest populations. Plant Physiol. 1992;86:379–383. [Google Scholar]
- Carpita NC, Gibeaut DM. Structural models of primary cell walls in flowering plants: consistency of molecular structure with the physical properties of the walls during growth. Plant J. 1993;3:1–30. doi: 10.1111/j.1365-313x.1993.tb00007.x. [DOI] [PubMed] [Google Scholar]
- Driouich A, Zhang GF, Staehelin LA. Effect of brefeldin A on the structure of the Golgi apparatus and on the synthesis and secretion of proteins and polysaccharides in sycamore maple suspension-cultured cells. Plant Physiol. 1993;101:1363–1373. doi: 10.1104/pp.101.4.1363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugars and related substances. Anal Chem. 1956;28:350–356. [Google Scholar]
- Edelmann HG, Fry SC. Kinetics of integration of xyloglucan into the walls of suspension-cultured rose cells. J Exp Bot. 1992;43:463–470. [Google Scholar]
- Edwards M, Dea ICM, Bulpin PV, Reid JSG. XG (amyloid) mobilization in the cotyledons of Tropaeolum majus L. seeds following germination. Planta. 1985;163:133–140. doi: 10.1007/BF00395907. [DOI] [PubMed] [Google Scholar]
- Faik A, Chileshe C, Sterling J, Maclachlan G. Xyloglucan galactosyl- and fucosyltransferase activities from pea epicotyl microsomes. Plant Physiol. 1997a;114:245–254. doi: 10.1104/pp.114.1.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faik A, Desveaux D, Maclachlan G. Xyloglucan galactosyl- and fucosyltransferase activities in the cotyledons of developing nasturtium seeds (abstract no. 716) Plant Physiol. 1997b;114:S-150. [Google Scholar]
- Fanutti C, Gidley MJ, Reid JSG. Substrate subsite recognition of the xyloglucan endo-transglycosylase or xyloglucan-specific endo-(1–4)-β-d-glucanase from cotyledons of germinated nasturtium (Tropaeolum majus L.) seeds. Planta. 1996;200:221–228. doi: 10.1007/BF00208312. [DOI] [PubMed] [Google Scholar]
- Farkas V, Hanna R, Maclachlan G. Xyloglucan oligosaccharide α-l-fucosidase activity from growing pea stems and germinating nasturtium seeds. Phytochemistry. 1991;30:3203–3207. doi: 10.1016/0031-9422(91)83176-l. [DOI] [PubMed] [Google Scholar]
- Fry SC, York WS, Albersheim P, Darvill AG, Hayashi T, Joseleau J-P, Kato Y, Lorences EP, Maclachlan GA, McNeil M and others. An unambiguous nomenclature for xyloglucan-derived oligosaccharides. Physiol Plant. 1993;89:1–3. [Google Scholar]
- Guillén R, York WS, Pauly M, An J, Impallomeni G, Albersheim P, Darvill AG. Metabolism of xyloglucan generates xylose-deficient oligosaccharide subunits of this polysaccharide in etiolated peas. Carbohydr Res. 1995;277:291–311. doi: 10.1016/0008-6215(95)00220-n. [DOI] [PubMed] [Google Scholar]
- Hanna R, Brummell DA, Camirand A, Hensel A, Russell EF, Maclachlan G. Solubilization and properties of GDP-fucose:xyloglucan 1,2-α-l-fucosyltransferase from pea epicotyl membranes. Arch Biochem Biophys. 1991;290:7–13. doi: 10.1016/0003-9861(91)90584-6. [DOI] [PubMed] [Google Scholar]
- Hayashi T. Xyloglucans in the primary wall. Annu Rev Plant Physiol Plant Mol Biol. 1989;49:139–168. [Google Scholar]
- Hayashi T, Maclachlan G. Pea xyloglucan and cellulose. I. Macromolecular organization. Plant Physiol. 1984;75:596–604. doi: 10.1104/pp.75.3.596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hensel A, Brummel DA, Hanna R, Maclachlan G. Auxin dependent breakdown of xyloglucan in germinating nasturtium seeds. Planta. 1991;183:321–326. doi: 10.1007/BF00197728. [DOI] [PubMed] [Google Scholar]
- Hoth A, Blaschek W, Franz G. Xyloglucan (amyloid) formation in the cotyledons of Tropaeolum majus L. seeds. Plant Cell Rep. 1986;5:9–12. doi: 10.1007/BF00269706. [DOI] [PubMed] [Google Scholar]
- Hoth A, Franz G (1986) Xyloglucan (amyloid) biosynthesis in the cotyledons of Tropaeolum majus seeds. In B Vian, D Reis, R Goldberg, eds, Proceedings of the 4th Cell Wall Meeting, Paris. pp 132–137
- Hsu DS, Reeves RE. The structure of nasturtium amyloid. Carbohydr Res. 1967;5:202–209. [Google Scholar]
- Kooiman P. A method for determination of amyloid in plant seeds. Recl Trav Chim Pays-Bas Belg. 1960;79:675–678. [Google Scholar]
- Levy S, Maclachlan G, Staehelin LA. Xyloglucan sidechains modulate binding to cellulose during in vitro binding assays as predicted by conformational dynamics simulations. Plant J. 1997;11:101–114. doi: 10.1046/j.1365-313x.1997.11030373.x. [DOI] [PubMed] [Google Scholar]
- Levy S, York WS, Stuike-Prill R, Meyer B, Staehelin LA. Simulations of the static and dynamic molecular conformations of xyloglucan: the role of the fucosylated side chain in surface-specific side chain folding. Plant J. 1991;1:195–215. [PubMed] [Google Scholar]
- Loewus FA. Phytate metabolism with special reference to its myoinositol component. Recent Adv Phytochem. 1982;17:173–192. [Google Scholar]
- Maclachlan G, Levy B, Farkas V. Acceptor requirements for GDP-fucose:xyloglucan 1,2-α-l-fucosyltransferase activity solubilized from pea epicotyl membranes. Arch Biochem Biophys. 1992;294:200–205. doi: 10.1016/0003-9861(92)90158-s. [DOI] [PubMed] [Google Scholar]
- Rao PS (1959) Tamarind. In RL Whistler, ed, Industrial Gums. Academic Press, New York, pp 418–504
- Reiter W-D, Chapple CCS, Somerville CR. Altered growth and cell walls in a fucose-deficient mutant of Arabidopsis. Science. 1993;261:1032–1035. doi: 10.1126/science.261.5124.1032. [DOI] [PubMed] [Google Scholar]
- Ruel K, Joseleau J-P, Franz G. Aspects cytologiques de la formation des xyloglucanes dans les cotyledons des graines de Tropaeolum majus L. C R Acad Sci Paris. 1990;310:89–95. [Google Scholar]
- Vincken J-P, Beldman G, Neissen WMA, Voragen AGJ. Degradation of apple fruit xyloglucan by endoglucanase. Carbohydr Polym. 1996;29:75–85. [Google Scholar]
- Vincken J-P, deKeiser A, Beldman G, Voragen AGJ. Fractionation of xyloglucan fragments and their interaction with cellulose. Plant Physiol. 1995;108:1579–1585. doi: 10.1104/pp.108.4.1579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zablackis E, Huang J, Muller B, Darvill AG, Albersheim P. Characterization of the cell wall polysaccharides of Arabidopsis thaliana leaves. Plant Physiol. 1995;107:1129–1138. doi: 10.1104/pp.107.4.1129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang GF, Staehelin LA. Functional compartment of the Golgi apparatus of plant cells. Plant Physiol. 1992;99:1070–1083. doi: 10.1104/pp.99.3.1070. [DOI] [PMC free article] [PubMed] [Google Scholar]