Abstract
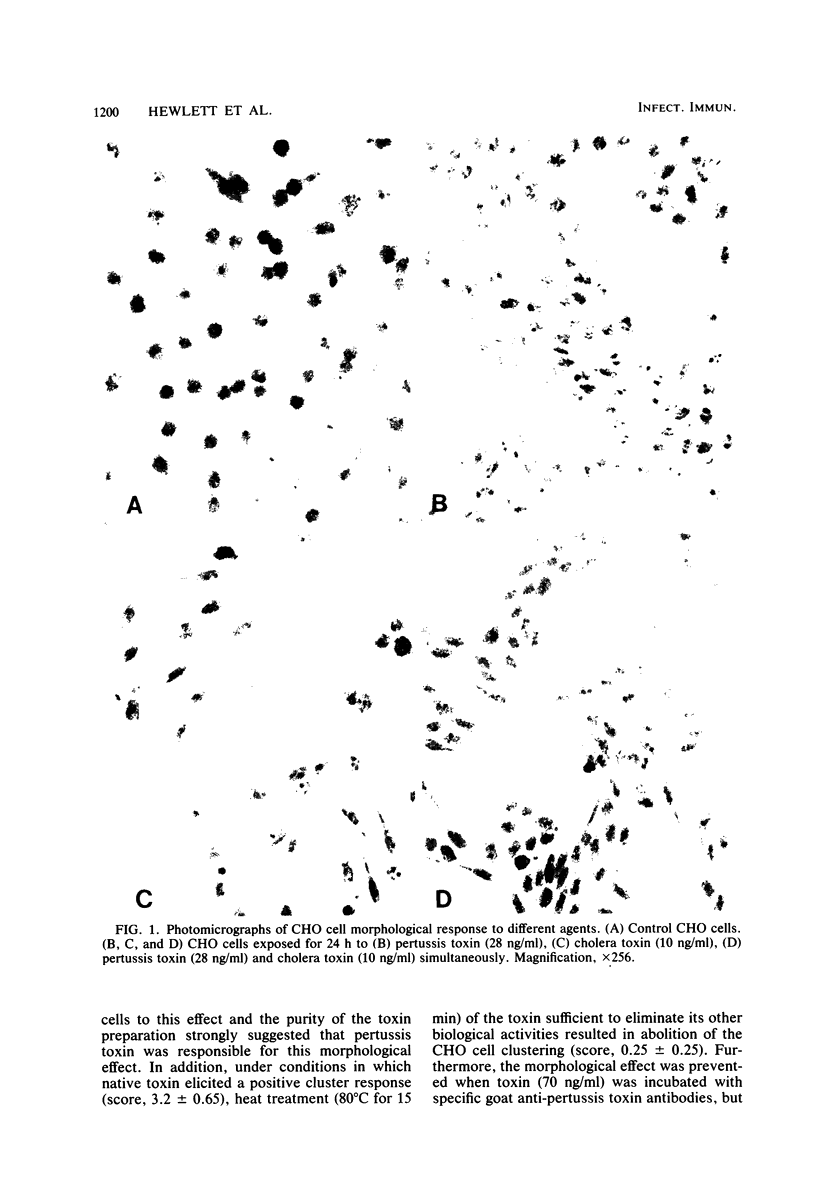
Exposing Chinese hamster ovary cells in culture to pertussis toxin resulted in a novel clustered growth pattern. The specificity of the response for pertussis toxin was shown by neutralization of the activity with specific anti-toxin antibody, heat lability (80 degrees C for 15 min), and absence of such activity by culture media from nontoxigenic Bordetella species. Although a lag of at least 16 h was required before clustered growth was seen, exposure to the toxin for as little as 10 min resulted in a full response 24 h later. The morphological effect appeared to be independent of the cyclic AMP-mediated cell elongation elicited by the heat-labile enterotoxin from Vibrio cholerae in that the pertussis toxin effect was seen in both the presence and absence of elongation. Although the mechanism by which this effect is mediated remains to be determined, it is already providing a useful in vitro assay for pertussis toxin.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Askelöf P., Gillenius P. Effect of lymphocytosis-promoting factor from Bordetella pertussis on cerebellar cyclic GMP levels. Infect Immun. 1982 Jun;36(3):958–961. doi: 10.1128/iai.36.3.958-961.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunson K. W., Nicolson G. L. Lipopolysaccharide effects on sensitive and resistant variant Chinese hamster ovary cell lines. J Supramol Struct. 1978;9(2):231–242. doi: 10.1002/jss.400090208. [DOI] [PubMed] [Google Scholar]
- Burns D. L., Hewlett E. L., Moss J., Vaughan M. Pertussis toxin inhibits enkephalin stimulation of GTPase of NG108-15 cells. J Biol Chem. 1983 Feb 10;258(3):1435–1438. [PubMed] [Google Scholar]
- DeBault L. E., Yoo T. J., Jensen K. L. Acute effects of Bordetella pertussis vaccine in vitro. J Reticuloendothel Soc. 1976 Feb;19(2):63–76. [PubMed] [Google Scholar]
- Endoh M., Soga M., Nakase Y. In vitro assay for histamine-sensitizing factor of Bordetella pertussis. Microbiol Immunol. 1980;24(9):887–890. doi: 10.1111/j.1348-0421.1980.tb02893.x. [DOI] [PubMed] [Google Scholar]
- Gill D. M., King C. A. The mechanism of action of cholera toxin in pigeon erythrocyte lysates. J Biol Chem. 1975 Aug 25;250(16):6424–6432. [PubMed] [Google Scholar]
- Guerrant R. L., Brunton L. L. Characterization of the Chinese hamster ovary cell assay for the enterotoxins of Vibrio cholerae and Escherichia coli and for specific antisera, and toxoid. J Infect Dis. 1977 May;135(5):720–728. doi: 10.1093/infdis/135.5.720. [DOI] [PubMed] [Google Scholar]
- Guerrant R. L., Brunton L. L., Schnaitman T. C., Rebhun L. I., Gilman A. G. Cyclic adenosine monophosphate and alteration of Chinese hamster ovary cell morphology: a rapid, sensitive in vitro assay for the enterotoxins of Vibrio cholerae and Escherichia coli. Infect Immun. 1974 Aug;10(2):320–327. doi: 10.1128/iai.10.2.320-327.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hazeki O., Ui M. Modification by islet-activating protein of receptor-mediated regulation of cyclic AMP accumulation in isolated rat heart cells. J Biol Chem. 1981 Mar 25;256(6):2856–2862. [PubMed] [Google Scholar]
- Hewlett E. L., Guerrant R. L., Evans D. J., Jr, Greenough W. B., 3rd Toxins of Vibrio cholerae and Escherichia coli stimulate adenyl cyclase in rat fat cells. Nature. 1974 May 24;249(455):371–373. doi: 10.1038/249371a0. [DOI] [PubMed] [Google Scholar]
- Hewlett E. L., Urban M. A., Manclark C. R., Wolff J. Extracytoplasmic adenylate cyclase of Bordetella pertussis. Proc Natl Acad Sci U S A. 1976 Jun;73(6):1926–1930. doi: 10.1073/pnas.73.6.1926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honda T., Shimizu M., Takeda Y., Miwatani T. Isolation of a factor causing morphological changes of chinese hamster ovary cells from the culture filtrate of Vibrio parahaemolyticus. Infect Immun. 1976 Oct;14(4):1028–1033. doi: 10.1128/iai.14.4.1028-1033.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsie A. W., Puck T. T. Morphological transformation of Chinese hamster cells by dibutyryl adenosine cyclic 3':5'-monophosphate and testosterone. Proc Natl Acad Sci U S A. 1971 Feb;68(2):358–361. doi: 10.1073/pnas.68.2.358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irons L. I., MacLennan A. P. Isolation of the lymphocytosis promoting factor-haemagglutinin of Bordetella pertussis by affinity chromatography. Biochim Biophys Acta. 1979 Sep 29;580(1):175–185. doi: 10.1016/0005-2795(79)90208-3. [DOI] [PubMed] [Google Scholar]
- KIND L. S. The altered reactivity of mice after inoculation with Bordetella pertussis vaccine. Bacteriol Rev. 1958 Sep;22(3):173–182. doi: 10.1128/br.22.3.173-182.1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katada T., Ui M. Direct modification of the membrane adenylate cyclase system by islet-activating protein due to ADP-ribosylation of a membrane protein. Proc Natl Acad Sci U S A. 1982 May;79(10):3129–3133. doi: 10.1073/pnas.79.10.3129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katada T., Ui M. In vitro effects of islet-activating protein on cultured rat pancreatic islets. Enhancement of insulin secretion, adenosine 3':5'-monophosphate accumulation and 45Ca flux. J Biochem. 1981 Apr;89(4):979–990. [PubMed] [Google Scholar]
- Katada T., Ui M. Islet-activating protein. A modifier of receptor-mediated regulation of rat islet adenylate cyclase. J Biol Chem. 1981 Aug 25;256(16):8310–8317. [PubMed] [Google Scholar]
- Katada T., Ui M. Slow interaction of islet-activating protein with pancreatic islets during primary culture to cause reversal of alpha-adrenergic inhibition of insulin secretion. J Biol Chem. 1980 Oct 25;255(20):9580–9588. [PubMed] [Google Scholar]
- Kong A. S., Morse S. I. The in vitro effects of Bordetella pertussis lymphocytosis-promoting factor on murine lymphocytes. I. Proliferative response. J Exp Med. 1977 Jan 1;145(1):151–162. doi: 10.1084/jem.145.1.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane A. G. Appearance of mouse-lethal toxin in liquid cultures of Bordetella pertussis. Appl Microbiol. 1968 Sep;16(9):1400–1405. doi: 10.1128/am.16.9.1400-1405.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merson M. H., Sack R. B., Kibriya A. K., Al-Mahmood A., Adamed Q. S., Huq I. Use of colony pools for diagnosis of enterotoxigenic Escherichia coli diarrhea. J Clin Microbiol. 1979 Apr;9(4):493–497. doi: 10.1128/jcm.9.4.493-497.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morse J. H., Morse S. I. Studies on the ultrastructure of Bordetella pertussis. I. Morphology, origin, and biological activity of structures present in the extracellular fluid of liquid cultures of Bordetella pertussis. J Exp Med. 1970 Jun 1;131(6):1342–1357. doi: 10.1084/jem.131.6.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morse S. I., Morse J. H. Isolation and properties of the leukocytosis- and lymphocytosis-promoting factor of Bordetella pertussis. J Exp Med. 1976 Jun 1;143(6):1483–1502. doi: 10.1084/jem.143.6.1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochiai T., Okumura K., Tada T., Iwasa S. Effect of lymphocytosis-promoting factor of Bordetella pertussis on the immune response. I. Suppression of cellular hypersensitivity reactions. Int Arch Allergy Appl Immunol. 1972;43(2):196–206. doi: 10.1159/000230837. [DOI] [PubMed] [Google Scholar]
- Parker C. D., Doyle S., Field L. H., Hewlett E. Variability in derivative strains of Bordetella pertussis. Dev Biol Stand. 1980;45:119–127. [PubMed] [Google Scholar]
- Pittman M. Pertussis toxin: the cause of the harmful effects and prolonged immunity of whooping cough. A hypothesis. Rev Infect Dis. 1979 May-Jun;1(3):401–412. doi: 10.1093/clinids/1.3.401. [DOI] [PubMed] [Google Scholar]
- Porter K., Prescott D., Frye J. Changes in surface morphology of Chinese hamster ovary cells during the cell cycle. J Cell Biol. 1973 Jun;57(3):815–836. doi: 10.1083/jcb.57.3.815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross R., Munoz J., Cameron C. Histamine-sensitizing factor, mouse-protective antigens, and other antigens of some members of the genus Bordetella. J Bacteriol. 1969 Jul;99(1):57–64. doi: 10.1128/jb.99.1.57-64.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudolph S. A., Schafer D. E., Greengard P. Effects of cholera enterotoxin on catecholamine-stimulated changes in cation fluxes, cell volume, and cyclic AMP levels in the turkey erythrocyte. J Biol Chem. 1977 Oct 25;252(20):7132–7139. [PubMed] [Google Scholar]
- Sandefur P. D., Peterson J. W. Neutralization of Salmonella toxin-induced elongation of Chinese hamster ovary cells by cholera antitoxin. Infect Immun. 1977 Mar;15(3):988–992. doi: 10.1128/iai.15.3.988-992.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato Y., Arai H., Suzuki K. Leukocytosis-promoting factor of Bordetella pertussis. II. Biological properties. Infect Immun. 1973 Jun;7(6):992–999. doi: 10.1128/iai.7.6.992-999.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato Y., Izumiya K., Sato H., Cowell J. L., Manclark C. R. Role of antibody to leukocytosis-promoting factor hemagglutinin and to filamentous hemagglutinin in immunity to pertussis. Infect Immun. 1981 Mar;31(3):1223–1231. doi: 10.1128/iai.31.3.1223-1231.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sumi T., Ui M. Potentiation of the adrenergic beta-receptor-mediated insulin secretion in pertussis-sensitized rats. Endocrinology. 1975 Aug;97(2):352–358. doi: 10.1210/endo-97-2-352. [DOI] [PubMed] [Google Scholar]
- Yajima M., Hosoda K., Kanbayashi Y., Nakamura T., Nogimori K., Mizushima Y., Nakase Y., Ui M. Islets-activating protein (IAP) in Bordetella pertussis that potentiates insulin secretory responses of rats. Purification and characterization. J Biochem. 1978 Jan;83(1):295–303. doi: 10.1093/oxfordjournals.jbchem.a131904. [DOI] [PubMed] [Google Scholar]
- Yajima M., Hosoda K., Kanbayashi Y., Nakamura T., Takahashi I., Ui M. Biological properties of islets-activating protein (IAP) purified from the culture medium of Bordetella pertussis. J Biochem. 1978 Jan;83(1):305–312. doi: 10.1093/oxfordjournals.jbchem.a131905. [DOI] [PubMed] [Google Scholar]




