Abstract
Congestive heart failure is associated with increased expression of pro-inflammatory cytokines, and the levels of these cytokines correlate with heart failure severity and prognosis. Chronic interleukin 6 (IL-6) stimulation leads to LV hypertrophy and dysfunction, and deletion of IL-6 reduces LV hypertrophy after angiotensin II infusion. In this study, we tested the hypothesis that IL-6 deletion has favorable effects on pressure-overloaded hearts. We performed transverse aortic constriction on IL-6-deleted (IL6KO) mice and C57BL/6J mice (CON) to induce pressure overload. Pressure overload was associated with similar LV hypertrophy, dilation, and dysfunction in CON and IL6KO mice. Re-activation of the fetal gene program was also similar in pressure-overloaded CON and IL6KO mice. There were no differences between CON and IL6KO mice in LV fibrosis or expression of extracellular matrix proteins after pressure overload. In addition, no group differences in apoptosis or autophagy were seen. These data indicate that IL-6 deletion does not block LV remodeling and dysfunction induced by pressure overload. Attenuated content of interleukin 11 appears to be a compensatory mechanism for IL-6 deletion in pressure-overloaded hearts. We infer from these data that limiting availability of IL-6 alone is not sufficient to attenuate LV remodeling and dysfunction in failing hearts.
Keywords: IL-6, inflammatory cytokine, LV function, hypertrophy, fibrosis, pressure overload, heart failure
INTRODUCTION
Congestive heart failure is an inexorable disease associated with high morbidity and mortality,1 despite advanced pharmaceutical and device therapies. Chronic pressure overload, as seen in persistent hypertension, is a major risk factor for heart failure.2 Although the underlying mechanisms are not fully understood, pro-inflammatory cytokines may be of mechanistic importance for left ventricular (LV) dysfunction and dilation.3
Serum levels of the pro-inflammatory interleukin 6 (IL-6) family cytokines [IL-6, leukemia inhibitory factor (LIF), ciliary neurotrophic factor (CNTF), oncostatin M, and cardiotrophin 1] are elevated in patients with heart failure regardless of etiology.4-10 Clinical and preclinical studies also show that increased levels of these pro-inflammatory IL-6 family cytokines correlate with high risk of developing LV hypertrophy, dilation, dysfunction, and fibrosis.4-6, 11-14 In addition, IL-6 perfusion directly inhibits contractility in isolated papillary muscle.15 Impaired contractility likely results from decreased SERCA2a expression after IL-6 stimulation.16 In vivo studies show that chronic IL-6 infusion is associated with LV hypertrophy, fibrosis, and dysfunction.17, 18 These observations indicate that increased IL-6 levels impair LV function. Finally, deletion of IL-6 reduces LV hypertrophy after angiotensin II infusion.19 Our hypothesis is that reduced IL-6 levels will decrease LV hypertrophy and improve function of the pressure-overloaded hearts. We induced pressure overload by transverse aortic constriction in a murine line with IL-6 deletion to test this hypothesis.
MATERIALS AND METHODS
Animals
This study was approved by Institutional Animal Care and Use Committee at the VA San Diego Healthcare System, in accordance with NIH and AAALAC guidelines. Male and female mice with germline IL-6 deletion (IL6KO; in congenic C57BL/6J background) were purchased from the Jackson Laboratories (Bar Harbor, ME). Two-month-old IL6KO mice were used for the study. Age and sex-matched wild type C57BL/6J mice were used as control (CON). Deletion of IL-6 gene was confirmed by PCR using mouse tail genomic DNA as template (Supplemental Material, Figure S1A). Absence of IL-6 mRNA expression and protein expression were further confirmed by quantitative real-time RT-PCR and enzyme-linked immunosorbent assay (ELISA), respectively (Supplemental Material, Figure S1).
Pressure Overload
Pressure overload was induced by transverse aortic constriction (TAC) as previously reported.20 Briefly, anesthetized mice were intubated and ventilated (pressure-controlled) with anesthesia maintained with 1% isoflurane in oxygen. The second intercostal space at the left upper sternal border was blunt dissected to expose the aortic arch. A 7-0 silk tie was applied against a 27-gauge needle between the innominate and left carotid artery. The needle was then removed, which leads to ~ 90% lumen narrowing. Surgery was performed without knowledge of group identity.
Echocardiography
Mouse anesthesia was induced with 5% isoflurane (at a flow rate of 1 L/min oxygen) and maintained with 1% isoflurane in oxygen. Echocardiographic images were obtained using a 16L MHz linear probe and Sonos 5500® Echocardiograph system (Philips Healthcare, Andover, MA). LV chamber dimensions and LV fractional shortening were acquired and analyzed without knowledge of group identity.
In Vivo Hemodynamics
A 1.4F conductance-micromanometer catheter (Millar Instruments) was inserted into the right carotid artery to access the LV cavity on anesthetized mice (80 mg/kg sodium pentobarbital, i.p.). LV pressure development (LV +dP/dt), relaxation (−dP/dt), and end-systolic pressure-volume relationship (ESPVR) were determined as previously described.20
Necropsy
Body and LV weight (including the septum), and tibial length were measured at necropsy. A short axis midwall LV ring was fixed in formalin; the remaining portion of LV was quickly frozen in liquid nitrogen and stored at −80°C.
Histology
Formalin-fixed LV samples were dehydrated and embedded in paraffin. Dewaxed LV sections (5 μm) were rehydrated and stained with hematoxylin and eosin. To measure collagen area, rehydrated LV sections were stained with picrosirius red. Fractional collagen area was calculated as percentage of picrosirius red-stained collagen area using NIH ImageJ software.
Quantitative RT-PCR
Total RNA was extracted, purified, and treated with RNase-free DNase to eliminate potential genomic DNA contamination. Quantitative real-time RT-PCR was conducted as previously described.20
Western Blotting
LV homogenates were used for Western blotting as previously described,20 using the following antibodies: Bcl2 (Cat #sc-7382, Santa Cruz Biotechnology, Santa Cruz, CA), α-smooth muscle actin (Cat #A5228, Sigma, St. Louis, MO), periostin (Cat #AF2955, R&D Systems, Minneapolis, MN), cathepsin D (Cat #AF1029, R&D Systems), LC3 (Cat #2775, Cell Signaling Technology, Danvers, MA), phospho-STAT3 (Cat #9145, Cell Signaling Technology), and STAT3 (Cat #9139, Cell Signaling Technology). Equal loading of samples and even transfer efficiency were monitored by re-probing stripped blots with the antibody to glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Cat #10R-G109A, Fitzgerald Industries International, Acton, MA). Quantification of protein expression was performed using Gel-Pro® Analyzer (Media Cybernetics, Silver Spring, MD).
Enzyme-Linked Immunosorbent Assay (ELISA)
Serum IL-6 and interleukin 11 (IL-11) levels in CON and IL6KO mice were measured using mouse IL-6 ELISA Ready-SET-Go kit (Cat #88-7064-22, eBioscience, Inc., San Diego, CA) and mouse IL-11 ELISA kit (Cat #ELM-IL11-001, RayBiotech, Inc., Norcross GA).
Terminal dUTP Nick End-labeling (TUNEL) Assay
Apoptotic cells were identified by TUNEL assay using CardioTACS In Situ Apoptosis Detection Kit (R&D Systems) as described previously.21
Statistical Analysis
Results are shown as mean±SE. Data were analyzed using 2-way ANOVA followed by Bonferroni post hoc test (for comparison of CON vs IL6KO mice, 2 weeks after TAC). The null hypothesis was rejected when P<0.05.
RESULTS
LV Function after Pressure Overload
Echocardiography showed reduced LV fractional shortening in CON mice 2 weeks after pressure overload (Table 1, Supplemental Material, Figure S2). These mice also had increased LV end-diastolic diameter and wall thickness, which was associated with increased mRNA expression of IL-6 in LV samples (no TAC: 100±10%, n=8; 2 weeks after TAC: 695±160%, n=8; P<0.002). IL6KO mice showed a similar degree reduction of LV fractional shortening 2 weeks after pressure overload (Table 1). There were no group differences in LV chamber dimensions between CON and IL6KO mice either before or 2 weeks after pressure overload. In addition, echocardiography showed similar changes in LV chamber dimensions and LV fractional shortening in CON and IL6KO mice after 4 weeks pressure overload (Table 2). In vivo hemodynamics study was performed to assess LV function 4 weeks after pressure overload in detail. There were no group differences in LV +dP/dt (CON: 4,159±557 mmHg/s, n=10; IL6KO: 3,521±396 mmHg/s, n=8; P=0.4) and LV −dP/dt (CON: -4,901±891 mmHg/s, n=10; IL6KO: -3,935±787 mmHg/s, n=8; P=0.4) 4 weeks after pressure overload. ESPVR, a measure relatively less dependent on load conditions, was not altered either (CON: 3.4±0.7 mmHg/μl, n=9; IL6KO: 2.3±0.5 mmHg/μl, n=9; P=0.3). Taken together, these data indicate that IL-6 deletion does not attenuate LV dilation and dysfunction associated with pressure overload.
Table 1.
Echocardiography Measurements 2 Weeks after TAC
| no TAC |
2 weeks after TAC |
P
|
|||||
|---|---|---|---|---|---|---|---|
| CON (n=10) |
IL6KO (n=10) |
CON (n=9) |
IL6KO (n=9) |
Interaction | IL-6 Deletion Effect |
TAC Effect |
|
| HR (bpm) | 627±10 | 622±15 | 631±14 | 606±18 | ns | ns | ns |
| LVEDD (mm) | 3.2±0.1 | 3.1±0.1 | 3.7±0.2 | 3.6±0.1 | ns | ns | <0.002 |
| LVESD (mm) | 1.8±0.1 | 1.8±0.1 | 2.6±0.3 | 2.5±0.2 | ns | ns | <0.0005 |
| PWTd (mm) | 0.6±0.01 | 0.6±0.01 | 0.9±0.04 | 0.9±0.05 | ns | ns | <0.0001 |
| IVSd (mm) | 0.6±0.01 | 0.6±0.01 | 0.8±0.03 | 0.9±0.04 | ns | ns | <0.0001 |
| FS (%) | 44±1 | 44±1 | 32±4 | 31±4 | ns | ns | <0.0001 |
HR, heart rate; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; PWTd, posterior wall thickness in diastole; IVSd, interventricular septum thickness in diastole; FS, fractional shortening; ns, not significant. Values shown are mean±SEM. Probability values are from 2-way ANOVA for interaction effect, IL-6 deletion effect, and transverse aortic constriction (TAC) effect.
Table 2.
Echocardiography Measurements 4 Weeks after TAC
| no TAC |
4 weeks after TAC |
P
|
|||||
|---|---|---|---|---|---|---|---|
| CON (n=8) |
IL6KO (n=8) |
CON (n=5) |
IL6KO(n=5) | Inter | IL-6 Deletion Effect |
TAC Effect |
|
| HR (bpm) | 528±25 | 521±28 | 463±17 | 474±31 | ns | ns | ns |
| LVEDD (mm) | 3.5±0.1 | 3.3±0.1 | 3.9±0.2 | 3.6±0.1 | ns | ns | <0.01 |
| LVESD (mm) | 2.0±0.1 | 1.9±0.1 | 2.5±0.2 | 2.2±0.2 | ns | ns | <0.004 |
| PWTd (mm) | 0.6±0.01 | 0.6±0.01 | 0.8±0.03 | 0.9±0.06 | ns | ns | <0.0001 |
| IVSd (mm) | 0.6±0.01 | 0.6±0.01 | 0.8±0.03 | 0.9±0.05 | ns | ns | <0.0001 |
| FS (%) | 42±3 | 41±2 | 25±3 | 29±4 | ns | ns | <0.0001 |
HR, heart rate; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; PWTd, posterior wall thickness in diastole; IVSd, interventricular septum thickness in diastole; FS, fractional shortening; ns, not significant. Values shown are mean±SEM. Probability values are from 2-way ANOVA for interaction effect, IL-6 deletion effect, and transverse aortic constriction (TAC) effect.
LV Size
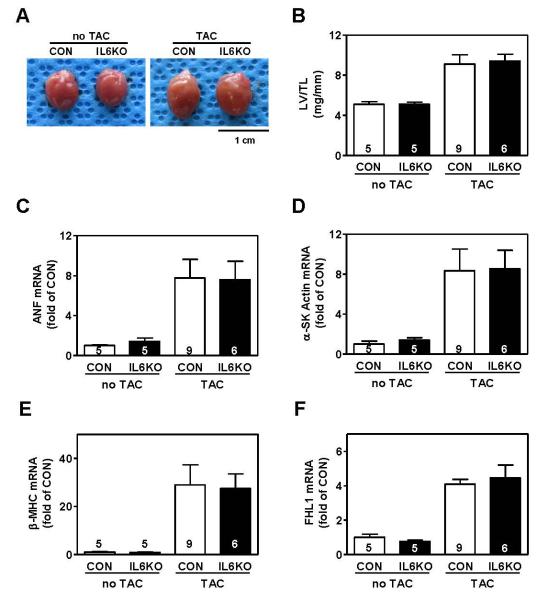
Pressure overload was associated with increased LV weight, LV weight/body weight ratio, LV weight/tibial length ratio in CON mice (Table 3; Figure 1A, B). The increase in lung weight after TAC indicates severe pulmonary congestion. IL6KO mice showed similar increases as CON mice in LV weight, lung weight, LV weight/body weight ratio, and LV weight/tibial length ratio after pressure overload (Table 3). By quantitative RT-PCR, we found comparable mRNA expression of the fetal gene program in LV samples from pressure-overloaded CON and IL6KO mice (Figure 1C, 1D, 1E). There were no differences in cardiac FHL1 mRNA (Figure 1F) or protein content [CON: 762±97 densitometric units (du), n=9; IL6KO: 658±120 du, n=6; P=0.51]. These data indicate that IL-6 deletion does not influence LV hypertrophy induced by pressure overload.
Table 3.
Morphometric Measurements
| no TAC |
2 weeks after TAC |
P
|
|||||
|---|---|---|---|---|---|---|---|
| CON (n=5) |
IL6KO (n=5) |
CON (n=9) |
IL6KO (n=6) |
Interaction | IL-6 Deletion Effect |
TAC Effect |
|
| Body Weight (g) | 22.5±0.3 | 23.6±1.1 | 25.5±0.7 | 24.5±0.5 | ns | ns | <0.02 |
| LV Weight (mg) | 87±3 | 88±4 | 154±16 | 157±12 | ns | ns | <0.0001 |
| Liver Weight (mg) | 1,160±66 | 1,406±92 | 1,144±79 | 1,151±65 | ns | ns | ns |
| Lung Weight (mg) | 134±3 | 136±7 | 254±32 | 260±32 | ns | ns | <0.0001 |
| Kidney (mg) | 322±10 | 331±18 | 300±14 | 289±7 | ns | ns | <0.04 |
| Spleen (mg) | 64±7 | 85±9 | 80±3 | 70±4 | ns | ns | ns |
| Tibial Length (mm) | 17.2±0.1 | 17.4±0.2 | 17.0±0.3 | 16.8±0.1 | ns | ns | ns |
| LV/BW (mg/g) | 3.9±0.1 | 3.8±0.3 | 6.1±0.7 | 6.4±0.5 | ns | ns | <0.001 |
| LV/TL (mg/mm) | 5.1±0.2 | 5.1±0.2 | 9.1±0.9 | 9.4±0.7 | ns | ns | <0.0001 |
BW, body weight; TL, tibial length; ns, not significant. Values shown are mean±SEM. Probability values are from 2-way ANOVA for interaction effect, IL-6 deletion effect, and transverse aortic constriction (TAC) effect.
Figure 1.
IL-6 deletion was insufficient to block pressure overload-induced LV hypertrophy. (A) Images of hearts from CON and IL6KO mice without TAC and 2 weeks after TAC. (B) There was no group difference in LV/tibial length ratio between CON and IL6KO mice without TAC and 2 weeks after TAC (P=0.82). No group differences in mRNA expression of atrial natriuretic factor (ANF, P=0.95) (C), α-skeletal muscle actin (α-SK Actin, P=0.95) (D), β-myosin heavy chain (β-MHC, P=0.90) (E), four-and-a-half LIM domain protein 1 (FHL1, P=0.61) (F) were observed in LV samples from CON and IL6KO mice 2 weeks after TAC. Probability values are from Bonferroni post hoc test (CON vs IL6KO, 2 weeks after TAC) after 2-way ANOVA. Error bars denote 1 SEM; numbers in bars indicate group size.
LV Fibrosis
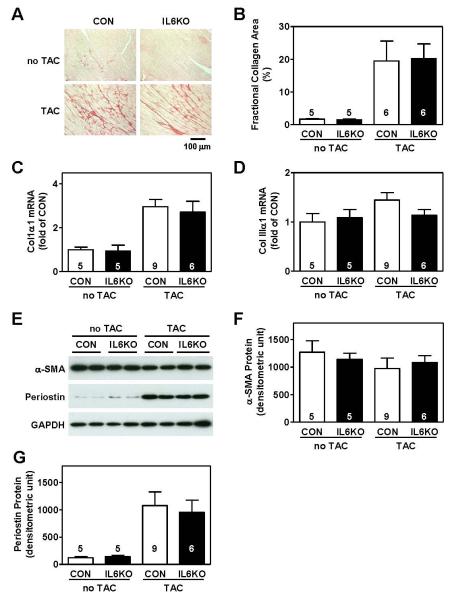
As anticipated, pressure overload increased LV fibrosis in CON mice, as shown by increased collagen deposition detected by picrosirius staining (Figure 2A). A similar degree of collagen deposition was found in LV samples from pressure-overloaded IL6KO mice (Figure 2A). Quantification of fractional collagen area showed no difference between CON and IL6KO mice after pressure overload (Figure 2B). There was no group difference in mRNA expression of extracellular matrix proteins collagen Iα1 (Figure 2C) and collagen IIIα1 (Figure 2D). We also found no difference in cardiac protein content of α-smooth muscle actin (Figure 2E, 2F) and periostin (Figure 2E, 2G). These results indicate that IL-6 deletion does not inhibit LV fibrosis induced by pressure overload.
Figure 2.
IL-6 deletion was insufficient to block LV fibrosis after pressure overload. (A) Representative photomicrographs show no difference in collagen deposition in LV samples from CON and IL6KO mice without TAC and 2 weeks after TAC. (B) The bar graph summarizes fractional collagen area in LV samples from CON and IL6KO mice without TAC and 2 weeks after TAC (P=0.92). No group differences were observed in mRNA expression of collagen Iα1 (P=0.63) (C) and collagen IIIα1 (P=0.13) (D) in LV samples from CON and IL6KO mice without TAC and 2 weeks after TAC. (C) Representative Western blots showing protein contents of α-smooth muscle actin (α-SMA) and periostin in LV samples from CON and IL6KO mice before and 2 weeks after TAC. There was no significant difference in protein expression of α-SMA (P=0.24) (F) and periostin (P=0.74) (G). Probability values are from CON vs IL6KO comparison by Bonferroni post hoc test (CON vs IL6KO, 2 weeks after TAC) after 2-way ANOVA. Error bars denote 1 SEM; numbers in bars indicate group size.
Apoptosis and Autophagy
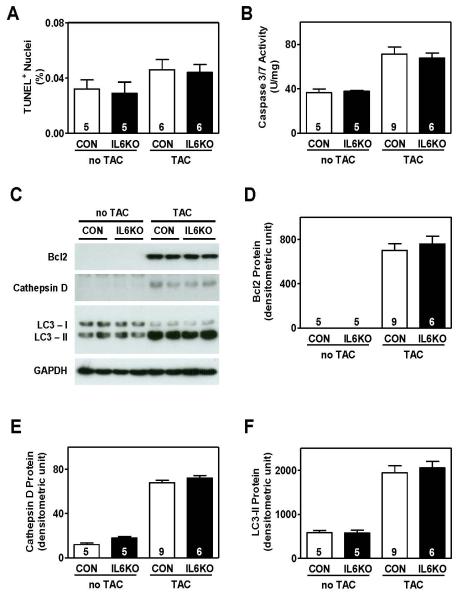
Pressure overload increased cardiac myocyte apoptosis (TUNEL staining) (Figure 3A), caspase 3/7 activity (Figure 3B), and Bcl2 protein expression (Figure 3C, D) in CON mice. IL6KO mice showed similar increases after pressure overload (Figure 3, Supplemental Material, Figure S3). Furthermore, there were no differences in cardiac protein levels of cathepsin D (Figure 3C, E) and LC3-II (Figure 3C, F) between CON and IL6KO mice 2 weeks after TAC. These data suggest that IL-6 deletion does not affect LV apoptosis or autophagy in hearts under pressure overload.
Figure 3.
IL-6 deletion was insufficient to suppress apoptosis and had no effect on autophagy in pressure-overloaded hearts. (A) There was no group difference in TUNEL positive cardiac myocytes in CON and IL6KO mice 2 weeks after TAC (P=0.83). (B) Caspase 3/7 activity was not significantly different in LV samples from CON and IL6KO 2 weeks after TAC (P=0.90). (C) Representative Western blots showing protein contents of Bcl2,cathepsin D, and LC3-II in LV samples from CON and IL6KO mice before and 2 weeks after TAC. There was no difference in protein content of Bcl2 (P=0.55) (D), cathepsin D (P=0.31) (E), and LC3-II (P=0.64) (F) in LV samples from CON and IL6KO mice 2 weeks after pressure overload. Probability values are from CON vs IL6KO comparison by Bonferroni post hoc test (CON vs IL6KO, 2 weeks after TAC) after 2-way ANOVA. Error bars denote 1 SEM; numbers in bars indicate group size.
STAT3 Activation
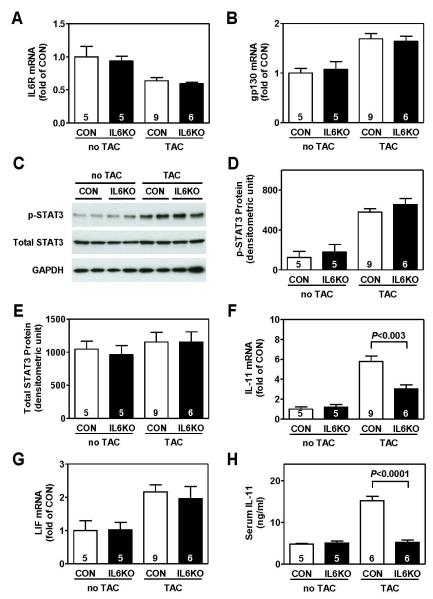
There were no group differences in mRNA expression of IL-6 receptor (IL-6R) (Figure 4A) and signal transducing component gp130 (Figure 4B) in LV samples from CON and IL6KO mice either before or 2 weeks after pressure overload. Pressure overload activated STAT3 signaling pathway in CON mice, as shown by increased STAT3 phosphorylation 2 weeks after TAC (Figure 4C, D). A similar degree of increase in STAT3 phosphorylation was found in LV samples from pressure-overloaded IL6KO mice (Figure 4C, D). No change was found in total STAT3 content (Figure 4C, E).
Figure 4.
IL-6 deletion was associated with decreased expression of IL-11, which may compensate the IL-6 deletion effects, in pressure-overloaded hearts. (A) There was no group difference in IL-6 receptor mRNA content in LV samples from CON and IL6KO mice 2 weeks after TAC (P=0.50). (B) There was no group difference in gp130 mRNA content in LV samples from CON and IL6KO mice 2 weeks after TAC (P=0.76). (C) Representative Western blots showing protein contents of phospho-STAT3 (p-STAT3) and total STAT3 in LV samples from CON and IL6KO mice before and 2 weeks after TAC. (D) There was no group difference in phospho-STAT3 protein content in LV samples from CON and IL6KO mice 2 weeks after TAC (P=0.25). (E) Total STAT3 protein content was not significantly different in LV samples from CON and IL6KO 2 weeks after TAC (P=1.00). (F) IL-6 deletion was associated with decreased mRNA expression of IL-11 in LV samples from IL6KO mice 2 weeks after TAC. (G) There was no group difference in leukemia inhibitory factor (LIF) mRNA content in LV samples from CON and IL6KO mice 2 weeks after TAC (P=0.61). (H) Decreased serum level of IL-11 was found in IL6KO mice compared to CON mice 2 weeks after TAC. Probability values are from CON vs IL6KO comparison by Bonferroni post hoc test (CON vs IL6KO, 2 weeks after TAC) after 2-way ANOVA. Error bars denote 1 SEM; numbers in bars indicate group size.
Expression of IL-6 Family Members
Pressure overload was associated with altered cardiac mRNA expression of other IL-6 family members: increased mRNA expression of IL-11 (Figure 4F), LIF (Figure 4G), and CNTF (Supplemental Material, Figure S4A); and decreased mRNA expression of cardiotrophin 1 (Supplemental Material, Figure S4B). Interestingly, IL-6 deletion only attenuated cardiac IL-11 mRNA expression in pressure-overloaded hearts, and had no effects on mRNA expression of LIF (Figure 4G), CNTF (Supplemental Material, Figure S4A), and cardiotrophin 1 (Supplemental Material, Figure S4B). Decreased serum IL-11 levels were also found in IL6KO mice 2 weeks after TAC (Figure 4H).
DISCUSSION
Epidemiology and animal studies show that heart failure is associated with increased expression of pro-inflammatory cytokine IL-6.4, 22 In this study, we showed that cardiac IL-6 expression was increased in pressure-overloaded hearts. However, IL-6 deletion did not influence pressure overload-induced LV dilation or dysfunction. There were no group differences in LV hypertrophy, fibrosis, apoptosis, or autophagy between CON and IL6KO mice after pressure overload. These data indicate that IL-6 deletion is dispensable for LV remodeling and dysfunction in response to pressure overload.
A previous study showed that deletion of IL-6 attenuated LV hypertrophy induced by infusion of angiotensin II.19 However, we found that IL-6 deletion did not affect LV hypertrophy and expression of the fetal gene program in pressure-overloaded hearts (Figure 2). The reason for this mismatch is not known, but may be due to differences in models (angiotensin II infusion vs pressure overload). Alternatively, the observed blockade of LV hypertrophy by IL-6 deletion in angiotensin II infusion may reflect the effect of genetic backgrounds, but not the effect of IL-6 deletion. Indeed, control mice in C57BL/6N genetic background (Charles River) were used to compared to IL6KO mice (in C57BL/6J genetic background, The Jackson Laboratories) in the previous study.19 We have previously shown profound differences in survival and cardiac function change in C57BL/6N and C57BL/6J mice (The Jackson Laboratories) in response to pressure overload.20
The IL-6 family cytokines include pro-inflammatory cytokines IL-6, LIF, CNTF, oncostatin M, and cardiotropin-1, and anti-inflammatory cytokine IL-11. Previous studies demonstrate that pro-inflammatory IL-6 family members induces LV hypertrophy, dilation, dysfunction, and fibrosis,4-6, 11-14 but anti-inflammatory cytokine IL-11 protects cardiac myocytes from ischemia/reperfusion injury23 and decreases cardiac fibrosis and apoptosis after myocardial infarction.24 Glycoprotein gp130 is a common signal transducing component for receptors for all IL-6 family members, and transduces signals from these cytokines to JAK-STAT3 signaling pathway. The inability of IL-6 deletion to attenuate LV remodeling and dysfunction after pressure overload may be due to the compensatory effects of other IL-6 family members. Indeed, deletion of IL-6 had no effect on cardiac STAT3 phosphorylation either before or after pressure overload (Figure 4C, 4D). Moreover, IL-6 deletion inhibited pressure overload-increased cardiac IL-11 mRNA content and circulating IL-11 level (Figure 4F, 4H), but did no affect expression of other IL-6 family members. Therefore, attenuated content of IL-11 appears to be a compensatory mechanism for IL-6 deletion in pressure overloaded hearts.
In addition to pressure overload, myocardial infarction is another major risk factor for congestive heart failure. Although IL-6 reduces apoptosis25 and is important for ischemic preconditioning,26 chronic elevation of IL-6 is associated with LV dilation and dysfunction.17, 27, 28 LV samples from the infarct border zone show elevated levels of IL-6,29 which correlates with LV size after myocardial infarction.30 Interestingly, IL-6 deletion did not prevent or worsen LV dysfunction and adverse remodeling in myocardial infarcted hearts.31 These results, together with our findings in the current study, demonstrate that IL-6 expression is not required for LV hypertrophy, dilation, and dysfunction in pathologically stressed hearts, although chronic elevation of IL-6 is sufficient to induce LV remodeling and dysfunction.
In conclusion, IL-6 deletion does not block LV remodeling and dysfunction in pressure-overloaded hearts. These data suggest that limiting availability of IL-6 alone (such as with immunodepletion with IL-6 antibody) is not sufficient to attenuate LV remodeling and dysfunction in failing hearts.
Supplementary Material
Acknowledgments
Sources of Support: This work was supported by Grants from the American Heart Association Western State Affiliates and the National Institutes of Health.
List of Abbreviations
- CNTF
ciliary neurotrophic factor
- CON mice
C57BL/6J mice
- ELISA
enzyme-linked immunosorbent assay
- ESPVR
end-systolic pressure-volume relationship
- IL-6
interleukin 6
- IL-11
interleukin 11
- IL6KO mice
IL-6-deleted mice
- LIF
leukemia inhibitory factor
- LV
left ventricular
- TAC
transverse aortic constriction
- TUNEL
terminal dUTP nick end-labeling
Footnotes
DISCLOSURE None.
REFERENCES
- 1.Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, Ferguson TB, Ford E, Furie K, Gillespie C, Go A, Greenlund K, Haase N, Hailpern S, Ho PM, Howard V, Kissela B, Kittner S, Lackland D, Lisabeth L, Marelli A, McDermott MM, Meigs J, Mozaffarian D, Mussolino M, Nichol G, Roger VL, Rosamond W, Sacco R, Sorlie P, Roger VL, Thom T, Wasserthiel-Smoller S, Wong ND, Wylie-Rosett J. Heart disease and stroke statistics--2010 update: a report from the American Heart Association. Circulation. 2010;121:e46–e215. doi: 10.1161/CIRCULATIONAHA.109.192667. [DOI] [PubMed] [Google Scholar]
- 2.Mann D. Pathophysiology of heart failure. In: Libby P, Bonow R, Mann D, Zipes D, editors. Braunwald’s Heart Disease. A textbook of cardiovascular medicine. Saunders; Philadelphia: 2008. pp. 541–560. [Google Scholar]
- 3.Bozkurt B, Mann DL, Deswal A. Biomarkers of inflammation in heart failure. Heart Fail Rev. 2010;15:331–341. doi: 10.1007/s10741-009-9140-3. [DOI] [PubMed] [Google Scholar]
- 4.Birner CM, Ulucan C, Fredersdorf S, Rihm M, Lowel H, Stritzke J, Schunkert H, Hengstenberg C, Holmer S, Riegger G, Luchner A. Head-to-head comparison of BNP and IL-6 as markers of clinical and experimental heart failure: Superiority of BNP. Cytokine. 2007;40:89–97. doi: 10.1016/j.cyto.2007.08.009. [DOI] [PubMed] [Google Scholar]
- 5.Tsutamoto T, Asai S, Tanaka T, Sakai H, Nishiyama K, Fujii M, Yamamoto T, Ohnishi M, Wada A, Saito Y, Horie M. Plasma level of cardiotrophin-1 as a prognostic predictor in patients with chronic heart failure. Eur J Heart Fail. 2007;9:1032–1037. doi: 10.1016/j.ejheart.2007.07.015. [DOI] [PubMed] [Google Scholar]
- 6.Hirota H, Izumi M, Hamaguchi T, Sugiyama S, Murakami E, Kunisada K, Fujio Y, Oshima Y, Nakaoka Y, Yamauchi-Takihara K. Circulating interleukin-6 family cytokines and their receptors in patients with congestive heart failure. Heart Vessels. 2004;19:237–241. doi: 10.1007/s00380-004-0770-z. [DOI] [PubMed] [Google Scholar]
- 7.Zolk O, Ng LL, O’Brien RJ, Weyand M, Eschenhagen T. Augmented expression of cardiotrophin-1 in failing human hearts is accompanied by diminished glycoprotein 130 receptor protein abundance. Circulation. 2002;106:1442–1446. doi: 10.1161/01.cir.0000033117.39335.df. [DOI] [PubMed] [Google Scholar]
- 8.Kalogeropoulos A, Georgiopoulou V, Psaty BM, Rodondi N, Smith AL, Harrison DG, Liu Y, Hoffmann U, Bauer DC, Newman AB, Kritchevsky SB, Harris TB, Butler J. Inflammatory markers and incident heart failure risk in older adults: the Health ABC (Health, Aging, and Body Composition) study. J Am Coll Cardiol. 2010;55:2129–2137. doi: 10.1016/j.jacc.2009.12.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Matsubara J, Sugiyama S, Nozaki T, Sugamura K, Konishi M, Ohba K, Matsuzawa Y, Akiyama E, Yamamoto E, Sakamoto K, Nagayoshi Y, Kaikita K, Sumida H, Kim-Mitsuyama S, Ogawa H. Pentraxin 3 is a new inflammatory marker correlated with left ventricular diastolic dysfunction and heart failure with normal ejection fraction. J Am Coll Cardiol. 2011;57:861–869. doi: 10.1016/j.jacc.2010.10.018. [DOI] [PubMed] [Google Scholar]
- 10.Kubota T, Miyagishima M, Alvarez RJ, Kormos R, Rosenblum WD, Demetris AJ, Semigran MJ, Dec GW, Holubkov R, McTiernan CF, Mann DL, Feldman AM, McNamara DM. Expression of proinflammatory cytokines in the failing human heart: comparison of recent-onset and end-stage congestive heart failure. J Heart Lung Transplant. 2000;19:819–824. doi: 10.1016/s1053-2498(00)00173-x. [DOI] [PubMed] [Google Scholar]
- 11.Cesari M, Penninx BW, Newman AB, Kritchevsky SB, Nicklas BJ, Sutton-Tyrrell K, Rubin SM, Ding J, Simonsick EM, Harris TB, Pahor M. Inflammatory markers and onset of cardiovascular events: results from the Health ABC study. Circulation. 2003;108:2317–2322. doi: 10.1161/01.CIR.0000097109.90783.FC. [DOI] [PubMed] [Google Scholar]
- 12.Fisman EZ, Benderly M, Esper RJ, Behar S, Boyko V, Adler Y, Tanne D, Matas Z, Tenenbaum A. Interleukin-6 and the risk of future cardiovascular events in patients with angina pectoris and/or healed myocardial infarction. Am J Cardiol. 2006;98:14–18. doi: 10.1016/j.amjcard.2006.01.045. [DOI] [PubMed] [Google Scholar]
- 13.Ohtsuka T, Hamada M, Inoue K, Ohshima K, Sujzuki J, Matsunaka T, Ogimoto A, Hara Y, Shigematsu Y, Higaki J. Relation of circulating interleukin-6 to left ventricular remodeling in patients with reperfused anterior myocardial infarction. Clin Cardiol. 2004;27:417–420. doi: 10.1002/clc.4960270712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yan AT, Yan RT, Cushman M, Redheuil A, Tracy RP, Arnett DK, Rosen BD, McClelland RL, Bluemke DA, Lima JA. Relationship of interleukin-6 with regional and global left-ventricular function in asymptomatic individuals without clinical cardiovascular disease: insights from the Multi-Ethnic Study of Atherosclerosis. Eur Heart J. 2010;31:875–882. doi: 10.1093/eurheartj/ehp454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Finkel MS, Oddis CV, Jacob TD, Watkins SC, Hattler BG, Simmons RL. Negative inotropic effects of cytokines on the heart mediated by nitric oxide. Science. 1992;257:387–389. doi: 10.1126/science.1631560. [DOI] [PubMed] [Google Scholar]
- 16.Villegas S, Villarreal FJ, Dillmann WH. Leukemia Inhibitory Factor and Interleukin-6 downregulate sarcoplasmic reticulum Ca2+ ATPase (SERCA2) in cardiac myocytes. Basic Res Cardiol. 2000;95:47–54. doi: 10.1007/s003950050007. [DOI] [PubMed] [Google Scholar]
- 17.Janssen SP, Gayan-Ramirez G, Van den Bergh A, Herijgers P, Maes K, Verbeken E, Decramer M. Interleukin-6 causes myocardial failure and skeletal muscle atrophy in rats. Circulation. 2005;111:996–1005. doi: 10.1161/01.CIR.0000156469.96135.0D. [DOI] [PubMed] [Google Scholar]
- 18.Melendez GC, McLarty JL, Levick SP, Du Y, Janicki JS, Brower GL. Interleukin 6 mediates myocardial fibrosis, concentric hypertrophy, and diastolic dysfunction in rats. Hypertension. 2010;56:225–231. doi: 10.1161/HYPERTENSIONAHA.109.148635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Coles B, Fielding CA, Rose-John S, Scheller J, Jones SA, O’Donnell VB. Classic interleukin-6 receptor signaling and interleukin-6 trans-signaling differentially control angiotensin II-dependent hypertension, cardiac signal transducer and activator of transcription-3 activation, and vascular hypertrophy in vivo. Am J Pathol. 2007;171:315–325. doi: 10.2353/ajpath.2007.061078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tang T, Lai NC, Hammond HK, Roth DM, Yang Y, Guo T, Gao MH. Adenylyl cyclase 6 deletion reduces LV hypertrophy, dilation, dysfunciton and fibrosis in pressure-overloaded female mice. J Am Coll Cardiol. 2010;55:1476–1486. doi: 10.1016/j.jacc.2009.11.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Takahashi T, Tang T, Lai NC, Roth DM, Rebolledo B, Saito M, Lew WY, Clopton P, Hammond HK. Increased cardiac adenylyl cyclase expression is associated with increased survival after myocardial infarction. Circulation. 2006;114:388–396. doi: 10.1161/CIRCULATIONAHA.106.632513. [DOI] [PubMed] [Google Scholar]
- 22.Tamariz L, Hare JM. Inflammatory cytokines in heart failure: roles in aetiology and utility as biomarkers. Eur Heart J. 2010;31:768–770. doi: 10.1093/eurheartj/ehq014. [DOI] [PubMed] [Google Scholar]
- 23.Kimura R, Maeda M, Arita A, Oshima Y, Obana M, Ito T, Yamamoto Y, Mohri T, Kishimoto T, Kawase I, Fujio Y, Azuma J. Identification of cardiac myocytes as the target of interleukin 11, a cardioprotective cytokine. Cytokine. 2007;38:107–115. doi: 10.1016/j.cyto.2007.05.011. [DOI] [PubMed] [Google Scholar]
- 24.Obana M, Maeda M, Takeda K, Hayama A, Mohri T, Yamashita T, Nakaoka Y, Komuro I, Takeda K, Matsumiya G, Azuma J, Fujio Y. Therapeutic activation of signal transducer and activator of transcription 3 by interleukin-11 ameliorates cardiac fibrosis after myocardial infarction. Circulation. 2010;121:684–691. doi: 10.1161/CIRCULATIONAHA.109.893677. [DOI] [PubMed] [Google Scholar]
- 25.Matsushita K, Iwanaga S, Oda T, Kimura K, Shimada M, Sano M, Umezawa A, Hata J, Ogawa S. Interleukin-6/soluble interleukin-6 receptor complex reduces infarct size via inhibiting myocardial apoptosis. Lab Invest. 2005;85:1210–1223. doi: 10.1038/labinvest.3700322. [DOI] [PubMed] [Google Scholar]
- 26.Dawn B, Xuan YT, Guo Y, Rezazadeh A, Stein AB, Hunt G, Wu WJ, Tan W, Bolli R. IL-6 plays an obligatory role in late preconditioning via JAK-STAT signaling and upregulation of iNOS and COX-2. Cardiovasc Res. 2004;64:61–71. doi: 10.1016/j.cardiores.2004.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kurdi M, Booz GW. Can the protective actions of JAK-STAT in the heart be exploited therapeutically? Parsing the regulation of interleukin-6-type cytokine signaling. J Cardiovasc Pharmacol. 2007;50:126–141. doi: 10.1097/FJC.0b013e318068dd49. [DOI] [PubMed] [Google Scholar]
- 28.Lecour S, James RW. When are pro-inflammatory cytokines SAFE in heart failure? Eur Heart J. 2011;32:680–685. doi: 10.1093/eurheartj/ehq484. [DOI] [PubMed] [Google Scholar]
- 29.Gwechenberger M, Mendoza LH, Youker KA, Frangogiannis NG, Smith CW, Michael LH, Entman ML. Cardiac myocytes produce interleukin-6 in culture and in viable border zone of reperfused infarctions. Circulation. 1999;99:546–551. doi: 10.1161/01.cir.99.4.546. [DOI] [PubMed] [Google Scholar]
- 30.Ono K, Matsumori A, Shioi T, Furukawa Y, Sasayama S. Cytokine gene expression after myocardial infarction in rat hearts: possible implication in left ventricular remodeling. Circulation. 1998;98:149–156. doi: 10.1161/01.cir.98.2.149. [DOI] [PubMed] [Google Scholar]
- 31.Fuchs M, Hilfiker A, Kaminski K, Hilfiker-Kleiner D, Guener Z, Klein G, Podewski E, Schieffer B, Rose-John S, Drexler H. Role of interleukin-6 for LV remodeling and survival after experimental myocardial infarction. FASEB J. 2003;17:2118–2120. doi: 10.1096/fj.03-0331fje. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.