Abstract
Obesity, an established risk factor for epithelial cancers, remains prevalent in the US and many other countries. In contrast to positive energy balance states (overweight, obesity), calorie restriction (CR) has been shown to act as a universal inhibitor of tumorigenesis in multiple animal models of human cancer. Unfortunately, the mechanisms underlying the enhancing effects of obesity or the inhibitory effects of CR on cancer etiology remain elusive. Here, we evaluated the impact of dietary energy balance manipulation on epithelial carcinogenesis and identified several potential mechanisms that may account for the differential effects of obesity and CR on cancer. Obesity enhanced tumor promotion during epithelial carcinogenesis, in part, due to altered IGF-1R/EGFR crosstalk and downstream signaling to effectors such as Akt/mTOR. Obesity-induced changes in cellular signaling subsequently led to altered levels of cell cycle proteins that favored enhanced epidermal proliferation during tumor promotion. In contrast, CR reduced susceptibility to tumor promotion, attenuated IGF-1R/EGFR crosstalk and downstream signaling and altered levels of cell cycle proteins that favored reduced epidermal proliferation during tumor promotion. Collectively, these findings suggest potential targets for the prevention of epithelial cancers, as well as for reversal of obesity-mediated cancer development and progression.
Keywords: energy balance, epithelial carcinogenesis, IGF-1R/EGFR crosstalk, cell cycle, proliferation
Introduction
Energy balance refers to the relationship between caloric consumption and energy expenditure (1). Epidemiologic and animal studies have established a direct correlation between positive energy balance states (e.g., overweight, obesity), and risk of developing multiple cancers (2–5). In contrast, negative energy balance states, such as calorie restriction (CR), have been shown to consistently inhibit tumorigenesis in animal models regardless of mode of tumor induction (1, 6–8). Previous studies have evaluated the impact of fat consumption and/or CR on two-stage skin carcinogenesis (8–10). This model of chemically-induced epithelial carcinogenesis enables mechanistic evaluation of dietary manipulation during all stages of tumor development, including tumor initiation, promotion and progression (11). Boutwell (8) found that CR inhibited skin tumor development using the two-stage carcinogenesis protocol and Birt and colleagues (9, 10) showed that 40% CR consistently inhibited tumor promotion by TPA, with little or no effects on either tumor initiation or tumor progression. Although these findings indicated a role for energy balance in the modulation of two-stage skin carcinogenesis, especially during tumor promotion, the effect of diet-induced obesity (DIO) per se has not been adequately studied in this model.
Several mechanisms have been proposed to explain the inhibitory effects of CR on tumor development (12, 13) including elevated serum corticosterone (6, 14) and alterations in cellular signaling. TPA-induced epidermal AP-1 activation and ERK phosphorylation was reduced in mice subject to 40% CR (15–17). Xie et al., (18) reported a reduction in TPA-mediated activation of epidermal PI3K and Ras signaling following 20% CR. Recently, we reported that both CR and DIO modulated steady state growth factor signaling pathways in mouse epidermis, liver and prostate (19). Additional studies suggested that these effects may be mediated by diet-induced changes in circulating IGF-1 levels and altered IGF-1R signaling (19, 20).
Diets of varying caloric density were used to induce changes in body mass and body fat content to determine the impact of both positive and negative energy balance on the promotion stage of multistage epithelial carcinogenesis in mouse skin. We provide novel evidence demonstrating that dietary energy balance affects susceptibility to epithelial carcinogenesis, in part, through diet-induced changes in IGF-1R and EGFR crosstalk and downstream signaling during tumor promotion.
Materials and Methods
Animals
ICR female mice (3–4 weeks of age, Harlan Teklad) were group housed; however, mice maintained on a 30% CR regimen were separated for one hour for feeding. Mice were weighed before randomization and then every two weeks for the duration of the experiments.
Diets
Four diets ranging in caloric density were utilized (Research Diets) and have been previously described: 30% CR diet, 15% CR diet, 10 Kcal% fat (ad libitum) and 60 Kcal% fat (ad libitum)(1). CR was achieved by administering a daily aliquot equivalent to 70% or 85% of the daily energy consumed by the 10 Kcal% fat group, as previously detailed (19).
Serum analysis and body fat composition
Blood was collected immediately following CO2 asphyxiation, serum was prepared and serum IGF-1, insulin, leptin and adiponectin levels were determined as previously described (19, 21). Corticosterone levels were determined by ELISA (Kamiya Biomedical Co.). Percent body fat was determined as previously described (22).
Two-stage skin carcinogenesis
ICR female mice were placed on the 10 Kcal% fat control diet at 7 weeks of age and initiated with 25 nmol of 7,12-dimethylbenz[a]anthracene (DMBA; Eastman Kodak Co.). Four weeks following initiation, mice were randomized and placed on the four diets (n=30/group). Four weeks later, mice received twice weekly topical treatments of 3.4 nmol of 12-O-tetradecanoylphorbol-13-acetate (TPA; Alexis Biochemicals) for 50 weeks. Tumor incidence (percent mice with papillomas) and tumor multiplicity (average number of tumors per mouse) were determined weekly until multiplicity reached a plateau (29 weeks). Carcinoma incidence and average carcinomas per mouse were determined weekly from initial detection until 50 weeks of tumor promotion. Squamous cell carcinomas (SCCs) were confirmed by histopathology.
Epidermal hyperproliferation
Female ICR mice were maintained on the diets described above for 15 weeks, after which they were treated twice weekly for two weeks with either acetone vehicle or 3.4 nmol of TPA (n=3/group). Mice were injected with bromodeoxyuridine (BrdU; Sigma Aldrich; 100 μg/g body weight) thirty minutes prior to sacrifice. Whole skin sections were excised, processed and evaluated for epidermal thickness and labeling index as previously described (20).
Epidermal protein lysates
For short-term in vivo experiments, ICR mice were maintained on the diets for 15 weeks, after which they received a single application of either acetone (vehicle) or 3.4 nmol of TPA. Mice were killed either 6 (acetone, TPA) or 18 hours (TPA) after treatment, epidermis was scraped and protein lysates were prepared as previously described (20).
Cell culture
C50 cells are a non-tumorigenic keratinocyte cell line derived from spontaneously immortalized normal mouse keratinocytes. The cells were obtained from Dr. Susan Fischer (UTMDACC) and were cultured as previously described (23) with no further characterizations. When cells reached ~80% confluency, they were serum and growth factor starved for 24 hours. Plates were then stimulated with either 25 ng/mL recombinant human IGF-1 (rhIGF-1; Sigma Aldrich) or 10 ng/mL epidermal growth factor (EGF; BD Biosciences) and harvested at multiple time points for protein or RNA isolation.
Immunoprecipitation and western blot analysis
Immunoprecipitation and Western blot analyses were performed using lysates prepared from either epidermis or cultured keratinocytes. For co-immunoprecipitation experiments, lysates were precipitated with IGF-1R (Cell Signaling) or EGFR (Millipore) antibodies using the Dynabead Protein G IP kit (Invitrogen). Western blots analyses were performed as previously described (20).
Real time quantitative reverse transcriptase polymerase chain reaction (qPCR)
For in vivo experiments, ICR mice were maintained on the diets for 15 weeks, after which they received a single application of either acetone vehicle or 3.4 nmol of TPA. Mice were killed either 6 (acetone, TPA) or 18 hours (TPA) after treatment and epidermis was scraped for RNA isolation (n=3/group). For in vitro experiments, C50 cells were stimulated with either IGF-1 or EGF, harvested for RNA and RNA was isolated using the Qiagen RNeasy protect mini kit. RNA quality was assessed using the Agilent 2100 Bioanalyzer (Agilent Technologies, Inc.) and qPCR was performed, as previously described, using assays on demand specific to TGF-α, HB-EGF, amphiregulin and EGFR (21). RNA was normalized to GAPDH. All measurements were duplicated.
Results
Dietary energy balance effects on skin tumor promotion
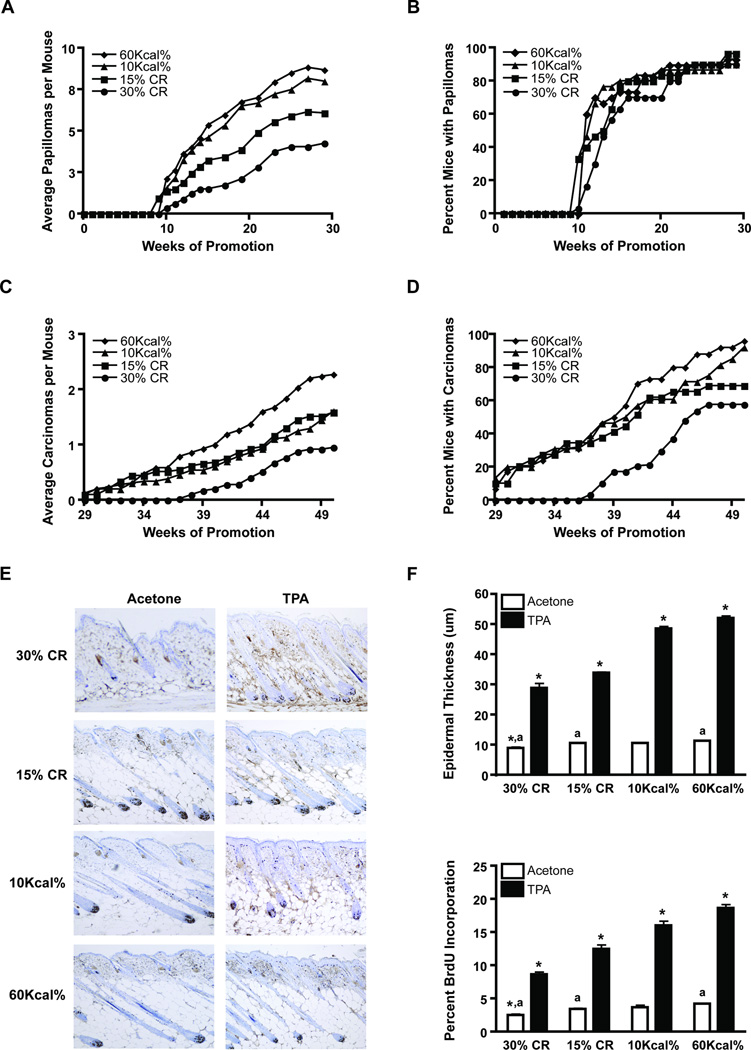
A two-stage skin carcinogenesis experiment was conducted utilizing four experimental diet groups to generate lean (30% CR), normal (15% CR), overweight (10Kcal% fat) and obese (DIO, 60Kcal% fat) body phenotypes, as previously described (5, 21, 24). Groups were initiated with DMBA, maintained on 10 Kcal% diet for 4 weeks, randomized into the experimental groups and promoted twice weekly with 3.4 nmol of TPA for 50 weeks. Tumor incidence and multiplicity (average number of papillomas per mouse) were calculated weekly until the latter reached a plateau at 29 weeks. CR (15% and 30%) significantly inhibited papilloma multiplicity when compared to both the overweight control and DIO groups (p < 0.05; Mann-Whitney U test); however, no significant differences were observed between the overweight control and DIO groups (Figure 1A, Supplemental Table 1). Dietary energy balance manipulation had no significant effect on papilloma incidence under these experimental conditions (Figure 1B, Supplemental Table 1).
Figure 1.
Effect of dietary energy balance on two-stage skin carcinogenesis and TPA-induced epidermal proliferation. Two-stage skin carcinogenesis was conducted using lean (30% CR), normal (15% CR), overweight control (10 Kcal% fat) and obese (60 Kcal% fat) mice (n=30/group). A, Tumor multiplicity. CR (30%, 15%) significantly reduced tumor multiplicity (p < 0.05; Mann-Whitney U test); B, Tumor incidence; C, Average carcinomas per mouse. 30% CR significantly reduced, while DIO (60 Kcal%) significantly increased the average number of carcinomas per mouse (p < 0.05; Mann-Whitney U test); D, Carcinoma incidence. CR (30%, 15%) significantly reduced carcinoma incidence (p < 0.05; χ2 test). E-F, ICR female mice were maintained on the four diets for 15 weeks and were treated with either acetone (white bars) or 3.4 nmol TPA (black bars), twice weekly for two weeks (n=3/group). E, Representative BrdU stained skin sections; F, Epidermal thickness (upper) and percent BrdU incorporation (lower). *Statistically different from all similarly treated groups; a significantly different from similarly labeled values (p < 0.05; Mann-Whitney U test).
Treatments continued for an additional 21 weeks to evaluate the impact of dietary energy balance on tumor progression. As shown in Figure 1 (panels C and D), the number and incidence of SCCs was significantly reduced in the 30% CR group (p < 0.05, Mann-Whitney U test and p < 0.05, χ2 test, respectively), compared to the 15% CR, overweight control and the DIO groups; however, dietary manipulation did not affect the overall rate of malignant conversion (see also Supplemental Table 1). A second two-stage skin carcinogenesis experiment was conducted that yielded nearly identical findings (data not shown).
Effect of long-term dietary manipulation on body profiles during two-stage skin carcinogenesis
Body mass, percent body fat and circulatory protein levels (i.e., IGF-1, insulin, leptin, adiponectin) were evaluated at the conclusion of the two-stage skin carcinogenesis study (Supplemental Table 2.I). Body mass and percent body fat was significantly different among all groups (p < 0.05, Mann-Whitney U test). CR (both 30% and 15%) significantly reduced levels of circulating IGF-1, insulin and leptin. In contrast, CR increased levels of adiponectin, as compared to both the overweight control and DIO groups; however, only the 30% CR group yielded a statistically significant difference (p < 0.05, Mann-Whitney U test). Despite significant differences in both body mass and percent body fat, no significant differences in the levels of circulatory proteins were observed between the overweight control and DIO groups at the end of the 50-week experimental period.
Dietary energy balance modulates TPA-induced epidermal hyperproliferation
To evaluate potential mechanisms whereby dietary energy balance modulates two-stage skin carcinogenesis, additional mice were maintained on the four diets for 15 weeks, thus generating lean, normal, overweight and obese body phenotypes. Body mass, percent body fat and levels of circulatory proteins were then determined at this time point (Supplemental Table 2.II). Body mass and percent body fat correlated directly with caloric consumption, yielding significant differences in both parameters among all groups (p < 0.05, Mann-Whitney U test). Similarly, levels of IGF-1, insulin, leptin and adiponectin were significantly different across the groups, with the greatest differences occurring between the caloric extremes (p < 0.05, Mann-Whitney U test). Specifically, CR reduced, while DIO increased levels of circulating IGF-1, insulin and leptin, findings consistent with our previous studies (19). Levels of adiponectin inversely correlated with caloric consumption, with significant differences occurring between each CR group and the overweight control and DIO groups (p < 0.05, Mann-Whitney U test). Additionally, 30% CR significantly increased circulating corticosterone levels relative to both the overweight control and DIO groups, however, no other statistically significant differences in corticosterone levels were observed.
Given the effect of dietary energy balance on tumor promotion, we evaluated diet-induced changes in the epidermal proliferative response following TPA treatment. Female ICR mice maintained on the four diets for 15 weeks were treated twice weekly for two weeks with either acetone or 3.4 nmol of TPA. Groups of mice were killed 48 hours following the final treatment and whole skin sections were removed and processed. Epidermal hyperplasia (epidermal thickness) and labeling index [LI; BrdU incorporation] were determined as previously described (20). Figure 1E shows representative BrdU stained skin sections, corresponding to both acetone and TPA treated skins excised from lean, normal, overweight and obese mice and quantitative analyses are shown in Figure 1F. In the absence of TPA treatment, 30% CR significantly reduced both epidermal thickness and LI, when compared to all other groups, however 15% CR only significantly reduced both parameters when compared to the DIO group (p < 0.05, Mann-Whitney U test). No differences in either epidermal thickness or LI were observed between the overweight control and DIO groups in the absence of TPA treatment. In contrast, both epidermal thickness and LI were significantly different among all dietary groups following TPA treatment, with the greatest differences again occurring between the 30% CR and DIO groups (p < 0.05, Mann-Whitney U test).
Effect of dietary manipulation on growth factor signaling and cell cycle regulatory proteins during tumor promotion
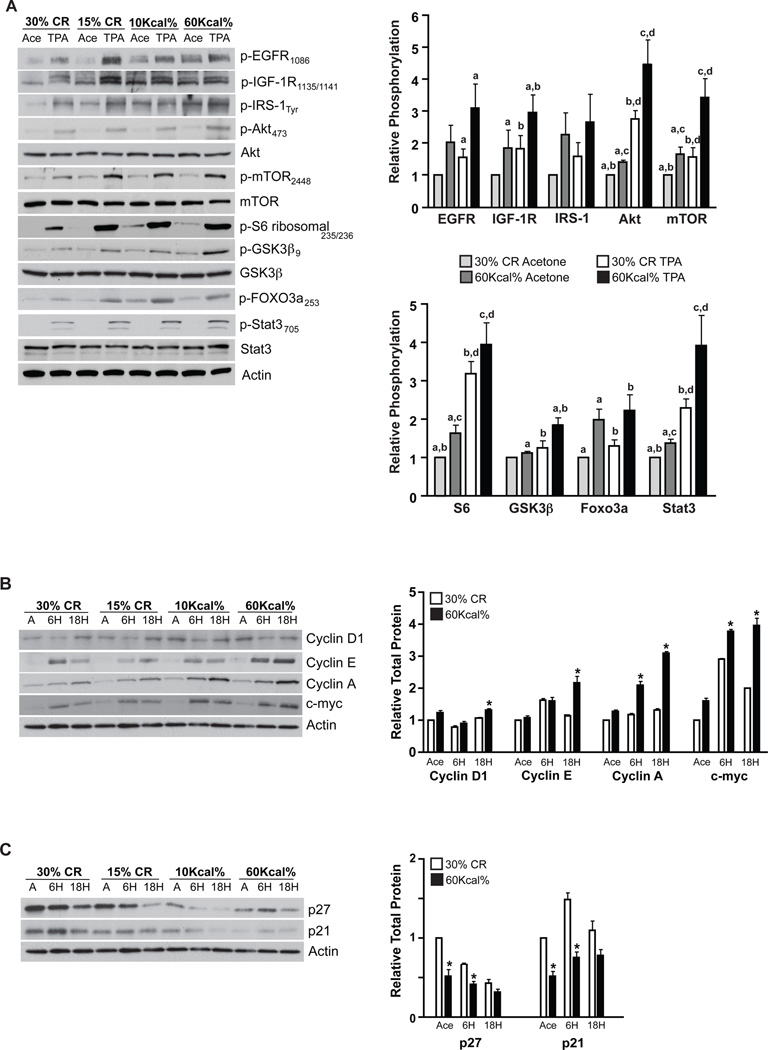
Diet-induced changes in growth factor signaling pathways were also assessed. ICR mice were maintained on the four diets for 15 weeks, treated with a single application of either acetone or 3.4 nmol of TPA and killed 6 hours later. Epidermal lysates were prepared for Western blot analyses as previously described (19, 20). As shown in Figure 2A, phosphorylation of the EGFR, IGF-1R and downstream signaling targets (i.e., Akt, mTOR, S6 ribosomal, GSK-3β, Foxo3a, Stat3) correlated directly with caloric density in the absence of TPA treatment, corroborating our previous findings indicating that dietary energy balance modulated steady-state growth factor signaling (19). Diet-induced changes in EGFR/IGF-1R activation and downstream signaling were further enhanced following TPA treatment, with significant differences in phosphorylation occurring between the 30% CR and DIO groups (Figure 2A). Specifically, 30% CR significantly reduced activation and/or phosphorylation of the EGFR, IGF-1R, Akt, mTOR, S6 ribosomal, GSK-3β, Foxo3a and Stat3, as compared to the DIO group (p < 0.05, Student’s t-test; Figure 2A).
Figure 2.
Effect of dietary manipulation on growth factor signaling and cell cycle regulatory proteins in epidermis following treatment with TPA. ICR female mice maintained on the four diets for 15 weeks were treated with a single application of acetone or 3.4 nmol TPA and killed 6 hours (growth factor signaling, cell cycle) or 18 hours (cell cycle) after treatment (n=6). Epidermal lysates were pooled for Western blot analyses. A, Growth factor signaling changes in epidermis 6 hours after TPA treatment (left) and densitometry graph representing mean ± SEM of three independent experiments (right) (30% CR acetone, light grey bars; 60Kcal% acetone, dark grey bars; 30% CR TPA, white bars; 60Kcal% TPA, black bars). Western blot data was normalized to both actin and total protein. a–d Significantly different from similarly labeled values (p < 0.05; Student’s t-test). B, Changes in positive and C, Changes in negative cell cycle regulatory proteins at 6 and 18 h following TPA treatment. Densitometry quantitation graphs (shown to the right of the Western blots in B and C represent mean ± SEM of three independent experiments (30% CR, white bars; 60Kcal%, black bars) in which Western blot data was normalized to actin. *Significantly different from the corresponding time point (p < 0.05; Student’s t-test).
We also evaluated diet-induced changes in cell cycle regulatory proteins. Epidermal protein lysates were prepared as above with the addition of an 18-hour time point. Initially, we analyzed changes in positive cell cycle regulatory protein levels. CR reduced epidermal levels of cyclin D1, cyclin E, cyclin A and c-myc compared to DIO, however, these differences were not statistically significant (Figure 2B). TPA treatment led to increased levels of cyclin D1 (18 h), cyclin E (6, 18 h), cyclin A (6, 18 h) and c-myc (6, 18 h) (25 and see Figure 2B). The TPA-mediated increases in epidermal cyclin D1, cyclin E, cyclin A and c-myc were significantly attenuated by 30% CR, compared to DIO, at 18 hours (p < 0.05, Student’s t-test). Diet-induced changes in cyclin A and c-myc levels following TPA treatment were also significant at the 6-hour time point (p < 0.05, Student’s t-test).
Additional analyses were performed to determine the impact of dietary energy balance on levels of negative cell cycle regulatory proteins. Levels of both p27 and p21 were significantly higher in 30% CR mice (vehicle control), as compared to DIO mice (Figure 2C). Following TPA treatment (6 h), both p27 and p21 levels were significantly higher in mice maintained on the 30% CR diet, as compared to mice maintained on the DIO diet (p < 0.05, Student’s t-test). A similar trend was observed at 18 hours, although these differences were not significantly different. Preliminary experiments (data not shown) demonstrated increased nuclear localization of both p27 and p21 in the epidermis of CR mice, as compared to mice maintained on the DIO regimen, suggesting that dietary energy balance may also modulate these negative cell cycle regulatory proteins through additional mechanisms.
Activation of the IGF-1R stimulates IGF-1R/EGFR crosstalk in cultured mouse keratinocytes
Both our current and previous data suggested a role for altered IGF-1 levels in modulating the effect of dietary energy balance on epithelial carcinogenesis. A reduction in circulating IGF-1, either due to genetic (LID mouse model) or dietary manipulation (CR) attenuated both IGF-1R and EGFR signaling (19, 20 and see Figure 2), indicating a potential for crosstalk between these two receptors. C50 cells were used to examine the role of IGF-1 in modulating both IGF-1R and EGFR signaling and crosstalk, as well as erbB2 activation. Cultured cells were stimulated with IGF-1 (25 ng/mL), harvested at multiple time points (0–120 minutes) and analyzed for IGF-1R activation (measured by IRS-1 phosphorylation) and for EGFR and erbB2 activation (measured by phosphorylation) using Western blot analysis. IGF-1 treatment significantly induced activation of all three cell surface receptors (Fig. 3). EGFR phosphorylation was rapid, with maximal activation occurring within 5 minutes and phosphorylation was maintained above the basal level up to 120 minutes. EGFR phosphorylation correlated directly with IGF-1 induced activation of the IGF-1R. Rapid phosphorylation of erbB2 also paralleled both IGF-1R and EGFR activation; however, erbB2 activation was maintained at a consistent level across all time points examined. Stimulation of C50 cells with EGF (10 ng/mL) over the same time course led to rapid activation of both the EGFR and erbB2, however, there was no significant effect on IGF-1R activation (Fig. 3).
Figure 3.
Effect of IGF-1 and EGF on receptor activation in keratinocytes. C50 cells were serum and growth factor starved for 24 hours, stimulated with either IGF-1 (25 ng/mL) or EGF (10 ng/mL) and harvested at multiple time points (0–120 minutes). A, Western blot analyses evaluating EGFR, erbB2 and IGF-1R activation in cell lysates. B, Quantitation. Data shown represents mean ± SEM of three independent experiments (IRS-1, white bars; EGFR, black bars; erbB2, gray bars). IGF-1 stimulation significantly increased activation of all targets at all time points, while EGF stimulation significantly increased EGFR and erbB2 activation at all time points (p < 0.05; Student’s t-test).
Previous studies have identified several potential mechanisms for IGF-1R and EGFR crosstalk (26–30). The ability of IGF-1 to induce heterodimerization between the IGF-1R and the EGFR was evaluated in co-immunoprecipitation experiments performed using lysates prepared from C50 cells treated with either IGF-1 or EGF. As shown in Figure 4A, IGF-1 treatment induced a statistically significant increase in IGF-1R/EGFR association from 5 to 60 minutes. In contrast, EGF had no observable effect. Additional experiments were performed to determine the impact of IGF-1 stimulation on levels of mRNA for the EGFR and EGFR ligands. qPCR was performed using RNA extracted from C50 cells (stimulated with IGF-1 or EGF and harvested at 0–120 minutes) to determine the relative expression of EGFR, TGF-α, HB-EGF and amphiregulin. Both IGF-1 and EGF stimulation significantly increased expression of HB-EGF and amphiregulin, with a greater induction occurring with EGF treatment (Fig. 4B). These relative increases in mRNA expression, however, were observed only at later time points (≥ 60 minutes). No increases in TGF-α and EGFR mRNA expression were observed following stimulation of C50 cells with either IGF-1 or EGF (Fig. 4B).
Figure 4.
Effect of IGF-1 and EGF on IGF-1R/EGFR association and EGFR ligand mRNA levels. C50 cells were cultured and harvested as described in Figure 4 and lysate was prepared for co-immunoprecipitation experiments and mRNA expression analyses. A, Effect of IGF-1 and EGF stimulation on IGF-1R/EGFR association. Right panel shows relative quantitation of mean ± SEM of three independent experiments (IGF-1, white bars; EGF, black bars). EGFR and IGF-1R were normalized to IgG and then to each other. *Denotes significant increase in IGF-1R/EGFR association, relative to the 0 time point (p < 0.05; Student’s t-test). B, Effect of IGF-1 (white bars) and EGF (black bars) stimulation on EGFR and EGFR ligand (TGF-α, HB-EGF, Amphiregulin, and EGFR) mRNA expression by qPCR analysis. Data shown represents mean ± SEM of three independent experiments. *Significant increase in EGFR ligand mRNA expression following EGF stimulation; **Significant increase in EGFR ligand mRNA following both IGF-1 and EGF stimulation (p < 0.05; Student’s t-test).
Dietary energy balance alters IGF-1R/EGFR crosstalk in vivo
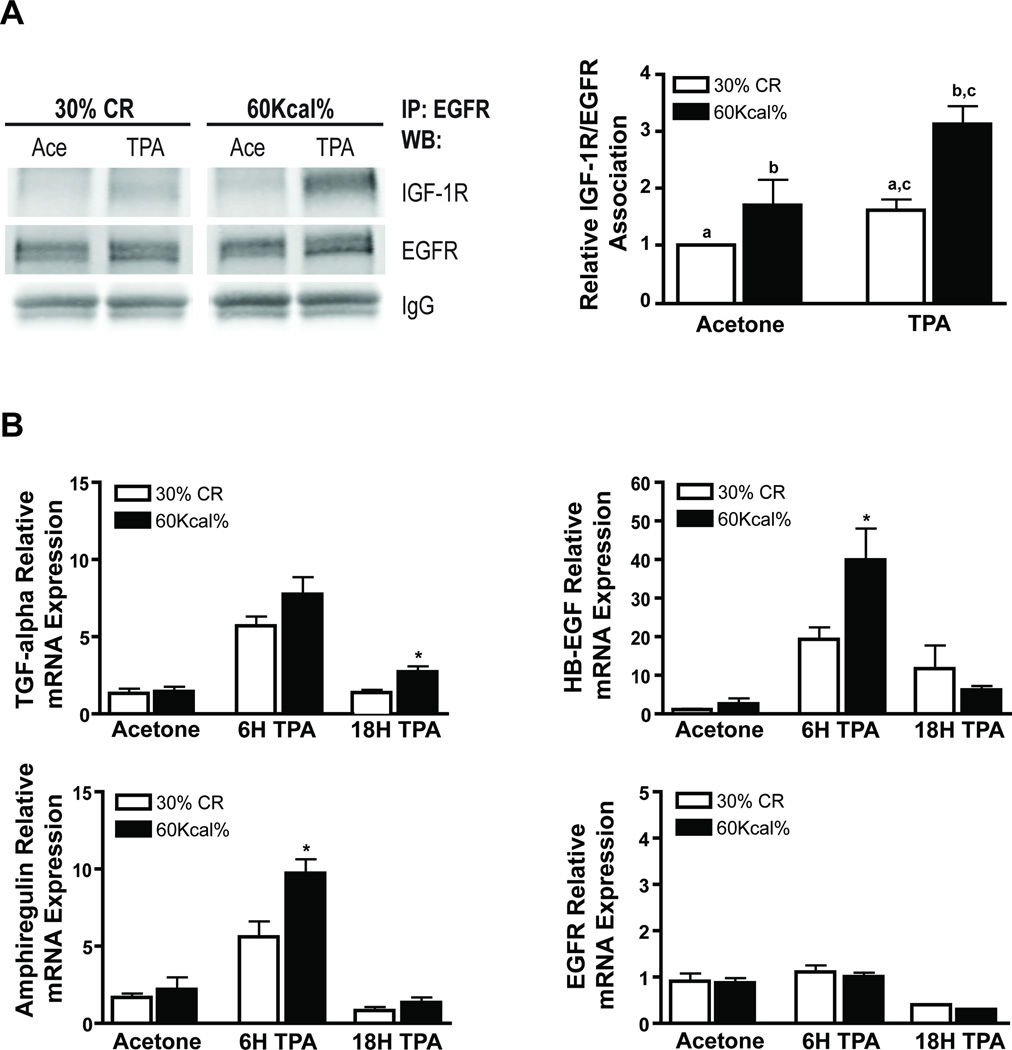
Finally, we evaluated the impact of dietary energy balance manipulation on IGF-1R/EGFR crosstalk during tumor promotion in mouse epidermis in vivo. Female ICR mice were maintained on the previously described 30% CR and DIO diets for 15 weeks. These lean and obese mice were then treated with a single application of acetone (vehicle) or 3.4 nmol of TPA and killed 6 or 18 hours later. Epidermal lysates and epidermal RNA were prepared for Western blot and qPCR analyses, respectively. Co-immunoprecipitation experiments were performed to evaluate diet-induced changes in IGF-1R/EGFR heterodimerization. As shown in Figure 5A, there was a greater degree of association between the IGF-1R and EGFR in obese mice relative to CR mice in the absence of TPA treatment. Furthermore, TPA (6 h) induced a significant increase in IGF-1R and EGFR association in both the CR and obese mice although the relative increase in heterodimerization was significantly greater in obese mice (p < 0.05, Student’s t-test).
Figure 5.
Effect of dietary manipulation on IGF-1R/EGFR heterodimerization and EGFR and EGFR ligand mRNA expression. ICR female mice were maintained on a 30% CR (white bars) and 60Kcal% fat (black bars) diet and treated with TPA as described in Figure 2 (n=6). Epidermal lysates were pooled and prepared for co-immunoprecipitation experiments and qPCR analysis. A, Co-immunoprecipitation with EGFR and subsequent Western blot analyses for EGFR and IGF-1R (left panel); representative densitometry graph (right panel). EGFR and IGF-1R were normalized to IgG and then to each other. Data shown represents mean ± SEM of three independent experiments. a–c Values with the same lettering indicate statistically significant differences (p < 0.05, Student’s t-test). B, qPCR analysis of EGFR and the EGFR ligands TGF-α, HB-EGF, Amphiregulin, and EGFR. Data represents mean ± SEM. * Significantly different from 30% CR group at the corresponding time point (P < 0.05, Mann-Whitney U test).
Dietary manipulation did not modulate mRNA levels of EGFR and EGFR ligands in the absence of TPA treatment; however, DIO significantly increased levels of mRNA for EGFR ligands compared to 30% CR, following treatment with TPA (Fig. 5B). Specifically, DIO significantly increased mRNA levels of TGF-α (18 h), HB-EGF (6 h) and amphiregulin (6 h) following TPA treatment. EGFR mRNA levels were not affected by diet or treatment with TPA. Collectively, the in vitro and in vivo results suggest that dietary energy balance modulates IGF-1R/EGFR crosstalk at least in part due to diet-induced changes in mRNA levels of EFGR ligands and changes in IGF-1R and EGFR heterodimerization.
Discussion
In this study potential mechanism(s) underlying the effects of dietary energy balance manipulation on skin tumor formation during the promotion phase of two-stage skin carcinogenesis were examined. Consistent with previously published data, both 15% and 30% CR significantly inhibited the promotion of papillomas by TPA compared to both the overweight control and DIO groups, but had no significant effect on tumor progression (8–10). Notably, no significant difference in tumor response was seen between the DIO and overweight control groups, although the DIO regimen significantly increased both body mass and percent body fat. Previous mammary carcinogenesis studies also reported that despite significant differences in body mass, no significant increase in tumor incidence occurred when dietary fat consumption exceeded 20–30Kcal% (31–33). These data support the hypothesis that in some model systems, there is an upper limit to the effects of a positive energy balance state above which further increases in total calories do not further increase tumor response.
Genetic reduction of circulating IGF-1 levels (i.e., LID mouse model) attenuated TPA-induced epidermal hyperproliferation, which correlated with reduced responsiveness to skin tumor promotion by TPA (20). In comparison to the overweight control and DIO groups both the 30% and 15% CR groups had significantly reduced epidermal thickness and LI in the absence of TPA. Following TPA treatment, we observed a significantly progressive effect of dietary energy balance across all the diet groups on the epidermal proliferative response, with 30% CR exhibiting the lowest response and DIO exhibiting the highest. Several other studies have demonstrated that energy balance impacts cellular proliferation in both normal and tumorigenic tissue (e.g., mammary, colon, liver, epidermis, and bladder) (34–38). Collectively, our results suggest that manipulating dietary energy balance impacts TPA-mediated epidermal hyperproliferation thus modulating epithelial carcinogenesis in mouse skin.
Previous studies have indicated an important role for IGF-1R and EGFR activation in TPA-induced epidermal hyperproliferation and tumor promotion in mouse skin (reviewed in 39). Inhibition of TPA-induced EGFR activation in vivo, using either RG13022 (tyrosine kinase inhibitor) or GW2974 (dual specific erbB2/EGFR inhibitor) significantly reduced TPA-induced epidermal hyperproliferation as well as skin tumor promotion (GW2974) (40, 41). Additionally, epidermal IGF-1 overexpression increased epidermal proliferation, both in the presence and absence of TPA, and increased sensitivity to two-stage skin carcinogenesis (42, 43). Furthermore, dietary energy balance induced changes in steady-state activation of these critical growth factor receptors in epidermis and other epithelial tissues of FVB/N and C57BL/6 mice (19). As shown in Figure 2A, 30% CR reduced activation and/or phosphorylation of the IGF-1R, EGFR and downstream signaling following treatment with TPA compared to the DIO group. Xie et al., reported a similar reduction in TPA-mediated epidermal Akt activation in CR mice (18).
The impact of dietary manipulation on levels of positive and negative cell cycle regulatory proteins was also evaluated. Notably, in mice treated with TPA, the levels of positive cell cycle regulatory proteins (cyclin D1, Cyclin E, Cyclin A, c-myc) correlated directly with caloric consumption, while the levels of negative cell cycle regulatory proteins (i.e., p21, p27) inversely correlated with caloric consumption. As reported earlier, elevated IGF-1R activation or increased Akt activity, respectively, leads to increased levels of positive cell cycle regulatory proteins (cyclins D, A, E, c-myc) during tumor promotion, providing a mechanism for the up-regulation of cellular proliferation observed in mouse epidermis after treatment with TPA (44, 45). Together, these observations suggest that dietary energy balance-induced alterations in epidermal proliferation seen during tumor promotion result from altered upstream growth factor signaling which modulates levels of both positive and negative cell cycle regulatory proteins.
Using C50 cells, we established that IGF-1 treatment specifically induced activation of not only the IGF-1R, but also the EGFR and erbB2. Similar to previous studies (29, 30), EGF stimulation did not activate the IGF-1R, suggesting that IGF-1R/EGFR crosstalk may be unidirectional. Thus, IGF-1 modulated activation of the EGFR in cultured keratinocytes through multiple mechanisms, including induction of IGF-1R/EGFR heterodimerization and heightened expression of EGFR ligand mRNA. Both mechanisms likely contribute to the sustained activation of the EGFR observed at later time points, however, they do not fully explain the rapid induction of EGFR and erbB2 phosphorylation observed following IGF-1 stimulation. EGFR phosphorylation may also occur as a result of IGF-1 mediated ectodomain shedding of preformed, membrane bound EGFR ligands (29, 30, 46), which may explain the early activation of the EGFR by IGF-1 observed in our studies. The activation (phosphorylation) of erbB2 seen in IGF-1 treated cells is likely due to EGFR:erbB2 heterodimerization, although we cannot completely rule out the possibility that other mechanisms may be involved, including heterodimerization between IGF-1R and erbB2 (47, 48). Finally, we conducted in vivo experiments to determine if dietary energy balance modulates IGF-1R/EGFR crosstalk. CR (30%) not only reduced IGF-1R/EGFR heterodimerization in epidermis following TPA treatment, but also significantly reduced mRNA expression of EGFR ligands, as compared to DIO. To our knowledge, this is the first report demonstrating that dietary energy balance impacts IGF-1R and EGFR crosstalk. These diet-related changes in receptor crosstalk account, at least in part, for the observed reduction (CR) or increase (DIO) in epidermal signaling that controls the proliferative response to TPA during tumor promotion.
In the current study, the two-stage skin carcinogenesis model was used primarily as a general model of epithelial carcinogenesis to evaluate mechanisms associated with dietary energy balance manipulation. Nevertheless, the observed changes may also be relevant to UVB-mediated skin carcinogenesis. In this regard, diet-induced obesity has been shown to increase UVB-mediated inflammation as well as inflammation and cell survival signaling (49). Akt activation (phosphorylation) was found to be elevated in the epidermis of obese mice exposed to UVB compared to mice on a control diet in this study. In a model of leptin deficiency-induced obesity (i.e., ob/ob mice), similar increases in UVB-induced inflammation signaling (NFκB, COX-2, TNFα) and survival signaling (Akt activation) were observed compared to wild-type mice (50). Elevated levels of PCNA and cyclin D1 were also observed in epidermis of the obese mice following exposure to UVB. Thus, a number of changes similar to those seen in our current study have been observed in skin/epidermis of obese mice (either diet-induced or genetically-induced obesity) following exposure to UVB. In a study by Hopper et al. (51), CR was reported to inhibit UVB-induced upregulation of AP-1:DNA binding and UVB-induced alteration in AP-1 constituent protein levels suggesting that CR inhibited events associated with UVB-mediated skin tumor promotion. Only limited information is available on the potential link between dietary energy balance and non-melanoma skin cancer (NMSC) in humans (reviewed in 52), however, stronger evidence points to a relationship between obesity and melanoma skin cancer (reviewed in 52). Further studies evaluating the role of dietary energy balance in both NMSC and melanoma skin cancer seem warranted based on the data presented in our current studies as well as those published on UV effects in mouse models of obesity as noted above.
In conclusion, we have shown that CR reduced, while DIO increased, signaling through the IGF-1R and EGFR following TPA treatment. Both in vitro and in vivo studies (20) suggest that levels of circulating IGF-1, which are modulated by dietary energy balance, regulate activation of the IGF-1R which in turn regulates crosstalk with the EGFR (and erbB2). These diet-induced changes in IGF-1R and EGFR activation subsequently altered downstream signaling to Akt and mTOR (and other signaling pathways), thus modulating levels and activity of cell cycle regulatory proteins. These changes in cell cycle regulatory proteins correlated directly with diet-induced changes in TPA-induced epidermal proliferation. Supplemental Figure 1 summarizes the effects of dietary energy balance on signaling pathways and cell cycle proteins observed in the present study in relation to altered epidermal proliferation and tumor promotion. Taken together, these effects of dietary energy balance on IGF-1R/EGFR signaling and crosstalk, cell cycle regulation and epidermal proliferation provide a plausible mechanism for the inhibitory effects of negative energy balance and the enhancing effects of positive energy balance on susceptibility to skin tumor promotion during two-stage skin carcinogenesis. These studies provide new information about potential molecular targets for prevention of epithelial cancers and for reversing the effects of obesity on cancer development.
Supplementary Material
Acknowledgments
Grant support: This work was supported by NIH Grant CA129409 and NIEHS Center Grant P30ES007784. T. Moore was supported by NIEHS training grant ES007247.
Footnotes
There are no conflicts of interest.
References
- 1.Patel AC, Nunez NP, Perkins SN, Barrett JC, Hursting SD. Effects of energy balance on cancer in genetically altered mice. J Nutr. 2004;134:3394S–3398S. doi: 10.1093/jn/134.12.3394S. [DOI] [PubMed] [Google Scholar]
- 2.Calle EE, Kaaks R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat Rev Cancer. 2004;4:579–591. doi: 10.1038/nrc1408. [DOI] [PubMed] [Google Scholar]
- 3.Park EJ, Lee JH, Yu GY, He G, Ali SR, Holzer RG, et al. Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell. 2010;140:197–208. doi: 10.1016/j.cell.2009.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yakar S, Nunez NP, Pennisi P, Brodt P, Sun H, Fallavollita L, et al. Increased Tumor Growth in Mice with Diet-Induced Obesity: Impact of Ovarian Hormones. Endocrinology. 2006 doi: 10.1210/en.2006-0311. [DOI] [PubMed] [Google Scholar]
- 5.Nunez NP, Perkins SN, Smith NC, Berrigan D, Berendes DM, Varticovski L, et al. Obesity accelerates mouse mammary tumor growth in the absence of ovarian hormones. Nutr Cancer. 2008;60:534–541. doi: 10.1080/01635580801966195. [DOI] [PubMed] [Google Scholar]
- 6.Stewart JW, Koehler K, Jackson W, Hawley J, Wang W, Au A, et al. Prevention of mouse skin tumor promotion by dietary energy restriction requires an intact adrenal gland and glucocorticoid supplementation restores inhibition. Carcinogenesis. 2005;26:1077–1084. doi: 10.1093/carcin/bgi051. [DOI] [PubMed] [Google Scholar]
- 7.Hursting SD, Nunez NP, Patel AC, Perkins SN, Lubet RA, Barrett JC. The utility of genetically altered mouse models for nutrition and cancer chemoprevention research. Mutat Res. 2005;576:80–92. doi: 10.1016/j.mrfmmm.2004.11.019. [DOI] [PubMed] [Google Scholar]
- 8.Boutwell RK. Some biological aspects of skin carcinogenesis. Prog Exp Tumor Res. 1964;4:207–250. doi: 10.1159/000385978. [DOI] [PubMed] [Google Scholar]
- 9.Birt DF, Pelling JC, White LT, Dimitroff K, Barnett T. Influence of diet and calorie restriction on the initiation and promotion of skin carcinogenesis in the SENCAR mouse model. Cancer Res. 1991;51:1851–1854. [PubMed] [Google Scholar]
- 10.Birt DF, Pinch HJ, Barnett T, Phan A, Dimitroff K. Inhibition of skin tumor promotion by restriction of fat and carbohydrate calories in SENCAR mice. Cancer Res. 1993;53:27–31. [PubMed] [Google Scholar]
- 11.Abel EL, Angel JM, Kiguchi K, DiGiovanni J. Multi-stage chemical carcinogenesis in mouse skin: fundamentals and applications. Nat Protoc. 2009;4:1350–1362. doi: 10.1038/nprot.2009.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hursting SD, Berger NA. Energy balance, host-related factors, and cancer progression. J Clin Oncol. 2010;28:4058–4065. doi: 10.1200/JCO.2010.27.9935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hursting SD, Smith SM, Lashinger LM, Harvey AE, Perkins SN. Calories and carcinogenesis: lessons learned from 30 years of calorie restriction research. Carcinogenesis. 2010;31:83–89. doi: 10.1093/carcin/bgp280. [DOI] [PubMed] [Google Scholar]
- 14.Yaktine AL, Vaughn R, Blackwood D, Duysen E, Birt DF. Dietary energy restriction in the SENCAR mouse: elevation of glucocorticoid hormone levels but no change in distribution of glucocorticoid receptor in epidermal cells. Mol Carcinog. 1998;21:62–69. [PubMed] [Google Scholar]
- 15.Birt DF, Yaktine A, Duysen E. Glucocorticoid mediation of dietary energy restriction inhibition of mouse skin carcinogenesis. J Nutr. 1999;129:571S–574S. doi: 10.1093/jn/129.2.571S. [DOI] [PubMed] [Google Scholar]
- 16.Birt DF, Przybyszewski J, Wang W, Stewart J, Liu Y. Identification of molecular targets for dietary energy restriction prevention of skin carcinogenesis: an idea cultivated by Edward Bresnick. J Cell Biochem. 2004;91:258–264. doi: 10.1002/jcb.10741. [DOI] [PubMed] [Google Scholar]
- 17.Liu Y, Duysen E, Yaktine AL, Au A, Wang W, Birt DF. Dietary energy restriction inhibits ERK but not JNK or p38 activity in the epidermis of SENCAR mice. Carcinogenesis. 2001;22:607–612. doi: 10.1093/carcin/22.4.607. [DOI] [PubMed] [Google Scholar]
- 18.Xie L, Jiang Y, Ouyang P, Chen J, Doan H, Herndon B, et al. Effects of dietary calorie restriction or exercise on the PI3K and RAS signaling pathways in the skin of mice. J Biol Chem. 2007 doi: 10.1074/jbc.M604857200. [DOI] [PubMed] [Google Scholar]
- 19.Moore T, Beltran L, Carbajal S, Strom S, Traag J, Hursting SD, et al. Dietary energy balance modulates signaling through the Akt/mammalian target of rapamycin pathways in multiple epithelial tissues. Cancer Prev Res. 2008;1:65–76. doi: 10.1158/1940-6207.CAPR-08-0022. [DOI] [PubMed] [Google Scholar]
- 20.Moore T, Carbajal S, Beltran L, Perkin SN, Yakar S, LeRoith D, et al. Reduced susceptibility to two-stage skin carcinogenesis in mice with low circulating IGF-1 levels. Cancer Res. 2008;68:3680–3688. doi: 10.1158/0008-5472.CAN-07-6271. [DOI] [PubMed] [Google Scholar]
- 21.Blando J, Moore T, Hursting S, Jiang G, Saha A, Beltran L, et al. Dietary energy balance modulates prostate cancer progression in hi-myc mice. Cancer Prev Res (Phila) 2011;4:2002–2014. doi: 10.1158/1940-6207.CAPR-11-0182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Berrigan D, Lavigne JA, Perkins SN, Nagy TR, Barrett JC, Hursting SD. Phenotypic effects of calorie restriction and insulin-like growth factor-1 treatment on body composition and bone mineral density of C57BL/6 mice: implications for cancer prevention. In Vivo. 2005;19:667–674. [PubMed] [Google Scholar]
- 23.He G, Thuillier P, Fischer SM. Troglitazone inhibits cyclin D1 expression and cell cycling independently of PPARgamma in normal mouse skin keratinocytes. J Invest Dermatol. 2004;123:1110–1119. doi: 10.1111/j.0022-202X.2004.23465.x. [DOI] [PubMed] [Google Scholar]
- 24.Nunez NP, Carpenter CL, Perkins SN, Berrigan D, Jaque SV, Ingles SA, et al. Extreme obesity reduces bone mineral density: complementary evidence from mice and women. Obesity (Silver Spring) 2007;15:1980–1987. doi: 10.1038/oby.2007.236. [DOI] [PubMed] [Google Scholar]
- 25.Segrelles C, Lu J, Hammann B, Santos M, Moral M, Cascallana L, et al. Deregulated activity of Akt in basal cells of stratified epithelia induces spontaneous tumors and heightened sensitivity to skin carcinogenesis. Cancer Res. 2007;67:10879–10888. doi: 10.1158/0008-5472.CAN-07-2564. [DOI] [PubMed] [Google Scholar]
- 26.Bor MV, Sorensen BS, Vinter-Jensen L, Flyvbjerg A, Pedersen SB, Nexo E. Epidermal growth factor and insulin-like growth factor I upregulate the expression of the epidermal growth factor system in rat liver. J Hepatol. 2000;32:645–654. doi: 10.1016/s0168-8278(00)80227-5. [DOI] [PubMed] [Google Scholar]
- 27.Nahta R, Yuan LX, Zhang B, Kobayashi R, Esteva FJ. Insulin-like growth factor-I receptor/human epidermal growth factor receptor 2 heterodimerization contributes to trastuzumab resistance of breast cancer cells. Cancer Res. 2005;65:11118–11128. doi: 10.1158/0008-5472.CAN-04-3841. [DOI] [PubMed] [Google Scholar]
- 28.Krane JF, Murphy DP, Carter DM, Krueger JG. Synergistic effects of epidermal growth factor (EGF) and insulin-like growth factor I/somatomedin C (IGF-I) on keratinocyte proliferation may be mediated by IGF-I transmodulation of the EGF receptor. J Invest Dermatol. 1991;96:419–424. doi: 10.1111/1523-1747.ep12469799. [DOI] [PubMed] [Google Scholar]
- 29.Roudabush FL, Pierce KL, Maudsley S, Khan KD, Luttrell LM. Transactivation of the EGF receptor mediates IGF-1-stimulated shc phosphorylation and ERK1/2 activation in COS-7 cells. J Biol Chem. 2000;275:22583–22589. doi: 10.1074/jbc.M002915200. [DOI] [PubMed] [Google Scholar]
- 30.El-Shewy HM, Kelly FL, Barki-Harrington L, Luttrell LM. Ectodomain shedding-dependent transactivation of epidermal growth factor receptors in response to insulin-like growth factor type I. Mol Endocrinol. 2004;18:2727–2739. doi: 10.1210/me.2004-0174. [DOI] [PubMed] [Google Scholar]
- 31.Cohen LA, Choi K, Weisburger JH, Rose DP. Effect of varying proportions of dietary fat on the development of N-nitrosomethylurea-induced rat mammary tumors. Anticancer Res. 1986;6:215–218. [PubMed] [Google Scholar]
- 32.Zevenbergen JL, Verschuren PM, Zaalberg J, van Stratum P, Vles RO. Effect of the amount of dietary fat on the development of mammary tumors in BALB/c-MTV mice. Nutr Cancer. 1992;17:9–18. doi: 10.1080/01635589209514168. [DOI] [PubMed] [Google Scholar]
- 33.Tang ZC, Shivapurkar N, Frost A, Alabaster O. The effect of dietary fat on the promotion of mammary and colon cancer in a dual-organ rat carcinogenesis model. Nutr Cancer. 1996;25:151–159. doi: 10.1080/01635589609514437. [DOI] [PubMed] [Google Scholar]
- 34.Lautenbach A, Budde A, Wrann CD, Teichmann B, Vieten G, Karl T, et al. Obesity and the associated mediators leptin, estrogen and IGF-I enhance the cell proliferation and early tumorigenesis of breast cancer cells. Nutr Cancer. 2009;61:484–491. doi: 10.1080/01635580802610115. [DOI] [PubMed] [Google Scholar]
- 35.James SJ, Muskhelishvili L. Rates of apoptosis and proliferation vary with caloric intake and may influence incidence of spontaneous hepatoma in C57BL/6 × C3H F1 mice. Cancer Res. 1994;54:5508–5510. [PubMed] [Google Scholar]
- 36.Steinbach G, Kumar SP, Reddy BS, Lipkin M, Holt PR. Effects of caloric restriction and dietary fat on epithelial cell proliferation in rat colon. Cancer Res. 1993;53:2745–2749. [PubMed] [Google Scholar]
- 37.Zhu Z, Jiang W, Thompson HJ. Effect of energy restriction on tissue size regulation during chemically induced mammary carcinogenesis. Carcinogenesis. 1999;20:1721–1726. doi: 10.1093/carcin/20.9.1721. [DOI] [PubMed] [Google Scholar]
- 38.Lok E, Scott FW, Mongeau R, Nera EA, Malcolm S, Clayson DB. Calorie restriction and cellular proliferation in various tissues of the female Swiss Webster mouse. Cancer Lett. 1990;51:67–73. doi: 10.1016/0304-3835(90)90232-m. [DOI] [PubMed] [Google Scholar]
- 39.Rho O, Kim DJ, Kiguchi K, Digiovanni J. Growth factor signaling pathways as targets for prevention of epithelial carcinogenesis. Mol Carcinog. 2010 doi: 10.1002/mc.20665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xian W, Kiguchi K, Imamoto A, Rupp T, Zilberstein A, DiGiovanni J. Activation of the epidermal growth factor receptor by skin tumor promoters and in skin tumors from SENCAR mice. Cell Growth Diff. 1995;6:1447–1455. [PubMed] [Google Scholar]
- 41.Kiguchi K, Kitamura T, Moore T, Rumi M, Chang H, Treece D, et al. Dual inhibition of both the EGFR and erbB2 effectively inhibits promotion of skin tumors during two-stage carcinogenesis. Cancer Prev Res. 2010 doi: 10.1158/1940-6207.CAPR-10-0010. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.DiGiovanni J, Bol DK, Wilker E, Beltran L, Carbajal S, Moats S, et al. Constitutive expression of insulin-like growth factor-1 in epidermal basal cells of transgenic mice leads to spontaneous tumor promotion. Cancer Res. 2000;60:1561–1570. [PubMed] [Google Scholar]
- 43.Wilker E, Bol D, Kiguchi K, Rupp T, Beltran L, DiGiovanni J. Enhancement of susceptibility to diverse skin tumor promoters by activation of the insulin-like growth factor-1 receptor in the epidermis of transgenic mice. Mol Carcinog. 1999;25:122–131. doi: 10.1002/(sici)1098-2744(199906)25:2<122::aid-mc7>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- 44.Segrelles C, Lu J, Hammann B, Santos M, Moral M, Cascallana L, et al. Deregulated activity of Akt in basal cells of stratified epithelia induces spotaneous tumors and heightened sensitivity to skin carcinogenesis. Cancer Res. 2007:10879–10888. doi: 10.1158/0008-5472.CAN-07-2564. [DOI] [PubMed] [Google Scholar]
- 45.Wilker E, Lu J, Rho O, Carbajal S, Beltran L, DiGiovanni J. Role of PI3K/Akt signaling in insulin-like growth factor-1 (IGF-1) skin tumor promotion. Mol Carcinog. 2005;44:137–145. doi: 10.1002/mc.20132. [DOI] [PubMed] [Google Scholar]
- 46.Saxena NK, Taliaferro-Smith L, Knight BB, Merlin D, Anania FA, O'Regan RM, et al. Bidirectional crosstalk between leptin and insulin-like growth factor-I signaling promotes invasion and migration of breast cancer cells via transactivation of epidermal growth factor receptor. Cancer Res. 2008;68:9712–9722. doi: 10.1158/0008-5472.CAN-08-1952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Earp H, Dawson T, Li X, Yu H. Heterodimerization and functional interaction between EGF receptor family members: A new signaling paradigm with implications for breast cancer research. Breast Cancer Research and Treatment. 1995;35:115–132. doi: 10.1007/BF00694752. [DOI] [PubMed] [Google Scholar]
- 48.Huang X, Gao L, Wang S, McManaman JL, Thor AD, Yang X, et al. Heterotrimerization of the growth factor receptors erbB2, erbB3, and insulin-like growth factor-i receptor in breast cancer cells resistant to herceptin. Cancer Res. 2010;70:1204–1214. doi: 10.1158/0008-5472.CAN-09-3321. [DOI] [PubMed] [Google Scholar]
- 49.Meeran SM, Singh T, Nagy TR, Katiyar SK. High-fat diet exacerbates inflammation and cell survival signals in the skin of ultraviolet B-irradiated C57BL/6 mice. Toxicol Appl Pharmacol. 2009;241:303–310. doi: 10.1016/j.taap.2009.09.003. [DOI] [PubMed] [Google Scholar]
- 50.Sharma SD, Katiyar SK. Leptin deficiency-induced obesity exacerbates ultraviolet B radiation-induced cyclooxygenase-2 expression and cell survival signals in ultraviolet B-irradiated mouse skin. Toxicol Appl Pharmacol. 2010;244:328–335. doi: 10.1016/j.taap.2010.01.010. [DOI] [PubMed] [Google Scholar]
- 51.Hopper BD, Przybyszewski J, Chen HW, Hammer KD, Birt DF. Effect of ultraviolet B radiation on activator protein 1 constituent proteins and modulation by dietary energy restriction in SKH-1 mouse skin. Mol Carcinog. 2009;48:843–852. doi: 10.1002/mc.20529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shipman AR, Millington GW. Obesity and the skin. Br J Dermatol. 2011;165:743–750. doi: 10.1111/j.1365-2133.2011.10393.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.