Abstract
The gut and Malpighian tubules of insects are the primary sites of active solute and water transport for controlling hemolymph and urine composition, pH, and osmolarity. These processes depend on ATPase (pumps), channels and solute carriers (Slc proteins). Maturation of genomic databases enables us to identify the putative molecular players for these processes. Anion transporters of the Slc4 family, AE1 and NDAE1, have been reported as HCO3− transporters, but are only part of the story. Here we report Dipteran (Drosophila melanogaster (d) and Anopheles gambiae (Ag)) anion exchangers, belonging to the Slc26 family, which are multi-functional anion exchangers. One Drosophila and two Ag homologues of mammalian Slc26a5 (prestin) and Slc26a6 (aka, PAT1, CFEX) were identified and designated dPrestin, AgPrestinA and AgPrestinB. dPrestin and AgPrestinB show electrogenic anion exchange (Cl−/nHCO3−, Cl−/SO42− and Cl−/oxalate2−) in an oocyte expression system. Since these transporters are the only Dipteran Slc26 proteins whose transport is similar to mammalian Slc26a6, we submit that Dipteran Prestin are functional and even molecular orthologues of mammalian Slc26a6. OSR1 kinase increases dPrestin ion transport, implying another set of physiological processes controlled by WNK/SPAK signaling in epithelia. All of these mRNAs are highly expressed in the gut and Malpighian tubules. Dipteran Prestin proteins appear suited for central roles in bicarbonate, sulfate and oxalate metabolism including generating the high pH conditions measured in the Dipteran midgut lumen. Finally, we present and discuss Drosophila genetic models that integrate these processes.
Keywords: Cl− transport, prestin, gut, Malpighian tubules, Slc26
Introduction
Ionic homeostasis is essential for all organisms to keep basic physiological functions. Compartmentalization of function is a second key concept of biology- at the level of organ systems, tissue, cells and even organelles. Regulating ion composition in these biological compartments is essential for proteins to act appropriately for establishing highly organized systems, and facilitating biological control: (a) of what moves into and out of the compartment; (b) sequestration and control of potentially toxic molecules needed for energy metabolism (e.g., O2, transition metals, H+, SO42−, oxalate2−, etc) ; (c) controlling rates of ion/solute movements. To achieve these functions, ion and solute transporters and channels are tightly regulated. Many of these functions are the domain of several kinds of proteins including solute carriers, known as Slc-proteins1. Active transport of ions by ATPases (pumps) maintains ionic gradients, while the presence and open probability of ion channels set membrane potentials. Secondary active transporters (i.e., Slc transporters) utilize one or more aspects of the membrane electrochemical gradient (individual [solute] gradient or electrical gradient, i.e., membrane potential, Vm) to specifically move ions and solutes into and out of the cellular compartments (for details and derivation, see (Romero et al., 2009)). The insect gut and Malpighian tubules carry out some of the most vital functions for maintenance of the solute composition, pH and osmolarity of the haemolymph within an appropriate range. At the same time, insect luminal pH of the gut (particularly the midgut) is also tightly regulated and shows high pH (> 10.0) (Boudko et al., 2001a), in part for the effective digestion of food. These findings indicate that insect gut is a place for active transport of ion, solute and water. This transport is performed by various Slc proteins, they are often poorly understood at molecular level, even in Diptera (Drosophila and Anopheles). However, the rapid increase in genomic information facilitates in silico identification of potential genes: Na+/K+-ATPase, V-ATPase, Na+/H+ antiporter (NHA) (Xiang et al., this issue), anion exchanger (AE) (Linser et al., this issue), Na+-driven Cl− HCO3− exchanger (NDAE1) (Romero et al., 2000b), etc. This study adds one more candidate molecule for understanding insect gut and Malpighian tubules physiology.
The Slc26 family contains multifunctional and highly versatile anion exchanger and channel proteins with intriguing roles in normal physiology and human pathophysiology (Dorwart et al., 2008; Mount and Romero, 2004a; Sindic et al., 2007). The human SLC26 transporter family consists of eleven members, with SLC26A10 likely being a pseudogene (Mount and Romero, 2004a). Among these members, three SLC26 genes are associated with human disease: SLC26A2 (DTDST), chondrodysplasis; SLC26A3 (DRA, CLD), congenital chloride-losing diarrhea; and SLC26A4 (Pendrin), Pendred syndrome and hereditary deafness (reviewed in (Mount and Romero, 2004a)). Most of the Slc26 proteins function primarily as anion exchangers transporting a wide variety of monovalent and divalent anions (sulfate, chloride, iodide, formate, oxalate, hydroxyl ion, and bicarbonate) (Kato and Romero, 2011; Mount and Romero, 2004a), whereas other Slc26 proteins function as chloride ion channels or anion-gated molecular motors (Dorwart et al., 2008; Mount and Romero, 2004a; Sindic et al., 2007). Slc26a6 is a well-characterized member, mediating Cl−/HCO3−, Cl−/SO42−, Cl−/oxalate2− and Cl−/formate− exchange. Slc26a6 is widespread and apically located in gut and renal epithelia. Because of these features, Slc26a6 can be involved in several physiological events, such as Cl− absorption and HCO3− secretion of pancreatic duct epithelia, sulfate absorption and bicarbonate secretion of intestinal epithelia, and oxalate excretion at intestine and renal tubules. Slc26a5, also known as prestin, is expressed in the outer hair cells (OHCs) of cochlea in the mammalian ear (Zheng et al., 2000) and involved in the auditory sensory system. Unlike other Slc26 transporters, mammalian Slc26a5 does not function as a transporter but rather as an anion- and voltage-sensitive molecular motor which alters OHC shape in response to Vm changes, i.e., the basis for cochlear amplification (Oliver et al., 2001). By contrast, non-mammalian Slc26a5 proteins (chicken and zebrafish) possesses Cl−, SO42− and oxalate2− transport activity (Schaechinger and Oliver, 2007).
The present manuscript reports the molecular cloning, phylogeny, localization and function of the first Dipteran (Drosophila and Anopheles gambiae) Slc26 members. Specifically, we report the localization pattern of their mRNA transcripts for dPrestin and AgPrestin. Dipteran Prestin function as Cl− exchange systems for HCO3−, oxalate2−, SO42− and formate. Potential signaling control via WNK/SPAK/OSR1 kinases and new Drosophila tools are discussed.
Results
Identification of candidate Dipteran HCO3− transporters
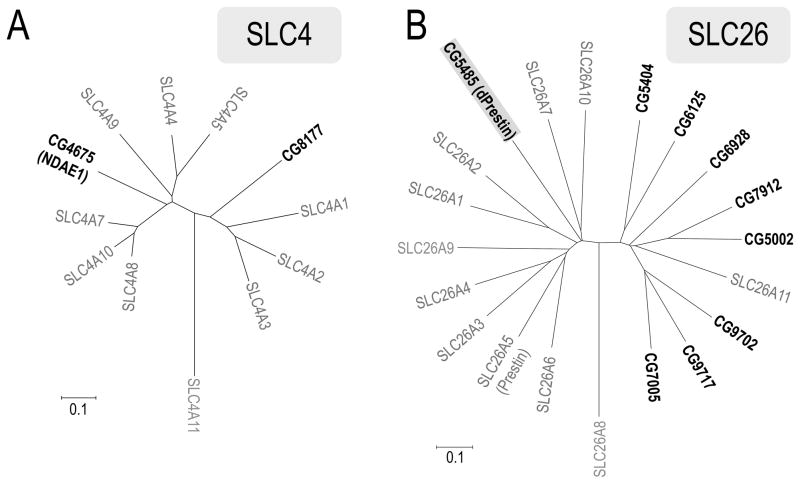
Presently there are 51 solute-leak carrier families (SLC) containing 378 transporter genes (categorized in the Human Genome Organization (HUGO) Nomenclature Committee Database (http://www.genenames.org/ and http://www.bioparadigms.org/slc/menu.asp) (Hediger et al., 2004). In these SLC families, SLC4 (Romero et al., 2004) and SLC26 (Mount and Romero, 2004b) contain mammalian HCO3− transporters. We therefore searched the Drosophila melanogaster genomic database for the orthologues of SLC4 and SLC26 family members and found two Slc4 and nine Slc26 homologues (Table 1). The Drosophila Slc4 proteins (CG4675/NDAE1 and CG8177) are summarized with human SLC4 members in the phylogenetic trees (Fig. 1A). Romero and coworkers functionally characterized CG4675 as a Na+-driven bicarbonate/chloride exchanger (NDAE1) (Romero et al., 2000a). By contrast, there are nine Drosophila Slc26 homologues. All Drosophila Slc26 sequences, except CG5485, are very close to SLC26A11 (Fig 1B), a human protein of unknown function. From Maximum Matching analysis using Genetyx software, CG5485 has a relatively high homology with human SLC26A5 (29.89%) and SLC26A6 (30.05%). The latter protein functions as a Cl−/HCO3− exchanger (Ko et al., 2002; Xie et al., 2002). CG5485 possesses a STAS (sulfate transporter anti-sigma) domain at the C-terminus which is a typical motif of Slc26 family members. This clone in Drosophila was previously called dPrestin (Weber et al., 2003)). Microarray analyses show that dPrestin is highly expressed in midgut, hindgut and Malpighian tubules (see discussion below and Fig 5A).
Table 1.
Slc4 and Slc26 genes in Drosophila and Anopheles
| Slc4
| ||
|---|---|---|
| Celera Gene ID | Clone name | Chr./Map posn |
|
| ||
| CG4675 | NDAE1 | 2L/27E6 |
| CG8177 | Cg8177 | 3L/67C1 |
|
| ||
| Slc26 | ||
|
| ||
| Celera Gene ID | Clone name | Chr./Map posn. |
| CG5002 | Slc26d5002 | 2R/54E7 |
| CG5404 | Slc26d 5404 | 3R/89A3-89A4 |
| CG5485 | dPrestin | 3L/75A8 |
| CG6125 | Slc26d6125 | 3R/88F3-88F4 |
| CG6928 | Slc26d6928 | 3L/68F1 |
| CG7005 | Slc26d7005 | 3R/96B2 |
| CG7912 | Slc26d7912 | 3R/99D3-99D4 |
| CG9702 | Slc26d9702 | 3R/100A1 |
| CG9717 | Slc26d9717 | 3R/100A1 |
Fig. 1. Phylogenetic tree of HCO3− transporters (A: SLC4 and B: SLC26) in Drosophila.
Results of the Drosophila melanogaster BLAST search revealed that there are two Slc4 clones and nine Slc26 clones. Neighbor-joining trees (Saitou 1987) were constructed based on the deduced amino acid sequences of Slc4 and Slc26 from Drosophila (black) and human (grey). Accession numbers are as follows: CG4675 (NDAE1), AF047468; CG8177, NM_140100; SLC4A1, NM_000342; SLC4A2, NM_003040; SLC4A3, NM_005070; SLC4A4, NM_003759; SLC4A5, NM_133478; SLC4A7, NM_003615; SLC4A8, NM_004858; SLC4A9, NM_031467; SLC4A10, NM_022058; SLC4A11, NM_032034; CG5485 (Drosophila Prestin), NM_140767; CG5002, AY240021; CG5404, NM_142225; CG6125, AY240022; CG6928, AY240023; CG7005, NM_079766; CG7912, NM_143504; CG9702, AY240025; CG9717, NM_143555; SLC26A1, AF297659; SLC26A2, BC059390; SLC26A3, BC025671; SLC26A4, AF030880; SLC26A5 (prestin), AF523354; SLC26A6, NM_134263; SLC26A7, AF331521; SLC26A8, BC025408; SLC26A9, BC136538; SLC26A10, NM_133489; SLC26A11, AF345195. Phylogenetic trees were constructed using the Clustal W computer program. The scale bar represents a genetic distance of 0.1 amino acid substitutions per site.
Fig. 5. Drosophila Prestin transport is activated by dOSR1.
A, B; Drosophila Prestin (dPrestin) activity at co-expression with dOSR1. Current-voltave (I-V) relationships of oocytes expressing dPrestin solely (filled square), dPrestin with wild-type dOSR1 (open square) and dOSR1 alone (open circle) in the presence of 0.33 mM oxalate (20 mM Cl−) (B) or 5 mM sulfate (20 mM Cl−) (C) are shown. Values are means ± SE, n = 3–5. Oxalate-elicited or sulfate-elicited currents are calculated as in Fig. 4
Drosophila Prestin transport function in Xenopus oocytes
To examine the dPrestin transport function, we expressed Prestin protein in Xenopus oocytes and performed electrophysiological experiments using ion-selective microelectrodes. First, cytosolic pH level (pHi) was monitored in response to exposure to Cl−-free medium (Fig 2Aa–c). Mouse Slc26a6 (mSlc26a6) was used as a positive ontrol. In oocytes expressing Drosophila Prestin and mSlc26a6, Cl− removal caused marked increase of pHi and a marked hyperpolarization. Readdition of Cl− elicited depolarization and suppression of pHi increase observed in Cl−-free medium (Fig 2Aa, b). Water-injected control oocytes had minimal background transport activity (Fig 2Ac). These results indicate that Drosophila Prestin mediate electrogenic Cl−/nHCO3− exchange (Fig 2Ad). Since Drosophila Prestin has high homology with SLC26A6, we next examined transport of oxalate, sulfate and formate (substrates for mSlc26a6 and SLC26A6) by monitoring intracellular chloride concentration ([Cl−]i) (Fig. 2B–D, a–c). In oocytes expressing Drosophila Prestin and mSlc26a6, Cl− removal caused marked reduction of [Cl−]i and a marked hyperpolarization. Readdition of Cl− elicited depolarization and recovery of [Cl−]i (Fig 2BCDa–c). Control oocytes did not show these responses (data not shown). These results indicate that Drosophila Prestin mediate electrogenic Cl−/oxalate2−, Cl−/sulfate2− and electroneutral Cl−/formate− exchange (Fig 2B–Dd). These activities are similar to those that we and others have reported for other species orthologues of Slc26a6 (Clark et al., 2008; Freel et al., 2006; Jiang et al., 2006; Jiang et al., 2002; Kato et al., 2009; Kurita et al., 2008; Monico et al., 2008; Wang et al., 2005; Xie et al., 2002). These data indicate that Drosophila Prestin is a functional orthologue of mammalian Slc26a6.
Fig. 2. Drosophila Prestin function in Xenopus oocytes.
A. Representative trances of intracellular pH (pHi) and membrane potential (Vm) of oocytes injected with Drosophila Prestin (dPrestin) cRNAs (a), mouse Slc26a6 (mSlc26a6) cRNAs (b), or water (control) (c) are shown. In the continuous presence of 33 mM HCO3−/5%CO2 (dot square), the Cl−/HCO3− exchange activities were monitored as changes in pHi and Vm when extracellular Cl− was removed (grey shading) and readded. Model illustrating the suggested transport activity (electrogenic Cl−/nHCO3− exchange activity) is shown in d. B. C. D. Representative traces of intracellular Cl− ([Cl−]i) and membrane potential (Vm) of oocytes injected with dPrestin cRNA (a), mSlc26a6 cRNAs (b) and water (control) (c) are shown in each tested substrates: oxalate (B), sulfate (C), and formate (D). Results for solution changes from 20 mM-Cl− ND96 (20Cl-ND96) to 20Cl-ND96 containing 0.33 mM oxalate, 5 mM sulfate, or 5 mM formate are indicated by dot square and results for solution changes to Cl−-free solution are indicated by gray shading. Models illustrating the suggested transport activity (electrogenic Cl−/oxalate2−, Cl−/sulfate2− and Cl−/formate− exchange activity) are shown in Bd, Cd and Dd, respectively.
Anopheles gambiae has two Prestin genes
A. gambiae has two Prestin homologous genes (AgPrestinA and AgPrestinB; both apparently paralogs of the same gene) with high homology to dPrestin (Fig 3A). AgPrestinB displays higher homology with dPrestin (46%) than AgPrestinA (37%). Both genes are located on chromosome 3. Microarray analysis (http://funcgen.vectorbase.org/ExpressionData/) (Neira Oviedo et al., 2009; Neira Oviedo et al., 2008) reveals that AgPrestinA mRNA is highly expressed in salivary glands and gastric caeca (Fig 3B). On the other hand AgPrestinB mRNA is highly expressed in posterior midgut and hindgut (Fig 3C).
Fig. 3. Phylogeny of Anopheles gambiae Prestin genes and their mRNA tissue distribution.
A. Phylogenetic tree of AgSls26a5/a6 including those of other several species. Results of the Anopheles gambiae BLAST search revealed that there are two Prestin related isoforms. Neighbor-joining trees (Saitou 1987) were constructed based on the deduced amino acid sequences of slc26a5 and slc26a6 from human (h), chicken (c), fugu (f), zebrafish (z), fly (d), and Anopheles gambiae (Ag). The bootstrap values from a 5,000-replicate analysis are given as % at the nodes. Accession numbers are as follows: hSLC26A5, AF523354; mSlc26a5, AF529192; cSlc26a5, EF028087; fSlc26a5; BAE75795, zSlc26a5, BC054604; AgPrestinA, GQ332421; AgPrestinB, AB671171; dPrestin, NM_140767; hSLC26A6, NM_134263; mSlc126a6, AY049076; fSlc26a6A, AB200328; fSlc26a6B, BAE75797; fSlc26a6C, BAE75798; zSlc26a6, BC155340. Phylogenetic trees were constructed using the Clustal W computer program. The scale bar represents a genetic distance of 0.05 amino acid substitutions per site. B. Tissue distribution of AgPrestinA and AgPrestinB. mRNA expression level (signal intensity in Y axis) of AgPrestinA and B was examined by microarray analysis using in each tissue: salivary gland (SG), gastric cacae (GC), anterior midgut (AMG), posterior midgut (PMG) and hindgut (HG). AgPrestinA is highly expressed in salivary glands and gastric cacae and AgPrestinB is expressed in posterior midgut and hindgut.
A. gambiae Prestin B has transport function
Oocytes were injected with AgPrestinA and AgPrestinB cRNAs and their intracellular pH level (pHi) was monitored in response to Cl−-removal as above for dPrestin experiments (Fig 4A). In oocytes expressing AgPrestinB, Cl− removal (0Cl) caused marked pHi increase and hyperpolarization (Fig 4Aa). By contrast, AgPrestinA-expressing oocytes and water-injected oocytes did not show these responses (Fig 4Bb). These results indicate that AgPrestinB possesses electrogenic Cl−/nHCO3− exchange (Fig 4Ac).
Fig. 4. AgPrestinB, but not AgPrestinA, has transport function.
A, Representative pHi/Vm experiments for oocytes injected with AgPrestinB cRNA (a) or water –control (b) are shown. In the continuous presence of 33 mM HCO3−/5%CO2 (dot square), the Cl−/HCO3− exchange activities were monitored as changes in pHi and Vm when extracellular Cl− is removed (grey shading) followed by readdition. Model illustrating the suggested transport activity (electrogenic Cl−/nHCO3− exchange activity) is shown in (c). (B) Current-voltage (IV) relationships of oocytes expressing AgPrestinB in the presence of 0.33 mM oxalate (20 mM Cl−) or (C) 5 mM sulfate (70 mM Cl−) are shown. Values are means ± SE, n = 3–5. Oxalate-elicited or sulfate-elicited currents are calculated as I (oxalate or sulfate) − I (no oxalate or ulfate).
To examine whether AgPrestinB mediates transport of oxalate and sulfate like dPrestin and mSlc26a6, we analyzed I-V relationships using two-electrode voltage-clamp (Fig 4BC). Both oxalate and sulfate caused voltage-dependent currents in AgPrestinB expressing oocytes, and these data indicate the electrogenic nature of Cl−/SO42− and Cl−/oxalate2− exchange (Fig 4BC).
Drosophila Prestin activity can be controlled by WNK/OSR1 signaling
Mammalian Slc26a6 and Slc26a9 have been reported to be regulated by WNK (with no lysine) kinases (Dorwart et al., 2007a; Plata et al., 2008; Yang et al., 2011). Therefore, we examined whether Drosophila Prestin is regulated by the WNK signaling pathway. Searching the Drosophila melanogaster genomic database reveals that Drosophila has only one WNK and only one SPAK-like protein, OSR1 (oxidative-stress-responsive kinase 1), while mammals have four WNK genes and several SPAK/OSR1 genes (Deaton et al., 2009; Delpire and Gagnon, 2008; Welling et al., 2010). Microarray analysis illustrates that these Drosophila WNK (dWNK) and Drosophila OSR1 (dOSR1) mRNAs are ubiquitously expressed in fly, including high levels in gut and Malpighian tubules (Table 2). Since Drosophila Prestin mRNA is co-localized with dWNK and dOSR1 mRNA in the gut and Malpighian tubules, we investigated if dPrestin function might be regulated by WNK and OSR1. Here we used dOSR1 as a molecule involved in the WNK signaling pathway. Thus, we co-expressed Drosophila Prestin and dOSR1 in Xenopus oocytes and compared I-V responses to oxalate and sulfate using two-electrode voltage-clamp (Fig 5A, B). Co-expression of dOSR1 with dPrestin increases both oxalate (Fig 5A) and sulfate transport currents (Fig 5B). To confirm whether dOSR1 itself mediates transport currents, we expressed dOSR1 solely and found that dOSR1 itself does not induce any currents (Fig 5A, B). These data indicate that the WNK/(SPAK/OSR1) signaling pathway can control dPrestin transport activities and may be physiologically important.
Table 2.
Drosophila Prestin, WNK and OSR1 mRNA expression
| Tissue | dPrestin | dWNK | dOSR1 |
|---|---|---|---|
| Head | 45 ± 2 | 114 ± 9 | 408 ± 41 |
| Salivary gland | 75 ± 8 | 206 ± 12 | 263 ± 14 |
| Midgut | 373 ± 29 | 141 ± 7 | 547 ± 13 |
| Tubule | 339 ± 34 | 124 ± 1 | 502 ± 41 |
| Hindgut | 266 ± 5 | 138 ± 1 | 892 ± 12 |
| Ovary | 160 ± 3 | 216 ± 7 | 609 ± 12 |
| Testis | 37 ± 2 | 140 ± 4 | 140 ± 5 |
| Larval CNS | 26 ± 4 | 146 ± 4 | 291 ± 19 |
| Larval Salivary | 50 ± 6 | 119 ± 7 | 674 ± 19 |
| Larval midgut | 313 ± 23 | 175 ± 13 | 507 ± 17 |
| Larval tubule | 318 ± 23 | 213 ± 3 | 421 ± 10 |
| Larval hindgut | 509 ± 30 | 169 ± 2 | 612 ± 43 |
| Larval fat body | 40 ± 6 | 110 ± 12 | 186 ± 3 |
| Larval trachea | 44 ± 4 | 89 ± 7 | 320 ± 28 |
| S2 cells | 230 ± 5 | 115 ± 0 | 541 ± 32 |
| Whole fly | 92 ± 9 | 97 ± 2 | 377 ± 15 |
Data from FlyAtlas.org (Chintapalli et al., 2007) quantifying the mRNA expression level of Drosophila Prestin (dPrestin), WNK (dWNK) and OSR1 (dOSR1) examined by microarray for adult tissues, larval tissues, S2 cells (growing) and whole fly. Noted in bold: dPrestin, dWNK and dOSR1 are coexpressed at midgut, hindgut and Malpighian tubules in both adult and larvae.
Discussion
Both SLC26 proteins (Dorwart et al., 2008; Mount and Romero, 2004a; Sindic et al., 2007) and certain SLC4 proteins exchange anions (Alper, 2010; Romero et al., 2009; Romero et al., 2004), with many causing serious human disorders when mutated. Database searches reveal that Slc26 family members exist in plants, animals and prokaryotes. Database searching of Drosophila melanogaster genomic DNA allowed us to find and then clone nine Slc26 homologues in the fly (Josephs et al., 2003; Josephs and Romero, 2004). Interestingly, eight paralogues are similar to SLC26A11 and only one orthologue (dPrestin) is similar to SLC26A1, A2, A3, A4, A5, A6 and A9, especially SLC26A5 and −A6 (Fig 2B). This analysis implies that vertebrate Slc26a5, Slc26a6 and fly (insect) Scl26a5/a6 share the same ancestral gene. At the molecular level, dScl26a5/a6 also has a sulfate transporter motif (amino acid residue 247–558) and a sulfate transporter anti-sigma factor antagonist (STAS) domain (amino acid residue 582–732) typical of Slc26 family members.
Functionally, dScl26a5/a6 can mediate the transport of Cl−, SO42−, oxalate2−, HCO3− and formate (Fig 2). In Drosophila Prestin mRNA is expressed in the midgut, hindgut and Malpighian tubules (Fig 5A). These same tissues also express dWNK and dOSR1 mRNA (Fig 5A). These features are very similar to those of vertebrate Slc26a6. Thus, it seems reasonable that dScl26a5/a6 is the functional orthologue of vertebrate Slc26a6.
Prestin is also present in Anopheles gambiae but there are two homologues (AgPrestinA and AgPrestinB) (Fig 3A). AgPrestinB is more similar to dPrestin than AgPrestinA at amino acid level. AgPrestinB shares mRNA expression patterns with dPrestin (midgut and hindgut) rather than AgPrestinA. Finally, AgPrestinB and dPrestin show similar physiological functions transporting Cl−, HCO3−, SO42− and oxalate2−.
The WNKs regulate many Na+, K+ and Cl− transporters involved in fluid and electrolyte homeostasis (Gamba, 2005; Subramanya et al., 2006), including Slc26 family members: Slc26a6 by WNK4 (Kahle et al., 2004) and Slc26a9 by WNK1, WNK3 and WNK4 (Dorwart et al., 2007b). The WNKs can regulate ion transporters directly by phosphorylating the transporters (Deaton et al., 2009; Delpire and Gagnon, 2008; Welling et al., 2010). Drosophila also has a WNK signaling system (frayed mutant). Interestingly Drosophila has only one WNK and one OSR1 homologue though vertebrates have several genes for each molecule. For example, human has four WNKs (WNK1-4) (Kahle et al., 2008) and two OSR1 related genes (OSR1 and SPAK) (Welling et al., 2010). Expression patterns of dPrestin mRNA overlap with dWNK and dOSR1 in the midgut, hindgut and Malpighian tubules (Table 2). In vitro expression with dPrestin illustrates that dOSR1 activates sulfate and oxalate transport activity of dPrestin (Fig 5A, B). Our results imply that Drosophila epithelial transport of Cl−, oxalate2−, SO42− (and perhaps HCO3−) in the gut and Malpighian tubule is regulated by a WNK/OSR1 signaling via dPrestin protein activation. These results further imply that Diptera (invertebrates) and Vertebrata share a common regulatory system to achieve highly organized epithelial transport.
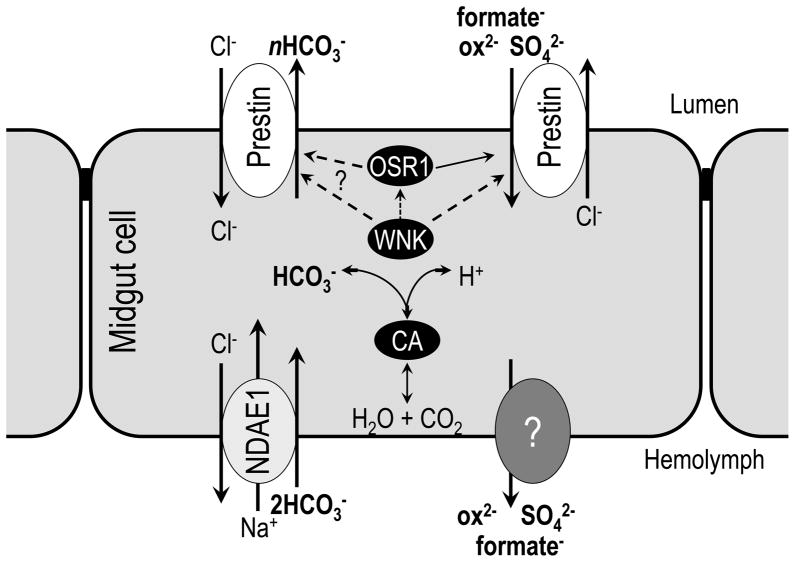
In Drosophila and mosquitos, luminal pH of the midgut is tightly regulated at each segmental region (Boudko et al., 2001a; Boudko et al., 2001b; Shanbhag and Tripathi, 2005, 2009). The luminal pH of the Drosophila middle midgut is less than 3.0, while the pH of the posterior midgut is greater than 10.0 (Boudko et al., 2001a; Boudko et al., 2001b; Jagadeshwaran et al., 2010; Onken and Moffett, 2009; Onken et al., 2008; Onken et al., 2009; Shanbhag and Tripathi, 2005, 2009). Moreover, the luminal pH of the gut segment between the gastric caeca and the anterior midgut in mosquito larvae is over 10.5 (Linser et al., 2011). Several ion carriers and enzymes are known to produce this wide range of pH differences in the gut lumen, such as V-type ATPase, Na+/K+ -ATPase, Na+/H+ antiporter (NHE, NHA), carbonic anhydrase (CA), etc (Linser et al., 2009; Okech et al., 2008). Since Diptera Prestin, elucidated in this study, transports bicarbonate (a major physiological buffer), it likely contributes to this luminal pH regulation in midgut. Fig. 6 presents our hypothesis of Prestin physiological function in the Diptera midgut for supplement of bicarbonate to gut luminal side. Prestin is expressed at the apical membrane of midgut cells and excretes bicarbonate which is produced by carbonic anhydrase or supplied by sodium-driven chloride-bicarbonate exchanger (NDAE1) from the basolateral side (Linser et al., 2011; Sciortino et al., 2001). WNK signaling may regulate this bicarbonate secretion thorough Prestin, allowing increased transport and thus increased buffering and increased midgut pH. Mutant analysis of Prestin and WNK as well as in vitro analysis of tubule and gut halide transport will allow us to test this hypothesis.
Fig 6.
A model of Slc26a5/a5 (prestin) function of HCO3−, SO42− and oxalate2− transport in Dipteran midgut.
Prestin also contributes to sulfate and oxalate metabolism at the gut and the Malpighian tubule, as it mediates transport of SO42− and oxalate2−. Since in Anopheles chondroitin sulfate biosynthesis is needed to support Plasmodium invasion of the midgut (Dinglasan et al., 2007), we speculate that AgPrestin – mediated SO42− uptake by the gut is also critical to support Plasmodium invasion and propagate malaria. Clearly further investigation will be needed to understand basic transport, transport regulation and how these processes control host-symbiont interaction.
Methods Summary
Drosophila
Flies were kept on standard medium in vials at 25°C, 12:12 h photoperiod, and 55% relative humidity. Wild-type (Oregon R) flies were used for obtaining total RNA for RT-PCR cloning.
Anopheles gambiae
Fertilized eggs of Anopheles gambiae were purchased from CDC-Atlanta. The eggs were hatched in deionized water and maintained at 28°C in an incubator with a 12/12 light dark cycle. Mosquitoes were grown to adult emergence and maintained in the lab on 30% sucrose/water for several days prior to harvest and tissue dissection.
Xenopus laevis
Frogs were housed and cared for in accordance and approval of the Institutional Care and Use committees of the Mayo Clnic.
Drosophila Prestin, Anopheles gambiae PrestinA and -B constructs
Complementary DNA was reverse-transcribed using oligo-dT primers and the SuperScript III First-Strand Synthesis System (Invitrogen, Carlsbad, CA). The sequence of Drosophila Prestin (CG5485) has been previously reported (Weber et al., 2003). The sequence of Anopheles Gambiae PrestinA and -B was predicted by genomic and EST database (http://www.ncbi.nlm.nih.gov/projects/mapview/map_search.cgi?taxid=7165) search first and then cloned by RT-PCR. We designed PCR primers to amplify the open reading frame plus restriction sites. Each sequence was verified.
Oocyte experiments
Drosophila Prestin, Anopheles gambiae PrestinA and -B were subcloned into the pGEMHE Xenopus expression vector, capped cRNA synthesized using the T7 mMESSAGE mMACHINE kit (Ambion, Austin, TX); oocytes injected with 50 nL cRNA (0.2 μg/μL, 10 ng/oocyte for dPrestin; 0.5 μg/μL, 25 ng/oocyte for AgPrestinA and -B) or water as previously for other transporters (Gunshin et al., 1997; Kato et al., 2009; Plata et al., 2007; Romero et al., 1998); and incubated at 16°C in OR3 media. Oocytes were studied 3–10 days after injection
Electrophysiology
Electrophysiology protocols were performed as we used for mouse Slc26a6 (Kato et al., 2009; Kurita et al., 2008; Xie et al., 2002). All solutions were either ND96 (96 mM NaCl, 2 mM KCl, 1.8 mM CaCl2, 1 mM MgCl2, 5 mM HEPES, pH 7.5) or iso-osmotic ion replacements (Sciortino and Romero, 1999). Cl− was replaced by gluconate. For HCO3− solutions, we used 5% CO2/33 mM HCO3− (pH 7.5). For sulfate, oxalate and formate solution, we used 5 mM SO42−, 0.33 mM oxalate2− and 5 mM formate−, respectively.
Two electrode voltage clamp
For these experiments, membrane currents were recorded with an OC-725C voltage clamp (Warner Instruments, Hamden, CT), filtered at 2–5 kHz, digitized at 10 kHz. I-V protocols consisted of 40 ms steps from Vh (−60 mV) to −140 mV and +60 mV in 20 mV steps (Kato et al., 2009; Sciortino and Romero, 1999).
Ion selective microelectrodes
Ion selective microelectrodes were used to monitor pHi and intracellular Cl− ([Cl−]i) of oocytes (Romero et al., 1998; Romero et al., 2000b). Intracellular pH and Cl− microelectrodes had slopes of at least −54 mV/pH unit or decade, respectively.
HIGHLIGHTS.
This is the first report of cloning and characterization of insect Slc26 transporters.
One Drosophila and two Anopheles gambiae orthologues display high homology with mammalian Slc26a5 and Slc26a6.
dPrestin and AgPrestin-B show similar substrate transport as mouse Slc26a6
dPrestin and AgPrestin-B are mainly expressed at gut and Malpighian tubules.
Drosophila OSR1 signaling activates dPrestin transport function.
Diptera prestin proteins are functional orthologues of mammalian Slc26a6 (i.e., PAT1, CFEX).
Acknowledgments
This work was supported by NIH: AI45098 (PJL), DK60845, EY017732 (MFR) and P50-DK083007 (to JC Lieske; MFR, TH). We thank Heather L. Holmes, Elyse M. Scileppi for excellent technical support as well as Dr. Zara M. Josephs and Dr. Min-Hwang Chang for initial cloning of non-prestin Slc26 Drosophila and Anopheles cDNAs.
Footnotes
“Slc” is HUGO nomenclature for Solute Leak Carrier. The convention is that human genes/proteins are designated “SLC#” and non-human genes/proteins are designated “[organism]Slc#.” The # refers to the particular gene/protein family (51 presently known) followed by “a*” where “*” particular gene/protein in the family. See http://www.bioparadigms.org/slc/menu.asp for details.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alper SL. Familial renal tubular acidosis. Journal of nephrology. 2010;23(Suppl 16):S57–76. [PubMed] [Google Scholar]
- Boudko DY, Moroz LL, Harvey WR, Linser PJ. Alkalinization by chloride/bicarbonate pathway in larval mosquito midgut. Proc Natl Acad Sci U S A. 2001a;98:15354–15359. doi: 10.1073/pnas.261253998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boudko DY, Moroz LL, Linser PJ, Trimarchi JR, Smith PJ, Harvey WR. In situ analysis of pH gradients in mosquito larvae using non-invasive, self-referencing, pH-sensitive microelectrodes. J Exp Biol. 2001b;204:691–699. doi: 10.1242/jeb.204.4.691. [DOI] [PubMed] [Google Scholar]
- Chintapalli VR, Wang J, Dow JA. Using FlyAtlas to identify better Drosophila melanogaster models of human disease. Nat Genet. 2007;39:715–720. doi: 10.1038/ng2049. [DOI] [PubMed] [Google Scholar]
- Clark JS, Vandorpe DH, Chernova MN, Heneghan JF, Stewart AK, Alper SL. Species differences in Cl- affinity and in electrogenicity of SLC26A6-mediated oxalate/Cl- exchange correlate with the distinct human and mouse susceptibilities to nephrolithiasis. J Physiol. 2008;586:1291–1306. doi: 10.1113/jphysiol.2007.143222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deaton SL, Sengupta S, Cobb MH. WNK kinases and blood pressure control. Curr Hypertens Rep. 2009;11:421–426. doi: 10.1007/s11906-009-0072-z. [DOI] [PubMed] [Google Scholar]
- Delpire E, Gagnon KB. SPAK and OSR1: STE20 kinases involved in the regulation of ion homoeostasis and volume control in mammalian cells. The Biochemical journal. 2008;409:321–331. doi: 10.1042/BJ20071324. [DOI] [PubMed] [Google Scholar]
- Dinglasan RR, Alaganan A, Ghosh AK, Saito A, van Kuppevelt TH, Jacobs-Lorena M. Plasmodium falciparum ookinetes require mosquito midgut chondroitin sulfate proteoglycans for cell invasion. Proc Natl Acad Sci U S A. 2007;104:15882–15887. doi: 10.1073/pnas.0706340104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorwart MR, Shcheynikov N, Wang Y, Stippec S, Muallem S. SLC26A9 is a Cl channel regulated by the WNK kinases. J Physiol. 2007a;584:333–345. doi: 10.1113/jphysiol.2007.135855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorwart MR, Shcheynikov N, Wang Y, Stippec S, Muallem S. SLC26A9 is a Cl(-) channel regulated by the WNK kinases. The Journal of physiology. 2007b;584:333–345. doi: 10.1113/jphysiol.2007.135855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorwart MR, Shcheynikov N, Yang D, Muallem S. The solute carrier 26 family of proteins in epithelial ion transport. Physiology (Bethesda) 2008;23:104–114. doi: 10.1152/physiol.00037.2007. [DOI] [PubMed] [Google Scholar]
- Freel RW, Hatch M, Green M, Soleimani M. Ileal oxalate absorption and urinary oxalate excretion are enhanced in Slc26a6 null mice. Am J Physiol Gastrointest Liver Physiol. 2006;290:G719–728. doi: 10.1152/ajpgi.00481.2005. [DOI] [PubMed] [Google Scholar]
- Gamba G. Molecular physiology and pathophysiology of electroneutral cation-chloride cotransporters. Physiological reviews. 2005;85:423–493. doi: 10.1152/physrev.00011.2004. [DOI] [PubMed] [Google Scholar]
- Gunshin H, Mackenzie B, Berger UV, Gunshin Y, Romero MF, Boron WF, Nussberger S, Gollan JL, Hediger MA. Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature. 1997;388:482–488. doi: 10.1038/41343. [DOI] [PubMed] [Google Scholar]
- Hediger MA, Romero MF, Peng JB, Rolfs A, Takanaga H, Bruford EA. The ABCs of solute carriers: physiological, pathological and therapeutic implications of human membrane transport proteinsIntroduction. Pflugers Arch. 2004;447:465–468. doi: 10.1007/s00424-003-1192-y. [DOI] [PubMed] [Google Scholar]
- Jagadeshwaran U, Onken H, Hardy M, Moffett SB, Moffett DF. Cellular mechanisms of acid secretion in the posterior midgut of the larval mosquito (Aedes aegypti) The Journal of experimental biology. 2010;213:295–300. doi: 10.1242/jeb.037549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayaraman S, Haggie P, Wachter RM, Remington SJ, Verkman AS. Mechanism and cellular applications of a green fluorescent protein-based halide sensor. The Journal of biological chemistry. 2000;275:6047–6050. doi: 10.1074/jbc.275.9.6047. [DOI] [PubMed] [Google Scholar]
- Jiang Z, Asplin JR, Evan AP, Rajendran VM, Velazquez H, Nottoli TP, Binder HJ, Aronson PS. Calcium oxalate urolithiasis in mice lacking anion transporter Slc26a6. Nat Genet. 2006;38:474–478. doi: 10.1038/ng1762. [DOI] [PubMed] [Google Scholar]
- Jiang Z, Grichtchenko II, Boron WF, Aronson PS. Specificity of anion exchange mediated by mouse Slc26a6. J Biol Chem. 2002;277:33963–33967. doi: 10.1074/jbc.M202660200. [DOI] [PubMed] [Google Scholar]
- Josephs Z, Satoh J-i, Chang MH, Mercado A, Zandi-Nejad K, Mount DB, Romero MF. Cloning and expression of Drosophila Slc4 and Slc26 homologs. FASEB J. 2003;17:A462. [Google Scholar]
- Josephs ZM, Romero MF. Identification & localization of Slc4 & Slc26 anion transporters in Drosophila and zebrafish. Faseb J. 2004;18:A317. [Google Scholar]
- Kahle KT, Gimenez I, Hassan H, Wilson FH, Wong RD, Forbush B, Aronson PS, Lifton RP. WNK4 regulates apical and basolateral Cl- flux in extrarenal epithelia. Proc Natl Acad Sci U S A. 2004;101:2064–2069. doi: 10.1073/pnas.0308434100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahle KT, Ring AM, Lifton RP. Molecular physiology of the WNK kinases. Annu Rev Physiol. 2008;70:329–355. doi: 10.1146/annurev.physiol.70.113006.100651. [DOI] [PubMed] [Google Scholar]
- Kato A, Kurita Y, Nakada T, Ogoshi M, Nakazato T, Doi H, Chang M-H, Hirose S, Romero MF. Identification of renal transporters involved in sulfate excretion in marine teleost fish. Am J Physiol Regul Integr Comp Physiol. 2009;297:R1647–1659. doi: 10.1152/ajpregu.00228.2009. (Editorial Focus R1645–1646) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato A, Romero MF. Regulation of Electroneutral NaCl Absorption by the Small Intestine. Annual Review of Physiology. 2011;73:261–281. doi: 10.1146/annurev-physiol-012110-142244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko SB, Shcheynikov N, Choi JY, Luo X, Ishibashi K, Thomas PJ, Kim JY, Kim KH, Lee MG, Naruse S, Muallem S. A molecular mechanism for aberrant CFTR-dependent HCO3− transport in cystic fibrosis. Embo J. 2002;21:5662–5672. doi: 10.1093/emboj/cdf580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurita Y, Nakada T, Kato A, Mistry AC, Chang MH, Romero MF, Hirose S. Identification of intestinal bicarbonate transporters involved in formation of carbonate precipitates to stimulate water absorption in marine teleost fish. Am J Physiol - Comp & Reg Physiol. 2008;284:R1402–1412. doi: 10.1152/ajpregu.00759.2007. [DOI] [PubMed] [Google Scholar]
- Linser PJ, Neira M, Piermarini PM, Hirata T, Romero MF. Slc4-like anion transporters of the larval mosquito alimentary canal. J Insect Physiol. 2011 doi: 10.1016/j.jinsphys.2012.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linser PJ, Smith KE, Seron TJ, Neira Oviedo M. Carbonic anhydrases and anion transport in mosquito midgut pH regulation. The Journal of experimental biology. 2009;212:1662–1671. doi: 10.1242/jeb.028084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monico CG, Weinstein A, Jiang Z, Rohlinger AL, Cogal AG, Bjornson BB, Olson JB, Bergstralh EJ, Milliner DS, Aronson PS. Phenotypic and functional analysis of human SLC26A6 variants in patients with familial hyperoxaluria and calcium oxalate nephrolithiasis. Am J Kidney Dis. 2008;52:1096–1103. doi: 10.1053/j.ajkd.2008.07.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mount DB, Romero MF. The SLC26 gene family of multifunctional anion exchangers. Pflügers Arch. 2004a;447:710–721. doi: 10.1007/s00424-003-1090-3. [DOI] [PubMed] [Google Scholar]
- Mount DB, Romero MF. The SLC26 gene family of multifunctional anion exchangers. Pflugers Arch. 2004b;447:710–721. doi: 10.1007/s00424-003-1090-3. [DOI] [PubMed] [Google Scholar]
- Neira Oviedo M, Ribeiro JM, Heyland A, VanEkeris L, Moroz T, Linser PJ. The salivary transcriptome of Anopheles gambiae (Diptera: Culicidae) larvae: A microarray-based analysis. Insect biochemistry and molecular biology. 2009;39:382–394. doi: 10.1016/j.ibmb.2009.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neira Oviedo M, Vanekeris L, Corena-McLeod MD, Linser PJ. A microarray-based analysis of transcriptional compartmentalization in the alimentary canal of Anopheles gambiae (Diptera: Culicidae) larvae. Insect molecular biology. 2008;17:61–72. doi: 10.1111/j.1365-2583.2008.00779.x. [DOI] [PubMed] [Google Scholar]
- Okech BA, Boudko DY, Linser PJ, Harvey WR. Cationic pathway of pH regulation in larvae of Anopheles gambiae. J Exp Biol. 2008;211:957–968. doi: 10.1242/jeb.012021. [DOI] [PubMed] [Google Scholar]
- Oliver D, He DZ, Klocker N, Ludwig J, Schulte U, Waldegger S, Ruppersberg JP, Dallos P, Fakler B. Intracellular anions as the voltage sensor of prestin, the outer hair cell motor protein. Science. 2001;292:2340–2343. doi: 10.1126/science.1060939. [DOI] [PubMed] [Google Scholar]
- Onken H, Moffett DF. Revisiting the cellular mechanisms of strong luminal alkalinization in the anterior midgut of larval mosquitoes. The Journal of experimental biology. 2009;212:373–377. doi: 10.1242/jeb.023580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onken H, Moffett SB, Moffett DF. Alkalinization in the isolated and perfused anterior midgut of the larval mosquito, Aedes aegypti. J Insect Sci. 2008;8:1–20. doi: 10.1673/031.008.4601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onken H, Patel M, Javoroncov M, Izeirovski S, Moffett SB, Moffett DF. Strong alkalinization in the anterior midgut of larval yellow fever mosquitoes (Aedes aegypti): involvement of luminal Na+/K+-ATPase. J Exp Zool A Ecol Genet Physiol. 2009;311:155–161. doi: 10.1002/jez.512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plata C, Sussman CR, Sindic A, Liang JO, Mount DB, Josephs ZM, Chang MH, Romero MF. Zebrafish Slc5a12 encodes an electroneutral sodium monocarboxylate transporter (SMCTn). A comparison with the electrogenic SMCT (SMCTe/Slc5a8) J Biol Chem. 2007;282:11996–12009. doi: 10.1074/jbc.M609313200. [DOI] [PubMed] [Google Scholar]
- Plata C, Vázquez N, Romero MF, Gamba G. Opposite effects of WNK3 and WNK4 upon the Slc26a9 anion transporter/channel. Faseb J. 2008;22:1202–1209. [Google Scholar]
- Romero MF, Chang MH, Mount DB. From cloning to structure, function, and regulation of chloride transporters. In: Alvarez-Leefmans FM Javier, Delpire Eric., editors. Physiology and Pathology of Chloride Transporters and Channels in the Nervous System: From molecules to diseases. Elsevier Academic Press; 2009. pp. 43–80. [Google Scholar]
- Romero MF, Fong P, Berger UV, Hediger MA, Boron WF. Cloning and functional expression of rNBC, an electrogenic Na+-HCO3− cotransporter from rat kidney. Am J Physiol. 1998;274:F425–432. doi: 10.1152/ajprenal.1998.274.2.F425. [DOI] [PubMed] [Google Scholar]
- Romero MF, Fulton CM, Boron WF. The SLC4 family of HCO 3 - transporters. Pflugers Arch. 2004;447:495–509. doi: 10.1007/s00424-003-1180-2. [DOI] [PubMed] [Google Scholar]
- Romero MF, Henry D, Nelson S, Harte PJ, Dillon AK, Sciortino CM. Cloning and characterization of a Na+-driven anion exchanger (NDAE1). A new bicarbonate transporter. The Journal of biological chemistry. 2000a;275:24552–24559. doi: 10.1074/jbc.M003476200. [DOI] [PubMed] [Google Scholar]
- Romero MF, Henry D, Nelson S, Harte PJ, Dillon AK, Sciortino CM. Cloning and characterization of a Na+ driven anion exchanger (NDAE1): a new bicarbonate transporter. J Biol Chem. 2000b;275:24552–24559. doi: 10.1074/jbc.M003476200. [DOI] [PubMed] [Google Scholar]
- Schaechinger TJ, Oliver D. Nonmammalian orthologs of prestin (SLC26A5) are electrogenic divalent/chloride anion exchangers. Proc Natl Acad Sci U S A. 2007;104:7693–7698. doi: 10.1073/pnas.0608583104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciortino CM, Fletcher BR, Shrode LD, Harte PJ, Romero MF. Localization of Endogenous and Recombinant Na+-driven Anion Exchanger Protein, NDAE1, from Drosophila melanogaster. Am J Physiol Cell Physiol. 2001;281:C449–463. doi: 10.1152/ajpcell.2001.281.2.C449. [DOI] [PubMed] [Google Scholar]
- Sciortino CM, Romero MF. Cation and voltage dependence of rat kidney, electrogenic Na+/HCO3− cotransporter, rkNBC, expressed in oocytes. Am J Physiol. 1999;277:F611–623. doi: 10.1152/ajprenal.1999.277.4.F611. [DOI] [PubMed] [Google Scholar]
- Shanbhag S, Tripathi S. Electrogenic H+ transport and pH gradients generated by a V-H+-ATPase in the isolated perfused larval Drosophila midgut. The Journal of membrane biology. 2005;206:61–72. doi: 10.1007/s00232-005-0774-1. [DOI] [PubMed] [Google Scholar]
- Shanbhag S, Tripathi S. Epithelial ultrastructure and cellular mechanisms of acid and base transport in the Drosophila midgut. The Journal of experimental biology. 2009;212:1731–1744. doi: 10.1242/jeb.029306. [DOI] [PubMed] [Google Scholar]
- Sindic A, Mount DB, Romero MF. Renal physiology of SLC26 anion exchangers. Curr Opin Nephrol Hypertens. 2007;16:484–490. doi: 10.1097/MNH.0b013e3282e7d7d0. [DOI] [PubMed] [Google Scholar]
- Subramanya AR, Yang CL, McCormick JA, Ellison DH. WNK kinases regulate sodium chloride and potassium transport by the aldosterone-sensitive distal nephron. Kidney international. 2006;70:630–634. doi: 10.1038/sj.ki.5001634. [DOI] [PubMed] [Google Scholar]
- Terhzaz S, Finlayson AJ, Stirrat L, Yang J, Tricoire H, Woods DJ, Dow JA, Davies SA. Cell-specific inositol 1,4,5 trisphosphate 3-kinase mediates epithelial cell apoptosis in response to oxidative stress in Drosophila. Cellular signalling. 2010;22:737–748. doi: 10.1016/j.cellsig.2009.12.009. [DOI] [PubMed] [Google Scholar]
- Wang Z, Wang T, Petrovic S, Tuo B, Riederer B, Barone S, Lorenz JN, Seidler U, Aronson PS, Soleimani M. Renal and intestinal transport defects in Slc26a6-null mice. Am J Physiol Cell Physiol. 2005;288:C957–965. doi: 10.1152/ajpcell.00505.2004. [DOI] [PubMed] [Google Scholar]
- Weber T, Gopfert MC, Winter H, Zimmermann U, Kohler H, Meier A, Hendrich O, Rohbock K, Robert D, Knipper M. Expression of prestin-homologous solute carrier (SLC26) in auditory organs of nonmammalian vertebrates and insects. Proc Natl Acad Sci U S A. 2003;100:7690–7695. doi: 10.1073/pnas.1330557100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welling PA, Chang YP, Delpire E, Wade JB. Multigene kinase network, kidney transport, and salt in essential hypertension. Kidney Int. 2010;77:1063–1069. doi: 10.1038/ki.2010.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie Q, Welch R, Mercado A, Romero MF, Mount DB. Molecular and functional characterization of the Slc26A6 anion exchanger, functional comparison to Slc26a1. Am J Physiol Renal Physiol. 2002;283:F826–838. doi: 10.1152/ajprenal.00079.2002. [DOI] [PubMed] [Google Scholar]
- Yang D, Li Q, So I, Huang CL, Ando H, Mizutani A, Seki G, Mikoshiba K, Thomas PJ, Muallem S. IRBIT governs epithelial secretion in mice by antagonizing the WNK/SPAK kinase pathway. J Clin Invest. 2011;121:956–965. doi: 10.1172/JCI43475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng J, Shen W, He DZ, Long KB, Madison LD, Dallos P. Prestin is the motor protein of cochlear outer hair cells. Nature. 2000;405:149–155. doi: 10.1038/35012009. [DOI] [PubMed] [Google Scholar]