Abstract
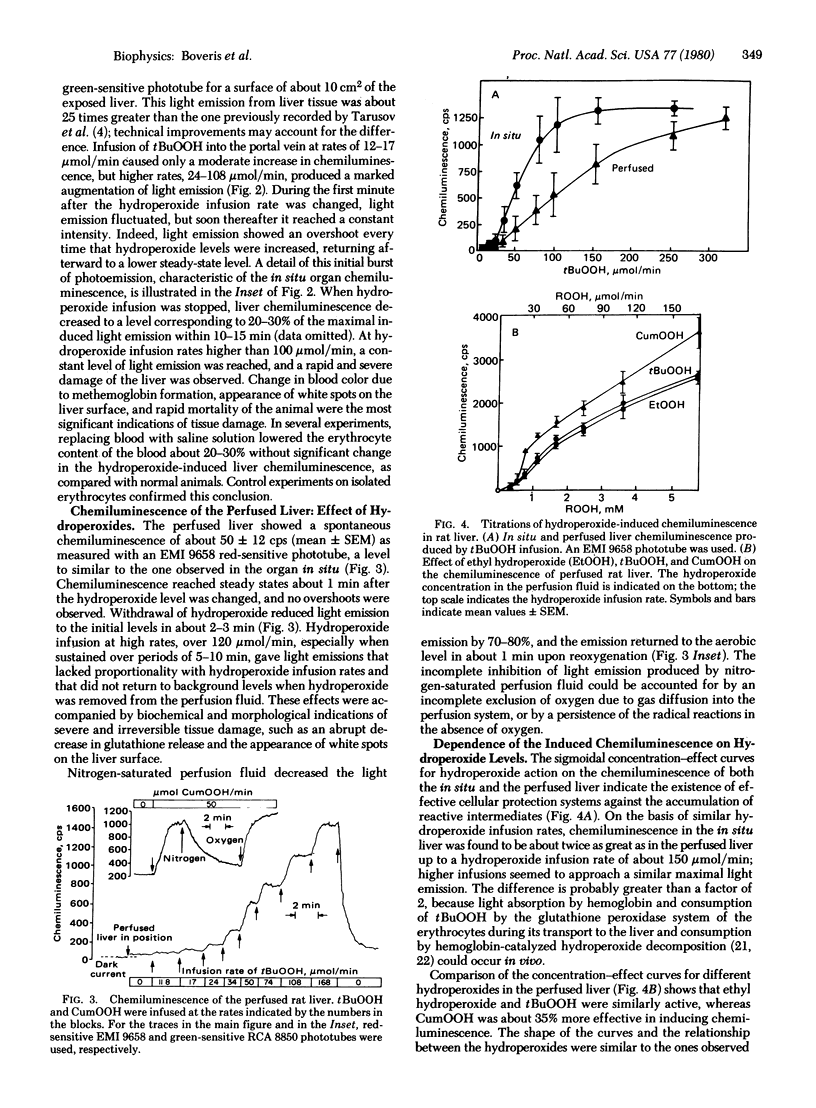
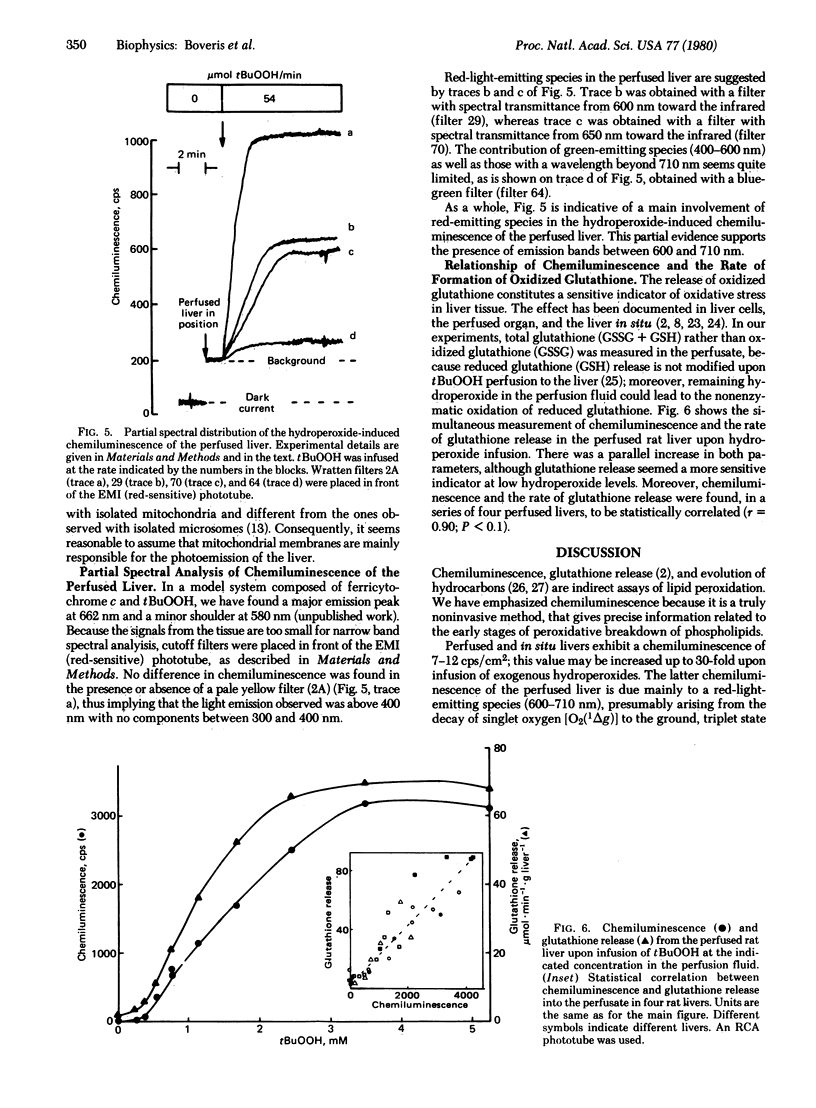
In situ and perfused rat livers showed a spontaneous chemiluminescence of 7-12 counts/sec . cm2 (corresponding to 7-12 x 10(3) photons/sec . cm2); chemiluminescence was increased up to 30 times by infusion of exogenous hydroperoxides. The chemiluminescence of the perfused liver was oxygen dependent. Ethyl, t-butyl, and cumene hydroperoxides were almost equally effective in inducing light emission in the perfused liver. Glutathione release and chemiluminescence showed a parallel increase upon hydroperoxide supply to the perfused liver. A partial spectral analysis of the chemiluminescence of the perfused liver showed a predominance of red-light-emitting species, presumably arising from the singlet oxygen dimol-emission peaks. Many side reactions derived from the complex free radical sequence of lipid peroxidation could afford the chemistry leading to light emission, which represents only about 10(-14) of the utilization of peroxide.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bartoli G. M., Sies H. Reduced and oxidized glutathione efflux from liver. FEBS Lett. 1978 Feb 1;86(1):89–91. doi: 10.1016/0014-5793(78)80105-7. [DOI] [PubMed] [Google Scholar]
- Chance B., Sies H., Boveris A. Hydroperoxide metabolism in mammalian organs. Physiol Rev. 1979 Jul;59(3):527–605. doi: 10.1152/physrev.1979.59.3.527. [DOI] [PubMed] [Google Scholar]
- Dillard C. J., Dumelin E. E., Tappel A. L. Effect of dietary vitamin E on expiration of pentane and ethane by the rat. Lipids. 1977 Jan;12(1):109–114. doi: 10.1007/BF02532981. [DOI] [PubMed] [Google Scholar]
- Halliwell B. Superoxide-dependent formation of hydroxyl radicals in the presence of iron chelates: is it a mechanism for hydroxyl radical production in biochemical systems? FEBS Lett. 1978 Aug 15;92(2):321–326. doi: 10.1016/0014-5793(78)80779-0. [DOI] [PubMed] [Google Scholar]
- Hamman J. P., Gorby D. R., Seliger H. H. A new type of biological chemiluminescence: the microsomal chemiluminescence of benzo[a]pyrene arises from the diol epoxide product of the 7,8-dihydrodiol. Biochem Biophys Res Commun. 1977 Apr 11;75(3):793–798. doi: 10.1016/0006-291x(77)91542-x. [DOI] [PubMed] [Google Scholar]
- Hanstein W. G., Hatefi Y. Lipid oxidation in biological membranes. II. Kinetics and mechanism of lipid oxidation in submitochondrial particles. Arch Biochem Biophys. 1970 May;138(1):87–95. doi: 10.1016/0003-9861(70)90287-0. [DOI] [PubMed] [Google Scholar]
- Hastings J. W., Wilson T. Bioluminescence and chemiluminescence. Photochem Photobiol. 1976 Jun;23(6):461–473. doi: 10.1111/j.1751-1097.1976.tb07282.x. [DOI] [PubMed] [Google Scholar]
- Howes R. M., Steele R. H. Microsomal ( S) chemiluminescence (CL) induced by NADPH and its relation to lipid peroxidation. Res Commun Chem Pathol Pharmacol. 1971 Jul-Sep;2(4):619–626. [PubMed] [Google Scholar]
- Howes R. M., Steele R. H. Microsomal (muS) chemiluminescence (CL) induced by NADPH and its relation to aryl-hydroxylations. Res Commun Chem Pathol Pharmacol. 1972 Mar;3(2):349–357. [PubMed] [Google Scholar]
- Kakinuma K., Cadenas E., Boveris A., Chance B. Low level chemiluminescence of intact polymorphonuclear leukocytes. FEBS Lett. 1979 Jun 1;102(1):38–42. doi: 10.1016/0014-5793(79)80923-0. [DOI] [PubMed] [Google Scholar]
- King M. M., Lai E. K., McCay P. B. Singlet oxygen production associated with enzyme-catalyzed lipid peroxidation in liver microsomes. J Biol Chem. 1975 Aug 25;250(16):6496–6502. [PubMed] [Google Scholar]
- Lloyd D., Boveris A., Reiter R., Filipkowski M., Chance B. Chemiluminescence of Acanthamoeba castellanii. Biochem J. 1979 Oct 15;184(1):149–156. doi: 10.1042/bj1840149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCord J. M., Day E. D., Jr Superoxide-dependent production of hydroxyl radical catalyzed by iron-EDTA complex. FEBS Lett. 1978 Feb 1;86(1):139–142. doi: 10.1016/0014-5793(78)80116-1. [DOI] [PubMed] [Google Scholar]
- Misra H. P., Fridovich I. A peroxide-dependent reduction of cytochrome c by NADH. Biochim Biophys Acta. 1973 Apr 5;292(3):815–824. doi: 10.1016/0005-2728(73)90028-5. [DOI] [PubMed] [Google Scholar]
- Nakano M., Noguchi T., Sugioka K., Fukuyama H., Sato M. Spectroscopic evidence for the generation of singlet oxygen in the reduced nicotinamide adenine dinucleotide phosphate-dependent microsomal lipid peroxidation system. J Biol Chem. 1975 Mar 25;250(6):2404–2406. [PubMed] [Google Scholar]
- Oliveira O. M., Haun M., Durán N., O'Brien P. J., O'Brien C. R., Bechara E. J., Cilento G. Enzyme-generated electronically excited carbonyl compounds, Acetone phosphorescence during the peroxidase-catalyzed aerobic oxidation of isobutanal. J Biol Chem. 1978 Jul 10;253(13):4707–4712. [PubMed] [Google Scholar]
- Oshino N., Chance B. Properties of glutathione release observed during reduction of organic hydroperoxide, demethylation of aminopyrine and oxidation of some substances in perfused rat liver, and their implications for the physiological function of catalase. Biochem J. 1977 Mar 15;162(3):509–525. doi: 10.1042/bj1620509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pryor W. A. The formation of free radicals and the consequences of their reactions in vivo. Photochem Photobiol. 1978 Oct-Nov;28(4-5):787–801. doi: 10.1111/j.1751-1097.1978.tb07020.x. [DOI] [PubMed] [Google Scholar]
- Rappaport A. M., Schneiderman J. H. The function of the hepatic artery. Rev Physiol Biochem Pharmacol. 1976;76:129–175. doi: 10.1007/BFb0027689. [DOI] [PubMed] [Google Scholar]
- Riely C. A., Cohen G., Lieberman M. Ethane evolution: a new index of lipid peroxidation. Science. 1974 Jan 18;183(4121):208–210. doi: 10.1126/science.183.4121.208. [DOI] [PubMed] [Google Scholar]
- Seliger H. H. The origin of bioluminescence. Photochem Photobiol. 1975 May;21(5):355–361. doi: 10.1111/j.1751-1097.1975.tb06684.x. [DOI] [PubMed] [Google Scholar]
- Sies H., Gerstenecker C., Menzel H., Flohé L. Oxidation in the NADP system and release of GSSG from hemoglobin-free perfused rat liver during peroxidatic oxidation of glutathione by hydroperoxides. FEBS Lett. 1972 Oct 15;27(1):171–175. doi: 10.1016/0014-5793(72)80434-4. [DOI] [PubMed] [Google Scholar]
- Sies H., Koch O. R., Martino E., Boveris A. Increased biliary glutathione disulfide release in chronically ethanol-treated rats. FEBS Lett. 1979 Jul 15;103(2):287–290. doi: 10.1016/0014-5793(79)81346-0. [DOI] [PubMed] [Google Scholar]
- Stauff J., Ostrowski J. Chemilumineszenz von Mitochondrien. Z Naturforsch B. 1967 Jul;22(7):734–740. [PubMed] [Google Scholar]
- Sugioka K., Nakano M. A possible mechanism of the generation of singlet molecular oxygen in nadph-dependent microsomal lipid peroxidation. Biochim Biophys Acta. 1976 Feb 16;423(2):203–216. doi: 10.1016/0005-2728(76)90179-1. [DOI] [PubMed] [Google Scholar]
- TAPPEL A. L. Studies of the mechanism of vitamin E action. III. In vitro copolymerization of oxidized fats with protein. Arch Biochem Biophys. 1955 Feb;54(2):266–280. doi: 10.1016/0003-9861(55)90039-4. [DOI] [PubMed] [Google Scholar]
- TARUSOV B. N., POLIVODA A. I., ZHURAVLEV A. I., SEKAMOVA E. N. [Ultraweak spontaneous luminescence in animal tissue]. Tsitologiia. 1962 Nov-Dec;4:696–699. [PubMed] [Google Scholar]
- Tietze F. Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: applications to mammalian blood and other tissues. Anal Biochem. 1969 Mar;27(3):502–522. doi: 10.1016/0003-2697(69)90064-5. [DOI] [PubMed] [Google Scholar]
- Tomoda A., Sugimoto K., Suhara M., Takeshita M., Yoneyama Y. Haemichrome formation from haemoglobin subunits by hydrogen peroxide. Biochem J. 1978 May 1;171(2):329–335. doi: 10.1042/bj1710329. [DOI] [PMC free article] [PubMed] [Google Scholar]



