Abstract
The accumulation of soluble carbohydrates resulting from growth under elevated CO2 may potentially signal the repression of gene activity for the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase (rbcS). To test this hypothesis we grew rice (Oryza sativa L.) under ambient (350 μL L−1) and high (700 μL L−1) CO2 in outdoor, sunlit, environment-controlled chambers and performed a cross-switching of growth CO2 concentration at the late-vegetative phase. Within 24 h, plants switched to high CO2 showed a 15% and 23% decrease in rbcS mRNA, whereas plants switched to ambient CO2 increased 27% and 11% in expanding and mature leaves, respectively. Ribulose-1,5-bisphosphate carboxylase/oxygenase total activity and protein content 8 d after the switch increased up to 27% and 20%, respectively, in plants switched to ambient CO2, but changed very little in plants switched to high CO2. Plants maintained at high CO2 showed greater carbohydrate pool sizes and lower rbcS transcript levels than plants kept at ambient CO2. However, after switching growth CO2 concentration, there was not a simple correlation between carbohydrate and rbcS transcript levels. We conclude that although carbohydrates may be important in the regulation of rbcS expression, changes in total pool size alone could not predict the rapid changes in expression that we observed.
The inevitability of the increase in atmospheric CO2 concentration makes understanding its potential effects on plant growth and development of great importance. Increased growth and yield responses for many plant species grown under elevated CO2 are well documented (Kimball, 1983; Cure and Acock, 1986). However, many C3 species when grown for long periods in elevated CO2 exhibit decreased photosynthetic capacity (Delucia et al., 1985; Sage et al., 1989; Besford et al., 1990). This acclimation response to elevated CO2 is often accompanied by an increase in soluble carbohydrate pools and a decrease in Rubisco protein content, activity, and activation state (Bowes, 1993; Drake et al., 1997).
Carbohydrate source-sink balance under growth at elevated CO2 is believed to play a major role in the regulation of photosynthesis through feedback inhibition (Arp, 1991; Stitt, 1991). Source-sink imbalances may occur during exposure to elevated CO2 when photosynthetic rate exceeds the export capacity or the capacity of sinks to use the photosynthate for growth, resulting in the accumulation of carbohydrates in photosynthetically active source leaves (Farrar and Williams, 1991; Stitt, 1991). It is hypothesized that accumulation of soluble carbohydrates in the cell may signal the repression of Rubisco small and large subunit gene expression (Sheen, 1994). Regulation of the expression of several genes coding for key photosynthetic enzymes has been shown to be influenced by soluble carbohydrate levels in plant cells (Koch, 1996). Transcription of the small subunit of Rubisco (rbcS) and to a lesser extent the large subunit (rbcL) has been shown to be strongly repressed by Glc and Suc (Sheen, 1990; Van Oosten and Besford, 1994). Repression of rbcS and rbcL occurs in field-grown wheat (Nie et al., 1995) and in growth-chamber-grown Arabidopsis (Cheng et al., 1998) exposed to elevated CO2. The signal transduction pathway for the regulation of sugar-sensing genes may involve the phosphorylation of hexose sugars via hexokinase (Jang and Sheen, 1997). However, the contributions that carbohydrate pool sizes or fluctuation of pools via metabolism make to the regulation of gene activity are still uncertain.
We reasoned that by nonintrusively modifying the source-sink balance via a cross-switching of growth [CO2], it should be possible to rapidly reverse the source-sink balance of plants grown at either ambient or elevated [CO2]. If photosynthetic acclimation to elevated CO2 results from carbohydrate feedback inhibition, then switching growth [CO2] from high to ambient should cause a shift from sink-limited to source-limited photosynthesis, and thus feedback-inhibited effects would disappear. Conversely, the opposite should be observed for plants switched from ambient to high CO2.
Rice (Oryza sativa L.) shows marked acclimation to growth under elevated atmospheric CO2 (Rowland-Bamford et al., 1991) and thus provides a good model system for studying the processes involved. In the present study rice was grown under field-like conditions in outdoor, sunlit, environment-controlled chambers under either ambient (350 μL L−1) or high (700 μL L−1) atmospheric [CO2] until the late-vegetative stage, at which time the cross-switching of growth [CO2] was imposed. It was hypothesized that altering the source-sink relationship would result in a change in the amount of rbcS mRNA, followed by corresponding changes in Rubisco total activity and protein content. Furthermore, if leaf carbohydrate pool size was a direct component of the signal transduction pathway, then changes in rbcS mRNA should correlate with changes in soluble carbohydrate pools.
MATERIALS AND METHODS
Plant Material and Growth Conditions
Rice (Oryza sativa L. cv Lemont) was grown in four sunlit environment-controlled chambers, known as Soil-Plant-Atmosphere-Research chambers, located in Gainesville, Florida. The chambers were constructed of an aluminum frame covered with transparent polyethylene terephthalate (Sixlight, Taiyo Kogyo Co., Tokyo, Japan), allowing approximately 90% of incident PAR to penetrate. Aboveground chamber dimensions were 2.0 × 1.0 m in the cross-section and 1.5 m in height. Chamber tops were attached to aluminum vats of 2.0 × 1.0 m in the cross-section and 0.6 m deep, providing a watertight, flooded rooting environment for rice culture. The chamber vats were filled with soil to a depth of 0.5 m. Pregerminated rice seed was sown in 1.0-m rows that were 0.17 m apart on October 3, 1996. Nine days after planting the rice was thinned to approximately 250 plants m−2 and a 0.05-m flood was applied and maintained for the duration of the experiment. Before planting, the soil was fertilized with 8.4 and 13.5 g m−2 phosphorous and potassium, respectively. Nitrogen as urea was applied to the soil at 12.6, 6.3, and 6.3 g m−2 at 8, 18, and 25 d after planting, respectively.
Two chambers of rice were grown at a daytime atmospheric [CO2] of 350 mL L−1 (ambient CO2) and the other two were grown at 700 mL L−1 (high CO2). At the late-vegetative phase of growth, 34 d after planting, a cross-switching of growth CO2 concentration was performed. One chamber of rice at ambient CO2 was switched to high CO2, whereas another chamber at high CO2 was switched to ambient CO2. Dry bulb temperatures were maintained at 28°C/21°C (day/night) and the dew point temperatures were maintained at 18°C/14°C (day/night). The data for microclimate conditions, canopy carbon-exchange rate, and evapotranspiration were collected every 2 s, and 300-s averages were computed and recorded. Each chamber [CO2] was monitored with a dedicated IR gas analyzer (Ultramat 21P, Siemens, Haguenan, France). Gas analyzers were calibrated and checked for linearity before and after the experiment with a range of standard [CO2] in nitrogen. Calibration was checked daily with a span gas. Daily printouts of environment-control conditions were monitored to ensure identical performance of the chambers throughout the study. Variability around the desired set points was ±0.25°C for dry bulb temperature, ±0.5°C for dew point temperature, and ±5 μL L−1 for [CO2]. Details of the methods used for controlling chamber environmental set points (hardware and control algorithms) are given by Pickering et al. (1994).
Photosynthesis Measurements
Photosynthesis was measured at the treatment growth [CO2] on the attached uppermost fully expanded leaf (mature leaf) and the emerging leaf just above it (expanding leaf) with a portable photosynthesis system (model LI-6200, Li-Cor, Lincoln, NE) equipped with a 0.25-L cuvette. The calibration of the instrument was checked daily with CO2 standards of 200, 400, 700, and 1000 μL L−1 CO2 in air. At 32 d after planting, 1 d before the start of the experiment, for each chamber the main culms of approximately 80 to 100 plants at the same developmental stage were tagged. The mature leaf was number 7, and the expanding leaf was number 8 on the main rice culm. Emerging leaves were approximately 25% expanded at the start of the measurements and fully expanded by the time the last measurement was taken. Photosynthesis was measured between 11:30 am and 12:30 pm eastern standard time, when the solar PPFD was saturating at 1200 to 1500 mmol m−2 s−1. Three mature and three expanding leaves were measured per chamber on the day before the CO2 switch (d 1), the day of the CO2 switch (d 2), and for several days thereafter.
Leaf Sampling
The mature and expanding leaves were detached at the ligule from 10 to 20 plants per treatment, at d 1, 3, and 10 of the CO2-switching experiment. Leaves were sampled between 1 pm and 3 pm eastern standard time. After detachment, the leaves were immediately immersed in liquid nitrogen. Sampled leaves were pooled by treatment and developmental stage (mature or expanding), ground to a fine powder in liquid nitrogen with a mortar and pestle, and stored in liquid nitrogen until analysis for Rubisco activity, Rubisco content, rbcS transcripts, and soluble carbohydrates. Ratios of fresh weight to leaf area were determined for a subset of plants at the same time that samples were taken for biochemical and molecular analysis. Leaf area was measured with a leaf area meter (model LI-3100, Li-Cor).
Rubisco Activity and Content Assays
Rubisco total activity was determined by the method described by Vu et al. (1997). Approximately 150 mg of liquid-nitrogen-frozen leaf powder was ground in a prechilled Ten Broek homogenizer at 4°C containing 3 mL of extraction medium consisting of 50 mm CO2-free Tricine-NaOH, pH 8.0, 10 mm MgCl2, 5 mm DTT, 10 mm isoascorbate, 0.1 mm EDTA, and 2% (w/v) PVP-40. The homogenate was microcentrifuged for 45 s at 4°C, an aliquot of the supernatant was immediately assayed for Rubisco activity, and a 0.2-mL aliquot of the supernatant was frozen in liquid nitrogen and later analyzed for Rubisco content. Assay reactions were performed at 30°C in a volume of 0.5 mL. The reaction mixture consisted of 50 mm CO2-free Tricine-NaOH, pH 8.0, 10 mm MgCl2, 5 mm DTT, 0.1 mm EDTA, 0.5 mm RuBP, and 10 mm NaH14CO3 (2 GBq mmol−1). A 0.1-mL aliquot of the supernatant was incubated with the reaction mixture with RuBP omitted for 5 min and the reaction initiated by the addition of RuBP. Reactions were stopped after 45 s by adding 0.1 mL of 6 n HCl. The assays were dried at 60°C and the acid-stable 14C radioactivity determined by scintillation spectrometry. Blank reactions consisting of the reaction mixture without the leaf extract were used to subtract background radioactivity from the assays.
Rubisco content was determined by a radioimmunoprecipitation procedure described by Vu et al. (1997). To a 0.2-mL aliquot of the leaf extract obtained from the Rubisco activity assays, NaHCO3 was added to 10 mm and the mixture allowed to incubate on ice for 20 min to activate Rubisco. A 0.025-mL aliquot of this mixture was added to 0.05 mL of buffer (100 mm Bicine, pH 7.8, 20 mm MgCl2, and 1 mm EDTA) containing 4 nmol of [2-14C] carboxyarabinitol bisphosphate and 0.05 mL of antiserum to purified tobacco Rubisco raised from rabbits. After incubation at 37°C for 2 h, the precipitate was collected on 0.45-mm pore-size Millipore cellulose acetate/nitrate filters and washed with 5 mL of a solution containing 145 mm NaCl and 10 mm MgCl2. The amount of bound 14C was determined by liquid-scintillation spectrometry. Blanks containing everything except the leaf extract were used to subtract background radioactivity. Assays were performed in triplicate on two to three extractions per treatment.
Rubisco Transcript Analysis
Total RNA was isolated from approximately 100 mg of liquid-nitrogen-frozen leaf tissue using a kit (RNeasy, Qiagen, Chatsworth, CA) following the manufacturer's instructions, but with the addition of two phenol/chloroform extraction steps. Absorbance of individual RNA samples was scanned between 320 and 220 nm and quantified by its A260. Total RNA (10 μg) was denatured in 50% (v/v) formamide, 18% (v/v) formaldehyde, and separated by electrophoresis on a 1.5% agarose gel containing 18% (v/v) formaldehyde. RNA was transferred by northern blotting to positively charged nylon membranes (Boehringer Mannheim). Electrophoresis and northern blotting were carried out using the procedures of Sambrook et al. (1989). Immediately after blotting, membranes were baked for 30 min at 120°C to achieve cross-linking. A duplicate set of samples was run on the same gel and stained with ethidium bromide (1 mg mL−1 stock solution). In addition, after hybridizations, the membranes were stained with ethidium bromide to verify integrity and equal loading of RNA.
Detection of mRNA for rbcS was performed by hybridization with a DIG-labeled DNA probe. A 176-bp probe was synthesized using PCR with a cDNA of the rice rbcS gene as the template (Xie and Wu, 1988). DIG labeling was performed using a probe-synthesis kit (PCR DIG, Boehringer Mannheim). Hybridization and detection of the probe were accomplished using the protocols described by the manufacturer. Hybridizations were carried out in 5× SSC (1× SSC = 150 mm NaCl, 15 mm sodium citrate), 50 mm sodium phosphate, pH 7.0, 50% (v/v) formamide, 7.0% (w/v) SDS, 0.1% (w/v) N-lauroylsarcosine, and 2% (w/v) blocking reagent (Boehringer Mannheim) for 16 h at 50°C. After hybridization, membranes were washed twice for 15 min at room temperature in 2× SSC, 0.1% (w/v) SDS, and twice for 15 min at 70°C in 0.1× SSC, 0.1% (w/v) SDS. The DIG label was detected by chemiluminescence (CSPD, Tropix, Bedford, MA) and the membranes were exposed to x-ray film (Fugi, Tokyo, Japan) at room temperature. Quantification of probe hybridized to rbcS mRNA of rice was achieved by image-density scanning of blots (IS-1000 version 2.0, Alpha Innotech Corp., San Leandro, CA). Northern-blot analysis was performed for two separate RNA extractions with similar results.
Carbohydrate Analysis
Soluble carbohydrates were extracted from approximately 150 mg of liquid-nitrogen-frozen leaf tissue using 80% (v/v) ethanol at 85°C. Glc, Fru, and Suc were quantified using the microtiter method described by Hendrix (1993). Carbohydrate assays were performed in triplicate on three separate extractions.
Statistical Methods
A repeated-measures analysis of variance was used to model Rubisco and soluble carbohydrate data according to a split-plot-in-time approach using the Mixed Repeated Measures Analysis procedure (SAS Institute, Cary, NC) to obtain the estimates for the model. The factors used for the model were [CO2] treatment, day of experiment, and [CO2] treatment × day of experiment.
RESULTS
Leaf Photosynthesis
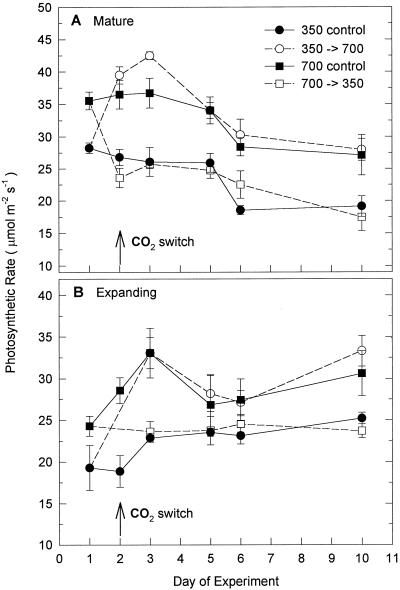
Light-saturated rates of photosynthetic CO2 assimilation by both mature and expanding leaves of rice, measured at their respective growth [CO2], were greatest for plants growing at high CO2 (700 μL L−1 CO2). Photosynthetic rates of mature leaves gradually declined during the course of the experiment, whereas those of expanding leaves tended to increase (Fig. 1). Values for rice grown continuously under ambient CO2 ranged from 19 to 28 and 19 to 25 μmol CO2 m−2 leaf area s−1 for mature and expanding leaves, respectively, whereas values for rice grown continuously under high CO2 were as high as 33 to 37 μmol m−2 s−1.
Figure 1.
Photosynthetic CO2-assimilation rate from mature (A) and expanding (B) leaves of rice. The switch in [CO2] was made on d 2. Values are the means ± se, n = 3.
Upon switching growth [CO2] from ambient to high and vice-versa there was a rapid adjustment in photosynthesis rate measured at the new [CO2]. Mature leaves of rice switched to ambient CO2 initially showed a 10% reduction in photosynthesis compared with ambient controls on d 2, the day of the CO2 switch, when both were measured at ambient [CO2]. By d 3, 1 d after the switch, the photosynthesis rate had increased to that of the ambient controls (Fig. 1A). Mature leaves of rice switched to high CO2 had an 8% higher photosynthetic rate on d 2 and a 15% higher rate on d 3, compared with the high-CO2 controls. By d 5, the rate had declined to that of the high-CO2 controls. The photosynthetic rates of expanding leaves of rice switched to either high or ambient CO2 follow the same pattern as their respective unswitched controls and were statistically indistinguishable from them (Fig. 1B).
Stomatal conductances of leaves for plants grown at high CO2 were 35% to 40% lower than those for plants grown at ambient CO2 (data not shown). For each of the CO2 treatments the Ci/Ca ratios were determined (data not shown). The Ci/Ca ratio of high-CO2 controls was 1% higher than that of the ambient controls throughout the study. In plants switched to either high or ambient CO2, there was a slight decline in Ci/Ca ratio of 3% compared with ambient controls on d 3, but by d 5 it was again within 1% of that of the ambient controls.
Rubisco Total Activity and Content
Rubisco total activity and content declined with age in mature leaves, with the greatest reduction being in plants grown continuously under high CO2 (Table I). During the course of the experiment, mature leaves of plants kept at ambient CO2 showed 28% and 33% decreases in Rubisco activity and content, respectively, whereas plants kept at high CO2 showed 43% and 41% decreases, respectively (Table I).
Table I.
Rubisco total activity and protein content for mature and expanding leaves of rice on d 1, 3, and 10 of the experiment
| Leaf Type and Day of Experiment | Rubisco Total Activity
|
Rubisco
Protein Content
|
||||||
|---|---|---|---|---|---|---|---|---|
| 350 | 350 → 700 | 700 | 700 → 350 | 350 | 350 → 700 | 700 | 700 → 350 | |
| μmol CO2 m−2 leaf area s−1 | g m−2 leaf area | |||||||
| Mature | ||||||||
| 1 | 88.3a ± 1.4 | 83.7a ± 0.8 | 2.9a ± 0.05 | 2.7b ± 0.05 | ||||
| 3 | 82.2a ± 0.7 | 81.0ab ± 2.0 | 65.8c ± 1.1 | 73.9b ± 2.2 | 2.8a ± 0.05 | 2.8a ± 0.03 | 2.4c ± 0.05 | 2.6b ± 0.03 |
| 10 | 63.8ab ± 3.2 | 57.5b ± 4.8 | 47.7c ± 3.6 | 65.5a ± 4.2 | 2.0a ± 0.03 | 2.1a ± 0.03 | 1.6b ± 0.03 | 2.0a ± 0.05 |
| Expanding | ||||||||
| 1 | 77.8a ± 2.0 | 75.4a ± 1.9 | 2.7a ± 0.05 | 2.5a ± 0.05 | ||||
| 3 | 74.6a ± 3.6 | 73.4a ± 5.1 | 67.8a ± 4.4 | 69.9a ± 4.4 | 2.6a ± 0.08 | 2.8a ± 0.07 | 2.5a ± 0.10 | 2.6a ± 0.07 |
| 10 | 75.7a ± 5.0 | 69.4ab ± 2.6 | 66.4b ± 4.1 | 76.8a ± 5.7 | 2.7ab ± 0.10 | 2.5b ± 0.07 | 2.3c ± 0.08 | 2.9a ± 0.10 |
The switch in [CO2] was made on d 2. Values are the means ± se, n = 6 to 9; values followed by a different letter across treatments on the same day are significantly different at the P ≤ 0.05 level.
At d 3, Rubisco activity and content in mature leaves of rice switched to ambient CO2 were significantly greater than those of high-CO2 controls but less than the ambient-CO2 controls. By d 10, the plants switched to ambient CO2 did not differ from those kept at ambient CO2, and had significantly higher activity and content than those kept at high CO2. Rubisco activity and content in mature leaves of rice switched to high CO2 differed little from those of the ambient-CO2 control, and remained greater than those of the high-CO2-grown plants, indicating a lack of adjustment despite the change in growth [CO2].
Expanding leaves of rice kept at ambient or high CO2 showed no significant differences in Rubisco total activity and content at d 1 and 3 (Table I). However, at d 10, both total activity and content were significantly less in the high-CO2 plants. One day after switching to high or ambient CO2 there were no changes in either total activity or content. By d 10, total activity and content in plants switched to ambient CO2 had increased to levels similar to those of plants kept at ambient CO2. Conversely, plants switched to high CO2 appeared to have less Rubisco activity and content than the ambient-CO2 plants, although the differences were not statistically significant.
Rubisco Small Subunit mRNA
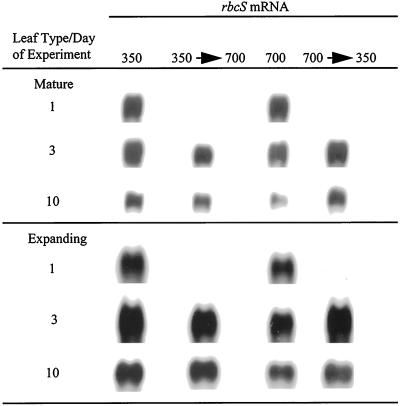
Differences in steady-state rbcS transcript levels between plants grown at ambient and high CO2 were detected in both mature and expanding leaves (Fig. 2). Levels of rbcS transcripts for mature leaves were similar in both ambient- and high-CO2-grown rice on d 1. However, by d 3 the amount of rbcS transcripts in plants kept at ambient CO2 was 13% greater than that of plants kept at high CO2, and by d 10 it was 43% greater (Table II).
Figure 2.
Northern blots of rbcS mRNA for mature and expanding leaves of rice on d 1, 3, and 10. The switch in [CO2] was made on d 2.
Table II.
Abundance of rbcS transcripts for mature and expanding leaves of rice on d 1, 3, and 10
| Leaf Type and Day of Experiment |
rbcS mRNA
|
|||
|---|---|---|---|---|
| 350 | 350 → 700 | 700 | 700 → 350 | |
| % of 350 control | ||||
| Mature | ||||
| 1 | 100 | 94 ± 4 | ||
| 3 | 100 | 77 ± 4 | 87 ± 3 | 98 ± 6 |
| 10 | 100 | 87 ± 4 | 57 ± 3 | 111 ± 5 |
| Expanding | ||||
| 1 | 100 | 85 ± 8 | ||
| 3 | 100 | 85 ± 6 | 70 ± 7 | 97 ± 4 |
| 10 | 100 | 107 ± 7 | 70 ± 12 | 86 ± 2 |
The switch in [CO2] was made on d 2. Values are the means ± sd of two experiments.
After switching the growth [CO2], changes in rbcS transcript levels occurred within 24 h. In mature leaves of rice switched to ambient CO2, the transcript level increased to that of ambient-CO2 controls by d 3, whereas for plants switched to high CO2 it decreased to 23% less than that of the ambient-CO2 controls. At d 10 the level of rbcS transcripts for rice switched to ambient CO2 was 11% greater than that of plants kept at ambient CO2, whereas plants switched to high CO2 showed 13% less.
In expanding leaves, rbcS transcript levels remained about 15% to 30% greater for plants kept at ambient CO2 compared with plants kept at high CO2 (Table II). After switching growth [CO2], expanding leaves showed a similar response to that of mature rice leaves. At d 3 plants switched to ambient CO2 showed an increase in transcripts. Conversely, plants switched to high CO2 showed a 15% decrease compared with plants kept at ambient CO2. At d 10 plants switched to high CO2 showed the same transcript level as plants kept at ambient CO2, whereas those switched to ambient CO2 showed less. Expanding leaves of rice were approximately 20% and 50% expanded on d 1 and 3, respectively, and were fully mature by d 10.
Soluble Carbohydrates
Mature leaves of plants kept in high CO2 had considerably greater Glc and Fru pool sizes than those of plants kept in ambient CO2 (Table III). However, Suc pool size did not differ until d 10, when it was significantly greater in plants kept in high CO2. Plants kept at ambient CO2 showed little change in Glc and Fru from d 1 to 10, whereas Glc increased in plants kept at high CO2. Suc pool sizes for both ambient- and high-CO2 plants were greatest at d 1 and declined thereafter.
Table III.
Glc, Fru, and Suc content for mature and expanding leaves of rice on d 1, 3, and 10
| Leaf Type and Sugar | Day of Experiment | Sugar Content
|
|||
|---|---|---|---|---|---|
| 350 | 350 → 700 | 700 | 700 → 350 | ||
| mg m−2 leaf area | |||||
| Mature | |||||
| Glc | 1 | 111b ± 10 | 190a ± 14 | ||
| 3 | 111b ± 12 | 95b ± 25 | 221a ± 11 | 79b ± 14 | |
| 10 | 79c ± 14 | 111bc ± 11 | 269a ± 33 | 158b ± 24 | |
| Fru | 1 | 221b ± 18 | 348a ± 25 | ||
| 3 | 253b ± 29 | 205b ± 8 | 411a ± 37 | 253b ± 24 | |
| 10 | 252b ± 24 | 284ab ± 32 | 363a ± 45 | 363a ± 27 | |
| Suc | 1 | 4377a ± 143 | 4550a ± 99 | ||
| 3 | 3887a ± 175 | 3271b ± 132 | 4266a ± 95 | 2481c ± 146 | |
| 10 | 3113c ± 82 | 3539bc ± 80 | 4076a ± 139 | 3681b ± 214 | |
| Expanding | |||||
| Glc | 1 | 340b ± 13 | 448a ± 23 | ||
| 3 | 394a ± 23 | 304b ± 26 | 430a ± 21 | 304b ± 10 | |
| 10 | 286b ± 26 | 537a ± 13 | 537a ± 31 | 251b ± 6 | |
| Fru | 1 | 412b ± 16 | 501a ± 37 | ||
| 3 | 412ab ± 41 | 358b ± 59 | 465a ± 42 | 376b ± 28 | |
| 10 | 555b ± 45 | 806a ± 44 | 770a ± 35 | 394c ± 17 | |
| Suc | 1 | 2541a ± 119 | 1915b ± 84 | ||
| 3 | 2775a ± 116 | 2775a ± 180 | 2523ab ± 147 | 2220b ± 113 | |
| 10 | 4618b ± 260 | 5871a ± 239 | 5334a ± 221 | 3437c ± 71 | |
The switch in [CO2] was made on d 2. Values are the means ± se, n = 9; values followed by a different letter across treatments on the same day are statistically different at the P ≤ 0.05 level.
Switching the growth [CO2] of rice from high to ambient resulted in a dramatic decline in soluble carbohydrates the day after the switch. Glc and Fru pools decreased to the same level as in the plants kept at ambient CO2, whereas Suc decreased to significantly less. Compared with plants kept at high CO2, Glc, Fru, and Suc pool sizes for plants switched to ambient CO2 were 64%, 38%, and 42% less, respectively. At d 10 soluble sugar pool sizes for plants switched to ambient CO2 increased to levels greater than those of plants kept at ambient CO2, but Glc and Suc pools were significantly less than those of high-CO2 controls.
Mature leaves of rice switched to high CO2 showed no significant difference in Glc and Fru pool sizes by d 3 compared with ambient-CO2 controls (Table III). However, Suc decreased dramatically by d 3, similar to that of plants switched to ambient CO2. By d 10 soluble sugar pool sizes in leaves of plants switched to high CO2 were not significantly different from those of plants kept at ambient CO2.
Glc and Fru pool sizes of expanding leaves generally exceeded those of mature leaves (Table III). Suc content increased continuously in all treatments during the course of the experiment, and by d 10 the high-CO2 plants and plants switched to high CO2 showed significantly greater amounts than plants kept in ambient CO2. By d 10 plants kept at high CO2 showed 47% and 23% greater Glc and Fru pool sizes, respectively, than plants kept at ambient CO2. Expanding leaves of plants switched to ambient CO2 showed a decrease in both Glc and Fru on d 3, with Glc attaining a level significantly less than that of plants kept at ambient CO2. From d 3 to 10 there was little change in the level of Glc and Fru in ambient-CO2-switched rice, and although Suc increased, it remained significantly less than in the other three treatments. Plants switched to high CO2 showed a small decline in Glc and Fru at d 3. However, by d 10 the amount of Glc, Fru, and Suc had increased dramatically to levels similar to that in plants kept at high CO2.
DISCUSSION
A cross-switching of growth [CO2] for rice previously grown at either ambient (350 μL L−1) or high (700 μL L−1) CO2 resulted in a rapid (24 h) change in rbcS gene expression. Both expanding and mature leaves of rice switched to high CO2 showed a decrease in transcripts with respect to plants maintained at ambient CO2. In contrast, high-CO2 plants switched to ambient CO2 showed an increase in rbcS transcripts. Whatever signaled the response in the expression of rbcS with the switching of growth [CO2] apparently was sensed by both expanding and mature leaves. This change in transcript levels persisted in mature leaves at d 10. Rapid responses in rbcS mRNA have been detected as a consequence of exogenously added sugars in detached tomato leaves (Van Oosten and Besford, 1994), maize mesophyll protoplasts (Sheen, 1990), and autotrophic cell-suspension cultures (Krapp et al., 1993). However, to our knowledge, this is the first report of a rapid response in rbcS expression for plants growing under field-like conditions and exposed to different [CO2].
Rubisco total activity and content measured at midday responded to the switching of rice to ambient CO2, but this response was not as rapid nor did it parallel the response in transcript levels. For expanding leaves, Rubisco total activity and content among all treatments were not significantly different on d 3, but by d 10 increased significantly for rice switched to ambient CO2 and decreased for plants switched to high CO2, although the change was not significantly different from that in plants kept at ambient CO2. A lag time between rbcS expression and changes in Rubisco total activity and content has been shown by others. Krapp et al. (1993) found that when Chenopodium rubrum cell-suspension cultures were supplied with Glc, the expression of rbcS transcripts decreased within 6 h, which was long before significant changes in Rubisco total activity and content occurred. In all CO2 treatments, mature leaves showed an overall trend of decreasing Rubisco total activity and content over time. The decrease was considerably greater in rice maintained at high CO2. Switching to ambient CO2 slowed this decline so that it paralleled that of plants maintained at ambient CO2. However, switching to high CO2 had no apparent effect on Rubisco total activity and content in mature leaves.
Synthesis of photosynthetically active Rubisco requires the coordination and assemblage of small and large subunits in the chloroplast. Therefore, changes in Rubisco synthesis may be regulated by changes in transcription of rbcS and rbcL, transcript stability, translation, and protein turnover, all of which may potentially be affected by growth [CO2] (Webber et al., 1994). The level and change in regulation through each of these steps may also vary with developmental stage (Deng and Gruissem, 1987). Besford et al. (1990) have shown for tomato leaves that Rubisco activity and protein content peak and then decline earlier during leaf expansion in high CO2 than in ambient CO2. Although transcriptional regulation of Rubisco small and large subunits has been shown to be affected by [CO2] in some instances (Winder et al., 1992), the level of regulation by translation and posttranslational turnover is complicated by the fact that photosynthetically competent Rubisco has a relatively slow turnover rate (Peterson et al., 1973).
Our study has shown that upon switching from high to ambient CO2, expanding leaves up-regulated rbcS mRNA, which apparently resulted in increased Rubisco synthesis, whereas switching to high CO2 resulted in down-regulation of rbcS mRNA and a decrease in Rubisco synthesis. Cheng et al. (1998) also showed that rbcS mRNA levels were substantially repressed in Arabidopsis 6 d after transferring them from a growth [CO2] of 360 to 1000 μL L−1, followed later by a significant decrease in Rubisco protein content at 9 and 12 d. The response of rice leaves to changes in growth [CO2] in our study is much more rapid than the acclimation to elevated CO2 reported in some other field studies (Körner and Diemer, 1994; Jacob et al., 1995).
Regulation of rbcS transcription was also apparent in mature leaves. Rubisco turnover appeared to be greater than synthesis, because content declined in all treatments from d 1 to 10. However, plants switched to ambient CO2 showed an increase in rbcS transcripts and by d 10 significantly greater Rubisco content than plants kept at high CO2, suggesting that synthesis had increased in this treatment. The processes of synthesis and turnover may have been affected differently by the switching of growth [CO2] depending on the developmental stage of the leaf. However, it is not evident from these data whether the turnover rate was affected by CO2 switching.
Increased amounts of nonstructural carbohydrates are often associated with photosynthetic acclimation (Bowes, 1993). The down-regulation of photosynthesis associated with acclimation has been attributed to an accumulation of carbohydrates in photosynthetically active source leaves caused when photosynthetic rate exceeds the capacity of sinks to use assimilate for growth (Arp, 1991; Farrar and Williams, 1991). In our study leaf photosynthesis was generally 25% to 30% greater in mature leaves and 20% to 24% greater in expanding leaves of plants grown under high CO2. Photosynthetic acclimation was evident in mature leaves of rice grown at high CO2. This can be inferred by comparing photosynthetic rates of plants switched to high CO2 with those of plants maintained at high CO2 on the day of the switch and 1 d after, when both were measured at high CO2. Photosynthesis was 15% greater in plants switched to high CO2. Also, plants switched to ambient CO2 showed a 10% lower photosynthetic rate than plants maintained at ambient CO2 on the day of the switch.
Growth at elevated CO2 often results in decreased stomatal conductance (Drake et al., 1997). To determine if stomatal conductance was a major limitation to leaf photosynthesis, in the present study the Ci/Ca ratios for various treatments were compared. Even though stomatal conductivity did decrease approximately 35% to 40% at high CO2, the Ci/Ca ratio of high-CO2 controls was nearly identical to that of ambient controls (1% higher) throughout the study. In plants switched to either high or ambient CO2, there was a slight decline in Ci/Ca ratio on d 3 (3% decrease), but by d 5 it was again within 1% of the ambient controls. These data indicate that stomatal conductance was not a major limiting factor to photosynthesis in the high-CO2-grown plants.
Pool sizes of Glc and Fru for mature leaves of plants kept in high CO2 were up to 2-fold greater than those of plants kept in ambient CO2, whereas Suc was only significantly higher on d 10. Transcript levels for rbcS did not differ on d 1, but were greater for the ambient controls on d 3 and 10. Differences in soluble carbohydrate pool sizes between ambient- and high-CO2-grown plants were not as great in expanding leaves as in mature leaves, at least until d 10. This might be expected, because at d 1 and 3 these leaves were growing and thus acting as carbohydrate sinks, whereas by d 10 they were fully expanded. However, at all three sampling times, plants kept at ambient CO2 contained a higher level of rbcS mRNA. Switching from high to ambient CO2 led to a significant decline in Glc and Fru pool sizes for both mature and expanding leaves and a dramatic decrease in Suc in mature leaves on d 3. This was accompanied by an increase in rbcS transcripts for both mature and expanding leaves, which suggests that decreased carbohydrate pool sizes may have signaled the activation of rbcS transcription. However, for plants switched to high CO2, even though photosynthesis increased and rbcS transcripts decreased, the pool sizes of Glc, Fru, and Suc either stayed the same or decreased in comparison with plants kept at ambient CO2. These data are at odds with the hypothesis that rbcS expression is regulated by soluble carbohydrate pool sizes. This does not rule out the possibility that soluble carbohydrates are involved in the regulation of rbcS expression, but our data indicate that the overall leaf pool sizes during leaf development cannot be used to predict the degree of rbcS expression.
Rice is known to be a Suc accumulator when photosynthetic rates are relatively high (Rowland-Bamford et al., 1990, 1996). During vegetative growth, elongating nodes and culms act as a major sink for photosynthetic assimilate (Yoshida and Aln, 1968). Rowland-Bamford et al. (1990) have shown during vegetative growth in rice that as growth [CO2] increases, so does the tendency for recently produced photosynthetic assimilate in leaves to be exported rather than stored. Because photosynthesis increased substantially in plants switched to high CO2, the most likely reason for the observed decrease in soluble carbohydrates, or the lack of accumulation, was an increase in export of recent photosynthetic assimilate.
The lack of correlation between soluble carbohydrate content and photosynthetic acclimation during growth under elevated CO2 seen here and in other studies (Wang and Nobel, 1996; Moore et al., 1997) suggests that fluctuations in soluble carbohydrate pools and metabolic cycling of carbon produced through photosynthesis, rather than total pool sizes themselves, may have a more vital role in signaling regulation of rbcS expression. The phosphorylation of hexose sugars via hexokinase has been hypothesized as a sugar-sensing and signal transduction mechanism for expression of sugar-sensitive genes in plants (Jang and Sheen, 1994). Cheng et al. (1998) showed that rbcS levels in Arabidopsis fluctuate diurnally, with a maximum occurring at the beginning and a minimum at the end of the light cycle; soluble carbohydrate levels generally followed an inverse pattern. Based on this and previous evidence (Moore et al., 1997), they suggest that hexokinase may act as a hexose flux sensor, primarily via nighttime activity, mediating the repression of rbcS transcription. Recently, Jang et al. (1997) have shown evidence for the involvement of hexokinase in the Glc-mediated repression of rbcS and cab1 (chlorophyll a/b-binding protein) and activation of nitrate reductase genes in transgenic Arabidopsis plants with altered hexokinase expression.
In conclusion, this study documents that a nonintrusive modification of atmospheric growth [CO2] in vegetatively growing rice under field-like conditions leads to a rapid change in expression of the rbcS in mature and expanding leaves. Switching the growth [CO2] eventually led to alterations of Rubisco protein content and total activity. Both mature and expanding leaves maintained the ability to up-regulate Rubisco upon being switched from high to ambient CO2, whereas exposure to high CO2 resulted in down-regulation of Rubisco. The signal for changing rbcS gene expression as a consequence of an alteration in [CO2] evidently is detected soon after switching, certainly within 24 h. However, changes in total leaf soluble carbohydrate pool sizes alone could not explain the rapid change in rbcS expression that we detected. Further work is under way to determine the flux of carbon through soluble carbohydrate pools under growth at elevated CO2 in an effort to elucidate the signal for rbcS expression as a function of [CO2].
ACKNOWLEDGMENTS
We thank Joan Anderson for her skillful technical assistance and Drs. Xie Yong and Ray Wu (Cornell University) for kindly providing the cDNA for rice rbcS.
Abbreviations:
- Ca
atmospheric CO2 concentration
- Ci
internal CO2 concentration
- DIG
digoxigenin
- RuBP
ribulose-1,5-bisphosphate
Footnotes
This work was supported by the U.S. Department of Agriculture/National Research Initiative Competitive Grants Program Plant Responses to the Environment (grant no. 95-37100-1597). This is Florida Agricultural Experiment Station journal series no. R-06325.
LITERATURE CITED
- Arp WJ. Effects of source-sink relations on photosynthetic acclimation to elevated CO2. Plant Cell Environ. 1991;14:869–875. [Google Scholar]
- Besford RT, Ludwig LJ, Withers AC. The greenhouse effect: acclimation of tomato plants growing in high CO2, photosynthesis and ribulose-1,5-bisphosphate carboxylase protein. J Exp Bot. 1990;41:925–931. [Google Scholar]
- Bowes G. Facing the inevitable: plants and increasing atmospheric CO2. Annu Rev Plant Physiol Plant Mol Biol. 1993;44:309–332. [Google Scholar]
- Cheng S-H, Moore BD, Seemann JR. Effects of short- and long-term elevated CO2 on the expression of ribulose-1,5-bisphosphate carboxylase/oxygenase genes and carbohydrate accumulation in leaves of Arabidopsis thaliana (L.) Heynh. Plant Physiol. 1998;116:715–723. doi: 10.1104/pp.116.2.715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cure JD, Acock B. Crop responses to carbon dioxide doubling: a literature survey. Agric For Meteorol. 1986;38:127–145. [Google Scholar]
- Delucia EH, Sasek TW, Strain BR. Photosynthetic inhibition after long-term exposure to elevated levels of atmospheric carbon dioxide. Photosynth Res. 1985;7:175–184. doi: 10.1007/BF00037008. [DOI] [PubMed] [Google Scholar]
- Deng X-W, Gruissem W. Control of plastid gene expression during development: the limited role of transcriptional regulation. Cell. 1987;49:379–387. doi: 10.1016/0092-8674(87)90290-x. [DOI] [PubMed] [Google Scholar]
- Drake BG, Gonzàlez-Meler MA, Long SP. More efficient plants: a consequence of rising atmospheric CO2? Annu Rev Plant Physiol Plant Mol Biol. 1997;48:609–639. doi: 10.1146/annurev.arplant.48.1.609. [DOI] [PubMed] [Google Scholar]
- Farrar JF, Williams ML. The effects of increased carbon dioxide and temperature on carbon partitioning, source-sink relations and respiration. Plant Cell Environ. 1991;14:819–830. [Google Scholar]
- Hendrix DL. Rapid extraction and analysis of nonstructural carbohydrates in plant tissues. Crop Sci. 1993;33:1306–1311. [Google Scholar]
- Jacob J, Greitner C, Drake BG. Acclimation of photosynthesis in relation to Rubisco and non-structural carbohydrate contents and in situ carboxylase activity in Scirpus olneyi grown at elevated CO2 in the field. Plant Cell Environ. 1995;18:875–884. [Google Scholar]
- Jang J-C, Leon P, Zhou L, Sheen J. Hexokinase as a sugar sensor in higher plants. Plant Cell. 1997;9:5–19. doi: 10.1105/tpc.9.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang J-C, Sheen J. Sugar sensing in higher plants. Plant Cell. 1994;6:1665–1679. doi: 10.1105/tpc.6.11.1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang J-C, Sheen J. Sugar sensing in higher plants. Trends Plant Sci. 1997;2:208–214. [Google Scholar]
- Kimball BA. Carbon dioxide and agricultural yield: assemblage and analysis of 430 prior observations. Agron J. 1983;75:779–788. [Google Scholar]
- Koch KE. Carbohydrate-modulated gene expression in plants. Annu Rev Plant Physiol Plant Mol Biol. 1996;47:509–540. doi: 10.1146/annurev.arplant.47.1.509. [DOI] [PubMed] [Google Scholar]
- Körner C, Diemer M. Evidence that plants from high altitudes retain their greater photosynthetic efficiency under elevated CO2. Funct Ecol. 1994;8:58–68. [Google Scholar]
- Krapp A, Hofmann B, Schafer C, Stitt M. Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates: a mechanism for the ‘sink regulation’ of photosynthesis. Plant J. 1993;3:817–828. [Google Scholar]
- Moore BD, Palmquis DE, Seeman JR. Influence of plant growth at high CO2 concentrations on leaf content of ribulose-1,5-bisphosphate carboxylase/oxygenase and intracellular distribution of soluble carbohydrates in tobacco, snapdragon, and parsley. Plant Physiol. 1997;115:241–248. doi: 10.1104/pp.115.1.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nie G-Y, Hendrix DL, Webber AN, Kimball BA, Long SP. Increased accumulation of carbohydrates and decreased photosynthetic gene transcript levels in wheat grown at an elevated CO2 concentration in the field. Plant Physiol. 1995;108:975–983. doi: 10.1104/pp.108.3.975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson LW, Kleinkopf GE, Huffaker RC. Evidence for lack of turnover of ribulose-1,5-diphosphate carboxylase in barley leaves. Plant Physiol. 1973;51:1042–1045. doi: 10.1104/pp.51.6.1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pickering NB, Allen LH Jr, Albrecht SL, Jones PJ, Jones JW, Baker JT (1994) Environmental plant chambers: control and measurement using CR-10T dataloggers. In DG Watson, FS Zazueta, TV Harrison, eds, Computers in Agriculture: Proceedings of the 5th International Conference on Computers in Agriculture, Orlando, Florida, February 5–9, 1994. American Society of Agricultural Engineers, St. Joseph, MI, pp 29–35
- Rowland-Bamford AJ, Allen LH, Jr, Baker JT, Boote KJ. Carbon dioxide effects on carbohydrate status and partitioning in rice. J Exp Bot. 1990;41:1601–1608. [Google Scholar]
- Rowland-Bamford AJ, Baker JT, Allen LH, Jr, Bowes G. Acclimation of rice to changing atmospheric carbon dioxide concentration. Plant Cell Environ. 1991;14:577–583. [Google Scholar]
- Rowland-Bamford AJ, Baker JT, Allen LH, Jr, Bowes G. Interactions of CO2 enrichment and temperature on carbohydrate accumulation and partitioning in rice. Environ Exp Bot. 1996;36:111–124. [Google Scholar]
- Sage RF, Sharkey TD, Seeman JR. Acclimation of photosynthesis to elevated CO2 in five C3 species. Plant Physiol. 1989;89:590–596. doi: 10.1104/pp.89.2.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sambrook J, Fritsch EF, Maniatis T. Molecular Cloning: A Laboratory Manual, Ed 2. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- Sheen J. Metabolic repression of transcription in higher plants. Plant Cell. 1990;2:1027–1038. doi: 10.1105/tpc.2.10.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheen J. Feedback control of gene expression. Photosynth Res. 1994;39:427–438. doi: 10.1007/BF00014596. [DOI] [PubMed] [Google Scholar]
- Stitt M. Rising CO2 levels and their potential significance for carbon flow in photosynthetic cells. Plant Cell Environ. 1991;14:741–762. [Google Scholar]
- Van Oosten JJ, Besford RT. Sugar feeding mimics effect of acclimation to high CO2-rapid down regulation of Rubisco small subunit transcripts but not of the large subunit transcripts. J Plant Physiol. 1994;143:306–312. [Google Scholar]
- Vu JCV, Allen LH, Jr, Boote KJ, Bowes G. Effects of elevated CO2 and temperature on photosynthesis and Rubisco in rice and soybean. Plant Cell Environ. 1997;20:68–76. [Google Scholar]
- Wang N, Nobel PS. Doubling the CO2 concentration enhanced the activity of carbohydrate-metabolism enzymes, source carbohydrate production, photoassimilate transport, and sink strength for Opuntia ficus-indica. Plant Physiol. 1996;110:893–902. doi: 10.1104/pp.110.3.893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webber AN, Nie G-Y, Long SP. Acclimation of photosynthetic proteins to rising atmospheric CO2. Photosynth Res. 1994;39:413–425. doi: 10.1007/BF00014595. [DOI] [PubMed] [Google Scholar]
- Winder TL, Anderson JC, Spalding MH. Translational regulation of the large and small subunits of ribulose bisphosphate carboxylase/oxygenase during induction of the CO2-concentrating mechanism in Chlamydomonas reinhardtii. Plant Physiol. 1992;98:1409–1414. doi: 10.1104/pp.98.4.1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie Y, Wu R. Nucleotide sequence of ribulose-1,5-bisphosphate carboxylase/oxygenase small subunit gene (rbcS) in rice. Nucleic Acids Res. 1988;16:7749. doi: 10.1093/nar/16.15.7749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida S, Aln SB. The accumulation process of carbohydrate in rice varieties in relation to their response to nitrogen in the tropics. Soil Sci Plant Nutr. 1968;14:153–162. [Google Scholar]