The body’s response to immune challenges and tissue damage involves a complex inflammatory process. The innate immune system acts as the initial barrier that protects from infectious agents and also senses endogenous danger molecules released after traumatic insults to mediate the response to injury. This latter role is particularly important in the central nervous system (CNS), where challenges like trauma, ischemia, or pathological neurodegeneration must be promptly contained. However, overactivation of the neuroinflammatory response can trigger deleterious effects and worsen the functional outcome of the injury, a process often observed after spinal cord injury (SCI) and stroke, as well as in neuropathic and inflammatory pain pathologies (Rice et al., 2007; Scholz and Woolf, 2007).
Many pathological neuroinflammatory reactions are characterized by increased production of the proinflammatory cytokine interleukin-1β (IL-1β). Although the involvement of IL-1β in CNS injury pathologies is widely accepted, the mechanisms behind its formation and release are incompletely understood. The inflammasome is emerging as the major determinant in the formation of mature IL-1β, which undergoes proteolytic processing from its immature pro–IL-1β form by the protease caspase-1 (Gross et al., 2011). Inflammasomes are multi-protein complexes formed of NOD-like receptor proteins (NLRPs) that act as scaffolds for the caspases necessary for cytokine processing. Of the several classes of inflammasomes that exist, both the NLRP1- and NLRP3-containing subfamilies assemble with caspase-1 to process IL-1β. Because of its role in IL-1β maturation, activation of the inflammasome is critical to the induction of neuroinflammatory cascades. NLRP inflammasomes are activated by recognizing pathogen-associated molecular patterns or, in the case of endogenous signals released after injury, danger-associated molecular patterns.
Among the many host-derived molecules that activate inflammasomes, ATP potently induces NLRP1- and NLRP3-mediated IL-1β processing. ATP is released or leaked from damaged cells after trauma, and the subsequent increase in extracellular levels of ATP is thought to play a major role in triggering the neuroinflammatory response in the CNS (Burnstock, 2008). However, the mechanism linking extracellular ATP to inflammasome activation to IL-1β release in CNS pathologies is unresolved. A recent report by de Rivero Vaccari et al. (2012) addresses several unanswered questions, including the cellular source of IL-1β in SCI models as well as the identity of the ATP receptor involved. Specifically, their results indicate a neuronal contribution to IL-1β release in the SCI response and implicate the P2X4 purinergic receptor in this system. Here, I discuss both of these results in light of previous work, suggesting microglial involvement in IL-1β processing and identifying P2X7 as the major purinergic receptor responsible for cytokine release. I propose a model in which P2X4 acts as an upstream trigger of P2X7 activation to enhance IL-1β release and the inflammatory response. I also discuss the potential implications of de Rivero Vaccari’s results regarding the role of neuroinflammation in other CNS pathologies.
Cellular source of IL-1β
One of the major findings of the study by de Rivero Vaccari et al. (2012) is the neuronal origin of IL-1β after SCI. In the periphery, inflammasome proteins involved in IL-1β maturation, which are found in monocytes, macrophages, dendritic cells, and neutrophils, are widely believed to be localized to cells of immune origin; however, the cellular localization of the inflammasome in the CNS has been unclear. NLRP1 components have been identified in neurons and astrocytes of the spinal cord, and microglia can also release IL-1β (Ferrari et al., 1997; Silverman et al., 2009). To demonstrate the involvement of neurons in IL-1β processing, de Rivero Vaccari et al. (2012) first showed that SCI-induced inflammasome responses were decreased in P2X4-deficient mice. They then used a knock-in of the β-gal reporter gene under the control of the P2X4 promoter in the P2X4-KO mice to investigate the cellular expression of P2X4. Strong colocalization of β-gal with neuronal markers was seen in control and SCI conditions, suggesting that P2X4, in normal WT mouse spinal cord, is exclusively expressed in neurons (de Rivero Vaccari et al., 2012).
However, this approach to P2X4 localization is rather indirect. Numerous groups have shown functional and immunohistochemical evidence of P2X4 in spinal microglia in resting and activated conditions (Tsuda et al., 2003; Ulmann et al., 2008), where it mediates the ATP-dependent release of brain-derived neurotrophic factor (Tsuda et al., 2010). P2X4 is directly involved in microglial chemotaxis (Ohsawa et al., 2007), and the developmental effects of P2X4-KO are unclear; thus, the failure to find P2X4 in microglia could arise from deficient microglia migration during development in the P2X4-KO model. This could possibly decrease the microglial population in the spinal cord. In a similar SCI model, it was shown that, although P2X4 was present in surviving neurons, most cells (>60%) expressing P2X4 were highly motile microglia and migrating macrophages that accumulate at the lesion site (Schwab et al., 2005), indicating that the absence of P2X4-positive microglia observed in the knockout animal by de Rivero Vaccari et al. might be a secondary effect of a cellular migratory defect. Furthermore, de Rivero Vaccari et al. observed a rapid and substantial increase in the abundance of P2X7 and inflammasome components such as caspase-1 after injury. The short (30-min) time frame of the increase indicates that it is more likely to derive from an infiltration of immune cells expressing high levels of inflammasome-signaling proteins rather than from de novo up-regulation, supporting the notion that migrating cells play an important role in the SCI response. The report by de Rivero Vaccari et al. (2012) thus demonstrates a contribution of neuronal P2X4 to the innate immune response in the spinal cord; however, P2X4 likely also plays a role in neuroinflammation via its expression at the surface of migrating and resident microglia.
P2X4 versus P2X7
A recent report showing that when activated with lipopolysaccharide (a process mimicking the microgliosis observed in various CNS injuries and pathologies) spinal microglia release IL-1β upon ATP stimulation (Clark et al., 2010) also suggests that spinal cord microglia play a prominent role in IL-1β release. Like most reports investigating ATP-activated innate immune responses in the CNS, Clark et al. (2010) identified the P2X7 subtype as the main purinergic receptor involved in cytokine release. P2X7-induced IL-1β release through activation of NLRP1 or NLRP3 inflammasome has been demonstrated extensively in immune cells (Ferrari et al., 1997; Di Virgilio, 2007) and in neurons (Silverman et al., 2009). de Rivero Vaccari et al. (2012) challenge the notion that P2X7 is the sole purinergic receptor involved in IL-1β release by showing that P2X4-KO mice exhibit lower levels of IL-1β when compared with WT mice, as well as a decrease in caspase-1 activity. Several functional differences between P2X4 and P2X7 purinergic receptors could affect the CNS response to injury. Mouse P2X4 receptor channels are ∼100 times more sensitive to ATP than P2X7 (North and Surprenant, 2000). P2X4 activation of the inflammasome might therefore allow for a graded neuroinflammatory response early after injury. Moreover, P2X7 activation of the caspase cascade induces apoptotic cell death, whereas sustained activation of P2X4 does not (Virginio et al., 1999; Bernier et al., 2012). One could therefore speculate that, after SCI, the surviving neurons around the lesion site, which are P2X4-positive (Schwab et al., 2005), contribute to the maintenance of the inflammatory environment by releasing IL-1β through stimulation of P2X4. In vitro experiments investigating the activation of inflammasome components in neurons exposed to low ATP concentrations (micromolar range), in the presence of the specific P2X4 potentiator ivermectin or P2X7 blockers (such as BBG), could help decipher the role that the purinergic receptor channels play in IL-1β release. Similar experiments performed on peritoneal macrophages from P2X7-KO mice provide evidence that, in the presence of ivermectin, P2X4 activation can indeed induce IL-1β release (Seil et al., 2010).
Molecular mechanism of ATP-induced inflammasome activation
Although the study by de Rivero Vaccari et al. (2012) does not directly address the molecular mechanism underlying ATP-dependent activation of the inflammasome, their results implicating P2X4 allow interesting parallels to be drawn with the P2X7-dependent mechanism. P2X7-mediated NLRP1 and NLRP3 inflammasome activation requires pannexin-1 (Panx1) hemichannels (Pelegrin and Surprenant, 2006; de Rivero Vaccari et al., 2008; Silverman et al., 2009). A large decrease in intracellular K+ concentration caused by P2X7-dependent opening of the Panx1 large pore is believed to stimulate caspase-1 activity. However, it is still unclear whether the P2X7 channel or the Panx1 channel, or a combination of the two, acts as the K+ efflux pathway. Although some evidence also points toward Panx1 directly activating caspase-1, the nature of the interaction remains unresolved (Silverman et al., 2009). In contrast, some groups have reported Panx1-independent assembly of inflammasome complexes and IL-1β release after ATP stimulation (Pelegrin et al., 2008; Qu et al., 2011). It was recently shown that P2X4 can mediate K+ efflux through a large conductance pore independently of Panx1 (Seil et al., 2010; Bernier et al., 2012). Therefore, P2X4-mediated permeabilization could elicit K+ efflux in a mechanism that could conceivably underlie the inflammasome-activating role assigned to neuronal P2X4.
Studies analyzing the P2X4 large conductance pore have consistently ruled out a Panx1 requirement for its formation; however, it is possible that Panx1 is implicated in inflammasome activation downstream of P2X4 stimulation. Although many studies hint at K+ efflux and the subsequent decrease in its intracellular concentration as a trigger for IL-1β processing in immune cells (Pétrilli et al., 2007), in neurons, high extracellular K+ can itself activate Panx1-dependent inflammasome activation (Silverman et al., 2009). As was hypothesized for P2X7 channels, release of K+ through the open P2X4 channel could trigger Panx1 activation, creating a positive feed-forward loop (Fig. 1). It would be interesting to investigate this possible mechanism using validated Panx1 blockers such as carbenoxolone, mefloquine, or the 10panx inhibitory peptide. Such experiments would not only provide information on a possible Panx1-independent role of P2X4 in inflammasome activation but could help determine whether intracellular K+ decrease and high extracellular K+-triggered Panx1 channel opening provide two distinct stimuli for inflammasome assembly.
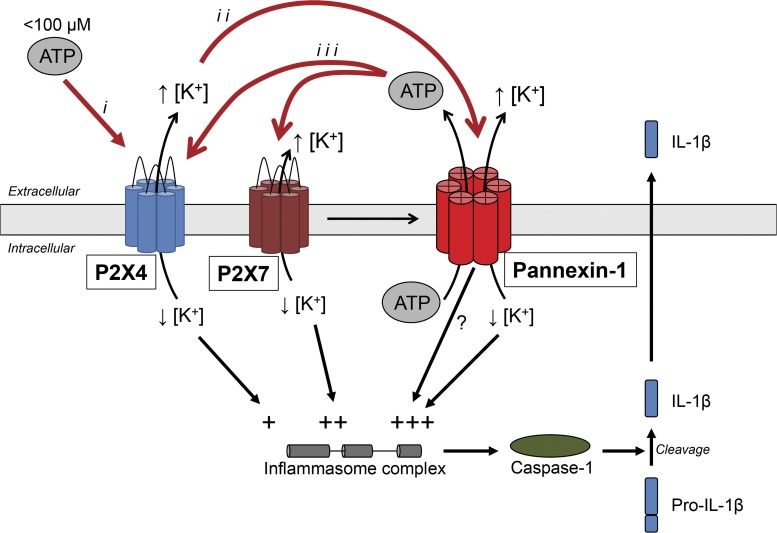
Figure 1.
Model representing a putative mechanism of ATP-induced inflammasome activation. Low extracellular ATP concentration (<100 µM) selectively activates the more sensitive P2X4 receptor channel (i). Opening of the P2X4 channel leads to K+ efflux, with the subsequent increase in [K+]o activating Panx1 (ii). Panx1 channel opening leads to the release of ATP, further activating P2X4 and P2X7 receptor channels (iii). Direct P2X7-mediated recruitment of Panx1 could also enhance such a positive feed-forward loop. P2X4, P2X7, and Panx1 would thereby all contribute to the assembly of the inflammasome complex via K+ efflux and to the consequent increase in [K+]o and decrease in [K+]i,. A direct interaction between Panx1 and inflammasome components may also occur. Depicted here is a putative mechanism for neuronal autocrine activation of the inflammasome; however, this amplification process could also participate in paracrine signaling as part of a neuron-to-immune cell cross talk.
Numerous recent studies have described Panx1 as an ATP release channel. Efflux of ATP through Panx1 has been shown in various cell types, including neurons (Bao et al., 2004; Huang et al., 2007; Xia et al., 2012). This process could therefore represent another positive feed-forward loop, whereby activated Panx1 would release ATP into the extracellular space, further activating the P2X channels and causing even greater changes in the intracellular K+ concentration (Fig. 1). This model could also explain how the P2X4 large pore and the P2X7–Panx1 pore complex are activated. These events would require the sustained presence of ATP; however, extracellular ATP is rapidly degraded by ectonucleotidases (Kukulski et al., 2011). ATP release via Panx1 could therefore provide a mechanism to locally regenerate the ATP necessary for P2X4 or P2X7 activation, in this way keeping the process active. It is also possible that the ATP generated in this process could affect surrounding cells, thereby creating a paracrine signaling pathway involving both neurons and immune cells.
With such a model containing several amplification steps, the role of P2X4 in the IL-1β maturation cascade identified by de Rivero Vaccari et al. (2012) raises the possibility that P2X4, a receptor channel more sensitive to ATP than P2X7, acts mainly to initiate the signaling cascade. This would be consistent with their results obtained using the P2X4-KO mice, where they observed acute and transient improvements, whereas other reports have found that P2X7 and Panx1 are involved in more chronic activation of the inflammasome complex. However, additional contributions of immune cells later recruited to the site of injury cannot be ruled out. This model for response amplification involves both P2X4 and P2X7, with the latter enhancing the reaction not only via increased K+ flux but also by directly coupling to Panx1 opening. The recruitment of P2X4, P2X7, and Panx1 in one coordinated, synergistic pathway could explain the considerable amount of evidence pointing toward the major role of P2X7 and Panx1 in the mechanism of ATP-triggered inflammasome assembly, while fitting with the recent results demonstrating P2X4 involvement (de Rivero Vaccari et al., 2012). However, their common characteristics in terms of K+ flux capabilities could make each component of the pathway somewhat redundant, explaining the reports of pannexin-independent inflammasome activation (Qu et al., 2011).
Physiological considerations
de Rivero Vaccari et al. reported that the decreased IL-1β release seen in the P2X4-KO mice after SCI translates into an improvement in functional outcome, measured as spinal cord lesion volume and locomotor activity. This adds to the considerable amount of evidence demonstrating a role for IL-1β in exacerbating damage in the CNS after trauma; notably, IL-1β blockade improves recovery from traumatic brain injury and peripheral nerve injury (de Rivero Vaccari et al., 2009; Nadeau et al., 2011). This highlights the essential role of innate immunity in the CNS. Furthermore, IL-1β signaling is involved in the generation of neuropathic pain, which frequently develops after SCI. Specifically blocking the release of IL-1β as part of the early treatment of SCI might prevent such a progression into chronic pain pathologies. Although more studies investigating IL-1β release mechanisms in the CNS are necessary, a better understanding of the signaling pathways underlying ATP-mediated inflammasome activation will likely lead to improved therapeutic treatment options in cases of brain and spinal trauma.
Please participate in a discussion of this Journal Club article on the JGP Facebook page (www.facebook.com/JGenPhysiol).
Acknowledgments
L.P. Bernier is supported by a Canada Graduate Scholarship from the Canadian Institutes of Health Research.
Philippe Séguéla served as faculty advisor.
Elizabeth M. Adler served as editor.
Footnotes
Abbreviations used in this paper:
- CNS
- central nervous system
- IL-1β
- interleukin-1β
- NLRP
- NOD-like receptor protein
- Panx1
- pannexin-1
- SCI
- spinal cord injury
References
- Bao L., Locovei S., Dahl G. 2004. Pannexin membrane channels are mechanosensitive conduits for ATP. FEBS Lett. 572:65–68 10.1016/j.febslet.2004.07.009 [DOI] [PubMed] [Google Scholar]
- Bernier L.P., Ase A.R., Boué-Grabot E., Séguéla P. 2012. P2X4 receptor channels form large noncytolytic pores in resting and activated microglia. Glia. 60:728–737 10.1002/glia.22301 [DOI] [PubMed] [Google Scholar]
- Burnstock G. 2008. Purinergic signalling and disorders of the central nervous system. Nat. Rev. Drug Discov. 7:575–590 10.1038/nrd2605 [DOI] [PubMed] [Google Scholar]
- Clark A.K., Staniland A.A., Marchand F., Kaan T.K., McMahon S.B., Malcangio M. 2010. P2X7-dependent release of interleukin-1beta and nociception in the spinal cord following lipopolysaccharide. J. Neurosci. 30:573–582 10.1523/JNEUROSCI.3295-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Rivero Vaccari J.P., Lotocki G., Marcillo A.E., Dietrich W.D., Keane R.W. 2008. A molecular platform in neurons regulates inflammation after spinal cord injury. J. Neurosci. 28:3404–3414 10.1523/JNEUROSCI.0157-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Rivero Vaccari J.P., Lotocki G., Alonso O.F., Bramlett H.M., Dietrich W.D., Keane R.W. 2009. Therapeutic neutralization of the NLRP1 inflammasome reduces the innate immune response and improves histopathology after traumatic brain injury. J. Cereb. Blood Flow Metab. 29:1251–1261 10.1038/jcbfm.2009.46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Rivero Vaccari J.P., Bastien D., Yurcisin G., Pineau I., Dietrich W.D., De Koninck Y., Keane R.W., Lacroix S. 2012. P2X4 receptors influence inflammasome activation after spinal cord injury. J. Neurosci. 32:3058–3066 10.1523/JNEUROSCI.4930-11.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Virgilio F. 2007. Liaisons dangereuses: P2X(7) and the inflammasome. Trends Pharmacol. Sci. 28:465–472 10.1016/j.tips.2007.07.002 [DOI] [PubMed] [Google Scholar]
- Ferrari D., Chiozzi P., Falzoni S., Hanau S., Di Virgilio F. 1997. Purinergic modulation of interleukin-1 β release from microglial cells stimulated with bacterial endotoxin. J. Exp. Med. 185:579–582 10.1084/jem.185.3.579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross O., Thomas C.J., Guarda G., Tschopp J. 2011. The inflammasome: an integrated view. Immunol. Rev. 243:136–151 10.1111/j.1600-065X.2011.01046.x [DOI] [PubMed] [Google Scholar]
- Huang Y.J., Maruyama Y., Dvoryanchikov G., Pereira E., Chaudhari N., Roper S.D. 2007. The role of pannexin 1 hemichannels in ATP release and cell-cell communication in mouse taste buds. Proc. Natl. Acad. Sci. USA. 104:6436–6441 10.1073/pnas.0611280104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kukulski F., Lévesque S.A., Sévigny J. 2011. Impact of ectoenzymes on p2 and p1 receptor signaling. Adv. Pharmacol. 61:263–299 10.1016/B978-0-12-385526-8.00009-6 [DOI] [PubMed] [Google Scholar]
- Nadeau S., Filali M., Zhang J., Kerr B.J., Rivest S., Soulet D., Iwakura Y., de Rivero Vaccari J.P., Keane R.W., Lacroix S. 2011. Functional recovery after peripheral nerve injury is dependent on the pro-inflammatory cytokines IL-1β and TNF: implications for neuropathic pain. J. Neurosci. 31:12533–12542 10.1523/JNEUROSCI.2840-11.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- North R.A., Surprenant A. 2000. Pharmacology of cloned P2X receptors. Annu. Rev. Pharmacol. Toxicol. 40:563–580 10.1146/annurev.pharmtox.40.1.563 [DOI] [PubMed] [Google Scholar]
- Ohsawa K., Irino Y., Nakamura Y., Akazawa C., Inoue K., Kohsaka S. 2007. Involvement of P2X4 and P2Y12 receptors in ATP-induced microglial chemotaxis. Glia. 55:604–616 10.1002/glia.20489 [DOI] [PubMed] [Google Scholar]
- Pelegrin P., Surprenant A. 2006. Pannexin-1 mediates large pore formation and interleukin-1beta release by the ATP-gated P2X7 receptor. EMBO J. 25:5071–5082 10.1038/sj.emboj.7601378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelegrin P., Barroso-Gutierrez C., Surprenant A. 2008. P2X7 receptor differentially couples to distinct release pathways for IL-1beta in mouse macrophage. J. Immunol. 180:7147–7157 [DOI] [PubMed] [Google Scholar]
- Pétrilli V., Papin S., Dostert C., Mayor A., Martinon F., Tschopp J. 2007. Activation of the NALP3 inflammasome is triggered by low intracellular potassium concentration. Cell Death Differ. 14:1583–1589 10.1038/sj.cdd.4402195 [DOI] [PubMed] [Google Scholar]
- Qu Y., Misaghi S., Newton K., Gilmour L.L., Louie S., Cupp J.E., Dubyak G.R., Hackos D., Dixit V.M. 2011. Pannexin-1 is required for ATP release during apoptosis but not for inflammasome activation. J. Immunol. 186:6553–6561 10.4049/jimmunol.1100478 [DOI] [PubMed] [Google Scholar]
- Rice T., Larsen J., Rivest S., Yong V.W. 2007. Characterization of the early neuroinflammation after spinal cord injury in mice. J. Neuropathol. Exp. Neurol. 66:184–195 10.1097/01.jnen.0000248552.07338.7f [DOI] [PubMed] [Google Scholar]
- Scholz J., Woolf C.J. 2007. The neuropathic pain triad: neurons, immune cells and glia. Nat. Neurosci. 10:1361–1368 10.1038/nn1992 [DOI] [PubMed] [Google Scholar]
- Schwab J.M., Guo L., Schluesener H.J. 2005. Spinal cord injury induces early and persistent lesional P2X4 receptor expression. J. Neuroimmunol. 163:185–189 10.1016/j.jneuroim.2005.02.016 [DOI] [PubMed] [Google Scholar]
- Seil M., El Ouaaliti M., Fontanils U., Etxebarria I.G., Pochet S., Dal Moro G., Marino A., Dehaye J.P. 2010. Ivermectin-dependent release of IL-1beta in response to ATP by peritoneal macrophages from P2X(7)-KO mice. Purinergic Signal. 6:405–416 10.1007/s11302-010-9205-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverman W.R., de Rivero Vaccari J.P., Locovei S., Qiu F., Carlsson S.K., Scemes E., Keane R.W., Dahl G. 2009. The pannexin 1 channel activates the inflammasome in neurons and astrocytes. J. Biol. Chem. 284:18143–18151 10.1074/jbc.M109.004804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuda M., Shigemoto-Mogami Y., Koizumi S., Mizokoshi A., Kohsaka S., Salter M.W., Inoue K. 2003. P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature. 424:778–783 10.1038/nature01786 [DOI] [PubMed] [Google Scholar]
- Tsuda M., Tozaki-Saitoh H., Inoue K. 2010. Pain and purinergic signaling. Brain Res. Rev. 63:222–232 10.1016/j.brainresrev.2009.11.003 [DOI] [PubMed] [Google Scholar]
- Ulmann L., Hatcher J.P., Hughes J.P., Chaumont S., Green P.J., Conquet F., Buell G.N., Reeve A.J., Chessell I.P., Rassendren F. 2008. Up-regulation of P2X4 receptors in spinal microglia after peripheral nerve injury mediates BDNF release and neuropathic pain. J. Neurosci. 28:11263–11268 10.1523/JNEUROSCI.2308-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virginio C., MacKenzie A., North R.A., Surprenant A. 1999. Kinetics of cell lysis, dye uptake and permeability changes in cells expressing the rat P2X7 receptor. J. Physiol. 519:335–346 10.1111/j.1469-7793.1999.0335m.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia J., Lim J.C., Lu W., Beckel J.M., Macarak E.J., Laties A.M., Mitchell C.H. 2012. Neurons respond directly to mechanical deformation with pannexin-mediated ATP release and autostimulation of P2X7 receptors. J. Physiol. 590:2285–2304 [DOI] [PMC free article] [PubMed] [Google Scholar]