Abstract
Due to the emergence of drug-resistance, first-line therapy with fluconazole (FLC) increasingly resulted in clinical failure for the treatment of candidemia. Our previous studies found that in vitro RTA2 was involved in the calcineurin-mediated resistance to FLC in C. albicans. In this study, we found that calcium-activated-calcineurin significantly reduced the in vitro sensitivity of C. albicans to FLC by blocking the impairment of FLC to the plasma membrane via Rta2p. Furthermore, we found that RTA2 itself was not involved in C. albicans virulence, but the disruption of RTA2 dramatically increased the therapeutic efficacy of FLC in a murine model of systemic candidiasis. Conversely, both re-introduction of one RTA2 allele and ectopic expression of RTA2 significantly reduced FLC efficacy in a mammalian host. Finally, we found that calcium-activated-calcineurin, through its target Rta2p, dramatically reduced the efficacy of FLC against candidemia. Given the critical roles of Rta2p in controlling the efficacy of FLC, Rta2p can be a potential drug target for antifungal therapies.
Introduction
Candida species are serious human fungal pathogens among immuno-compromised individuals such as patients with AIDS and patients undergoing organ transplantation, parenteral nutrition and radiation treatment for cancer [1]. Candida blood stream infections (candidemia) are life threatening among hospitalized immune-compromised patients, including neonates [2], [3]. Despite available therapy options, the mortality rate directly attributable to candidemia is high, ranging from 32% to 54%, depending on the population and the species studied [4], [5], [6]. Today, candidemia leads to additional ICU length of stay and costs, the annual treatment cost is at least £16.2 million [7], [8]. Although infections with non-albicans Candida species have emerged in recent years, Candida albicans is still responsible for the majority of cases [9], [10].
To date, only four classes of antifungal drugs are clinically available for treatment of systemic fungal infections: polyenes (amphotericin B), pyrimidines (particularly 5-fluorocytosine), azoles (e.g. fluconazole, FLC) and echinocandins (e.g. caspofungin) [11], [12]. Among these, azole antifungal drugs, such as FLC, are commonly used in the treatment of invasive candidiasis, and the mechanism of action of these drugs involves the blockade of ergosterol synthesis through the competitive inhibition of 14α-demethylase, which is encoded by ERG11 gene. Consequently, the inhibition of the enzyme leads to accumulation of 14-methylated sterols that can cause disruption of fungal membranes [13]. Although these drugs have advanced the management of fungal infections, the rate of therapeutic failure remains high. Most importantly, the small number of treatment options available has resulted in wide-spread drug resistance in pathogenic species. For each of the four classes of antifungals drugs (polyenes, pyrimidines, azoles, echinocandins), drug-resistant clinical strains has been reported [14]; Azole-resistant Candida spp. is now particularly common among isolates from HIV-positive patients [15]. Therefore, the need of developing new antifungal strategies is pressing.
Inhibition of calcineurin-mediated azole resistance has been proposed as a novel therapeutic approach [16]. Although the precise mechanisms involved remain elusive, extensive experiments have further confirmed the benefits of such combinatorial approaches. For instance, inhibitors of calcineurin (CsA, FK506) or inhibitors of calcineurin adaptor protein HSP90 (geldanamycin, radicicol) can chemosensitize C. albicans cells to azoles [17], [18], [19]. Although these examples sufficiently demonstrate the potential for combination antifungal therapy, suppression of the human immune system by CsA/FK506 currently precludes the use of such inhibitors in clinics [20], [21]. While a non-immunosuppressive FK506 analogue (L-685, 818) has been identified, proprietary restrictions have currently prevented further testing [20]. Given the importance of serious mycoses in clinical medicine and the need of alternative therapies for use in patients with fungal infections, research designed to identify new antifungal targets remains a priority.
We previously established that calcium-upregulated expression of RTA2 dramatically reduced the sensitivity of C. albicans to FLC by blocking its impairment to the fungal plasma membrane [22]. The expression of RTA2 was under the control of the calcium-activated-calcineurin via its transcriptional factor Crz1p in C. albicans [22]. However, it is unclear whether calcium-activated-calcineurin mediates in vitro and in vivo responses to FLC via Rta2p in C. albicans. Moreover, the therapeutic potential of targeting Rta2p remains elusive. Here, we investigated the possible influences of RTA2 expression on the in vitro and in vivo responses to FLC mediated by the calcium-activated-calcineurin in C. albicans.
Results
Calcium-activated-calcineurin Dramatically Reduces in vitro Sensitivity to FLC via Rta2p in C. albicans
It has been well documented that the phosphatase activity of calcineurin, consisted of a catalytic subunit A (encoded by CNA) and a regulatory subunit B (encoded by CNB1), can be activated by calcium ions and then regulate its target gene expression via transcriptional factor Crz1p [23]. In order to identify whether calcium-activated-calcineurin mediates the in vitro response to FLC via Rta2p in C. albicans, we sequentially disrupted two alleles of RTA2 gene in either cnaΔ/Δ or crz1Δ/Δ null mutant strains (Table 1), which were verified by Southern blot analysis with a probe specific to the RTA2 promoter (Figure S1). And then RTA2, CNA or CRZ1 was selectively re-introduced into either cnaΔ/Δ rta2Δ/Δ or crz1Δ/Δ rta2Δ/Δ mutant at the ade2 locus as determined by PCR (Figure S2A and S2B). Calcium treatment significantly up-regulated the expression of UTR2, a known target gene of the calcineurin pathway [23] in both rta2Δ/Δ cnaΔ/Δ::CNA and rta2Δ/Δ crz1Δ/Δ::CRZ1 complemented strains (Figure S3A), which confirmed that the function of the calcineurin pathway had been restored. The expression of RTA2 had no changes in both cnaΔ/Δ rta2Δ/Δ::RTA2 and crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains when treated with calcium (Figure S3B). These results confirmed that the expression of RTA2 was under the control of the calcium-activated-calcineurin via its transcriptional factor Crz1p in C. albicans.
Table 1. C. albicans strains used in this study.
| Strain | Parental strain | Genotype | Reference |
| RM1000 | RM100 | ura3Δ::imm434/ura3Δ::imm434, his1Δ::HisG/his1Δ::HisG, iro1Δ::imm434/iro1Δ::imm434 | [55] |
| RM1000U | RM1000 | RM1000 * ADE2/ade2::URA3 | This study |
| JXM101 | JXM100 | RM1000 * rta2Δ::hisG/rta2Δ::hisG | [22] |
| JXM201 | JXM101 | RM1000 * rta2Δ::hisG/rta2Δ::hisG ADE2/ade2::RTA2/URA3 | [37] |
| JXM101U | JXM101 | RM1000 * rta2Δ::hisG/rta2Δ::hisG ADE2/ade2::URA3 | This study |
| CAF2-1 | SC5314 | ura3Δ::imm434/URA3 | [56] |
| DSY2091 | CAF4-2 | cnaΔ::hisG/cnaΔ::hisG::URA3::hisG | [23] |
| DSY2115 | DSY2101 | cnaΔ::hisG/cnaΔ::hisG; LEU2::CNA::URA3 | [18] |
| DSY2195 | DSY2188 | crz1Δ::hisG/crz1Δ::hisG::URA3::hisG | [23] |
| MKY268 | MKY59 | crz1Δ::hisG/crz1Δ::hisG LEU2::CRZ1/URA3 | [23] |
| DSY2101 | DSY2091 | cnaΔ::hisG/cnaΔ::hisG | [18] |
| DJY201 | DSY2101 | rta2Δ::hisG/rta2Δ::hisG cnaΔ::hisG/cnaΔ::hisG | This study |
| DJYCNA | DJY201 | rta2Δ::hisG/rta2Δ::hisG cnaΔ::hisG/cnaΔ::hisG ADE2/ade2::CNA/URA3 | This study |
| DJYRTA2 | DJY201 | cnaΔ::hisG/cnaΔ::hisG rta2Δ::hisG/rta2Δ::hisG ADE2/ade2::RTA2/URA3 | This study |
| MKY59 | DSY2195 | crz1Δ::hisG/crz1Δ::hisG | [23] |
| MJY201 | MKY59 | rta2Δ::hisG/rta2Δ::hisG crz1Δ::hisG/crz1Δ::hisG | This study |
| MJYCRZ1 | MJY201 | rta2Δ::hisG/rta2Δ::hisG crz1Δ::hisG/crz1Δ::hisG ADE2/ade2::CRZ1/URA3 | This study |
| MJYRTA2 | MJY201 | crz1Δ::hisG/crz1Δ::hisG rta2Δ::hisG/rta2Δ::hisG ADE2/ade2::RTA2/URA3 | This study |
| DSY9u | DSY1024 | cdr1Δ::hisG/cdr1Δ::hisG cdr2Δ::hisG/cdr2Δ::hisG camdr1Δ::hisG/camdr1Δ::hisG flu1Δ::hisG/flu1Δ::hisG | This study |
| DSJ101 | DSJ100 | DSY9u # rta2Δ::hisG/rta2Δ::hisG | [57] |
| DSJRTA2 | DSJ101 | DSY9u # rta2Δ::hisG/rta2Δ::hisG ADE2/ade2::pCDR2-RTA2/URA3 | This study |
| DSJ101U | DSJ101 | DSY9u # rta2Δ::hisG/rta2Δ::hisG ADE2/ade2::URA3 | This study |
RM1000 background;
DSY9u background.
And then the influences of calcium signaling on the sensitivities of above-constructed mutants to FLC were examined. The addition of calcium (1 mM) significantly reduced the sensitivities of FLC to both cnaΔ/Δ::CNA and crz1Δ/Δ::CRZ1 complemented strains, with an increase of MIC80s(Minimum Inhibitory Concentration for 80%)from 0.5 to 16 µg/ml (Table 2). However, re-introduction of anyone of CNA, CRZ1 and RTA2 could not restore the effect of calcium on reducing FLC sensitivities to complemented strains including rta2Δ/Δ cnaΔ/Δ::CNA, cnaΔ/Δ rta2Δ/Δ::RTA2, rta2Δ/Δ crz1Δ/Δ::CRZ1 and crz1Δ/Δ rta2Δ/Δ::RTA2 (Table 2). But the effect of calcium on reducing FLC sensitivity was completely restored in the rta2Δ/Δ::RTA2 complemented strain, with an increase of MIC80s from 0.5 to 64 µg/ml (Table 2). In contrast to the results presented above, the addition of calcium (1 mM) had no effect on the antifungal activity of caspofungin in all tested strains (Table 2). Taken together, these results suggested that calcium-activated-calcineurin dramatically reduced the in vitro sensitivity to FLC via Rta2p in C. albicans (Figure S4).
Table 2. Effects of calcium on the antifungal activity versus mutant C. albicans strains.
| Antifungal MIC80 (µg/ml)a | Strains | |||||||
| cnaΔ/Δ::CNA | rta2Δ/Δ cnaΔ/Δ::CNA | cnaΔ/Δ rta2Δ/Δ::RTA2 | crz1Δ/ Δ::CRZ1 | rta2Δ/Δ crz1Δ/ Δ::CRZ1 | crz1Δ/Δ rta2Δ/ Δ::RTA2 | rta2Δ/Δ::RTA2 | ||
| Fluconazole | – | 0.5 | 0.125 | 0.06 | 0.5 | 0.125 | 0.125 | 0.5 |
| +1mM CaCl2 | 16 | 0.25 | 0.125 | 16 | 0.25 | 0.25 | 64 | |
| Caspofungin | – | 0.3 | 0.6 | 0.6 | 0.3 | 0.3 | 0.15 | 0.3 |
| +1mM CaCl2 | 0.3 | 0.6 | 0.6 | 0.6 | 0.6 | 0.3 | 0.3 | |
MIC80s were determined after 48 h incubation and all experiments were performed in triplicate.
Calcium-activated-calcineurin Blocks the Impairment of FLC to the Plasma Membrane of C. albicans via Rta2p
In our previous study, it was found that the depletion of RTA2 made the plasma membranes of C. albicans liable to be destroyed by FLC and calcium-upregulated expression of RTA2 attenuated the destroying effects [22]. In the present study, transmission electron microscopy was performed to observe the ultra-structure changes of crz1Δ/Δ::CRZ1, rta2Δ/Δ crz1Δ/Δ::CRZ1 and crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains in the presence or absence of FLC or FLC plus calcium. High magnification of these three untreated or calcium-treated complemented strains showed intact cell membranes (Figure 1 A to F). As shown in Figure 1G and 1M, the cell membranes of the crz1Δ/Δ::CRZ1 complemented strain remained intact when treated with 2 µg/ml FLC and were modestly damaged when treated by 8 µg/ml FLC. Surprisingly, the addition of calcium could completely attenuate the destroying effects of FLC to the plasma membrane of crz1Δ/Δ::CRZ1 complemented strain (Figure 1N). However, re-introduction of one CRZ1 or RTA2 allele into crz1Δ/Δ rta2Δ/Δ mutant failed to attenuate the destroying effects of 2 µg/ml FLC to the plasma membranes of either rta2Δ/Δ crz1Δ/Δ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains (Figure 1I and 1K). And extensive solubilization of intracytoplasmic inclusion bodies was seen in either rta2Δ/Δ crz1Δ/Δ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains when treated by 8 µg/ml FLC (Figure 1O and 1Q). The addition of calcium also failed to attenuate the destroying effects of FLC to the plasma membranes of either rta2Δ/Δ crz1Δ/Δ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains (Figure 1 P and R). Taken together, these results suggested a model in which the calcium-activated-calcineurin blocked the impairment of FLC to the plasma membrane of C. albicans via Rta2p.
Figure 1. Ultra-structure of C. albicans cells.
(A to R) Ultra-structural images of crz1Δ/Δ::CRZ1, rta2Δ/Δ crz1Δ/Δ::CRZ1 and crz1Δ/Δ rta2Δ/Δ::RTA2 complemented C. albicans strains in the presence or absence of agents at indicated concentrations shown on the lower. Arrow heads indicated the cytoplasmic membrane and arrows indicated the extensive solubilization of the cytoplasmic membrane. The bar represents a length of 0.2 µm.
Rta2p is Critical for the Therapeutic Efficacy of FLC Against Candidemia
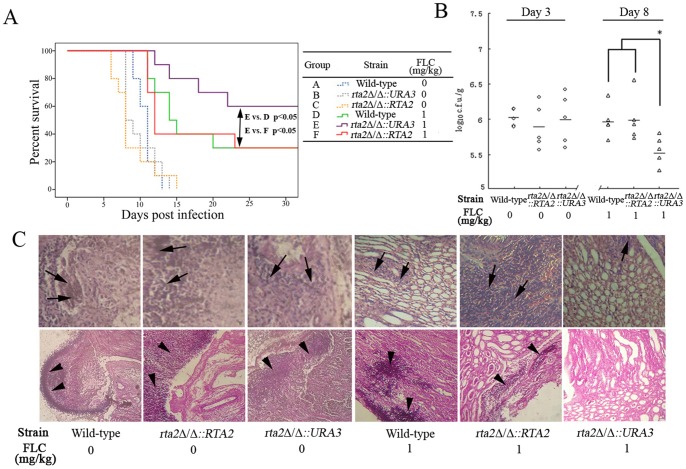
It has been well documented that calcineurin is essential for the virulence of C. albicans in systemic infection models and for the emergence of FLC resistance to C. albicans [24], [25]. As a target gene of the calcineurin pathway [22], the influence of RTA2 on either virulence or efficacy of FLC remains unknown. To examine the influence of RTA2 on C. albicans virulence, groups of 20 mice were intravenously injected with the wild type, rta2Δ/Δ mutant and rta2Δ/Δ::RTA2 complemented strains, and the survival of mice was monitored for 30 days. As shown in Figure 2A, the comparison of the survival times among groups of mice infected by the wild-type, rta2Δ/Δ mutant and rta2Δ/Δ::RTA2 complemented strains showed no significant differences. Furthermore, both the tissue burdens and histological analysis determined at day 3 post-inoculation consistently showed similar phenotypes (Figure 2 B and 2C). These findings proved that RTA2 was not involved in the virulence of C. albicans.
Figure 2. Rta2p played an important role in FLC efficacy against systemic candidiasis.
(A) Survival curves of different groups of mice infected by C. albicans strains. If indicated, groups of mice received intraperitoneal FLC therapy at 1 mg/kg or placebo once a day for a week. (B) Kidney c.f.u. assay in mice with systemic candidiasis. Paired kidneys of untreated and FLC-treated mice were removed aseptically for c.f.u. assay on day 3 and 8 post infection, respectively. * P<0.05 compared with groups of mice infected with either wild-type or rta2Δ/Δ::RTA2 complemented strains after receiving FLC therapy. (C) Histopathological analysis of kidneys from mice with systemic candidiasis. Paired kidneys of untreated and FLC-treated mice were removed aseptically for histopathological analysis on day 3 and 8 post infection, respectively. The top panels show H&E sections; the bottom panels show PAS staining in kidney sections. Arrows indicate abscesses; arrow heads indicate fungi.
We then investigated the influences of RTA2 on the efficacy of FLC against C. albicans in a murine model of systemic candidiasis. FLC treatment resulted in about 30% survival rate of mice infected with wild-type strain in 30 days (Figure 2A). In contrast, the disruption of RTA2 significantly improved the survival of mice infected with rta2Δ/Δ mutant to 60% (Figure 2A). More importantly, this improved survival dropped to 30% when infected with rta2Δ/Δ::RTA2 complemented strain (Figure 2A). When the fungal burden in renal tissues was counted at day 8 post-inoculation, we noticed that, due to FLC treatment, mice infected with rta2Δ/Δ mutant exhibited a lower fungal load than mice infected with either wild-type or rta2Δ/Δ::RTA2 complemented strains (P<0.05; Figure 2B), which was consistent with the results of survival analysis. A similar trend was also shown in kidney pathological changes analysis (Figure 2C). Therefore, these results suggested that Rta2p plays a critical role on the efficacy of FLC against systemic candidiasis.
Ectopic Expression of RTA2 Reduced the Efficacy of FLC Against Candidemia
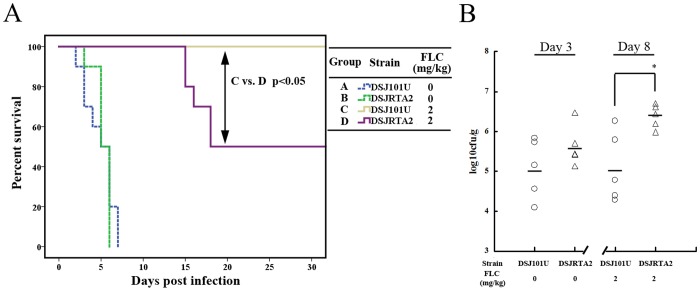
In order to verify the direct influences of RTA2 on the efficacy of FLC, the expression of RTA2 ORF was put under the control of the oestradiol-inducible CDR2 promoter (pCDR2) and the plasmid pCDR2-RTA2 was introduced into a C. albicans mutant (DSJ101) with deletion of drug-resistance-related genes including CDR1, CDR2, CaMDR1, FLU1 and RTA2 at the ade2 locus as determined by PCR (Figure S2C). Up-regulation of RTA2 in DSJRTA2 harboring the plasmid pCDR2-RTA2 by oestradiol was confirmed by quantitative RT-PCR (Figure S3C).
Survival analysis showed that DSJRTA2 and its parental strain (DSJ101U) had similar virulence, and all mice died within 10 days (Figure 3A). The kidney fungal burdens, determined at day 3 post-inoculation, consistently reflected virulence phenotypes observed between DSJRTA2 and DSJ101U (Figure 3B), which confirmed that RTA2 was not involved in C. albicans virulence. Of note, FLC treatment made all DSJ101U-infected mice survive during the experiments (Figure 3A). However, only 50% of the DSJRTA2-infected mice survived after treatment with FLC (Figure 3A). Kidneys of mice infected with DSJRTA2 exhibited a significantly higher fungal burden than mice infected with its parental strain DSJ101U (P<0.05, Figure 3B). Therefore, these results suggested that Rta2p directly affects the efficacy of FLC against candidiasis.
Figure 3. Ectopic expression of RTA2 in vivo significantly reduced FLC efficacy against candidiasis.
(A) Survival curves of different groups of mice infected by DSJ101U with deletion of drug-resistance-related genes (CDR1, CDR2, CaMDR1, FLU1 and RTA2) or DSJRTA2 carrying the plasmid pCDR2-RTA2. If indicated, groups of mice received intraperitoneal FLC therapy at 2 mg/kg or placebo once a day for a week. (B) Kidney c.f.u. assay in mice with systemic candidiasis. Paired kidneys of untreated and FLC-treated mice were removed aseptically for c.f.u. assay on day 3 and 8 post infection, respectively. * P<0.05 compared with groups of mice infected by DSJ101U with FLC therapy.
Calcium-activated-calcineurin, through its Target Rta2p, Significantly Reduces the Efficacy of FLC Against Candidemia
The expression of RTA2 has been reported to be under the control of the calcium-activated-calcineurin via its transcriptional factor Crz1p in C. albicans [22], [23]. Since mammalian contains a range of ionized calcium in whole blood (0.94–1.33 mM) [26], it was examined whether the calcineurin pathway of C. albicans could be activated by ionized calcium in serum and then the calcium-activated-calcineurin mediated in vivo responses of C. albicans to FLC via Rta2p.
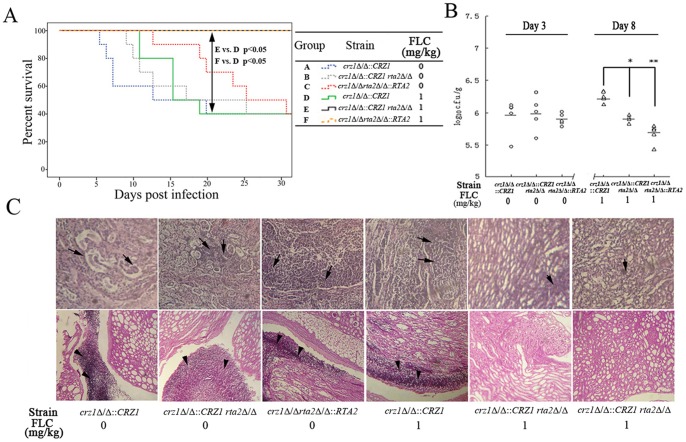
After receiving FLC treatment, survival analysis showed that re-introduction of either one CRZ1 or RTA2 allele into crz1Δ/Δ rta2Δ/Δ mutant led to 100% of survival (Figure 4A). However, re-introduction of one CRZ1 allele into crz1Δ/Δ mutant significantly reduced survival rate to 50% (Figure 4A). The fungal burdens of mice infected with the crz1Δ/Δ::CRZ1 complemented strain were significantly higher than those infected with either the rta2Δ/Δ crz1Δ/Δ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains (P<0.05 and P<0.01, respectively; Figure 4B). Histological analysis with HE staining also showed that kidney damage was aggravated in mice infected with crz1Δ/Δ::CRZ1 complemented strain infected when compared with either rta2Δ/Δ crz1Δ/Δ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains (Figure 4C). PAS staining also revealed a large number of fungi in kidneys from the crz1Δ/Δ::CRZ1 complemented strain infected mice and hardly any fungi in mice infected with either the rta2Δ/Δ crz1Δ/Δ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains (Figure 4C). Therefore, the results of H&E and PAS staining were consistent with those of survival analysis and kidney fungal burdens. Overall, ionized calcium in mice serum could not reduce the efficacy of FLC against bloodstream infection with C. albicans mutants due to their lack of either CRZ1 or RTA2, which indicated that calcium-activated-calcineurin affects the efficacy of FLC against candidemia via Rta2p.
Figure 4. Calcium-activated-calcineurin mediated the in vivo response to FLC via Rta2p in C. albicans.
(A) Survival curves of among groups of mice infected by crz1Δ/Δ::CRZ1, rta2Δ/Δcrz1Δ/Δ ::CRZ1 or crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains. If indicated, groups of mice received received intraperitoneal FLC therapy at 1 mg/kg or placebo once a day for a week. (B) Kidney c.f.u. assay in mice with systemic candidiasis. Paired kidneys of untreated and FLC-treated mice were removed aseptically for c.f.u. assay on day 3 and 8 after the infection, respectively. * P<0.05 or * P<0.01 compared with groups of mice infected by crz1Δ/Δ::CRZ1 after receiving FLC therapy. (C) Histopathological analysis of kidneys from mice with systemic candidiasis. Paired kidneys of untreated and FLC-treated mice were removed aseptically for histopathological analysis on day 3 and 8 post infection, respectively. The top panels show H&E sections; the bottom panels show PAS staining in kidney sections. Arrows indicate abscesses; arrowheads indicate fungi.
Discussion
FLC has long been the drug of choice for candidemia, owing to its excellent safety profile and efficacy against key causative pathogens. However, because of the associated rise in FLC-resistant Candida infections and the increased risk of death due to FLC failure as first-line therapy [9], [27], the need of identifying new antifungal strategies to enhance FLC efficacy against Candida infections is pressing. A major limitation of FLC is its emergence of tolerance. As a result, many individuals require long-term treatment of FLC, and this in turn frequently results in the selection of FLC-resistant fungal strains. In C. albicans, calcineurin has been shown to be essential for virulence and emergence of FLC tolerance [24], [25]. Inactivation of the pathway by CsA or FK506, or by deletion of the gene encoding one of the calcineurin subunits, in combination with FLC resulted in the killing of cells by this otherwise fungistatic drug [18], [28], [29]. Amiodarone (a calcium channels inhibitor) was also reported to be significantly synergistic with FLC against fluconazole-resistant C. albicans in a calcineurin/Crz1p dependent manner [30], [31]. Several lines of evidence suggested that iron, AGE3 and HSP90 mediated antifungal drug susceptibilities cross talk with the calcineurin signaling pathway [17], [32], [33], [34] and calcineurin could be targeted to enhance FLC therapy [16], [35], [36]. Our previous study showed that the disruption of RTA2 could block the emergence of calcium-mediated tolerance to FLC by aggravating its impairment to the plasma membrane of C. albicans [22]. The expression of RTA2 was found to be under the control of the Ca2+-activated-calcineurin via its transcriptional factor Crz1p in C. albicans [22], [23].
In the present study, we found that RTA2 itself was not involved in C. albicans virulence (Figure 2). However, the disruption of RTA2 significantly augmented the therapeutic efficacy of FLC against systemic candidiasis. Conversely, both re-introduction of one RTA2 allele and ectopic expression of RTA2 significantly reduced FLC efficacy in a murine model of systemic candidiasis (Figure 2 and 3). Furthermore, calcium-activated-calcineurin could not complement the in vitro calcium-mediated tolerance to FLC and attenuate its impairment to the plasma membrane of C. albicans when genetically compromising the function of Rta2p (Table 2 and Figure 1). The consistent conclusion was drawn by the in vivo experiments that FLC treatment was significantly more efficacious when CRZ1 was restored in crz1Δ/Δ rta2Δ/Δ mutant but not crz1Δ/Δ mutant (Figure 4). Taken together, our study implies that calcium-activated-calcineurin mediates the in vitro and in vivo responses of C. albicans to FLC via Rta2p, which is an important determinant of FLC tolerance in C. albicans. Given the critical roles by Rta2p in controlling the efficacy of FLC, Rta2p can be an attractive target for the development of powerful and effective therapy strategies against life-threatening fungal infections. However, the molecular mechanisms involved remain unknown. Our previous studies showed that Rta2p acted as a sphingolipid long chain base transporter protein and was critical for the association of certain transmembrane (TM) and glycosylphosphatidylinositol (GPI)-anchored proteins with lipid rafts in C. albicans [22], [37]. And ceramide biosynthesis is also essential for the association of TM and GPI-anchored proteins with lipid rafts [38], [39], [40]. In Saccharomyces cerevisiae, calcineurin signaling is necessary for normal sphingolipid metabolism by regulating ceramide production [41], [42]. Therefore, we speculate that Rta2p, as a target of the calcineurin pathway, may mediate in vitro and in vivo responses of C. albicans to FLC by regulating the biosynthesis of ceramides.
During an infection, the C. albicans cells exist in the host body and are surrounded by blood and other body fluid, where they encounter the antifungal drugs. Previous studies have demonstrated that biological fluids such as serum can have profound effects on antifungal susceptibilities [43], [44], [45], [46]. Of particular note was the blood ionized calcium (0.94–1.33 mM) [26], which was in vitro sufficient to activate the protein phosphatase activity of calcineurin in S. cerevisiae and C. albicans [22], [47], [48]. However, the standard antifungal susceptibility testing [49], which is conducted in RPMI-1640 media containing only 0.42 mM ionized calcium [50], is unreliable in predicting the clinical outcome of therapies, especially for systemic infections [51], [52]. Our finding may partially explain the existence and persistence of such a discrepancy between the susceptibilities of in vivo and in vitro environments.
Materials and Methods
Drugs and Agents
FLC was from Pfizer Inc (New York, N.Y.). Caspofungin was purchased from Merck (Whitehouse Station, N.J.). RPMI 1640 medium (with L-glutamine and without sodium bicarbonate), morpholinepropanesulfonic acid (MOPS), and CaCl2 were purchased from Sigma Chemical Co. (Cleveland, Ohio).
C. albicans Strains and Culture Media
C. albicans strains used in this study are listed in Table 1 and cultured in YPD medium (1% yeast extract, 2% Bacto peptone, and 2% dextrose) or SC medium (0.67% yeast nitrogen base with ammonium sulfate without amino acids, 2% glucose, 0.077% complete supplement mixture minus uracil) supplemented with 50 µg/mluridine as required.
Construction of Mutant Strains
All the primer sequences are listed in Table S1. The XhoI digested fragment of pUC-RTA2-URA3, constructed by us previously [22], was transformed into the ura3Δ/Δ cnaΔ/Δ (DSY2101) or ura3Δ/Δ crz1Δ/Δ (MKY59) mutant (Table 1) by standard methods [53].
Construct of the CNA and CRZ1 Revertant Strains
The genes (CNA, CRZ1 and RTA2) containing their ORFs and 1 kb up/downstream were amplified by PCR with Pyrobest polymerase (TaKaRa). The PstI-KpnI digested PCR fragments of CNA and RTA2 was ligated into plasmid pBes116 [54], and then plasmids pBes-CNA and pBes-RTA2 were obtained.The NotI-KpnI digested PCR fragment of CRZ1 was ligated into pBes116, and plasmid pBes-CRZ1 was obtained. And then, the AscI digested fragment of pBes-CNA, pBes-CRZ1 or pBes-RTA2 was transformed into DJY201 (cnaΔ/Δ rta2Δ/Δ), MJY201 (crz1Δ/Δ rta2Δ/Δ) or JXM101 (rta2Δ/Δ) as before [22].
Construct of RTA2 Fused to pCDR2 in RTA2 Mutants
The fragment containing CDR2 promoter sequence and ORF of RTA2 was obtained by the Fusion PCR methods. The PstI-KpnI digested PCR fragment was ligated into pBes116, and recombinant plasmid pBesCDR2-RTA2 was obtained. DNA sequencing confirmed that the sequence of the insert was identical to CDR2 and RTA2 sequence reported in the Candida Genome Database (http://www.candidagenome.org/). The C. albicans mutant DSJ101 with deletion of drug-resistance related genes including CDR1, CDR2, CaMDR1, FLU1 and RTA2 was transformed with the linearized plasmid pBesCDR2-RTA2 by AscI and selected on SC medium without uridine as before [22].
Relative Quantification of Genes by Quantitative RT-PCR
All the primer sequences are listed in Table S1. Quantitative RT-PCR was done as described previously [22].
Susceptibility Testing
Drug sensitivities were assayed by the modified broth microdilution method as described previously [22]. Briefly, RPMI 1640 medium was adjusted to pH 7 at 25°C using 3-[N-morpholino]propanesulphonic acid (MOPS).The initial concentration of the fungal suspension in the RPMI 1640 medium was 1–5×103 c.f.u./mL, 0.1 ml of cells were inoculated into successive wells of a 96-well microtiter plate containing serial two-fold dilutions of antifungal drugs. Drug-free medium with fungi and a fungi-free medium were used as the positive and negative controls, respectively. After incubation at 35°C for 48 h, absorbance at 630 nm was determined in a microplate reader. MIC80 were estimated. Assays for susceptibility to FLC and caspofungin were also performed in the presence of a fixed concentration (1 mM) of CaCl2.
Transmission Electron Microscopy
With or without treatment of FLC, CaCl2 or FLC plus CaCl2, transmission electron microscopies of C. albicans cells including the crz1Δ/Δ::CRZ1, rta2Δ/Δ crz1Δ/Δ::CRZ1 and crz1Δ/Δ rta2Δ/Δ::RTA2 complemented strains (Table 1) were obtained according to our previously described protocol [22].
Systemic Murine Candidiasis Model
Groups of ICR female mice (18–22 g) were inoculated via lateral tail vein with 200 µl of a suspension containing 1×106 c.f.u different C. albicans strains in sterile saline. The following regimens were administered to the infected mice: FLC at 1 mg/kg or 2 mg/kg once a day intraperitoneally for a week, or placebo once a day. Mice were monitored daily for survival for a period of 30 days. Duplicate independent experiments were conducted (n = 10 in each group). Kaplan–Meier and Life Table analyses were used to estimate survival probabilities. Log-rank testing was used to evaluate the equality of survival curves. P<0.05 was considered significant.
Kidney c.f.u. Assay and Histopathological Analysis
Paired kidneys of untreated and FLC-treated mice were removed aseptically on day 3 and 8 post inoculation, which were weighed and homogenized in 5 ml sterile physiological saline. Serial dilutions were carried out in Sabouraud chloramphenicol agar to determine the c.f.u./g kidney. Statistical analyses were performed using anova and post hoc (Bonferroni and Student–Newman–Keuls’) tests. P<0.05 was considered significant. For histopathological analysis, paired kidneys of untreated and FLC-treated mice were removed aseptically on day 3 and 8 post infection from each mouse before being fixed in 10% neutral buffered formalin. Kidneys were stained using haematoxylin and eosin (H&E) and periodic acid-Schiff (PAS) to reveal inflammatory infiltration, microabscesses, abscesses and necrosis (H&E staining) and the hyphal structure of the fungal pathogens (PAS). Tissues were examined microscopically.
Ethics Statement
The animal experiments were approved by the Animal Ethics Committee of Second Military Medical University (Shanghai, China). All surgery was performed under sodium pentobarbital anesthesia, and all efforts were made to minimize suffering of the animals.
Supporting Information
Schematic representation of disruption of RTA2 using the URA3 selection marker. (A) Sequential targeted disruption of the two RTA2 alleles in C. albicans with the disruption cassette. (B) Southern analysis of the genomic DNA digested with BglII and SalI. The exact size and genotype of the expected hybridizing DNA fragment are indicated on the right.
(TIF)
(A) Strains, with one allele of RTA2 reintroduced into ADE2 locus, yielded only one 3.8 kb PCR product by PCR analysis with primer specific to ADE2 and primer specific to RTA2, with plasmid pBes-RTA2 as control. (B) Strains, with one allele of CNA or CRZ1 reintroduced into ADE2 locus, yielded only one 2.5 kb PCR product by PCR analysis with primer specific to ADE2 and primer specific to URA3, with plasmid pBes116 as control. (C) Strains, with the fusion fragment of CDR2 promoter and ORF of RTA2 reintroduced into ADE2 locus, yielded only one 3.9 kb PCR product by PCR analysis with primer specific to ADE2 and primer specific to RTA2, with plasmid pBesCDR2-RTA2 as control.
(TIF)
(A) Expression levels of UTR2 were examined by quantitative RT-PCR in the wild-type strain (CAF2-1), DJYCNA (rta2Δ/Δ cnaΔ/Δ::CNA) and MJYCRZ1 (rta2Δ/Δ crz1Δ/Δ::CRZ1) after exposure to CaCl2 (200 mM) for 2 h, with their corresponding drug-free strains as controls. (B) Expression levels of RTA2 were examined by quantitative RT-PCR in the wild-type strain (CAF2-1), DJYRTA2 (cnaΔ/Δ rta2Δ/Δ::RTA2) and MJYRTA2 (crz1Δ/Δ rta2Δ/Δ::RTA2) after exposure to CaCl2 (1 mM) for 16 h, with their corresponding drug-free strains as controls. (C) Expression levels of RTA2 were examined by quantitative RT-PCR in the parental strain (DSJ101U), DSJ-RTA2 carrying the fusion of the oestradiol-inducible CDR2 promoter and ORF of RTA2, in the presence of oestradiol (OST, 10 ug/ml), with their corresponding drug-free strains as controls. Data are represented as means ± standard deviation.
(TIF)
Schematic view of the calcineurin pathway in Candida albicans.
(TIF)
Primers used in this study.
(DOCX)
Acknowledgments
We thank J. Pla for kindly providing the C. albicans strain RM1000, W. A. Fonzi for plasmid p5921, Jiang-Ye Chen for plasmid pBes116, D. Sanglard for the gifts of DSY1024 and MKY59.
Funding Statement
This study was supported by the National Science Foundation of China (30900055, 30825041 and 90913008). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Schelenz S, Abdallah S, Gray G, Stubbings H, Gow I, et al. (2011) Epidemiology of oral yeast colonization and infection in patients with hematological malignancies, head neck and solid tumors. J Oral Pathol Med 40: 83–89. [DOI] [PubMed] [Google Scholar]
- 2. Pagano L, Caira M, Valentini CG, Posteraro B, Fianchi L (2010) Current therapeutic approaches to fungal infections in immunocompromised hematological patients. Blood Rev 24: 51–61. [DOI] [PubMed] [Google Scholar]
- 3. Zaoutis T (2010) Candidemia in children. Curr Med Res Opin 26: 1761–1768. [DOI] [PubMed] [Google Scholar]
- 4. Ortega M, Marco F, Soriano A, Almela M, Martinez J, et al. (2011) Candida species bloodstream infection: epidemiology and outcome in a single institution from 1991 to 2008. J Hosp Infect 77: 157–161. [DOI] [PubMed] [Google Scholar]
- 5. Bassetti M, Taramasso L, Nicco E, Molinari MP, Mussap M, et al. (2011) Epidemiology, Species Distribution, Antifungal Susceptibility and Outcome of Nosocomial Candidemia in a Tertiary Care Hospital in Italy. PLoS One 6: e24198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Jutiamornlerd N, Chusri S, Siripaitoon P (2011) Epidemiology of Candidemia in Songklanagarind Hospital. J Med Assoc Thai 94: 927–932. [PubMed] [Google Scholar]
- 7. Wilson LS, Reyes CM, Stolpman M, Speckman J, Allen K, et al. (2002) The direct cost and incidence of systemic fungal infections. Value Health 5: 26–34. [DOI] [PubMed] [Google Scholar]
- 8. Hassan I, Powell G, Sidhu M, Hart W, Denning D (2009) Excess mortality, length of stay and cost attributable to candidaemia. J Infect 59: 360–365. [DOI] [PubMed] [Google Scholar]
- 9. Horn DL, Neofytos D, Anaissie EJ, Fishman JA, Steinbach WJ, et al. (2009) Epidemiology and outcomes of candidemia in 2019 patients: data from the prospective antifungal therapy alliance registry. Clin Infect Dis 48: 1695–1703. [DOI] [PubMed] [Google Scholar]
- 10. Cortes JA, Reyes P, Gómez C, Buitrago G, Leal AL (2011) Fungal bloodstream infections in tertiary care hospitals in Colombia. Rev Iberoam Micol 28: 74–78. [DOI] [PubMed] [Google Scholar]
- 11. Cowen LE (2008) The evolution of fungal drug resistance: modulating the trajectory from genotype to phenotype. Nat Rev Microbiol 6: 187–198. [DOI] [PubMed] [Google Scholar]
- 12. Chapman SW, Sullivan DC, Cleary JD (2008) In search of the holy grail of antifungal therapy. Trans Am Clin Climatol Assoc 119: 197–215. [PMC free article] [PubMed] [Google Scholar]
- 13. Sionov E, Chang YC, Garraffo HM, Dolan MA, Ghannoum MA, et al. (2012) Identification of a Cryptococcus neoformans Cytochrome P450 Lanosterol 14α-Demethylase (Erg11) Residue Critical for Differential Susceptibility between Fluconazole/Voriconazole and Itraconazole/Posaconazole. Antimicrob Agents Chemother 56: 1162–1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Cannon RD, Lamping E, Holmes AR, Niimi K, Baret PV, et al. (2009) Efflux-mediated antifungal drug resistance. Clin Microbiol Rev 22: 291–321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Traeder C, Kowoll S, Arastéh K (2008) Candida infection in HIV positive patients 1985–2007. Mycoses 51: 58–61. [DOI] [PubMed] [Google Scholar]
- 16. Steinbach WJ, Reedy JL, Cramer RA, Perfect JR, Heitman J (2007) Harnessing calcineurin as a novel anti-infective agent against invasive fungal infections. Nat Rev Microbiol 5: 418–430. [DOI] [PubMed] [Google Scholar]
- 17. Singh SD, Robbins N, Zaas AK, Schell WA, Perfect JR, et al. (2009) Hsp90 governs echinocandin resistance in the pathogenic yeast Candida albicans via calcineurin. PLoS pathogens 5: e1000532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Sanglard D, Ischer F, Marchetti O, Entenza J, Bille J (2003) Calcineurin A of Candida albicans: involvement in antifungal tolerance, cell morphogenesis and virulence. Mol Microbiol 48: 959–976. [DOI] [PubMed] [Google Scholar]
- 19. Cowen LE, Lindquist S (2005) Hsp90 potentiates the rapid evolution of new traits: drug resistance in diverse fungi. Science 309: 2185–2189. [DOI] [PubMed] [Google Scholar]
- 20. Hoon S, Smith AM, Wallace IM, Suresh S, Miranda M, et al. (2008) An integrated platform of genomic assays reveals small-molecule bioactivities. Nat Chem Biol 4: 498–506. [DOI] [PubMed] [Google Scholar]
- 21. Cowen LE, Singh SD, K hlerJR, Collins C, Zaas AK, et al. (2009) Harnessing Hsp90 function as a powerful, broadly effective therapeutic strategy for fungal infectious disease. Proc Natl Acad Sci U S A 106: 2818–2823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Jia XM, Wang Y, Jia Y, Gao PH, Xu YG, et al. (2009) RTA2 is involved in calcineurin-mediated azole resistance and sphingoid long-chain base release in Candida albicans. Cell Mol Life Sci 66: 122–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Karababa M, Valentino E, Pardini G, Coste AT, Bille J, et al. (2006) CRZ1, a target of the calcineurin pathway in Candida albicans. Mol Microbiol 59: 1429–1451. [DOI] [PubMed] [Google Scholar]
- 24. Blankenship JR, Wormley FL, Boyce MK, Schell WA, Filler SG, et al. (2003) Calcineurin is essential for Candida albicans survival in serum and virulence. Eukaryot Cell 2: 422–430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Bader T, Bodendorfer B, Schroppel K, Morschhauser J (2003) Calcineurin is essential for virulence in Candida albicans. Infect Immun 71: 5344–5354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Moore EW (1970) Ionized calcium in normal serum, ultrafiltrates, and whole blood determined by ion-exchange electrodes. J Clin Invest 49: 318–334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Pfaller MA, Diekema DJ, Rinaldi MG, Barnes R, Hu B, et al. (2005) Results from the ARTEMIS DISK Global Antifungal Surveillance Study: a 6.5-year analysis of susceptibilities of Candida and other yeast species to fluconazole and voriconazole by standardized disk diffusion testing. J Clin Microbiol 43: 5848–5859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Cruz MC, Goldstein AL, Blankenship JR, Del Poeta M, Davis D, et al. (2002) Calcineurin is essential for survival during membrane stress in Candida albicans. Embo J 21: 546–559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sun S, Li Y, Guo Q, Shi C, Yu J, et al. (2008) In vitro interactions between tacrolimus and azoles against Candida albicans determined by different methods. Antimicrob Agents Chemother 52: 409–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gamarra S, Rocha EMF, Zhang YQ, Park S, Rao R, et al. (2010) Mechanism of the synergistic effect of amiodarone and fluconazole in Candida albicans. Antimicrob Agents Chemother 54: 1753–1761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Zhang YQ, Gamarra S, Garcia-Effron G, Park S, Perlin DS, et al. (2010) Requirement for ergosterol in V-ATPase function underlies antifungal activity of azole drugs. PLoS pathogens 6: e1000939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Epp E, Vanier G, Harcus D, Lee AY, Jansen G, et al. (2010) Reverse genetics in Candida albicans predicts ARF cycling is essential for drug resistance and virulence. PLoS Pathog 6: e1000753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Cowen LE (2009) Hsp90 orchestrates stress response signaling governing fungal drug resistance. PLoS Pathog 5: e1000471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Hameed S, Dhamgaye S, Singh A, Goswami SK, Prasad R (2011) Calcineurin signaling and membrane lipid homeostasis regulates iron mediated multidrug resistance mechanisms in Candida albicans. PLoS One 6: e18684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Marchetti O, Entenza JM, Sanglard D, Bille J, Glauser MP, et al. (2000) Fluconazole plus cyclosporine: a fungicidal combination effective against experimental endocarditis due to Candida albicans. Antimicrob Agents Chemother 44: 2932–2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Onyewu C, Afshari NA, Heitman J (2006) Calcineurin promotes infection of the cornea by Candida albicans and can be targeted to enhance fluconazole therapy. Antimicrob Agents Chemother: 3963–3965. [DOI] [PMC free article] [PubMed]
- 37. Wang L, Jia Y, Tang RJ, Xu Z, Cao YB, et al. (2012) Proteomic Analysis of Rta2p-Dependent Raft-Association of Detergent-Resistant Membranes in Candida albicans. PLoS One 7: e37768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Lee MCS, Hamamoto S, Schekman R (2002) Ceramide biosynthesis is required for the formation of the oligomeric H+-ATPase Pma1p in the yeast endoplasmic reticulum. J Biol Chem 277: 22395–22401. [DOI] [PubMed] [Google Scholar]
- 39. Gaigg B, Timischl B, Corbino L, Schneiter R (2005) Synthesis of sphingolipids with very long chain fatty acids but not ergosterol is required for routing of newly synthesized plasma membrane ATPase to the cell surface of yeast. J Biol Chem 280: 22515–22522. [DOI] [PubMed] [Google Scholar]
- 40. Gaigg B, Toulmay A, Schneiter R (2006) Very long-chain fatty acid-containing lipids rather than sphingolipids per se are required for raft association and stable surface transport of newly synthesized plasma membrane ATPase in yeast. J Biol Chem 281: 34135–34145. [DOI] [PubMed] [Google Scholar]
- 41. Mulet JM, Martin DE, Loewith R, Hall MN (2006) Mutual antagonism of target of rapamycin and calcineurin signaling. Journal of Biological Chemistry 281: 33000–33007. [DOI] [PubMed] [Google Scholar]
- 42. Aronova S, Wedaman K, Aronov PA, Fontes K, Ramos K, et al. (2008) Regulation of ceramide biosynthesis by TOR complex 2. Cell metabolism 7: 148–158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Odabasi Z, Paetznick V, Rex JH, Ostrosky-Zeichner L (2007) Effects of serum on in vitro susceptibility testing of echinocandins. Antimicrob Agents Chemother 51: 4214–4216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Yang YL, Lin YH, Tsao MY, Chen CG, Shih HI, et al. (2006) Serum repressing efflux pump CDR1 in Candida albicans. BMC Mol Biol 7: 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Nakayama H, Tanabe K, Bard M, Hodgson W, Wu S, et al. (2007) The Candida glabrata putative sterol transporter gene CgAUS1 protects cells against azoles in the presence of serum. J Antimicrob Chemother 60: 1264–1272. [DOI] [PubMed] [Google Scholar]
- 46. Paderu P, Garcia-Effron G, Balashov S, Delmas G, Park S, et al. (2007) Serum differentially alters the antifungal properties of echinocandin drugs. Antimicrob Agents Chemother 51: 2253–2256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Edlind T, Smith L, Henry K, Katiyar S, Nickels J (2002) Antifungal activity in Saccharomyces cerevisiae is modulated by calcium signalling. Mol Microbiol 46: 257–268. [DOI] [PubMed] [Google Scholar]
- 48. Withee JL, Mulholland J, Jeng R, Cyert MS (1997) An essential role of the yeast pheromone-induced Ca2+ signal is to activate calcineurin. Mol Biol Cell 8: 263–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Galgiani JM, Standards NCfCL (1997) Reference method for broth dilution antifungal susceptibility testing yeast: approved standard: National Committee for Clinical Laboratory Standards.
- 50. Kawaai H, Seino H, Yamazaki S, Taki K (2009) Changes in leukocyte migration during carbonic anhydrase activity inhibition. Burns 35: 397–404. [DOI] [PubMed] [Google Scholar]
- 51. Rex JH, Pfaller MA, Galgiani JN, Bartlett MS, Espinel-Ingroff A, et al. (1997) Development of interpretive breakpoints for antifungal susceptibility testing: conceptual framework and analysis of in vitro-in vivo correlation data for fluconazole, itraconazole, and Candida infections. Clin Infect Dis 24: 235–247. [DOI] [PubMed] [Google Scholar]
- 52. White TC, Marr KA, Bowden RA (1998) Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev 11: 382–402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Sanglard D, Ischer F, Monod M, Bille J (1996) Susceptibilities of Candida albicans multidrug transporter mutants to various antifungal agents and other metabolic inhibitors. Antimicrob Agents Chemother 40: 2300–2305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Feng Q, Summers E, Guo B, Fink G (1999) Ras Signaling Is Required for Serum-Induced Hyphal Differentiation in Candida albicans. J Bacteriol 181: 6339–63469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Negredo A, Monteoliva L, Gil C, Pla J, Nombela C (1997) Cloning, analysis and one-step disruption of the ARG5,6 gene of Candida albicans. Microbiology 143 (Pt 2): 297–302. [DOI] [PubMed] [Google Scholar]
- 56. Fonzi WA, Irwin MY (1993) Isogenic strain construction and gene mapping in Candida albicans. Genetics 134: 717–728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Jia X, Ma Z, Jia Y, Gao P, Zhang J, et al. (2008) RTA2, a novel gene involved in azole resistance in Candida albicans. Biochem Biophys Res Commun 373: 631–636. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Schematic representation of disruption of RTA2 using the URA3 selection marker. (A) Sequential targeted disruption of the two RTA2 alleles in C. albicans with the disruption cassette. (B) Southern analysis of the genomic DNA digested with BglII and SalI. The exact size and genotype of the expected hybridizing DNA fragment are indicated on the right.
(TIF)
(A) Strains, with one allele of RTA2 reintroduced into ADE2 locus, yielded only one 3.8 kb PCR product by PCR analysis with primer specific to ADE2 and primer specific to RTA2, with plasmid pBes-RTA2 as control. (B) Strains, with one allele of CNA or CRZ1 reintroduced into ADE2 locus, yielded only one 2.5 kb PCR product by PCR analysis with primer specific to ADE2 and primer specific to URA3, with plasmid pBes116 as control. (C) Strains, with the fusion fragment of CDR2 promoter and ORF of RTA2 reintroduced into ADE2 locus, yielded only one 3.9 kb PCR product by PCR analysis with primer specific to ADE2 and primer specific to RTA2, with plasmid pBesCDR2-RTA2 as control.
(TIF)
(A) Expression levels of UTR2 were examined by quantitative RT-PCR in the wild-type strain (CAF2-1), DJYCNA (rta2Δ/Δ cnaΔ/Δ::CNA) and MJYCRZ1 (rta2Δ/Δ crz1Δ/Δ::CRZ1) after exposure to CaCl2 (200 mM) for 2 h, with their corresponding drug-free strains as controls. (B) Expression levels of RTA2 were examined by quantitative RT-PCR in the wild-type strain (CAF2-1), DJYRTA2 (cnaΔ/Δ rta2Δ/Δ::RTA2) and MJYRTA2 (crz1Δ/Δ rta2Δ/Δ::RTA2) after exposure to CaCl2 (1 mM) for 16 h, with their corresponding drug-free strains as controls. (C) Expression levels of RTA2 were examined by quantitative RT-PCR in the parental strain (DSJ101U), DSJ-RTA2 carrying the fusion of the oestradiol-inducible CDR2 promoter and ORF of RTA2, in the presence of oestradiol (OST, 10 ug/ml), with their corresponding drug-free strains as controls. Data are represented as means ± standard deviation.
(TIF)
Schematic view of the calcineurin pathway in Candida albicans.
(TIF)
Primers used in this study.
(DOCX)