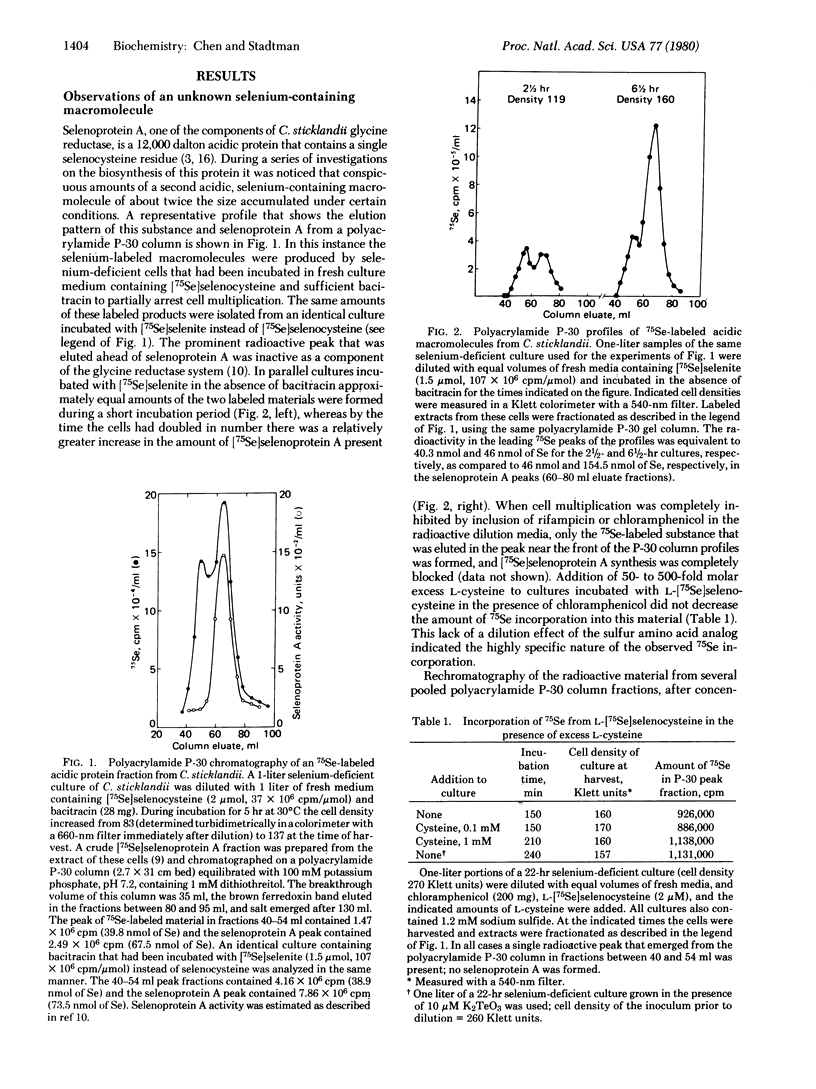
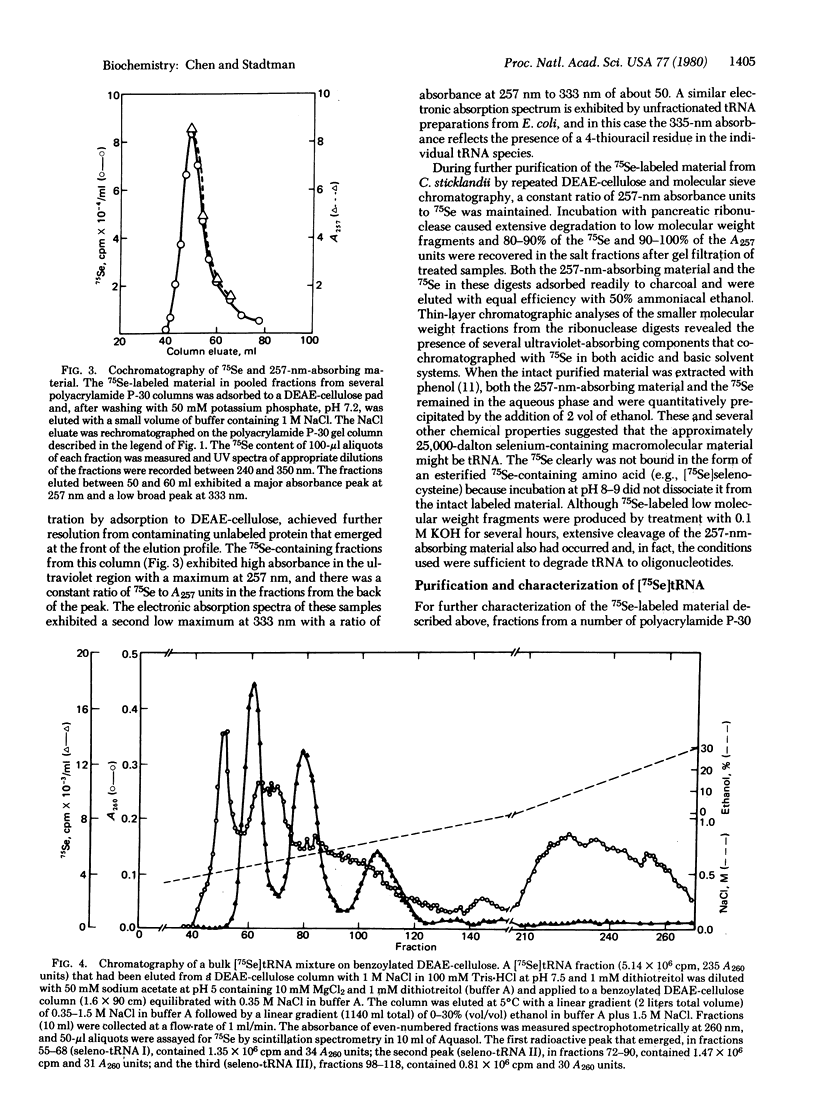
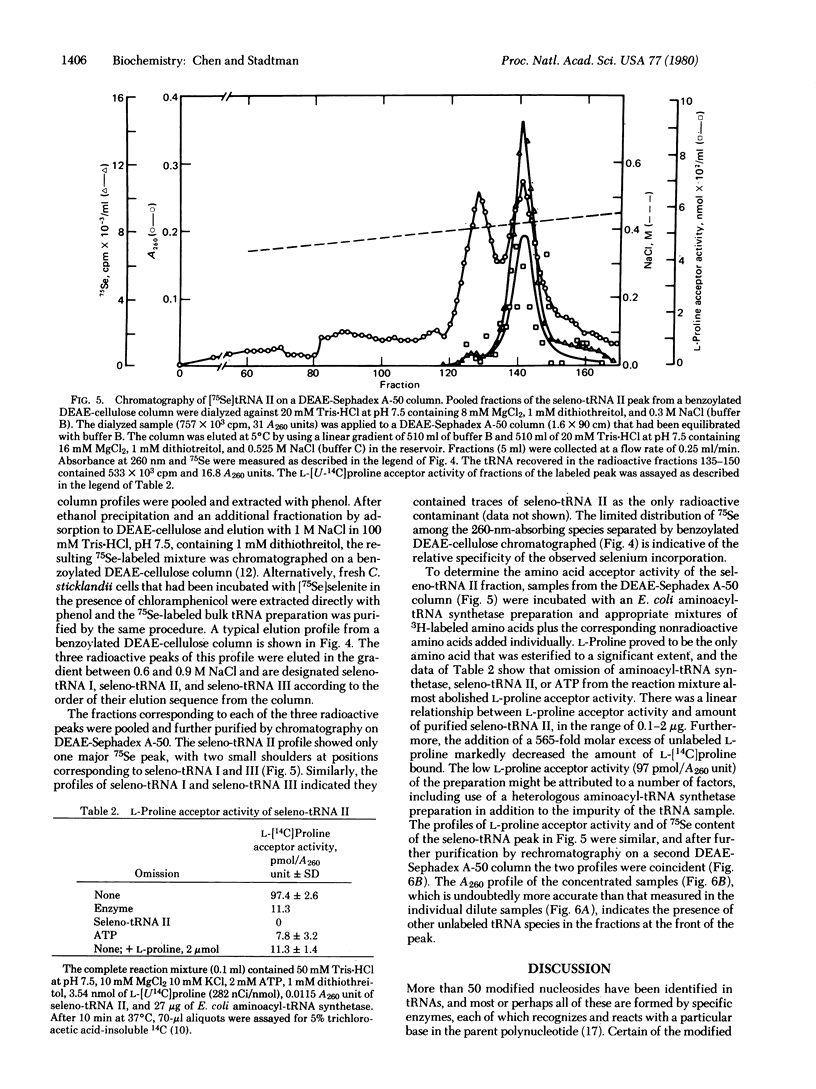
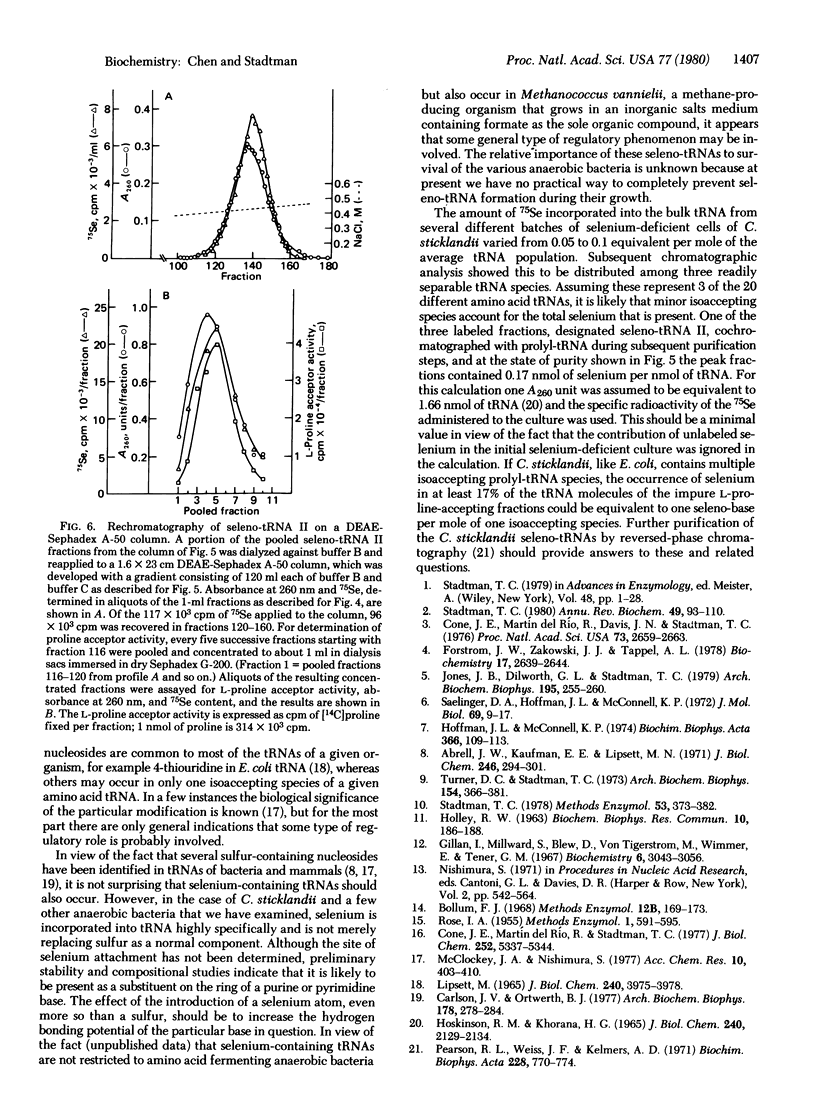
Abstract
75Se-Labeled tRNAs were synthesized by Clostridium sticklandii cultures supplemented with 1 microM sodium [75Se]selenite or [75Se]selenocysteine. This process is highly specific for selenium; it occurred in the presence of 1.2 mM sodium sulfide and was not decreased by the further addition of a 500-fold molar excess of cysteine. The 75Se in these tRNAs was located in the polynucleotide portion of the molecules and not in esterified (alkali-labile) selenocysteine. Inhibition of cell multiplication by antibiotics that block either protein synthesis or DNA-dependent RNA synthesis did not prevent this 75Se incorporation. Three [75Se]tRNAs were separated from C. sticklandii cells labeled in the presence of chloramphenicol and were partially purified by chromatography on benzoylated DEAE-cellulose and DEAE-Sephadex A-50 columns. These were designated seleno-tRNAs I, II, and III according to their elution sequence from benzoylated DEAE-cellulose. Cochromatography of purified seleno-tRNA II on DEAE-Sephadex A-50 with an L-proline-accepting species suggests that it is a selenium-containing L-prolyl-tRNA.
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrell J. W., Kaufman E. E., Lipsett M. N. The biosynthesis of 4-thiouridylate. Separation and purification of two enzymes in the transfer ribonucleic acid-sulfurtransferase system. J Biol Chem. 1971 Jan 25;246(2):294–301. [PubMed] [Google Scholar]
- Cone J. E., Del Río R. M., Davis J. N., Stadtman T. C. Chemical characterization of the selenoprotein component of clostridial glycine reductase: identification of selenocysteine as the organoselenium moiety. Proc Natl Acad Sci U S A. 1976 Aug;73(8):2659–2663. doi: 10.1073/pnas.73.8.2659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cone J. E., del Río R. M., Stadtman T. C. Clostridial glycine reductase complex. Purification and characterization of the selenoprotein component. J Biol Chem. 1977 Aug 10;252(15):5337–5344. [PubMed] [Google Scholar]
- Forstrom J. W., Zakowski J. J., Tappel A. L. Identification of the catalytic site of rat liver glutathione peroxidase as selenocysteine. Biochemistry. 1978 Jun 27;17(13):2639–2644. doi: 10.1021/bi00606a028. [DOI] [PubMed] [Google Scholar]
- Gillam I., Millward S., Blew D., von Tigerstrom M., Wimmer E., Tener G. M. The separation of soluble ribonucleic acids on benzoylated diethylaminoethylcellulose. Biochemistry. 1967 Oct;6(10):3043–3056. doi: 10.1021/bi00862a011. [DOI] [PubMed] [Google Scholar]
- HOLLEY R. W. Large-scale preparation of yeast "soluble" ribonucleic acid. Biochem Biophys Res Commun. 1963 Jan 31;10:186–188. doi: 10.1016/0006-291x(63)90048-2. [DOI] [PubMed] [Google Scholar]
- HOSKINSON R. M., KHORANA H. G. STUDIES ON POLYNUCLEOTIDES. XLI. PURIFICATION OF PHENYLALANINE-SPECIFIC TRANSFER RIBONUCLEIC ACID FROM YEAST BY COUNTERCURRENT DISTRIBUTION. J Biol Chem. 1965 May;240:2129–2134. [PubMed] [Google Scholar]
- Hoffman J. L., McConnell K. P. The presence of 4-selenouridine in Escherichia coli tRNA. Biochim Biophys Acta. 1974 Sep 27;366(1):109–113. doi: 10.1016/0005-2787(74)90323-2. [DOI] [PubMed] [Google Scholar]
- Jones J. B., Dilworth G. L., Stadtman T. C. Occurrence of selenocysteine in the selenium-dependent formate dehydrogenase of Methanococcus vannielii. Arch Biochem Biophys. 1979 Jul;195(2):255–260. doi: 10.1016/0003-9861(79)90351-5. [DOI] [PubMed] [Google Scholar]
- Lipsett M. N. The isolation of 4-thiouridylic acid from the soluble ribonucleic acid of Escherichia coli. J Biol Chem. 1965 Oct;240(10):3975–3978. [PubMed] [Google Scholar]
- Ortwerth B. J., Carlson J. V. Lysine transfer RNA from liver: a sulfur-containing species that codes for AAG. Arch Biochem Biophys. 1977 Jan 15;178(1):278–284. doi: 10.1016/0003-9861(77)90192-8. [DOI] [PubMed] [Google Scholar]
- Pearson R. L., Weiss J. F., Kelmers A. D. Improved separation of transfer RNA's on polychlorotrifuoroethylene-supported reversed-phase chromatography columns. Biochim Biophys Acta. 1971 Feb 11;228(3):770–774. doi: 10.1016/0005-2787(71)90748-9. [DOI] [PubMed] [Google Scholar]
- Saelinger D. A., Hoffman J. L., McConnell K. P. Biosynthesis of selenobases in transfer RNA by Escherichia coli. J Mol Biol. 1972 Aug 14;69(1):9–17. doi: 10.1016/0022-2836(72)90020-4. [DOI] [PubMed] [Google Scholar]
- Stadtman T. C. Selenium-dependent clostridial glycine reductase. Methods Enzymol. 1978;53:373–382. doi: 10.1016/s0076-6879(78)53043-7. [DOI] [PubMed] [Google Scholar]
- Stadtman T. C. Selenium-dependent enzymes. Annu Rev Biochem. 1980;49:93–110. doi: 10.1146/annurev.bi.49.070180.000521. [DOI] [PubMed] [Google Scholar]
- Turner D. C., Stadtman T. C. Purification of protein components of the clostridial glycine reductase system and characterization of protein A as a selenoprotein. Arch Biochem Biophys. 1973 Jan;154(1):366–381. doi: 10.1016/0003-9861(73)90069-6. [DOI] [PubMed] [Google Scholar]


