Abstract
Enhanced Cl− efflux during acidosis in plants is thought to play a role in cytosolic pH (pHc) homeostasis by short-circuiting the current produced by the electrogenic H+ pump, thereby facilitating enhanced H+ efflux from the cytosol. Using an intracellular perfusion technique, which enables experimental control of medium composition at the cytosolic surface of the plasma membrane of charophyte algae (Chara corallina), we show that lowered pHc activates Cl− efflux via two mechanisms. The first is a direct effect of pHc on Cl− efflux; the second mechanism comprises a pHc-induced increase in affinity for cytosolic free Ca2+ ([Ca2+]c), which also activates Cl− efflux. Cl− efflux was controlled by phosphorylation/dephosphorylation events, which override the responses to both pHc and [Ca2+]c. Whereas phosphorylation (perfusion with the catalytic subunit of protein kinase A in the presence of ATP) resulted in a complete inhibition of Cl− efflux, dephosphorylation (perfusion with alkaline phosphatase) arrested Cl− efflux at 60% of the maximal level in a manner that was both pHc and [Ca2+]c independent. These findings imply that plasma membrane anion channels play a central role in pHc regulation in plants, in addition to their established roles in turgor/volume regulation and signal transduction.
Plasma membrane anion channels play fundamental roles in several areas of plant cell biology (Tyerman, 1992; Hedrich, 1994; Schroeder, 1995). Salt loss from guard cells during stomatal closure and from euryhaline algae responding to hypoosmotic stress is effected principally by the opening of anion channels (Okazaki and Tazawa, 1990; Okazaki and Iwasaki, 1992; Schroeder et al., 1993; Schwartz et al., 1995). Thus, because the equilibrium potential of permeant anions is positive of 0 mV, the opening of anion channels depolarizes the membrane and brings the membrane potential into a range in which outward-rectifying K+ channels open to provide accompanying cation loss.
A second important function of anion channels is likely to relate to the early stages of signal transduction. A wide range of stimuli, including fungal elicitors (Nürnberger et al., 1994), red light (Ermolayeva et al., 1997), blue light (Cho and Spalding, 1996), and Nod factors (Ehrhardt et al., 1992), evoke a rapid depolarization of the plasma membrane, which, based on the measurements of fluxes, ionic currents, and inhibitor sensitivity, indicates the opening of anion channels. Ward et al. (1995) have proposed that one function of these anion-channel-evoked depolarizations might be to open voltage-dependent Ca2+ channels, which, by allowing entry of external Ca2+, would elevate [Ca2+]c. As befits these important roles in cell biology, the activities of anion channels are tightly regulated by voltage (Keller et al., 1989; Okihara et al., 1991; Schroeder and Keller, 1992), [Ca2+]c (Shiina and Tazawa 1987, 1988; Schroeder and Hagiwara, 1989; Hedrich et al., 1990; Okihara et al., 1991), nucleotides (Hedrich et al., 1990; Schulz-Lessdorf et al., 1996; Thomine et al., 1997), and phosphorylation/dephosphorylation cascades (Zimmermann et al., 1994; Schmidt et al., 1995; Pei et al., 1997).
A further function for anion channels in the control of pHc is hinted at by the observation that cytosolic acidification with weak acids leads to an enhancement of cellular Cl− efflux in charophyte algae (Sanders, 1980a; Smith and Reid, 1991) and higher plants (Beffagna et al., 1995). Smith and Reid (1991) proposed that the activation of anion channels in these conditions would provide a shunt pathway for charge compensation of H+ moved electrogenically out of the cell by the activated H+-ATPase. The failure to provide such a shunt pathway would severely compromise the ability of the pump to effect control of pHc because activation of the pump by cytosolic H+ would be countered by membrane hyperpolarization (Blatt et al., 1990). The membrane depolarizations and conductance increases reported for fungi and lower plants in conditions of cytosolic acidification might also have their origins in the opening of anion channels, with similar implications for pHc regulation (Sanders et al., 1981; Frachisse et al., 1988; Roberts et al., 1997).
A pivotal question relating to the role of anion channels in responding to pHc concerns the mechanism of action of cytosolic H+. Does H+ directly activate anion efflux, or are the effects mediated by one of the other well-established regulators of anion channels? In the present study we have addressed this question using internodal cells of charophyte algae. These large and robust cells can be internally perfused, thereby permitting experimental control of medium composition at the inner surface of the plasma membrane in a manner analogous to the whole-cell patch-clamp technique. The results reveal not only that cytosolic H+ activates anion efflux directly, but also that indirect effects can prevail, which are mediated through a pH-induced decrease in KCa. Furthermore, a profound influence of phosphorylation state on anion efflux has been discovered that might underlie the refractory period that follows electrical excitation of these cells.
MATERIALS AND METHODS
Plant Material
Chara corallina was cultured at 16°C in 60-L tanks that were illuminated with blue-enriched light (one 15-W Gro-lux [Sylvania] and one 15-W warm-white fluorescent tube). Internodal cells that showed no pronounced calcification, with diameters between 0.6 and 1 mm, and lengths between 5 and 8 cm, were harvested as described by Sanders (1980b).
Media
The composition of APW used as an external medium in perfusion experiments was as follows: 0.5 mm CaSO4, 0.2 mm K2SO4, 0.5 mm Na2SO4, 0.5 mm Hepes-NaOH, pH 7.5, and 200 mm sorbitol. Final free K+ concentration measured with an ion-selective electrode was 0.38 mm. Perfusion media for tonoplast-free perfusion were based on media described by Sanders (1980c) and Williamson (1975) and contained the following: either 5 mm EGTA (used with [Ca2+]c = 0.001–1.0 μm), 5 mm HEDTA (for [Ca2+]c = 1.0–10 μm), or 5 mm NTA (for [Ca2+]c = 10–100 μm); 0.5 mm MgSO4; 300 mm sorbitol, 0.5 mm Na36Cl (specific activity, 97 kBq mL−1); or 50 mm Hepes plus KOH. Final free [K+] ranged from 20 mm at pH 7.8 to 11 mm at pH 6.8. Final free Ca2+ concentrations were calculated using iterative software (SOLCON, D.C.S. White, University of York, UK, and Y.E. Goldman, University of Pennsylvania, Philadelphia). The dissociation constants (25°C) for the Ca2+ buffers were as follows (Martell and Smith, 1974, 1982; Smith and Martell, 1976):
[H+][EGTA]/[H·EGTA] = 3.39 × 10−10 m; [H+]2[EGTA]/[H2·EGTA] = 4.57 × 10−19 m2; [H+]3[EGTA]/[H3·EGTA] = 1.05 × 10−21 m3; [H+]4[EGTA]/[H4·EGTA] = 1.05 × 10−23 m4; [Mg2+][EGTA]/[Mg·EGTA] = 6.18 × 10−6 m; [Mg2+][H+] [EGTA]/[Mg·H·EGTA] = 1.20 × 10−13 m2; [Ca2+][EGTA]/[Ca·EGTA] = 1.07 × 10−11 m; [Ca2+][H+][EGTA]/[Ca·H·EGTA] = 1.74 × 10−15 m2; [H+][HEDTA]/[H·HEDTA] = 3.39 × 10−10 m; [H+]2[HEDTA]/[H2·HEDTA] = 4.57 × 10−19 m2; [H+]3[HEDTA]/[H3·HEDTA] = 1.05 × 10−21 m3; [H+]4[HEDTA]/[H4·HEDTA] = 1.05 × 10−23 m4; [Mg2+][HEDTA]/[Mg·HEDTA] = 6.18 × 10−6 m; [Mg2+][H+][HEDTA]/[Mg·H·HEDTA] = 1.20 × 10−13 m2; [Ca2+][HEDTA]/[Ca·HEDTA] = 1.07 × 10−11 m; [Ca2+][H+][HEDTA]/[Ca·H·HEDTA] = 1.74 × 10−15 m2; [H+][NTA]/[H·NTA] = 2.24 × 10−10 m; [H+]2[NTA]/[H2·NTA] = 7.41 × 10−13 m2; [H+]3[NTA]/[H3·NTA] = 1.175 × 10−14 m3; [H+]4[NTA]/[H4·NTA] = 1.86 × 10−15 m4; [Mg2+][NTA]/[Mg·NTA] = 3.39 × 10−6 m; [Ca2+][NTA]/[Ca·NTA] = 3.63 × 10−7 m; [Ca2+][NTA]2/[Ca·NTA2] = 1.55 × 10−9 m2.
For phosphorylation experiments 1 μm PKA catalytic subunit (isolated from bovine heart) and 3 mm BTP-ATP were added to the perfusion medium. PKA was reconstituted in deionized water containing 6 mg/mL DTT and was used the same day or the day after. BTP-ATP was prepared by adding 1 g of Dowex 50-X8 cation-exchange resin to 50 mL of a 30 mm solution of Na2ATP. After stirring, filtration, and addition of BTP, the resulting solution was stored at −20°C. Alkaline phosphatase (from bovine intestinal mucosa, prepared in deionized water), ATP-γ-S, and anion channel antagonists were added to the perfusion medium as indicated.
When tested externally, anion-channel antagonists were added to APW and introduced into the central chamber of the perfusion apparatus (see below) after the 2-min recovery period. The cell was then left for another 10 min before addition of fresh medium for monitoring efflux. The inhibitors IAA-94/95, ISA-94/95, NPPB, and NPPB-S were made up in 0.5% (v/v) DMSO, which on its own had no discernible effect. NPPB, NPPB-S, IAA-94/95, and ISA-94/95 were a kind gift of Dr. A. Pope (SmithKline Beecham Pharmaceuticals, Welwyn, Hertfordshire, UK). Na36Cl was obtained from Amersham, and all other chemicals were obtained from Sigma.
Intracellular Perfusion and Removal of Vacuolar Membrane and Streaming Cytoplasm
Intracellular perfusion of the internodal cells was performed as described by Sanders (1980b) and Tazawa et al. (1976). Internodal cells of the required dimension were cut from neighboring cells the day before an experiment and kept under white light overnight. The cell was blotted before perfusion, loosely tied with six ligatures of waxed dental floss or silk sutures (Ethicon, Somerville, NJ), and placed on the perfusion platform. Three chambers, two for perfusion medium at either end and one for APW in the center, were placed on top and firmly held in position by vacuum grease, which was also applied beneath the cell and at its contact with the chamber edges to prevent leakage. Turgor reduction in air was aided by placing a few drops of perfusion medium on each cell end. When the cell had wilted very slightly, perfusion medium was introduced to the left-hand chamber and both ends of the cell were removed. Through the constant removal of perfusion medium from the right-hand chamber and refilling of the left-hand chamber a pressure difference of 6 mm of water was maintained across the ends of the cell, resulting in a flow rate of 4 to 8 mm s−1. The resulting shear forces caused the removal of the vacuolar membrane and streaming cytoplasm, which was completed within 2 min of the start of the perfusion. Then the two outermost ligatures were tied and APW was introduced into the central chamber. After 2 min of recovery, the remaining four ligatures were tied and the APW in the central chamber was replaced. Five minutes later, APW was removed from the central chamber and, providing that the cells appeared healthy, transferred to a scintillation vial. Cells exhibiting disruption in chloroplast structure at this stage were discarded. Typically, such cells amounted to around 10% of those perfused. Efflux of 36Cl− from the cell to the APW was measured in a scintillation counter (model 1212 Minibeta, Amersham-Pharmacia Biotech). Fluxes are expressed as picomoles per square centimeter per second. Preliminary experiments indicated that Cl− efflux was invariant with time for at least 15 min, providing that cells appeared healthy, with nondisrupted chloroplasts. The area of cell in the central chamber was approximately 0.5 cm2.
Membrane Potential Measurements
The perfusion platform was securely clamped and a binocular microscope was set up horizontally to focus onto the central chamber. Perfused cells were impaled after the 2-min recovery period with a glass microelectrode filled with 0.1 m KCl and connected with a Ag/AgCl half-cell to a high-impedance amplifier (FD-223, World Precision Instruments, Hamden, CT). The circuit within the chamber was completed by a KCl-filled agar bridge. Micromanipulation was performed with a Huxley-Goodfellow micromanipulator (Goodfellow Metals, Cambridge, UK). Signals were recorded on a chart recorder for at least 7 min, during which a stable membrane potential was reached after the first 1 to 2 min. Results are expressed as means ± se for between three and five determinations for each condition.
RESULTS
To determine whether reverse action of the previously described Cl−-H+ symporter (Sanders, 1980c) contributes to efflux of radiolabeled Cl−, the effect of external Cl− in the range 0 to 5 mm was tested. For [Ca2+]c = 5 μm and pHc = 7.8, negligible variation in Cl− efflux was found (mean value of approximately 40 pmol cm−2 s−1) at all Cl− concentrations tested (data not shown). The absence of self-exchange of Cl− supports the conclusion that the Cl−-H+ symporter is not effectively reversible (Sanders and Hansen, 1981). It can be argued, therefore, that the observed Cl− efflux through the plasma membrane of C. corallina cells is mediated by anion channels.
To test the possibility that variation in the membrane potential might contribute to modulation of Cl− efflux in the experimental conditions of the present study, membrane potential measurements were performed on perfused cells. At the extremes of both pHc and [Ca2+]c (i.e. at pHc values of 6.8 and 7.8 and [Ca2+]c values of 0.01 and 100 μm), variations in the membrane potential were not very pronounced. For example, at low [Ca2+]c (0.01 μm), the membrane potential was not significantly different over the pH range tested (−83 ± 5 mV at pH 6.8; −85 ± 10 mV at pH 7.8). At high [Ca2+]c (100 μm), the membrane potential was generally about 20 mV more negative (e.g. −103 ± 8 mV at pH 7.8) but still fell within a range in which channel activity would be expected to be mainly unchanged, since Ca2+-dependent anion channels in C. corallina show maximum activity in the range between −80 and −100 mV (Okihara et al., 1991).
Direct Effect of pHc
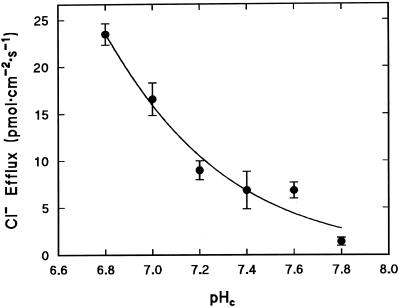
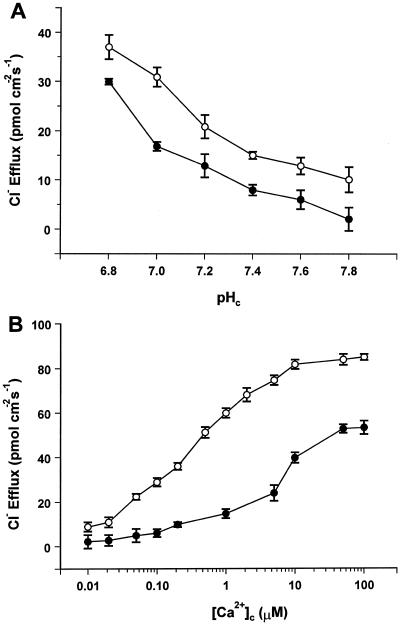
Direct involvement of pHc in regulation of Cl− efflux was tested by perfusing the cells with Ca2+-free media buffered over a range of physiologically relevant pH values (resting conditions, pH 7.8; acidosis, pH 6.8) (Reid et al., 1989). Figure 1 shows that efflux of 36Cl− was enhanced as a first-order function of intracellular H+ as the pH was lowered over the range of 7.8 (1.4 pmol cm−2 s−1) to 6.8 (24 pmol cm−2 s−1). The effect of pHc on Cl− efflux can be adequately described by the binding of a single H+ with a pK of 6.1 ± 0.5, which could indicate the involvement of the imidazole ring of His (pKa = 6.2).
Figure 1.
Direct effect of pHc on Cl− efflux. Cl− efflux was measured in the absence of Ca2+ in the perfusion medium. Each point is the mean of three replicates. The fitted curve describes the relationship JCl = Jmax/(1 + 10(pH-pK)), where JCl is the Cl− efflux, Jmax is the Cl− efflux at the optimum pH (fitted as 132 pmol cm−2 s−1), and pK = 6.1 ± 0.5.
In intact cells, Cl− efflux increases from <1 to 16 pmol cm−2 s−1 when pH is decreased from 7.5 to 6.8 by the uptake of butyric acid (Smith and Reid, 1991), compared with 4 to 24 pmol cm−2 s−1 in perfused cells over the same pH range. These values are in reasonable agreement given the known variability of different C. corallina cultures.
pHc Modulates Activation by [Ca2+]c
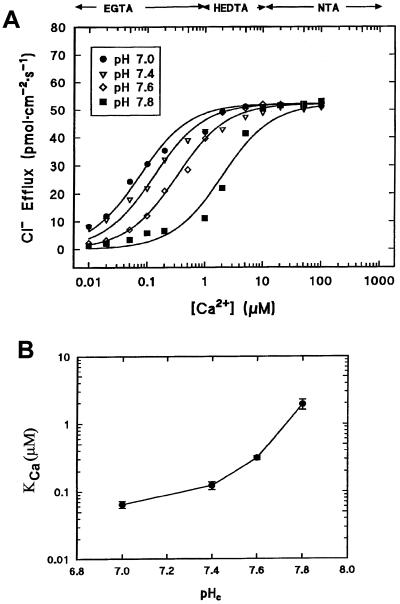
To test for interaction between H+ and Ca2+ in activation of Cl− efflux, cells were perfused in media of defined Ca2+ activities at fixed pH values in the range of 7.0 to 7.8. The Ca2+ activities were selected to embrace the range of [Ca2+]c that occurs physiologically, i.e. 0.2 μm in resting cells to 6 μm during the action potential (Williamson and Ashley, 1982).
Figure 2A shows that at the resting pH of 7.8, Cl− efflux increased as a function of [Ca2+]c, as shown previously by Shiina and Tazawa (1988). It is noteworthy that a large portion (70%) of the Cl− efflux was activated by an increase in [Ca2+]c over the physiological range of 0.2 to 6.0 μm. As pH was progressively decreased from 7.8 to 7.0, two important things occurred: First, Cl− efflux saturated to a common pH-independent value of about 53 pmol cm−2 s−1 at maximum permissive levels of free Ca2+ despite the fact that the flux in the absence of Ca2+ was pH sensitive (Fig. 1). Second, KCa declined as pH was decreased (Fig. 2B).
Figure 2.
Modulation of Ca2+-dependent Cl− efflux by pHc. A, Cl− efflux as a function of [Ca2+]c over the physiological pH range. Each point is the mean of three replicates: se values, omitted for clarity, were between 5% and 10%. Data were fitted by Equation 3 with S = 0.95, α = 55, KH = 7.94 × 10−7, and variable Kca values. B, Affinity of Ca2+ binding (KCa) as a function of pHc. Values were derived from the fits shown in A.
The data can be described by a function based on a model that assumes competition between H+ and Ca2+ for a regulatory binding site according to the scheme:
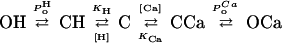 |
where C and O represent the closed and open states of the channel, respectively, Po denotes the open probability, and KCa and KH are the affinities of the binding site for Ca2+ and H+, respectively. The Cl− flux JCl can be described as:
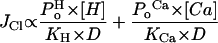 |
1 |
with
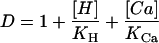 |
2 |
Let 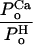 = α and S be a scaling factor (which includes
PoH). Then
= α and S be a scaling factor (which includes
PoH). Then
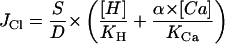 |
3 |
The data are well described by Equation 3 when pH dependence is incorporated into the KCa component, as shown in Figure 2B. At low [Ca2+]c, channel opening is pH dependent, but this component becomes insignificant at high [Ca2+]c, where Cl− efflux saturates at a common value independent of pHc. The affinity for Ca2+ binding is dramatically enhanced when cytosolic [H+] increases. The pH-dependent increase in Ca2+ affinity was strongest for small decreases in pHc in the vicinity of the resting pH of 7.8 and became progressively less pronounced for stronger cytosolic acidification.
Effect of Phosphorylation/Dephosphorylation and Nucleotides
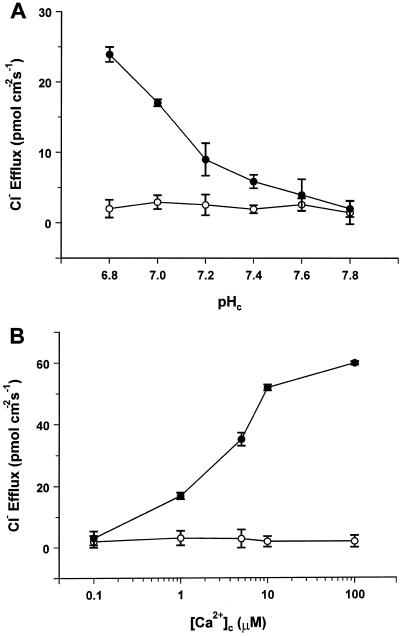
The effect of phosphorylation on Cl− efflux was tested by addition of 1 μm PKA catalytic subunit (Reinhart et al., 1991) and 3 mm ATP in the perfusion medium. Figure 3A shows that in the absence of Ca2+, internal perfusion with PKA and ATP resulted in a dramatic inhibition of Cl− efflux over the whole pHc range tested (7.8–6.8). The Ca2+-responsive Cl− efflux was also inhibited in these conditions. Figure 3B demonstrates that when cells were perfused with defined [Ca2+]c (0.1–100 μm) at pHc = 7.8 in the presence of PKA/ATP, Cl− efflux was barely detectable throughout the [Ca2+]c range tested. This shows that phosphorylation completely overrides the opening effects of H+ and Ca2+ on the Cl− efflux pathway. It should be noted that the inclusion of ATP alone in the perfusion medium stimulates rather than inhibits Cl− efflux, as demonstrated previously by Shiina and Tazawa (1988).
Figure 3.
Inhibition of Cl− efflux in phosphorylating conditions. A, pH dependence of Cl− efflux in Ca2+-free medium. B, Ca2+ dependence of Cl− efflux at pHc = 7.8. •, Control; ○, plus 1 μm PKA catalytic subunit and 3 mm ATP. Each point is the mean ± se of three independent replicates.
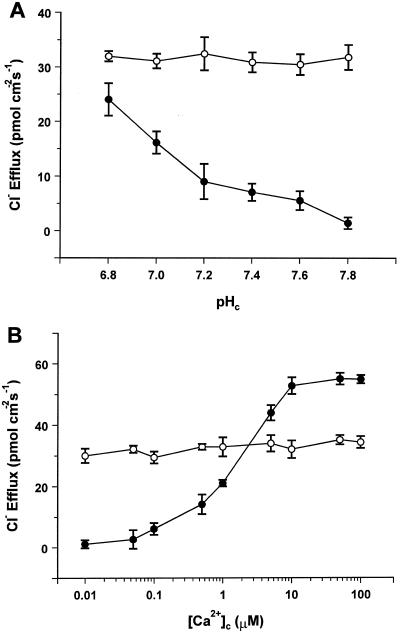
The effect of dephosphorylation was tested by inclusion of 30 nm alkaline phosphatase in the perfusion medium. When tested in the absence of Ca2+ in the pH range of 7.8 to 6.8, the phosphatase caused a pH-independent increase in Cl− efflux to levels of 30 to 35 pmol cm−2 s−1 (Fig. 4A). Figure 4B shows that in dephosphorylating conditions Cl− efflux is insensitive to [Ca2+]c. In the presence of defined [Ca2+]c (0.1–100 μm) tested at pHc = 7.8, addition of the phosphatase to the perfusion medium reduced Cl− efflux to the same level as in the absence of internal Ca2+, which was about 60% of the maximal level obtained in the control with optimal [Ca2+]c. Thus, depending on the [Ca2+]c level, dephosphorylating conditions can either stimulate ([Ca2+]c < 2 μm) or inhibit ([Ca2+]c > 2 μm) Cl− efflux.
Figure 4.
Effect of dephosphorylating conditions on Cl− efflux. A, pH dependence of Cl− efflux in Ca2+-free medium. B, Ca2+ dependence of Cl− efflux at pHc = 7.8. •, Control; ○, plus 30 nm nonspecific alkaline phosphatase. Each point is the mean ± se of three independent replicates.
Plant anion channels have repeatedly been shown to be subject to regulation by nucleotides (Hedrich et al., 1990; Schulz-Lessdorf et al., 1996; Thomine et al., 1997). This possibility was tested for the Cl− efflux pathway in C. corallina using the nonhydrolyzable analog ATP-γ-S, which was included in the perfusion medium in preference to ATP to eliminate possible inhibition by endogenous kinases. Figure 5A illustrates that in the absence of Ca2+, addition of ATP-γ-S elevated Cl− efflux by a constant value of 11.0 ± 0.6 pmol cm−2 s−1 over the whole pH range tested (7.8–6.8). The effect of ATP-γ-S on Cl− efflux in the presence of [Ca2+]c over the range of 0.1 to 100 μm (at a constant pHc of 7.8) is shown in Figure 5B. The presence of ATP-γ-S enhanced the sensitivity of Cl− efflux to [Ca2+]c such that saturation of the flux, which occurred at a 1.5-fold higher level than in the control, was achieved at a [Ca2+]c of 10 μm compared with 50 μm in the control.
Figure 5.
Stimulation of Cl− efflux by ATP-γ-S. A, pH dependence of Cl− efflux in Ca2+-free medium. B, Ca2+ dependence of Cl− efflux at pHc = 7.8. •, Control; ○, plus 3 mm ATP-γ-S. Each point is the mean ± se of three independent replicates.
Pharmacology
Anion-channel blockers were applied to the external and the internal face of the plasma membrane and were tested in conditions in which maximum Cl− efflux occurs (pHc = 6.8 and [Ca2+]c = 100 μm). Table I summarizes the effects of various anion-channel antagonists on Cl− efflux. All antagonists tested (except ethacrynic acid) were much more effective when applied internally. Most potent inhibition was achieved by internal application of IAA-94/95 (90% inhibition at 1 μm, 50% inhibition at 53 nm) followed by NPPB-S (80% inhibition at 10 μm), A-9-C (60% inhibition at 10 μm), NPPB (50% inhibition at 10 μm), ethacrynic acid (40% inhibition at 100 μm), and ISA-94/95 (33% inhibition at 10 μm).
Table I.
Effect of anion-channel blockers on Cl− efflux
| Inhibitor | Concentration | Internal Inhibition | External Inhibition |
|---|---|---|---|
| μm | % | ||
| IAA-94/95 | 1 | 90 ± 4 | Not determined |
| 10 | Leak | 4 ± 2 | |
| 100 | Not determined | 36 ± 2 | |
| ISA-94/95 | 1 | 8 ± 3 | Not determined |
| 10 | 33 ± 4 | 5 ± 2 | |
| 100 | Not determined | 20 ± 1 | |
| NPPB | 1 | 5 ± 3 | Not determined |
| 10 | 50 ± 2 | 8 ± 1 | |
| 100 | Not determined | 19 ± 2 | |
| NPPB-S | 1 | 80 ± 2 | Not determined |
| 10 | Leak | 5 ± 3 | |
| 100 | Not determined | 29 ± 3 | |
| A-9-C | 10 | 60 ± 2 | 10 ± 2 |
| 100 | 70 ± 2 | 20 ± 1 | |
| Ethacrynic acid | 10 | 12 ± 2 | 30 ± 2 |
| 100 | 40 ± 2 | 20 ± 3 | |
Anion-channel antagonists were either added to the perfusion medium (Internal Inhibition) or to APW (External Inhibition). When applied externally, the inhibitors were present for 10 min. Three replicates were performed for each condition. Data are means ± se.
DISCUSSION
Activation of Cl− Efflux by Cytosolic Ca2+ and H+
Perfused internodal cells of C. corallina constitute a powerful system for investigation of cytosolic control of solute fluxes across the plasma membrane. Not only can the composition of the internal solution be subject to experimental control, in addition, ionic fluxes can be measured unambiguously using radioisotopes. Cl− release measured radiometrically from C. corallina bears the characteristics of a channel-mediated flux. The unidirectional flux is insensitive to the external Cl− concentration and is activated by [Ca2+]c. Extensive investigations of the ionic basis of charophyte action potentials resulted in the conclusion that Ca2+-activated anion channels must reside at the plasma membrane (Beilby, 1984; Shiina and Tazawa 1987, 1988). These conclusions were supported by the characterization in planar lipid bilayers of a Ca2+-activated Cl− channel from charophytes (Lunevsky et al., 1983; Berestovsky et al., 1987) and by patch-clamp studies on the plasma membrane of C. corallina internodal cells, which demonstrated Ca2+-activated Cl− channels with a maximum open frequency in the range of −80 to −100 mV (Okihara et al., 1991). Furthermore, Ca2+-activated anion channels are widely distributed at the plasma membranes of higher plants (Tyerman, 1992; Ward et al., 1995).
In the present work the perfusion solutions were selected to render the membrane potential relatively insensitive to changes in pHc and [Ca2+]c by clamping it in the region of the K+ equilibrium potential (−85 to −100 mV in these experiments; see Sanders, 1980c). This ensured that the effects of changes in ligand composition could be interpreted without the complication of associated changes in membrane potential, to which anion channels are known to be markedly sensitive (Keller et al., 1989; Okihara et al., 1991; Schroeder and Keller, 1992).
The present results demonstrate that Cl− efflux from C. corallina is activated directly by cytosolic H+. Additionally, elevation in cytosolic H+ markedly decreased the Ca2+ activation constant for Cl− release. In resting conditions at pHc = 7.8 and [Ca2+]c = 200 nm, Cl− efflux is low. However, the Cl− efflux pathway is poised such that as either [Ca2+]c or [H+]c is elevated, a marked increase in Cl− efflux results. Physiologically, the H+-induced decrease in the Ca2+ activation constant is primarily responsible for this marked sensitivity to H+. Thus, the net effect is that the increase in cytosolic acidity sensitizes the Cl− release channels to activation by Ca2+.
Physiological Role of Activation by H+
Control of pHc by transport events (biophysical pH regulation; Smith and Raven, 1979) is achieved principally by activation of the H+-pumping ATPase. Nevertheless, simple substrate (H+)-induced activation of the enzyme in the absence of activation of other transport systems will have a negligible effect on the net rate of H+ removal from the cytosol, because the hyperpolarization resulting from activation of this electrogenic transport system will tend to offset any increase in activity. Thus, by providing a shunt conductance for the H+-ATPase, anion channels indirectly enhance H+ export from the cell. The current-voltage relationship for the C. corallina plasma membrane H+-ATPase exhibits considerable voltage sensitivity over the physiological range (Blatt et al., 1990), so it is possible that membrane depolarization resulting from activation of anion channels increases H+ pump activity to counteract cytosolic acidification.
Physiological Role of Activation by Ca2+
The potent sensitivity of charophyte plasma membrane anion channels to changes in [Ca2+]c has been reported previously in internally perfused cells (Shiina and Tazawa, 1987, 1988). In tonoplast-free cells of Nitellopsis obtusa, Shiina and Tazawa (1988) observed a massive increase in Cl− efflux upon elevation of Ca2+ in the perfusion medium. After increasing [Ca2+]c from 1 to 4 and 16 μm, they found an enhancement of Cl− efflux from 0.05 to 0.58 and 1.28 nmol cm−2 s−1 (measured at an internal pH of 7.0), whereas in our study Cl− efflux saturated at a lower rate of 0.053 nmol cm−2 s−1 and showed a higher affinity for Ca2+ (KCa = 64 ± 7 nm at pHc = 7.0). This difference in Cl− efflux and Ca2+ sensitivity could be the result of different experimental conditions, e.g. the exclusion of ATP in the perfusion medium used for the present study. Removal of ATP from the cytoplasmic medium has been reported to prevent excitability of tonoplast-free C. corallina cells (Lühring and Tazawa, 1985). Furthermore, in contrast to Shiina and Tazawa (1988), we did not observe the dramatic depolarization upon elevation of [Ca2+]c but rather a small hyperpolarization, which can also account for the differences in the Cl− flux.
Ca2+-sensitive anion channels that open upon depolarization have been characterized in excised plasma membrane patches of C. corallina (Okihara et al., 1991). Whether these channels have a role in the Ca2+- and pH-dependent Cl− efflux observed in the present report is difficult to judge, because the pH dependence and pharmacology have not been tested in the patch-clamp study. Simultaneous measurements of action potentials and patch currents (cell-attached mode) in the C. corallina plasma membrane recently revealed two additional types of Cl− conductances that are closely associated with excitability (Homann and Thiel, 1994). These channels were active only during the action potential and could not be directly gated open either by depolarizing voltage changes or by elevation of [Ca2+]c, nor could they be observed in excised patches (Thiel et al., 1993; Homann and Thiel, 1994). It has been argued, therefore, that these channels are mainly responsible for the rapid, massive Cl− fluxes during the charophyte action potential. Whether the pH- and Ca2+-sensitive Cl− efflux pathway under investigation in this study has a direct role in excitability is not clear, but the [Ca2+]c changes that occur during the action potential are sufficient to activate Cl− efflux through these channels.
Phosphorylation and the Refractory Period of the Charophyte Action Potential
Previous reports have stated, on the basis of electrophysiological data, that phosphorylation will either inhibit (Shiina and Tazawa, 1986; Shiina et al., 1988) or activate (Zherelova, 1989) plasma membrane Ca2+ channels from charophytes. The action potential of charophytes typically lasts for about 2 s and exhibits a refractory period of about 6 to 60 s (Beilby and Coster, 1979). The present results demonstrate an effective inhibition of Cl− efflux in phosphorylating conditions like those thought to prevail after elevation of [Ca2+]c during the action potential (Shiina et al., 1988). Our findings suggest that the refractory period that follows stimulation could result from inactivation of anion channels through phosphorylation.
Pharmacology of Cl− Efflux
The pharmacology of Cl− efflux can provide insight into the physiological role of the anion channels involved (Ward et al., 1995). High-affinity ligands such as IAA-94 also provide a tool for purifying Cl− channels and have been used successfully in animal cells (Landry et al., 1989). A range of anion-channel antagonists have been tested to provide additional indications that Cl− efflux is channel mediated, and also to identify high-affinity antagonists that can be used in future studies to further elucidate the physiological role of the channel and its single-channel properties.
Anion channel antagonists such as A-9-C and ethacrynic acid, which are known to block Ca2+-activated Cl− efflux and action potentials in charophytes (Shiina and Tazawa, 1987, 1988), inhibited Cl− efflux in the present study by 70% and 40% when applied to the cytoplasmic side. The most potent inhibitors of the pH and Ca2+-sensitive Cl− efflux in C. corallina, however, were the high-affinity ligands IAA-94/95 (90% inhibition, Ki = 53 nm) and NPPB-S (80% inhibition at 10 μm). IAA-94 and NPPB have also been reported to inhibit fast and slow anion channels in guard cells at micromolar concentrations (Marten et al., 1992; Schroeder et al., 1993), although in these cases an extracellular site of action seemed likely. So far, the effect of these high-affinity ligands on charophyte responses involving Cl− channel activation, such as excitability and turgor regulation, as well as their effect on single-channel currents, remains to be elucidated.
ACKNOWLEDGMENT
We are very grateful to Dr. A. Pope (SmithKline Beecham Pharmaceuticals) for the kind gift of NPPB, NPPB-S, IAA-94/95, and ISA-94/95.
Abbreviations:
- A-9-C
anthracene-9-carboxylic acid
- APW
artificial pond water
- BTP
bis-tris propane
- [Ca2+]c
cytosolic free Ca2+ concentration
- HEDTA
N-hydroxyethylethylenediaminetriacetic acid
- IAA-94/95
[(2-cyclopentyl-6,7-dichloro-2-methyl-2,3-dihydro-1-oxo-1H-inden-5-yl)oxyl]acetic acid
- ISA-94/95
[(2-cyclopentyl-6,7-dichloro-2-methyl-2,3-dihydro-1-oxo-1H-inden-5-yl)oxy]methanesulfonic acid
- KCa
half-activation constant for Ca2+
- KH
half-activation constant for H+
- NPPB
5-nitro-2-(3-phenylpropylamino)benzoic acid
- NPPB-S
5-nitro-2-(3-phenylpropylamino)benzene sulfonic acid
- NTA
nitrilotriacetic acid
- pHc
cytosolic pH
- PKA
protein kinase A
Footnotes
This work was supported by the Biotechnology and Biological Sciences Research Council (studentship to A.C. and research fellowship to E.J.).
LITERATURE CITED
- Beffagna N, Romani G, Gatti L. Changes in chloride fluxes and cytosolic pH induced by abscisic acid in Elodea densa leaves. Bot Acta. 1995;108:74–79. [Google Scholar]
- Beilby MJ. Calcium and plant action potentials. Plant Cell Environ. 1984;7:415–421. [Google Scholar]
- Beilby MJ, Coster HGL. The action potential in Chara corallina. III. The Hodgkin-Huxley parameters for the plasmalemma. Aust J Plant Physiol. 1979;6:337–353. [Google Scholar]
- Berestovsky GN, Zherelova OM, Katev AA. Ionic channels in characean algal cells. Biophysics. 1987;32:1101–1120. [Google Scholar]
- Blatt MR, Beilby MJ, Tester M. Voltage dependence of the Chara proton pump revealed by current-voltage measurement during rapid metabolic blockade with cyanide. J Membr Biol. 1990;114:205–224. doi: 10.1007/BF01869215. [DOI] [PubMed] [Google Scholar]
- Cho MH, Spalding EP. An anion channel in Arabidopsis hypocotyls activated by blue light. Proc Natl Acad Sci USA. 1996;93:8134–8138. doi: 10.1073/pnas.93.15.8134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrhardt DW, Atkinson EM, Long SR. Depolarization of alfalfa root hair membrane potential by Rhizobium meliloti nod factors. Science. 1992;256:998–1000. doi: 10.1126/science.10744524. [DOI] [PubMed] [Google Scholar]
- Ermolayeva E, Sanders D, Johannes E. Ionic mechanism and role of phytochrome-mediated membrane depolarization in caulonemal side branch initial formation in the moss Physcomitrella patens. Planta. 1997;201:109–118. [Google Scholar]
- Frachisse JM, Johannes E, Felle H. The use of weak acids as physiological tools: a study of the effects of fatty acids on internal pH and electrical plasma membrane properties of Riccia fluitans rhiziod cells. Biochim Biophys Acta. 1988;938:199–210. [Google Scholar]
- Hedrich R. Voltage-dependent chloride channels in plant cells: identification, characterization, and regulation of a guard cell anion channel. Curr Top Membr Transp. 1994;42:1–33. [Google Scholar]
- Hedrich R, Busch H, Raschke K. Ca2+ and nucleotide dependent regulation of a voltage dependent anion channel in the plasma membrane of guard cells. EMBO J. 1990;9:3889–3892. doi: 10.1002/j.1460-2075.1990.tb07608.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homann U, Thiel G. Cl− and K+ channel currents during the action potential in Chara: simultaneous recording of membrane voltage and patch currents. J Membr Biol. 1994;141:297–309. doi: 10.1007/BF00235139. [DOI] [PubMed] [Google Scholar]
- Keller UB, Hedrich R, Raschke K. Voltage-dependent anion channels in the plasma membrane of guard cells. Nature. 1989;341:450–453. doi: 10.1002/j.1460-2075.1990.tb07608.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landry DW, Akabas MH, Redhead C, Edelmann A, Cragoe EJ, Al-Awqati Q. Purification and reconstitution of chloride channels from kidney and trachea. Science. 1989;244:1469–1472. doi: 10.1126/science.2472007. [DOI] [PubMed] [Google Scholar]
- Lühring H, Tazawa M. Cytoplasmic Ca2+ has no effect on the excitability of Chara plasmalemma. Plant Cell Physiol. 1985;26:769–774. [Google Scholar]
- Lunevsky VZ, Zherelova OM, Vostikov I, Berestovsky GN. Excitation of Characeae cell membranes as a result of activation of Ca2+ and Cl− channels. J Membr Biol. 1983;87:177–190. [Google Scholar]
- Martell AE, Smith RM (1974) Critical Stability Constants, Vol 1. Plenum Press, New York
- Martell AE, Smith RM (1982) Critical Stability Constants, Vol 4. Plenum Press, New York
- Marten I, Zeilinger C, Redhead C, Landry DW, Al-Awqati Q, Hedrich R. Identification and characterisation of a voltage-dependent anion channel in the plasma membrane of guard cells by high affinity ligands. EMBO J. 1992;11:3569–3575. doi: 10.1002/j.1460-2075.1992.tb05440.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nürnberger T, Nennstiel D, Jabs T, Sacks WR, Hahlbrock K, Scheel D. High affinity binding of fungal oligopeptide elicitors to parsley plasma membranes triggers multiple defense responses. Cell. 1994;78:449–460. doi: 10.1016/0092-8674(94)90423-5. [DOI] [PubMed] [Google Scholar]
- Okazaki Y, Iwasaki N. Net efflux of Cl− during hypotonic turgor regulation in a brackish water alga Lamprothamnium. Plant Cell Environ. 1992;15:61–70. doi: 10.1093/pcp/pcf127. [DOI] [PubMed] [Google Scholar]
- Okazaki Y, Tazawa M. Calcium-ion and turgor regulation in plant cells. J Membr Biol. 1990;114:189–194. doi: 10.1007/BF01869213. [DOI] [PubMed] [Google Scholar]
- Okihara K, Okhawa T, Tsutsui I, Kasai M. A Ca2+ and voltage-dependent Cl−-sensitive anion channel in the Chara plasmalemma: a patch clamp study. Plant Cell Physiol. 1991;32:593–601. [Google Scholar]
- Pei Z-M, Kuchitsu K, Ward JM, Schwarz M, Schroeder JI. Differential abscisic acid regulation of guard cell slow anion channels in Arabidopsis wild type and abi1 and abi2 mutants. Plant Cell. 1997;9:409–423. doi: 10.1105/tpc.9.3.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid RJ, Smith FA, Whittington J. Control of intracellular pH in Chara corallina during uptake of a weak acid. J Exp Bot. 1989;40:883–890. [Google Scholar]
- Reinhart PH, Cung C, Martin BL, Brautigan DL, Levitan IB. Modulation of calcium-activated potassium channels from rat brain by protein kinase A and phosphatase 2A. J Neurosci. 1991;11:1627–1635. doi: 10.1523/JNEUROSCI.11-06-01627.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts SK, Dixon GK, Dunbar SJ, Sanders D. Laser ablation of the cell wall and localised patch clamping of the plasma membrane in the filamentous fungus Aspergillus: characterisation of an anion-selective efflux channel. New Phytol. 1997;137:579–585. [Google Scholar]
- Sanders D. Control of plasma membrane Cl− fluxes in Chara corallina by external Cl− and light. J Exp Bot. 1980a;31:105–118. [Google Scholar]
- Sanders D. Control of Cl− influx in Chara by cytoplasmic Cl− concentration. J Membr Biol. 1980b;52:51–60. [Google Scholar]
- Sanders D. The mechanism of Cl− transport at the plasma membrane of Chara corallina. I. Cotransport with H+ J Membr Biol. 1980c;53:129–153. [Google Scholar]
- Sanders D, Hansen U-P. Mechanism of Cl− transport at the plasma membrane of Chara corallina. II. Transinhibition and the determination of H+/Cl− binding order from a reaction kinetic model. J Membr Biol. 1981;58:139–153. [Google Scholar]
- Sanders D, Hansen U-P, Slayman CL. Role of the plasma membrane proton pump in the pH regulation in non-animal cells. Proc Natl Acad Sci USA. 1981;78:5903–5907. doi: 10.1073/pnas.78.9.5903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt C, Schelle I, Liao Y-J, Schroeder JI. Strong regulation of slow anion channels and abscisic acid signaling in guard cells by phosphorylation and dephosphorylation events. Proc Natl Acad Sci USA. 1995;92:9535–9539. doi: 10.1073/pnas.92.21.9535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroeder JI. Anion channels as central mechanisms for signal transduction in guard cells and putative functions in roots for plant-soil interactions. Plant Mol Biol. 1995;28:353–361. doi: 10.1007/BF00020385. [DOI] [PubMed] [Google Scholar]
- Schroeder JI, Hagiwara S. Cytosolic calcium regulates ion channels in the plasma membrane of Vicia faba guard cells. Nature. 1989;338:427–430. [Google Scholar]
- Schroeder JI, Keller BU. Two types of anion currents in guard cells with distinct voltage regulation. Proc Natl Acad Sci USA. 1992;89:5025–5029. doi: 10.1073/pnas.89.11.5025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroeder JI, Schmidt C, Schaefer J. Identification of high-affinity slow anion channel blockers and evidence for stomatal regulation by slow anion channels in guard cells. Plant Cell. 1993;5:1831–1841. doi: 10.1105/tpc.5.12.1831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulz-Lessdorf B, Lohse G, Hedrich R. GCAC1 recognizes the pH gradient across the plasma membrane: a pH-sensitive and ATP-dependent anion channel links guard cell membrane potential to acid and energy metabolism. Plant J. 1996;10:993–1004. [Google Scholar]
- Schwartz A, Ilan N, Schwarz M, Schaeffer J, Assmann SM, Schroeder JI. Anion-channel blockers inhibit S-type anion channels and abscisic acid responses in guard cells. Plant Physiol. 1995;109:651–658. doi: 10.1104/pp.109.2.651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiina T, Tazawa M. Regulation of membrane excitation by protein phosphorylation. Protoplasma. 1986;134:60–61. [Google Scholar]
- Shiina T, Tazawa M. Ca2+-activated Cl− channel in the plasmalemma of Nitellopsis obtusa. J Membr Biol. 1987;99:137–146. [Google Scholar]
- Shiina T, Tazawa M. Ca2+-dependent Cl− efflux in tonoplast-free cells of Nitellopsis obtusa. J Membr Biol. 1988;106:135–139. [Google Scholar]
- Shiina T, Wayne R, Lim Tung HY, Tazawa M. Possible involvement of protein phosphorylation/dephosphorylation in the modulation of Ca2+ channels of Nitellopsis. J Membr Biol. 1988;102:255–264. [Google Scholar]
- Smith FA, Raven JA. Intracellular pH and its regulation. Annu Rev Plant Physiol. 1979;30:289–311. [Google Scholar]
- Smith FA, Reid RJ. Biophysical and biochemical regulation of cytoplasmic pH in Chara corallina during acid loads. J Exp Bot. 1991;42:173–182. [Google Scholar]
- Smith RM, Martell AE (1976) Critical Stability Constants, Vol 4. Plenum Press, New York
- Tazawa M, Kikuyama M, Shimmen T. Electric characteristics and cytoplasmic streaming of Characeae cells lacking tonoplast. Cell Struct Funct. 1976;1:165–176. [Google Scholar]
- Thiel G, Homann U, Gradmann D. Microscopic elements of electrical excitation in Chara: transient activity of Cl− channels in the plasma membrane. J Membr Biol. 1993;134:53–66. doi: 10.1007/BF00233475. [DOI] [PubMed] [Google Scholar]
- Thomine S, Guern J, Barbier-Brygoo H. Voltage-dependent anion channel in Arabidopsis hypocotyls: nucleotide regulation and pharmacological properties. J Membr Biol. 1997;159:71–82. doi: 10.1007/s002329900270. [DOI] [PubMed] [Google Scholar]
- Tyerman SD. Anion channels in plants. Annu Rev Plant Physiol Plant Mol Biol. 1992;43:351–373. [Google Scholar]
- Ward JM, Pei Z-M, Schroeder JI. Roles of ion channels in initiation of signal transduction in higher plants. Plant Cell. 1995;7:833–844. doi: 10.1105/tpc.7.7.833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson RE. Cytoplasmic streaming in Chara: a cell model activated by ATP and inhibited by cytochalasin B. J Cell Sci. 1975;17:655–668. doi: 10.1242/jcs.17.3.655. [DOI] [PubMed] [Google Scholar]
- Williamson RE, Ashley CC. Free Ca2+ and cytoplasmic streaming in the alga Chara. Nature. 1982;296:647–651. doi: 10.1038/296647a0. [DOI] [PubMed] [Google Scholar]
- Zherelova OM. Protein kinase C is involved in regulation of Ca2+ channels in the plasma membrane of Nitella syncarpa. FEBS Lett. 1989;242:330–332. doi: 10.1016/0014-5793(89)80495-8. [DOI] [PubMed] [Google Scholar]
- Zimmermann S, Thomine S, Guern J, Barbier-Brygoo H. An anion current at the plasma membrane of tobacco protoplasts shows ATP-dependent voltage regulation and is modulated by auxin. Plant J. 1994;6:707–716. [Google Scholar]